Published online Mar 16, 2026. doi: 10.4253/wjge.v18.i3.114327
Revised: November 17, 2025
Accepted: January 6, 2026
Published online: March 16, 2026
Processing time: 176 Days and 11.2 Hours
Acute caustic ingestion is a medical emergency that can cause a wide spectrum of gastrointestinal injuries, making timely and accurate triage essential. Early eso
Core Tip: Endoscopy best assesses mucosal injury, but it cannot reliably evaluate transmural or extra-luminal complications. Computed tomography provides complementary cross-sectional information, identifying necrosis, perforation, and mediastinal or peritoneal involvement, and is especially valuable when endoscopy is contraindicated or inconclusive. Together, they provide a comprehensive evaluation that improves risk stratification and guides management in acute caustic ingestion.
- Citation: Singh AK, Birda CL, Kochhar R. Optimizing triage in acute caustic ingestion: Choosing between endoscopy and computed tomography. World J Gastrointest Endosc 2026; 18(3): 114327
- URL: https://www.wjgnet.com/1948-5190/full/v18/i3/114327.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i3.114327
Acute caustic ingestion is a potentially life-threatening emergency that can cause a wide spectrum of gastrointestinal (GI) injuries, ranging from superficial mucosal burns to full-thickness necrosis, perforation, and mediastinitis[1,2]. Early and accurate assessment of both the extent and depth of injury is crucial for guiding appropriate management, whether that involves surgical intervention, intensive care monitoring, or conservative therapy. Traditionally, upper GI endoscopy performed within the first 12-24 hours has been regarded as the gold standard for evaluating mucosal damage[3,4]. However, endoscopy has important limitations, including its inability to reliably assess transmural and extra-luminal involvement, as well as the potential risk of iatrogenic perforation in patients with severe injury.
In recent years, computed tomography (CT) has emerged as a valuable complementary tool in the evaluation of acute caustic ingestion[5-7]. CT not only provides a non-invasive assessment of hollow organ wall involvement but also offers critical information regarding peri-visceral and extra-luminal complications. Furthermore, CT aids in risk stratification, helps identify patients at high risk for adverse outcomes, and guides the choice between conservative vs surgical ma
The present review focuses on the role of CT in the assessment, management, and prognostication of acute caustic injury, highlighting its advantages, limitations, and integration with endoscopic findings.
Attempts to predict the severity of esophageal and gastric injury based on symptoms have yielded inconsistent results. While some correlations exist, the absence of symptoms does not rule out severe injury[8]. Several clinical scores have been used with inconsistent results[9-13]. Clinical grading, when used alone also, has significant limitations. External signs such as oral burns, drooling, or vomiting may not correlate with the extent of esophageal or gastric injury, leading to both underestimation and overestimation of the risk[14]. Moreover, reliance solely on clinical parameters does not provide information about the depth of injury or the presence of complications such as perforation or mediastinitis[15]. Consequently, exclusive dependence on clinical grading can delay timely intervention and compromise prognosis.
Along with clinical assessment, all patients require baseline laboratory tests, including complete blood counts, liver and renal function tests, serum lactate and electrocardiogram. Leukocytosis, elevated alanine aminotransferase, and th
Early esophagogastroduodenoscopy (EGD) or upper GI endoscopy has traditionally been used for the assessment of injury and remains the initial approach at most centers[17]. Endoscopy helps in guiding further treatment, prognosis and timing of discharge. Recommendation for early endoscopy is based on the safety of the procedure in caustic injury before the injured segment enters the weaker reparative phase with increased risk of perforation. A large United States database study showed that all clinical outcomes (death, perforation, sepsis, aspiration pneumonia, parenteral feeding, GI bleed, systemic complications) were less common in the early EGD (within 48 hours) group compared to the delayed EGD (after 48 hours) group[4]. Delayed EGD also had poor clinical outcomes and increased the cost of hospital admissions. Thus, EGD should be done at the earliest and preferably in the initial 24-48 hours of caustic ingestion in all patients who are stable. The principle of gentle handling during endoscopy with the use of carbon dioxide insufflation and conscious sedation should be followed. In selected cases when a patient presents late, endoscopy can be safely performed after 48 hours in expert hands to assess the extent and severity of injury[17-19].
