Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. May 15, 2026; 18(5): 118319
Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.118319
Published online May 15, 2026. doi: 10.4251/wjgo.v18.i5.118319
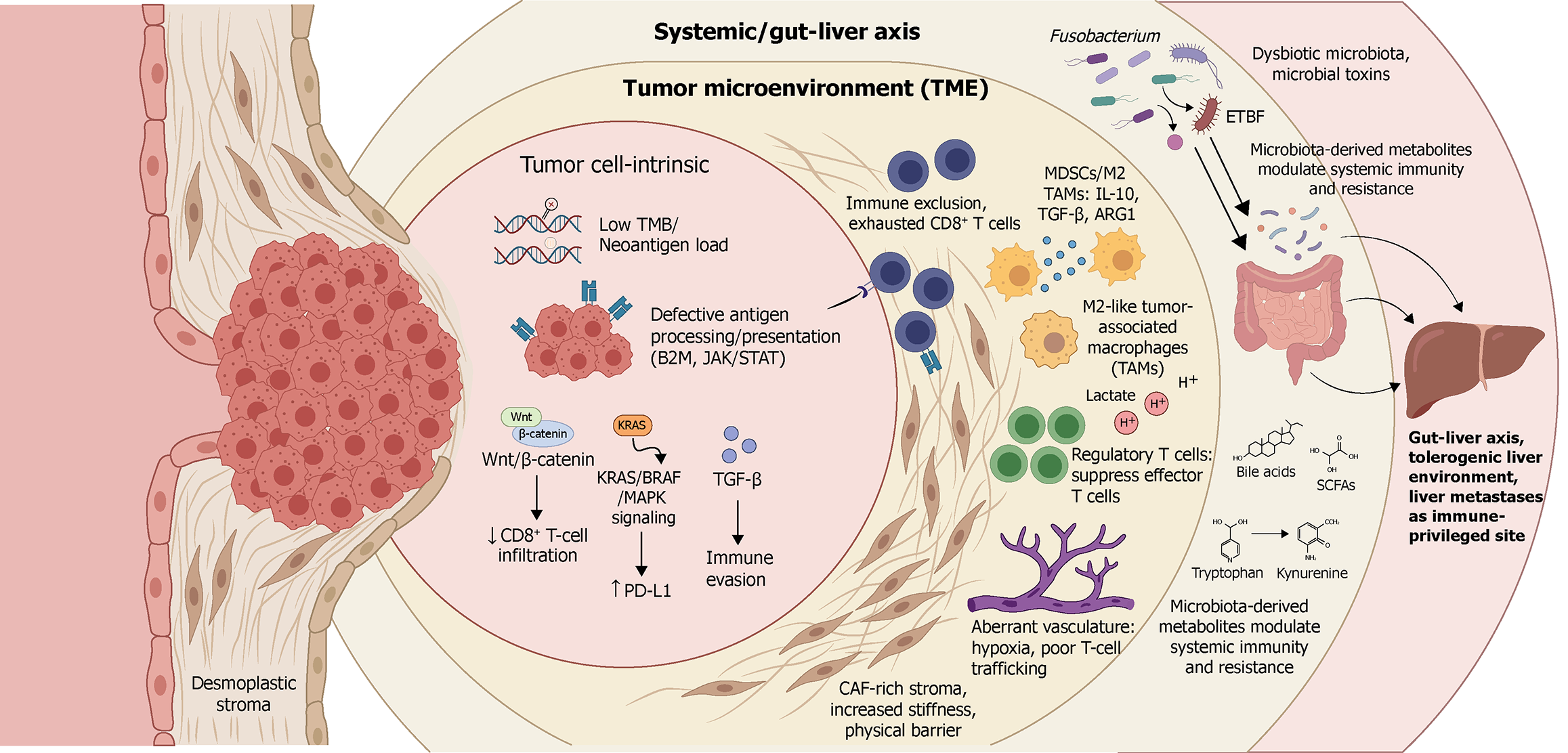
Figure 1 Immunobiology of “cold” microsatellite-stable/mismatch repair-proficient colorectal cancer and multilevel barriers to immune checkpoint blockade.
