Copyright: ©Author(s) 2026.
World J Gastrointest Endosc. Apr 16, 2026; 18(4): 117976
Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.117976
Published online Apr 16, 2026. doi: 10.4253/wjge.v18.i4.117976
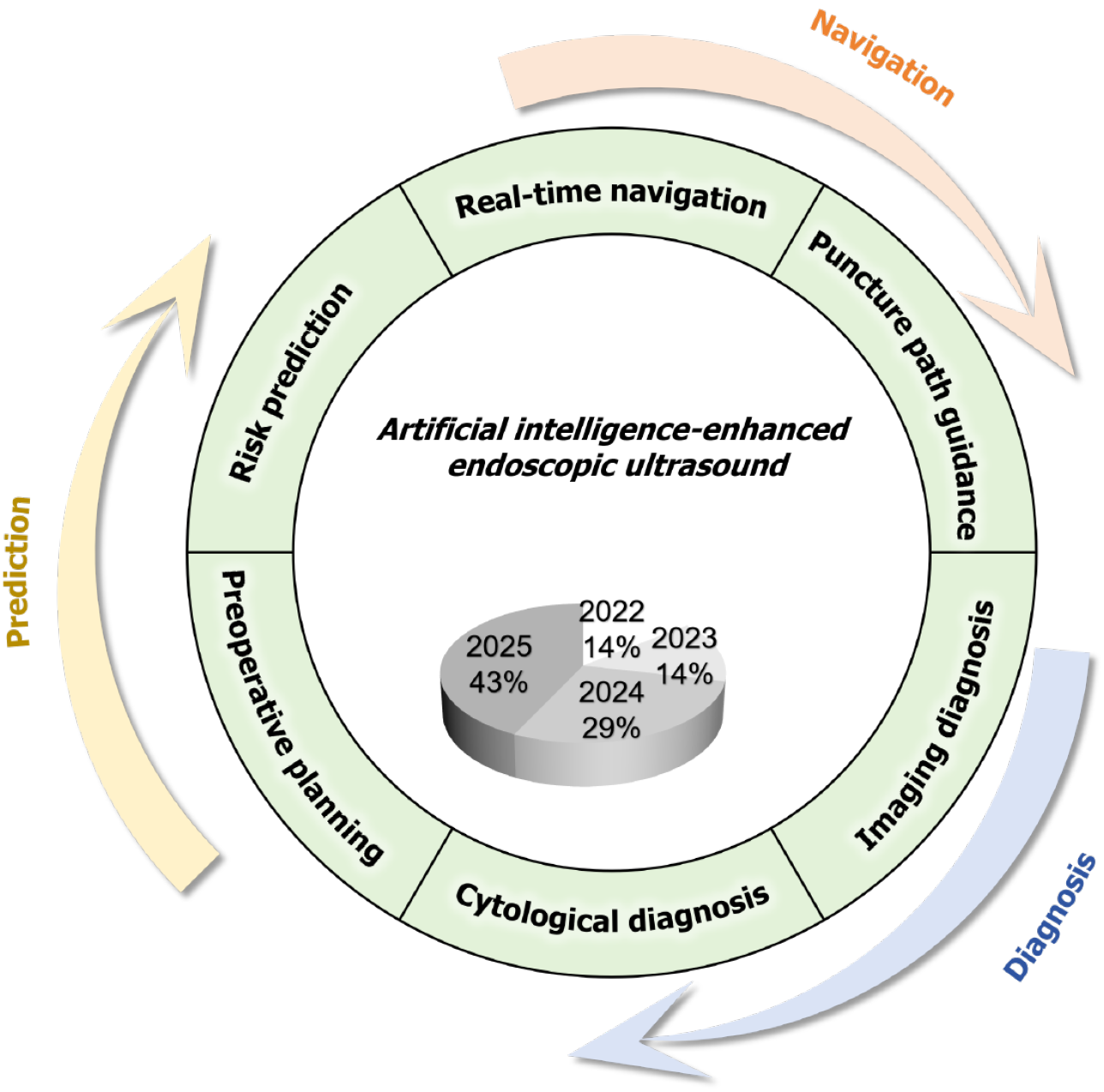
Figure 1 Integrated artificial intelligence framework in the endoscopic ultrasound workflow.
This schematic depicts a three-part closed-loop framework, termed “Prediction-Navigation-Diagnosis” (indicated by arrows), through which artificial intelligence (AI) enhances endoscopic ultrasound. Six core application domains (shown in boxes) include AI-driven preoperative prediction (preoperative planning and risk stratification), intraoperative navigation (real-time navigation and puncture path guidance), and postoperative diagnosis (cytological diagnosis and imaging diagnosis). The central pie chart demonstrates that the representative AI models cited in this review predominantly originate from 2024-2025 (over 70%), highlighting the recent and advanced nature of developments in this field.
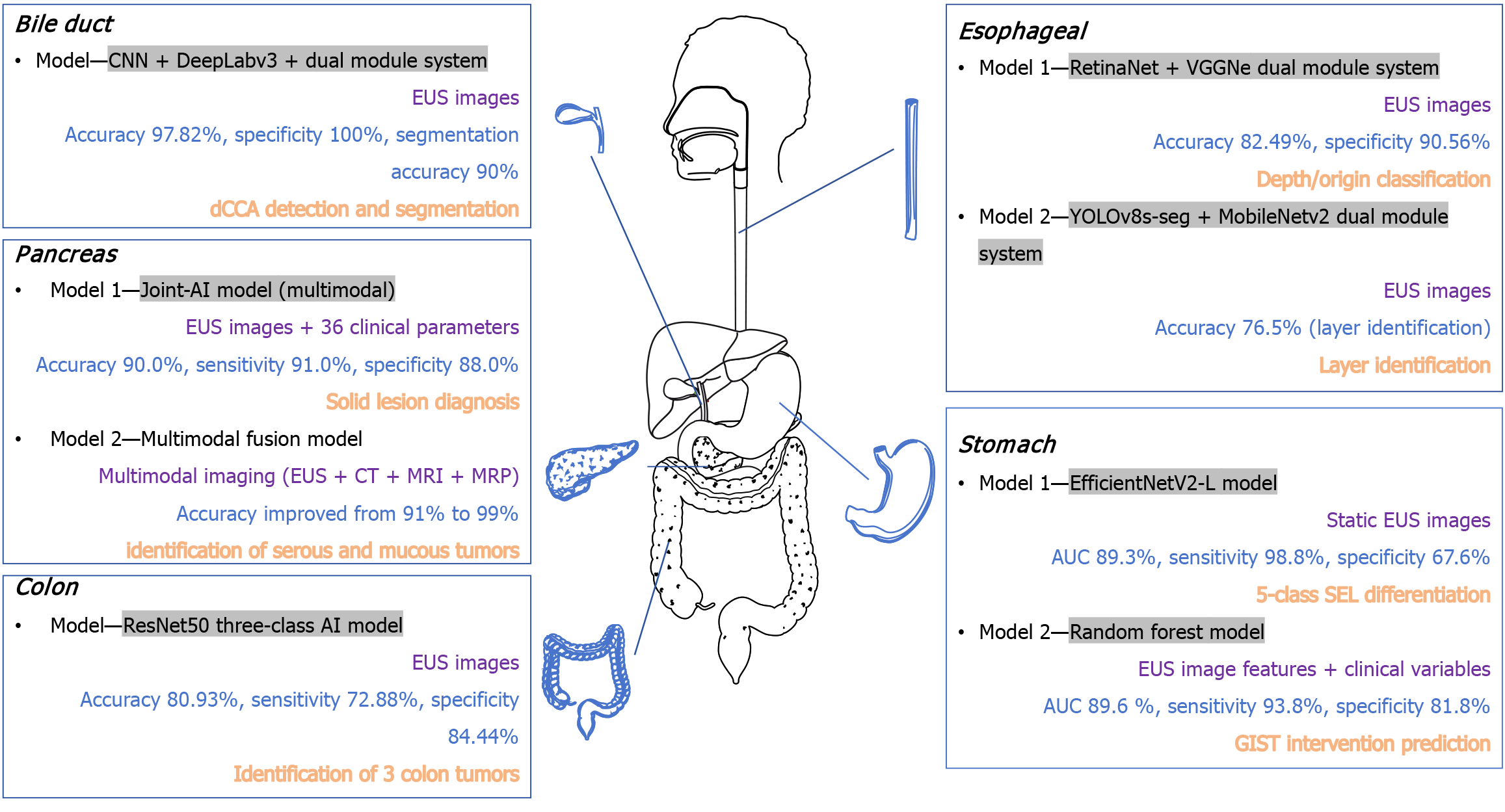
Figure 2 Distribution of artificial intelligence diagnostic models across the digestive system.
This figure summarizes representative artificial intelligence diagnostic models for endoscopic ultrasound, organized by anatomical location within the digestive tract. For each model, four key elements are presented: Model (grey background) specifies the algorithm architecture; data modality (purple) indicates the type of input data used; key metrics (blue) report the primary performance indicators; and application (orange) describes the diagnostic task. The models demonstrate the application of Artificial Intelligence across diverse gastrointestinal pathologies, including subepithelial lesions, tumors, and specific malignancies. AI: Artificial intelligence; AUC: Area under the curve; CNN: Convolutional neural network; CT: Computed tomography; dCCA: Distal cholangiocarcinoma; EUS: Endoscopic ultrasound; GIST: Gastrointestinal stromal tumor; MRI: Magnetic resonance imaging; MRP: Magnetic resonance pancreatography; SEL: Subepithelial lesions.
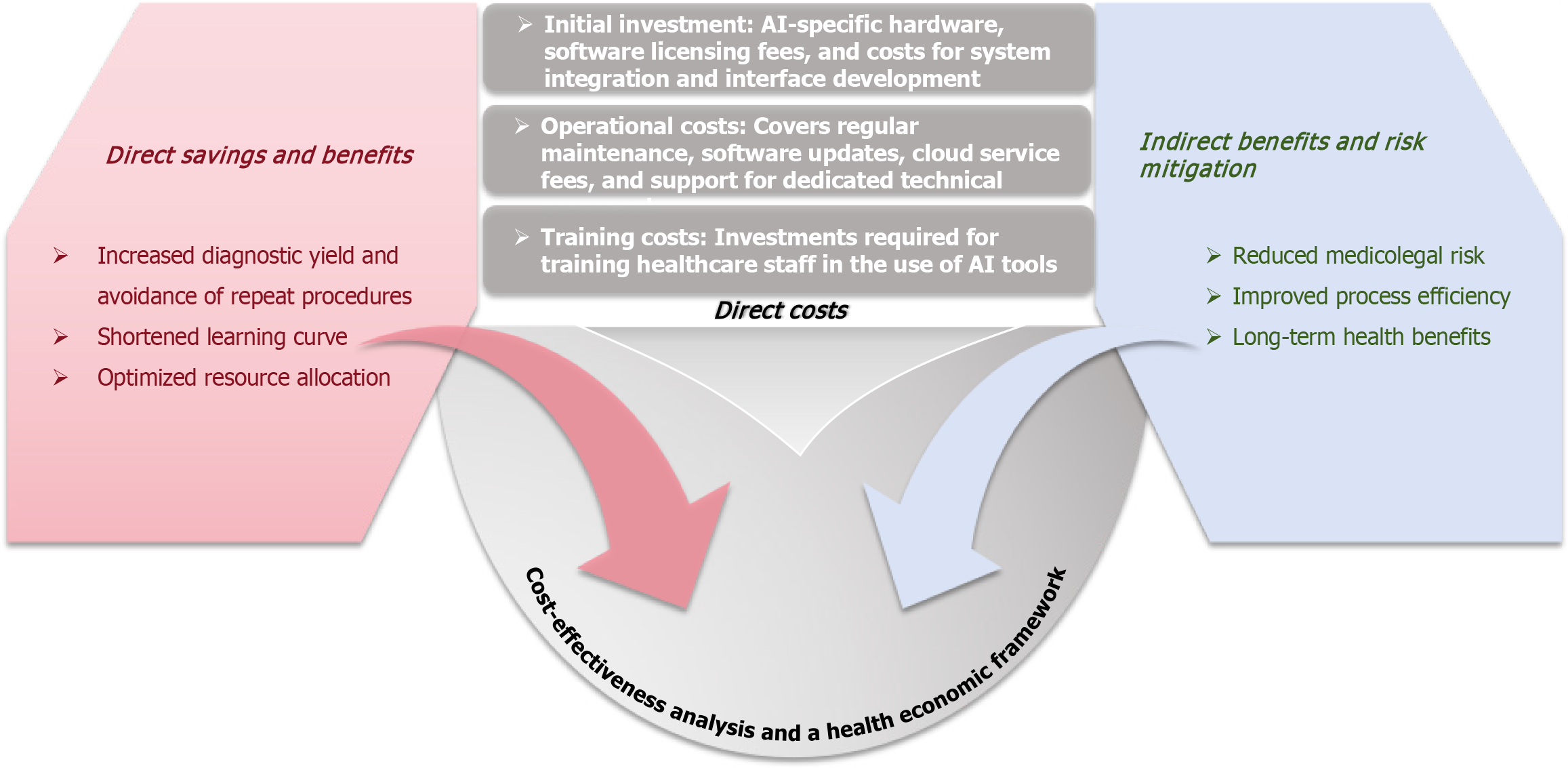
Figure 3 Health economic framework for artificial intelligence-enhanced endoscopic ultrasound workflow.
This flowchart outlines the procedural integration of artificial intelligence in endoscopic ultrasound, accompanied by a structured health economic framework. it details direct costs (e.g., maintenance, cloud services) and indirect costs (e.g., staff training), while highlighting potential savings from reduced repeat procedures, improved diagnostic yield, shortened learning curves, and lower medicolegal risks. This framework supports the cost-benefit analysis essential for the adoption of artificial intelligence in clinical settings. AI: Artificial intelligence.
