Published online Oct 27, 2020. doi: 10.4254/wjh.v12.i10.850
Peer-review started: April 29, 2020
First decision: July 5, 2020
Revised: July 15, 2020
Accepted: August 24, 2020
Article in press: August 24, 2020
Published online: October 27, 2020
Processing time: 177 Days and 4.8 Hours
Hepatitis C virus (HCV) infection may affect lipid metabolism by enhancing the circulating levels of inflammatory cytokines, together with its impact on endothelial function.
To evaluate the potential correlation of changes in lipid profile, carotid intima-media thickness (CIMT), and ankle-brachial index with the severity of fibrosis, grades of esophageal varices (EVs), and fibrosis indices.
The study included 240 subjects who were divided into 3 groups; group 1 (n = 90, HCV-related cirrhotic patients with EVs), group 2 (n = 90, HCV-related cirrhotic patients without EVs), and group 3 (n = 60, served as the healthy control group). All patients underwent routine laboratory tests, including a lipid profile assay. Low-density lipoproteins (LDL)/platelet count and platelet/splenic diameter ratios were calculated. Abdominal ultrasonography, CIMT by carotid Doppler, bedside ankle-brachial index (ABI), liver stiffness measurement, and upper gastrointestinal endoscopy were performed.
Multivariate logistic regression revealed that very-low-density lipoprotein (VLDL) (β = 0.988, odds ratio 2.5, P = 0.001), LDL/platelet count ratio (β = 1.178, odds ratio 3.24, P = 0.001), CIMT (β = 1.37, odds ratio 3.9, P = 0.001), and ABI (β = 2.3, odds ratio 5.9, P = 0.001) were the key variables associated with significant fibrosis, EVs and endothelial dysfunction. CIMT and LDL/platelet count ratio were predictive of advanced fibrosis and EVs at cutoff values of 1.1 mm and 1 mm, respectively, with an area under the curve (AUC) of 0.966 and 0.960 (P = 0.001), while VLDL and ABI at a cutoff of 16.5 mg/dL and 0.94 were predictive of advanced fibrosis and EVs with an AUC of 0.891 and 0.823, respectively (P = 0.001).
CIMT, ABI, VLDL, LDL/platelet count ratio are good non-invasive predictors of advanced fibrosis, presence of EVs, and endothelial dysfunction in liver cirrhosis.
Core Tip: Hepatitis C virus (HCV) infection may affect lipid metabolism by enhancing the circulating levels of inflammatory cytokines. HCV may induce endothelial dysfunction. Carotid intima-media thickness, low-density lipoproteins (LDL)/platelet count ratio, ankle-brachial index, and very LDL were predictive of advanced fibrosis, esophageal varices, and endothelial dysfunction at cutoff values of 1.1 mm, 1, 0.94, and 16.5 mg/dL, respectively. Novel markers provide information beyond the previous traditional markers such as platelet count, FIB-4, and platelet/splenic diameter ratio with additional data regarding endothelial dysfunction and subclinical atherosclerosis.
- Citation: Hanafy AS, Basha MAK, Wadea FM. Novel markers of endothelial dysfunction in hepatitis C virus-related cirrhosis: More than a mere prediction of esophageal varices. World J Hepatol 2020; 12(10): 850-862
- URL: https://www.wjgnet.com/1948-5182/full/v12/i10/850.htm
- DOI: https://dx.doi.org/10.4254/wjh.v12.i10.850
Hepatitis C virus (HCV) infection represents a major public health burden with an estimated global prevalence of 2.8% resulting in more than 185 million infected patients[1]. Prompt recognition of vascular changes in patients with significant liver fibrosis is required for directing therapy and follow-up against both cardiovascular disease and esophageal varices (EVs).
Chronic HCV infection is associated with a chronic inflammatory state leading to a disproportion of pro-inflammatory/anti-inflammatory cytokines ratio. Also, lipid abnormalities, insulin resistance (IR), and increased risk of atherosclerosis have been described in chronic HCV[2,3].
HCV may trigger atherosclerosis through the production of intracellular adhesion molecules, anti-endothelial antibodies, oxidative stress generation, and IR[4,5]. Increased carotid intima-media thickness (CIMT) represents an initial ultrasonographic sign of atherosclerosis that can be easily evaluated at the bedside. Previously, HCV RNA was isolated from carotid plaques from patients infected with HCV[6,7].
HCV is a hybrid molecule composed of viral and lipoprotein components (lipoviroparticles); the latter resemble very low-density lipoproteins (VLDLs) and low-density lipoproteins (LDLs)[8], and conjugate with cell surface receptors mainly scavenger receptor class B member 1 protein and the LDL receptor[9], the latter binds to either the lipoprotein component of the viral particle or the hyper-variable region 1 of glycoprotein E2[10].
HCV infection causes a defect in intrahepatic cholesterol synthesis due to its employment in viral replication, with a later decrease in the available cholesterol for peripheral delivery via VLDL and this stimulates more expression of LDL receptors and an increase in LDL uptake by the liver accounting for the decreased serum LDL; therefore, sustained virological response (SVR) may cause a rebound increase in lipid levels[11]. VLDL level may be low in advanced liver diseases due to decreased synthesis and therefore it can be used as a marker of advanced fibrosis[12,13].
In addition to predicting EVs and fibrosis severity, the objective of this study was to determine predictive markers of vascular changes and endothelial dysfunction in HCV-related cirrhosis.
Evaluation of the association of changes in lipid profile, CIMT and ankle-brachial index (ABI) with the severity of fibrosis, grades of EVs, and fibrosis indices.
This cross-sectional case-control study was conducted in the Gastroenterology and Hepatology Clinic, Department of Internal Medicine, Faculty of Medicine, Zagazig University Hospitals during the period from November 2018 to December 2019. The research protocol (IRB-255-2018) was accepted by the Zagazig Faculty of Medicine Ethical Committee.
All procedures were carried out under the Zagazig University's ethical principles and in compliance with the Helsinki declaration and its more recent modifications. Informed consent was obtained from each patient who participated in the study.
The study included chronic HCV infected patients (n = 180) and 60 healthy subjects as a control group, all were matched for age, sex, and body mass index.Diagnosis of liver cirrhosis was based on clinical, laboratory, ultrasonographic, and FibroScan features. Eligible patients obtained a diagnosis of liver cirrhosis secondary to HCV infection proven by a positive anti-HCV test and HCV-RNA in serum, and were divided into 2 groups: Group 1 which included 90 cirrhotic patients complicated with EVs and group 2 which included 90 cirrhotic patients without EVs. Group 3 included the control subjects.
