Published online Mar 27, 2024. doi: 10.4254/wjh.v16.i3.452
Peer-review started: December 26, 2023
First decision: January 11, 2024
Revised: January 19, 2024
Accepted: February 28, 2024
Article in press: February 28, 2024
Published online: March 27, 2024
Processing time: 92 Days and 11.6 Hours
A new nomenclature consensus has emerged for liver diseases that were previously known as non-alcoholic fatty liver disease (NAFLD) and metabolic dysfunction-associated fatty liver disease (MAFLD). They are now defined as metabolic dysfunction-associated steatotic liver disease (MASLD), which includes cardiometabolic criteria in adults. This condition, extensively studied in obese or overweight patients, constitutes around 30% of the population, with a steady increase worldwide. Lean patients account for approximately 10%-15% of the MASLD population. However, the pathogenesis is complex and is not well understood.
To systematically review the literature on the diagnosis, pathogenesis, characteristics, and prognosis in lean MASLD patients and provide an interpretation of these new criteria.
We conducted a comprehensive database search on PubMed and Google Scholar between January 2012 and September 2023, specifically focusing on lean NAFLD, MAFLD, or MASLD patients. We include original articles with patients aged 18 years or older, with a lean body mass index categorized according to the World Health Organization criteria, using a cutoff of 25 kg/m2 for the general population and 23 kg/m2 for the Asian population.
We include 85 studies in our analysis. Our findings revealed that, for lean NAFLD patients, the prevalence rate varied widely, ranging from 3.8% to 34.1%. The precise pathogenesis mechanism remained elusive, with associations found in genetic variants, epigenetic modifications, and adaptative metabolic response. Common risk factors included metabolic syndrome, hypertension, and type 2 diabetes mellitus, but their prevalence varied based on the comparison group involving lean patients. Regarding non-invasive tools, Fibrosis-4 index outperformed the NAFLD fibrosis score in lean patients. Lifestyle modifications aided in reducing hepatic steatosis and improving cardiometabolic profiles, with some medications showing efficacy to a lesser extent. However, lean NAFLD patients exhibited a worse prognosis compared to the obese or overweight counterpart.
MASLD is a complex disease comprising epigenetic, genetic, and metabolic factors in its pathogenesis. Results vary across populations, gender, and age. Limited data exists on clinical practice guidelines for lean patients. Future studies employing this new nomenclature can contribute to standardizing and generalizing results among lean patients with steatotic liver disease.
Core Tip: Steatotic liver disease, extensively studied in overweight/obese patients, poses a unique challenge in lean individuals due to limited data on its pathogenesis, diagnosis, management, and risk factors. The lack of consensus in nomenclature impedes the comprehension and application of findings. To address this gap, we conducted a systematic review focusing on lean individuals with steatotic liver disease. This review interprets the new approach, introducing the term metabolic dysfunction-associated steatotic liver disease in alignment with current literature. We aim to enhance the understanding of steatotic liver disease in lean populations, contributing to a precise approach in research and clinical settings.
- Citation: Sato-Espinoza K, Chotiprasidhi P, Huaman MR, Díaz-Ferrer J. Update in lean metabolic dysfunction-associated steatotic liver disease. World J Hepatol 2024; 16(3): 452-464
- URL: https://www.wjgnet.com/1948-5182/full/v16/i3/452.htm
- DOI: https://dx.doi.org/10.4254/wjh.v16.i3.452
In 1980, Ludwig, Viggiano, McGill, and Oh introduced the term non-alcoholic fatty liver disease (NAFLD), defining the disease as the presence of hepatic fat in the absence of significant alcohol intake. It was characterized as hepatic steatosis observed through imaging or histology, excluding other causes of chronic liver disease and steatosis, such as substantial alcohol consumption, prolonged use of steatogenic medication, or hereditary monogenic disorders[1]. By utilizing this exclusionary criterion, the differential diagnosis of NAFLD was formed. In 2020, the concept of metabolic dysfunction-associated fatty liver disease (MAFLD) emerged, encompassing individuals previously excluded due to alcohol consumption or other liver diseases[2]. This represented a shift towards a "positive" diagnosis, moving away from an exclusory approach. However, even with this new terminology, patient stigmatization persisted due to the continued use of the term "fatty." Consequently, a collaborative effort involving the following groups: American Association for the Study of Liver Disease, European Association for the Study of the Liver, and Latin American Association for the Study of the Liver, utilizing the Delphi method, led to the development of a novel nomenclature metabolic dysfunction-associated steatotic liver disease (MASLD)[3]. The recent consensus reclassified NAFLD and MAFLD[4,5] to MASLD[3]. To meet the new MASLD criteria, individuals must exhibit at least 1 of 5 cardiometabolic risk factors linked to insulin resistance (IR). MASLD constitutes approximately 30% of the global population, and its prevalence is steadily increasing worldwide[6]. Despite this condition being extensively researched in overweight and obese individuals, 10%-15% of MASLD patients will exhibit normal weight and are classified as either lean or non-obese[7]. The categorization depends on ethnicity; the World Health Organization (WHO) categorizes a normal body mass index (BMI) for the general population with a cutoff of 25 kg/m2 and 23 kg/m2 for the Asian population[8]. Most studies have predominantly focused on BMI when investigating patients with lean MASLD. However, BMI has been proven to be an imperfect marker of adiposity[9-13]. Vilarinho et al[14] have proposed a classification system for patients with lean MASLD, distinguishing two phenotypes based on epidemiological characteristics, natural history, and prognosis. Type 1 includes individuals with visceral adiposity and insulin resistance. While type 2 comprises of those with hepatic steatosis resulting from monogenic diseases, this requires a nuanced understanding of the pathophysiology.
The pathophysiology of MASLD is intricate and diverse. The clinical spectrum of this disease ranges from simple steatosis to cirrhosis and is influenced by diverse factors, including the overconsumption of carbohydrates and dietary sugars such as fructose, sucrose, and glucose[15]. Dysbiosis, bacterial translocation, and pro-inflammatory factors in the liver also contribute to its complexity[16]. It is proposed that the disease phenotype arises from intricate interactions between genetic and environmental factors[17]. Despite the various potential mechanism proposed, the literature supports that IR and lipotoxicity play a key role in the pathogenesis[18]. This interplay results in a chronic elevation of plasma levels of non-esterified fatty acids, which are ectopically deposited in the liver, promoting the development of steatosis. Additionally, triglycerides (TG) within hepatocytes further increase the accumulation of toxic lipids, such as ceramides and diacylglycerols, intensifying IR and activating inflammatory pathways. Furthermore, it has been reported that lean MASLD patients experience increased concentrations of serum bile acids and elevated farnesoid X receptor (FXR) activity as an initial metabolic response[16-19].
Genes have been identified as modulators of insulin sensitivity and regulators of the intracellular flow of fatty acids, TG, oxidative stress, endotoxin response, cytokine activity, and the development of fibrosis[18]. The most studied single nucleotide polymorphisms (SNPs) associated with steatosis across diverse ethnicities are rs58542926 in the TM6SF2 gene (transmembrane 6 superfamily member 2)[20] and rs738409 in the PNPLA3 gene (patatin-like phospholipase domain-containing protein 3)[21]. The I148M polymorphism of PNPLA3 disrupts triglyceride lipolysis in lipid droplets[22]. Polymorphism in TM6SF2 plays a pivotal role in hepatic and cholesterol metabolism[20]. Additionally, MBOAT7 influ
Regarding the diagnosis of steatotic liver disease in lean patients, it is typically conducted through[18,24] imaging modalities such as abdominal ultrasound (US)[25,26], computed tomography (CT)[27,28], or magnetic resonance imaging (MRI)[29]. Additionally, FibroScan, assessing the controlled attenuation parameter (CAP)[30-32] and liver stiffness measurement (LSM)[31,33], is employed. However, liver biopsy is usually reserved for patients with an unclear diagnosis. Conversely, non-invasive scores are also utilized for diagnosis, which will be discussed later in this review.
The development of the new MASLD nomenclature consensus has been proven helpful for accurately classifying patients with liver steatosis, allowing individuals previously classified as "lean NAFLD" to be categorized as lean MASLD, facilitating uniform studies in the future, particularly for those presenting with cardiometabolic risk[34,35]. These new approaches broaden the focus regarding the metabolic pathogenesis of the disease. However, individuals not meeting these criteria and have no known cause of liver disease have been classified as having cryptogenic steatotic liver disease[3]. This distinction is significant because some patients previously labeled as NAFLD are now reclassified as cryptogenic steatotic liver disease. Discussing this reclassification is important because this new approach does not imply that other causes of steatosis should not be considered, and it also allows for a more in-depth characterization of fibrosis severity using a non-invasive test. Due to the homogenization of the concept of steatotic liver disease, this has been a significant step forward in understanding and addressing this complex disease. As establishing a consensus on how to categorize these patients is essential for future studies, ensuring that results are comparable across different research endeavors.
