Published online Mar 16, 2026. doi: 10.4253/wjge.v18.i3.115257
Revised: December 9, 2025
Accepted: January 15, 2026
Published online: March 16, 2026
Processing time: 150 Days and 23.3 Hours
Endoscopic submucosal dissection (ESD) is a viable therapeutic approach for laterally spreading tumors; however, delayed intramural hematoma remains a rare but serious complication. This report describes a successfully managed case, adding valuable evidence to the existing literature. This study demonstrates that conservative management can be both safe and effective, despite previous reports of fatal outcomes. This experience underscores the importance of early recognition and appropriate management of this complication by endoscopists.
A 60-year-old male underwent ESD due to a rectal laterally spreading tumor. The procedure was completed successfully, and the initial postoperative course was uneventful. On postoperative day 4, the patient developed hematochezia acc
Delayed intramural hematoma following ESD can be effectively managed with conservative therapy, resulting in complete recovery.
Core Tip: This report describes a case of delayed intramural hematoma occurring on the fourth day after endoscopic submucosal dissection of a rectal laterally spreading tumor. The intramural hematoma was successfully treated non-surgically, resulting in complete recovery. In contrast to previously reported fatal outcomes, this case provides valuable clinical evidence that careful non-surgical management can achieve excellent results, highlighting an important strategy for endoscopists to avoid unnecessary invasive interventions.
- Citation: Xu YL, Zhang ML, Zhou HJ, Gao PJ, Zhang XN, Rao M. Giant rectal intramural hematoma following endoscopic submucosal dissection successfully treated with conservative therapy: A case report. World J Gastrointest Endosc 2026; 18(3): 115257
- URL: https://www.wjgnet.com/1948-5190/full/v18/i3/115257.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i3.115257
Laterally spreading tumors (LSTs) of the colorectum constitute a unique group of superficial mucosal lesions. Endoscopic submucosal dissection (ESD) for LSTs has demonstrated high rates of en bloc and complete resection (89.8% and 81.9%, respectively), confirming its efficacy and safety as a treatment modality[1]. Common complications linked to ESD include bleeding and perforation, whereas delayed hematoma occurs infrequently. Tanaka et al[2] reported a fatal case of intra
A 60-year-old man developed hematochezia following ESD, and diagnosed with a large intramural hematoma four days later.
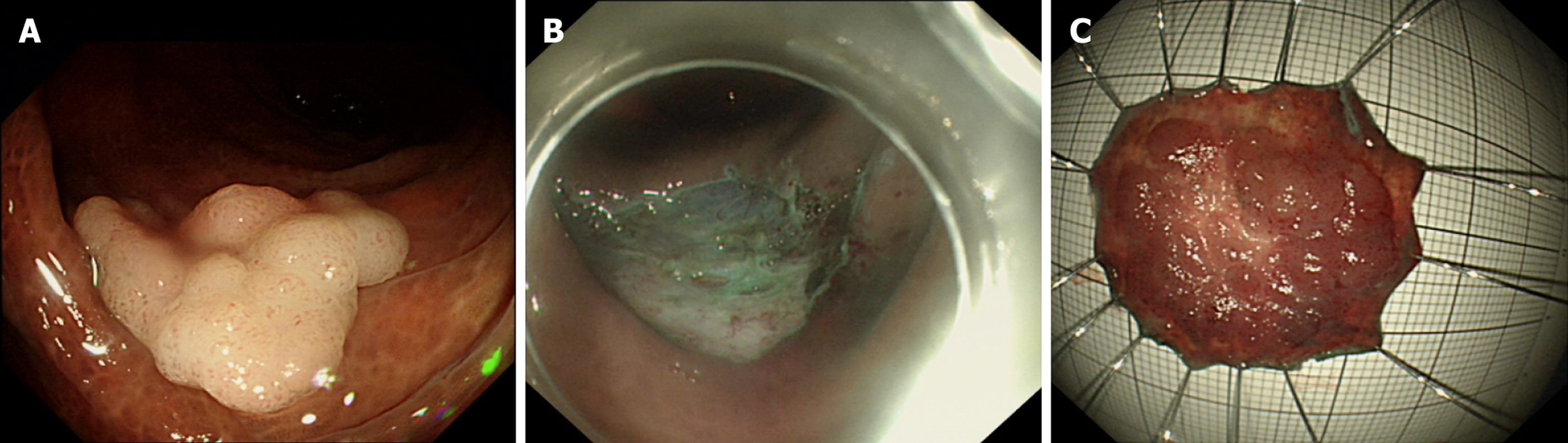
The patient underwent standard colonoscopy which revealed a granular nodular mixed-type LST measuring 2.5 cm × 2.5 cm in the rectum, approximately 15 cm from the anal verge. After careful bowel preparation, ESD was performed (Figure 1). Histopathological analysis of the excised tissue verified the presence of a tubular adenoma. The initial post
The patient denied any significant past medical history.