Smoking, obesity, patients with other diseases that may alter serum lipid levels such as diabetes, non-alcoholic fatty liver disease or chronic alcohol consumption, any patients who had previously received anti-viral therapy for HCV or had been cured of HCV which may induce a rebound increase in serum lipids, lipid-lowering medications, recipients of solid organ transplantation and patients who had refused to participate in the study.
Routine laboratory tests were performed including liver and kidney function tests, full blood count, and coagulation profile. LDL-C was calculated using the Friedewald formula: LDL-C (mg/dL) = Total cholesterol–(HDL-C)–(triglycerides/5)[14]. The LDL/platelet count ratio was also calculated. VLDL cholesterol was estimated by dividing the TGs/5if the TGs were lower than 450 mg/dL[15].
The ultrasonographic features of liver cirrhosis or the presence of ascites were documented. Criteria for portal hypertension were defined as portal vein diameter greater than 13 mm, splenic bipolar diameter greater than 130 mm or the presence of portal venous collaterals[16].
Common carotid arteries were evaluated on both sides by an experienced radiologist who was blinded to clinical data using B-mode duplex ultrasound with a 7.5MHz linear probe (Siemens G60®). CIMT was measured from the intima lumen interface to the media adventitia interface; with a value > 0.9 mm considered abnormal. Three measures were obtained on either side; the mean CIMT was defined as the mean right and left CIMT[17].
Liver stiffness measurement: Liver stiffness measurement was performed by an experienced physician who was blinded to the clinical data of the patients using FibroScan®. Fibrosis stages F0-1; F2; F3 and F4 or cirrhosis were defined by the spectrum of liver stiffness values 2.5-7, 7-9.5, 9.5-12.5 and > 12.5 kPa, respectively[18].
Fibrosis index-4: This was calculated by the following equation using 4 factors: [age (year)×aspartate aminotransferase (U/L)]/[platelet count (PLT) (109/L)]×[alanine aminotransferase (U/L)]. A fibrosis index-4 (FIB-4) score < 1.45 displayed a negative predictive value of 90% for advanced fibrosis, however, a FIB-4 > 3.25 had a 97% specificity and a positive predictive value of 65% for advanced fibrosis[19].
PLT/splenic diameter ratio: The PLT/splenic diameter (PLT/SD) ratio was calculated by dividing the number of platelets (μL) by the maximum bipolar diameter of the spleen in millimeters, detected by abdominal ultrasound.
EVs were diagnosed and graded from grade I to grade IV, using the Paquet grading system[20].
Endothelial dysfunction can be evaluated by flow-mediated dilatation in the brachial artery using high-resolution ultrasound or simply by bedside ABI which is considered an easy and cost-effective method for assessing endothelial dysfunction[21].The patient remained in the supine position for 5 min and the blood pressure cuff was applied to the arm and lower calf, then the stethoscope was used to measure the systolic pressure in the brachial and dorsalis pedis arteries of both sides. ABI was calculated by dividing the highest value of pressure in the dorsalis pedis arteries by the highest brachial pressure. The normal ABI is over 1 and the cutoff value to diagnose peripheral arterial disease is ≤ 0.90 at rest with a sensitivity of 95% and specificity of 100%, values > 1.40 suggest arteriosclerosis mainly seen in diabetes or chronic renal failure[22].
All data were statistically analyzed using SPSS 20 for Windows (SPSS Inc., Chicago, IL, United States). Quantitative data were expressed as the mean ± SD. Qualitative data were expressed as absolute frequencies (number) and (percentage). The F test was performed to compare between more than two groups of normally distributed variables. LSD with Bonferroni correction was applied to detect the difference between groups.
Spearman’s correlation coefficient (r) was performed for ordinal variables and Pearson correlation for continuous variables. Logistic regression analysis was performed by forwarding selection to identify variables independently associated with advanced fibrosis and endothelial dysfunction. All variables with P < 0.05 were considered statistically significant.
Receiver operating characteristic curves were plotted and the area under the curve (AUC) was calculated, the performance of the cutoff value was judged by calculation of Youden’s J value; values near 1 indicated good performance (J = sensitivity + specificity – 1). Sensitivity/specificity and positive/negative predictive values for the non-invasive diagnosis of fibrosis, EVs, and endothelial dysfunction were assessed considering liver stiffness as the reference for stages of fibrosis.
The current study included 240 subjects, and the baseline demographic, laboratory, and endoscopic findings of all subjects are summarized in Table 1. A highly significant statistical difference regarding serum transaminases and platelet count was found among the groups with the key significant difference between group 1 (cirrhosis with EVs) and group 2 (cirrhosis without EVs), and group 1 (cirrhosis with EVs) and group 3 (control group) (P = 0.001 and 0.001, respectively).
| Variable | Liver cirrhosis with EVs | Liver cirrhosis without EVs | Control group | P value1 |
| n | 90 | 90 | 60 | - |
| Sex (M/F) | 62/28 | 60/30 | 40/20 | 0.4 |
| Age (yr) | 49.6 ± 8.4 | 47.7 ± 9.7 | 46 ± 6.7 | 0.32 |
| BMI (kg/m2) | 26.3 ± 2.1 | 25.8 ± 1.3 | 26.6 ± 0.9 | 0.21 |
| ABI | 0.94 ± 0.09 | 1.08 ± 0.13 | 1.16 ± 0.11 | 0.001 |
| AST (IU/L) | 53.7 ± 10.2 | 43.7 ± 5.6 | 31.1 ± 10.8 | 0.001 |
| ALT (IU/L) | 47.2 ± 7.8 | 36.2 ± 8.5 | 30.6 ± 7.7 | 0.001 |
| Platelet count × 103 | 79.2 ± 21.2 | 189.3 ± 43.2 | 197.7 ± 18.7 | 0.001 |
| Total cholesterol (mg/dL) | 177.6 ± 18.8 | 199.2 ± 27.4 | 210.4 ± 12.6 | 0.01 |
| HDL (mg/dL) | 39.5 ± 4.3 | 44.6 ± 7.1 | 35.4 ± 5.3 | 0.004 |
| TGs (mg/dL) | 135.7 ± 14.4 | 168.8 ± 12.8 | 180.8 ± 8.4 | 0.01 |
| VLDL (mg/dL) | 16.6 ± 4.3 | 26.5 ± 5.3 | 25 ± 5.7 | 0.001 |
| LDL (mg/dL) | 105.8 ± 14.5 | 128.9 ± 20.6 | 138.9 ± 16.5 | 0.03 |
| FIB-4 | 4.97 ± 1.8 | 1.54 ± 0.47 | 1.51 ± 0.3 | 0.001 |
| Platelets/SD ratio | 449 ± 167 | 1375 ± 380 | 1702 ± 238 | 0.001 |
| LDL/platelet count ratio | 1.41 ± 0.4 | 0.71 ± 0.21 | 0.75 ± 0.09 | 0.001 |
| Child-Pugh class (n) | A (26), B (46), C (18) | A (88), B (2) | Non-cirrhotic | - |
| Endoscopy | - | - | - | - |
| EVs | - | - | - | - |
| Grade I-II, n (%) | 32 (35.5) | - | - | - |
| Grade III, n (%) | 42 (46.7) | - | - | - |
| Grade IV, n (%) | 16 (17.8) | - | - | - |
| 2Fundal varix, n (%) | 12 (13.3) | - | - | - |
The total cholesterol and LDL levels were significantly lower in group 1 with a significant difference between groups 1 & 2, and 1 & 3 (P = 0.001 and 0.001, respectively).
