Published online Mar 16, 2026. doi: 10.4253/wjge.v18.i3.116958
Revised: January 7, 2026
Accepted: January 21, 2026
Published online: March 16, 2026
Processing time: 108 Days and 9.3 Hours
Although Helicobacter pylori (H. pylori) infection is widely known to induce chronic gastric inflammation, its contribution to low-grade sustained systemic inflammation remains unclear.
To investigate the effect of H. pylori eradication on systemic inflammatory activity through the analysis of serum cytokine levels in patients with either gastritis (GA) or duodenal ulcer (DU).
We included fifty-four patients who underwent upper gastrointestinal endoscopy for dyspepsia and were diagnosed with either H. pylori-positive GA or DU. Serum cytokine levels were measured before and after bacterial eradication using enzyme-linked immunosorbent assay. Paired comparisons within each group (GA and DU) were performed using Wilcoxon signed-rank test. Differences between groups at baseline and post-treatment were analyzed using Mann-Whitney U test. To account for multiple testing across cytokines, P values were adjusted using the false discovery rate approach according to the Benjamini-Hochberg procedure.
In the GA group, all measured cytokines: Interleukin (IL)-1α, IL-1β, IL-2, IL-6, IL-8, IL-10, IL-12p70, IL-23, interferon gamma (IFN-γ), and tumor necrosis factor-alpha (TNF-α)-showed significant post-treatment reductions. In the DU group, most cytokines decreased, but IL-2 and IL-12p70 did not reach sta
Our findings suggest that H. pylori eradication is associated with reduced pro-inflammatory cytokine levels in patients with GA and with a distinct shift toward an anti-inflammatory profile in patients with DU, highlighting a broader systemic immunological context beyond the gastric niche.
Core Tip: Helicobacter pylori (H. pylori) infection is consistently associated with chronic gastric inflammation and may also relate to sustained low-grade systemic inflammatory activity. In this study, distinct circulating cytokine patterns were observed in patients with gastritis (GA) and duodenal ulcer (DU) following bacterial eradication, with attenuation of pro-inflammatory mediators in GA and a regulatory-skewed cytokine profile in DU. These findings highlight a potential systemic immunological dimension of H. pylori-associated disease, with relevance for inflammation-related conditions beyond the gastric compartment.
- Citation: de Melo FF, Lemos FFB, Leal RAOS, Rocha GA, de Magalhães Queiroz DM. Helicobacter pylori eradication is associated with systemic anti-inflammatory shift in duodenal ulcer patients compared to those with gastritis. World J Gastrointest Endosc 2026; 18(3): 116958
- URL: https://www.wjgnet.com/1948-5190/full/v18/i3/116958.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i3.116958
Helicobacter pylori (H. pylori) is a spiral-shaped, gram-negative bacterium that colonizes nearly half of the global po
Persistent antigenic stimulation during H. pylori colonization sustains a chronic state of inflammation in the gastric mucosa, initiated through pro-inflammatory cytokines such as interleukin (IL)-1β, IL-6, and tumor necrosis factor-alpha (TNF-α)[7]. Cytokines such as IL-12 induce differentiation of Th1 cells, which produce interferon gamma (IFN-γ), es
Beyond the gastric niche, the broader systemic impact of this cytokine network remains incompletely understood. Nonetheless, H. pylori infection appears to contribute to a state of clinically relevant low-grade systemic inflammation, associated with extra-gastric conditions such as metabolic, cardiovascular, and autoimmune diseases[15-17]. Supporting this view, our group previously demonstrated that bacterial eradication in patients with idiopathic thrombocytopenic purpura improved platelet counts and altered circulating cytokine profiles, suggesting that clearance of the infection can influence immune responses beyond the stomach[18].
Although eradication reverses gastric mucosal inflammation, its effects on systemic cytokine profiles in patients with gastric manifestations is unclear. It is also uncertain whether individuals with GA and those with duodenal ulcer (DU) exhibit distinct systemic immunological responses after treatment. In this study, we investigated circulating cytokine profiles in patients with GA or DU before and after H. pylori eradication. Comparative analysis of pre- and post-treatment cytokine profiles may uncover broader systemic immunological signatures associated with bacterial clearance, with potential implications for low-grade systemic inflammation and extra- gastric conditions.