The patient did not report any notable personal or family history.
Upon admission, abdominal examination showed slight tenderness without rebound tenderness.
Preoperatively, the patient exhibited normal complete blood count, coagulation profile, liver function, and renal function results. Dynamic monitoring of clinical parameters and symptom progression throughout the hematoma treatment period are shown in Table 1.
| Postoperative day | Main symptoms | Blood pressure (mmHg) | WBC (× 109/L; reference range: 3.50-9.50) | Hemoglobin (g/L; reference range: 130-175) | Urea (mmol/L; reference range: 3.6-9.5) | Creatinine (μmol/L; reference range: 57-111) | Liver function tests |
| Day 4 | Hematochezia | 102/58 | 14.02 | 121 | 9.0 | 137 | Normal |
| Day 5 | Abdominal distension and constipation | 108/65 | 9.81 | 98 | 8.9 | 121 | Normal |
| Day 6 | No significant improvement | 110/74 | 6.46 | 88 | 8.6 | 85 | - |
| Day 7 | Abdominal distension relieved | 105/76 | 5.77 | 86 | - | - | - |
| Day 8 | No abdominal distension or constipation | 115/80 | 5.5 | 92 | 6.9 | 68 | Normal |
| Day 11 | Recovering well | 109/67 | 5.8 | 112 | - | - | - |
