Published online Jun 27, 2013. doi: 10.4254/wjh.v5.i6.332
Revised: May 1, 2013
Accepted: May 17, 2013
Published online: June 27, 2013
Processing time: 117 Days and 11.5 Hours
The use of direct-acting antiviral agents (e.g., telaprevir, boceprevir) has improved response rates in patients with hepatitis C virus (HCV) genotype 1 infections. Substantial number of drug-drug interactions are anticipated with the use of telaprevir, a cytochrome P450 3A and P-glycoprotein substrate and inhibitor. Herein we describe a patient with HCV-associated hepatocellular carcinoma treated simultaneously with a telaprevir-containing regimen and localized chemotherapy (transcatheter arterial chemoembolization) with doxorubicin. No clinically relevant interactions or adverse events developed while on antiviral therapy.
Core tip: This case suggests that therapy for chronic hepatitis C virus (HCV) infection may be given simultaneously with localized chemotherapy in patients with hepatocellular carcinoma (HCC). The use of telaprevir has improved response rates in patients with HCV genotype 1 infections. Substantial number of drug-drug interactions are anticipated with the use of telaprevir, a cytochrome P450 3A and P-glycoprotein substrate and inhibitor. Herein, we describe a patient with HCV-associated HCC treated simultaneously with a telaprevir-containing regimen and localized chemotherapy (transcatheter arterial chemoembolization). No clinically relevant interactions or adverse events developed while on antiviral therapy.
- Citation: Torres HA, Mahale P, Miller ED, Oo TH, Frenette C, Kaseb AO. Coadministration of telaprevir and transcatheter arterial chemoembolization in hepatitis C virus-associated hepatocellular carcinoma. World J Hepatol 2013; 5(6): 332-335
- URL: https://www.wjgnet.com/1948-5182/full/v5/i6/332.htm
- DOI: https://dx.doi.org/10.4254/wjh.v5.i6.332
Core tip: This case suggests that therapy for chronic hepatitis C virus (HCV) infection may be given simultaneously with localized chemotherapy in patients with hepatocellular carcinoma (HCC). The use of telaprevir has improved response rates in patients with HCV genotype 1 infections. Substantial number of drug-drug interactions are anticipated with the use of telaprevir, a cytochrome P450 3A and P-glycoprotein substrate and inhibitor. Herein, we describe a patient with HCV-associated HCC treated simultaneously with a telaprevir-containing regimen and localized chemotherapy (transcatheter arterial chemoembolization). No clinically relevant interactions or adverse events developed while on antiviral therapy.
The use of direct-acting antiviral agents (e.g., telaprevir, boceprevir) has improved response rates and in many cases shortened treatment durations in patients with hepatitis C virus (HCV) genotype 1 infections[1-3]. In the wake of this development, the American Association for the Study of Liver Diseases recently updated its practice guidelines for the treatment of chronic HCV infections[4]. However, owing to a lack of information about the safety and interactions of these direct-acting antiviral agents, these new guidelines failed to address the issue of HCV infection in special populations, including patients with cancer.
In HCV-infected cancer patients, antiviral therapy is not recommended during chemotherapy or immunosuppressive therapy for the fear of potentiating myelosuppression and causing life-threatening cytopenia. Authors have reported hepatic dysfunction because of acute exacerbation of chronic HCV infection in cancer patients necessitating discontinuation of chemotherapy or immunosuppressive therapy owing to persistently increased aminotransferase levels[5-7]. Even though this acute exacerbation of chronic HCV infection does not lead to fulminant hepatitis, it interrupts the administration of potentially life-saving chemotherapy[8,9].
Information about concomitant HCV treatment in cancer patients undergoing chemotherapy or immunosuppressive therapy is scarce. A small case series looked at management of severe hepatic dysfunction with pegylated interferon-alpha-2a and ribavirin in three children with hematologic malignancies and HCV infections[10]. The investigators started HCV therapy only after dose reduction and intermittent discontinuation of chemotherapy failed to control the hepatic dysfunction and made administration of chemotherapy impossible. Two of the three patients had sustained virological responses, both of whom had genotype 1b infections. More importantly, oncologists were able to initiate chemotherapy in all three cases.
HCV-related hepatocellular carcinoma (HCC) represents a particular challenge, since the majority of patients have poor liver reserve. Therefore, systemic chemotherapy trials in HCC indicated poor tolerance with no survival benefit. Recently, transcatheter arterial chemoembolization (TACE) modality was approved for unresectable HCC, and was extended to patients awaiting liver transplantation as a “bridging therapy”[11,12].
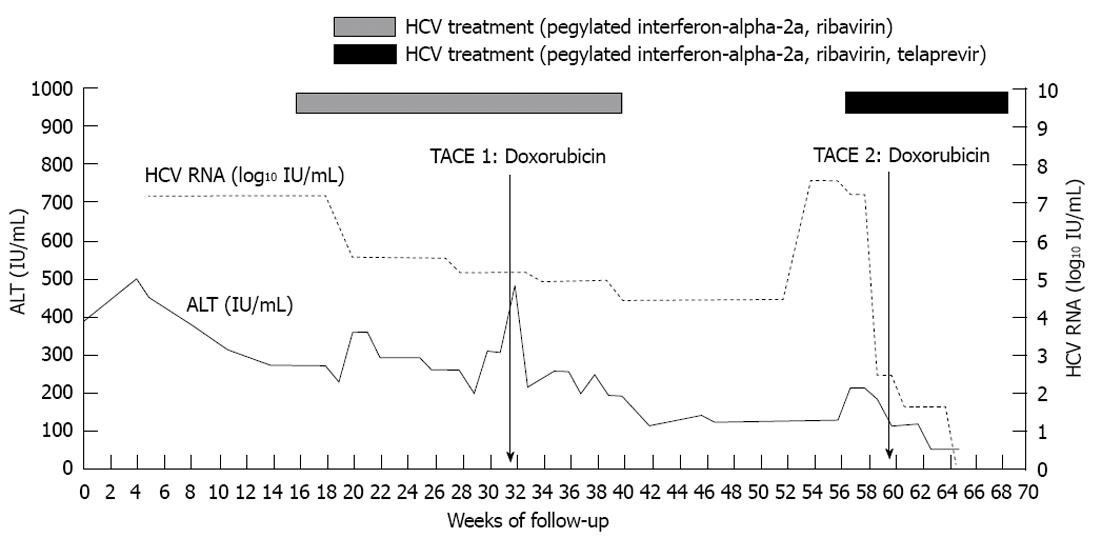
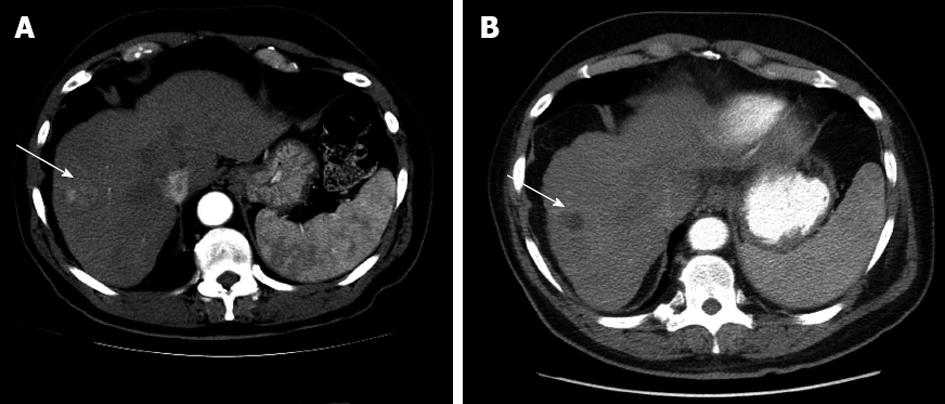
Herein we describe the case of a 68-year-old male with chronic HCV infection with genotype 1b and HCC deemed to be a candidate for TACE and liver transplantation (Table 1). We initiated HCV therapy with pegylated interferon-alpha-2a and ribavirin to prevent allograft infection and normalize alanine aminotransferase levels, as the elevated transaminases were preventing the administration of TACE. Three months after initiation of HCV-targeted treatment with pegylated interferon-alpha-2a and ribavirin as salvage therapy, the patient’s transaminases improved, and we were able to perform the first TACE procedure with doxorubicin drug-eluding beads (Figure 1). The patient had only a partial virological response (reduction in HCV RNA level < 2 log10 IU/mL below baseline) at 24 wk after treatment initiation; therefore, HCV therapy was discontinued. Three months later, we restarted treatment of HCV, now with telaprevir, pegylated interferon-alpha-2a, and ribavirin following the standard guidelines for patients without cancer[4]. Less than a month after treatment initiation, the patient underwent a second TACE procedure with doxorubicin (Figure 2). The transaminases continued to improve after TACE, and the patient’s HCV RNA level was below 43 IU/mL at week 4 of therapy and became undetectable 8 wk after treatment initiation. The patient received dual therapy with pegylated interferon-alpha-2a and ribavirin following 12 wk of the triple combination therapy described above.
| Characteristics | |
| Age, yr | 69 |
| Sex | Male |
| Race | African-American |
| HCV treatment history | Partial responder |
| IL-28B polymorphism | CT |
| Treatment duration before liver transplant, wk | 24 |
| Antiviral treatment toxicity | Hematologic (anemia, neutropenia, thrombocytopenia) |
Patient met the Milan criteria[13] and he underwent liver transplantation when a suitable donor organ became available at week 24 of total antiviral therapy. Liver transplantation was complicated with viremic recurrence first noted 4 wk after transplant, without abnormalities of liver enzymes or graft dysfunction.
During the 24 wk of antiviral therapy, adverse side effects of HCV therapy included anemia, thrombocytopenia, and neutropenia that necessitated administration of growth factors and reduction of the pegylated interferon-alpha-2a and ribavirin doses. HCV therapy with telaprevir-containing regimen did not lead to side effects that were different than those encountered during dual therapy with pegylated interferon-alpha-2a and ribavirin.
The increased incidence of HCC in the United States has resulted in a dramatic rise of patients listed for orthotopic liver transplantation[14] with concomitant increased waiting-times for all patients. This prolonged wait time allows consideration of treatment of HCV while waiting for transplantation in those patients with adequate liver function who are listed for HCC-related reasons as achievement of sustained virological response prior to transplantation could eliminate the risk of HCV recurrence post-transplant[15,16].
A recent study by Garg et al[17] concluded that co-administration of telaprevir, a cytochrome P450 3A and P-glycoprotein substrate and inhibitor, and cyclosporine or tacrolimus was associated with major interactions with significantly increased blood concentrations of these immunosuppressive agents, which could lead to serious or life-threatening adverse events. We did not encounter any clinically relevant adverse effects of administration of the telaprevir-containing regimen during TACE with doxorubicin used and loco-regional therapy.
To our knowledge, pharmacokinetic studies have not been conducted looking for drug-drug interactions between doxorubicin (used as part of TACE) and telaprevir. Although such interactions remain theoretical, they are possible as systemic concentrations of doxorubicin can be identified following TACE. For example, in one study using a rabbit liver tumor model, plasma concentration of doxorubicin after TACE was as high as 360.5 ng/mL[18]. In a phase 2 study designed to establish the efficacy and safety of drug eluting beads loaded with doxorubicin for the TACE treatment of HCC patients, doxorubicin Cmax and AUC were 78.97 ± 38.3 ng/mL and 662.6 ± 417.6 ng/mL respectively[19]. However, TACE procedure with doxorubicin drug-eluding beads is associated with minimal systemic exposure of the chemotherapeutic agent with negligible systemic toxicity[20].
This case suggests that therapy for chronic HCV infection may be given simultaneously with a localized chemotherapeutic modality (TACE) in patients with HCC, given the tolerability of this procedure in compensated liver cirrhosis. However, this intervention should be used with caution and only in the salvage setting, as more studies are required to evaluate the interactions of direct-acting antiviral agents with chemotherapeutic agents.
| 1. | Poordad F, McCone J, Bacon BR, Bruno S, Manns MP, Sulkowski MS, Jacobson IM, Reddy KR, Goodman ZD, Boparai N. Boceprevir for untreated chronic HCV genotype 1 infection. N Engl J Med. 2011;364:1195-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2017] [Cited by in RCA: 1923] [Article Influence: 128.2] [Reference Citation Analysis (1)] |
| 2. | Bacon BR, Gordon SC, Lawitz E, Marcellin P, Vierling JM, Zeuzem S, Poordad F, Goodman ZD, Sings HL, Boparai N. Boceprevir for previously treated chronic HCV genotype 1 infection. N Engl J Med. 2011;364:1207-1217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1314] [Cited by in RCA: 1268] [Article Influence: 84.5] [Reference Citation Analysis (0)] |
| 3. | Jacobson IM, McHutchison JG, Dusheiko G, Di Bisceglie AM, Reddy KR, Bzowej NH, Marcellin P, Muir AJ, Ferenci P, Flisiak R. Telaprevir for previously untreated chronic hepatitis C virus infection. N Engl J Med. 2011;364:2405-2416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1926] [Cited by in RCA: 1783] [Article Influence: 118.9] [Reference Citation Analysis (0)] |
| 4. | Ghany MG, Nelson DR, Strader DB, Thomas DL, Seeff LB. An update on treatment of genotype 1 chronic hepatitis C virus infection: 2011 practice guideline by the American Association for the Study of Liver Diseases. Hepatology. 2011;54:1433-1444. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 822] [Cited by in RCA: 824] [Article Influence: 54.9] [Reference Citation Analysis (1)] |
| 5. | Gigliotti AR, Fioredda F, Giacchino R. Hepatitis B and C infection in children undergoing chemotherapy or bone marrow transplantation. J Pediatr Hematol Oncol. 2003;25:184-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 6. | Firpi RJ, Nelson DR. Management of viral hepatitis in hematologic malignancies. Blood Rev. 2008;22:117-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 7. | Zuckerman E, Zuckerman T, Douer D, Qian D, Levine AM. Liver dysfunction in patients infected with hepatitis C virus undergoing chemotherapy for hematologic malignancies. Cancer. 1998;83:1224-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Mahale P, Kontoyiannis DP, Chemaly RF, Jiang Y, Hwang JP, Davila M, Torres HA. Acute exacerbation and reactivation of chronic hepatitis C virus infection in cancer patients. J Hepatol. 2012;57:1177-1185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 101] [Article Influence: 7.2] [Reference Citation Analysis (3)] |
| 9. | Torres HA, Davila M. Reactivation of hepatitis B virus and hepatitis C virus in patients with cancer. Nat Rev Clin Oncol. 2012;9:156-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 121] [Article Influence: 8.6] [Reference Citation Analysis (3)] |
| 10. | Papaevangelou V, Varsami M, Papadakis V, Zellos A, Parcharidou A, Papargyri S, Karentzou O, Manolaki N, Roma E, Polychronopoulou S. Hepatitis C treatment concomitant to chemotherapy as “salvage” therapy in children with hematologic malignancies. Pediatr Infect Dis J. 2010;29:277-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Farmer RW, Kralj I, Valdata A, Urbano J, Enguix DP, García Mónaco R, Scoggins CR, McMasters KM, Rustein L, Martin RC. Hepatic arterial therapy as a bridge to ablation or transplant in the treatment of hepatocellular carcinoma. Am Surg. 2011;77:868-873. [PubMed] |
| 12. | De Luna W, Sze DY, Ahmed A, Ha BY, Ayoub W, Keeffe EB, Cooper A, Esquivel C, Nguyen MH. Transarterial chemoinfusion for hepatocellular carcinoma as downstaging therapy and a bridge toward liver transplantation. Am J Transplant. 2009;9:1158-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 101] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 13. | Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, Montalto F, Ammatuna M, Morabito A, Gennari L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5914] [Cited by in RCA: 5277] [Article Influence: 175.9] [Reference Citation Analysis (12)] |
| 14. | Wertheim JA, Petrowsky H, Saab S, Kupiec-Weglinski JW, Busuttil RW. Major challenges limiting liver transplantation in the United States. Am J Transplant. 2011;11:1773-1784. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 143] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 15. | Terrault NA. Hepatitis C therapy before and after liver transplantation. Liver Transpl. 2008;14 Suppl 2:S58-S66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 44] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Everson GT, Trotter J, Forman L, Kugelmas M, Halprin A, Fey B, Ray C. Treatment of advanced hepatitis C with a low accelerating dosage regimen of antiviral therapy. Hepatology. 2005;42:255-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 243] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 17. | Garg V, van Heeswijk R, Lee JE, Alves K, Nadkarni P, Luo X. Effect of telaprevir on the pharmacokinetics of cyclosporine and tacrolimus. Hepatology. 2011;54:20-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 189] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 18. | Gupta S, Wright KC, Ensor J, Van Pelt CS, Dixon KA, Kundra V. Hepatic arterial embolization with doxorubicin-loaded superabsorbent polymer microspheres in a rabbit liver tumor model. Cardiovasc Intervent Radiol. 2011;34:1021-1030. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 19. | Varela M, Real MI, Burrel M, Forner A, Sala M, Brunet M, Ayuso C, Castells L, Montañá X, Llovet JM. Chemoembolization of hepatocellular carcinoma with drug eluting beads: efficacy and doxorubicin pharmacokinetics. J Hepatol. 2007;46:474-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 761] [Cited by in RCA: 686] [Article Influence: 36.1] [Reference Citation Analysis (5)] |
P- Reviewers Lonardo A, Ramos S, Wong GLH S- Editor Huang XZ L- Editor A E- Editor Li JY