Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.111687
Revised: August 26, 2025
Accepted: January 20, 2026
Published online: April 16, 2026
Processing time: 280 Days and 23 Hours
Endoscopic ultrasound (EUS)-guided tissue acquisition, including both fine needle aspiration and fine needle biopsy (EUS-FNB), has been widely used to obtain samples from pancreatic and non-pancreatic lesions.
To summarize the incidence of complications from this procedure in a single endoscopic center.
Data were retrospectively collected from patients who underwent diagnostic EUS at our endoscopy center over a 1-year period. We enrolled 88 cases with solid pan
A total of 88 EUS-FNB procedures were performed (48.9% female; mean age 67.8 ± 12.7 years). Complications occurred in 9.1% of cases, including bleeding (3.4%), perforation (4.5%), and pancreatitis (1.1%), with no infections or deaths. Endo
Adverse events after EUS-FNB were higher than reported in literature; endoscopist experience and specific diagnoses, rather than overall comorbidity burden, were key predictors of complications.
Core Tip: This study highlighted that complications following fine needle biopsy are infrequent and primarily influenced by endoscopist experience rather than the overall burden of comorbidities. Although most patients had at least one comorbidity, no significant association was found between comorbidities and complication rates. Cardiovascular disease emerged as a specific predictor of risk while arterial hypertension showed a surprising negative association. These findings emphasize the importance of individual clinical factors and endoscopist expertise over cumulative comorbidity scores when assessing procedural risk and ensuring patient safety during fine needle biopsy.
- Citation: Peruhova M, Stoitsov V, Mirchev MB, Velikova T. Complication incidence after endoscopic ultrasound-guided fine needle biopsy: A single-center experience. World J Gastrointest Endosc 2026; 18(4): 111687
- URL: https://www.wjgnet.com/1948-5190/full/v18/i4/111687.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i4.111687
For the first time in 1980, an ultrasonography probe was connected to an endoscope, and endoscopic ultrasound (EUS) was introduced in clinical practice[1]. Initially, EUS was utilized as a diagnostic imaging technique for various gas
The development of EUS-FNA revolutionized the field and enhanced the diagnostic capabilities of EUS. The primary benefit is that it enables the safe and accurate sampling of lesions that were previously inaccessible or required more invasive methods for pathological diagnosis[5]. EUS-FNA is a diagnostic procedure that in most cases can provide adequate cytological samples from pathological tissue for the final diagnosis. Still, it is insufficient for allowing immunohistochemistry analysis of the pathological probe[6]. With EUS-FNB adequate tissue acquisition can be achieved with a small number of punctures, allowing the supply of core tissue for immunohistochemical staining and gene profiling[7]. To enhance the diagnostic yield of EUS-FNA/EUS-FNB, various methodological advancements have been suggested, including altering the needle tip, employing a larger gauge (G) needle, increasing the number of passes within the lesion, and adjusting the tissue sampling technique (fanning method vs simple “to and fro” movement, dry vs wet technique, and slow-pull stylet vs standard suction)[8].
It is a well-known fact that routine EUS with or without FNA/FNB is a safe and efficient procedure. Despite routine EUS being associated with adverse events (AEs), their overall incidence is modest. To enhance intraprocedural app
This study aimed to comprehensively assess the complications associated with EUS-FNB at our endoscopic center and identify the risk factors for these complications. We collected data on the comorbidities of the patients, endoscopic experience of the endoscopists, and different biopsy targets according to their location in the abdominal cavity. We evaluated the potential connections between these factors and the rate of complications after EUS-FNB and the occurrence of complications following FNB and identified possible predictors, including patient comorbidities and endoscopist experience, using statistical modeling and cluster analysis.
This study represented a retrospective analysis of a prospectively maintained database of patients who underwent diagnostic EUS for the evaluation of solid pancreatic or non-pancreatic lesions over 1 year. We collected medical data that were documented, including patient demographics, presumptive diagnosis, comorbidities of the patient, technical success of the procedure, years of endoscopic experience of the endoscopist, and occurrence of AEs or complications. We observed early complications after EUS-FNA/FNB that were detected immediately after the procedure and up to 3 days after it. The complications were detected by post-procedural monitoring. During the follow-up period, we evaluated patients’ clinical conditions and performed a conventional abdominal ultrasound. All the patients had a lab test 12 hours after the procedure. We measured the level of hemoglobin, leucocytes, pancreatic enzymes, and C-reactive protein. If we had a suspicion of acute pancreatitis, bleeding, or perforation, we performed a CT scan. According to international endoscopic guidelines, EUS-FNB is a high-risk endoscopic procedure related to a high risk of periprocedural and post-procedural bleeding. From this perspective, all patients included in the study were evaluated based on their coagulation status and antiplatelet or anticoagulant therapy.