Considering the significant implication of this complex disease, we intended to conduct a systematic review of the literature pertaining to the diagnosis, pathogenesis, characteristics, and complications associated with lean MASLD patients. Additionally, our goal is to provide an interpretation of this new criteria.
We conducted a database search on PubMed and Google Scholar, selecting papers published between January 2012 and September 2023 in the English language. The last access to PubMed and Google Scholar occurred on 25 September 2023. The keywords and terms utilized in our search were as follows: (1) NAFLD or non-alcoholic liver disease; (2) MASLD or metabolic dysfunction association steatotic liver disease; (3) guidelines; (4) management; (5) characteristics; and (6) lean. The specific search terms included "non-alcoholic fatty liver disease"[MeSH Terms] OR nafld [All Fields], "guide
We included original articles that featured patients aged 18 years or older, with BMI categorized by the WHO for both the general and Asian populations. In the general population, BMI was described as normal (18.5-24.9 kg/m2), overweight (25-29.9 kg/m2), and obese (> 30 kg/m2). In the Asian population, BMI was described as normal (18.5-22.9 kg/m2), overweight (23-24.9 kg/m2), and obese (> 25 kg/m2). In this review, normal BMI is referred to as lean, non-obese, or normal weight. We included studies that diagnose steatosis liver disease using abdominal US, abdominal CT, or MRI, in conjunction with FibroScan, which incorporates CAP and/or LSM, as well as histological diagnosis via biopsy. Diagnosis may also involve clinically identifying steatosis liver disease based on elevated liver enzymes, while ruling out other liver diseases.
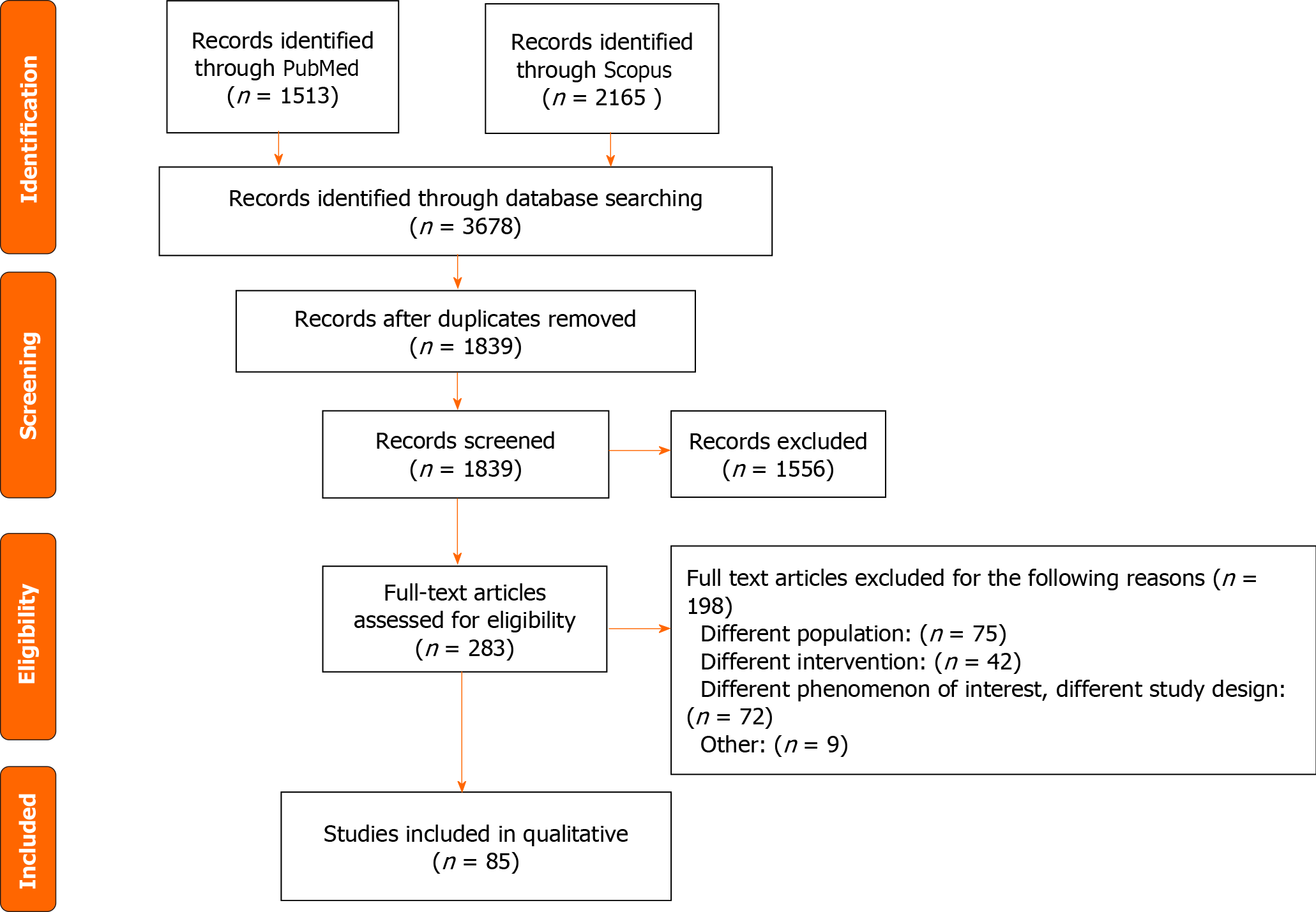
We excluded systematic reviews, review articles, case reports, poster presentations, conference abstracts, editorials, letters to the editor, studies involving patients under 18 years old, studies which utilizes animals, and studies categorizing BMI differently than the WHO. After removing duplicates and applying our inclusion and exclusion criteria, a total of 85 papers were identified. Refer to Figure 1 for more details.
Only one expert review on clinical practice updates for lean MASLD patients was found in the literature[24]. The review offered practical advice for physicians. The evaluation of MASLD patients should include routine assessments for hypertension (HTN), type 2 diabetes mellitus (T2DM), dyslipidemia, and a comprehensive alcohol consumption history. Regarding screening lean patients, only patients older than 40 years old with T2DM require recommended evaluation. It is essential to investigate and rule out alternative causes of liver steatosis, starting with non-invasive methods such as serum scores or imaging; liver biopsy should be reserved for undetermined diagnosis. NAFLD fibrosis score (NFS) and fibrosis-4 score (FIB-4) were the two non-invasive scores recommended. The recommended imaging modalities were transient elastography (FibroScan) and magnetic resonance elastography. While no specific treatment exists for lean patients, it is recommended that lifestyle modifications advocating a modest weight loss of 3%-5% (less than in overweight or obese patients) be pursued. Surveillance for liver cancer is crucial, and it involves employing abdominal ultrasound, with or without alpha-fetoprotein, in patients with cirrhosis.
Genetic variants and epigenetic modifications have been correlated in lean NAFLD patients. However, the precise mechanisms have yet to be fully elucidated, and in some cases, have produced contradictory results. Zeng et al[36] described that in the Chinese population, there was no significant difference in SNPs in the SIRT1, APOC3, PNPLA3, AGTR1, and PPARGC1A genes between lean patients with and without NAFLD. They concluded that metabolic factors played a vital role in the occurrence and progression of NAFLD rather than genetic factors.
On the other hand, Wei et al[37] found that a SNP in PNPLA3 (rs738409) had a higher prevalence in non-obese patients compared to obese patients with NAFLD. Carrying the GG allele in PNPLA3 (rs738409) increases the risk of NAFLD in the general population, especially in patients without metabolic syndrome (MetS). This SNP appeared to be independent of dietary factors or metabolic conditions[38]. Despite these contradictory results, the GG variant of patatin-like phospholipase domain 3 (PNPLA3), encodes adiponutrin and plays a crucial role in lipid metabolism. It has been identified as an independent variable, and it has been associated with a higher risk of NAFLD and significant fibrosis in lean patients[37-39].
Alharthi et al[16] described an alteration in adaptive metabolic response characterized by elevated concentrations of serum bile acids and increased activity of the FXR in lean NAFLD patients. Models of metabolic maladaptation loss have been proposed for these patients[16,19]. The Western diet may alter intestinal permeability, increase exposure to bacterial products, and lipopolysaccharides. In lean patients with NAFLD, this could lead to higher endotoxemia, increased expression of macrophage TLR4, and higher production of inflammatory cytokines compared to healthy thin individuals.