We included fifty-four adult patients with confirmed H. pylori infection who demonstrated successful eradication following standard therapy. All participants had undergone upper gastrointestinal endoscopy due to dyspeptic sy
This research was conducted in full compliance with the Declaration of Helsinki and received approval from the Ethics Committee of the Federal University of Minas Gerais, Brazil. Written informed consent was obtained from all participants prior to inclusion.
Clinical and demographic data, including histopathological diagnosis, age, and sex of the patients, were meticulously collected from medical records. Concurrently, blood samples were obtained from all patients at the time of upper gastrointestinal endoscopy and subsequently collected again six months post-eradication therapy, coinciding with the ¹³C-UBT. These samples were immediately processed to isolate serum, which was carefully aliquoted and preserved at -80 °C until further analysis.
Serum levels of IL-1α, IL-1β, IL-2, IL-6, IL-8, IL-10, IL-12p70, IL-23, IFN-γ, and TNF-α were measured using commercially available enzyme-linked immunosorbent assay kits (Biosource, Camarillo, CA, United States), following the manu
All statistical analyses were performed using GraphPad Prism (GraphPad Software, San Diego, CA, United States). Categorical variables were compared using Pearson’s χ2 test or Fisher’s exact test, as appropriate, and effect sizes were expressed as odds ratios. Given the non-normal distribution of cytokine data and the moderate sample sizes (GA, n = 33; DU, n = 21), nonparametric tests were used throughout. Paired pre- and post-treatment comparisons within each clinical group were performed using the Wilcoxon signed-rank test. Comparisons between independent groups (GA vs DU), at both baseline and post-treatment, were conducted using the Mann-Whitney U test. All tests were two-tailed.
To account for multiple testing across cytokines, P-values were adjusted using the false discovery rate (FDR) according to the Benjamini-Hochberg procedure. FDR correction was applied separately to each predefined family of comparisons: (1) GA vs DU at baseline; (2) GA vs DU post-treatment; (3) Pre- vs post-treatment comparisons within the GA group; and (4) Pre- vs post-treatment comparisons within the DU group. An FDR-adjusted P-value < 0.05 was considered statistically significant. Effect sizes for nonparametric comparisons were calculated using the r coefficient (r = z/√n), where n denotes the total number of observations for Mann-Whitney tests and the number of paired observations for Wilcoxon tests[19].
Age distribution was similar between groups, with median ages of 45.0 years [interquartile range (IQR) = 16] in the GA group and 38.0 years (IQR = 25) in the DU group. Mean age was 43.5 ± 13.4 years for GA and 42.5 ± 15.0 years for DU patients, with overlapping 95%CI (37.4-49.6 and 37.2-47.9, respectively). Females accounted for 68.5% of the total sample, representing 81.0% of GA patients and 60.6% of DU patients. No statistically significant differences were observed between groups in terms of age or sex distribution.