In our study patients who were taking warfarin were discontinued 5 days before the procedure with bridging therapy considered for patients at high risk. The international normalized ratio was checked before the procedure to ensure it was below 1.2. Patients who were on P2Y12 receptor antagonists (e.g., clopidogrel) discontinued 5 days before the procedure, and their thrombotic risk was evaluated. Before performing EUS-FNA it is crucial to rule out pre-existing hematological disorders, such as coagulopathy (international normalized ratio > 1.2), thrombocytopenia (platelet count < 50000), and recent or chronic use of thienopyridines (e.g., clopidogrel), to minimize the risk of complications. All patients receiving anticoagulant or antiplatelet therapy discontinued their medication in accordance with established clinical guidelines prior to the procedure. A control coagulogram was performed in each case to confirm that coagulation parameters were within the normal reference range at the time of biopsy. Regarding the EUS-FNB sampling process, two two-needle passes were utilized for the cell block. The EUS-FNB samples underwent macroscopic on-site assessment that was verified as sufficient by physically examining the ejected tissue for its presence. Until the endosonographer felt that an adequate sample had been acquired, needle passes were repeated.
We enrolled 88 cases with solid pancreatic or non-pancreatic lesions. The mean age of the subjects was 67.8 ± 12.7 years (32-89 years). Forty-five were male (51.1%), and 43 were female (48.9%). Eighty of them (90.9%) reported comorbidities.
EUS investigations were performed using linear echoendoscopes (GF-UCT 180, Olympus Medical Systems, Tokyo) coupled with the corresponding US processor (Aloka Hitachi Aloka 750 ultrasound system). Commercially available 22G FNB needles (Acquire, Boston Scientific, Marlborough, MA, United States; Trident, Micro-Tech, CA, United States) were used. All the procedures were performed under sedation with propofol.
The study protocol received approval from the institutional research ethics board and was conducted in accordance with the principles of the Declaration of Helsinki. All patients signed informed consent before the procedure.
All statistical analyses were conducted using IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp., Armonk, NY, United States). Descriptive statistics were presented as absolute numbers and percentages for categorical variables. Differences between groups were assessed using Pearson’s χ2 test or Fisher’s exact test as appropriate. For comparisons involving continuous variables across categories, one-way analysis of variance was used. Pearson correlation coefficients were calculated to investigate the relationships between complication rates and predictor variables, including years of endoscopist experience and the number of comorbidities. To further examine predictors of complications, a multiple linear regression model was constructed, including individual comorbid conditions [e.g., cardiovascular disease (CVD), diabetes, cancer] and prior medical interventions (e.g., previous surgery, chemotherapy, radiation therapy) as in