The prevalence of lean NAFLD exhibits a wide range, varying from 3.8% to 34.1%[7,40-56]. Refer to Table 1, for more details.
| Ref. | Population | Prevalence | Characteristics | Cardiometabolic risk | Laboratory values | Anthropometric values |
| Younossi et al[7], 2012 | Compared lean with overweight/obese NAFLD patients (n = 11613) | 18% | < 40 yr, female | ↓ T2DM, IR, HTN, Hypercholesterolemia | ↓ AST, ALT, platelets | Not reported |
| Wei et al[37], 2015 | Compared lean with overweight/obese NAFLD patients (n = 911) | 19.3% | < 50 years: male, > 50 years: No difference between sexes | ↓ T2DM, HTN, MetS and liver stiffness | ↓ ALT, HOMA-IR, ↑ HDL | ↓ WC, WHR |
| Fracanzani et al[66], 2017 | Compare lean with overweight/obese NAFLD patients (n = 669) | 21.3% | Not reported | ↓ T2DM, MetS, HTN | ↓ HOMA-IR, ↑ HDL, platelet | ↓ WC |
| Golabi et al[65], 2019 | Compare lean with and without NAFLD patients (n = 5375) | Not reported | Not reported | ↑ Risk cardiovascular and all-cause of mortality | Not reported | Not reported |
| Shao et al[63], 2020 | Compare lean with obese NAFLD patients (n = 543) | Not reported | No difference between sexes or age | ↓ BP | ↓ AST, ALT, LDL, total cholesterol, FBG, HOMA-IR, ↑ HDL | ↓ BMI, WC, WHR |
| Aneni et al[40], 2020 | Compared lean with and without NAFLD patients (n = 9137) | 3.8% | > 40 yr, male | ↑ Risk of AD, BP | ↑ FBG, total cholesterol, LDL, TG, AST, ALTl; Low HDL | ↑ WC |
| Rahman et al[41], 2020 | Compared lean with and without NAFLD patients (n = 1305) | 4.4% | > 40 yr, male | ↑ MetS, T2DM | ↑ TG, Total cholesterol, FBG | ↑ Abdominal obesity, BMI |
| Semmler et al[46], 2021 | Compared lean with and without NAFLD patients (n = 3043) | 6.7% | > 40 yr, male | ↑ Dyslipidemia, IR, T2DM, MetS, cardiovascular risk | Not reported | ↑ WC |
| Weinberg et al[60], 2021 | Compared lean with overweight/obese NAFLD patients (n = 3386) | Not reported | > 60 yr No difference between sexes | ↓ Cirrhosis, CVD, HTN, T2DM, dyslipidemia | ↓ AST, ALT; ↑ Albumin | Not reported |
| Aneni et al[58], 2022 | Compared lean with overweight/obese NAFLD patients (n = 6513) | Not reported | < 45 yr, female | ↓ HTN, T2DM, hyperlipidemia, MetS, AD, ↑ risk of all-cause of mortality | ↓ FBG, total cholesterol, LDL, TG, AST, ALT; ↑ HDL | ↓ WC |
| Razouki et al[45], 2022 | Describe lean NAFLD (n = 1049) | 5.8% | > 60 yr, male, Asian American | ↑ MetS, Inadequate physical activity | ↑ FBG, TG | |
| Zhang et al[56], 2022 | Compare lean with obese NAFLD patients (n = 2708) | 34.1% | > 40 yr | ↓ BP | ↓ HOMA-IR; ↑ HDL | ↓ WC |
| Ahmed et al[50], 2022 | Compared lean with overweight/obese NAFLD patients (n = 4834) | 8.6% | Females, Asian and African American | ↓ HTN, T2DM, hyperlipidemia | Not reported | Not reported |
| Nabi et al[42], 2023 | Compared lean with non-lean NAFLD patients (n = 25753) | 5.3% | < 40 yr, no difference between sexes | ↑ Risk of CVD, liver-related events, CKD and all-cause of death | ↑ AST | ↓ WC |
| De et al[52], 2023 | Compared lean with non-lean NAFLD patients (n = 1040) | 14.3% | No difference between sexes and age | ↓ HTN, MetS | No significant difference | ↓ Central obesity |
| Wijarnpreecha et al[62], 2023 | Compared lean with non-lean NAFLD patients (n = 18594) | 11.4% | Female, no difference between age | ↓ MetS, HTN, T2DM, CKD, cerebrovascular accident | ↓ AST, ALT, total cholesterol, LDL and TG, ↑ HDL | Not reported |
| Biswas et al[59], 2023 | Compared lean with overweight/obese NAFLD patients (n = 1051) | 12.1% | < 40 yr, males | ↓ HTN | Not difference in ALT and AST | ↓ WC |
| Kawanaka et al[57], 2023 | Compared lean with non-lean NAFLD patients (n = 782) | 11% | > 50 yr, no difference between sexes | ↓ HTN | ↓ AST, ALT, TG, HOMA-IR, HbA1C | Not reported |
| Ishido et al[61], 2023 | Compared lean with non-lean NAFLD patients (n = 581) | Not reported | Males, no difference between age | ↓ HTN, T2DM | ↓ AST, ALT, TG; ↑ HDL | ↓ BMI |
Many studies have indicated that lean NAFLD occurs to people that are older than 40 years old[40,41,46,47,53,55-57]. However, conflicting findings exist, with some studies suggesting that patients are younger than 40 years old[7,42,58,59]. While other studies report patients being older than 60 years old[45,60]. One study demonstrated, by stratifying the prevalence of lean NAFLD by age and sex, that males under 50 years old have an increased likelihood of developing the lean NAFLD phenotype; however, beyond 50 years old, no significant differences between the sexes were observed[37].
When examining the sexes separately, some studies reported a high prevalence of lean NAFLD in males[40,41,45,46,59,61], while others indicated a higher prevalence in females[7,50,58,62]. Nevertheless, there are studies reporting no significant differences in prevalence between females and males[42,51,52,57,60,63].
These variations highlight the heterogeneity of lean NAFLD prevalence in different cohorts and across distinct populations.
Studies have compared lean patients with and without NAFLD. These studies have demonstrated that lean NAFLD patients are at a higher risk of atherogenic dyslipidemia[40,64], MetS, T2DM[41,46], dyslipidemia, and cardiovascular complications[46]. Additionally, these patients manifest elevated cardiovascular and all-cause mortality rates[65]. When laboratory values were compared, this revealed elevated levels of TG, total cholesterol, and fasting blood glucose (FBG) for patients with lean NAFLD[41]. Regarding anthropometric measurements, the studies showed higher waist circumference (WC)[40,41,44,46] and BMI[41] in lean NAFLD patients compared to those without NAFLD.
When comparing lean patients with NAFLD and overweight/obese patients with NAFLD, studies reported that lean NAFLD patients have a lower prevalence of T2DM[7,37,50,58,60-62,66], dyslipidemia[7,50,58,60] , HTN[7,49,50,52,56-58,60,63,66,67], MetS[49,52,62,66], cardiovascular disease[60], and cirrhosis[60,62]. Laboratory values were compared, indicating lower levels of aspartate aminotransferase (AST)[7,53,57,59,62,63,67], alanine aminotransferase (ALT)[7,53,57,59,62,63,67], platelet count[7,66], FBG[53,58,63], TG[53,57,58,61,62], homeostatic model assessment for insulin resistance (HOMA-IR)[57,63,68], and total cholesterol[57,58,61-63], as well as higher levels of high density lipoproteins (HDL)[56,61-63,66,69]. Regarding anthropometric measurements, the studies reported lower WC[52,56,63,66], BMI[63,70], and waist-to-hip ratio (WHR)[63,70] in lean NAFLD compared to overweight/obese counterparts.
In studies where BMI was compared, lean NAFLD patients exhibited a lower prevalence of comorbidities and a more favorable laboratory profile when compared to overweight or obese patients with NAFLD. Conversely, in studies comparing individuals with and without NAFLD, lean NAFLD patients displayed a worse profile with the highest rates of comorbidities and adverse laboratory values compared to healthy lean individuals without NAFLD. This consideration holds significant importance in the interpretation and application of risk factor concepts in clinical practice. These heterogeneous results underscore the need for regular monitoring in patients who are lean and have NAFLD, given the elevated risk of metabolic diseases compared to those who are lean and do not have NAFLD.