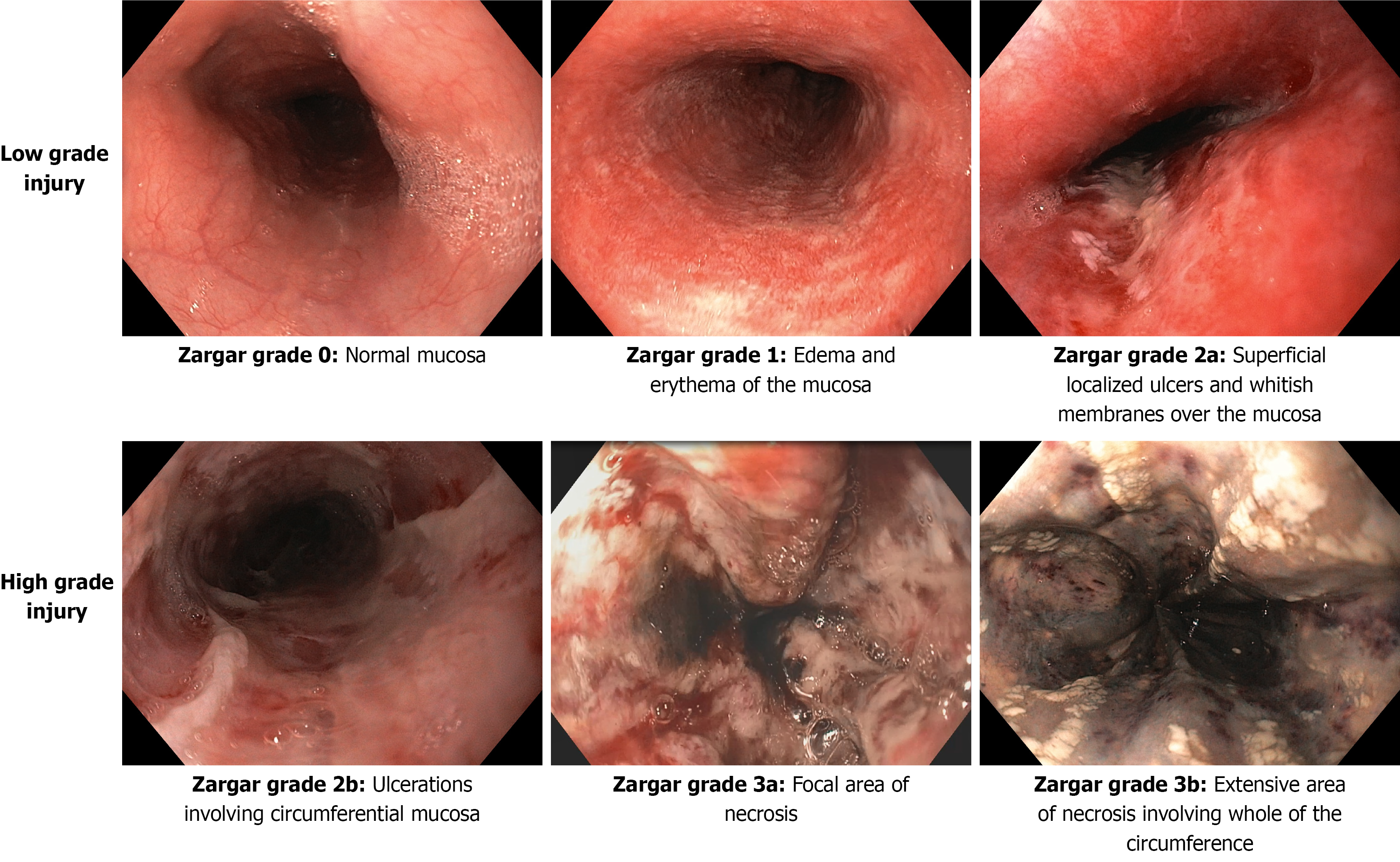
During endoscopic examination of the upper GI tract, Zargar’s grading (Figure 1 and Table 1) should be used to assess the extent and severity of injury for the esophagus, stomach and duodenum[18]. Use of grading systems standardizes the reporting of injury and also helps in prognosticating. The injury should be described using standard definitions, words and phrases (Table 1). Classification of injury into low (grade ≤ 2a) and high (grade ≥ 2b) grade helps in identifying the patients who can be allowed early discharge from the hospital, and also those who require long-term hospitalization. Kochhar et al[20] showed that patients with low-grade injury are less likely to develop follow-up complications and could be discharged early. Patients with high-grade injury require long-term hospitalization, intensive care unit admission, nutritional support and are also likely to develop long-term complications.
| Endoscopic grading | Findings |
| Grade 0 | Normal findings |
| Grade 1 | Edema and hyperemia of the mucosa |
| Grade 2a | Superficial localized ulcerations, friability, hemorrhage and blisters |
| Grade 2b | Deep ulcerations (either discrete or circumferential), in addition to friability, hemorrhage, and superficial ulcerations |
| Grade 3a | Small, scattered areas of necrosis |
| Grade 3b | Extensive necrosis |
Although early endoscopy remains the gold standard for evaluating mucosal damage in acute caustic ingestion, its role has important limitations. As an invasive procedure, it may not be feasible in unstable patients, particularly those with psychiatric problems, respiratory distress or hemodynamic instability. Early endoscopy may also overestimate the depth of injury, leading to overtreatment. In a retrospective series of 62 patients, Kochhar et al[20] demonstrated that endoscopy within 24 hours tended to overpredict severity, whereas reassessment on day 5 showed higher specificity for predicting both esophageal (83% vs 65%) and antropyloric strictures (95% vs 61%). Factors such as blood, food debris, mucosal slough, and charring were noted to contribute to overgrading during early examinations. Endoscopic scoring is further limited by inter-observer variability, with inconsistent terminology and interpretation. Additional concerns include the risk of iatrogenic perforation in fragile tissues and the limited reliability in delayed presenters, where mucosal healing or sloughing can obscure the true extent of injury.