VLDL was significantly lower in group 1 with a significant difference between groups 1 & 2, and 1 & 3 (P = 0.001 and 0.001, respectively) and between group 2 & 3 (P = 0.001). Also, the LDL/platelet count ratio was significantly higher in group 1 with a significant difference between groups 1 & 2, and 1 & 3 (P = 0.001 and 0.001, respectively).
FIB-4 values were significantly higher in group 1 with a significant difference between group 1 & 2, and 1 & 3 (P = 0.001 and 0.001, respectively) as presented in Table 1. In addition, ABI was significantly lower in group 1 when compared with group 2 and 3 (Table 1).
Carotid-intima media thickness was significantly higher in group 1 when compared with group 2 and 3. These results are presented in Figure 1 and Table 2. SD and FibroScan readings (kPa) were significantly higher in cirrhotic patients with EVs with a significant difference between group 1 & 2, and 1 & 3 (P = 0.001 and 0.001), however, the PLT/SD ratio was significantly lower in the same group as presented in Table 2. The cutoff value of the PLT/SD ratio associated with advanced fibrosis and presence of EVs was 872 with a sensitivity of 100%, specificity of 94%, AUC of 0.991, P = 0.001, and Youden's J value of 0.94.
| Variable | Liver cirrhosis with EVs | Liver cirrhosis without EVs | Control group | P value |
| Splenic diameter | 182.9 ± 21.4 | 141 ± 12.8 | 111.2 ± 9.7 | 0.003 |
| Platelets/SD ratio | 449 ± 166 | 1374.9 ± 380 | 1701 ± 239 | 0.001 |
| FibroScan (kPa) | 24.4 ± 6.02 | 14.8 ± 1.9 | 5.2 ± 0.82 | 0.001 |
| CIMT (mm) | 1.2 ± 0.13 | 0.69 ± 0.14 | 0.62 ± 0.11 | 0.001 |
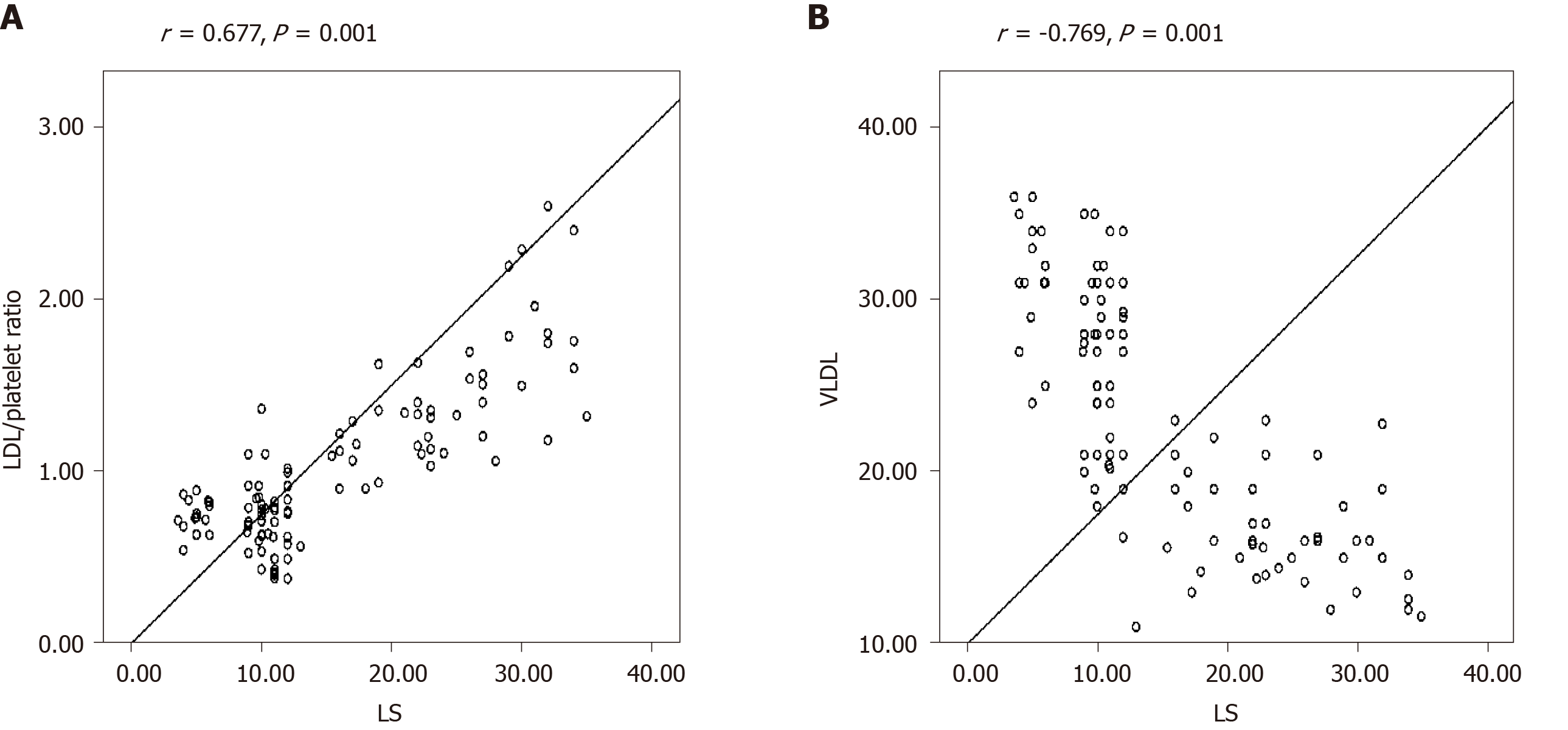
Liver stiffness significantly correlated with platelet count (r = -0.615, P = 0.001), VLDL (r = -0.619, P = 0.001), SD (r = 0.534, P = 0.001), FIB-4 (r = 0.588, P = 0.001), CIMT (0.712, P = 0.001), LDL/PLT ratio (r = 0.677, P = 0.001), and ABI (r = -0.546, P = 0.001).
