Published online Mar 16, 2026. doi: 10.4253/wjge.v18.i3.116060
Revised: November 23, 2025
Accepted: January 15, 2026
Published online: March 16, 2026
Processing time: 132 Days and 8.9 Hours
Self-expandable metallic stent (SEMS) placement is a palliative treatment for ino
To evaluate the technical success of using the vertebral column and diaphragm landmarks for SEMS placement.
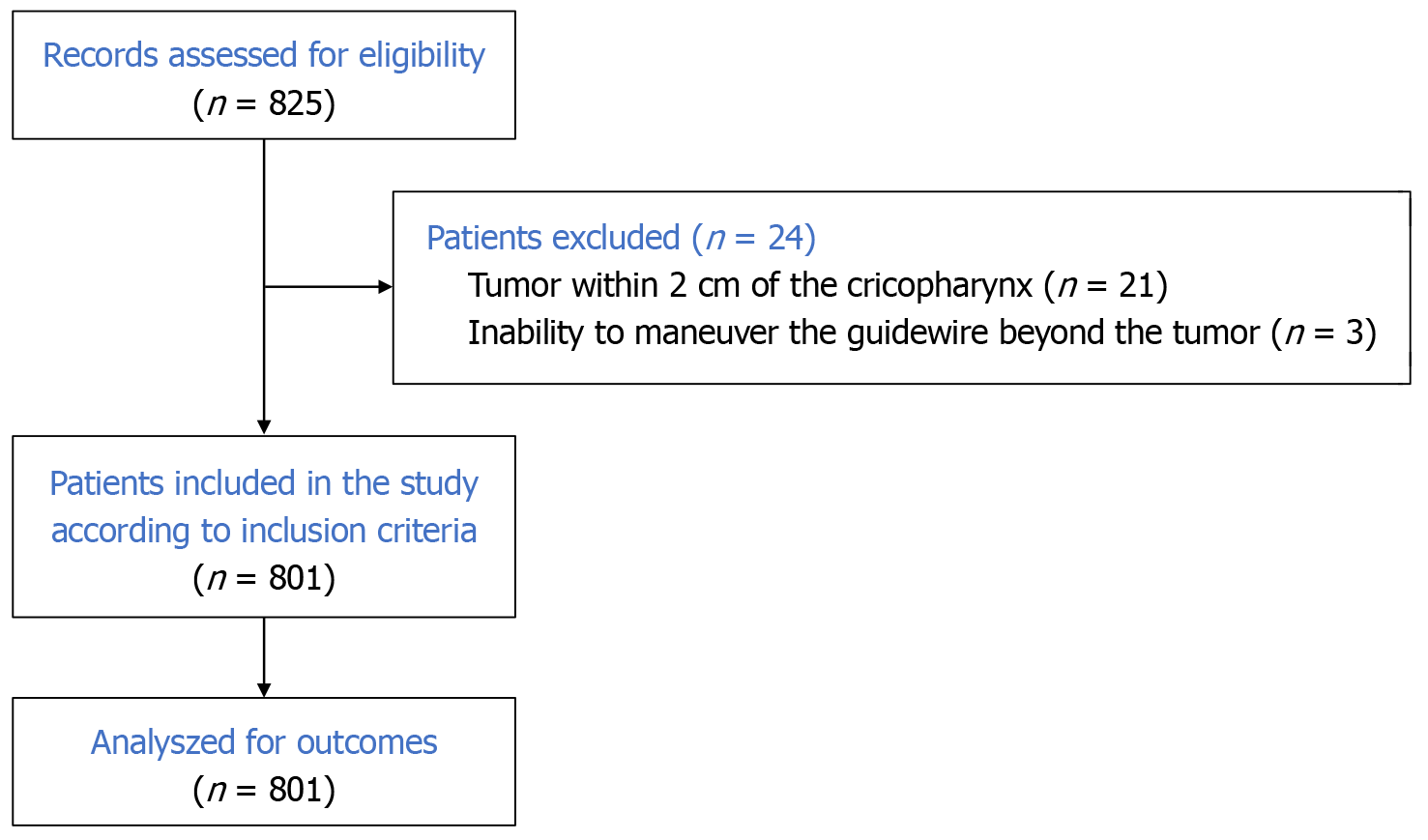
This retrospective cross-sectional study included 801 patients with inoperable esophageal malignancies who underwent SEMS placement for dysphagia due to malignant esophageal strictures from January 1, 2006, to December 31, 2024. The SEMS placement procedure utilized the vertebral column and diaphragm as land
Among 801 patients (18-95 years; mean 50 ± 15 years), 408 (50.90%) were female. All presented with dysphagia (average duration: 3.84 months). Squamous cell carcinoma was the predominant etiology (594; 74.16%), followed by adenocarcinoma (207; 25.84%). Tumors involved the middle third of the esophagus in 365 patients (45.57%), and distant metastasis was the leading cause of inoperability (52.81%). Gastroesophageal junction involvement was observed in 240 patients (30.00%). Tracheoesophageal fistula was present in 48 cases (6.00%). Mean stricture length was 8.4 cm. 139 patients (17.40%) required pre-stent dilatation. SEMS length ranged from 8 cm to 18 cm. Technical success was 100% without major immediate adverse events. Repeat stenting was needed in 15 patients (1.87%) due to tumor overgrowth, and 11 (1.37%) required dilatation for tumor ingrowth.