Because of the inherent limitation of endoscopy in assessing the full extent of damage, adjunct imaging techniques are often needed in acute caustic injury. Early interest in using endoscopic ultrasound to assess caustic injury depth was tempered by its limited ability to evaluate beyond the GI wall and its procedural risks comparable to standard en
A variety of imaging modalities can aid in the evaluation of acute caustic injury. Plain chest and abdominal X-rays are useful for detecting complications such as free air, aspiration pneumonia, or gross perforation, but provide no in
Timing of CT: The timing of CT in acute caustic injury is crucial to maximize its diagnostic and prognostic value. In the acute phase, submucosal inflammatory infiltration and edema manifest as hollow organ wall thickening with edematous mucosa. With progression, mucosal sloughing results in thinning or denudation of the mucosa. Thus, CT is most in
CT technique and protocol: A CT should include the neck, chest, and abdomen, with intravenous contrast being essential to assess mural enhancement patterns and differentiate viable tissue from necrosis. The use of oral contrast remains debatable, with two protocols described[7]. The first suggests an initial non-contrast CT to detect air around the GI tract; if pneumomediastinum or pneumoperitoneum is present, then a contrast-enhanced CT with positive oral contrast is performed to localize the perforation and aid surgical decision-making. If no free air is seen, oral contrast is avoided[7,23]. The second protocol involves a single venous-phase contrast-enhanced CT of the neck, chest, and abdomen with oral contrast[5]. No direct comparison exists between these protocols, and selection should depend on the clinical context, type of grading system, balancing diagnostic yield against radiation exposure, and the likelihood of surgical intervention. At our center, we follow the modified second protocol - a single venous-phase acquisition - which provides sufficient information for most patients. Although this approach cannot precisely localize the perforation, it balances the risk of radiation exposure and allows approximation of the necrotic site based on the distribution of free air, with definitive localization achieved during surgical exploration.
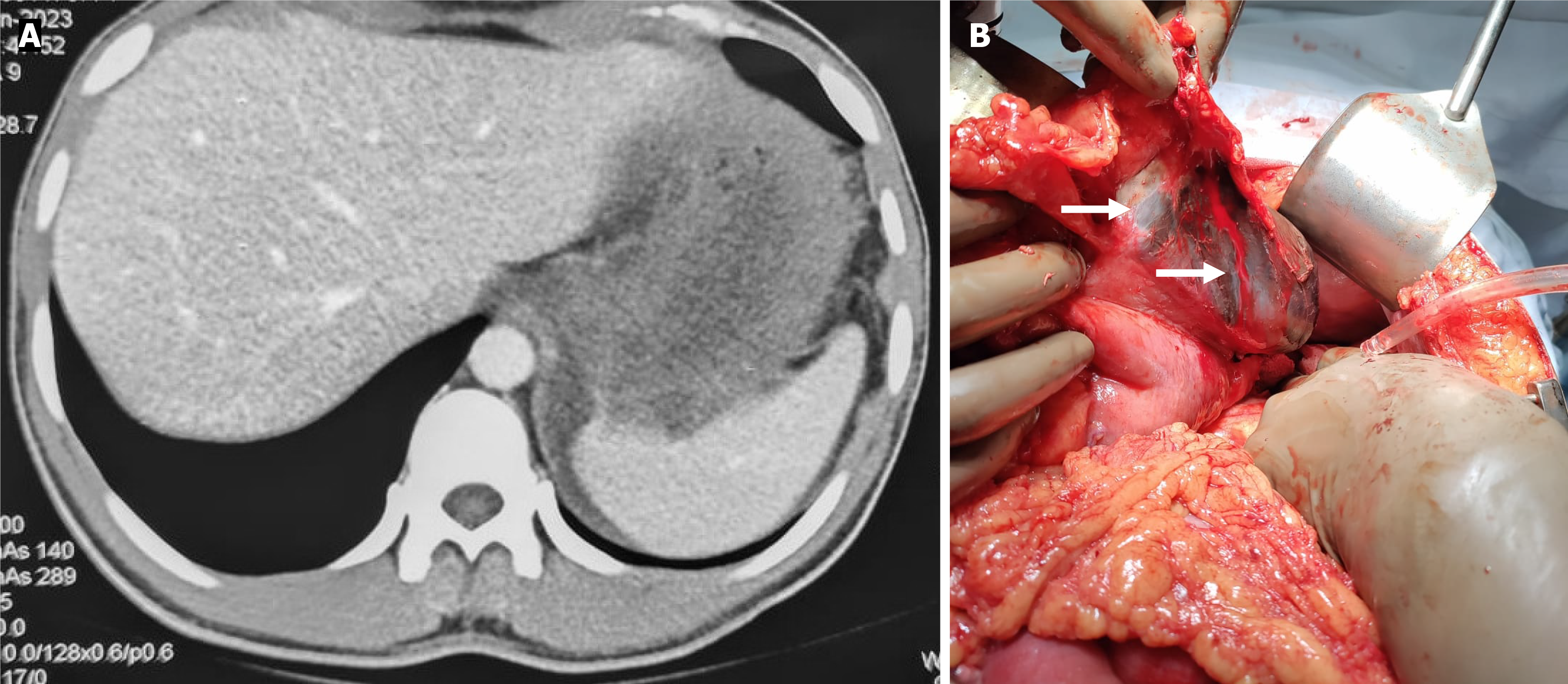
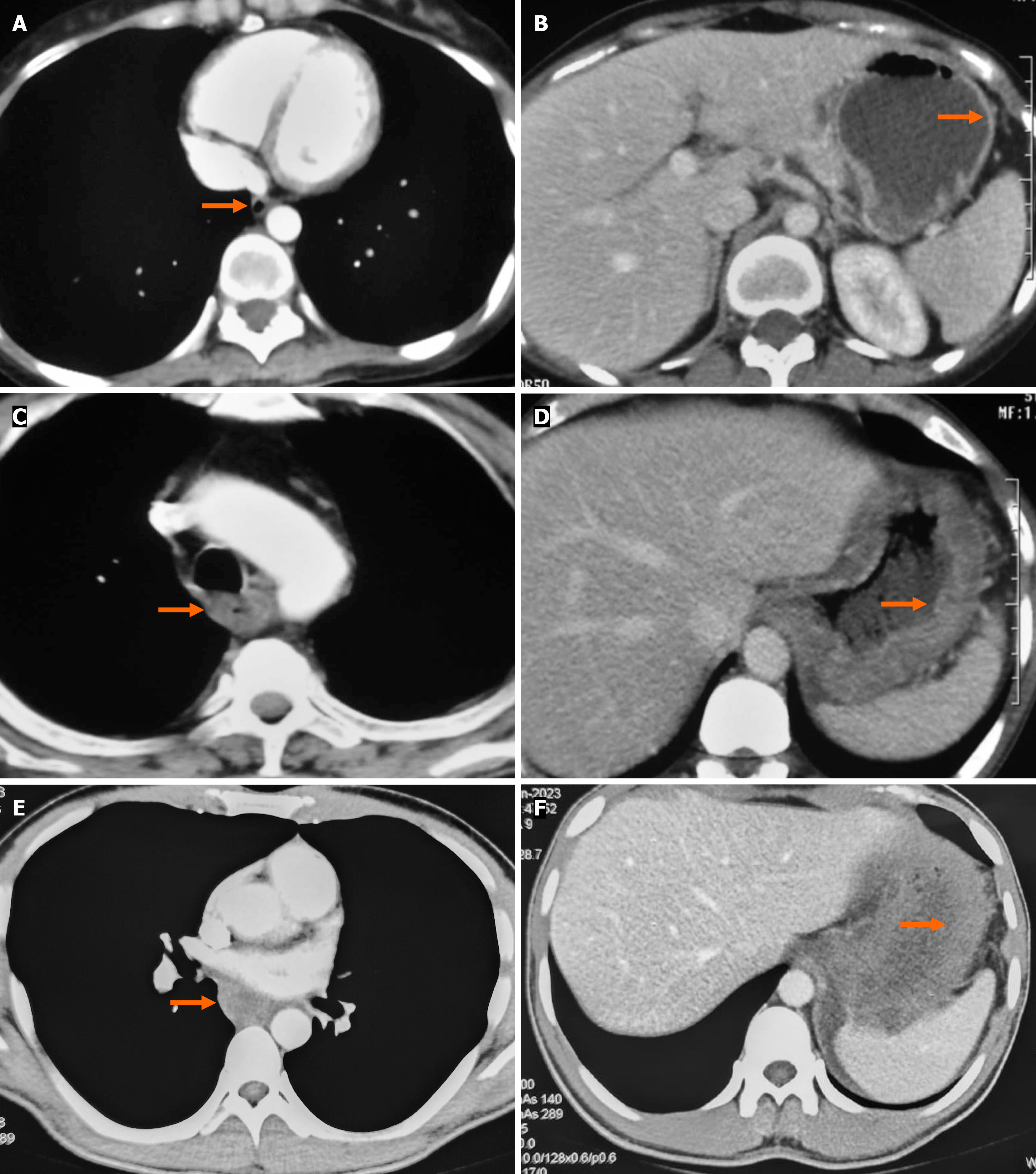
CT grading systems in caustic injury: Several CT-based grading systems have been proposed to standardize the radiological assessment of caustic injuries, the most commonly cited being those by Ryu et al[5] and Chirica et al[7] The Ryu system is a 4-point classification based on esophageal wall thickening and peri-organ collections, with additional assessment of surrounding tissue infiltration and inflammation (Table 2). In contrast, the Chirica system is a simplified 3-point classification that evaluates wall thickening and enhancement, while also incorporating peri-visceral inflammatory changes (Table 2 and Figure 3). The Chirica grading is versatile, as it can be applied to the esophagus, stomach, or other hollow organ, whereas the Ryu grading is specific to esophageal injury. Both systems are useful in predicting stricture development of the esophagus, but Chirica grading also provides valuable short-term prognostic information, including the likelihood of intensive care unit admission and surgical exploration in cases of impending perforation[7].
| Grade | CT findings | Clinical implication |
| Ryu classification | ||
| Grade 1 | No definite swelling of esophagus wall (< 3 mm, within normal limit) | No injury |
| Grade 2 | Edematous wall thickening (> 3 mm) without periesophageal soft tissue infiltration | Usually mild injury; often managed conservatively; low risk of complications |
| Grade 3 | Edematous wall thickening with periesophageal soft tissue infiltration plus well-demarcated tissue interface | Moderate injury; risk of stricture formation; close monitoring required |
| Grade 4 | Edematous wall thickening with periesophageal | Severe injury; high risk of progression; surgical evaluation considered |
| Soft tissue infiltration plus blurring of the tissue | ||
| Interface or localized fluid collection around esophagus or descending aorta | ||
| Chirica classification | ||
| Grade 1 | Normal appearing GI tract | No injury |
| Grade 2 | Wall edema, surrounding soft tissue inflammation and increased postcontrast wall enhancement | Mild to moderate risk of injury; moderate to high risk of stricture formation |
| Grade 3 | Transmural necrosis, as shown by the absence of postcontrast wall enhancement with surrounding soft tissue inflammation | Moderate injury; risk of stricture formation; close monitoring required |
Correlation between CT and endoscopic grading and clinical outcomes: The correlation between CT and endoscopic grading has been studied to evaluate the reliability of CT in predicting the severity and outcomes of caustic injury. Several studies have compared Ryu’s CT grading with Zargar’s endoscopic classification. Ryu et al[5] in their initial report, found poor concordance between the two modalities. Later, Bahrami-Motlagh et al[24] demonstrated only mo
In contrast, studies applying the Chirica grading system have shown improved predictive performance of CT, particularly for identifying necrosis and stricture formation. In a cohort of 120 patients, Chirica et al[7] reported that 19 (15.8%) had injuries overestimated as necrosis on endoscopy, whereas CT correctly excluded necrosis and avoided unnecessary surgical exploration. The same group also demonstrated superior performance of CT compared to endoscopy in pre
Overall, the available evidence suggests that while endoscopy remains more sensitive for detecting mucosal injury and predicting early mortality, CT offers greater specificity for identifying transmural necrosis, stricture risk, and extra-luminal complications such as perforation, mediastinitis, and peritonitis. Importantly, CT may underestimate subtle mucosal changes that are better visualized endoscopically. Thus, the two modalities are best viewed as complementary: Endoscopy remains the gold standard for mucosal grading, whereas CT provides crucial information on depth of injury and extra-luminal pathology, enhancing prognostic accuracy and guiding surgical decision-making.
CT offers several important advantages in the evaluation of acute caustic injury. It is a rapid, widely available, and non-invasive tool that can be performed even in unstable or critically ill patients. Despite these strengths, CT also has several disadvantages. First, the majority of patients with caustic ingestion have low-grade injury[15]. The generalization of CT as an initial assessment modality for injury should be weighed against the risk of radiation exposure. Additionally, the use of intravenous contrast carries risks of nephrotoxicity and allergic reactions. Second, CT is less sensitive than endoscopy in detecting subtle mucosal changes, particularly superficial injuries such as Zargar grade I and IIa lesions, which may be underestimated on imaging[6]. Third, CT grading has been validated during the initial period of injury i.e., for the initial 24-48 hours. A significant proportion of patients present late in the course, and the validity of CT for their assessment of injury is not known. Fourth, most of the available studies on the use of CT for caustic injury include alkali as the main damaging substance. Whether the results of CT apply to patients with acid injury is a matter of research. Fifth, using CT in unstable patients may delay surgical exploration, but this must be balanced against its ability to define the extent of injury and identify other potential causes of hemodynamic instability.
Furthermore, the correlation between CT and endoscopic grading is variable, with some studies showing only mo
The choice between EGD and CT as the initial modality for injury assessment in acute caustic ingestion remains an area of ongoing research. Comparative studies evaluating early endoscopy (within 24-48 hours) and CT of the chest and abdomen have reported variable results, though CT appears to offer greater accuracy in detecting GI necrosis[6,7,21,23,24]. Both modalities have distinct advantages and limitations (Table 3). Endoscopy has traditionally served as the primary tool for assessing the severity and extent of mucosal injury. Until stronger evidence becomes available, endoscopy should remain the initial modality for evaluating the extent and grade of injury. The recently published Indian Society of Gastroenterology guidelines recommend CT as a complementary tool in specific situations[27]: (1) When there is high clinical suspicion or signs of mediastinitis or peritonitis; (2) For assessing GI tract viability in patients with high-grade injury (Zargar grade 3B); (3) When endoscopy is not feasible or contraindicated; and (4) For evaluating suspected injuries beyond the stomach.