| Day 15 | Tolerating liquid diet well | 112/78 | 5.96 | 115 | 3.6 | 63 | Normal |
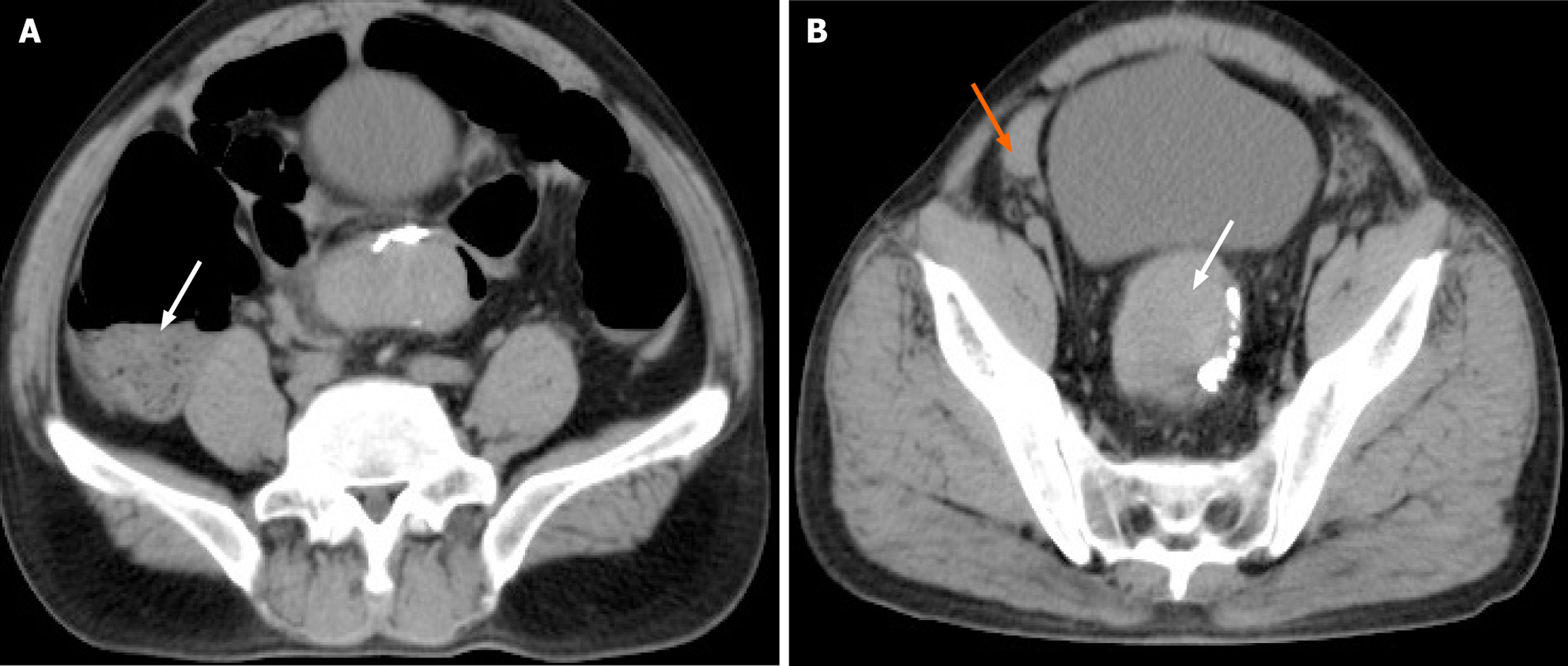
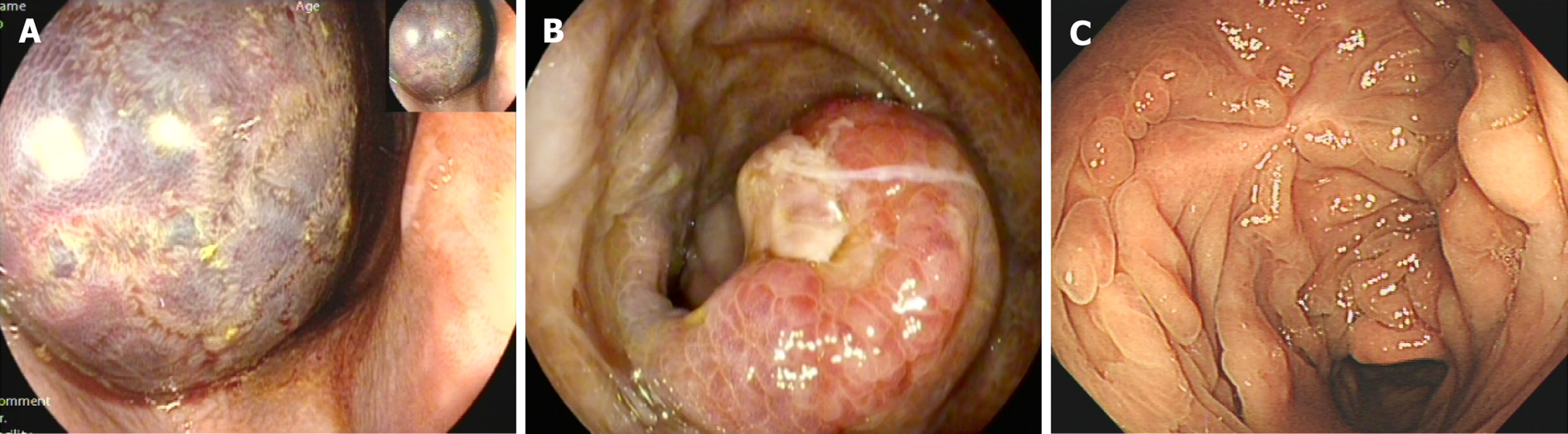
On postoperative day 4, the onset of hematochezia prompted an emergent computed tomography scan, which revealed intestinal obstruction (Figure 2A), rectal hematoma and hemoperitoneum (Figure 2B). Emergency colonoscopy revealed a large, purple-walled hematoma at the ESD site, almost completely occluding the lumen and preventing further adv
The patient was diagnosed with a delayed post-ESD intramural hematoma secondary to the ESD procedure.
Immediate conservative management was initiated, including fasting, bed rest, intravenous antibiotic therapy with cefminox (2.0 g every 12 hours), and intramuscular administration of the hemostatic agent hemocoagulase atrox (2.0 KU every 12 hours).
By postoperative day 15, the patient’s symptoms had improved significantly. Follow-up colonoscopy demonstrated marked resolution of the hematoma (Figure 3B). He was discharged symptom-free on postoperative day 18. During the 6-month follow-up, the patient was asymptomatic, and colonoscopy showed a well-healed scar at the site of resection (Figure 3C).