Concentric layers summarize how tumor cell-intrinsic programs, the local tumor microenvironment (TME), and systemic gut-liver crosstalk converges to generate an immune-excluded, immune checkpoint inhibitor (ICI)-refractory phenotype in microsatellite-stable (MSS) colorectal cancer (CRC). Tumor cell-intrinsic mechanisms include low tumor mutational burden and limited neoantigenicity, impaired antigen processing/presentation (e.g., β2-microglobulin or Janus kinase/signal transducer and activator of transcription pathway alterations), and oncogenic signaling (Wnt/β-catenin and KRAS/BRAF/mitogen-activated protein kinase) that reduces cluster of differentiation 8+ T-cell infiltration and promotes programmed death ligand-1 upregulation and immune evasion; tumor- and stroma-derived transforming growth factor-β (TGF-β) further reinforces immune suppression and exclusion. In the TME, dense cancer-associated fibroblast-rich desmoplastic stroma increases tissue stiffness and forms a physical barrier, while aberrant vasculature and hypoxia impair T-cell trafficking. Immunosuppressive populations including myeloid-derived suppressor cells and M2-like tumor-associated macrophages producing interleukin-10, TGF-β, and arginase-1, along with regulatory T cells blunt effector function. Metabolic stress (glycolysis-driven lactate accumulation and acidic potential of hydrogen) further suppresses T-cell activity. At the sys
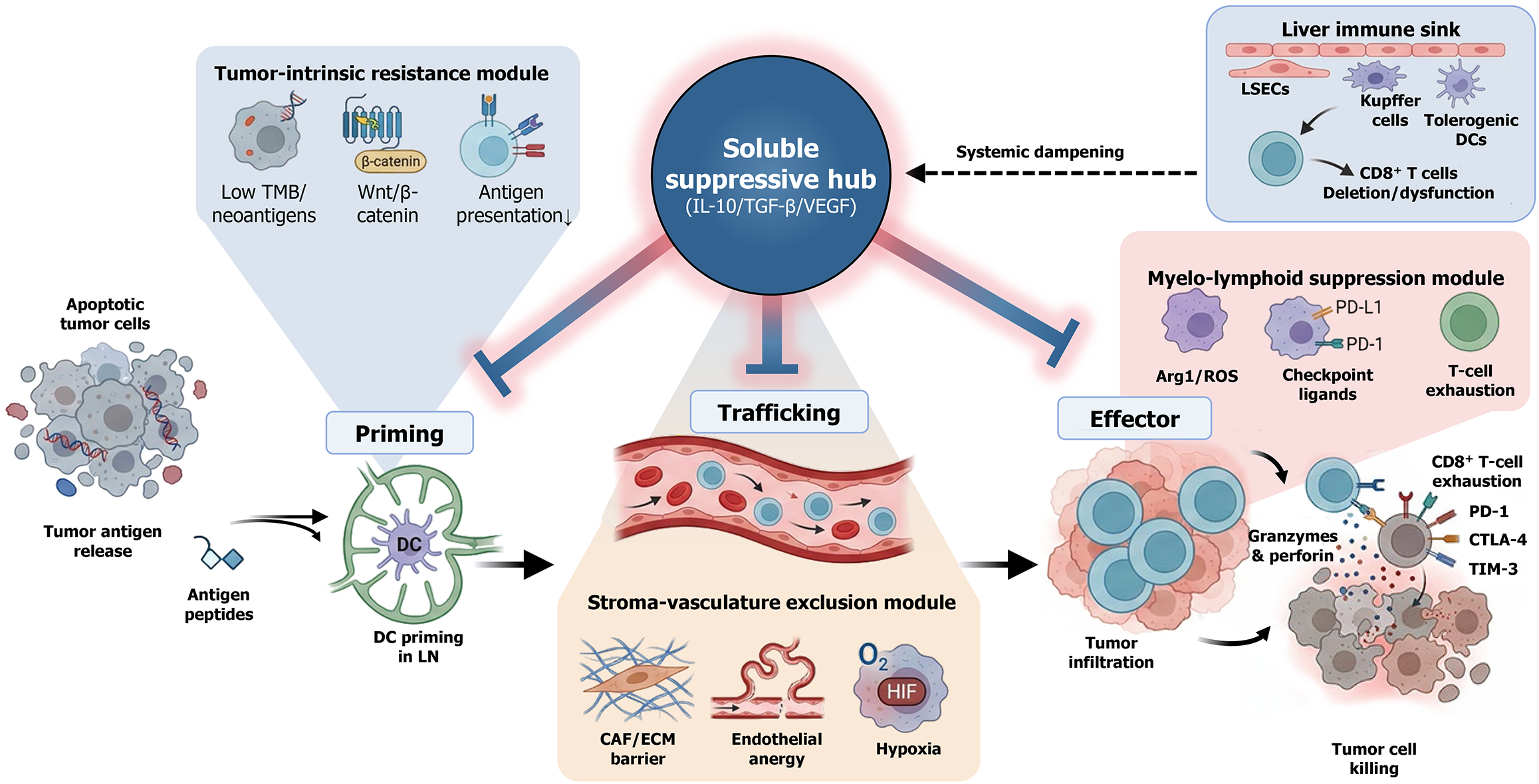
Figure 2 Multi-scale immunobiological barriers to immune checkpoint inhibitor response in microsatellite-stable/proficient mismatch repair colorectal cancer.