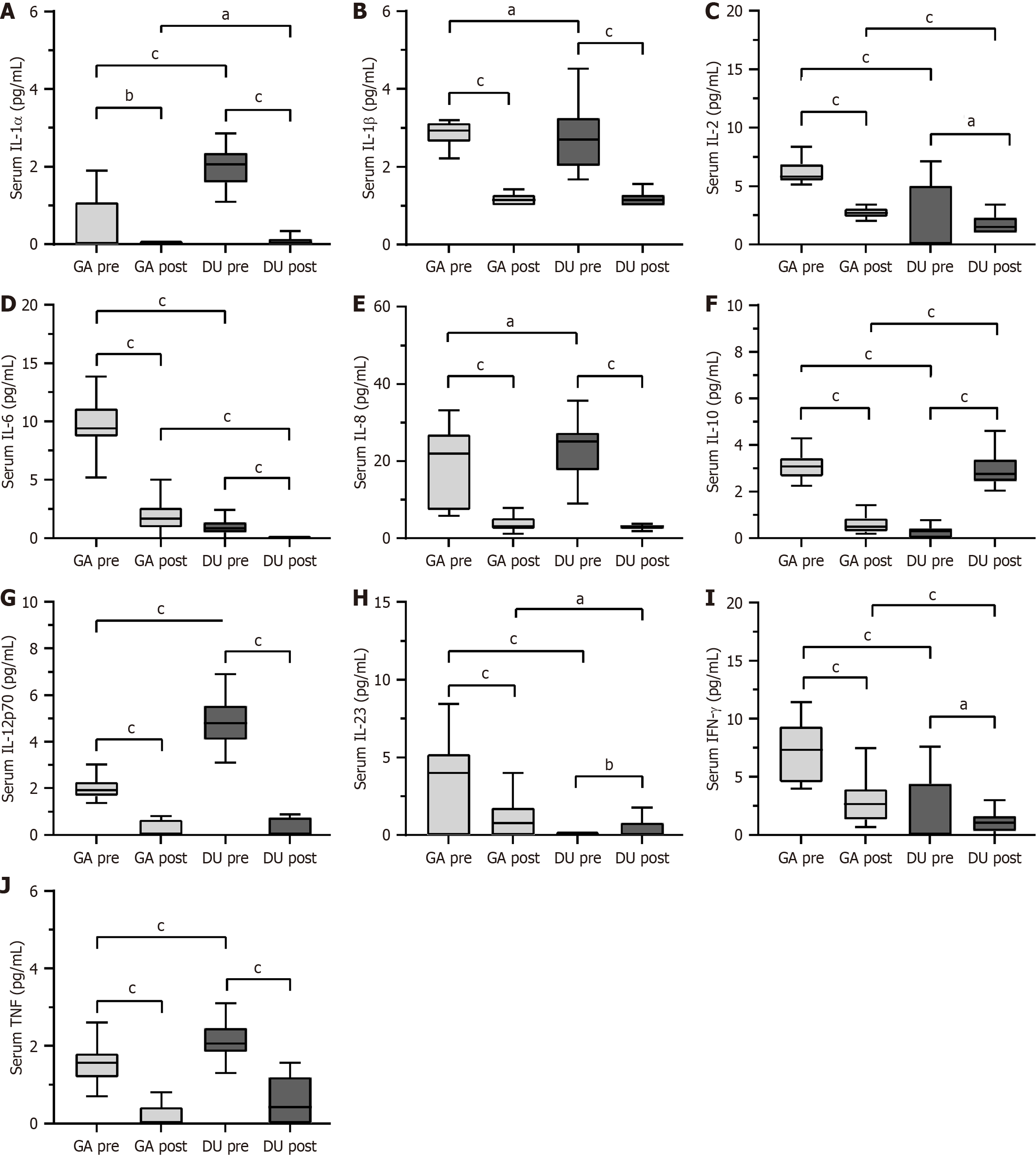
In patients with GA, H. pylori eradication was associated with a consistent decrease across all measured circulating cytokines (Figure 1 and Table 1). These decreases involved both pro-inflammatory and regulatory mediators and were predominantly associated with large within-group effect sizes, with moderate effects observed for a smaller subset of cytokines.