An increasingly significant direct correlation was detected between the LDL/PLT ratio, LS (r = 0.677, P = 0.001) and FIB4 (r = 0.763, P = 0.001). On the other hand, a high significant negative correlation was detected between VLDL and LS (r = -0.769, P = 0.001) as well as FIB4 (r = -0.533, P = 0.001) as shown in Figure 2 and Supplementary Figure 1.
Multivariate logistic regression was carried out to identify variables independently associated with fibrosis severity, EVs and endothelial dysfunction and revealed that VLDL (β = 0.988, odds ratio 2.5, P = 0.001), LDL/PLT ratio (β = 1.178, odds ratio 3.24, P = 0.001), CIMT (β = 1.37, odds ratio 3.9, P = 0.001), and ABI (β = 2.3, odds ratio 5.9, P = 0.001) were independently associated with fibrosis severity.
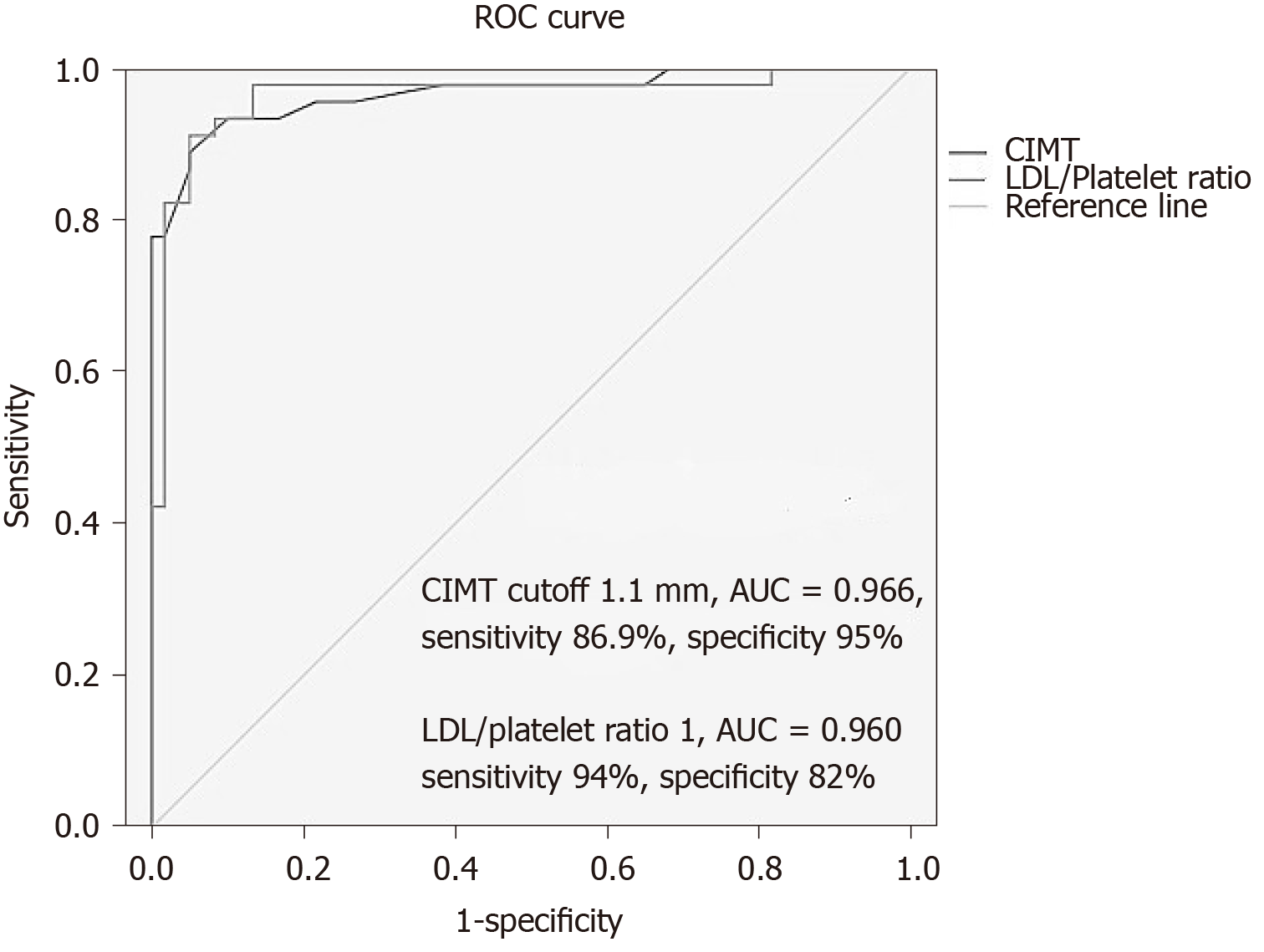
CIMT and LDL/PLT ratio were predictive of advanced fibrosis, EVs and endothelial dysfunction at cutoff values 1.1 mm and 1, respectively, with an AUC of 0.966 and 0.960, 95%CI(0.913-1 and 0.916-1), sensitivity of 86.9% and 94%, specificity of 95% and 82% as presented in Figure 3, and Youden’s J value = 0.819 and 0.76, respectively.