| Parameters | EGD | CT scan |
| Easy availability | - | - |
| Can be used for Zargar grading for steroid therapy | - | - |
| Visualization of the entire GIT and other systems | - | - |
| Easily identifies perforation | - | - |
| Identifies the candidates for emergency surgery | - | - |
| Facilitates insertion of the feeding tube | - | - |
| Risk of radiation | No | Yes |
| Validity beyond 48-72 hours of ingestion | - | - |
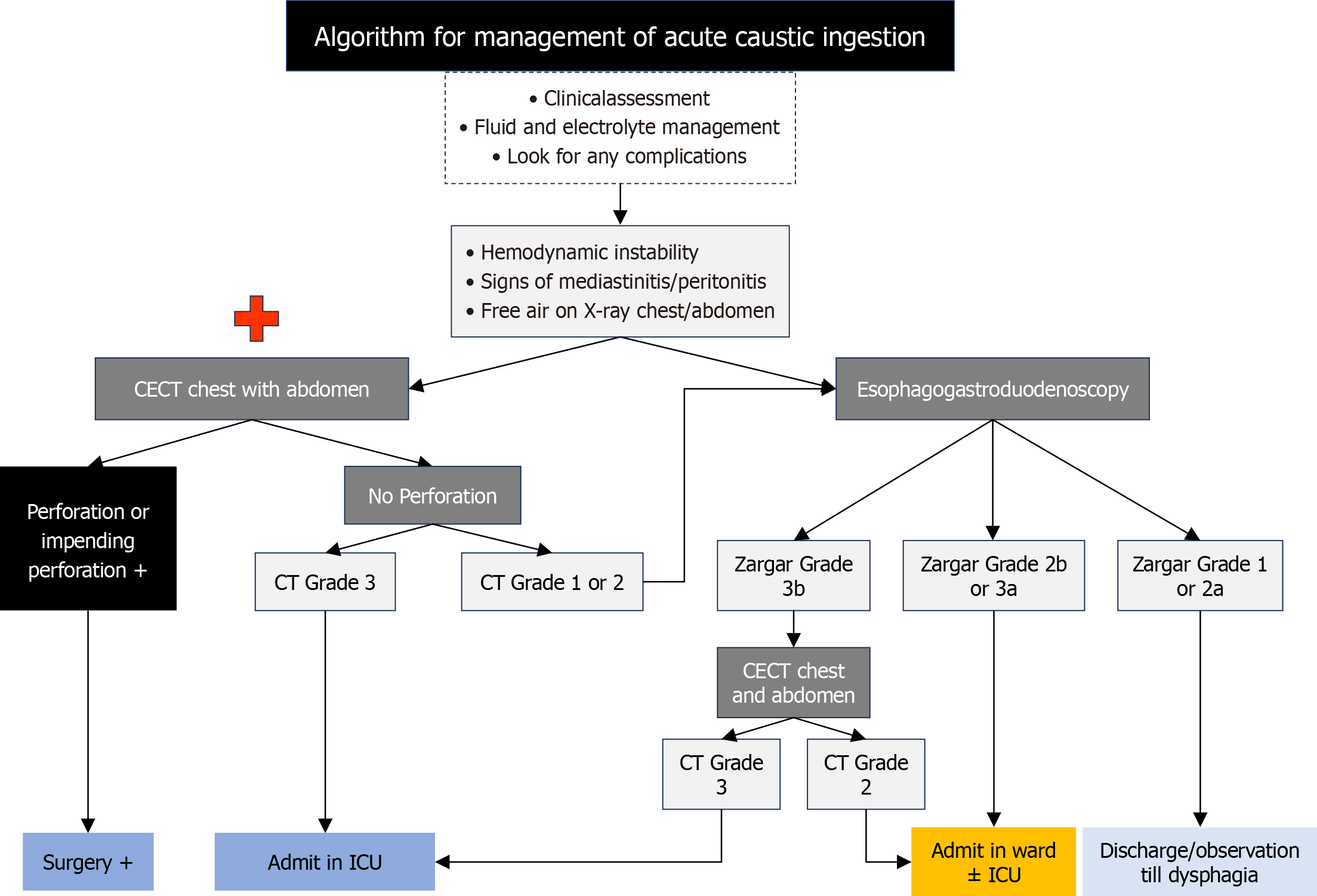
The Indian Society of Gastroenterology guidelines on caustic injury, integrating available evidence and acknowledging the strengths and limitations of each modality, recommend a management algorithm for acute cases in which CT is emphasized as a complementary tool (Figure 4).
Dual-energy CT (DECT) is an emerging technique with potential to enhance the evaluation of acute caustic injury. By acquiring images at two different energy levels, DECT allows a superior ability to differentiate tissues and thus enhance visualization[28]. This can help differentiate viable from necrotic hollow organ wall based on iodine uptake, better assess subtle mucosal and submucosal injury, and detect early ischemia that may be missed on conventional CT. Virtual non-contrast and iodine maps may reduce the need for separate non-contrast scans, thereby minimizing radiation exposure. Although clinical data on DECT in caustic ingestion are not available, its ability to provide functional information on perfusion and tissue viability suggests a promising role in improving diagnostic accuracy, guiding timely surgical in
MRI is a non-invasive imaging modality that can complement the evaluation of caustic GI injury, although supporting literature is currently lacking. It can provide a detailed assessment of transmural and peri-visceral involvement, offer superior soft-tissue contrast to better identify wall edema and necrosis, and allow evaluation of adjacent mediastinal and abdominal structures - features not accessible through endoscopy. However, these advantages overlap with those of CT, and the choice between modalities should be guided by clinical urgency, availability, and radiation considerations. MRI may be particularly valuable in delayed presentations with suspected strictures, fistulas, or vascular involvement, and its lack of ionizing radiation makes it a safer option for children and for repeated follow-up imaging.