| Cytokine | Group | Pre-treatment | Post-treatment | FDR-adjusted P value1 | r2 | ||||
| Median | Q1 (25th) | Q3 (75th) | Median | Q1 (25th) | Q3 (75th) | ||||
| IL-1α | GA | 0.00 | 0.00 | 1.08 | 0.00 | 0.00 | 0.04 | 0.001 | 0.52 |
| DU | 1.79 | 1.59 | 2.39 | 0.04 | 0.00 | 0.14 | < 0.001 | 0.85 | |
| IL-1β | GA | 2.93 | 2.65 | 3.13 | 1.01 | 1.01 | 1.28 | < 0.001 | 0.88 |
| DU | 2.70 | 2.07 | 3.30 | 1.15 | 1.01 | 1.28 | 0.017 | 0.87 | |
| IL-2 | GA | 5.82 | 5.65 | 6.86 | 2.72 | 2.55 | 3.06 | < 0.001 | 0.86 |
| DU | 0.00 | 0.00 | 4.90 | 1.34 | 1.00 | 1.68 | < 0.001 | 0.33 | |
| IL-6 | GA | 9.81 | 9.14 | 11.66 | 1.66 | 1.11 | 3.33 | < 0.001 | 0.88 |
| DU | 0.67 | 0.43 | 1.16 | 0.00 | 0.00 | 0.18 | < 0.001 | 0.78 | |
| IL-8 | GA | 16.62 | 7.20 | 25.87 | 3.20 | 2.53 | 5.20 | < 0.001 | 0.86 |
| DU | 25.00 | 17.52 | 27.56 | 3.20 | 2.53 | 3.20 | < 0.001 | 0.87 | |
| IL-10 | GA | 2.96 | 2.45 | 3.17 | 0.49 | 0.28 | 0.54 | < 0.001 | 0.88 |
| DU | 0.30 | 0.00 | 0.42 | 2.76 | 2.45 | 3.17 | < 0.001 | 0.87 | |
| IL-12p70 | GA | 1.93 | 1.80 | 2.40 | 0.00 | 0.00 | 0.35 | < 0.001 | 0.83 |
| DU | 4.68 | 4.09 | 5.69 | 0.00 | 0.00 | 0.77 | < 0.001 | 0.87 | |
| IL-23 | GA | 4.00 | 2.00 | 6.22 | 0.00 | 0.00 | 1.28 | < 0.001 | 0.76 |
| DU | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.80 | 0.007 | 0.38 | |
| IFN-γ | GA | 8.57 | 5.53 | 10.89 | 3.57 | 2.14 | 4.28 | < 0.001 | 0.88 |
| DU | 4.00 | 0.00 | 5.80 | 0.71 | 0.35 | 1.42 | 0.011 | 0.37 | |
| TNF-α | GA | 1.57 | 1.19 | 1.96 | 0.42 | 0.03 | 0.80 | < 0.001 | 0.87 |
| DU | 1.90 | 1.85 | 2.26 | 0.42 | 0.03 | 1.19 | < 0.001 | 0.87 | |
In contrast, patients with DU exhibited a more heterogeneous cytokine response to eradication (Figure 1 and Table 1). Post-treatment decreases were observed for IL-1α, IL-1β, IL-6, IL-8, IL-12p70, IFN-γ, and TNF-α, with large effects for several cytokines and moderate-to-small effects for others. Notably, IL-10 showed a marked post-treatment increase with a large effect size, whereas IL-2 and IL-23 showed small-effect post-treatment increases.
At baseline, distinct differences in circulating cytokine profiles were observed between clinical phenotypes (Figure 1 and Table 2). Patients with DU exhibited higher serum levels of IL-1α, associated with a moderate between-group effect, and IL-12p70, associated with a large effect, whereas patients with GA showed higher baseline concentrations of IL-6 and IL-10, both with large effects, and higher levels of IL-2, IFN-γ, IL-23, and TNF-α, predominantly with moderate effects. No significant differences between groups were found for IL-1β and IL-8.