Using the vertebral column and diaphragm as landmarks provides a simplified, safe, and reproducible method for SEMS placement. Larger prospective studies are needed to validate these results and assess long-term outcomes.
Core Tip: A novel, landmark-based technique for self-expandable metallic stent placement in patients with inoperable esophageal cancer was investigated in this study. By utilizing the vertebral column and diaphragm as anatomical guides, the procedure became more streamlined, cost-effective, and reproducible without compromising safety or technical success. We achieved a 100% success rate and minimal complications in 801 patients, indicating that this approach offers a practical al
- Citation: Siyal M, Asim M, Qureshi S, Ghazanfar S, Siddiqui AR, Ahmed N, Altaf A, Zakaria N, Yaseen A, Kakar F, Kadir S, Hasan MK, Niaz SK. Navigating self-expandable metallic stent placement in inoperable esophageal malignancies: A landmark-based technique using the vertebral column and diaphragm. World J Gastrointest Endosc 2026; 18(3): 116060
- URL: https://www.wjgnet.com/1948-5190/full/v18/i3/116060.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i3.116060
Esophageal cancer is the seventh most prevalent malignancy globally and the sixth-leading cause of cancer-related mortality[1]. The overall 5-year survival rate is less than 20%[2]. Esophageal cancer poses a significant disease burden in Pakistan, where its incidence is relatively high[3]. The presence of distant metastases renders most cases inoperable and results in a dismal 5-year survival rate of 5%[4]. For patients with inoperable esophageal cancer (50%-60% at presen
Multiple strategies are available for SEMS placement, employing either fluoroscopic or endoscopic guidance. Fluoroscopic techniques often rely on indirect methods to define tumor length, including the use of external radio-opaque markers[8], injecting contrast into the submucosal plane to create an internal landmark[9], or instilling dye within the esophageal lumen to outline the stricture[10]. While these approaches assist in estimating the extent of the lesion, each carries practical drawbacks and potential risks. Hence, there is a need for performing the fluoroscopy-guided procedure via a streamlined method. Employing the vertebral column and diaphragm as landmarks for SEMS placement under fluoroscopic guidance offers a novel, simplified, easy-to-learn, and safe approach. This technique reduces procedural complexity and potential risks. This study evaluated the technical success of using the vertebral column and diaphragm as landmarks for SEMS placement under fluoroscopic guidance in patients with inoperable esophageal malignancies.
This was a retrospective study conducted at Dr. Ruth K.M Pfau, Civil Hospital Karachi, in the endoscopy department of Surgical Unit-IV and the Sindh Institute of Advanced Endoscopy and Gastroenterology. This study included all patients with endoscopically, histologically, and radiologically proven inoperable esophageal malignancy who received esophageal SEMS placement for amelioration of dysphagia from January 1, 2006, to December 31, 2024. Inoperable esophageal malignancy was defined as unresectable esophageal carcinoma due to locally advanced disease, distant metastasis, or comorbidities rendering the patients unfit for surgery.