The majority of patients reported comorbidities (90.9%) as follows: 46 (52.3%) arterial hypertension (AH); 25 (28.4%) other CVD; 19 (21.6%) diabetes; 4 (4.5%) liver cirrhosis; 14 (15.9%) liver metastases; 6 (6.8%) respiratory disease; 18 (20.5%) cancer; 31 (35.2%) previous surgery; 2 (2.3%) previous chemotherapy; 3 (3.4%) previous radiation therapy; and 9 (10.2%) pathology in thyroid gland. A total of 88 FNB procedures were analyzed, targeting a wide range of anatomical sites. The most commonly biopsied organ was the pancreas, accounting for 45.5% (n = 40) of all procedures. Other frequently sampled sites included the liver (14.8%, n = 13), lymph nodes (14.8%, n = 13), duodenum (8.0%, n = 7), and stomach (8.0%, n = 7). Less commonly biopsied sites included the mediastinum, adrenal gland, mediastinal lymph nodes, papilla vateri, liver and lymph nodes, and retroperitoneal tumor, each contributing 1.1% to 2.3% of cases. Two procedures had undocumented biopsy sites. This distribution reflects the clinical diversity of FNB indications in our cohort.
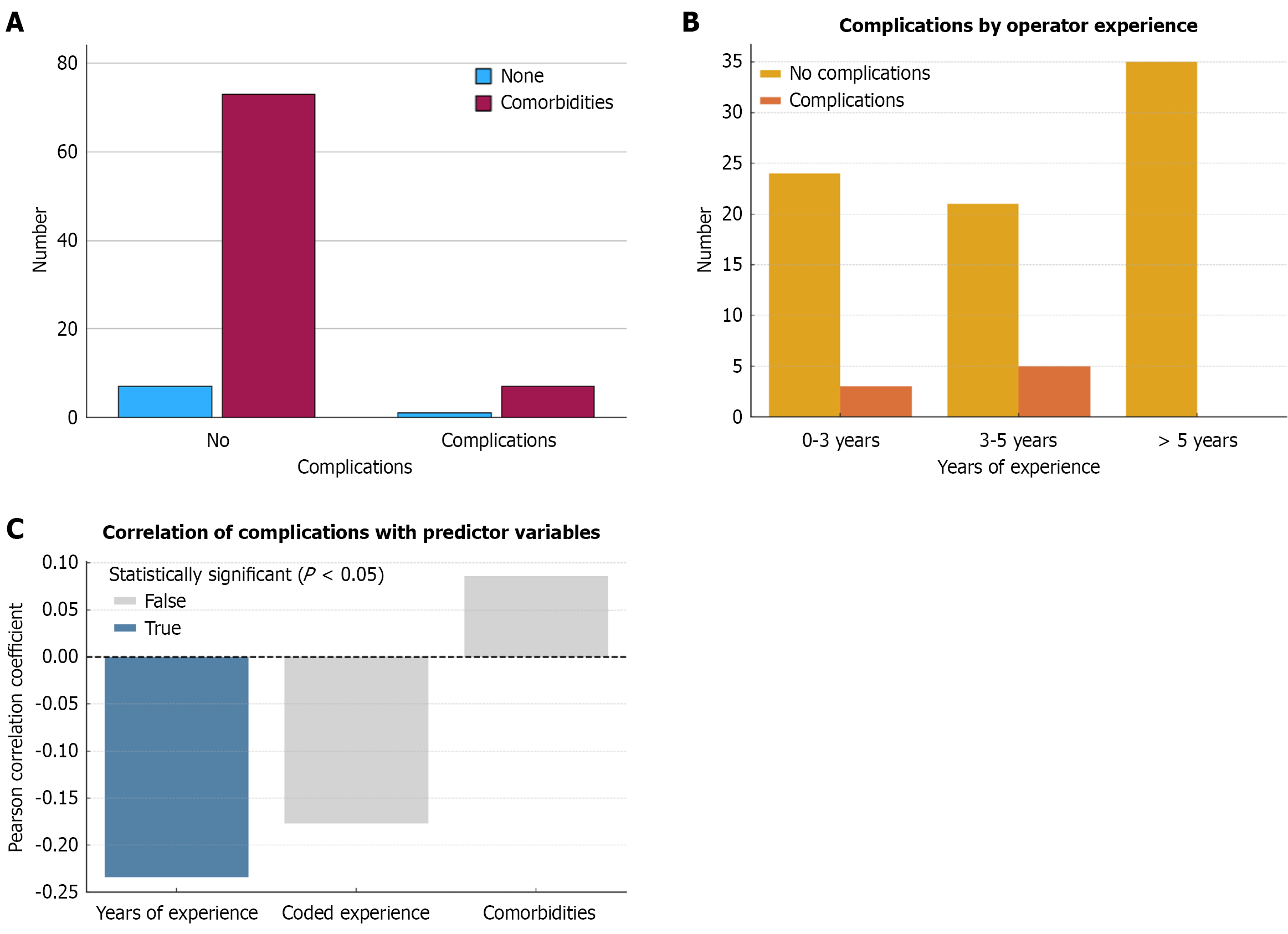
Histological analysis was successfully performed in the majority of cases with only 3 patients (3.4%) having no biopsy sample obtained. Among the 88 procedures, the most common pathological diagnosis was adenocarcinoma, identified in 31 cases (35.2%). Normal tissue was observed in 25 patients (28.4%) while another 25 samples (28.4%) were classified as other histological findings. Less frequent diagnoses included lymphoma (2.3%, n = 2) and GI stromal tumor (GIST) (2.3%, n = 2). These findings reflect the diagnostic versatility of FNB in evaluating both malignant and benign conditions across diverse tissue types. Complications were observed in 8 (9.1%) patients who underwent the procedure. These included bleeding in 3 (3.4%), perforation in 4 (4.5%), and pancreatitis in 1 (1.1%) case. No infections and no lethal cases were reported in the studied group. We found that patients with comorbidities were more likely to experience complications during the FNB procedure. Specifically, 8 patients had complications of whom 1 (12.5%) had no comorbidities and 7 (87.5%) had comorbidities; however, this difference was not statistically significant (P = 0.725) (Figure 1A). Interestingly, a trend towards more frequent bleeding and pancreatitis was found when the pancreas was punctured [odds ratio = 9.98; 95% confidence interval (CI): 0.51-191.90]. Furthermore, the perforation was a more common observation if another organ was the target of FNB (OR = 5.89; 95%CI: 0.29-117.90) but without reaching significance.
Regarding the experience of the three endoscopists, one reported 0-3 years of experience, one reported 3-5 years of experience, and one reported more than 5 years of experience. The procedures were distributed among the three specialists: The one with 0-3 years of experience performed 27 procedures (30.7%), the one with 3-5 years of experience performed 26 procedures (29.5%), and the most experienced one (with more than 5 years of experience) performed 35 procedures (39.8%). We found that the rate of complications was the lowest in the procedures performed by the endoscopist with more than 5 years of experience, and the highest in the procedures performed by the endoscopist with 3-5 years of experience (P = 0.032, Pearson’s χ2) with a likelihood ratio of 9.322 (df = 2, P = 0.009) (Table 1, Figure 1B).