| Cytokine | Group | Pre-treatment | FDR-adjusted P value (pre-tx)1 | r (pre-tx)2 | Post-treatment | FDR-adjusted P value (post-tx)1 | r (post-tx)2 | ||||
| Median | Q1 | Q3 | Median | Q1 | Q3 | ||||||
| IL-1α | GA | 0.00 | 0.00 | 1.08 | < 0.001 | 0.70 | 0.00 | 0.00 | 0.04 | 0.012 | 0.34 |
| DU | 1.79 | 1.59 | 2.39 | 0.04 | 0.00 | 0.14 | |||||
| IL-1β | GA | 2.93 | 2.65 | 3.13 | 0.013 | 0.25 | 1.01 | 1.01 | 1.28 | 0.95 | 0.01 |
| DU | 2.70 | 2.07 | 3.30 | 1.15 | 1.01 | 1.28 | |||||
| IL-2 | GA | 5.82 | 5.65 | 6.86 | < 0.001 | 0.61 | 2.72 | 2.55 | 3.06 | < 0.001 | 0.66 |
| DU | 0.00 | 0.00 | 4.90 | 1.34 | 1.00 | 1.68 | |||||
| IL-6 | GA | 9.81 | 9.14 | 11.66 | < 0.001 | 0.83 | 1.66 | 1.11 | 3.33 | < 0.001 | 0.78 |
| DU | 0.67 | 0.43 | 1.16 | 0.00 | 0.00 | 0.18 | |||||
| IL-8 | GA | 16.62 | 7.20 | 25.87 | 0.013 | 0.25 | 3.20 | 2.53 | 5.20 | 0.14 | 0.20 |
| DU | 25.00 | 17.52 | 27.56 | 3.20 | 2.53 | 3.20 | |||||
| IL-10 | GA | 2.96 | 2.45 | 3.17 | < 0.001 | 0.84 | 0.49 | 0.28 | 0.54 | < 0.001 | 0.84 |
| DU | 0.30 | 0.00 | 0.42 | 2.76 | 2.45 | 3.17 | |||||
| IL-12p70 | GA | 1.93 | 1.80 | 2.40 | < 0.001 | 0.82 | 0.00 | 0.00 | 0.35 | 0.15 | 0.20 |
| DU | 4.68 | 4.09 | 5.69 | 0.00 | 0.00 | 0.77 | |||||
| IL-23 | GA | 4.00 | 2.00 | 6.22 | < 0.001 | 0.72 | 0.00 | 0.00 | 1.28 | 0.035 | 0.29 |
| DU | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.80 | |||||
| IFN-γ | GA | 8.57 | 5.53 | 10.89 | < 0.001 | 0.66 | 3.57 | 2.14 | 4.28 | < 0.001 | 0.56 |
| DU | 4.00 | 0.00 | 5.80 | 0.71 | 0.35 | 1.42 | |||||
| TNF-α | GA | 1.57 | 1.19 | 1.96 | < 0.001 | 0.53 | 0.42 | 0.03 | 0.80 | 0.08 | 0.24 |
| DU | 1.90 | 1.85 | 2.26 | 0.42 | 0.03 | 1.19 | |||||
Post-eradication, cytokine profiles showed a broadly convergent distribution between groups (Figure 1 and Table 2). No differences were observed for IL-1β, IL-8, IL-12p70, or TNF-α. IL-1α remained marginally higher in the DU group, with a small between-group effect. Conversely, IL-6, IL-2, and IFN-γ levels remained higher in the GA group after eradication, corresponding to moderate between-group effects. Notably, IL-10 displayed an inverse pattern, with higher post-treatment levels observed in patients with DU, reflecting a large between-group effect (Figure 1).
Our findings suggest that H. pylori eradication is associated with significant modulation of systemic cytokine levels in patients with GA and DU, reinforcing the hypothesis that the bacterium may have immunological effects beyond the gastric mucosa. The consistent decline in pro-inflammatory cytokines following bacterial clearance highlights the broader immunomodulatory impact of eradication therapy and may be relevant to low-grade systemic inflammation. Impor
Among patients with GA, eradication was associated with a marked reduction across all measured cytokines, including key pro-inflammatory mediators such as IL-1β, IL-6, IL-8, TNF-α, and IFN-γ. The decline in IL-12p70 and IL-23-cytokines central to Th1 and Th17 polarization—may be consistent with broader changes in T-helper-related signaling associated with bacterial persistence and clearance[20-22]. Interestingly, the decline in IL-10 levels, traditionally regarded as anti-inflammatory, may reflect the resolution of an immunoregulatory feedback loop previously activated to restrain chronic inflammation[13]. This pattern aligns with the systemic immune downregulation expected after pathogen clearance.