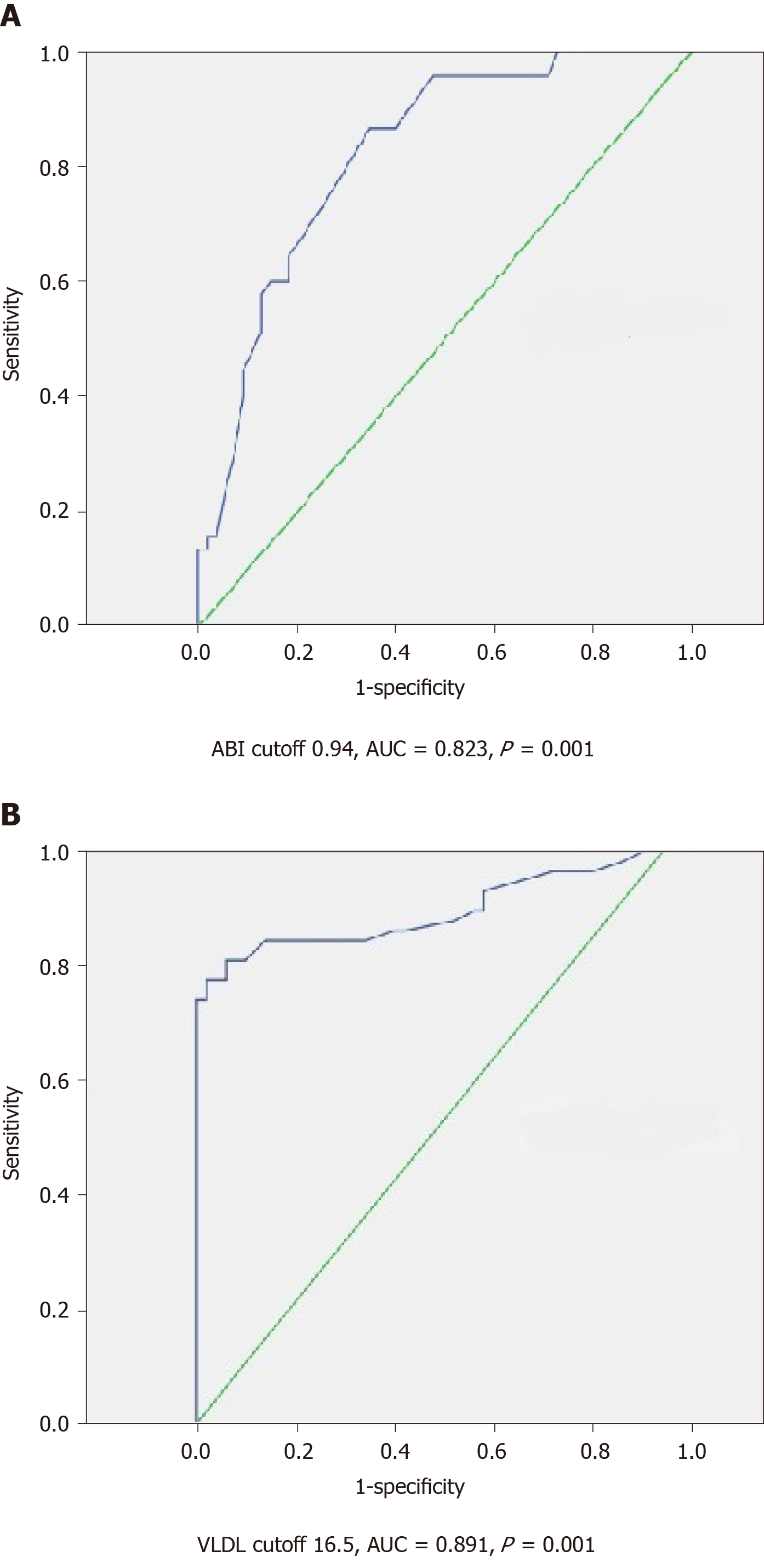
VLDL and ABI cutoff values predictive of advanced fibrosis, EVs and endothelial dysfunction were 16.5 mg/dL and 0.94 with an AUC of 0.891 and 0.823, sensitivity of 74.1% and 90.5%, and specificity of 100% and 92%, and corresponding to Youden’s J value = 0.741 and 0.82, respectively, as presented in Figure 4.
When patients were categorized based on the determined cutoff values of VLDL, CIMT, ABI, and the LDL/platelet ratio; they displayed an extremely significant discriminating ability for lipid profile, platelet count, SD, FIB-4 and the PLT/SD ratio as presented in Table 3.
| Variable | VLDL | CIMT | LDL/platelet ratio | ABI | ||||||||
| Cutoff value | Below 16.5 | Above 16.5 | P value | Below 1.1 | Above 1.1 | P value | Below 1 | Above 1 | P value | Below 0.94 | Above 0.94 | P value |
| n | 76 | 164 | - | 96 | 144 | - | 98 | 142 | - | 70 | 170 | - |
| Sex (M/F) | 56/20 | 106/58 | 0.2 | 64/32 | 98/46 | 0.86 | 66/32 | 96/46 | 0.83 | 47/23 | 115/55 | 0.45 |
| Age (yr) | 50.3 ± 10 | 48.5 ± 8.3 | 0.3 | 49.6 ± 8.6 | 48.7 ± 9 | 0.56 | 49.8 ± 8.8 | 48 ± 9 | 0.78 | 51.5 ± 8.9 | 48.2 ± 7.4 | 0.2 |
| AST (IU/L) | 46 ± 11.6 | 38.4 ± 12.1 | 0.01 | 47.5 ± 11.8 | 34.9 ± 8.4 | 0.01 | 52 ± 11 | 35 ± 9 | 0.001 | 49 ± 12 | 38 ± 11 | 0.03 |
| ALT (IU/L) | 45 ± 8.4 | 37.5 ± 8.5 | 0.03 | 46 ± 8 | 35.4 ± 7 | 0.03 | 45 ± 7.8 | 34 ± 5.6 | 0.02 | 43 ± 8 | 32 ± 8 | 0.047 |
| Platelet count × 103 | 83.6 ± 47 | 165.5 ± 62.2 | 0.001 | 82.5 ± 41.4 | 182 ± 40 | 0.001 | 81 ± 26 | 188 ± 41 | 0.001 | 87 ± 23 | 160 ± 61 | 0.001 |
| Total cholesterol | 166 ± 21 | 198 ± 24 | 0.02 | 170 ± 18 | 200.5 ± 26 | 0.034 | 168 ± 26 | 197 ± 28 | 0.04 | 173 ± 27 | 196 ± 25 | 0.023 |
| Triglycerides (mg/dL) | 135.7 ± 15 | 164 ± 19 | 0.03 | 137 ± 15 | 168 ± 15 | 0.032 | 140 ± 19 | 168 ± 16 | 0.045 | 143 ± 18 | 162 ± 24 | 0.01 |
| VLDL (mg/dL) | 14.6 ± 1.5 | 26.2 ± 5.6 | 0.001 | 17 ± 4.9 | 28 ± 7 | 0.001 | 15 ± 4 | 32 ± 6 | 0.001 | 17 ± 5 | 25 ± 12 | 0.03 |
| LDL (mg/dL) | 106 ± 15 | 126 ± 21 | 0.001 | 102 ± 13 | 130 ± 20 | 0.023 | 110 ± 17 | 122 ± 20 | 0.06 | 109 ± 19 | 129 ± 22 | 0.021 |
| HDL (mg/dL) | 40 ± 4 | 43 ± 8 | 0.89 | 39 ± 5 | 42 ± 8.2 | 0.45 | 40 ± 5 | 42 ± 8 | 0.82 | 39 ± 4 | 42 ± 8 | 0.34 |
| FIB-4 | 5.03 ± 1.8 | 2.2 ± 1.6 | 0.001 | 5 ± 2 | 1.7 ± 0.6 | 0.001 | 4.9 ± 1.9 | 1.6 ± 0.5 | 0.001 | 4.2 ± 2 | 2.4 ± 1.9 | 0.01 |
| LDL/platelet count ratio | 1.5 ± 0.48 | 0.84 ± 0.3 | 0.001 | 1.4 ± 0.4 | 0.75 ± 0.21 | 0.001 | 1.5 ± 0.3 | 0.7 ± 0.15 | 0.001 | 1.24 ± 0.21 | 0.91 ± 0.34 | 0.001S |
| Splenic diameter (mm) | 186.3 ± 25.4 | 141.5 ± 24 | 0.001 | 185.4 ± 20 | 133 ± 17 | 0.001 | 181 ± 24 | 122 ± 11 | 0.001 | 171 ± 29 | 136 ± 13 | 0.001 |
| Platelets/SD | 492.6 ± 76.2 | 1237.7 ± 502 | 0.001 | 463.3 ± 43 | 1400 ± 410 | 0.001 | 471 ± 213 | 1440 ± 402 | 0.001 | 678 ± 298 | 1173 ± 554 | 0.001 |
CIMT > 1.1 mm, LDL/PLT ratio > 1, and VLDL < 16.5 were associated with lower ABI values and in this way endothelial dysfunction (P = 0.001) when compared with other non-invasive tools. Also, CIMT > 1.1 mm, LDL/PLT ratio > 1, ABI < 0.94 and VLDL < 16.5 showed comparable results for liver stiffness and were more efficient in identifying EVs (P = 0.001) and equally large EVs (P = 0.048) when compared with traditional tools such as FIB-4 and the platelets/SD ratio as shown in Table 4.
| Cutoff value | PLT/SD ratio above 872 | FIB-4 above 3.25 | ABI below 0.94 | LDL/PLT ratio above 1 | CIMT above 1.1 | VLDL below 16.5 | P value |
| n | 90 | 90 | 70 | 98 | 96 | 76 | |
| LS (kPa) | 24.4 ± 6.2 | 26. 5 ± 5.2 | 22 ± 8.2 | 23 ± 7 | 24.6 ± 6.7 | 25.3 ± 6.3 | F = 1.87, P = 0.117 |
| ABI | 1.08 ± 0.09 | 1.09 ± 0.1 | 0.887 ± 0.04 | 0.93 ± 0.1 | 0.88 ± 0.1 | 0.96 ± 0.08 | F = 30, P = 0.001 |
| Endoscopy | - | - | - | - | - | - | - |
| EVs, n (%) | 82/90 (91.1) | 56/90 (62.2) | 65/70 (92.9) | 92/98 (94) | 90/96 (93.8) | 68/76 (89.5) | P = 0.001 |
| Grade I-II, n (%) | 26/32 (81.3) | 14/32 (35) | 20/32 (62.5) | 24/32 (75) | 24/32 (75) | 10/32 (31.3) | χ2 = 13.3, P = 0.009 |
| Grade III, n (%) | 32/42 (76.2) | 30/42 (71.4) | 30/42 (71.4) | 42/42 (100) | 40/42 (95) | 34/42 (81) | χ2= 7.67, P = 0.104 |
| Grade IV, n (%) | 14/16 (87.5) | 6/16 (37.5) | 9/16 (56.3) | 16/16 (100) | 16/16 (100) | 12/16 (75) | χ2= 9.55, P = 0.048 |
| Fundal varix, n (%) | 10/12 (83.3) | 6/12 (50) | 6/12 (50) | 10/12 (83) | 10/12 (83) | 12/12 (100) | χ2= 3.47,P = 0.48 |
Compensated cirrhosis may be difficult to differentiate from chronic hepatitis[23,24]. Accurate assessment of the fibrosis stage requires screening for complications mainly EVs to commence appropriate treatment[25].
Tools to estimate the extent and severity of liver fibrosis may be invasive such as liver biopsy, or non-invasive such as serological tests and imaging[26]. Liver biopsy is the best available standard modality of reference, but it has some limits and it is refused by most patients[27,28].
Could endothelial dysfunction be linked to histological severity and EVs development in HCV-related cirrhosis? This was the research question in the current study, which revealed that cirrhotic patients with and without EVs experienced significantly more reduced levels of total cholesterol, triglycerides, LDL, VLDL and several studies determined the changes in the lipid profile of patients with chronic liver disease and correlated them with the severity of liver disease. Abbasi et al[29] stated that serum cholesterol and triglycerides levels were inversely proportional to the histological severity. Ghadir et al[30] and Boemeke et al[31] observed a significant decrease in LDL, triglyceride, VLDL, and total cholesterol in cirrhotic patients compared with controls.
In advanced cirrhosis, IR is more common and associated with dyslipidemia with enhanced systemic inflammation[32,33]. There are no available reports that link lipid profile changes, CIMT, and ABI as non-invasive tools to the presence and size of EVs.
The current study is the first to describe the capability of predicting the presence and grading of EVs using lipid profile, CIMT, and ABI and to link the emergence of EVs with underlying endothelial dysfunction that may increase morbidity through an added cardiovascular risk.
In a study which enrolled patients with HCV and liver cirrhosis, the CIMT and epicardial fat thickness were significantly increased in the cirrhotic and non-cirrhotic HCV groups when compared with the control group, and CIMT and epicardial fat thickness were significantly increased with the progression of Child's class, spleen span, portal vein diameter and negatively associated with PLT[34].
Another study showed that CIMT was significantly higher in HCV-positive patients (1.04) than in HCV-negative patients (0.71) with more frequent plaque formation and therefore chronic HCV was an independent risk factor for stroke[35]. Patients with chronic liver disease and cirrhosis have more elevated risks of acute coronary syndrome and peripheral arterial disease than those without chronic liver disease and cirrhosis[36,37] .
In another study conducted in patients with HCV-related liver cirrhosis without a previous history of cerebrovascular disease, cardiac and peripheral vascular diseases, a decrease in the brachial-ankle pulse wave velocity was reported to be directly in proportion to the severity of cirrhosis (F = 4.90, P < 0.05)[38].
Due to the proven lipid changes in cirrhotic patients and thrombocytopenia as a well-known non-invasive predictor of liver cirrhosis and EVs; a new promising non-invasive predictor (LDL/PLT ratio) was calculated, and our results confirmed that this ratio was significantly higher in cirrhotic patients complicated with EVs (P < 0.001).
Using multivariate logistic regression to detect variables independently associated with significant fibrosis, EVs, and endothelial dysfunction; CIMT, VLDL, LDL/PLT ratio, and ABI were the most significant variables.
CIMT > 1.1 mm, LDL/PLT ratio > 1,VLDL < 16.5 mg/d, and ABI < 0.94 were significantly associated with higher liver stiffness values, FIB4, SD and increased incidence of larger varices (P < 0.001) with a significant discriminating ability for the degree of liver stiffness and grades of EVs with the advantage of providing information on endothelial dysfunction assessed by ABI when they were compared with the traditional non-invasive tools such as FIB-4 and PLT/SD ratio.
The limitation of the current study is that it was conducted in a single center and specific cardiac investigations should be performed to diagnose cardiovascular disease risk such as electrocardiography, echocardiography and coronary CT angiography which need to be conducted in high risk cirrhotic patients in future studies.
In conclusion, based on the current results, this may offer the chance for these markers to serve as non-invasive predictors of cirrhosis, EVs and endothelial dysfunction in this category of patients. The study raised an issue worthy of research, as patients with advanced fibrosis and larger varices had higher CIMT and lower ABI; therefore, placing these patients at an increased cardiovascular risk added to the risk of variceal bleeding; thus, prevention and treatment should be discussed in other studies.
Hepatitis C virus (HCV) infection may affect lipid metabolism by enhancing the circulating levels of inflammatory cytokines. HCV may induce endothelial dysfunction.
We believe that there is a potential correlation between the changes in lipid profile, carotid intima-media thickness (CIMT) and ankle-brachial index with the severity of fibrosis, grades of esophageal varices (EVs), and fibrosis indices.
To identify predictive markers of vascular changes and endothelial dysfunction in HCV-related cirrhosis
HCV infected cirrhotic patients with and without EVs were evaluated by routine laboratory tests, including lipid profile assay, abdominal ultrasonography, carotid intima-media thickness (CIMT) by carotid Doppler, bedside ankle-brachial index (ABI), liver stiffness measurement, and upper gastrointestinal endoscopy and compared to the healthy control group. Logistic regression analysis was performed to identify variables independently associated with advanced fibrosis and endothelial dysfunction.
CIMT, low-density lipoproteins (LDL)/platelet ratio, ABI, and very LDL (VLDL) were predictive of advanced fibrosis, EVs and endothelial dysfunction. They were effective at cutoff values of 1.1 mm, 1, 0.94, and 16.5 mg/dL, respectively.
CIMT, ABI, VLDL, and LDL/platelet count ratio are good non-invasive predictors of advanced fibrosis, presence of EVs, and endothelial dysfunction in liver cirrhosis.
The proposed markers serve as non-invasive predictors of cirrhosis, EVs and endothelial dysfunction, and patients with advanced fibrosis and larger varices had higher CIMT and lower ABI consequently, they bear an increased cardiovascular risk added to the risk of variceal bleeding. The study was designed and validated in a single-center. External, prospective validation is required to determine the widespread applicability and utility of this model.
Special thanks to Zagazig University's main Lab and Diagnostic Radiology Department for their help in this research.
| 1. | MohdHanafiah K, Groeger J, Flaxman AD, Wiersma ST. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57:1333-1342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1926] [Cited by in RCA: 1807] [Article Influence: 139.0] [Reference Citation Analysis (2)] |
| 2. | Negro F, Forton D, Craxì A, Sulkowski MS, Feld JJ, Manns MP. Extrahepatic morbidity and mortality of chronic hepatitis C. Gastroenterology. 2015;149:1345-1360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 276] [Article Influence: 25.1] [Reference Citation Analysis (2)] |
| 3. | Lauer GM, Walker BD. Hepatitis C virus infection. N Engl J Med. 2001;345:41-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2159] [Cited by in RCA: 1968] [Article Influence: 78.7] [Reference Citation Analysis (4)] |
| 4. | Lindenbach BD. Measuring HCV infectivity produced in cell culture and in vivo. Methods MolBiol. 2009;510:329-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 5. | Jacobson Brown PM, Neuman MG. Immunopathogenesis of hepatitis C viral infection: Th1/Th2 responses and the role of cytokines. ClinBiochem. 2001;34:167-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 64] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 6. | Boddi M, Abbate R, Chellini B, Giusti B, Giannini C, Pratesi G, Rossi L, Pratesi C, Gensini GF, Paperetti L, Zignego AL. Hepatitis C virus RNA localization in human carotid plaques. J ClinVirol. 2010;47:72-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 117] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 7. | Barone M, Viggiani MT, Amoruso A, Schiraldi S, Zito A, Devito F, Cortese F, Gesualdo M, Brunetti N, Di Leo A, Scicchitano P, Ciccone MM. Endothelial dysfunction correlates with liver fibrosis in chronic HCV infection. Gastroenterol Res Pract. 2015;2015:682174. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 8. | Ploss A, Dubuisson J. New advances in the molecular biology of hepatitis C virus infection: towards the identification of new treatment targets. Gut. 2012;61 Suppl 1:i25-i35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 9. | Bartosch B, Verney G, Dreux M, Donot P, Morice Y, Penin F, Pawlotsky JM, Lavillette D, Cosset FL. An interplay between hypervariable region 1 of the hepatitis C virus E2 glycoprotein, the scavenger receptor BI, and high-density lipoprotein promotes both enhancement of infection and protection against neutralizing antibodies. J Virol. 2005;79:8217-8229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 227] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 10. | Petit JM, Benichou M, Duvillard L, Jooste V, Bour JB, Minello A, Verges B, Brun JM, Gambert P, Hillon P. Hepatitis C virus-associated hypobetalipoproteinemia is correlated with plasma viral load, steatosis, and liver fibrosis. Am J Gastroenterol. 2003;98:1150-1154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 88] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 11. | Syed GH, Tang H, Khan M, Hassanein T, Liu J, Siddiqui A. Hepatitis C virus stimulates low-density lipoprotein receptor expression to facilitate viral propagation. J Virol. 2014;88:2519-2529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 82] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 12. | Chrostek L, Supronowicz L, Panasiuk A, Cylwik B, Gruszewska E, Flisiak R. The effect of the severity of liver cirrhosis on the level of lipids and lipoproteins. ClinExp Med. 2014;14:417-421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 13. | Duseja A, Singh SP, Saraswat VA, Acharya SK, Chawla YK, Chowdhury S, Dhiman RK, Jayakumar RV, Madan K, Misra SP, Mishra H, Modi SK, Muruganathan A, Saboo B, Sahay R, Upadhyay R. Non-alcoholic Fatty Liver Disease and Metabolic Syndrome-Position Paper of the Indian National Association for the Study of the Liver, Endocrine Society of India, Indian College of Cardiology and Indian Society of Gastroenterology. J ClinExpHepatol. 2015;5:51-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 129] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 14. | Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. ClinChem. 1972;18:499-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20005] [Cited by in RCA: 20605] [Article Influence: 381.6] [Reference Citation Analysis (3)] |