Patients with NAFLD are at risk of progressing to non-alcoholic steatohepatitis (NASH) and developing other complications[71]. We will now present literature that has evaluated and characterized NASH patients, refer to Table 2 for more details. The most used score in studies diagnosing NASH in patients is the NAFLD Activity Score (NAS), which has been proposed and validated by the NASH Clinical Research Network[72]. This score assesses three characteristics in liver histology: Steatosis Grade, Lobular Inflammation, and Hepatocellular Ballooning. The score ranges from 0 to 8, with a score < 3 correlating with not-NASH, and a score > 5 correlating with a diagnosis of NASH.
| Ref. | Population | Results |
| Leung et al[73], 2017 | Histology (n = 307), lean vs obese NAFLD patients | Low NAS (steatosis and hepatocyte ballooning), low stiffness |
| Denkmayr et al[76], 2018 | Histology (n = 466), lean vs overweight/obese NAFLD patients | High lobular inflammation and hepatocellular ballooning |
| Li et al[82], 2019 | Scores (n = 898), lean vs overweight/obese NAFLD patients | WHR and FLI accurate in lean and obese patients, ZJU and HSI accurate in lean patients |
| Kim et al[75], 2019 | Histology (n = 542), lean vs obese NAFLD patients | Low grade steatosis and NAS, high stage of fibrosis |
| Fu et al[81], 2020 | Scores (n = 709), non-obese vs obese NAFLD patients | FIB-4, NFS, APRI, BARD score and AST-to-ALT ratio had similar accurate in obese and non-obese patients |
| Eren et al[79], 2022 | Scores (n = 560), lean vs overweight vs severely and morbid obese NAFLD patients | FIB-4 and NFS cannot discriminate advance fibrosis in lean patients |
| Park et al[80], 2023 | Scores (n = 1501), lean vs non-lean NAFLD patients | FIB-4 and NFS accurate in identify advance fibrosis in lean and non-lean NAFLD patients |
| Iwaki et al[74], 2022 | Histology (n = 223), lean vs obese NAFLD patients | Low grade lobular inflammation, steatosis |
| Rastogi et al[77], 2022 | Histology (n = 1273), lean vs overweight/obese NAFLD patients | High hepatocyte ballooning, early-stage fibrosis |
Leung et al[73] reported that non-obese patients with NASH exhibited lower NAS due to reduced steatosis and hepatocyte ballooning, along with lower liver stiffness. Furthermore, Iwaki et al[74] observed a low grade of lobular inflammation and fibrosis stage, with no significant differences in steatosis, ballooning, and overall NAS in non-obese compared to obese patients. Additionally, Kim et al[75] found that lean patients displayed a low grade of steatosis and overall NAS, but a higher stage of fibrosis compared to their obese counterparts with NAFLD.
On the contrary, Denkmayr et al[76] identified a higher proportion of lobular inflammation and hepatocellular ballooning, with a notable prevalence of cirrhosis in lean patients. However, the degree of steatosis was similar across the groups. Also, Rastogi et al[77] found a high proportion of hepatocyte ballooning but a high prevalence in none/early-stage fibrosis.
The results of histology in different studies are inconclusive. They indicate that histological characteristics could vary, showing either worse or better outcomes in lean vs overweight or obese individuals. However, this emphasizes the importance of careful evaluation for lean patients, similar to the rest of the population. These contradictory results may be influenced by the different types of patients undergoing liver biopsy. Leung, Kim, and Denkymar assessed histology in the following types of patients: those exhibiting abnormal liver enzyme levels, those with suspected NAFLD, and those with a confirmed diagnosis of NAFLD through non-invasive tools. In contrast, Iwaki examined the histology in a tertiary center where referrals were received, particularly for patients with more severe liver conditions. Moreover, the differen
In the context of interpreting non-invasive tools in lean patients with NAFLD or NASH, a critical consideration is the selection of the most suitable scoring system or algorithm for clinical application. We will now present literature that has evaluated accuracy of those scores, refer to Table 2 for more details.
The accuracy of FIB-4 and NFS was compared in patients who underwent liver biopsy[78]. FIB-4 assessed age, levels of AST, ALT, and platelets, while NFS considered age, BMI, impaired fasting glucose or diabetes, levels of AST, ALT, platelets, and albumin. In a study by Eren et al[79], it was observed that both FIB-4 and NFS were ineffective in discriminating against advanced fibrosis in both lean and morbidly obese patients. Contrastingly, a study by Park et al[80] revealed that the diagnostic performance of FIB-4 and NFS in identifying advanced hepatic fibrosis was comparable, irrespective of BMI. The sensitivity of NFS in lean patients was inferior to that of FIB-4. In addition to comparing FIB-4 and NFS, Fu et al[81] included AST-to-platelet, BARD score, and the AST-to-ALT ratio in the comparison. They found that all non-invasive scores performed equally for both obese and non-obese patients. The negative predictive value (NPV) was higher in non-obese patients due to the lower prevalence of advanced fibrosis. Moreover, Li et al[82] compared 8 NAFLD-related algorithms, finding that WHR and Fatty Liver Index exhibited diagnostic accuracy for NAFLD in both lean and overweight/obese populations, but Zhejiang University Index and Hepatic Steatosis Index demonstrated exclusively positive associations in lean patients.
In summary, the review of accuracy and performance across different non-invasive tools in patients with NAFLD revealed that FIB-4 outperformed NFS in this specific population. However, it is crucial to note that this result was observed in only one study. Nonetheless, this finding does hold significance, considering that the only clinical guideline for lean MASLD recommends FIB4 and NFS equally. Thus, it is imperative that new studies compare these non-invasive tools in patients with MASLD due to the updated guidelines.
Clinical trials were conducted to explore potential treatments for NAFLD. In the literature reviewed, we found two types of treatment: pharmacological and non-pharmacological.
Pharmacological: In a one-year follow-up study involving 8 lean patients with NAFLD, half received ursodeoxycholic acid, and the other half received 10 mg of the Niemann-Pick C1 Like 1 (NPC1L1) inhibitor, ezetimibe. The findings revealed that patients treated with ezetimibe for 12 months experienced decreased levels of AST and low-density lipoprotein, but no significant changes were observed in HDL, TG, HOMA-IR, or liver fat attenuation in abdominal US[83]. In another study involving 50 patients, 25 received a synbiotic capsule, and 25 received a placebo capsule. Both groups received advice on maintaining a balanced diet and engaging in physical activity. After 28 wk of treatment and follow-up, both groups exhibited reduced hepatic steatosis and inflammatory markers, with the synbiotic group having a higher mean reduction in FBS, TG, and AST[84].
Pemafibrate, a selective peroxisome proliferator-activated receptor-αmodulator, dosed at 0.1 mg twice daily was studied. The first study by Shinozaki et al[85] treated 71 patients for 6 months, finding that lean patients experienced a greater reduction in ALT and serum mac-2 binding protein glycosylation isomer than obese patients. The second study by Suzuki et al[86] treated 38 patients for 12 months and found a strong association in the decrease of ALT, AST, hepatic steatosis, and fibrosis in both lean and obese patients. Canagliflozin at a dosage of 100 mg once daily was evaluated in 20 patients with T2DM and NAFLD, but due to only one patient being lean, the results were inconclusive in this population[87].
Various pharmacological treatments and interventions have been investigated in patients with lean MASLD, demonstrating some degree of efficacy in improving the metabolic profile or reducing hepatic steatosis. However, longitudinal clinical trials with large study populations are still warranted to identify a promising drug for treating both lean MASLD and MASH. On the other hand, the literature supports that lifestyle modification is an effective therapy in lean patients with MASLD, similarly to overweight/obese patients.
Non-pharmacological: Lifestyle changes such as exercise and diet modification were evaluated in lean patients with NAFLD. Jin et al[88] followed patients for 14 years and found a reduction in hepatic steatosis, total cholesterol levels, and body weight. Wong et al[89] followed patients for 12 months and found that 50% of non-obese patients achieved NAFLD remission with a 3%-5% weight reduction, which was maintained over 6 years of follow-up. However, 50% of the obese group achieved remission with a higher percentage of weight loss (7%-10%). Hamurcu et al[90] and Sinn et al[91] found a decrease in body weight and hepatic steatosis, as well as improvement in anthropometric parameters in both lean and obese patients.
A retrospective study compared post-transplant outcomes in lean and obese patients with NASH from the United Network for Organ Sharing (UNOS)[92]. The study concluded that lean individuals experienced lower survival rates and graft survival at 10 years follow up compared to their obese counterparts. Although no distinguishable trends in the cause of death based on BMI were identified, early multiorgan failure was more prevalent in lean patients[92]. A recent retrospective study including NAFLD patients of the UNOS, found that patients with normal weight and who maintained a stable weight during the wait period for a liver transplant had a worse survival rate than patients with stable obesity during this period at 3 and 5 years. Also, patients with stable normal weight compared to stable obese, had high risk of all-cause mortality and graft failure[93].
Overall, the findings of these studies reveal a poorer survival rate and graft failure in lean patients compared to their overweight/obese counterparts. However, this may have been influenced by the baseline conditions of these individuals. For example, conditions such as sarcopenia, which demonstrated a strong correlation in lean patients[9-12], were not assessed in these studies due to the exclusive consideration of BMI rather than skeletal muscle mass. Sarcopenia could serve as a potential contributor to the worse prognosis in lean patients. Another factor highlighted in the study is that lean patients exhibited a higher rate of ascites and worse functional status, necessitating total assistance. These factors could potentially explain the heightened risk of complications during and post liver transplant. While these variables could explain the worse outcomes in lean patients, there remains a gap in knowledge concerning the exact reasons underlying the adverse outcomes. Further research is needed to elucidate the specific mechanisms and factors that contribute to the observed disparities in transplantation outcomes between lean and overweight/obese patients.
MASLD is a complex disease that comprised of epigenetic, genetic, and metabolic factors in its pathogenesis. The prevalence varies among populations, ranging from approximately 4% to 34%. The current literature reveals disparities in sex and age, with older male patients being the most at-risk group. Furthermore, when metabolic conditions were examined in lean patients with NAFLD vs without NAFLD, lean patients with NAFLD were associated with a higher prevalence of metabolic diseases and a worse metabolic profile. However, when BMI was compared among NAFLD patients, lean patients showed a lower prevalence of metabolic disease, a better metabolic profile, but in some cases, worse histologic results with advanced fibrosis. In evaluating the accuracy and performance of non-invasive tools for diagnosing steatotic liver disease in this population, FIB-4 appears to be the most ideal score to use. Regarding prognosis and outcomes, lean patients with NAFLD have a better metabolic profile and clinical characteristics than overweight/obese patients. However, lean NAFLD patients experience a higher mortality rate, primarily due to cardiovascular disease or all-cause mortality, and faster progression to advanced liver disease. It is important to note that metabolic diseases were a significant variable in past studies of NAFLD patients, indicating that the new concept of MASLD that includes cardiometabolic risk criteria provides a more accurate diagnosis for patients with liver steatosis. Future studies utilizing this new nomenclature can contribute to standardizing and generalizing study results among lean patients with steatotic liver diseases.
Metabolic dysfunction-associated steatotic liver disease (MASLD) is the new nomenclature of non-alcoholic fatty liver disease (NAFLD) and metabolic dysfunction-associated fatty liver disease (MAFLD). It is a complex condition, and its mechanism is poorly understood. There are several studies involving overweight/obese patients but there is very limited literature available regarding lean patients.
Only one clinical guideline is available for physicians to diagnosis and manage lean patients with MASLD. However, the pathogenesis, accurate treatment, risk factor and outcomes remain unknown.
The aim of this systematic review is to report literature of diagnosis, pathogenesis, characteristics, and prognosis in lean MASLD patients in diverse populations, and provide an interpretation of the new MASLD criteria.
A search on two large databases was conducted, PubMed and Google Scholar, selecting original articles published between January 2012 and September 2023 specifically focusing on lean NAFLD, MAFLD, or MASLD patients.
85 articles met the eligibility criteria and underwent further analysis. The prevalence of lean MASLD among diverse populations ranges from 4% to 34%. The pathogenesis of lean MASLD involves genetic, epigenetic, and metabolic factors; however, the mechanism remains elusive. Although adequate treatment remains challenging to identify, lifestyle modifications have proven effective in reducing hepatic steatosis and improving cardiometabolic profiles. Some medications have shown efficacy to a lesser extent.
MASLD is a complex condition that requires attention, especially in lean patients. Risk factors and metabolic conditions are associated with this condition independently of BMI. Therefore, investigations aimed at decreasing the risk of future complications, such as cirrhosis or the development of hepatocellular carcinoma in lean MASLD patient, are necessary with the same relevance as in overweight/obese counterparts.
Future studies using this new nomenclature of MASLD can contribute to standardizing and generalizing study results in lean patients with steatotic liver diseases. It is also important to take into consideration other values, such as muscle mass or waist circumference and not only BMI, to make a more accurate evaluation of the lean patients.
| 1. | Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434-438. [PubMed] |
| 2. | Eslam M, Sanyal AJ, George J; International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158:1999-2014.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2794] [Cited by in RCA: 2497] [Article Influence: 416.2] [Reference Citation Analysis (5)] |
| 3. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Narro GEC, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Ann Hepatol. 2024;29:101133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 443] [Article Influence: 221.5] [Reference Citation Analysis (3)] |
| 4. | Chung GE, Yu SJ, Yoo JJ, Cho Y, Lee KN, Shin DW, Kim D, Kim YJ, Yoon JH, Han K, Cho EJ. Lean or diabetic subtypes predict increased all-cause and disease-specific mortality in metabolic-associated fatty liver disease. BMC Med. 2023;21:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (3)] |
| 5. | Semmler G, Wernly S, Bachmayer S, Leitner I, Wernly B, Egger M, Schwenoha L, Datz L, Balcar L, Semmler M, Stickel F, Niederseer D, Aigner E, Datz C. Metabolic Dysfunction-Associated Fatty Liver Disease (MAFLD)-Rather a Bystander Than a Driver of Mortality. J Clin Endocrinol Metab. 2021;106:2670-2677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 6. | Le MH, Yeo YH, Li X, Li J, Zou B, Wu Y, Ye Q, Huang DQ, Zhao C, Zhang J, Liu C, Chang N, Xing F, Yan S, Wan ZH, Tang NSY, Mayumi M, Liu X, Rui F, Yang H, Yang Y, Jin R, Le RHX, Xu Y, Le DM, Barnett S, Stave CD, Cheung R, Zhu Q, Nguyen MH. 2019 Global NAFLD Prevalence: A Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2022;20:2809-2817.e28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 519] [Cited by in RCA: 477] [Article Influence: 119.3] [Reference Citation Analysis (2)] |
| 7. | Younossi ZM, Stepanova M, Negro F, Hallaji S, Younossi Y, Lam B, Srishord M. Nonalcoholic fatty liver disease in lean individuals in the United States. Medicine (Baltimore). 2012;91:319-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 441] [Cited by in RCA: 410] [Article Influence: 29.3] [Reference Citation Analysis (5)] |
| 8. | WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9413] [Cited by in RCA: 8600] [Article Influence: 390.9] [Reference Citation Analysis (3)] |
| 9. | Kashiwagi K, Takayama M, Fukuhara K, Shimizu-Hirota R, Chu PS, Nakamoto N, Inoue N, Iwao Y, Kanai T. A significant association of non-obese non-alcoholic fatty liver disease with sarcopenic obesity. Clin Nutr ESPEN. 2020;38:86-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 10. | Byeon JH, Kang MK, Kim MC. Association of Low Skeletal Muscle Mass with the Phenotype of Lean Non-Alcoholic Fatty Liver Disease. Healthcare (Basel). 2022;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Cho Y, Chang Y, Ryu S, Jung HS, Kim CW, Oh H, Kim MK, Sohn W, Shin H, Wild SH, Byrne CD. Skeletal muscle mass to visceral fat area ratio as a predictor of NAFLD in lean and overweight men and women with effect modification by sex. Hepatol Commun. 2022;6:2238-2252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 12. | Kim HK, Bae SJ, Lee MJ, Kim EH, Park H, Kim HS, Cho YK, Jung CH, Lee WJ, Choe J. Association of Visceral Fat Obesity, Sarcopenia, and Myosteatosis with Non-Alcoholic Fatty Liver Disease without Obesity. Clin Mol Hepatol. 2023;29:987-1001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 13. | Saponaro C, Sabatini S, Gaggini M, Carli F, Rosso C, Positano V, Armandi A, Caviglia GP, Faletti R, Bugianesi E, Gastaldelli A. Adipose tissue dysfunction and visceral fat are associated with hepatic insulin resistance and severity of NASH even in lean individuals. Liver Int. 2022;42:2418-2427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 14. | Vilarinho S, Ajmera V, Zheng M, Loomba R. Emerging Role of Genomic Analysis in Clinical Evaluation of Lean Individuals With NAFLD. Hepatology. 2021;74:2241-2250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (4)] |
| 15. | Fuke N, Nagata N, Suganuma H, Ota T. Regulation of Gut Microbiota and Metabolic Endotoxemia with Dietary Factors. Nutrients. 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 215] [Cited by in RCA: 192] [Article Influence: 27.4] [Reference Citation Analysis (6)] |
| 16. | Alharthi J, Pan Z, Gloss BS, McLeod D, Weltman M, George J, Eslam M. Loss of metabolic adaptation in lean MAFLD is driven by endotoxemia leading to epigenetic reprogramming. Metabolism. 2023;144:155583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 17. | Xu R, Pan J, Zhou W, Ji G, Dang Y. Recent advances in lean NAFLD. Biomed Pharmacother. 2022;153:113331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 18. | Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, Abdelmalek MF, Caldwell S, Barb D, Kleiner DE, Loomba R. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology. 2023;77:1797-1835. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1980] [Cited by in RCA: 1770] [Article Influence: 590.0] [Reference Citation Analysis (6)] |
| 19. | Chen F, Esmaili S, Rogers GB, Bugianesi E, Petta S, Marchesini G, Bayoumi A, Metwally M, Azardaryany MK, Coulter S, Choo JM, Younes R, Rosso C, Liddle C, Adams LA, Craxì A, George J, Eslam M. Lean NAFLD: A Distinct Entity Shaped by Differential Metabolic Adaptation. Hepatology. 2020;71:1213-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 266] [Article Influence: 44.3] [Reference Citation Analysis (9)] |
| 20. | Luo F, Oldoni F, Das A. TM6SF2: A Novel Genetic Player in Nonalcoholic Fatty Liver and Cardiovascular Disease. Hepatol Commun. 2022;6:448-460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 103] [Article Influence: 25.8] [Reference Citation Analysis (4)] |
| 21. | Wang Y, Kory N, BasuRay S, Cohen JC, Hobbs HH. PNPLA3, CGI-58, and Inhibition of Hepatic Triglyceride Hydrolysis in Mice. Hepatology. 2019;69:2427-2441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 169] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 22. | Petta S, Vanni E, Bugianesi E, Rosso C, Cabibi D, Cammà C, Di Marco V, Eslam M, Grimaudo S, Macaluso FS, McLeod D, Pipitone RM, Abate ML, Smedile A, George J, Craxì A. PNPLA3 rs738409 I748M is associated with steatohepatitis in 434 non-obese subjects with hepatitis C. Aliment Pharmacol Ther. 2015;41:939-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 23. | Meroni M, Longo M, Fracanzani AL, Dongiovanni P. MBOAT7 down-regulation by genetic and environmental factors predisposes to MAFLD. EBioMedicine. 2020;57:102866. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 24. | Long MT, Noureddin M, Lim JK. AGA Clinical Practice Update: Diagnosis and Management of Nonalcoholic Fatty Liver Disease in Lean Individuals: Expert Review. Gastroenterology. 2022;163:764-774.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 247] [Cited by in RCA: 235] [Article Influence: 58.8] [Reference Citation Analysis (0)] |
| 25. | Lan Y, Lu Y, Li J, Hu S, Chen S, Wang Y, Yuan X, Liu H, Wang X, Wu S, Wang L. Outcomes of subjects who are lean, overweight or obese with nonalcoholic fatty liver disease: A cohort study in China. Hepatol Commun. 2022;6:3393-3405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 26. | Kwon YM, Oh SW, Hwang SS, Lee C, Kwon H, Chung GE. Association of nonalcoholic fatty liver disease with components of metabolic syndrome according to body mass index in Korean adults. Am J Gastroenterol. 2012;107:1852-1858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 156] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 27. | Raghu Teja KJSS, Durgaprasad BK, Vijayalakshmi P. Evaluation and Comparative Correlation of Abdominal Fat Related Parameters in Obese and Non-obese Groups Using Computed Tomography. Curr Med Imaging. 2021;17:417-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 28. | Yu AH, Duan-Mu YY, Zhang Y, Wang L, Guo Z, Yu YQ, Wang YS, Cheng XG. Correlation between Non-Alcoholic Fatty Liver Disease and Visceral Adipose Tissue in Non-Obese Chinese Adults: A CT Evaluation. Korean J Radiol. 2018;19:923-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 29. | Zheng M, Huang DQ, Konkwo C, Agrawal S, Khera AV, Loomba R, Vilarinho S, Ajmera V. Genomic analysis of lean individuals with NAFLD identifies monogenic disorders in a prospective cohort study. JHEP Rep. 2023;5:100692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 30. | Shida T, Oshida N, Suzuki H, Okada K, Watahiki T, Oh S, Kim T, Isobe T, Okamoto Y, Ariizumi SI, Yamamoto M, Shoda J. Clinical and anthropometric characteristics of non-obese non-alcoholic fatty liver disease subjects in Japan. Hepatol Res. 2020;50:1032-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 31. | Unger LW, Forstner B, Muckenhuber M, Scheuba K, Eigenbauer E, Scheiner B, Pfisterer N, Paternostro R, Trauner M, Mandorfer M, Reiberger T. Hepatic Steatosis in Lean Patients: Risk Factors and Impact on Mortality. Dig Dis Sci. 2020;65:2712-2718. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 32. | Yari Z, Fotros D, Hekmatdoost A. Comparison of cardiometabolic risk factors between obese and non-obese patients with nonalcoholic fatty liver disease. Sci Rep. 2023;13:14531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 33. | Shi YW, Wang QY, Zhao XY, Sun YM, Kong YY, Ou XJ, Jia JD, Wu SS, You H. Non-obese patients with nonalcoholic fatty liver disease may use a lower liver stiffness cut-off to assess fibrosis stages. J Dig Dis. 2020;21:279-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 34. | Lee SR, Han KD, Choi EK, Oh S, Lip GYH. Nonalcoholic fatty liver disease and the risk of atrial fibrillation stratified by body mass index: a nationwide population-based study. Sci Rep. 2021;11:3737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 35. | Cong F, Zhu L, Deng L, Xue Q, Wang J. Correlation between nonalcoholic fatty liver disease and left ventricular diastolic dysfunction in non-obese adults: a cross-sectional study. BMC Gastroenterol. 2023;23:90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 36. | Zeng J, Yang RX, Sun C, Pan Q, Zhang RN, Chen GY, Hu Y, Fan JG. Prevalence, clinical characteristics, risk factors, and indicators for lean Chinese adults with nonalcoholic fatty liver disease. World J Gastroenterol. 2020;26:1792-1804. [PubMed] [DOI] [Full Text] |
| 37. | Wei JL, Leung JC, Loong TC, Wong GL, Yeung DK, Chan RS, Chan HL, Chim AM, Woo J, Chu WC, Wong VW. Prevalence and Severity of Nonalcoholic Fatty Liver Disease in Non-Obese Patients: A Population Study Using Proton-Magnetic Resonance Spectroscopy. Am J Gastroenterol. 2015;110:1306-14; quiz 1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 251] [Article Influence: 22.8] [Reference Citation Analysis (3)] |
| 38. | Shen J, Wong GL, Chan HL, Chan HY, Yeung DK, Chan RS, Chim AM, Chan AW, Choi PC, Woo J, Chu WC, Wong VW. PNPLA3 gene polymorphism accounts for fatty liver in community subjects without metabolic syndrome. Aliment Pharmacol Ther. 2014;39:532-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 99] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 39. | Tobari M, Hashimoto E. Characteristic Features of Nonalcoholic Fatty Liver Disease in Japan with a Focus on the Roles of Age, Sex and Body Mass Index. Gut Liver. 2020;14:537-545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 40. | Aneni EC, Bittencourt MS, Teng C, Cainzos-Achirica M, Osondu CU, Soliman A, Al-Mallah M, Buddoff M, Parise ER, Santos RD, Nasir K. The risk of cardiometabolic disorders in lean non-alcoholic fatty liver disease: A longitudinal study. Am J Prev Cardiol. 2020;4:100097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 41. | Rahman MM, Kibria MG, Begum H, Haque M, Sultana N, Akhter M, Rowshon AHM, Ahmed F, Hasan M. Prevalence, risk factors and metabolic profile of the non-obese and obese non-alcoholic fatty liver disease in a rural community of South Asia. BMJ Open Gastroenterol. 2020;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 42. | Nabi O, Lapidus N, Boursier J, de Ledinghen V, Petit JM, Kab S, Renuy A, Zins M, Lacombe K, Serfaty L. Lean individuals with NAFLD have more severe liver disease and poorer clinical outcomes (NASH-CO Study). Hepatology. 2023;78:272-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 90] [Article Influence: 30.0] [Reference Citation Analysis (3)] |
| 43. | Chung GE, Lee JH, Lee H, Kim MK, Yim JY, Choi SY, Kim YJ, Yoon JH, Kim D. Nonalcoholic fatty liver disease and advanced fibrosis are associated with left ventricular diastolic dysfunction. Atherosclerosis. 2018;272:137-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 44. | Boonchai P, Kositamongkol C, Jitrukthai S, Phothirat S, Mepramoon E, Nimitpunya P, Srivanichakorn W, Chaisathaphol T, Washirasaksiri C, Auesomwang C, Sitasuwan T, Tinmanee R, Sayabovorn N, Charatcharoenwitthaya P, Phisalprapa P. Clinical Differences and Non-Alcoholic Fatty Liver Disease-Related Factors of Lean and Non-Lean Patients with Metabolic Syndrome. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 45. | Razouki ZA, Zhang X, Hwang JP, Heredia NI. Clinical Factors Associated with Non-Obese Nonalcoholic Fatty Liver Disease Detected among US Adults in the NHANES 2017-2018. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 46. | Semmler G, Wernly S, Bachmayer S, Wernly B, Schwenoha L, Huber-Schönauer U, Stickel F, Niederseer D, Aigner E, Datz C. Nonalcoholic Fatty Liver Disease in Lean Subjects: Associations With Metabolic Dysregulation and Cardiovascular Risk-A Single-Center Cross-Sectional Study. Clin Transl Gastroenterol. 2021;12:e00326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 47. | Kalligeros M, Vassilopoulos S, Vassilopoulos A, Shehadeh F, Lazaridou I, Mylonakis E, Promrat K, Wands JR. Prevalence and risk factors of nonalcoholic fatty liver disease, high-risk nonalcoholic steatohepatitis, and fibrosis among lean United States adults: NHANES 2017-2020. Ann Gastroenterol. 2023;36:670-677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 48. | Li M, Zhang W, Li X, Liang S, Zhang Y, Mo Y, Rao S, Zhang H, Huang Y, Zhu Y, Zhang Z, Yang W. Metabolic and Risk Profiles of Lean and Non-Lean Hepatic Steatosis among US Adults. Nutrients. 2023;15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 49. | Akyuz U, Yesil A, Yilmaz Y. Characterization of lean patients with nonalcoholic fatty liver disease: potential role of high hemoglobin levels. Scand J Gastroenterol. 2015;50:341-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (4)] |
| 50. | Ahmed OT, Gidener T, Mara KC, Larson JJ, Therneau TM, Allen AM. Natural History of Nonalcoholic Fatty Liver Disease With Normal Body Mass Index: A Population-Based Study. Clin Gastroenterol Hepatol. 2022;20:1374-1381.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 51. | Dai W, Zhang Z, Zhao S. The Risk of Type 2 Diabetes and Coronary Artery Disease in Non-obese Patients With Non-alcoholic Fatty Liver Disease: A Cohort Study. Front Cardiovasc Med. 2021;8:680664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 52. | De A, Mehta M, Singh P, Bhagat N, Mitra S, Das A, Duseja A. Lean Indian patients with non-alcoholic fatty liver disease (NAFLD) have less metabolic risk factors but similar liver disease severity as non-lean patients with NAFLD. Int J Obes (Lond). 2023;47:986-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 53. | Hagström H, Nasr P, Ekstedt M, Hammar U, Stål P, Hultcrantz R, Kechagias S. Risk for development of severe liver disease in lean patients with nonalcoholic fatty liver disease: A long-term follow-up study. Hepatol Commun. 2018;2:48-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 242] [Article Influence: 30.3] [Reference Citation Analysis (7)] |
| 54. | Wei L, Cheng X, Luo Y, Yang R, Lei Z, Jiang H, Chen L. Lean non-alcoholic fatty liver disease and risk of incident diabetes in a euglycaemic population undergoing health check-ups: A cohort study. Diabetes Metab. 2021;47:101200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 55. | Huang JF, Tsai PC, Yeh ML, Huang CF, Huang CI, Hsieh MH, Dai CY, Yang JF, Chen SC, Yu ML, Chuang WL, Chang WY. Risk stratification of non-alcoholic fatty liver disease across body mass index in a community basis. J Formos Med Assoc. 2020;119:89-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (4)] |
| 56. | Zhang Z, Zhang L, Jiang W, Du T, Yuan G. Non-obese NAFLD had no better cardio-metabolic risk profile than obese NAFLD in type 2 diabetic patients. Cardiovasc Diabetol. 2022;21:210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 57. | Kawanaka M, Nishino K, Kawada M, Ishii K, Tanikawa T, Katsumata R, Urata N, Nakamura J, Suehiro M, Haruma K, Kawamoto H. Lean nonalcoholic fatty liver disease: Age-dependent differences in pathology, prognosis, and liver-related events. Hepatol Res. 2023;53:829-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 58. | Aneni EC, Saeed GJ, Bittencourt MS, Cainzos-Achirica M, Osondu CU, Budoff M, Parise ER, Santos RD, Nasir K. Cardiometabolic disorders, inflammation and the incidence of non-alcoholic fatty liver disease: A longitudinal study comparing lean and non-lean individuals. PLoS One. 2022;17:e0266505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 59. | Biswas S, Vaishnav M, Farooqui N, Aggarwal A, Pathak P, Yadav R, Das P, Elhence A, Goel A, Mishra AK, Shalimar. Impact of body mass index on disease progression and outcomes in patients with nonalcoholic fatty liver disease. Postgrad Med J. 2023;99:1094-1103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 60. | Weinberg EM, Trinh HN, Firpi RJ, Bhamidimarri KR, Klein S, Durlam J, Watkins S, Reddy KR, Weiss M, Zink RC, Lok AS. Lean Americans With Nonalcoholic Fatty Liver Disease Have Lower Rates of Cirrhosis and Comorbid Diseases. Clin Gastroenterol Hepatol. 2021;19:996-1008.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 61. | Ishido S, Tamaki N, Takahashi Y, Uchihara N, Suzuki K, Tanaka Y, Miyamoto H, Yamada M, Matsumoto H, Nobusawa T, Keitoku T, Takaura K, Tanaka S, Maeyashiki C, Yasui Y, Tsuchiya K, Nakanishi H, Kurosaki M, Izumi N. Risk of cardiovascular disease in lean patients with nonalcoholic fatty liver disease. BMC Gastroenterol. 2023;23:211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 62. | Wijarnpreecha K, Li F, Lundin SK, Suresh D, Song MW, Tao C, Chen VL, Lok ASF. Higher mortality among lean patients with non-alcoholic fatty liver disease despite fewer metabolic comorbidities. Aliment Pharmacol Ther. 2023;57:1014-1027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (4)] |
| 63. | Shao C, Ye J, Li F, Lin Y, Wu T, Wang W, Feng S, Zhong B. Early Predictors of Cardiovascular Disease Risk in Nonalcoholic Fatty Liver Disease: Non-obese Versus Obese Patients. Dig Dis Sci. 2020;65:1850-1860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 64. | Dong BY, Mao YQ, Li ZY, Yu FJ. The value of the atherogenic index of plasma in non-obese people with non-alcoholic fatty liver disease: a secondary analysis based on a cross-sectional study. Lipids Health Dis. 2020;19:148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 65. | Golabi P, Paik J, Fukui N, Locklear CT, de Avilla L, Younossi ZM. Patients With Lean Nonalcoholic Fatty Liver Disease Are Metabolically Abnormal and Have a Higher Risk for Mortality. Clin Diabetes. 2019;37:65-72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 105] [Article Influence: 15.0] [Reference Citation Analysis (4)] |
| 66. | Fracanzani AL, Petta S, Lombardi R, Pisano G, Russello M, Consonni D, Di Marco V, Cammà C, Mensi L, Dongiovanni P, Valenti L, Craxì A, Fargion S. Liver and Cardiovascular Damage in Patients With Lean Nonalcoholic Fatty Liver Disease, and Association With Visceral Obesity. Clin Gastroenterol Hepatol. 2017;15:1604-1611.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 177] [Article Influence: 19.7] [Reference Citation Analysis (6)] |
| 67. | Trifan A, Rotaru A, Stafie R, Stratina E, Zenovia S, Nastasa R, Huiban L, Cuciureanu T, Muzîca C, Chiriac S, Gîrleanu I, Sîngeap AM, Sfarti C, Cojocariu C, Stanciu C. Clinical and Laboratory Characteristics of Normal Weight and Obese Individuals with Non-Alcoholic Fatty Liver Disease. Diagnostics (Basel). 2022;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 68. | Zhang S, Liu XH, Wang G, Zhang L, Zhu YJ, Wu J, Liu YL, Zhang J. Population differences of metabolic associated fatty liver disease and nonalcoholic fatty liver disease based on a com¬munity elderly population. Linchuang Gandanbing Zazhi. 2022;547. [DOI] [Full Text] |
| 69. | Zhang YN, Wang QQ, Chen YS, Shen C, Xu CF. Association between Serum Uric Acid to HDL-Cholesterol Ratio and Nonalcoholic Fatty Liver Disease in Lean Chinese Adults. Int J Endocrinol. 2020;2020:5953461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 70. | Shao C, Ye J, Li F, Feng S, Wang W, Zhong B. Different predictors of steatosis and fibrosis severity among lean, overweight and obese patients with nonalcoholic fatty liver disease. Dig Liver Dis. 2019;51:1392-1399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (4)] |
| 71. | Alam S, Gupta UD, Alam M, Kabir J, Chowdhury ZR, Alam AK. Clinical, anthropometric, biochemical, and histological characteristics of nonobese nonalcoholic fatty liver disease patients of Bangladesh. Indian J Gastroenterol. 2014;33:452-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (4)] |
| 72. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A, Yeh M, McCullough AJ, Sanyal AJ; Nonalcoholic Steatohepatitis Clinical Research Network. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9231] [Cited by in RCA: 8598] [Article Influence: 409.4] [Reference Citation Analysis (7)] |
| 73. | Leung JC, Loong TC, Wei JL, Wong GL, Chan AW, Choi PC, Shu SS, Chim AM, Chan HL, Wong VW. Histological severity and clinical outcomes of nonalcoholic fatty liver disease in nonobese patients. Hepatology. 2017;65:54-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 289] [Article Influence: 32.1] [Reference Citation Analysis (3)] |
| 74. | Iwaki M, Kessoku T, Tanaka K, Ozaki A, Kasai Y, Yamamoto A, Takahashi K, Kobayashi T, Nogami A, Honda Y, Ogawa Y, Imajo K, Oyamada S, Kobayashi N, Aishima S, Saito S, Nakajima A, Yoneda M. Comparison of long-term prognosis between non-obese and obese patients with non-alcoholic fatty liver disease. JGH Open. 2022;6:696-703. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 75. | Kim D, Kim W, Joo SK, Kim JH, Harrison SA, Younossi ZM, Ahmed A. Predictors of nonalcoholic steatohepatitis and significant fibrosis in non-obese nonalcoholic fatty liver disease. Liver Int. 2019;39:332-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (3)] |
| 76. | Denkmayr L, Feldman A, Stechemesser L, Eder SK, Zandanell S, Schranz M, Strasser M, Huber-Schönauer U, Buch S, Hampe J, Paulweber B, Lackner C, Haufe H, Sotlar K, Datz C, Aigner E. Lean Patients with Non-Alcoholic Fatty Liver Disease Have a Severe Histological Phenotype Similar to Obese Patients. J Clin Med. 2018;7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 94] [Article Influence: 11.8] [Reference Citation Analysis (4)] |
| 77. | Rastogi A, Rath I, Varadarajan A, Ramakrishna G, Bihari C, Maiwall R. Non-alcoholic fatty liver disease (NAFLD) in lean individuals - Single centre large cohort clinicopathologic and immunophenotypic study. Pathol Res Pract. 2022;238:154112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 78. | Tan EX, Lee JW, Jumat NH, Chan WK, Treeprasertsuk S, Goh GB, Fan JG, Song MJ, Charatcharoenwitthaya P, Duseja A, Imajo K, Nakajima A, Seki Y, Kasama K, Kakizaki S, Lesmana LA, Zheng KI, Zheng MH, Koh CJ, Ho KY, Goh KL, Wong VW, Dan YY. Non-obese non-alcoholic fatty liver disease (NAFLD) in Asia: an international registry study. Metabolism. 2022;126:154911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 10.8] [Reference Citation Analysis (1)] |
| 79. | Eren F, Kaya E, Yilmaz Y. Accuracy of Fibrosis-4 index and non-alcoholic fatty liver disease fibrosis scores in metabolic (dysfunction) associated fatty liver disease according to body mass index: failure in the prediction of advanced fibrosis in lean and morbidly obese individuals. Eur J Gastroenterol Hepatol. 2022;34:98-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 58] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 80. | Park H, Yoon EL, Ito T, Jo AJ, Kim M, Lee J, Kim HL, Arai T, Atsukawa M, Kawanaka M, Toyoda H, Ishigami M, Yu ML, Jun DW, Nguyen MH. Diagnostic Performance of the Fibrosis-4 Index and Nonalcoholic Fatty Liver Disease Fibrosis Score in Lean Adults With Nonalcoholic Fatty Liver Disease. JAMA Netw Open. 2023;6:e2329568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 81. | Fu C, Wai JW, Nik Mustapha NR, Irles M, Wong GL, Mahadeva S, Shili S, Chan AW, Merrouche W, Chan HL, Foucher J, Le Bail B, Wong VW, Chan WK, de Lédinghen V. Performance of Simple Fibrosis Scores in Nonobese Patients With Nonalcoholic Fatty Liver Disease. Clin Gastroenterol Hepatol. 2020;18:2843-2845.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 82. | Li C, Guo P, Zhang R, Zhang M, Li Y, Huang M, Ji X, Jiang Y, Wang C, Li R, Feng R; Nutrition and Health Institute. Both WHR and FLI as Better Algorithms for Both Lean and Overweight/Obese NAFLD in a Chinese Population. J Clin Gastroenterol. 2019;53:e253-e260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (5)] |
| 83. | Enjoji M, Machida K, Kohjima M, Kato M, Kotoh K, Matsunaga K, Nakashima M, Nakamuta M. NPC1L1 inhibitor ezetimibe is a reliable therapeutic agent for non-obese patients with nonalcoholic fatty liver disease. Lipids Health Dis. 2010;9:29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 54] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 84. | Mofidi F, Poustchi H, Yari Z, Nourinayyer B, Merat S, Sharafkhah M, Malekzadeh R, Hekmatdoost A. Synbiotic supplementation in lean patients with non-alcoholic fatty liver disease: a pilot, randomised, double-blind, placebo-controlled, clinical trial. Br J Nutr. 2017;117:662-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 176] [Article Influence: 19.6] [Reference Citation Analysis (1)] |
| 85. | Shinozaki S, Tahara T, Miura K, Lefor AK, Yamamoto H. Pemafibrate therapy for non-alcoholic fatty liver disease is more effective in lean patients than obese patients. Clin Exp Hepatol. 2022;8:278-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (2)] |
| 86. | Suzuki Y, Maekawa S, Yamashita K, Osawa L, Komiyama Y, Nakakuki N, Takada H, Muraoka M, Sato M, Takano S, Fukasawa M, Yamaguchi T, Funayama S, Morisaka H, Onishi H, Enomoto N. Effect of a combination of pemafibrate and a mild low-carbohydrate diet on obese and non-obese patients with metabolic-associated fatty liver disease. J Gastroenterol Hepatol. 2023;38:921-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 87. | Inoue M, Hayashi A, Taguchi T, Arai R, Sasaki S, Takano K, Inoue Y, Shichiri M. Effects of canagliflozin on body composition and hepatic fat content in type 2 diabetes patients with non-alcoholic fatty liver disease. J Diabetes Investig. 2019;10:1004-1011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 88. | Jin YJ, Kim KM, Hwang S, Lee SG, Ha TY, Song GW, Jung DH, Kim KH, Yu E, Shim JH, Lim YS, Lee HC, Chung YH, Lee Y, Suh DJ. Exercise and diet modification in non-obese non-alcoholic fatty liver disease: analysis of biopsies of living liver donors. J Gastroenterol Hepatol. 2012;27:1341-1347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 83] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 89. | Wong VW, Wong GL, Chan RS, Shu SS, Cheung BH, Li LS, Chim AM, Chan CK, Leung JK, Chu WC, Woo J, Chan HL. Beneficial effects of lifestyle intervention in non-obese patients with non-alcoholic fatty liver disease. J Hepatol. 2018;69:1349-1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 244] [Article Influence: 30.5] [Reference Citation Analysis (1)] |
| 90. | Hamurcu Varol P, Kaya E, Alphan E, Yilmaz Y. Role of intensive dietary and lifestyle interventions in the treatment of lean nonalcoholic fatty liver disease patients. Eur J Gastroenterol Hepatol. 2020;32:1352-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 91. | Sinn DH, Kang D, Cho SJ, Paik SW, Guallar E, Cho J, Gwak GY. Weight change and resolution of fatty liver in normal weight individuals with nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2021;33:e529-e534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 92. | Satapathy SK, Jiang Y, Agbim U, Wu C, Bernstein DE, Teperman LW, Kedia SK, Aithal GP, Bhamidimarri KR, Duseja A, Maiwall R, Maliakkal B, Jalal P, Patel K, Puri P, Ravinuthala R, Wong VW, Abdelmalek MF, Ahmed A, Thuluvath PJ, Singal AK; Global NAFLD Consortium. Posttransplant Outcome of Lean Compared With Obese Nonalcoholic Steatohepatitis in the United States: The Obesity Paradox. Liver Transpl. 2020;26:68-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 93. | Ochoa-Allemant P, Trivedi HD, Saberi B, Bonder A, Fricker ZP. Waitlist and posttransplantation outcomes of lean individuals with nonalcoholic fatty liver disease. Liver Transpl. 2023;29:145-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology & hepatology
Country/Territory of origin: Peru
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kelleni MT, Egypt S-Editor: Liu JH L-Editor: A P-Editor: Guo X