| Years of experience of the endoscopist | Significance1, P value | |||
| 0-3 | 3-5 | More than 5 | ||
| No complications | 24 (30.0) | 21 (26.3) | 35 (43.8) | 0.028 |
| Complications | 3 (37.5) | 5 (62.5) | 0 | |
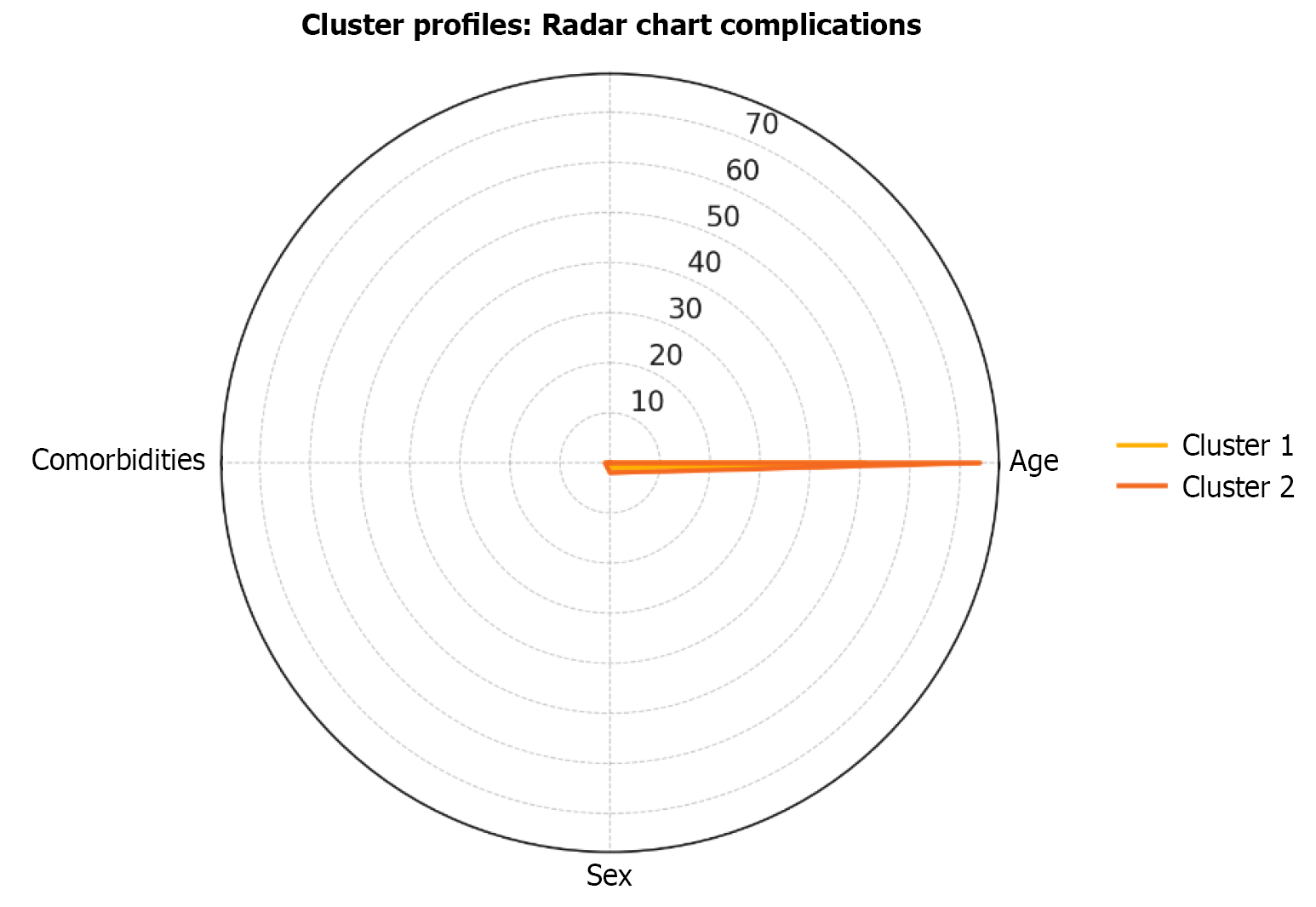
Pearson correlation showed a weak but statistically significant negative association between years of experience and the occurrence of complications (r = -0.234, P = 0.014), indicating that more experienced endoscopists had fewer complications. A similar trend was observed with categorized experience (r = -0.177, P = 0.050). No significant correlation was found between complications and patient comorbidities (r = 0.086, P = 0.214) (Figure 1C). Specific complications according to the endoscopist’s years of experience are presented in Table 2. Cluster analysis using a two-cluster solution revealed distinct patient profiles primarily based on age and sex. Both clusters had no complications and shared identical values for comorbidities (mean age = 1), indicating these variables did not differentiate the groups. Cluster 1 was characterized by a mean age of 51 years and predominantly male patients (coded as 1) while cluster 2 included older patients (mean age = 74) and was composed mainly of female patients (coded as 2). Analysis of variance results confirmed significant between-cluster differences for age (F = 196.50, P < 0.001) and sex (F = 6.39, P = 0.013) while complications and comorbidities did not differ significantly between groups. This suggests that age and sex were the primary drivers of cluster formation (Figure 2).
| Complications | Years of experience of the endoscopist | Significance, P value | ||
| 0-3 | 3-5 | More than 5 | ||
| Pancreatitis | 0 | 1 | 0 | 0.299 |
| Bleeding | 1 | 2 | 0 | 0.260 |
| Perforations | 2 | 2 | 0 | 0.250 |
| Infections | 0 | 0 | 0 | N/A |
| Lethality | 0 | 0 | 0 | N/A |
Indeed, the mean age of patients with complications was higher than that of patients without complications (70.88 ± 13.206 vs 67.35 ± 12.750, P = 0.981), but this difference was not statistically significant in our study. A multiple linear regression analysis examining the relationship between complications and clinical predictors (n = 88) revealed that the overall model was not statistically significant [F (12, 75) = 1.26, P = 0.262, R2 = 0.168]. Among the predictors, CVD (B = 0.208, P = 0.014) and AH (B = -0.167, P = 0.031) were significant while the total number of comorbidities was not associated with complications (P = 0.828). These findings suggest that specific diagnoses rather than the burden of comorbidities may better predict the risk of complications (Table 3).
| Variable | B (unstandardized) | Beta (standardized) | t | P value | Interpretation |
| CVD | 0.208 | 0.327 | 2.504 | 0.014 | Significant positive predictor |
| AH (hypertension) | -0.167 | -0.291 | -2.195 | 0.031 | Significant negative predictor |
| Cancer | 0.179 | 0.251 | 1.896 | 0.062 | Trend-level significance |
| Previous surgery | -0.128 | -0.212 | -1.823 | 0.072 | Near significance |
| Comorbidities (total) | 0.026 | 0.026 | 0.218 | 0.828 | Not significant |
Our observation for more frequent perforations after FNB could be associated to endoscopist capability and older patients. Hemorrhage could be justified by the equal needle size used in all circumstances. In some of the cases, the needle diameter could be incorrectly bigger. Blood-thinning drugs are proven risk factors for hemorrhage. Therefore, they were ceased in all patients before the procedure. Additionally, we documented various complications, depending on the punctured organ. Due to the close anatomical relationship between the pancreas and major blood vessels, postprocedural bleeding may occur. Pancreatic puncture can also result in pancreatitis. In contrast, perforations were observed more frequently in cases that were performed by less experienced endoscopists. One of the patients with duodenal perforation had a medical history of a duodenal ulcer in the past, indicating a risk factor for this type of complication. Another strong positive correlation was found between the level of experience and the incidence of perforation. The highly experienced (with more than 5 years of experience) endoscopist had no perforation.
This study examined complication rates following FNB procedures in a cohort of 88 patients with a high prevalence of comorbid conditions. While 90.9% of patients had at least one comorbidity, complications were observed in only 9.1% of cases, including bleeding, perforation, and pancreatitis. Importantly, no infections or fatal outcomes were reported, suggesting an overall acceptable safety profile of the FNB procedure in this population. A meta-analysis by Tian et al[12] on complication incidence of EUS-guided pancreas biopsy from data of 78 studies from 71 articles comprising 11652 patients showed that bleeding and pancreatitis complication incidences was low [0.210 × 10-4 (95%CI: -0.648 × 10-4 to 1.068 × 10-4)] while abdominal pain, fever, infections, duodenal perforation, pancreatic fistula, abscess, and sepsis were extremely low.
Despite the high comorbidity burden, no significant association was found between the total number of comorbidities and the occurrence of complications. Among the 8 patients who experienced complications, 87.5% had one or more comorbidities. However, this observation did not reach statistical significance (P = 0.725), and Pearson correlation analysis confirmed the absence of a meaningful linear relationship (r = 0.086, P = 0.214). These results imply that comorbidity burden alone may not be a reliable predictor of complications during FNB. This finding aligns with previous reports emphasizing the importance of individualized risk profiling over simple comorbidity counts[3]. Furthermore, although EUS-FNA has a low complication rate, the increasing number of procedures may lead to a higher frequency of AEs. The main complications linked to EUS-FNA include bleeding, perforation, infection, and organ-specific events such as acute pancreatitis following pancreatic puncture. In rare cases needle tract seeding has also been reported. Although complication rates vary depending on the lesion and study, they are generally low, ranging from 0% to 2.5%, with a mortality rate between 0.1% and 0.8%. Fatal outcomes are typically due to severe hemorrhage or complications following duodenal perforation[13-17].
In contrast, endoscopist experience emerged as a statistically significant factor. The lowest complication rate was observed in procedures performed by the most experienced endoscopist (> 5 years). In comparison, the highest rate (62.5% of all complications) was reported for procedures performed by an endoscopist with intermediate experience (3-5 years of experience). This relationship was statistically significant (P = 0.032), and the likelihood ratio test (P = 0.009) further supported the robustness of this finding. Moreover, Pearson correlation analysis confirmed a negative association between years of experience and complication rates (r = -0.234, P = 0.014). These findings underscore the role of clinical expertise in minimizing procedure-related risks and are in line with studies demonstrating learning curve effects in interventional endoscopy and ultrasound-guided procedures[18-20].
One of our study limitations was that it represents a small number of patients (88) who underwent EUS-FNB by three endoscopists with different levels of experience. It is essential to note that our center has a limited number of highly qualified endoscopists who are capable of performing EUS, and patients were randomly assigned to these three endoscopists. From that perspective, the lack of a protocol for allocating patients amongst endoscopists could be highlighted as a disadvantage of the study. Current evidence suggests that EUS-guided transmural interventions are generally safe and effective with lower complication rates compared with percutaneous radiological methods and less invasiveness than surgery, particularly when performed by experienced endoscopists[21]. Given the complexity of these advanced therapeutic techniques, an additional 12-24 months of specialized endoscopic training is often necessary to achieve proficiency[22-24].
Cluster analysis revealed two distinct patient profiles that differed primarily in terms of age and sex. Cluster 1 consisted of younger, predominantly male patients (mean age: 51 years) while cluster 2 comprised older, predominantly female patients (mean age: 74 years). Neither the comorbidity burden nor complication rates differed between clusters, suggesting that demographic characteristics rather than clinical risk factors defined patient grouping in this context. While the cluster analysis did not yield actionable predictors of complications, it provided useful insight into the demographic stratification of the FNB population. The cluster analysis was performed as an exploratory tool to assess potential patterns among patient-related and lesion-related factors. Although methodologically interesting, it did not identify clinically significant differences in complication risk. Thus, its value remains primarily hypothesis generating rather than conclusive, and future studies with larger cohorts are needed to explore such stratifications further.