| 15. | Nanda SK, Bharathy M, Dinakaran A, Ray L, Ravichandran K. Correlation of Friedewald's calculated low-density lipoprotein cholesterol levels with direct low-density lipoprotein cholesterol levels in a tertiary care hospital. Int J Appl Basic Med Res. 2017;7:57-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | González-Ojeda A, Cervantes-Guevara G, Chávez-Sánchez M, Dávalos-Cobián C, Ornelas-Cázares S, Macías-Amezcua MD, Chávez-Tostado M, Ramírez-Campos KM, Ramírez-Arce Adel R, Fuentes-Orozco C. Platelet count/spleen diameter ratio to predict esophageal varices in Mexican patients with hepatic cirrhosis. World J Gastroenterol. 2014;20:2079-2084. [PubMed] [DOI] [Full Text] |
| 17. | Finn AV, Kolodgie FD, Virmani R. Correlation between carotid intimal/medial thickness and atherosclerosis: a point of view from pathology. ArteriosclerThrombVascBiol. 2010;30:177-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 266] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 18. | Wong VW, Vergniol J, Wong GL, Foucher J, Chan HL, Le Bail B, Choi PC, Kowo M, Chan AW, Merrouche W, Sung JJ, de Lédinghen V. Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology. 2010;51:454-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1098] [Cited by in RCA: 988] [Article Influence: 61.8] [Reference Citation Analysis (13)] |
| 19. | Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, Montaner J, S Sulkowski M, Torriani FJ, Dieterich DT, Thomas DL, Messinger D, Nelson M; APRICOT Clinical Investigators. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4179] [Cited by in RCA: 3913] [Article Influence: 195.7] [Reference Citation Analysis (7)] |
| 20. | Paquet KJ. Prophylactic endoscopic sclerosing treatment of the esophageal wall in varices -- a prospective controlled randomized trial. Endoscopy. 1982;14:4-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 306] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 21. | Kajikawa M, Maruhashi T, Iwamoto Y, Iwamoto A, Matsumoto T, Hidaka T, Kihara Y, Chayama K, Nakashima A, Goto C, Noma K, Higashi Y. Borderline ankle-brachial index value of 0. Circ J. 2014;78:1740-1745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Carmo GA, Mandil A, Nascimento BR, Arantes BD, Bittencourt JC, Falqueto EB, Ribeiro AL. Can we measure the ankle-brachial index using only a stethoscope? Fam Pract. 2009;26:22-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Garcia-Tsao G, Lim JK; Members of Veterans Affairs Hepatitis C Resource Center Program. Management and treatment of patients with cirrhosis and portal hypertension: recommendations from the Department of Veterans Affairs Hepatitis C Resource Center Program and the National Hepatitis C Program. Am J Gastroenterol. 2009;104:1802-1829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 165] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 24. | Garcia-Tsao G, Friedman S, Iredale J, Pinzani M. Now there are many (stages) where before there was one: In search of a pathophysiological classification of cirrhosis. Hepatology. 2010;51:1445-1449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 427] [Cited by in RCA: 389] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 25. | de Franchis R. Evolving consensus in portal hypertension. J Hepatol. 2005;43:167-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 855] [Cited by in RCA: 714] [Article Influence: 34.0] [Reference Citation Analysis (1)] |
| 26. | Soresi M, Giannitrapani L, Cervello M, Licata A, Montalto G. Non invasive tools for the diagnosis of liver cirrhosis. World J Gastroenterol. 2014;20:18131-18150. [PubMed] [DOI] [Full Text] |
| 27. | Maharaj B, Maharaj RJ, Leary WP, Cooppan RM, Naran AD, Pirie D, Pudifin DJ. Sampling variability and its influence on the diagnostic yield of percutaneous needle biopsy of the liver. Lancet. 1986;1:523-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 490] [Cited by in RCA: 449] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 28. | Regev A, Berho M, Jeffers LJ, Milikowski C, Molina EG, Pyrsopoulos NT, Feng ZZ, Reddy KR, Schiff ER. Sampling error and intraobserver variation in liver biopsy in patients with chronic HCV infection. Am J Gastroenterol. 2002;97:2614-2618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1681] [Cited by in RCA: 1537] [Article Influence: 64.0] [Reference Citation Analysis (4)] |
| 29. | Abbasi A, Bhutto AR, Butt N, Lal K, Munir SM. Serum cholesterol: could it be a sixth parameter of Child-Pugh scoring system in cirrhotics due to viral hepatitis? J Coll Physicians Surg Pak. 2012;22:484-487. [PubMed] |
| 30. | Ghadir MR, Riahin AA, Havaspour A, Nooranipour M, Habibinejad AA. The relationship between lipid profile and severity of liver damage in cirrhotic patients. Hepat Mon. 2010;10:285-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 31. | Boemeke L, Bassani L, Marroni CA, Gottschall CB. Lipid profile in cirrhotic patients and its relation to clinical outcome. Arq Bras Cir Dig. 2015;28:132-135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 32. | Leavens KF, Easton RM, Shulman GI, Previs SF, Birnbaum MJ. Akt2 is required for hepatic lipid accumulation in models of insulin resistance. Cell Metab. 2009;10:405-418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 241] [Article Influence: 14.2] [Reference Citation Analysis (4)] |
| 33. | Marchesini G, Brizi M, Bianchi G, Tomassetti S, Bugianesi E, Lenzi M, McCullough AJ, Natale S, Forlani G, Melchionda N. Nonalcoholic fatty liver disease: a feature of the metabolic syndrome. Diabetes. 2001;50:1844-1850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1918] [Cited by in RCA: 1717] [Article Influence: 68.7] [Reference Citation Analysis (3)] |
| 34. | Barakat AAE, Nasr FM, Metwaly AA, Morsy S, Eldamarawy M. Atherosclerosis in chronic hepatitis C virus patients with and without liver cirrhosis. Egypt Heart J. 2017;69:139-147. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 35. | Osama A, Ashour Y, Abd El-Razek R, Monir D. Assessment of carotid intima-media thickness and carotid plaque formation among patients with ischemic stroke and hepatitis C virus infection. Egy J NeurolPsyNeurosurg. 2019;55:1-6. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 36. | Lin SY, Lin CL, Lin CC, Wang IK, Hsu WH, Kao CH. Risk of acute coronary syndrome and peripheral arterial disease in chronic liver disease and cirrhosis: A nationwide population-based study. Atherosclerosis. 2018;270:154-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 37. | Patel SS, Nabi E, Guzman L, Abbate A, Bhati C, Stravitz RT, Reichman T, Matherly SC, Driscoll C, Lee H, Luketic VA, Sterling RK, Sanyal AJ, Patel V, Levy M, Siddiqui MS. Coronary artery disease in decompensated patients undergoing liver transplantation evaluation. Liver Transpl. 2018;24:333-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 38. | Kawakami M, Koda M, Murawaki Y. The prevalence of atherosclerosis in cirrhotic patients: assessment of two aspects of atherosis and sclerosis. Hepatol Res. 2007;37:152-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Egypt
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Poturoglu S, Yao DF S-Editor: Yan JP L-Editor: Webster JR P-Editor: Wang LL