Artificial intelligence (AI) and automated image analysis hold promise for improving the evaluation of acute caustic injury. Advanced algorithms, particularly those based on deep learning, can assist in detecting subtle imaging features such as early wall thickening, loss of stratification, or microperforations that may be overlooked on routine CT inter
Despite growing evidence supporting the role of CT in acute caustic injury, significant gaps remain in its standardization and validation. Future research should focus on establishing uniform CT acquisition techniques and contrast protocols, and on developing validated grading systems with consistent definitions for mural injury assessment. The use of AI-based, radiologist-independent tools may further enhance diagnostic accuracy and reduce inter-observer variability. Integrating imaging features with clinical and laboratory data could improve outcome prediction, particularly for complications such as necrosis, perforation, or stricture formation. In addition, studies should evaluate sequential or combined use of CT and endoscopy to optimize management strategies. By addressing these priorities, future studies can refine diagnostic accuracy, improve prognostication, and optimize management pathways, ultimately leading to better outcomes in patients with acute caustic ingestion (Figure 4).
While CT demonstrates superior specificity in predicting necrosis, endoscopy remains more sensitive for detecting subtle mucosal changes. Thus, rather than being competitive, CT and endoscopy should be regarded as complementary modalities - endoscopy for mucosal grading and CT for assessing transmural extension and extra-luminal pathology. The integration of both approaches enables more accurate risk stratification, optimizes decision-making, and ultimately improves patient outcomes.
| 1. | Zargar SA, Kochhar R, Nagi B, Mehta S, Mehta SK. Ingestion of corrosive acids. Spectrum of injury to upper gastrointestinal tract and natural history. Gastroenterology. 1989;97:702-707. [PubMed] |
| 2. | Zargar SA, Kochhar R, Nagi B, Mehta S, Mehta SK. Ingestion of strong corrosive alkalis: spectrum of injury to upper gastrointestinal tract and natural history. Am J Gastroenterol. 1992;87:337-341. [PubMed] |
| 3. | Cheng HT, Cheng CL, Lin CH, Tang JH, Chu YY, Liu NJ, Chen PC. Caustic ingestion in adults: the role of endoscopic classification in predicting outcome. BMC Gastroenterol. 2008;8:31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 123] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 4. | Abbas A, Brar TS, Zori A, Estores DS. Role of early endoscopic evaluation in decreasing morbidity, mortality, and cost after caustic ingestion: a retrospective nationwide database analysis. Dis Esophagus. 2017;30:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 5. | Ryu HH, Jeung KW, Lee BK, Uhm JH, Park YH, Shin MH, Kim HL, Heo T, Min YI. Caustic injury: can CT grading system enable prediction of esophageal stricture? Clin Toxicol (Phila). 2010;48:137-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 6. | Lurie Y, Slotky M, Fischer D, Shreter R, Bentur Y. The role of chest and abdominal computed tomography in assessing the severity of acute corrosive ingestion. Clin Toxicol (Phila). 2013;51:834-837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (2)] |
| 7. | Chirica M, Resche-Rigon M, Pariente B, Fieux F, Sabatier F, Loiseaux F, Munoz-Bongrand N, Gornet JM, Brette MD, Sarfati E, Azoulay E, Zagdanski AM, Cattan P. Computed tomography evaluation of high-grade esophageal necrosis after corrosive ingestion to avoid unnecessary esophagectomy. Surg Endosc. 2015;29:1452-1461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 8. | Crain EF, Gershel JC, Mezey AP. Caustic ingestions. Symptoms as predictors of esophageal injury. Am J Dis Child. 1984;138:863-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 62] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 9. | Uygun I, Aydogdu B, Okur MH, Arayici Y, Celik Y, Ozturk H, Otcu S. Clinico-epidemiological study of caustic substance ingestion accidents in children in Anatolia: the DROOL score as a new prognostic tool. Acta Chir Belg. 2012;112:346-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 10. | Sharif AF, Gameel DEGE, Abdo SAE, Elgebally EI, Fayed MM. Evaluation of Pediatric Early Warning System and Drooling Reluctance Oropharynx Others Leukocytosis scores as prognostic tools for pediatric caustic ingestion: a two-center, cross-sectional study. Environ Sci Pollut Res Int. 2022;29:5378-5395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Gorman RL, Khin-Maung-Gyi MT, Klein-Schwartz W, Oderda GM, Benson B, Litovitz T, McCormick M, McElwee N, Spiller H, Krenzelok E. Initial symptoms as predictors of esophageal injury in alkaline corrosive ingestions. Am J Emerg Med. 1992;10:189-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 52] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Havanond C, Havanond P. Initial signs and symptoms as prognostic indicators of severe gastrointestinal tract injury due to corrosive ingestion. J Emerg Med. 2007;33:349-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 13. | Tosca J, Villagrasa R, Sanahuja A, Sanchez A, Trejo GA, Herreros B, Pascual I, Mas P, Peña A, Minguez M. Caustic ingestion: development and validation of a prognostic score. Endoscopy. 