Intramural colorectal hematoma is a rare clinical entity. Reported etiologies in the literature include abdominal trauma[3], anticoagulant use[4], and hematologic disorders[5]. With advancements in endoscopic techniques, cases have also been reported following routine colonoscopy[6] and polypectomy[7]. These endoscopy-related occurrences are often associated with factors such as operator experience, excessive instrument manipulation, or intestinal traction. The clinical pre
Delayed post-ESD bleeding, reported to occur in approximately 2.9% of procedures, is associated with several well-established risk factors, including large lesions (≥ 3 cm), a history of antiplatelet medication use, and tumor location within the rectosigmoid region[8]. In the present case, electrocoagulation was applied to visible vessel stumps during dissection, and no intraoperative bleeding was observed. However, the patient developed postoperative hematochezia, which resolved with supportive care and advice to avoid physical exertion. Notably, the patient’s coagulation profile was normal, and he was not receiving anticoagulation therapy. The lesion in this case did not exhibit any of the previously identified risk factors. The postoperative bleeding was therefore considered to be related to inadequate management of a vascular stump, resulting in delayed hemorrhage. Intraoperatively, all visible vessels at the resection site that were considered potential sources of bleeding were treated with prophylactic electrocautery. It was confirmed that all active bleeding had ceased before terminating the procedure. Therefore, we hypothesize that the source of bleeding originated from a vessel within the deeper submucosal layer. A potential mechanism involves temporary hemostasis achieved by intraoperative electrocoagulation, followed by retraction of the vessel stump. This may have obscured the bleeding site from the endoscopic field, allowing concealed hemorrhage into the loose submucosal space, which subsequently deve
Following hematoma formation, the large intramural hematoma obstructed the intestinal lumen, resulting in abdominal distension and cessation of flatus, clinical manifestations consistent with intestinal obstruction. Given the patient’s stable condition, absence of peritonitis, and lack of anticoagulant use that could increase bleeding risk, conservative management was selected. The patient remained on bed rest with limited fluid intake and received intravenous fluids, in addition to intravenous antibiotics to prevent infection and intramuscular hemostatic treatment. Using this approach, the hematoma progressively decreased in size and eventually resolved completely. Hematoma is an unco
This report describes a rare case of intramucosal hematoma following ESD that was successfully managed with conservative therapy, offering valuable clinical insights. To reduce the likelihood of injury to deep vessels and hematoma development, the intraoperative technique should emphasize avoiding excessive electrocoagulation and repeated dissection within the same deep tissue plane. In addition, meticulous hemostasis is essential to prevent blood loss from obscuring the resection bed, an especially critical consideration for large lesions or those with submucosal invasion, where inadequate visualization may compromise the assessment of vascular stump integrity. If a postoperative hemat
ESD is primarily indicated for colorectal lesions with submucosal invasion or for large lesions, particularly those ≥ 2 cm[11]. With its increasing clinical adoption, postoperative complications have drawn increasing attention. Current evidence indicates a perforation rate of approximately 5.5%, a bleeding rate of about 4.1%, and surgical intervention required in roughly 1.8% of cases[12]. However, postoperative intramural hematoma is rarely reported and is yet to be well characterized. We identified five published cases (Table 2), of which only two received conservative treatment, both patients were receiving anticoagulation. One patient[2] resumed anticoagulants on postoperative day 1 for atrial fibrillation, deteriorated into diffuse peritonitis, and ultimately died - conservative therapy was chosen against medical recomm
| Ref. | Complication | Year | Age (years) | Sex | Anticoagulant therapy | Lesion size (mm) | Device | Time to hematoma onset | Treatment | Outcome |
| [2] | Hematoma | 2023 | 82 | Male | Yes | 25 | ESD | Postoperative day 4 | Conservative treatments | Died on postoperative day 25 |
| [6] | Hematoma | 2022 | 50 | Female | No | No available | Colonoscopy | Three hours | Surgery | Discharged |
| [7] | Hematoma | 2023 | 81 | Female | Yes | 7 | Cold snare polypectomy | Following day | Conservative treatments | Discharged 20 days after admission |
| [9] | Hematoma | 2022 | 61 | Female | No | 8 | Cold snare polypectomy | Following day | Endoscopic intervention | Hematoma absorption 3 days after intervention |
| [10] | Hematoma + perforation | 2025 | 43 | Female | No | 3 | Cold snare polypectomy | Two hours | Surgery | Discharged on postoperative day 14 |
The success of conservative therapy in the present case hinged on the following favorable factors: Good general condition, stable hemodynamics, absence of peritoneal signs, no radiologic evidence of perforation, and incomplete lumi
For post-ESD hematoma, conservative management may be considered the first-line approach in patients who are hemodynamically stable, without perforation or diffuse peritonitis, with limited hematoma extent, and when multidisciplinary monitoring and intervention are available. Conversely, endoscopic or surgical intervention should be promptly undertaken if perforation, worsening infection, progressive hematoma enlargement, or persistent active bleeding occurs.
| 1. | Zhu M, Xu Y, Yu L, Niu YL, Ji M, Li P, Shi HY, Zhang ST. Endoscopic submucosal dissection for colorectal laterally spreading tumors: Clinical outcomes and predictors of technical difficulty. J Dig Dis. 2022;23:228-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (6)] |
| 2. | Tanaka K, Yoshikawa T, Yazumi S. A huge colonic intramural hematoma caused by endoscopic submucosal dissection. Gastrointest Endosc. 2023;98:460-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 3. | Hou MM, Tsou YK. Education and imaging. Gastrointestinal: Acute colonic intramural hematoma after blunt abdominal trauma. J Gastroenterol Hepatol. 2009;24:494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 4. | Kula J, Szmigiel P, Rusinowski C, Mrowiec S, Wosiewicz P. Extensive spontaneous intramural hematoma of rectum and sigmoid colon in a patient undergoing anticoagulant therapy: A case report. Medicine (Baltimore). 2025;104:e42428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Jarry J, Biscay D, Lepront D, Rullier A, Midy D. Spontaneous intramural haematoma of the sigmoid colon causing acute intestinal obstruction in a haemophiliac: report of a case. Haemophilia. 2008;14:383-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 6. | Jalal A, Khatiwada P, Shoreibah M, Dauer R. Intramural Hematoma and Retroperitoneal Hematoma Following a Routine Colonoscopy: An Uncommon Complication of a Common Procedure. Case Rep Gastroenterol. 2022;16:515-520. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Arakane N, Yoshida K, Murata S, Mizuno R, Furuta K, Tojo M, Tamagawa H, Miyanaga R, Watanabe K, Fukuhara S. Large submucosal hematoma after cold snare polypectomy for colorectal adenoma. DEN Open. 2023;3:e199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 8. | Seo M, Song EM, Cho JW, Lee YJ, Lee BI, Kim JS, Jeon SW, Jang HJ, Yang DH, Ye BD, Byeon JS. A risk-scoring model for the prediction of delayed bleeding after colorectal endoscopic submucosal dissection. Gastrointest Endosc. 2019;89:990-998.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (3)] |
| 9. | Negishi R, Muramoto T, Ohata K. Hemostasis using purse-string reefing with endoscopic clip and endoloop for a huge hematoma after cold snare polypectomy. Dig Endosc. 2022;34:e68-e70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Kodama Y, Mizokami Y, Toyama Y, Kusaka H, Maeda G, Asahara S, Nagahama R, Horiguchi SI, Aoyama H. A case of gastrointestinal perforation following transarterial embolization for an intramural hematoma after cold snare polypectomy of an adenoma in the transverse colon. DEN Open. 2025;5:e70017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Pimentel-Nunes P, Libânio D, Bastiaansen BAJ, Bhandari P, Bisschops R, Bourke MJ, Esposito G, Lemmers A, Maselli R, Messmann H, Pech O, Pioche M, Vieth M, Weusten BLAM, van Hooft JE, Deprez PH, Dinis-Ribeiro M. Endoscopic submucosal dissection for superficial gastrointestinal lesions: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2022. Endoscopy. 2022;54:591-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 617] [Cited by in RCA: 522] [Article Influence: 130.5] [Reference Citation Analysis (1)] |
| 12. | Singh RR, Nanavati J, Gopakumar H, Kumta NA. Colorectal endoscopic submucosal dissection in the West: A systematic review and meta-analysis. Endosc Int Open. 2023;11:E1082-E1091. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |