Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.110217
Revised: July 14, 2025
Accepted: January 21, 2026
Published online: March 27, 2026
Processing time: 296 Days and 14.8 Hours
Still, in most endemic countries, hepatitis A (HA) is a serious socio-medical problem due to high morbidity, leading to frequent hospitalizations and financial burdens on health systems.
To find out the clinical spectrum of HA infection in children, the relationship between atypical manifestations and laboratory findings and the outcome of patients with atypical HA virus (HAV) manifestations.
A total of 115 patients aged 0-14 years (mean 5.2 ± 3.5 years) were admitted to the hospital with symptoms of HA and a positive IgM anti-HAV test result during the period from July 2018 to December 2023. Patients were followed up weekly until they had made a complete recovery.
Atypical features were present in 30 (26%) children; prolonged cholestasis 11 (9.56%), acute acalculous cholecystitis (AAC) 15 (13.04%), ascites 2 (1.73%), thrombocytopenia 1 (0.86%) and fulminant hepatitis 1 (0.86%) were observed. Pruritus, as well as higher serum total and direct bilirubin levels, were statistically significant in children with atypical manifestations of the disease. Moreover, this group had a prolonged mean duration of jaundice and hospital course.
Atypical manifestations such as protracted cholestasis and AAC are not uncommon in children with HA.
Core Tip: We showed that most cases of hepatitis A (HA) 30/115 (26.08%) can occur with atypical symptoms that require longer hospitalization. Most cases occur in patients with higher serum bilirubin levels, gamma-glutamyl transferase levels, and persistent icterus. High serum bilirubin levels and their prolonged retention, as well as the thickening of the gallbladder wall, can be perceived as prognostic signs of a possible transition into an atypical clinical course for the patient with HA. Atypical manifestations such as prolonged cholestasis and acute acalculous cholecystitis are not uncommon in children with HA.
- Citation: Velev V, Tomov T, Popov M, Tomov L, Golkocheva-Markova EN, Pavlova M, Velikova T, Mitova-Mineva Y. Some peculiarities in the abnormal course of hepatitis A in childhood. World J Hepatol 2026; 18(3): 110217
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/110217.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.110217
Acute hepatitis (AH) is an inflammatory disease of the liver, most often of viral origin. Hepatitis A (HA) is the most common viral liver infection worldwide. It is caused by the RNA HA virus (HAV), which is responsible for enteral transmission of disease. HA is ubiquitous, but its incidence is highest in countries with low hygiene standards and is relatively rare in highly industrialized countries[1,2]. In most cases, the encounter with HAV occurs relatively early in an individual's development, and therefore, children and adolescents are most often affected[2].
Still, in most endemic countries, HA is a serious socio-medical problem due to high morbidity, leading to frequent hospitalizations and financial burdens on health systems. The reported annual incidence worldwide is about 1.5 million[2]. This likely represents only a limited portion of the reported cases, as surveillance systems in many endemic countries are insufficiently effective. Furthermore, the disease often occurs in asymptomatic or abortive forms, remaining undiagnosed[3]. In the European Union (EU) since 2016, cases have increased mainly due to men having sex with men and epidemic outbreaks among minority groups in Southeast and Southern Europe[1,4]. For 2022, 4.43 cases per 100000 people were reported in Bulgaria, which ranks our country 4th after Croatia, Hungary and Romania. The average incidence in the EU is 1.0 per 100000 for the same year. In Bulgaria, HA is most common among 5-14-year-old children (12.71/100000), followed by 5-24-year-olds (8.50/100000) and 0-4-year-old (6.16/100000)[5]. The disease usually proceeds as a self-limiting infection. Acute liver failure (ALF) and/or death are extremely rare. Chronification has not been described[1,2,6]. However, in rare cases, the infection may deviate from its usual clinical course and proceed atypically, more severely, with a protracted course[2,7]. Cholestatic hepatitis, edematous-ascites forms, and some extrahepatic features, such as ascites, pleural or pericardial effusions, acute acalculous cholecystitis, thrombocytopenia, acute renal failure (AKI), pancreatitis, neurological manifestations, and ALF, have been described[1,7,8]. There is still limited literature data on the atypical clinical spectrum in children with HA. Our study aims to investigate the correlation between specific clinical and laboratory manifestations in a child population in Bulgaria.
The retrospective study was conducted at the Specialized Hospital for Infectious and Parasitic Diseases “Prof. Iv. Kirov”, a tertiary hospital, from July 2018 to December 2023. The studies were performed in accordance with the local ethics committee of the Hospital “Prof. Iv. Kirov” and with informed consent of the parents/legal guardians in accordance with the Helsinki Declaration.
One hundred fifteen persons who fulfill the following criteria were hospitalized: (1) Age dup to 14 years; (2) Clinical and biochemical characteristics suggestive of HA; (3) Positive serology for the disease (positive anti-HAV-IgM) as determined through ELISA or CLIA; and (4) Clinical signs of complications, including persistent vomiting, dehydration, cholestatic jaundice, hepatomegaly with elevated transaminases, suspicion of fulminant hepatitis, or atypical symptoms requiring closer monitoring.
As part of the pre-set diagnostic criteria, serological testing was repeated within 7 days following an initial negative anti-HAV-IgM result. Strict measures were taken to ensure the quality of the sales kits met the manufacturer's specifications. The results are considered valid when these control and quality measures are strictly implemented. Fresh serum or plasma samples were used and, if necessary, stored according to laboratory practice at 2 °C-8 °C for no more than 48 hours. All patients were regularly monitored for hematological and basic biochemical parameters at least three times during bedtime. Molecular testing for HAV RNA was not available during the study period, and long-term follow-up data could not be retrieved due to the retrospective design.
Each child underwent an abdominal ultrasound (US) using a Samsung Medison Convex Array US Transducer with a Frequency Range of 3.5-7 MHz. The patients fasted 12 hours before the planned US, which was performed by two licensed US specialists with qualifications in the field of infectious diseases. Longitudinal, transverse, and inclined sections were created in real-time. For the accuracy of the image parameters, we utilized the data from Konuş et al[9] and the European Federation of US in Medicine and Biology (EFSUMB)-EFSUMB Pediatric Registry[9,10]. Some children underwent X-rays. For each patient, an individual card was completed with clinical, laboratory, and imaging indicators.
The study was conducted in accordance with the Declaration of Helsinki (2000) and was approved by the university’s ethics committee at the hospital. Children with twice negative anti-HAV-IgM markers, with underlying other liver disease, or whose parents refused to participate were excluded from the study. In most children, a control clinical examination was performed 30 days after discharge, which included a study of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and total bilirubin (TB). If necessary, gamma-glutamyl transferase (GGT) and serum albumin levels were also examined, and a control abdominal US was performed. In some children, a second follow-up exa
HA is defined as an acute infectious disease, most often preceded by prodromal manifestations-fatigue, nausea, lack of appetite, vomiting, fever associated with elevated liver enzymes at least twice the norm, in most cases and increased total bilirubin (≥ 2 mg/dL) in the absence of underlying other liver or biliary disease. Typical HA was classified as a disease characterized by prodromal manifestations preceding the icteric phase, which resolves within 2-3 weeks. Atypical HA-as prolonged cholestasis ≥ 12 weeks, accompanied by itching, total serum bilirubin ≥ 10 mg/dL, and direct should be > 50% of it. A new deterioration in the clinical and biochemical recovery of the patient is defined as a relapse within 6 months from the onset of acute illness[8,11].
Also, cases of oblivious seroconversion, which we defined as negative anti-HAV-IgM serology during hospitalization and positivity and during retesting after 7 days. A thickened gallbladder wall was defined as a wall that is ultra soundly more than 3 mm, with or without pericholecystic edema, together with spontaneous or palpable pain in the right hypochondrium and fever. Ascites is determined by US detection of freely mobile fluid in the abdominal cavity. Pleural effusion is defined as the presence of free fluid in the pleural cavity, as evidenced by a chest X-ray.
Intravenous hemolysis was defined as a case of a drop in hemoglobin combined with an increased number of reticulocytes. Thrombocytopenia-reduced platelet count below 150.000/mm3, excluding other causes, including drug interactions. ALF was defined as an acute deterioration of objective and laboratory status during HA with an international normalized ratio ≥ 1.5 and evidence of hepatic encephalopathy within 4 weeks of symptom onset[8,11,12].
The statistical analysis was performed using the SPSS, version 23, and Octave 8.3. Continuous data were represented as the arithmetic mean with SD or the median, and discrete variables were defined as n (%). To compare continuous variables between two groups, a student t-test was used. The categorical variables were compared using the Fisher exact test and the χ2 test. Variables with a sample size greater than 50 and P < 0.05 were considered statistically significant. In the analysis of the data comparing the two groups, the Breslow-Day and Tarone’s tests for homogeneity of odds ratios, as well as the Cochran-Mantel-Haenszel test for conditional independence, were also used.
A total of 115 patients up to 14 years of age were admitted to the hospital with symptoms of acute HA in the period July 2018 to December 2023.
Table 1 presents the primary clinical and laboratory indicators, as well as the types of atypical courses we have observed.
| Symptoms | n (%) | |
| Clinical features of hepatitis A | Interval from symptom onset to hospital admission (days) | 5.5 (2-9) |
| Average length of hospitalization (days) | 6.86 (5-18) | |
| Аverage time of pre icteric period (days) | 4.3 (1-7) | |
| Presenting symptoms (%) | General weakness | 72 |
| Fever | 52 | |
| Jaundice | 98 | |
| Nausea/vomiting | 73 | |
| Аbdominal pain | 44 | |
| Diarrhea | 18 | |
| Laboratory parameters | AST (IU/L), initial/peak level (normal range) | 910/1350 (< 40) |
| ALT (IU/L), initial/peak level (normal range) | 1020/1410 (< 40) | |
| Bilirubin (mg/dL), initial/peak level (normal range) | 3.4/6.8 (0.2-1.2) | |
| WBC (103/mm3), initial level (normal range) | 6.1 (4-10) | |
| Hemoglobin (g/dL), initial level (normal range) | 12.5 (12-16) | |
| Platelet (103/mm3), initial level (normal range) | 166 (130-360) | |
| Clinical outcomes | Typical course/recovery without complications | 85 (73.91) |
| Prolonged icteric period | 11 (9.56) | |
| Acute acalculous cholecystitis | 15 (13.04) | |
| Ascites | 2 (1.73) | |
| Fulminant hepatitis, spontaneous recovery | 1 (0.86) | |
| Thrombocytopenia | 1 (0.86) |
In addition to acalculous cholecystitis, hepatomegaly was present in 64% of patients, splenomegaly in 9%, and ascites in 2%. Periportal edema was documented in 2.5% of the cases.
The patients were classified into two groups: Group A, characterized by typical manifestations, and group B, with atypical manifestations (Table 2).
| Parameter | Typical | Atypical | P value |
| Clinical manifestation | 85 (73.9) | 30 (26) | N/A |
| Mean age (years) | 4.15 | 8.28 | 0.002 |
| Males | 42 (49.41) | 16 (53.33) | N/A |
| Average length of hospitalization (days) | 5.14 (3-7) | 8.64 (5-18) | 0.005 |
| Fever | 46 (54.11) | 27 (90) | 0.002 |
| Nausea/vomiting | 51 (60.0) | 25 (83.33) | 0.001 |
| Pruritus | 32 (37.64) | 23 (76.66) | 0.03 |
| Аbdominal pain | 40 (47.05) | 17 (56.66) | NS |
| Diarrhea | 13 (15.29) | 8 (26.66) | 0.02 |
| Headache | 28 (32.94) | 15 (50) | 0.005 |
| AST (IU/L), peak level | 1250 | 1640 | 0.01 |
| ALT (IU/L), peak level | 1035 | 2268 | 0.02 |
| Bilirubin (mg/dL), peak level | 3.4 | 8.9 | 0.02 |
| WBCs (103/mm3), initial level | 6.7 | 13.9 | 0.007 |
| Hemoglobin (g/dL), initial level | 11.4 | 8.9 | NS |
| Platelet (103/mm3), initial level | 155 | 148 | NS |
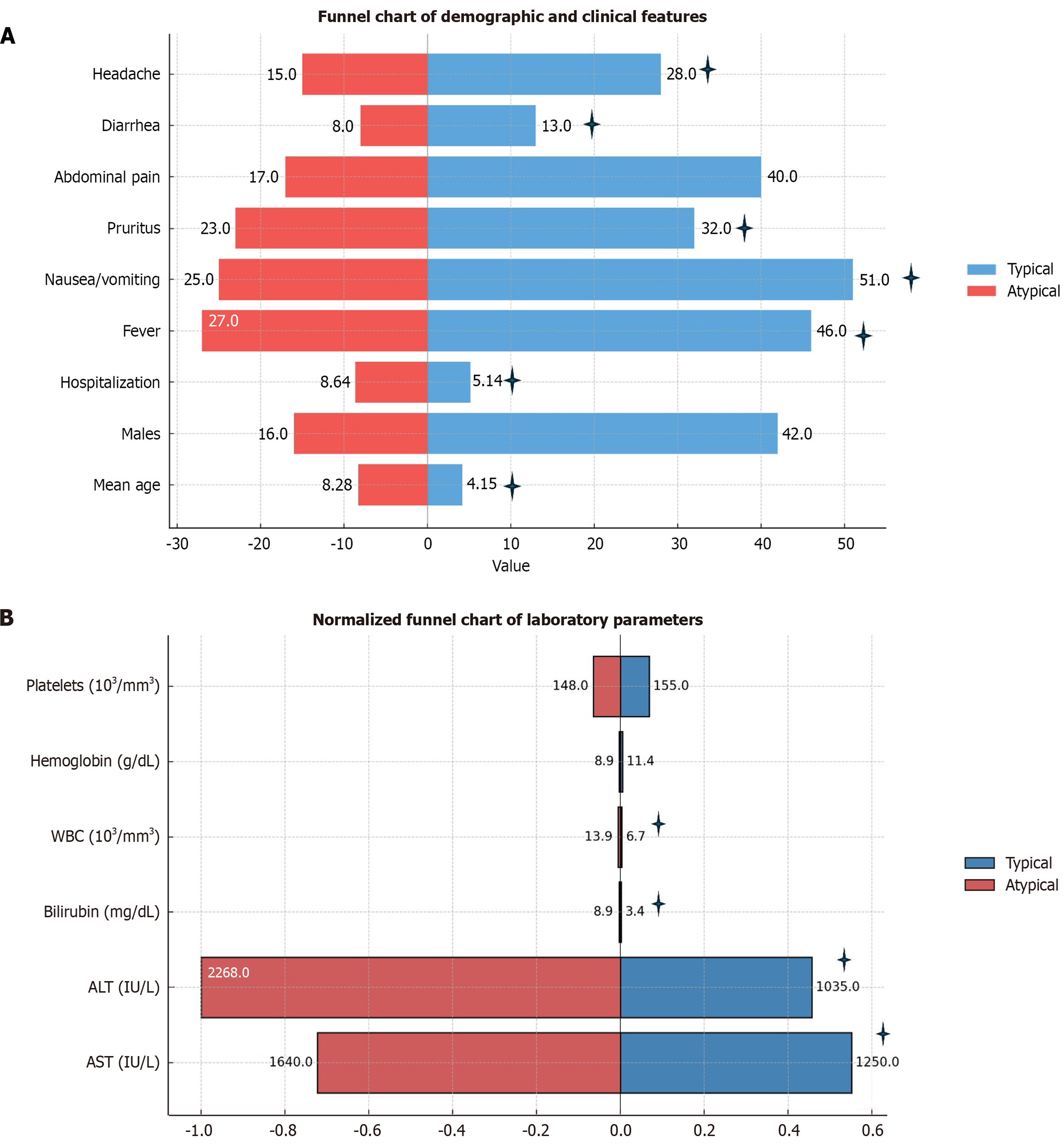
In patients in the second group, additional laboratory and/or imaging tests were sometimes required. In terms of clinical symptoms ingroup B, fever, itching and headache are significantly more common, with a pronounced statistical relationship (Figure 1A).
The longer hospital stay period in these patients is also highly statistically dependent. The situation is similar to the symptoms of nausea, vomiting, and diarrhea, although with a lower statistical dependence. We did not observe a statistical relationship in terms of sex; however, an age-related difference exists-the atypical course is more common in older children.
Regarding laboratory parameters, as shown in Figure 1B, children with an atypical course exhibit increased peak values of AST and ALT, particularly serum total bilirubin, with a clear statistical relationship. Leukocytosis also exhibits a smaller but statistically significant association. We did not find a statistically significant difference in platelet count and hemoglobin between groups A and B.
We also reported differences in prolonged cholestasis between the two categories. In children of category B, it was present in 11 children (all of whom had other clinical and laboratory changes), with an average duration of 12.5 days. In contrast, group A had only 1 child with a longer icteric period, approximately 7 days (P < 0.02), without any other atypical clinical or laboratory changes. The total duration of the icteric period in group B children differed significantly statistically compared to those in group A (P < 0.006). It was accompanied by higher serum total and direct bilirubin levels, persistently elevated GGT values, the presence of itching, and more intense dark urine. This so-called cholestasis syndrome was observed mainly in children over 6 years of age, and in 5 of them (45.45%), it was combined with AH, and in 8 (72.72%) and with a thickened gallbladder wall (mine). In 6 of these patients, Cynarae folium extractum was added to the therapy, and in one 14-year-old, ursodeoxyfolic acid was added for 30 days. In 15 patients, we observed an atypical course with the classic signs of AAS-thickened gallbladder wall > 3 mm (with or without pericholecystic edema or wall dissection), fever, abdominal pain in the lower right quadrant-spontaneous and/or palpation, often combined with vomiting. The US of the bile gum found an average thickness of 5.4 mm in children. Of these, 9 (60%) children had leukocytosis, and 7 (50%) had higher-than-average total and direct serum bilirubin levels, with an average duration of 8.2 days (P < 0.02) compared to category A. In these children, intravenous Ampicillin was added to the therapy for 10 days.
We did not observe statistically significant differences in other clinical and laboratory indicators compared to category A. Ascites in the abdominal cavity were observed in 2 (26.66%) of the children in category B, and both patients were diagnosed by US. In patients with ascites, serum albumin levels were below normal, and the difference was statistically significant compared to the rest of the category B children in whom this indicator was examined. The complication was overcome solely through dietary salt restriction, without the need for diuretics or parenteral albumin preparations.
We found asymptomatic thrombocytopenia in one child with 90.000103/mm3. Other causes of the condition (malaria, medication, underlying disease) were ruled out. Bone marrow puncture was not necessary, as thrombocytopenia subsided spontaneously in 9 days. Again, in one child, during a relapse, readmitted due to deterioration of the condition 2 weeks after complete cure, ALF developed. The patient had a 4-year-old girl with low-grade encephalopathy, low albumin values and pericholecystic ascites. Serum transaminases did not show statistically significant improvement; however, a very high total bilirubin level (15.4 mg/dL) was observed, as well as prolonged icterus (14.6 days). The child finally recovered after the repeated 21-day hospital stay with standard symptomatic and pathogenetic therapy. Normalization of total bilirubin (P < 0.008) took longer in children with atypical manifestations, and their average hospital stay was 5.8 days longer.
We found no statistically significant differences in the values of hepatic transaminases (AST, ALT). In children with AH, white blood cell (WBC) also normalized more slowly, and two had short-term hemoglobin values below normal. Among our 30 cases with atypical manifestations, we had an average hospital stay of 8.64 days, and in 3 of the children, it exceeded 20 days. As shown in Table 2, the difference is statistically significant.
Вe identified only 2 patients with mild pleural effusion detected during chest imaging, likely secondary to systemic inflammation, and illustrating the extra-abdominal manifestations in our cohort. Other described atypical manifestations (pancreatitis, autoimmune disorders, neurological manifestations, AKI) etc. have not been reported in our atypical cases.
According to the available literature sources, this is the first comprehensive study of the atypical course of HA in childhood in Bulgaria. We demonstrated that most cases (30/115, 26.08%) can present with atypical symptoms, necessitating longer hospitalization. Most cases occur in individuals with higher serum bilirubin levels, GGT levels, and a prolonged icteric phase, as seen in some individuals with higher WBC counts and lower albumin values.
The proportion of cases with an atypical course inpatients with HA (26.08%) is relatively high compared to the world literature, where the percentage fluctuates between 15% and 20%[7,8,11]. This may be because, in our work, we administer only to the child population and due to the randomness of the sample. Most authors assume that the frequency and severity of an atypical course depend on the immunological state of the body, and children have been shown to have a more reactive immune system[8,11,13]. Of course, there is still far from consensus on the causes of such abnormal forms of HA in any age group[1,2,7,11].
In terms of clinical findings, in category B children, we observed an extended mean duration of jaundice, as well as an increased average hospital stay, a high incidence of pruritus, and, to some extent, headache. From laboratory tests, total and direct bilirubin, GGT, and albumin (where applicable) also showed statistically significant abnormalities in many cases. Similar manifestations are described by other world authors[8,11]. Itis clear from our study that high serum bilirubin levels and their prolonged retention, as well as the thickening of the gallbladder wall, can be perceived as prognostic signs of a possible transition into an atypical clinical course for the patient with HA[7,14].
Atypical manifestations are more common in children approaching or beginning the first decade of life (8.28 years) compared to 4.15 years for children in category A[2,8,13]. We would rather not draw definite conclusions about gender. The relatively high number of children with AA does not surprise us, as we have conducted a similar study in the pediatric population, and some literature data confirm that the condition among children with HA is not at all as rare as previously thought[14]. This suggests that itis prudent to consider such a complication at the onset of the disease and to perform more frequent abdominal ultrasonography for specific indications. Notinfrequently, such a condition, in addition to exacerbating the course of infection, can also necessitate surgical intervention in advanced cases[7,14].
We observed ascites in 2/30 (26.66%) of the children in the abnormal group, which corresponded with the observations of other authors (3%-30%)[7,11]. The reason for the US-only ascites is likely due to the lower serum albumin level, especially since a poor diet exacerbated the condition. Regarding the two children with asymptomatic thrombocytopenia, we believe that it was more of a transient immunological phenomenon[8]. We found only one child who developed ALF. Fortunately, despite the high total and direct bilirubin, leukocytosis, ascites and prolonged icterus syndrome, the child came out of the serious condition after standard therapy in the intensive infectious sector. According to the data of relatives, before the relapse, the child used unregulated herbal preparations, which could be the reason for the development of this rare complication of HA, especially in a child without underlying diseases[8,12].
In line with this, Cynarae folium extractum (artichoke leaf extract)[15] was administered as part of supportive hepatoprotective therapy, based on clinical judgment and institutional practice. Artichoke extract is known for its antioxidant, choleretic, and hepatoregenerative properties, and has been traditionally used in the management of liver dysfunction. Its inclusion in the therapeutic regimen reflects increasing interest in evidence-based phytotherapy as an adjunct to standard medical care in hepatic conditions[15,16]. Although not part of a universally standardized treatment protocol, its use was consistent with national phytotherapeutic guidelines and administered under physician supervision. No adverse effects were observed in the patients who received it.
In our cohort, a subset of children presented with atypical clinical courses of HA, including prolonged cholestasis, ascites, thrombocytopenia, and acute acalculous cholecystitis[17]. Although the precise mechanisms underlying these atypical presentations remain unclear, several potential risk factors may contribute to their development. Younger age, underlying nutritional deficiencies, or impaired immune responses may predispose certain children to more severe or atypical manifestations[18]. Lee et al[19] in their study of 770 HA patients performed a multivariate analysis, and showed that clinical parameters such as age, peak creatinine, bilirubin, ALT, baseline LDH, and total cholesterol were identified as independent predictors of severe acute HA, notably, the HA viral load showed a strong association with disease severity[20].
Additionally, while viral load was not assessed in this study, previous literature suggests that higher HAV viral loads may be associated with a more aggressive clinical course[19]. These variables merit further investigation in prospective studies with larger sample sizes and extended follow-up. Understanding the predictors of atypical disease courses could help identify at-risk children earlier and tailor their clinical management accordingly[20].
Our study has a few limitations. First, our study is a single-center retrospective analysis, and only symptomatic cases requiring hospitalization were included, where the hospital-based design limits generalizability to broader pediatric populations, especially asymptomatic or mild outpatient cases, which limits the external validity of the findings. We included only symptomatic hospitalized patients because it was not possible to work with symptomatic patients, as well as with outpatients. A limited number of patients from only one tertiary center in the country were examined. Second, we have used standard laboratory tests in all the described children, and only in some of those with an atypical course were the laboratory tests extended. Additionally, we have not done a preliminary study in the community on the number and type of atypical cases of HA, which can largely distort attempts to draw statistical conclusions. And lastly, ribonucleic acid of HAV (RNA reverse-transcription PCR) was not in demand in the laboratory, which prevented molecular epidemiological analysis from being conducted in our study.
The findings of our work show that childhood HA should not be overlooked. In most cases, it is benign, sometimes asymptomatic, and self-limiting.However, complications should be monitored, especially in the first ten yearsof life.
| 1. | Abutaleb A, Kottilil S. Hepatitis A: Epidemiology, Natural History, Unusual Clinical Manifestations, and Prevention. Gastroenterol Clin North Am. 2020;49:191-199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 2. | Jeong SH, Lee HS. Hepatitis A: clinical manifestations and management. Intervirology. 2010;53:15-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 126] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 3. | Aggarwal R, Goel A. Hepatitis A: epidemiology in resource-poor countries. Curr Opin Infect Dis. 2015;28:488-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (3)] |
| 4. | Epidemiological update: Hepatitis A outbreak in the EU/EEA mostly affecting men who have sex with men. ECDC. 2018. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/HEPA_AER_2018_Report.pdf. |
| 5. | Surveillance Atlas of Infectious Diseases. ECDC. Available from: https://atlas.ecdc.europa.eu/public/. |
| 6. | Kojaoghlanian T. Hepatitis A. Pediatr Rev. 2010;31:348-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 7. | Muñoz-Martínez SG, Díaz-Hernández HA, Suárez-Flores D, Sánchez-Ávila JF, Gamboa-Domínguez A, García-Juárez I, Torre A. Atypical manifestations of hepatitis A virus infection. Rev Gastroenterol Mex (Engl Ed). 2018;83:134-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 8. | Alam R, Karim ASMB, Mazumder MW, Das SR, Benzamin M, Sonia ZF, Rahman SMH. Atypical manifestations of acute viral hepatitis A in children in Bangladesh: Are these really uncommon? Indian J Gastroenterol. 2021;40:470-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (2)] |
| 9. | Konuş OL, Ozdemir A, Akkaya A, Erbaş G, Celik H, Işik S. Normal liver, spleen, and kidney dimensions in neonates, infants, and children: evaluation with sonography. AJR Am J Roentgenol. 1998;171:1693-1698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 195] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 10. | Dietrich CF, Augustiniene R, Batko T, Cantisani V, Cekuolis A, Deganello A, Dong Y, Franke D, Harkanyi Z, Humphries PD, Jędrzejczyk M, Jüngert J, Kinkel H, Koller O, Kosiak W, Kunze C, Ljutikov A, Madzik J, Mentzel HJ, Piskunowicz M, Rafailidis V, Schreiber-Dietrich D, Sellars ME, Stenzel M, Taut H, Yusuf GT, Sidhu PS. European Federation of Societies for Ultrasound in Medicine and Biology (EFSUMB): An Update on the Pediatric CEUS Registry on Behalf of the "EFSUMB Pediatric CEUS Registry Working Group". Ultraschall Med. 2021;42:270-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 11. | Jain J, Jain M. Atypical manifestations of viral hepatitis A and E. Trop Doct. 2013;43:17-18. [PubMed] [DOI] [Full Text] |
| 12. | Bucuvalas J, Yazigi N, Squires RH Jr. Acute liver failure in children. Clin Liver Dis. 2006;10:149-168, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 13. | Rawat A, Tiwari S, Saxena A. Atypical Presentation of Hepatitis A Infection in an Infant. Indian J Pediatr. 2024;91:207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 14. | Velev V, Popov M, Tomov L, Golemanov B. Involvement of the gallbladder in the course of viral hepatitis A in childhood. Trop Doct. 2019;49:271-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 15. | Cynarae folium - herbal medicinal product. EMA 2024 revision. Available from: https://www.ema.europa.eu/en/medicines/herbal/cynarae-folium. |
| 16. | Akçakaya A. Phytotherapy in Liver Diseases. Bezmialem Sci. 2024;12:491-497. [DOI] [Full Text] |
| 17. | Migueres M, Lhomme S, Izopet J. Hepatitis A: Epidemiology, High-Risk Groups, Prevention and Research on Antiviral Treatment. Viruses. 2021;13:1900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 147] [Cited by in RCA: 104] [Article Influence: 20.8] [Reference Citation Analysis (1)] |
| 18. | Kim JI, Kim YS, Jung YK, Kwon OS, Kim YS, Ku YS, Choi DJ, Kim JH. Factors influencing the severity of acute viral hepatitis A. Korean J Hepatol. 2010;16:295-300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 19. | Lee HW, Chang DY, Moon HJ, Chang HY, Shin EC, Lee JS, Kim KA, Kim HJ. Clinical Factors and Viral Load Influencing Severity of Acute Hepatitis A. PLoS One. 2015;10:e0130728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 20. | Gholizadeh O, Akbarzadeh S, Ghazanfari Hashemi M, Gholami M, Amini P, Yekanipour Z, Tabatabaie R, Yasamineh S, Hosseini P, Poortahmasebi V. Hepatitis A: Viral Structure, Classification, Life Cycle, Clinical Symptoms, Diagnosis Error, and Vaccination. Can J Infect Dis Med Microbiol. 2023;2023:4263309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (1)] |