2021;53:784-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Bautista Casasnovas A, Estevez Martinez E, Varela Cives R, Villanueva Jeremias A, Tojo Sierra R, Cadranel S. A retrospective analysis of ingestion of caustic substances by children. Ten-year statistics in Galicia. Eur J Pediatr. 1997;156:410-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 49] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 15. | Chirica M, Resche-Rigon M, Zagdanski AM, Bruzzi M, Bouda D, Roland E, Sabatier F, Bouhidel F, Bonnet F, Munoz-Bongrand N, Marc Gornet J, Sarfati E, Cattan P. Computed Tomography Evaluation of Esophagogastric Necrosis After Caustic Ingestion. Ann Surg. 2016;264:107-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 16. | Vezakis AI, Pantiora EV, Kontis EA, Sakellariou V, Theodorou D, Gkiokas G, Polydorou AA, Fragulidis GP. Clinical Spectrum and Management of Caustic Ingestion: A Case Series Presenting Three Opposing Outcomes. Am J Case Rep. 2016;17:340-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 17. | De Lusong MAA, Timbol ABG, Tuazon DJS. Management of esophageal caustic injury. World J Gastrointest Pharmacol Ther. 2017;8:90-98. [PubMed] [DOI] [Full Text] |
| 18. | Zargar SA, Kochhar R, Mehta S, Mehta SK. The role of fiberoptic endoscopy in the management of corrosive ingestion and modified endoscopic classification of burns. Gastrointest Endosc. 1991;37:165-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 286] [Article Influence: 8.2] [Reference Citation Analysis (2)] |
| 19. | Bonnici KS, Wood DM, Dargan PI. Should computerised tomography replace endoscopy in the evaluation of symptomatic ingestion of corrosive substances? Clin Toxicol (Phila). 2014;52:911-925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 20. | Kochhar R, Ashat M, Reddy YR, Dhaka N, Manrai M, Sinha SK, Dutta U, Yadav TD, Gupta V. Relook endoscopy predicts the development of esophageal and antropyloric stenosis better than immediate endoscopy in patients with caustic ingestion. Endoscopy. 2017;49:643-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Khan HK, Rathi V, Shreshtha S, Tandon A, Gupta A. CT appearances of gastric injury due to caustic ingestion and associated findings (a study of 30 cases). Emerg Radiol. 2023;30:539-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 22. | Chirica M, Kelly MD, Siboni S, Aiolfi A, Riva CG, Asti E, Ferrari D, Leppäniemi A, Ten Broek RPG, Brichon PY, Kluger Y, Fraga GP, Frey G, Andreollo NA, Coccolini F, Frattini C, Moore EE, Chiara O, Di Saverio S, Sartelli M, Weber D, Ansaloni L, Biffl W, Corte H, Wani I, Baiocchi G, Cattan P, Catena F, Bonavina L. Esophageal emergencies: WSES guidelines. World J Emerg Surg. 2019;14:26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 258] [Cited by in RCA: 179] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 23. | Kaewlai R, Noppakunsomboon N, Tongsai S, Tamrakar B, Kumthong N, Teerasamit W, Kongkaewpaisan N, Pisanuwongse A, Amornsitthiwat R, Maitriwong W, Khanutwong C, Apisarnthanarak P. Performance of computed tomography and its reliability for the diagnosis of transmural gastrointestional necrosis in a setting of acute ingestion of predominantly strong acid substances in adults. Clin Toxicol (Phila). 2023;61:346-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 24. | Bahrami-Motlagh H, Hadizadeh-Neisanghalb M, Peyvandi H. Diagnostic Accuracy of Computed Tomography Scan in Detection of Upper Gastrointestinal Tract Injuries Following Caustic Ingestion. Emerg (Tehran). 2017;5:e61. [PubMed] |
| 25. | Bruzzi M, Chirica M, Resche-Rigon M, Corte H, Voron T, Sarfati E, Zagdanski AM, Cattan P. Emergency Computed Tomography Predicts Caustic Esophageal Stricture Formation. Ann Surg. 2019;270:109-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 26. | Chen YJ, Seak CJ, Cheng HT, Chen CC, Chen TH, Sung CM, Ng CJ, Kang SC, Su MY, Hsieh SY. Evaluation of a Diagnostic and Management Algorithm for Adult Caustic Ingestion: New Concept of Severity Stratification and Patient Categorization. J Pers Med. 2022;12:989. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 27. | Singh AK, Gunjan D, Dash NR, Poddar U, Gupta P, Jain AK, Lahoti D, Nayer J, Goenka M, Philip M, Chadda R, Singh RK, Appasani S, Zargar SA, Broor SL, Nijhawan S, Shukla S, Gupta V, Kate V, Makharia G, Kochhar R. Short-term and long-term management of caustic-induced gastrointestinal injury: An evidence-based practice guidelines. Indian J Gastroenterol. 2025;44:646-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 28. | Higuchi Y, Watanabe T, Tabeta A, Yamana H, Tanaka Y, Tsutsumi Y. Diagnostic Accuracy of Dual-Energy Computed Tomography for Bowel Necrosis in Acute Abdomen With Bowel Ischemia. Cureus. 2025;17:e81057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |