Published online Nov 27, 2020. doi: 10.4254/wjh.v12.i11.965
Peer-review started: July 16, 2020
First decision: September 14, 2020
Revised: September 16, 2020
Accepted: October 5, 2020
Article in press: October 5, 2020
Published online: November 27, 2020
Processing time: 130 Days and 14.3 Hours
The morbidity and mortality of human immunodeficiency virus (HIV)-infection is often associated with liver disease, which progresses slowly into severe liver dysfunction. There are multiple insults which exacerbate HIV-related liver injury, including HIV-associated dysregulation of lipid metabolism and fat turnover, co-infections with hepatotropic viruses and alcohol abuse. As we reported before, exposure of hepatocytes to HIV and alcohol metabolites causes high oxidative stress, impairs proteasomal and lysosomal functions leading to accumulation of HIV in these cells, which end-ups with apoptotic cell death and finally promotes development of liver fibrosis.
To study whether obeticholic acid (OCA) prevents HIV/ethanol metabolism-induced hepatotoxicity and subsequent activation of hepatic stellate cells (HSC) by HIV+ apoptotic hepatocyte engulfment.
Huh7.5-CYP (RLW) cells were exposed to HIV and acetaldehyde-generating system (AGS) in the presence or absence of OCA. In the cells, we measured the expression of HIV-related markers: HIVgagRNA-by real-time polymerase chain reaction (PCR), p24- by western blot, HIV DNA-by semi-nested PCR, integrated HIV DNA-by ddPCR. Lysosomal and proteasomal activities were measured using fluorometrically-labeled substrates. For hepatocyte apoptosis, cleaved caspase 3 and cleaved PARP were visualized by western blot and cytokeratin 18- by M30 ELISA-in supernatants. Apoptotic bodies were generated from untreated and HIV-treated RLW cells exposed to UV light. Pro-fibrotic activation of HSC was characterized by Col1A1 and transforming growth factor-β mRNAs, while inflammasome activation- by NLRP3, caspase 1, interleukin (IL)-6, IL-1β mRNA levels.
In RLW cells, OCA treatment attenuated HIV-AGS-induced accumulation of HIVgagRNA, HIV DNA and p24. OCA suppressed reactive oxygen species production and restored chymotrypsin-like proteasome activity as well as cathepsin B lysosome activity. OCA also decreased HIV-AGS-triggered apoptosis in RLW cells. Exposure of HIV-containing apoptotic hepatocytes to HSC prevented activation of inflammasome and induced pro-fibrotic activation in these cells.
We conclude that by suppressing oxidative stress and restoring proteasomal and lysosomal functions impaired by HIV and ethanol metabolism, OCA decreases accumulation of HIV in hepatocytes, leading to down-regulation of apoptosis in these cells. In addition, OCA reverses pro-fibrotic and inflammasome-related activation of HSC triggered by engulfment of HIV-containing apoptotic hepatocytes, potentially contributing to suppression of liver fibrosis development.
Core Tip: We investigated the ability of obeticholic acid (OCA) to reverse pro-fibrotic effects of human immunodeficiency virus (HIV) and ethanol metabolism in liver cells. Based on our previous studies, hepatocyte apoptosis occurs under combined exposure of cells to HIV and ethanol metabolites. The subsequent engulfment of HIV-containing apoptotic hepatocytes by hepatic stellate cells induced pro-fibrotic activation in these cells, thereby promoting fibrosis development. Here, we demonstrated that OCA attenuates hepatocyte apoptosis by preventing accumulation of HIV components in liver cells exposed to virus and acetaldehyde-generating system (AGS) mimicking natural ethanol metabolism in primary hepatocytes. These beneficial effects of OCA are attributed to suppression of oxidative stress leading to restoration of HIV-AGS-impaired proteasomal and lysosomal functions in liver cells. OCA also reduces activation of inflammasome in hepatic stellate cells and their pro-fibrotic activation. Thus, anti-fibrotic properties of OCA can be used for combined treatment of HIV-infected alcohol abusers with a high risk of liver fibrosis development.
- Citation: New-Aaron M, Ganesan M, Dagur RS, Kharbanda KK, Poluektova LY, Osna NA. Obeticholic acid attenuates human immunodeficiency virus/alcohol metabolism-induced pro-fibrotic activation in liver cells. World J Hepatol 2020; 12(11): 965-975
- URL: https://www.wjgnet.com/1948-5182/full/v12/i11/965.htm
- DOI: https://dx.doi.org/10.4254/wjh.v12.i11.965
Liver disease is a second-leading cause of mortality in human immunodeficiency virus (HIV)-infected patients[1]. While effective anti-retroviral treatment (ART) has dramatically decreased the onset of acquired immunodeficiency syndrome, the morbidity and mortality are often associated with liver disease, which progresses slowly into severe liver dysfunction. This progression is related not only to the ART-induced hepatotoxicity, but to HIV properties likely associated with progression of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis (NASH)[2,3]. There are multiple insults, which promote HIV-related liver injury, including dysregulation of lipid metabolism and fat turnover, co-infections with hepatotropic viruses and alcohol abuse[4,5].
Recently, we have shown that alcohol metabolite, acetaldehyde promotes accumulation of HIV proteins in hepatocytes, inducing oxidative stress and cell apoptosis[6]. Furthermore, the engulfment of these HIV-containing hepatocytes by non-parenchymal cells, macrophages and hepatic stellate cells (HSC) causes inflammasome activation in macrophages and pro-fibrotic activation of HSC[6]. This indicates that massive apoptosis of infected hepatocytes may induce continuous activation of HSC leading to liver fibrosis development. Since acetaldehyde-triggered HIV replication in hepatocytes is abortive[6], HIV accumulation cannot be fully controlled by ART and requires the treatment with additional anti-fibrotic drugs.
One of the promising drugs with anti-fibrotic activity is obeticholic acid (OCA). In 2016, OCA became the United States Food and Drug Administration-approved to treat primary biliary cholangitis and currently is at phase 3 trial (Regenerate) to treat fibrosis caused by NASH[7]. OCA is a farnesoid-X receptor (FXR) agonist, which binds to the FXR in the nucleus of liver and intestinal cells. Multiple genes are activated by this pathway, including the control of metabolism of bile acids, lipids, glucose, and amino acids. FXR is highly expressed on hepatocytes and immune cells, and is involved in pathogenesis of viral hepatitis, alcohol-and non-alcohol-induced liver disease[8]. In NASH-fibrosis, OCA regulates liver injury progression via targeting of gut microbiota[9]. Currently, it is not quite clear whether OCA directly modulates hepatocyte apoptosis: While some studies reported the lack of anti-apoptotic effects of OCA on these cells[10], other studies demonstrated the reduction of hepatocyte apoptosis by OCA-mediated suppression of metabolic stress and prevention of subsequent p53 activation, with further anti-fibrotic and anti-inflammatory downstream effects[11]. These studies were mainly performed on experimental in vivo models, which makes difficult to exclude the effects of OCA on gut microbiota, narrowing down the mechanisms to only OCA-regulated hepatocyte apoptosis. None of published in vivo or in vitro studies have been performed in the context of the effects of OCA on HIV- and alcohol-induced liver injury. However, activation of HSC by engulfment of HIV-expressing apoptotic hepatocytes generated as a downstream event in HIV and ethanol metabolism-induced oxidative stress is one of the reasons for liver fibrosis progression, which serves as an important target to prevent end-stage liver disease development. Thus, based on already characterized mechanisms of liver injury progression triggered by the combination of HIV with acetaldehyde[6], we aimed to investigate whether OCA protects from apoptotic hepatocyte death and from activation of HSC by engulfment of apoptotic HIV-infected hepatocytes.
This is the original (basic) study performed at University of Nebraska Medical Center, Omaha, NE, United States. Here, for the first time, in vitro approach is used to characterize the ability of OCA to reverse the pathology induced by HIV and ethanol metabolism in liver parenchymal and non-parenchymal cells. In our study, OCA has been tested as an anti-fibrotic drug, which affects the pathogenesis of HIV-alcohol-induced liver fibrosis development.
High glucose Dulbecco’s modified eagle medium and fetal bovine serum were purchased from Invitrogen (Carlsbad, CA, United States), Trizol was from Life Technologies, primer probes and real-time polymerase chain reaction (RT-PCR) reagents were from Applied Biosystems by Thermo Fisher Scientific, CA , United States. Other reagents, all analytical grade quality, were from Sigma (St. Louis, MO, United States).
As experimental prototype of human primary hepatocytes, we used Huh7.5-CYP (RLW) cells. These cells have reduced innate immunity and can be infected with HIV. To metabolize ethanol, they were stably transfected by CYP2E1, but do not express alcohol dehydrogenase (ADH). To overcome this limitation and mimic natural ethanol metabolism in primary hepatocytes, we treated RLW cells with acetaldehyde-generating system (AGS), which contains yeast ADH as a source of enzyme, NAD as a co-factor, and 50 mmol/L ethanol as a substrate for ADH and continuously produce physiologically relevant amount of acetaldehyde (Ach) without toxic effects. We have characterized and successfully used these cells and AGS for HCV-based ethanol liver studies[12-14]. Cells were pre-treated for 24 h with AGS and then exposed to HIVADA (MOI 0.1) for 48 h. To investigate the effects of OCA, cells were pre-treated with OCA (50 μmol/L) for 4 h before experiment.
HIV RNA was detected by RT-PCR; HIV DNA was detected by semi-nested PCR; integrated HIV DNA was measured by digital droplet PCR; reactive oxygen species (ROS) were quantified by DCF (2’,7’–dichlorofluorescein fluorescence method); western blot was performed as described. The details of all these methods were already published[6].
Apoptosis in RLW cells was measured by caspase 3 and PARP cleavage in cell lysates (western blot) and M30 Apoptosense ELISA (Duapharma group, Inc. West Chester, OH, United States) in cell supernatants.
Proteasome activities and cathepsin B and L activities were assayed fluorometrically as described previously[15,16].
Apoptotic bodies (AB) were generated from uninfected and HIV-infected RLW cells by exposure to UV light and characterized as shown previously[13].
As the source of human HSC, we used commercially available human cell line, LX2 (EMD Millipore, cat SCC064) grown based on instructions from the manufacturer. AB from RLW cells (AB Hep), both uninfected (control) and HIV-infected (ABHIV), were incubated with LX2 cells for 2-8 h at 1: 3 ratio and then pro-fibrotic markers [Col1A1, transforming growth factor (TGF)-β] and inflammasome-related parameters [NLRP3, caspase 1, interleukin (IL)-6, IL-1β] were quantified by RT-PCR.
Data were analyzed using GraphPad Prism v7.03 software (GraphPad, La Jolla, CA, United States). Data from at least three duplicate independent experiments were expressed as mean ± SEM. Comparisons among multiple groups were performed by one-way ANOVA, using a Tukey post-hoc test. For comparisons between two groups, we used Student’s t-test. A P value of 0.05 or less was considered significant.
Our previous studies on primary human hepatocytes exposed to ethanol and RLW cells treated with AGS demonstrated, first, that incubation of ADH-non-expressing RLW cells with AGS recapitulated the effects of ethanol on ethanol-metabolizing hepatocytes and second, that the highest levels of apoptotic hepatocyte death were observed when cells were exposed to both insults (ethanol/AGS and HIV)[6]. That is why in this paper, we presented the data on ability of OCA to protect cells from the harmful effects of AGS on HIV-infected hepatocyte-like RLW cells to mimic ethanol metabolism observed in primary human hepatocytes. For OCA screening, we did not use primary human hepatocytes since their supply was limited and they quickly (in 24 h) de-differentiate to loose ethanol-metabolizing capacity.
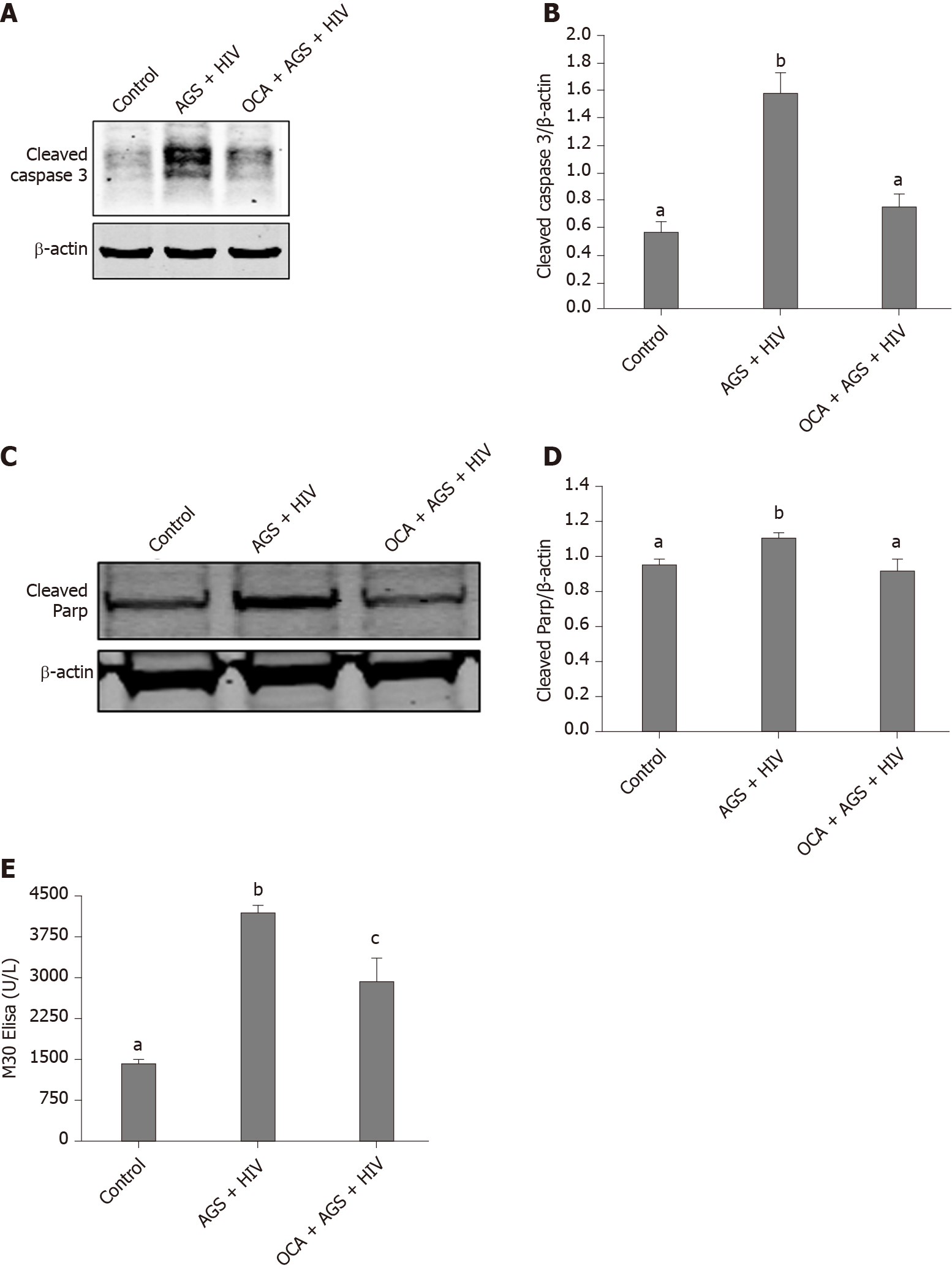
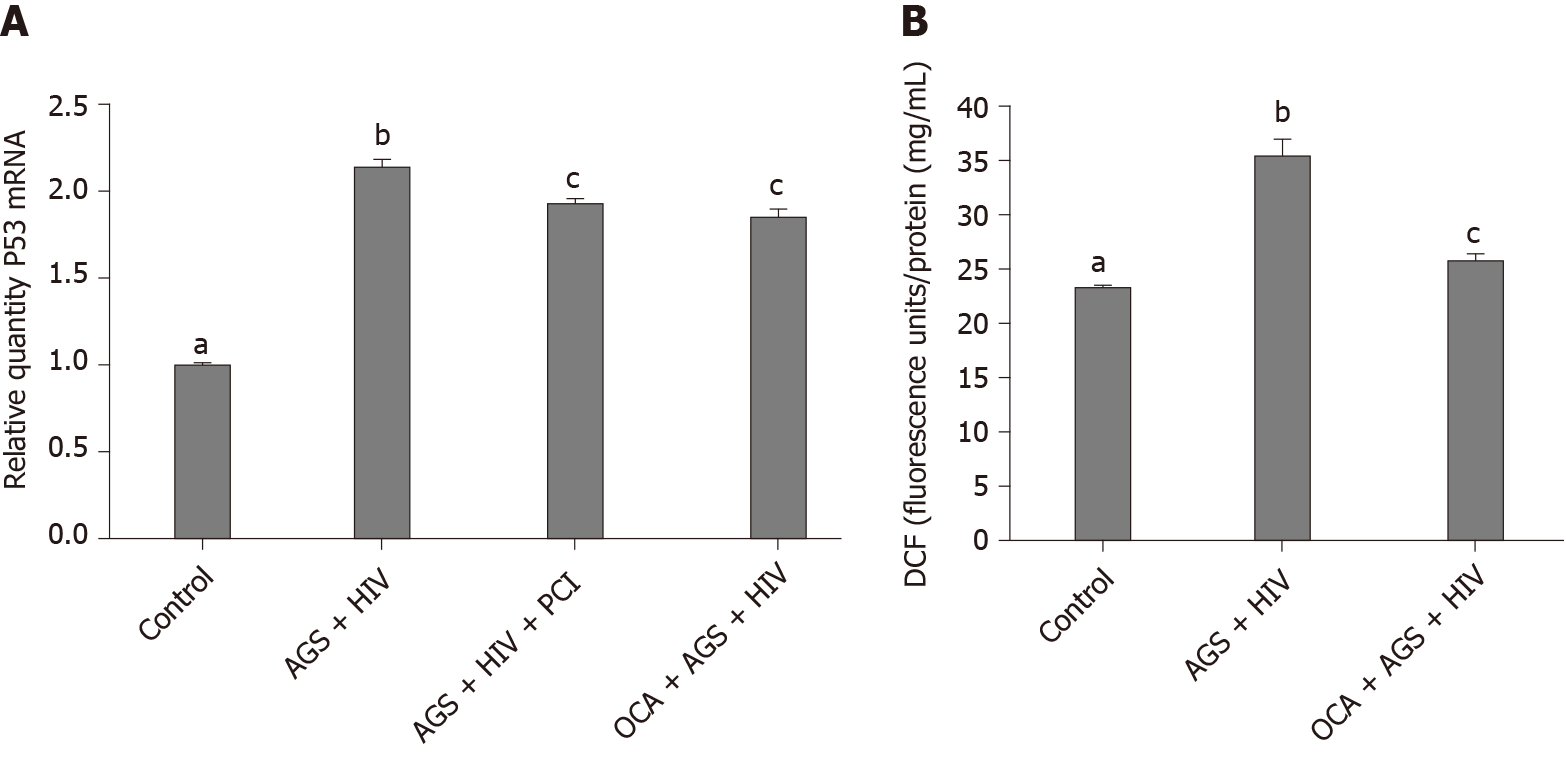
We found that while AGS + HIV induced caspase 3 cleavage almost three-fold, pre-treatment with OCA suppresses these effects (Figure 1A and B). The same trend (but with lower magnitude of response to AGS) was observed on cleaved PARP, and OCA also suppressed it (Figure 1C and D). Measuring cleaved cytokeratin 18 by M30 ELISA in cell supernatants, we found that OCA attenuated AGS-HIV-induced apoptosis in hepatocytes (Figure 1E). Since apoptosis may be induced by elevated p53, we measured the induction of p53 mRNA by AGS+HIV, in the presence or absence of pan-caspase inhibitor or OCA. As shown on Figure 2A, while AGS and HIV induced p53 mRNA level two-fold, both OCA and pan-caspase inhibitor reduced p53 expression only by 14%. In addition, OCA attenuated AGS-HIV-induced ROS production (Figure 2B), thereby protecting liver cells from oxidative stress.
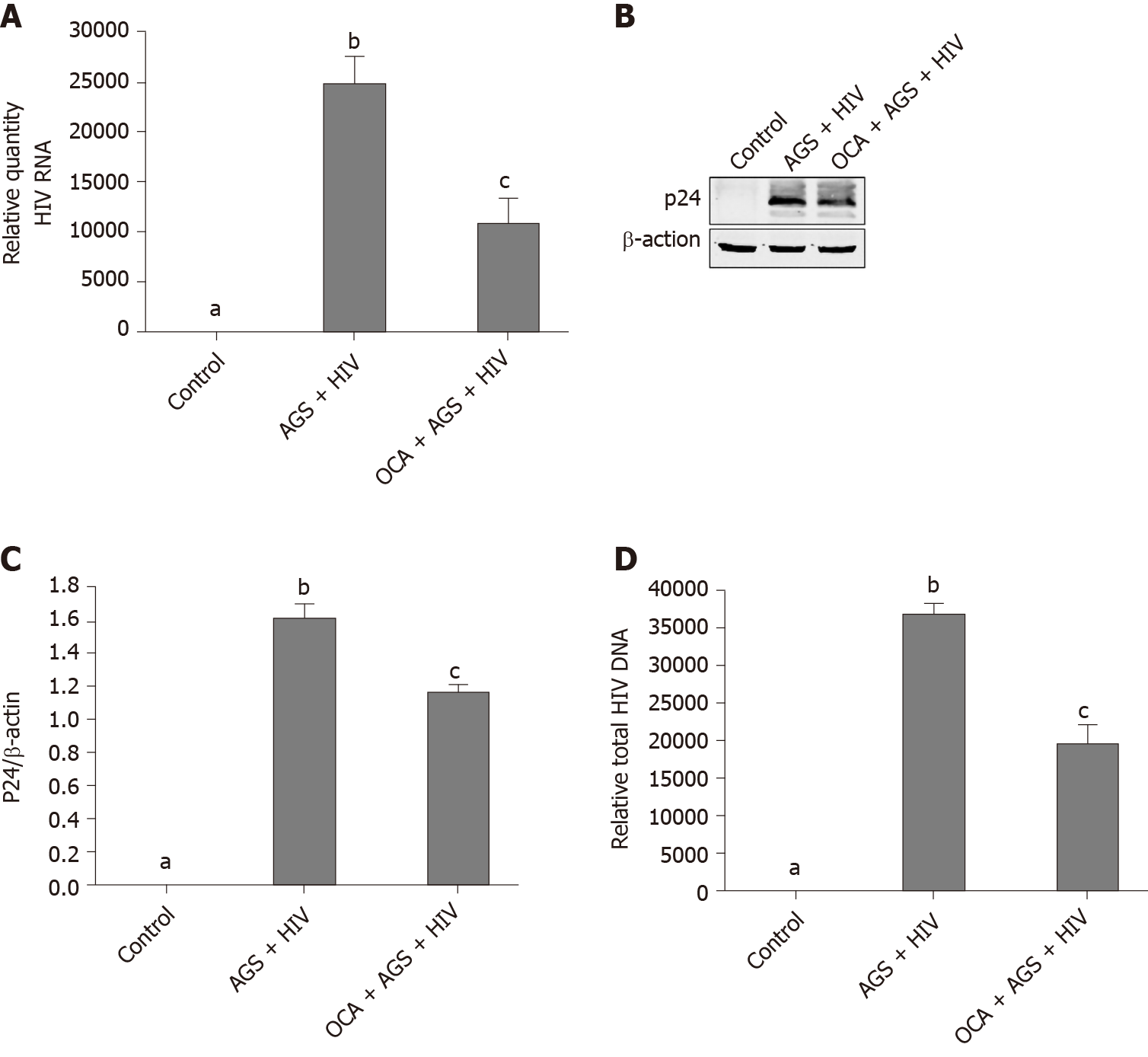
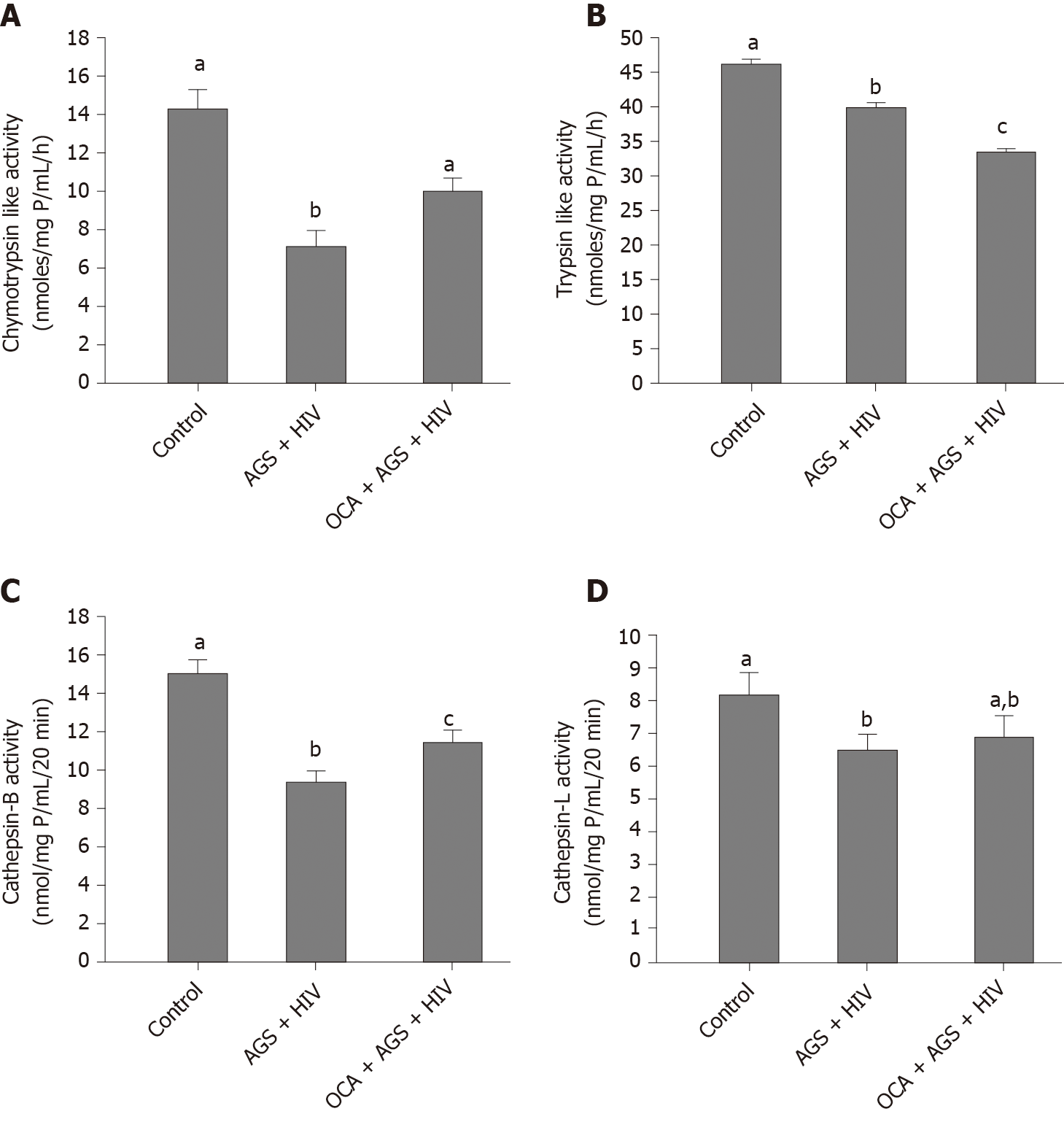
We measured the effects of OCA on accumulation of HIVgag RNA (RT-PCR) and p24 (western blot) induced by AGS + HIV exposure to RLW cells. In these experiments, OCA suppressed the treatment-induced HIVgag RNA levels by 50% (Figure 3A) and p24 –by 26% (Figure 3B and C). OCA also decreased the level of HIV DNA in RLW cells by about 60% (Figure 3D), with no integration of HIV DNA in cell genome. As has been shown by us before, the accumulation of HIV components in RLW cells is controlled by HIV-AGS-decreased lysosomal and proteasomal activities[6]. In fact, we observed partial restoration of chymotrypsin-like proteasome and cathepsin B and L lysosomal activities by OCA; however, OCA did not restore AGS-HIV-affected trypsin-like proteasome activity (Figure 4A-D).
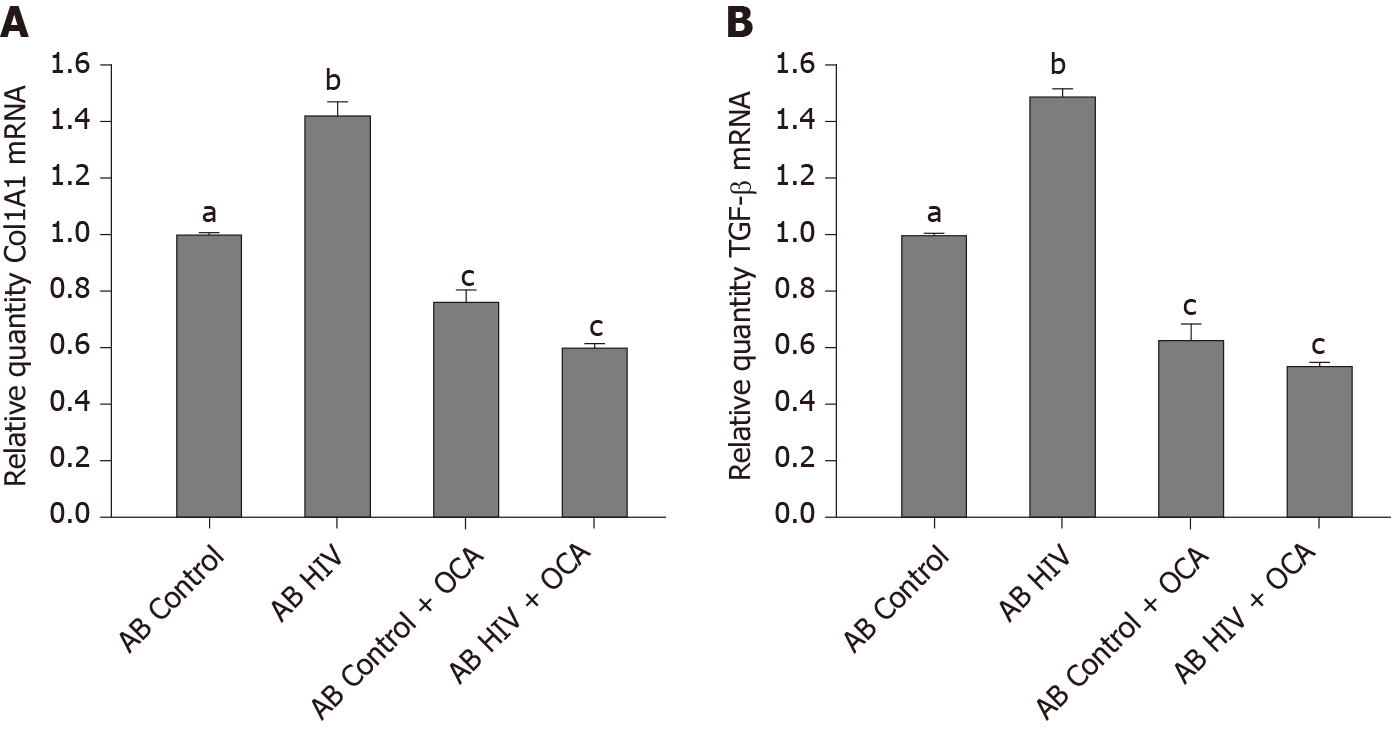
AB generated from uninfected (control) or HIV-infected RLW cells were exposed to LX2 cells, followed by measurement of Col1A1 and TGF-β mRNAs by RT-PCR. We found that HIV-containing apoptotic cells induced more prominent pro-fibrotic activation of HSC than uninfected apoptotic cells; OCA suppressed this pro-fibrotic HSC activation by uninfected AB and to most extend, by ABHIV engulfment (Figure 5A and B).
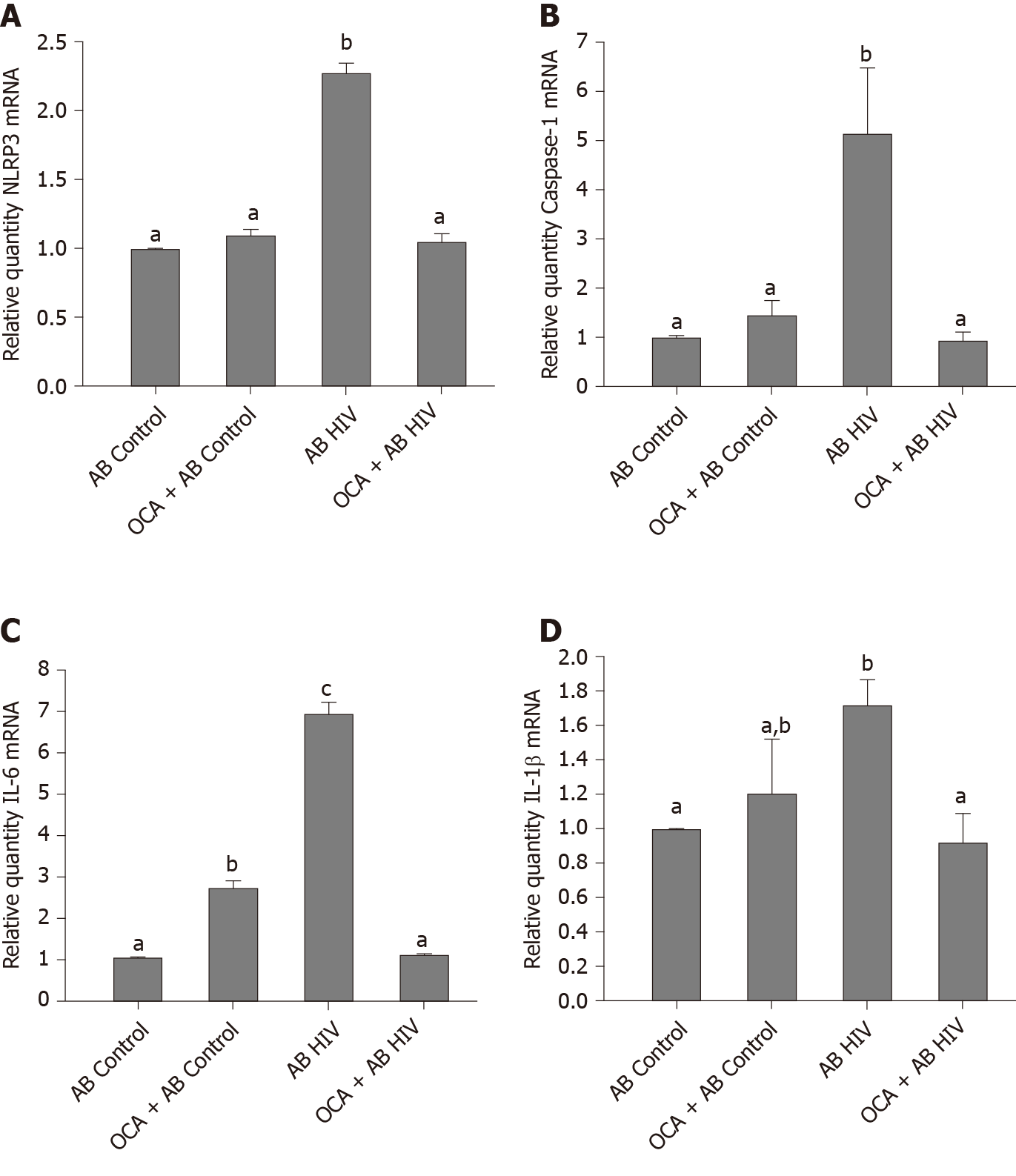
To characterize the effects of OCA on inflammasome activation in HSC, we measured NRLP3, caspase 1 and pro-inflammatory cytokines, IL-6 and IL-1β mRNA expression induced by engulfment of uninfected AB and HIV-containing AB, Up-regulation of NRLP3, caspase 1, IL-6 and IL-1 β levels by HIV-containing AB was successfully prevented by OCA treatment (Figure 6).
As previously shown, HIV accumulation induced by pre-exposure of cells to ethanol metabolites, mainly acetaldehyde produced by AGS, induces oxidative stress and apoptosis in hepatocytes. This is beneficial due to infected hepatocytes clearance before the integration of HIV DNA into human genome occurs. However, intensive hepatocyte apoptosis may have detrimental outcomes since HIV-containing apoptotic hepatocytes induce pro-fibrotic activation of HSC, thereby promoting fibrosis development[6]. Here, we investigated whether OCA protects from AGS-HIV-induced hepatocyte apoptosis, which causes HSC activation by ABHIV engulfment to drive liver fibrosis development.
In our model, OCA pre-treatment attenuated apoptosis (caspase 3 and PARP cleavage as well as cleaved cytokeratin 18 expression) in AGS-HIV-exposed liver cells. Unlike suppression of p53 expression on liver cells by OCA reported in HIV-non-infected cells by others[11], here, OCA mildly suppressed AGS-HIV-induced p53 mRNA. Apoptotic hepatocyte death was triggered by oxidative stress induced by AGS and HIV in CYP2E1-expressing RLW cells. In our hands, OCA indeed suppressed ROS production, thereby attenuating oxidative stress, which corroborated the data obtained on different models[17,18]. Here, the suppression of oxidative stress by OCA restores proteasome and lysosome functions, which increases the degradation of HIV proteins[19,20] and thus, diminishes the expression of HIV gag RNA and p24 gag protein in infected RLW cells. However, as we established before, the prevention of HIV and AGS- induced apoptotic hepatocyte death by exposure to pan-caspase inhibitor causes accumulation of cells with integrated HIV DNA[6], which is an unwanted event. Importantly, while OCA pre-treatment suppresses apoptosis in HIV-infected hepatocytes, there was no increase in hepatocytes expressing integrated HIV DNA.
In addition to beneficial effects of OCA on attenuation of HIV-AGS-induced hepatocyte death and reduction of HIV markers expression, OCA also reverses pro-fibrotic activation (based on Col1A1 and TGF-β mRNA levels) of HSC by engulfment of HIV-containing apoptotic hepatocytes. Similar event was observed by[10] on CCl4-injured mice, indicating that OCA controls HSC activation triggered via multiple mechanisms. One of this mechanisms contributing to liver fibrosis progression is an activation of inflammasome pathway in HSC[21]. In fact, we observed the reversing effect of OCA on this pathway in HSC, which supports anti-fibrotic activity of OCA. Thus, the suppression of pro-fibrotic activation by OCA in HSC is crucial for liver fibrosis development.
In fact, in vivo protection from early alcohol-induced liver damage by OCA has been already demonstrated on alcohol-fed uninfected mice without disclosing the mechanisms, by which this happened[22]. Furthermore, in monocytes from peripheral blood of HIV-infected patients, the expression of nuclear receptors, including FXR, was reported to be low[23], and the treatments with FXR agonists like OCA might play a beneficial role. Importantly, in HIV-infected alcohol-abused patients, the efficacy of OCA treatment has never been tested. Furthermore, these studies have not been done in the context of HIV-affected liver function. Our experiments provides in vitro evidence for protective effects of OCA from liver fibrosis progression induced by HIV and ethanol metabolism. The major limitation of this innovative study is that the results are currently based only on in vitro, but not in vivo experiments. Nevertheless, these in vitro experiments are necessary to characterize the exact mechanisms, by which OCA prevents HIV/ethanol metabolism-induced liver injury. These mechanisms are difficult to identify by in vivo studies, due to multiple triggers of liver fibrosis progression. We plan to confirm the in vitro effects of OCA in future by in vivo studies on humanized mice model since only these mice can be infected with human live HIV and fed the liquid ethanol-containing diet. Thus, our current in vitro study pioneers in justifying OCA inclusion to the treatment scheme of HIV-infected alcohol abusers with high risk of liver fibrosis development.
In conclusion, in HIV-infected hepatocytes exposed to continuously released acetaldehyde, OCA attenuates apoptotic death of infected cells and pro-fibrotic activation of HSC by engulfment of apoptotic HIV+ hepatocytes, thereby protecting from liver fibrosis development.
Due to frequent association of morbidity and mortality of human immunodeficiency virus (HIV)-infection with liver injury, the inclusion of drugs with anti-fibrotic activity to the treatment of people living with HIV (PLWH) is pathogenically important. Alcohol consumption is known to speed up liver fibrosis development. Previously, we have shown that the exposure of hepatocytes to HIV and ethanol metabolites causes high oxidative stress, impairs proteasomal and lysosomal functions leading to accumulation of HIV in these cells, which end-ups with apoptotic cell death and finally, promotes progression to liver fibrosis.
The combined exposure of hepatocytes to HIV and alcohol induces hepatotoxicity and pro-fibrotic activation of hepatic stellate cells (HSC). Since HIV replication in hepatocytes is abortive, it cannot be fully controlled by antiretroviral therapy (ART) and thus, to prevent liver fibrosis progression in alcohol-abused PLWH, ART should be combined with the drugs suppressing apoptosis without enhancing HIV DNA integration in hepatocytes and decreasing pro-fibrotic activation of liver non-parenchymal cells.
The objective of this study was to investigate whether obeticholic acid (OCA) prevents HIV/ethanol metabolism-induced activation of HSC by HIV+ apoptotic hepatocyte engulfment, thereby diminishing liver fibrosis.
The study was performed on hepatocyte-like Huh7.5-CYP (RLW) cells infected with HIV ADA and exposed to acetaldehyde-generating system (AGS) in the presence or absence of OCA. As an end-point, we have measured expression of HIV-related markers (HIVgagRNA-by real-time polymerase chain reaction (PCR), p24- by western blot, HIV DNA-by semi-nested PCR, integrated HIV DNA-by ddPCR) and non-HIV-related parameters (lysosomal and proteasomal activities, hepatocytes apoptosis). We also characterized pro-fibrotic activation and inflammasome induction in HSC (LX2 cells) by HIV-containing apoptotic hepatocytes internalization.
We found that OCA attenuated HIV-AGS-induced accumulation of HIVgagRNA, HIV DNA and p24. It suppressed ROS production, restored chymotrypsin-like proteasome activity as well as cathepsin B lysosome activity and decreased apoptosis in RLW cells. Exposure of HIV-containing apoptotic hepatocytes to OCA prevented activation of inflammasome and pro-fibrotic activation of HSC.
By suppressing oxidative stress and restoring proteasomal and lysosomal functions impaired by HIV and ethanol metabolism, OCA decreases accumulation of HIV in hepatocytes, leading to down-regulation of apoptosis in these cells. OCA also reverses pro-fibrotic and inflammasome-related activation of HSC triggered by engulfment of HIV-containing apoptotic hepatocytes, potentially contributing to suppression of liver fibrosis development.
Our in vitro studies are in the frame of pre-clinical characterization of anti-fibrotic effects of OCA in alcohol-exposed PLWH with a high risk of liver fibrosis development. At the next step, these in vitro effects will be confirmed by in vivo experiments on humanized mice infected with HIV and fed ethanol diet.
| 1. | Sherman KE, Rockstroh J, Thomas D. Human immunodeficiency virus and liver disease: An update. Hepatology. 2015;62:1871-1882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 2. | Sterling RK, Smith PG, Brunt EM. Hepatic steatosis in human immunodeficiency virus: a prospective study in patients without viral hepatitis, diabetes, or alcohol abuse. J Clin Gastroenterol. 2013;47:182-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 3. | Seth A, Sherman KE. Fatty liver disease in persons with HIV infection. Top Antivir Med. 2019;27:75-82. [PubMed] |
| 4. | van Welzen BJ, Mudrikova T, El Idrissi A, Hoepelman AIM, Arends JE. A Review of Non-Alcoholic Fatty Liver Disease in HIV-Infected Patients: The Next Big Thing? Infect Dis Ther. 2019;8:33-50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 5. | Ganesan M, Poluektova LY, Kharbanda KK, Osna NA. Liver as a target of human immunodeficiency virus infection. World J Gastroenterol. 2018;24:4728-4737. [PubMed] [DOI] [Full Text] |
| 6. | Ganesan M, New-Aaron M, Dagur RS, Makarov E, Wang W, Kharbanda KK, Kidambi S, Poluektova LY, Osna NA. Alcohol Metabolism Potentiates HIV-Induced Hepatotoxicity: Contribution to End-Stage Liver Disease. Biomolecules. 2019;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 7. | Shah RA, Kowdley KV. Obeticholic acid for the treatment of nonalcoholic steatohepatitis. Expert Rev Gastroenterol Hepatol. 2020;14:311-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 8. | Wang H, He Q, Wang G, Xu X, Hao H. FXR modulators for enterohepatic and metabolic diseases. Expert Opin Ther Pat. 2018;28:765-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 9. | Zhang DY, Zhu L, Liu HN, Tseng YJ, Weng SQ, Liu TT, Dong L, Shen XZ. The protective effect and mechanism of the FXR agonist obeticholic acid via targeting gut microbiota in non-alcoholic fatty liver disease. Drug Des Devel Ther. 2019;13:2249-2270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 10. | Zhou J, Huang N, Guo Y, Cui S, Ge C, He Q, Pan X, Wang G, Wang H, Hao H. Combined obeticholic acid and apoptosis inhibitor treatment alleviates liver fibrosis. Acta Pharm Sin B. 2019;9:526-536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 11. | Goto T, Itoh M, Suganami T, Kanai S, Shirakawa I, Sakai T, Asakawa M, Yoneyama T, Kai T, Ogawa Y. Obeticholic acid protects against hepatocyte death and liver fibrosis in a murine model of nonalcoholic steatohepatitis. Sci Rep. 2018;8:8157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 12. | Ganesan M, Dagur RS, Makarov E, Poluektova LI, Kidambi S, Osna NA. Matrix stiffness regulate apoptotic cell death in HIV-HCV co-infected hepatocytes: Importance for liver fibrosis progression. Biochem Biophys Res Commun. 2018;500:717-722. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 13. | Ganesan M, Natarajan SK, Zhang J, Mott JL, Poluektova LI, McVicker BL, Kharbanda KK, Tuma DJ, Osna NA. Role of apoptotic hepatocytes in HCV dissemination: regulation by acetaldehyde. Am J Physiol Gastrointest Liver Physiol. 2016;310:G930-G940. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 14. | Ganesan M, Tikhanovich I, Vangimalla SS, Dagur RS, Wang W, Poluektova LI, Sun Y, Mercer DF, Tuma D, Weinman SA, Kharbanda KK, Osna NA. Demethylase JMJD6 as a New Regulator of Interferon Signaling: Effects of HCV and Ethanol Metabolism. Cell Mol Gastroenterol Hepatol. 2018;5:101-112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | Ganesan M, Krutik VM, Makarov E, Mathews S, Kharbanda KK, Poluektova LY, Casey CA, Osna NA. Acetaldehyde suppresses the display of HBV-MHC class I complexes on HBV-expressing hepatocytes. Am J Physiol Gastrointest Liver Physiol. 2019;317:G127-G140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 16. | Thomes PG, Ehlers RA, Trambly CS, Clemens DL, Fox HS, Tuma DJ, Donohue TM. Multilevel regulation of autophagosome content by ethanol oxidation in HepG2 cells. Autophagy. 2013;9:63-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 17. | Zhu JB, Xu S, Li J, Song J, Luo B, Song YP, Zhang ZH, Chen YH, Xie DD, Yu DX, Xu DX. Farnesoid X receptor agonist obeticholic acid inhibits renal inflammation and oxidative stress during lipopolysaccharide-induced acute kidney injury. Eur J Pharmacol. 2018;838:60-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 18. | Zhang DG, Zhang C, Wang JX, Wang BW, Wang H, Zhang ZH, Chen YH, Lu Y, Tao L, Wang JQ, Chen X, Xu DX. Obeticholic acid protects against carbon tetrachloride-induced acute liver injury and inflammation. Toxicol Appl Pharmacol. 2017;314:39-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 70] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 19. | Lata S, Mishra R, Banerjea AC. Proteasomal Degradation Machinery: Favorite Target of HIV-1 Proteins. Front Microbiol. 2018;9:2738. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 20. | Wei BL, Denton PW, O'Neill E, Luo T, Foster JL, Garcia JV. Inhibition of lysosome and proteasome function enhances human immunodeficiency virus type 1 infection. J Virol. 2005;79:5705-5712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 96] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 21. | Inzaugarat ME, Johnson CD, Holtmann TM, McGeough MD, Trautwein C, Papouchado BG, Schwabe R, Hoffman HM, Wree A, Feldstein AE. NLR Family Pyrin Domain-Containing 3 Inflammasome Activation in Hepatic Stellate Cells Induces Liver Fibrosis in Mice. Hepatology. 2019;69:845-859. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 122] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 22. | Iracheta-Vellve A, Calenda CD, Petrasek J, Ambade A, Kodys K, Adorini L, Szabo G. FXR and TGR5 Agonists Ameliorate Liver Injury, Steatosis, and Inflammation After Binge or Prolonged Alcohol Feeding in Mice. Hepatol Commun. 2018;2:1379-1391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 86] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 23. | Renga B, Francisci D, D'Amore C, Schiaroli E, Carino A, Baldelli F, Fiorucci S. HIV-1 infection is associated with changes in nuclear receptor transcriptome, pro-inflammatory and lipid profile of monocytes. BMC Infect Dis. 2012;12:274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: MD DEA S-Editor: Zhang H L-Editor: A P-Editor: Ma YJ