Regression analysis further explored the predictive value of individual comorbidities. The model did not reach statistical significance overall (R2 = 0.168, P = 0.262), but two predictors stood out. CVD was a significant positive predictor of complications (B = 0.208, P = 0.014), suggesting that patients with established CVD may be at elevated risk. Interestingly, AH was identified as a significant negative predictor (B = -0.167, P = 0.031). This finding may reflect confounding factors or underlying protective mechanisms, such as increased clinical monitoring or treatment. This unexpected inverse relationship warrants further investigation in larger cohorts. CVD was a statistically significant positive predictor of complications (P = 0.014), suggesting that patients with CVD are more likely to develop complications. At the same time, AH was a significant negative predictor (P = 0.031), which is counterintuitive and may require further investigation (e.g., confounding). We can speculate that confounding factors likely influenced this result.
Furthermore, this finding was counterintuitive and likely reflected confounding factors rather than a true protective effect. Possible explanations include selection bias with patients with hypertension potentially being managed more cautiously, more rigorous periprocedural monitoring in this subgroup, or even medication-related effects. Patients with hypertension in our cohort were generally subject to closer medical monitoring and stricter preprocedural optimization, potentially reducing their overall risk of complications. Nonetheless, residual confounding cannot be excluded, and further studies with larger cohorts are needed to confirm or refute this observation, thereby clarifying whether this is a spurious association or reflects a reproducible clinical trend.
Comorbidities (total) were not a significant predictor (P = 0.828), indicating that the overall burden of comorbidities as measured here did not independently predict complications when specific diagnoses are considered simultaneously. The overall explanatory power of the model was limited, suggesting that other clinical or procedural factors might play a larger role. Other factors, such as cancer and previous surgery, showed trend-level associations but did not achieve statistical significance[25]. Duodenal perforation was an uncommon but significant complication that can occur during EUS-FNB. Although the general occurrence rate was low, certain factors may heighten the risk. These factors comprise the inexperience of the operator, older patient age, and the possible existence of pre-existing duodenal conditions such as peptic ulcer disease or duodenal diverticula[26].
The study published data related to the correlation between specific endoscopic EUS techniques and the incidence of EUS-induced duodenal perforations. In our study three of the perforations were associated with a particular endoscopic technique, known as the “Forgotten Knob” technique. The endoscopists with low endoscopic experience during EUS-FNB in the descending duodenum retracted the scope without releasing the scope’s fixed knob, leading to perforation. Our more frequent perforations could be attributed to endoscopist inexperience and patients’ older age. However, Benson et al[25] showed that EUS and endoscopic retrograde cholangiopancreatography complication rates were not increased in elderly patients. They demonstrated that the complication rate for EUS was 4.8% among elderly patients compared with 3.1% in younger patients, a difference that was not statistically significant (P = 0.49). Overall complication rates were the same in both groups at 7.6% (P = 1.0). Elderly patients received significantly lower doses of sedation (P < 0.001) while the incidence of procedural bleeding was significantly higher in this group (P = 0.016)[8].
The occurrence of hemorrhage could be attributed to the uniform use of a single needle size, which may have been excessive in diameter for certain cases. The characteristics of the target lesion frequently influenced needle selection. For example, 22G needles are commonly used to obtain tissue from pancreatic tumors, whereas 25G needles may be more suitable for small or difficult-to-reach targets, and 19G needles are chosen for larger specimens when fragmentation is a concern. Unfortunately, we do not have 25G needles available at our center. For this reason, we were unable to tailor the needle size to the lesion type or location and the risk of bleeding.
Among the 88 procedures, the most common pathological diagnosis was adenocarcinoma, identified in 31 cases (35.2%). Normal tissue was observed in 25 patients (28.4%) while another 25 samples (28.4%) were classified as other histological findings. Less frequent diagnoses included lymphoma (2.3%, n = 2) and GIST (2.3%, n = 2). We established that only 3 patients from our study had clinically significant hemorrhage. These 3 patients had EUS-FNB for pancreatic head lesions. Histologically, these lesions were confirmed as pancreatic adenocarcinoma. Many studies established that hypervascular tumors, such as neuroendocrine tumors and GIST, are one of the major risk factors for bleeding after EUS-FNB. Clinically, bleeding may present as intra-abdominal or bleeding from the upper GI tract. Unfortunately, we did not evaluate the vascularity of the tumors by contrast CT/magnetic resonance imaging and Doppler EUS before the procedures[27]. Two of the patients who had bleeding after EUS-FNB had taken anticoagulation therapy for atrial fibrillation. Blood-thinning agents were replaced with low molecular weight heparin 2 days before the procedure. It is a well-known fact that blood-thinning agents are proven risk factors for bleeding after EUS-FNB[28]. We performed a control coagulation test for all patients before the procedure, and all the patients were within the normal range at the time of the biopsy.
In addition, we observed distinct patterns of complications depending on the target organ. The close proximity of major blood vessels surrounding the pancreas predisposes to postprocedural bleeding[28] while direct pancreatic puncture may result in pancreatitis. In contrast, the higher incidence of perforations associated with non-pancreatic targets was likely unrelated to the biopsy itself but rather to the rigidity of the echoendoscope and the increased mobility of the duodenum when it is not affected by pancreatic disease[26,29]. Taken together, these findings highlight the complexity of predicting complications in FNB. Rather than the total comorbidity count, individual diagnoses and procedural variables, particularly the endoscopist’s experience, appear to have greater predictive value. These results support the need for individualized risk assessment, careful training of endoscopists, and further prospective studies to validate these associations.
In this study the overall complication rate following FNB was low (9.1%) despite a high prevalence of comorbidities among patients. Overall, the incidence of AEs observed after EUS-FNB in our study appeared to be slightly higher than that reported in previous studies. While the presence of comorbidities was common, it was not independently associated with an increased risk of complications. In contrast, endoscopist experience emerged as a significant and consistent predictor with fewer complications observed in procedures performed by the most experienced specialist. Additionally, CVD was identified as a potential risk factor for complications while AH showed an unexpected inverse association. These findings suggest that specific clinical conditions and procedural expertise may play a more significant role in determining the risk of complications than the overall comorbidity burden. Ensuring procedures are performed by experienced endoscopists and identifying high-risk diagnoses could improve patient safety. Future research with larger sample sizes and a prospective design is warranted to validate these observations and refine risk stratification strategies in FNB.
| 1. | Wiersema MJ, Vilmann P, Giovannini M, Chang KJ, Wiersema LM. Endosonography-guided fine-needle aspiration biopsy: diagnostic accuracy and complication assessment. Gastroenterology. 1997;112:1087-1095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 888] [Cited by in RCA: 716] [Article Influence: 24.7] [Reference Citation Analysis (4)] |
| 2. | Dumonceau JM, Deprez PH, Jenssen C, Iglesias-Garcia J, Larghi A, Vanbiervliet G, Aithal GP, Arcidiacono PG, Bastos P, Carrara S, Czakó L, Fernández-Esparrach G, Fockens P, Ginès À, Havre RF, Hassan C, Vilmann P, van Hooft JE, Polkowski M. Indications, results, and clinical impact of endoscopic ultrasound (EUS)-guided sampling in gastroenterology: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline - Updated January 2017. Endoscopy. 2017;49:695-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 244] [Article Influence: 27.1] [Reference Citation Analysis (1)] |
| 3. | Mizuide M, Ryozawa S, Fujita A, Ogawa T, Katsuda H, Suzuki M, Noguchi T, Tanisaka Y. Complications of Endoscopic Ultrasound-Guided Fine Needle Aspiration: A Narrative Review. Diagnostics (Basel). 2020;10:964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 4. | Zhang L, Sanagapalli S, Stoita A. Challenges in diagnosis of pancreatic cancer. World J Gastroenterol. 2018;24:2047-2060. [PubMed] [DOI] [Full Text] |
| 5. | Akahoshi K, Oya M, Koga T, Shiratsuchi Y. Current clinical management of gastrointestinal stromal tumor. World J Gastroenterol. 2018;24:2806-2817. [PubMed] [DOI] [Full Text] |
| 6. | Cazacu IM, Luzuriaga Chavez AA, Saftoiu A, Vilmann P, Bhutani MS. A quarter century of EUS-FNA: Progress, milestones, and future directions. Endosc Ultrasound. 2018;7:141-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 7. | Kongkam P, Orprayoon T, Yooprasert S, Sirisub N, Klaikaew N, Sanpawat A, Safa S, Ridtitid W, Kullavanijaya P, Rerknimitr R. Endoscopic ultrasound guided fine needle biopsy (EUS-FNB) from peritoneal lesions: a prospective cohort pilot study. BMC Gastroenterol. 2021;21:400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 8. | Cherciu Harbiyeli IF, Constantin A, Cazacu IM, Burtea DE, Gheorghe EC, Popescu CF, Bejinariu N, Georgescu CV, Pirici D, Ungureanu BS, Copăescu C, Săftoiu A. Technical Performance, Overall Accuracy and Complications of EUS-Guided Interventional Procedures: A Dynamic Landscape. Diagnostics (Basel). 2022;12:1641. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 9. | Conti Bellocchi MC, Bernuzzi M, Brillo A, Bernardoni L, Amodio A, De Pretis N, Frulloni L, Gabbrielli A, Crinò SF. EUS-FNA versus EUS-FNB in Pancreatic Solid Lesions ≤ 15 mm. Diagnostics (Basel). 2024;14:427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (3)] |
| 10. | Jenssen C, Alvarez-Sánchez MV, Napoléon B, Faiss S. Diagnostic endoscopic ultrasonography: assessment of safety and prevention of complications. World J Gastroenterol. 2012;18:4659-4676. [PubMed] [DOI] [Full Text] |