This schematic summarizes how microsatellite-stable/proficient mismatch repair (MSS/pMMR) colorectal cancer (CRC) resists immune checkpoint inhibitors (ICIs) across the major phases of antitumor immunity priming, trafficking, and effector function. Following tumor cell death, released antigenic peptides are captured by dendritic cells (DCs) and presented in draining lymph nodes to initiate T-cell priming. In MSS/pMMR CRC, tumor-intrinsic resistance (low tumor mutational burden/neoantigen scarcity, oncogenic Wnt/β-catenin signaling, and impaired antigen processing/presentation) limits productive priming and T-cell generation. Even when primed, T cells frequently fail to reach or penetrate tumor nests due to a stroma-vasculature exclusion module, characterized by cancer-associated fibroblast/extracellular matrix physical barriers, dysfunctional/endothelial anergy that reduces adhesion and transmigration, and hypoxia/hypoxia-inducible factor-driven vascular and metabolic constraints. Within tumors, a myelo-lymphoid suppression module (e.g., myeloid-derived suppressor cells/tumor-associated macrophages/regulatory T cell) further suppresses immunity through arginase-1/reactive oxygen species and upregulation of inhibitory checkpoint ligands, fostering cluster of differentiation (CD) 8+ T-cell exhaustion marked by programmed cell death protein 1, cytotoxic T-lymphocyte-associated protein-4, and T cell im
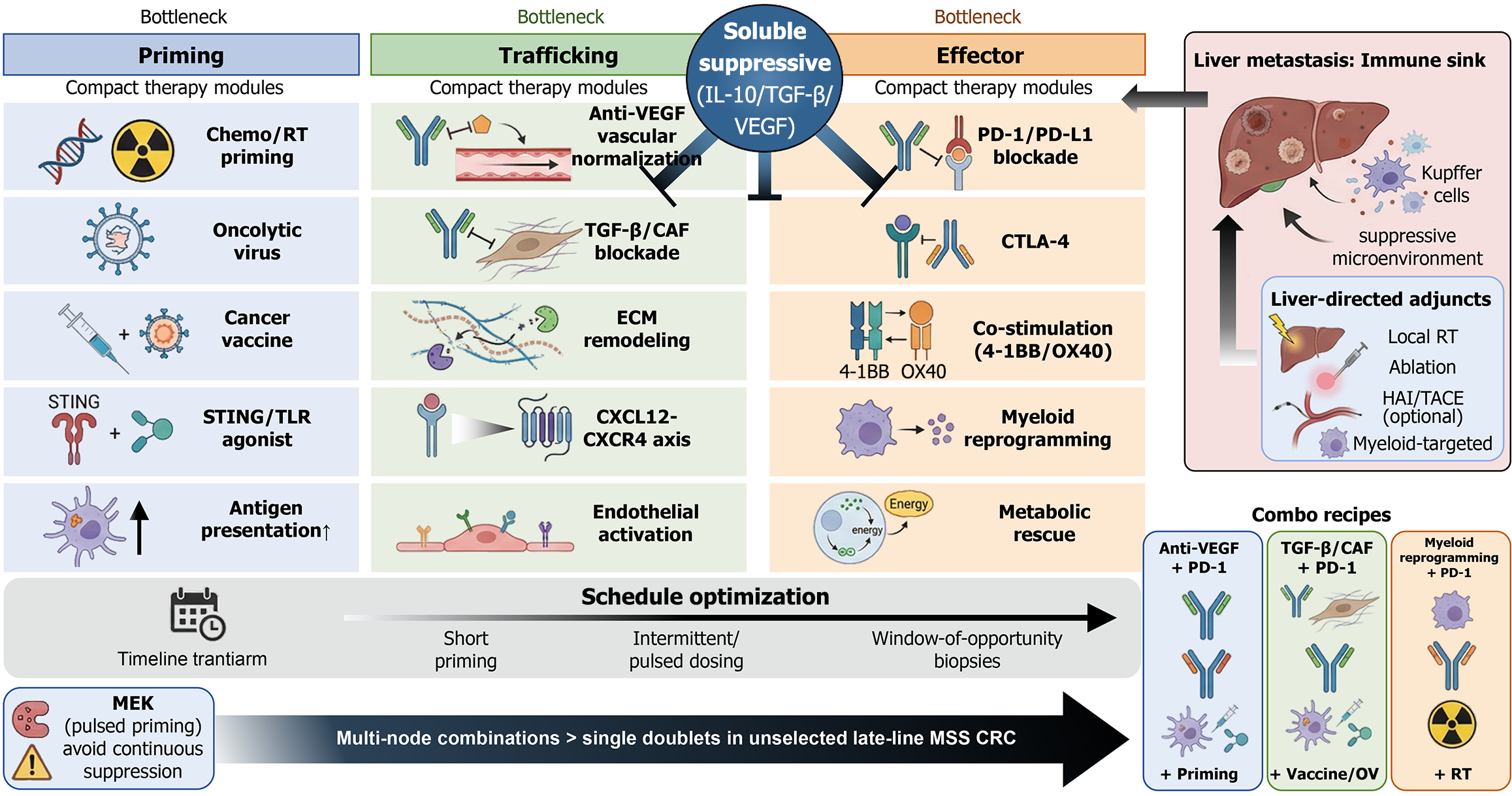
Figure 3 Bottleneck-guided “cold-to-hot” combination design for microsatellite-stable/proficient mismatch repair colorectal cancer.