In the DU group, cytokine modulation also followed a predominantly suppressive trend, with reductions in IL-1α, IL-1β, IL-6, IL-8, IL-12p70, and TNF-α. However, distinct from the GA group, these patients exhibited a post-eradication increase in IL-10 and IL-23, despite an overall reduction in most pro-inflammatory markers. The increase in IL-10 may indicate an active immunoregulatory response aimed at restoring homeostasis in the aftermath of ulcer-associated mucosal injury[23,24]. In addition, IL-2 showed a modest post-treatment increase, whereas IFN-γ decreased to a lesser extent than observed in GA (Table 1).
Concurrently, the modest elevation of IL-23, together with the increase in IL-2 and a less pronounced decrease in IFN-γ, may suggest a shift from a predominantly Th1-associated profile toward a more regulatory phenotype during re
Baseline comparisons further reinforce this distinction: Patients with DU exhibited significantly higher levels of IL-1α and IL-12p70 and notably lower levels of IL-6, IL-2, IL-10, and IFN-γ, as well as IL-23. This pattern suggests a strong Th1-driving stimulus in DU, as indicated through elevated IL-12p70, but paradoxically lower systemic IFN-γ levels may reflect increased local consumption or sequestration of IFN-γ at ulcer sites, or tighter immunoregulatory control limiting its systemic presence. In contrast, higher circulating IFN-γ in GA may indicate more diffuse or sustained systemic Th1 activation without focal tissue damage. The markedly lower IL-10 levels in DU patients further suggest reduced systemic regulatory tone, which, along with localized IFN-γ activity, may contribute to ulcer-associated inflammatory patterns.
After treatment, the convergence of cytokine profiles between groups-particularly for IL-1β, IL-8, IL-12p70, and TNF-α is consistent with partial normalization of systemic immune parameters after eradication therapy, regardless of the initial clinical manifestation. Nevertheless, some differences persisted: The GA group maintained higher levels of IL-6, IL-2, and IFN-γ, whereas IL-10 remained elevated in the DU group. These lingering disparities may reflect residual immune imprinting or distinct healing trajectories in the gastric vs duodenal mucosa.
Altogether, our data support that H. pylori eradication is associated with disease-specific systemic cytokine changes in GA and DU. The distinct profiles observed in GA and DU may reflect partially divergent immunopathological processes, despite their origin in a shared infection. Moreover, the profound changes in circulating cytokines reinforce the systemic reach of H. pylori associated immune activation and underscore the potential relevance of eradication therapy for un
Our study has some limitations. Because this was an observational analysis without a non-eradication comparator and without H. pylori-negative or healthy controls, causal inference is limited and the independent contribution of infection vs underlying phenotype cannot be fully separated. Given the modest sample size and evaluation of multiple cytokines, the analyses should be considered exploratory despite FDR adjustment, and validation in larger, independent cohorts is warranted. In addition, key potential confounders (e.g., smoking status, body mass index, and medication exposures such as nonsteroidal anti-inflammatory drugs) were not comprehensively assessed, and may have influenced between-group differences. We also did not evaluate clinical outcomes, and the clinical threshold significance of the observed cytokine shifts remains undefined. Finally, treatment-related factors (antibiotics and proton pump inhibition) may influence cy
Our findings suggest that the eradication of H. pylori is associated with lower circulating levels of pro-inflammatory cytokines in patients with GA and is associated with a shift toward an anti-inflammatory cytokine profile in those with DU. These distinct immunomodulatory patterns may be consistent with differences in systemic immune activation across clinical phenotypes but should be interpreted cautiously given the observational design. The observed post-eradication changes may be relevant to low-grade systemic inflammation, which has been implicated in the pathogenesis of various chronic diseases; however, clinical outcomes were not assessed, and the clinical implications of these cytokine changes remain to be established. These results reinforce the systemic relevance of H. pylori eradication beyond its local impact on gastric mucosa and support further validation in larger controlled studies.
| 1. | Li Y, Choi H, Leung K, Jiang F, Graham DY, Leung WK. Global prevalence of Helicobacter pylori infection between 1980 and 2022: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2023;8:553-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 392] [Cited by in RCA: 332] [Article Influence: 110.7] [Reference Citation Analysis (9)] |
| 2. | Malfertheiner P, Camargo MC, El-Omar E, Liou JM, Peek R, Schulz C, Smith SI, Suerbaum S. Helicobacter pylori infection. Nat Rev Dis Primers. 2023;9:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 773] [Cited by in RCA: 639] [Article Influence: 213.0] [Reference Citation Analysis (18)] |
| 3. | Usui Y, Taniyama Y, Endo M, Koyanagi YN, Kasugai Y, Oze I, Ito H, Imoto I, Tanaka T, Tajika M, Niwa Y, Iwasaki Y, Aoi T, Hakozaki N, Takata S, Suzuki K, Terao C, Hatakeyama M, Hirata M, Sugano K, Yoshida T, Kamatani Y, Nakagawa H, Matsuda K, Murakami Y, Spurdle AB, Matsuo K, Momozawa Y. Helicobacter pylori, Homologous-Recombination Genes, and Gastric Cancer. N Engl J Med. 2023;388:1181-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 175] [Article Influence: 58.3] [Reference Citation Analysis (3)] |
| 4. | Lemos FFB, de Castro CT, Calmon MS, Silva Luz M, Pinheiro SLR, Faria Souza Mendes Dos Santos C, Correa Santos GL, Marques HS, Delgado HA, Teixeira KN, Souza CL, Oliveira MV, Freire de Melo F. Effectiveness of Helicobacter pylori eradication in the treatment of early-stage gastric mucosa-associated lymphoid tissue lymphoma: An up-to-date meta-analysis. World J Gastroenterol. 2023;29:2202-2221. [PubMed] [DOI] [Full Text] |
| 5. | Almadi MA, Lu Y, Alali AA, Barkun AN. Peptic ulcer disease. Lancet. 2024;404:68-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 47] [Article Influence: 23.5] [Reference Citation Analysis (1)] |
| 6. | Wu X, Gou G, Wen M, Wang F, Liu Y, Li L, Xu J, Xie R. The immunoregulatory role of helper T cells in Helicobacter pylori infection. Front Immunol. 2025;16:1593727. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 7. | Gobert AP, Wilson KT. Induction and Regulation of the Innate Immune Response in Helicobacter pylori Infection. Cell Mol Gastroenterol Hepatol. 2022;13:1347-1363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 8. | Bamford KB, Fan X, Crowe SE, Leary JF, Gourley WK, Luthra GK, Brooks EG, Graham DY, Reyes VE, Ernst PB. Lymphocytes in the human gastric mucosa during Helicobacter pylori have a T helper cell 1 phenotype. Gastroenterology. 1998;114:482-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 415] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 9. | D'Elios MM, Manghetti M, De Carli M, Costa F, Baldari CT, Burroni D, Telford JL, Romagnani S, Del Prete G. T helper 1 effector cells specific for Helicobacter pylori in the gastric antrum of patients with peptic ulcer disease. J Immunol. 1997;158:962-967. [PubMed] [DOI] [Full Text] |
| 10. | Bagheri N, Salimzadeh L, Shirzad H. The role of T helper 1-cell response in Helicobacter pylori-infection. Microb Pathog. 2018;123:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (2)] |
| 11. | Khamri W, Walker MM, Clark P, Atherton JC, Thursz MR, Bamford KB, Lechler RI, Lombardi G. Helicobacter pylori stimulates dendritic cells to induce interleukin-17 expression from CD4+ T lymphocytes. Infect Immun. 2010;78:845-853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 85] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 12. | Mizuno T, Ando T, Nobata K, Tsuzuki T, Maeda O, Watanabe O, Minami M, Ina K, Kusugami K, Peek RM, Goto H. Interleukin-17 levels in Helicobacter pylori-infected gastric mucosa and pathologic sequelae of colonization. World J Gastroenterol. 2005;11:6305-6311. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 78] [Cited by in RCA: 81] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 13. | Freire de Melo F, Rocha AM, Rocha GA, Pedroso SH, de Assis Batista S, Fonseca de Castro LP, Carvalho SD, Bittencourt PF, de Oliveira CA, Corrêa-Oliveira R, Magalhães Queiroz DM. A regulatory instead of an IL-17 T response predominates in Helicobacter pylori-associated gastritis in children. Microbes Infect. 