| 11. | Conti CB, Cereatti F, Grassia R. Endoscopic ultrasound-guided sampling of solid pancreatic masses: the fine needle aspiration or fine needle biopsy dilemma. Is the best needle yet to come? World J Gastrointest Endosc. 2019;11:454-471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 17] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 12. | Tian G, Ye Z, Zhao Q, Jiang T. Complication incidence of EUS-guided pancreas biopsy: A systematic review and meta-analysis of 11 thousand population from 78 cohort studies. Asian J Surg. 2020;43:1049-1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 13. | Eloubeidi MA, Chen VK, Eltoum IA, Jhala D, Chhieng DC, Jhala N, Vickers SM, Wilcox CM. Endoscopic ultrasound-guided fine needle aspiration biopsy of patients with suspected pancreatic cancer: diagnostic accuracy and acute and 30-day complications. Am J Gastroenterol. 2003;98:2663-2668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 284] [Article Influence: 12.3] [Reference Citation Analysis (3)] |
| 14. | Eloubeidi MA, Tamhane A, Varadarajulu S, Wilcox CM. Frequency of major complications after EUS-guided FNA of solid pancreatic masses: a prospective evaluation. Gastrointest Endosc. 2006;63:622-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 207] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 15. | Carrara S, Arcidiacono PG, Mezzi G, Petrone MC, Boemo C, Testoni PA. Pancreatic endoscopic ultrasound-guided fine needle aspiration: complication rate and clinical course in a single centre. Dig Liver Dis. 2010;42:520-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 52] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 16. | Hamada T, Yasunaga H, Nakai Y, Isayama H, Horiguchi H, Matsuda S, Fushimi K, Koike K. Rarity of severe bleeding and perforation in endoscopic ultrasound-guided fine needle aspiration for submucosal tumors. Dig Dis Sci. 2013;58:2634-2638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 17. | Cheng B, Zhang Y, Chen Q, Sun B, Deng Z, Shan H, Dou L, Wang J, Li Y, Yang X, Jiang T, Xu G, Wang G. Analysis of Fine-Needle Biopsy vs Fine-Needle Aspiration in Diagnosis of Pancreatic and Abdominal Masses: A Prospective, Multicenter, Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2018;16:1314-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 113] [Article Influence: 14.1] [Reference Citation Analysis (2)] |
| 18. | de Moura DTH, McCarty TR, Jirapinyo P, Ribeiro IB, Flumignan VK, Najdawai F, Ryou M, Lee LS, Thompson CC. EUS-guided fine-needle biopsy sampling versus FNA in the diagnosis of subepithelial lesions: a large multicenter study. Gastrointest Endosc. 2020;92:108-119.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 19. | Saumoy M, Kahaleh M. Safety and Complications of Interventional Endoscopic Ultrasound. Clin Endosc. 2018;51:235-238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 20. | Lakhtakia S. Complications of diagnostic and therapeutic Endoscopic Ultrasound. Best Pract Res Clin Gastroenterol. 2016;30:807-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (5)] |
| 21. | ASGE Training Committee; Jorgensen J, Kubiliun N, Law JK, Al-Haddad MA, Bingener-Casey J, Christie JA, Davila RE, Kwon RS, Obstein KL, Qureshi WA, Sedlack RE, Wagh MS, Zanchetti D, Coyle WJ, Cohen J. Endoscopic retrograde cholangiopancreatography (ERCP): core curriculum. Gastrointest Endosc. 2016;83:279-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 22. | Rosenthal LS. Is a fourth year of training necessary to become competent in EUS and ERCP? Notes from the 2008 class of advanced endoscopy fellows. Gastrointest Endosc. 2008;68:1150-1152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 23. | Wang F, Hao J, Wei K, Zhou C, Geng Z, Du Z, Sun H, Wang Z, Ma Q, Wu Z. Comparative diagnostic efficacy and safety of ultrasound-guided percutaneous transhepatic biopsy and endoscopic ultrasound-guided fine-needle aspiration biopsy for gallbladder tumors. Sci Rep. 2025;15:12155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 24. | Lee H, Seo D, Song T, Oh D, Huh G, Cho S. Clinical outcomes, complications and risk factors for comorbidities of eus-guided through-the-needle biopsy in pancreatic cystic lesions in a large cohort. Gastrointest Endosc. 2024;99:AB788-AB789. [DOI] [Full Text] |
| 25. | Benson ME, Byrne S, Brust DJ, Manning B 3rd, Pfau PR, Frick TJ, Reichelderfer M, Gopal DV. EUS and ERCP complication rates are not increased in elderly patients. Dig Dis Sci. 2010;55:3278-3283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 26. | Amini A, Lopez RA. Duodenal Perforation. 2023 Mar 13. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. [PubMed] |
| 27. | Lin YC, Yen HH, Huang SP, Shih KL, Chen YY. Comparison of Adverse Events of Different Endoscopic Ultrasound-Guided Tissue Acquisition Methods: A Single-Center Retrospective Analysis. Diagnostics (Basel). 2022;12:2123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 28. | Kitano M, Sakamoto H, Kudo M. Endoscopic ultrasound: contrast enhancement. Gastrointest Endosc Clin N Am. 2012;22:349-358, xi. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 29. | Saftoiu A, Napoleon B, Arcidiacono PG, Braden B, Burmeister S, Carrara S, Cui XW, Fusaroli P, Gottschalk U, Hocke M, Hollerbach S, Iglesias-Garcia J, Jenssen C, Kitano M, Larghi A, Oppong KW, Sahai AV, Sun S, Burmester E, Di Leo M, Petrone MC, Santos E, Teoh AYB, Hwang JH, Rimbas M, Sharma M, Puri R, Kahaleh M, Dietrich CF. Do we need contrast agents for EUS? Endosc Ultrasound. 2020;9:361-368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (1)] |