Schematic framework that maps rational combination immunotherapy to the dominant rate-limiting step (“bottleneck”) along the cancer-immunity cycle in microsatellite-stable (MSS)/proficient mismatch repair colorectal cancer (CRC): Priming, trafficking, and effector function. For each bottleneck, “compact therapy modules” are shown that are intended to restore productive antitumor immunity rather than simply add a second agent to programmed death ligand-1 (PD-L1) blockade. Priming modules aim to increase immunogenic antigen availability and innate sensing (chemo/radiotherapy-mediated priming, oncolytic viruses, cancer vaccines, stimulator of interferon genes/Toll like receptor agonists, and strategies that enhance antigen presentation). Trafficking modules target stromal/vascular exclusion and immune cell access [anti-vascular endothelial growth factor (VEGF)-mediated vascular normalization, transforming growth factor-β (TGF-β)/cancer-associated fibroblast (CAF) blockade, extracellular matrix remodeling, disruption of the CXCL12-CXCR4 retention axis, and endothelial activation to support adhesion/extravasation]. Effector modules focus on reversing intratumoral dysfunction and exhaustion [programmed cell death protein-1 (PD-1)/PD-L1 blockade, cytotoxic T-lymphocyte-associated protein-4 blockade, co-stimulatory agonism such as 4-1BB/OX40, myeloid reprogramming, and metabolic rescue]. These interventions are coordinated against a shared soluble suppressive hub (interleukin-10/TGF-β/VEGF) that can concurrently dampen priming, trafficking, and effector cytotoxicity. The right panel highlights liver metastasis as an immune sink, where a suppressive hepatic microenvironment (e.g., Kupffer cell-driven tolerogenicity) can systemically blunt immune checkpoint inhibitor efficacy; “liver-directed adjuncts” (local radiotherapy, ablation, optional hepatic arterial infusion/transarterial chemoembolization approaches, and myeloid-targeted strategies) are positioned as context-specific intensifiers to overcome hepatic immune resistance. The bottom timeline emphasizes schedule optimization short immune priming, intermittent/pulsed dosing (illustrated by pulsed mitogen-activated protein kinase kinase priming to avoid continuous immune suppression), and window-of-opportunity biopsies to mechanistically validate target engagement and refine sequencing. Overall, the model underscores that multi-node combinations (addressing more than one bottleneck) are more likely to outperform single doublets in unselected, late-line MSS CRC, with example “combo recipes” illustrated (anti-VEGF + PD-1 with priming; TGF-β/CAF + PD-1 with vaccine/oncolytic priming; myeloid reprogramming + PD-1 with radiotherapy). IL: Interleukin; TGF-β: Transforming growth factor-β; VEGF: Vascular endothelial growth factor; RT: Radiotherapy; STING: Stimulator of interferon genes; TLR: Toll like receptor; MEK: Mitogen-activated protein kinase kinase; ECM: Extracellular matrix; CAF: Cancer-associated fibroblast; PD-1: Programmed cell death protein 1; PD-L1: Programmed death-ligand 1; CTLA-4: Cytotoxic T-lymphocyte-associated protein-4; MSS: Microsatellite stable; CRC: Colorectal cancer; HAI: Hepatic arterial infusion; TACE: Transarterial chemoembolization; OV: Oncolytic viruses.
- Citation: Chi F, Liu CB, Li J, Xia XW, Hua QJ, Wang W. Converting cold to hot: Strategies to sensitize microsatellite-stable colorectal cancer to immunotherapy. World J Gastrointest Oncol 2026; 18(5): 118319
- URL: https://www.wjgnet.com/1948-5204/full/v18/i5/118319.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i5.118319