Data from a total of 801 patients were reviewed in this study. Figure 1 depicts the selection, exclusion, and final in
Technical success was defined as the precise deployment and expansion of SEMS at the fluoroscopic anatomical landmarks, effectively bridging the strictured area, covering at least 2 cm of normal mucosa on either side of the tumor, without any immediate adverse events like major bleeding or perforation. Immediate adverse events were defined as complications occurring during the procedure or within 24 hours of stent placement and included major bleeding requiring transfusion or intervention, perforation, aspiration pneumonia, severe chest pain, or death. Re-intervention was defined as any additional endoscopic or radiological procedure required to restore luminal patency or manage a stent-related complication. Inoperability was defined as esophageal carcinoma deemed unresectable due to locally advanced disease, distant metastasis, or significant comorbidities rendering the patient unfit for surgery, as determined by the treating multidisciplinary team.
Stent insertion was performed under fluoroscopic guidance by endoscopists who had each independently performed a high volume of complex endoscopic therapeutic procedures, including esophageal SEMS placements, with documented proficiency and low complication rates. In practice, junior faculty members and advanced gastrointestinal fellows had also performed SEMS placements under the direct supervision of experienced faculty members. All procedures were performed under conscious sedation (midazolam and nalbuphine) or monitored anesthesia care. An upper gastroin
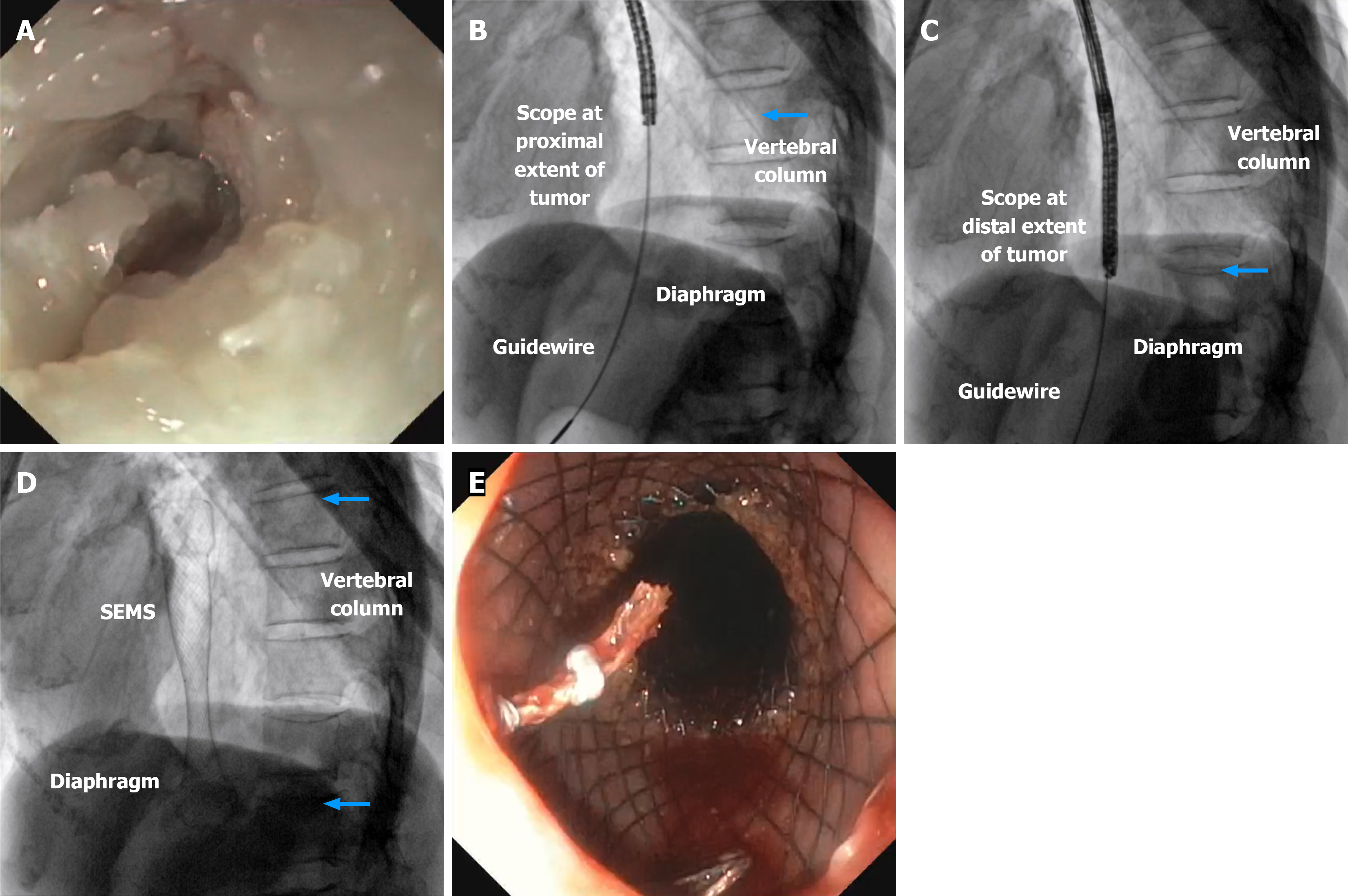
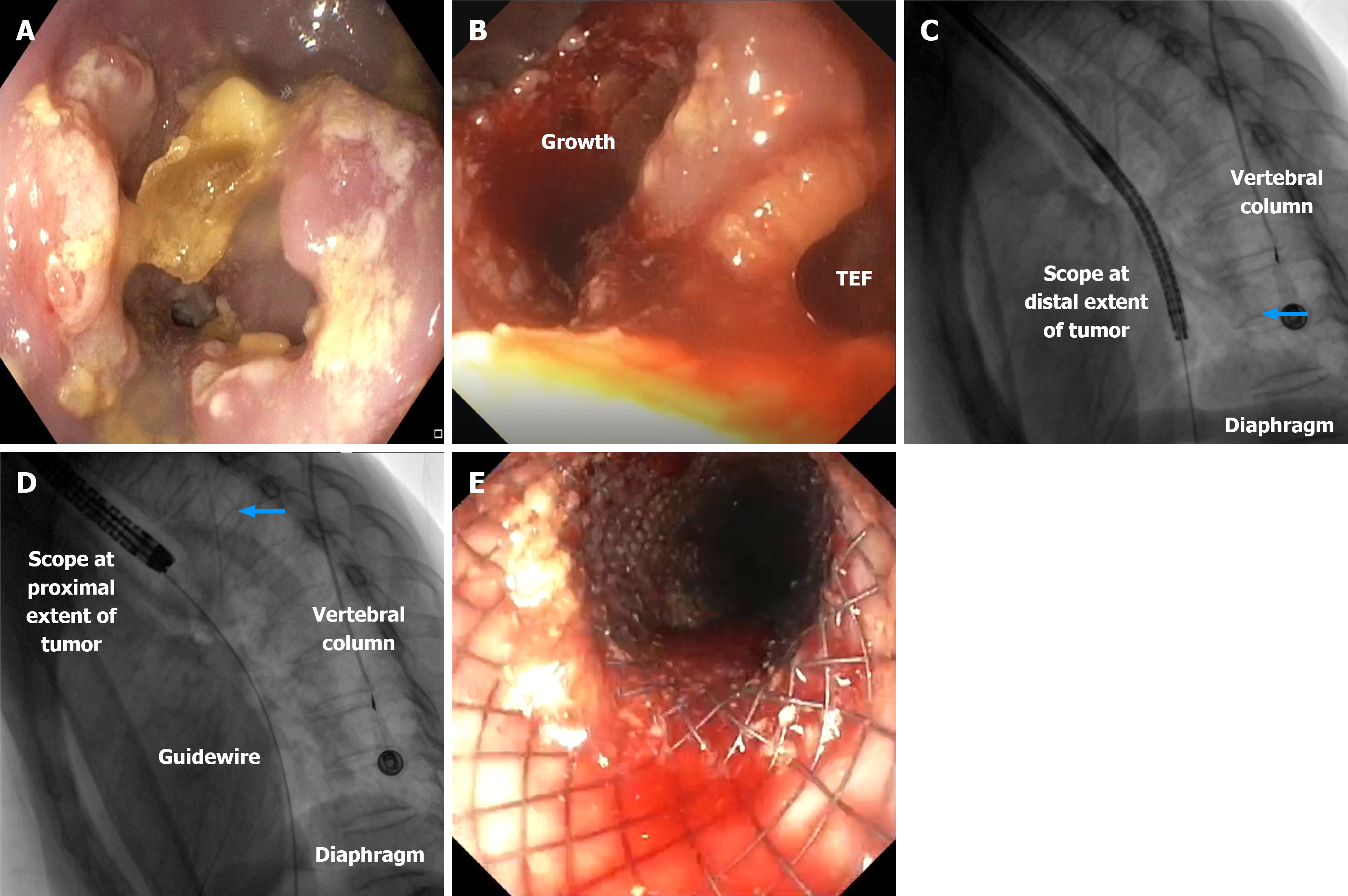
Representative images of the SEMS placement of 2 patients are presented in Figures 2 and 3.
For proximal or mid-esophageal tumors: (1) Identify key anatomical landmarks using fluoroscopy; (2) With the scope in place under fluoroscopic guidance, identify the vertebra in line with the distal extent of the tumor using the diaphragm as a reference, and identify the vertebra at the proximal extent of the tumor. Count the vertebrae between the distal and proximal extent of the tumor. Assign a length of 2.5 cm per vertebral body. Re-confirm the desired size of the SEMS by calculating the total length of the stricture by counting the vertebrae and adding one vertebral body above the proximal extent and one vertebral body below the distal extent of the tumor to obtain the proximal and distal vertebral markers respectively; and (3) Using fluoroscopy, position the stent. Place the distal end of the stent at the distal vertebral marker. Position the proximal end of the stent at the proximal vertebral marker. Release the stent at the appropriate position to ensure full coverage of the stricture.
For tumors extending beyond the gastroesophageal junction: (1) Identify key anatomical landmarks using fluoroscopy; (2) Identify the proximal extent of the tumor. Count the vertebra in line with the proximal extent of the tumor using the diaphragm as a reference for precise alignment. Identify the vertebra in line with the distal extent of the tumor beyond the gastroesophageal junction. Measure the distance from the proximal end of the tumor to the proximal stomach. Add one vertebra above and one vertebra below the fluoroscopic landmarks and reconfirm the required size of the stent; and (3) Place the stent under fluoroscopic guidance. Position the upper end of the stent at the proximal vertebral marker. Extend the distal end of the stent into the stomach past the gastroesophageal junction to ensure proper coverage. Release the stent.
Data were entered and analyzed in Statistical Package for the Social Sciences version 30 (IBM Corp., Armonk, NY, United States). Demographics for categorical variables were reported as n (%), whereas numerical variables were reported as mean ± SD.
A total of 801 patients with biopsy-proven, inoperable esophageal malignancy were included in the analysis (Table 1). Of these, 408 patients (50.90%) were female. The overall age ranged from 18 years to 95 years, with a mean age of 50 ± 15 years. All patients presented with dysphagia (100%), followed by unintentional weight loss (83.64%), vomiting (69.41%), and hematemesis (9.24%). Histopathological examination demonstrated squamous cell carcinoma in 594 pa
| Characteristics | Values |
| Age | 50.25 ± 14.91 |
| Sex | |
| Male | 393 (49.10) |
| Female | 408 (50.90) |
| Presenting complaint | |
| Dysphagia | 801 (100) |
| Weight loss | 670 (83.64) |
| Vomiting | 556 (69.41) |
| Hematemesis | 74 (9.24) |
| Cause of inoperability | |
| Distant metastasis | 423 (52.81) |
| Locally advanced disease | 316 (39.46) |
| Unfit for surgery | 62 (7.73) |
| Histopathology | |
| Squamous cell carcinoma | 594 (74.16) |
| Adenocarcinoma | 207 (25.84) |
| Location of the tumor | |
| Upper one-third of the esophagus | 83 (10.36) |
| The middle one-third of the esophagus | 365 (45.57) |
| The lower one-third of the esophagus | 353 (44.07) |
| Involvement of GEJ | |
| Yes | 240 (30.00) |
| No | 561 (70.00) |
| TEF | |
| Yes | 48 (6.00) |
| No | 753 (94.00) |
| Length of stricture in cm | 8.40 ± 2.44 |
| Dilatation | |
| Yes | 139 (17.40) |
| No | 662 (82.60) |
| Type of stent | |
| Partially covered | 726 (90.64) |
| Fully covered | 75 (9.36) |
| Size of stent in cm | 12.49 ± 2.26 |
Esophageal SEMS placement is an effective palliative measure for patients with malignant esophageal strictures[11]. Our study highlighted a simplified technique for SEMS placement under fluoroscopy by using the vertebral column and diaphragm as landmarks. To the best of our knowledge, this study represents the largest single-center study on esophageal SEMS placement. Previously, Włodarczyk and Kużdżał[12] reported a cohort of 442 patients. There are several techniques for SEMS placement that utilize fluoroscopic or endoscopic approaches. Fluoroscopic-guided techniques include ascertaining the extent of the tumor by placement of external radio-opaque markers[8], submucosal injection of a contrast material serving as an internal marker[9], and the luminal instillation of dye for delineating the stricture length[10]. However, the external markers can detach, leading to malposition. Additionally, submucosal contrast injection in
Unlike the simplified landmark-guided deployment described by Kini et al[14], which relied solely on endoscopic distance measurements without fluoroscopic confirmation, our technique integrates both endoscopic delineation and fluoroscopic guidance, using vertebral bodies and the diaphragm as stable reference points. This dual-modality approach not only allows for accurate calculation of stricture length, tailored stent sizing, and precise deployment but also provides objective fluoroscopic markers that remain useful even in patients with spinal deformities or prior surgical alterations. As a result, the risks of misplacement, incomplete coverage, or migration are minimized, particularly for tumors involving the gastroesophageal junction.