2012;14:341-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 14. | Rocha GA, de Melo FF, Cabral MMDA, de Brito BB, da Silva FAF, Queiroz DMM. Interleukin-27 is abrogated in gastric cancer, but highly expressed in other Helicobacter pylori-associated gastroduodenal diseases. Helicobacter. 2020;25:e12667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 15. | Azami M, Baradaran HR, Dehghanbanadaki H, Kohnepoushi P, Saed L, Moradkhani A, Moradpour F, Moradi Y. Association of Helicobacter pylori infection with the risk of metabolic syndrome and insulin resistance: an updated systematic review and meta-analysis. Diabetol Metab Syndr. 2021;13:145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 16. | Sun L, Zheng H, Qiu M, Hao S, Liu X, Zhu X, Cai X, Huang Y. Helicobacter pylori infection and risk of cardiovascular disease. Helicobacter. 2023;28:e12967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 17. | Wang L, Cao ZM, Zhang LL, Dai XC, Liu ZJ, Zeng YX, Li XY, Wu QJ, Lv WL. Helicobacter Pylori and Autoimmune Diseases: Involving Multiple Systems. Front Immunol. 2022;13:833424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 74] [Article Influence: 18.5] [Reference Citation Analysis (1)] |
| 18. | Rocha AM, Souza C, Melo FF, Clementino NC, Marino MC, Rocha GA, Queiroz DM. Cytokine profile of patients with chronic immune thrombocytopenia affects platelet count recovery after Helicobacter pylori eradication. Br J Haematol. 2015;168:421-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 19. | Lakens D. Calculating and reporting effect sizes to facilitate cumulative science: a practical primer for t-tests and ANOVAs. Front Psychol. 2013;4:863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7466] [Cited by in RCA: 5734] [Article Influence: 441.1] [Reference Citation Analysis (1)] |
| 20. | Athie-Morales V, Smits HH, Cantrell DA, Hilkens CM. Sustained IL-12 signaling is required for Th1 development. J Immunol. 2004;172:61-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 171] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 21. | Stritesky GL, Yeh N, Kaplan MH. IL-23 promotes maintenance but not commitment to the Th17 lineage. J Immunol. 2008;181:5948-5955. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 339] [Cited by in RCA: 323] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 22. | Moyat M, Velin D. Immune responses to Helicobacter pylori infection. World J Gastroenterol. 2014;20:5583-5593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 73] [Cited by in RCA: 96] [Article Influence: 8.0] [Reference Citation Analysis (2)] |
| 23. | Nguyen HD, Aljamaei HM, Stadnyk AW. The Production and Function of Endogenous Interleukin-10 in Intestinal Epithelial Cells and Gut Homeostasis. Cell Mol Gastroenterol Hepatol. 2021;12:1343-1352. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (5)] |
| 24. | Liu S, Deng Z, Zhu J, Ma Z, Tuo B, Li T, Liu X. Gastric immune homeostasis imbalance: An important factor in the development of gastric mucosal diseases. Biomed Pharmacother. 2023;161:114338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 25. | Schnell A, Littman DR, Kuchroo VK. T(H)17 cell heterogeneity and its role in tissue inflammation. Nat Immunol. 2023;24:19-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 203] [Article Influence: 67.7] [Reference Citation Analysis (0)] |
| 26. | Wang Y, Xue N, Wang Z, Zeng X, Ji N, Chen Q. Targeting Th17 cells: a promising strategy to treat oral mucosal inflammatory diseases. Front Immunol. 2023;14:1236856. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 27. | Cui H, Wang N, Li H, Bian Y, Wen W, Kong X, Wang F. The dynamic shifts of IL-10-producing Th17 and IL-17-producing Treg in health and disease: a crosstalk between ancient "Yin-Yang" theory and modern immunology. Cell Commun Signal. 2024;22:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 51] [Article Influence: 25.5] [Reference Citation Analysis (0)] |