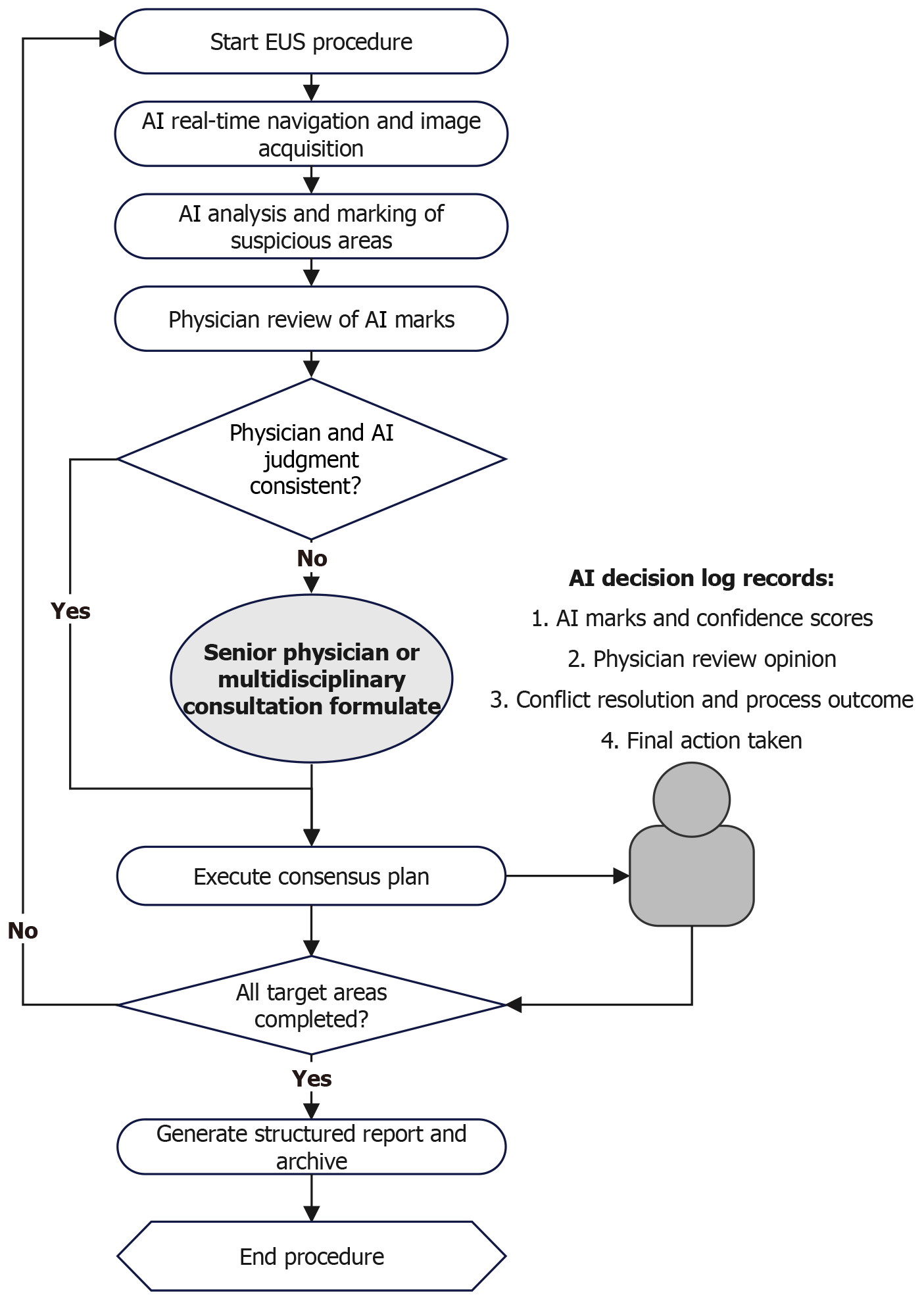
Figure 4 Closed-loop workflow of artificial intelligence-augmented endoscopic ultrasound procedure.
This flowchart delineates the operational integration of artificial intelligence (AI) into the endoscopic ultrasound procedure. Beginning with AI-assisted navigation and image acquisition, it proceeds through sequential steps of AI analysis, physician review, consensus decision-making (with multidisciplinary consultation if needed), and final structured reporting. The diagram visually encapsulates the synergistic human-AI collaboration required to implement the “Prediction-Navigation-Diagnosis” framework in clinical practice. AI: Artificial intelligence; EUS: Endoscopic ultrasound.
- Citation: Chen ZY, Wang YQ, Tan XZ, Liu P, Peng Y. Artificial intelligence in endoscopic ultrasound: Clinical translation of a prediction, navigation, and diagnosis framework. World J Gastrointest Endosc 2026; 18(4): 117976
- URL: https://www.wjgnet.com/1948-5190/full/v18/i4/117976.htm
- DOI: https://dx.doi.org/10.4253/wjge.v18.i4.117976