In contrast with previous studies[15] where male patients predominated (69.3%), the majority of our patients were female (50.90%). This discrepancy may be due to late presentation, limited access to healthcare, biological factors, so
A partially covered SEMS was placed in 90.64% of patients due to a decreased risk of stent migration compared with fully covered SEMS[20] and the low prevalence of restenosis due to tumor ingrowth compared with uncovered SEMS[21]. This choice likely led to better long-term clinical outcomes and decreased the requirement for re-intervention. Ad
While the mean thoracic vertebral body height is typically reported between 20 mm and 22 mm in Indian cohorts, and Kunkel et al[23] reported vertebral body heights ranging from 14.2 mm to 21.96 mm across thoracic levels with higher values distally, Singh et al[24] documented anterior heights approaching 29-30 mm at T12 in an Indian cadaveric population. Thoracic intervertebral discs contribute an additional 3-5 mm in height, particularly in lower thoracic levels[25]. Thus, approximating each vertebral level (vertebral body + disc) as 2.5 cm is consistent with published anatomical data and provides a pragmatic safety margin to ensure complete stricture coverage and minimize the risk of uncovered tumor margins during stent deployment. We recognize this represents an upper-range approximation and acknowledge anatomical variability; therefore, this study was consistently supplemented by real-time endoscopic and fluoroscopic verification to ensure accurate stent placement.
This landmark-based method offers notable cost advantages in resource-limited settings by eliminating the need for specialized consumables and contrast agents. Its shorter procedure time further enhances cost-effectiveness by reducing fluoroscopy exposure, staff demands, and room occupancy, making it particularly suitable for high-volume public hospitals. Moreover, this approach is easily teachable to junior endoscopists. Consistent vertebral-diaphragm alignment allows predictable fluoroscope orientation, enabling trainees to identify key anatomical landmarks and deploy stents accurately under supervision. Its simplicity and minimal operator-dependent variability result in a short learning curve, with most juniors achieving proficiency after a few supervised procedures, making it a valuable training tool in resource-limited centers.
A key finding of our study was the high technical success rate of 100%, highlighting the reliability and reproducibility of this technique across multiple endoscopists with minimal variability. No immediate procedure-related adverse events, such as bleeding or perforation, which can occur in up to 6% of patients[26], were observed in our patients. However, stent misdeployment could not be reliably commented upon, as it is possible that such events, if they occurred, may not have been consistently documented in the available records. 15 patients (1.87%) required placement of a stent within a stent due to tumor overgrowth and disease progression, which was previously reported by Na et al[27] in 5% of their patients. Stent blockage due to tumor ingrowth requiring Savary Gilliard dilatation was observed in 11 patients (1.37%). The technical ease, cost-effectiveness, minimal manpower requirements, and time efficiency make it a valuable alternative to current SEMS placement techniques. This technique is accessible to a broad range of endoscopists, including those working in resource-limited settings. These attributes collectively support the widespread adoption and standardization of this technique in clinical practice.
Our study had several strengths. First, the sample size was sufficient. Second, our results aligned with studies de
Despite these strengths and key findings, we should acknowledge the limitations of the study. The data were reviewed retrospectively, and the improvement in dysphagia, malnutrition, and quality of life, which are essential to judge clinical impact, could not be estimated. Head-to-head comparison with existing techniques for SEMS placement under fluoroscopic guidance could not be made, limiting our ability to demonstrate superiority, non-inferiority, or incremental benefit of the proposed technique. Moreover, there exists the possibility of underestimation of stated late adverse events, in
Our method of SEMS placement using the vertebral column and diaphragm as landmarks is a novel approach. We reported the safety and reproducibility of this technique on a large scale with multiple endoscopists performing the procedure. This technique offers a simplified, reliable, and reproducible approach with a high success rate, minimal inter-endoscopist variability, and procedure-related adverse events, making it an attractive option for standardization. Future prospective, multi-center studies are warranted to compare this method with existing techniques, evaluate long-term clinical outcomes, and establish standardized training protocols for wider adoption.
We acknowledge the valuable services and brilliant procedural skills of all the endoscopists who performed SEMS placement in the department of Surgical Unit-IV and Sindh Institute of Advanced Endoscopy and Gastroenterology. Their sincere contribution to serve in the best interest of the patients is appreciated.
| 1. | Lander S, Lander E, Gibson MK. Esophageal Cancer: Overview, Risk Factors, and Reasons for the Rise. Curr Gastroenterol Rep. 2023;25:275-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 84] [Article Influence: 28.0] [Reference Citation Analysis (3)] |
| 2. | Li J, Xu J, Zheng Y, Gao Y, He S, Li H, Zou K, Li N, Tian J, Chen W, He J. Esophageal cancer: Epidemiology, risk factors and screening. Chin J Cancer Res. 2021;33:535-547. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 145] [Article Influence: 29.0] [Reference Citation Analysis (5)] |
| 3. | Asghar MS, Khan NA, Kazmi SJH, Hassan M, Rasheed U, Jawed R, Yaseen R, Naqvi SAA. Clinical, epidemiological, and diagnostic characteristics of esophageal carcinoma in a Pakistani population. Ann Saudi Med. 2021;41:91-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 4. | Ghazy HF, El-Hadaad HA, Wahba HA, Abbas R, Abbas OA. Metastatic Esophageal Carcinoma: Prognostic Factors and Survival. J Gastrointest Cancer. 2022;53:446-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 5. | Besharat S, Jabbari A, Semnani S, Keshtkar A, Marjani J. Inoperable esophageal cancer and outcome of palliative care. World J Gastroenterol. 2008;14:3725-3728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 35] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 6. | Mohapatra S, Santharaman A, Gomez K, Pannala R, Kachaamy T. Optimal Management of Dysphagia in Patients with Inoperable Esophageal Cancer: Current Perspectives. Cancer Manag Res. 2022;14:3281-3291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 7. | Sharma B, Raina S, Sharma R, Bodh V, Raina SK, Sharma N. Self-Expanding Metallic Stents (SEMS) in Inoperable Esophageal Cancer: A Prospective Analysis of Morbidity and Survival Outcomes. Indian J Palliat Care. 2019;25:398-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 8. | Fujita T, Tanabe M, Shimizu K, Iida E, Matsunaga N. Radiological image-guided placement of covered Niti-S stent for palliation of dysphagia in patients with cervical esophageal cancer. Dysphagia. 2013;28:253-259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 9. | Mangiavillano B, Auriemma F, Bianchetti M, Mantovani N, Pilati S, Reggio D, Barletti C, Forcignanò E, Pentassuglia G, Arezzo A, Cavargini E, Fabbri C, Mezzanzanica M, De Luca L, Conigliaro R, Repici A. Through-the-scope esophageal stent for the relief of malignant dysphagia: Results of a multicentric study (with video). Dig Endosc. 2021;33:118-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 10. | Kim KY, Tsauo J, Song HY, Kim PH, Park JH. Self-Expandable Metallic Stent Placement for the Palliation of Esophageal Cancer. J Korean Med Sci. 2017;32:1062-1071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 11. | Hindy P, Hong J, Lam-Tsai Y, Gress F. A comprehensive review of esophageal stents. Gastroenterol Hepatol (N Y). 2012;8:526-534. [PubMed] |
| 12. | Włodarczyk JR, Kużdżał J. Stenting in Palliation of Unresectable Esophageal Cancer. World J Surg. 2018;42:3988-3996. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 13. | Därr R, Lenders JW, Stange K, Kindel B, Hofbauer LC, Bornstein SR, Eisenhofer G. [Diagnosis of pheochromocytoma and paraganglioma: the clonidine suppression test in patients with borderline elevations of plasma free normetanephrine]. Dtsch Med Wochenschr. 2013;138:76-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 14. | Kini R, Ramanathan S, Thangavel P, Karunakaran P, Mohamed KS, Ananthavadivelu M. A simplified technique of esophageal self-expandable metallic stent placement without fluoroscopic and endoscopic guidance for treating esophageal carcinoma. Turk J Gastroenterol. 2018;29:82-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 15. | Heutlinger O, Acharya N, Kharabaf S, Acharya N, Perez-Menendez S, Kirby K, Smith B, Nguyen NT. A systematic review and meta-analysis of factors associated with esophageal stent migration and a comparison of antimigration techniques. J Gastrointest Surg. 2025;29:101977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 16. | Qureshi S, Khan S, Quraishy MS, Zaheer S. Esophageal cancer patient survival: A retrospective study from a tertiary care hospital in Pakistan. Pak J Med Sci. 2024;40:811-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 17. | Butt N, Soomro S, Mahesar GB, Abbasi A, Haider G. 2863 Epidemiological and Clinical Characteristics of Esophageal Carcinoma: A Four-Year Experience From Largest Referral Centre of Karachi, Pakistan. Am J Gastroenterol. 2019;114:S1571-S1571. [DOI] [Full Text] |
| 18. | Qumseya B, Yang S, Guo Y. Trends in prevalence of esophageal adenocarcinoma: Findings from a statewide database of over 6 million patients. Endosc Int Open. 2024;12:E218-E226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (1)] |
| 19. | Yu Z, Chen T, Peng H, Li A, Wei Y, Xiao S. Trends in incidence, treatment modalities and prognosis of esophageal adenocarcinoma in the US population. Cancer Epidemiol. 2024;93:102683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 20. | Das KK, Hasak S, Elhanafi S, Visrodia KH, Ginsberg GG, Ahmad NA, Hollander T, Lang G, Kushnir VM, Mullady DK, Abu Dayyeh BK, Buttar NS, Wong Kee Song LM, Kochman ML, Chandrasekhara V. Performance and Predictors of Migration of Partially and Fully Covered Esophageal Self-Expanding Metal Stents for Malignant Dysphagia. Clin Gastroenterol Hepatol. 2021;19:2656-2663.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 21. | Ahmed O, Lee JH, Thompson CC, Faulx A. AGA Clinical Practice Update on the Optimal Management of the Malignant Alimentary Tract Obstruction: Expert Review. Clin Gastroenterol Hepatol. 2021;19:1780-1788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 22. | Silva R. Esophageal Stenting: How I Do It. GE Port J Gastroenterol. 2023;30:35-44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 23. | Kunkel ME, Herkommer A, Reinehr M, Böckers TM, Wilke HJ. Morphometric analysis of the relationships between intervertebral disc and vertebral body heights: an anatomical and radiographic study of the human thoracic spine. J Anat. 2011;219:375-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 24. | Singh R, Srivastva SK, Prasath CS, Rohilla RK, Siwach R, Magu NK. Morphometric measurements of cadaveric thoracic spine in Indian population and its clinical applications. Asian Spine J. 2011;5:20-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 25. | Fletcher JG, Stringer MD, Briggs CA, Davies TM, Woodley SJ. CT morphometry of adult thoracic intervertebral discs. Eur Spine J. 2015;24:2321-2329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 26. | Teyangesikayi G, Scriba MF, Viranna S, Jonas EG, Chinnery GE. The incidence and management of complications following stenting of oesophageal malignancies. S Afr J Surg. 2023;61:27-32. [PubMed] [DOI] [Full Text] |
| 27. | Na HK, Song HY, Kim JH, Park JH, Kang MK, Lee J, Oh SJ. How to design the optimal self-expandable oesophageal metallic stents: 22 years of experience in 645 patients with malignant strictures. Eur Radiol. 2013;23:786-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 28. | Wang C, Wei H, Li Y. Comparison of fully-covered vs partially covered self-expanding metallic stents for palliative treatment of inoperable esophageal malignancy: a systematic review and meta-analysis. BMC Cancer. 2020;20:73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (1)] |