Published online May 14, 2026. doi: 10.3748/wjg.v32.i18.118499
Revised: February 2, 2026
Accepted: February 24, 2026
Published online: May 14, 2026
Processing time: 122 Days and 22.3 Hours
Colonic anastomotic varices are extremely rare, particularly in patients without portal hypertension. Their clinical presentation is often nonspecific, and stan
A 65-year-old man with a history of laparoscopic transverse colectomy presented with recurrent melena at 30-, 35-, and 38-month postoperatively. Colonoscopy repeatedly demonstrated active bleeding from dilated submucosal veins at the anastomotic site, and endoscopic clipping provided only temporary hemostasis. Laboratory tests revealed preserved hepatic function, and imaging revealed no findings suggestive of portal hypertension. Due to refractory bleeding and the invasive nature of the reoperation, an interventional radiology (IR) approach was selected. Computed tomography portography revealed venous convergence in both the superior and inferior mesenteric systems at the anastomotic site. Tar
IR is an effective and minimally invasive treatment option for refractory anastomotic colonic varices in patients without portal hypertension.
Core Tip: Colonic anastomotic varices are extremely rare, particularly in patients without cirrhosis or portal hypertension, and optimal management has not been established. We describe a unique case of recurrent bleeding from transverse colonic anastomotic varices after colectomy, in which several endoscopic attempts at hemostasis were unsuccessful. Angiography revealed localized venous congestion at the anastomosis, prompting targeted embolization using a transhepatic interventional radiological approach. This case underscores the need to consider anastomotic varices as a potential late postoperative bleeding source and shows that coordinated treatment planning across multiple specialties is essential for selecting the most appropriate therapeutic strategy.
- Citation: Tomita D, Yamamoto M, Wakamatsu M, Shibayama R, Maeda Y, Hiramatsu K, Hanaoka Y, Toda S, Ueno M, Matoba S, Kuroyanagi H. Anastomotic colonic varices after colectomy treated by obliteration via a transhepatic portal approach: A case report. World J Gastroenterol 2026; 32(18): 118499
- URL: https://www.wjgnet.com/1007-9327/full/v32/i18/118499.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i18.118499
Ectopic varices may occur at any site along the gastrointestinal tract and account for approximately 5% of all variceal bleeding episodes, most often in the setting of portal hypertension[1]. Among these, colonic varices and varices arising at gastrointestinal anastomotic sites are exceedingly uncommon compared to duodenal or rectal varices[2,3]. Only a few cases of colonic-related anastomotic varices involving the rectosigmoid or ileocolic anastomosis have been reported in the literature[4-6].
Diagnosis and management of colonic anastomotic varices present substantial clinical challenges. Their endoscopic appearance may mimic postoperative vascular changes and active bleeding may be intermittent or difficult to localize. Owing to the rarity of this condition, no standardized therapeutic approach has been established, and interventions range from endoscopic therapy to transjugular intrahepatic portosystemic shunt (TIPS) placement or surgical resection. Interventional radiology (IR) has been used for selected types of ectopic varices[7-9], but experience with colonic anasto
Here, we describe a unique case of recurrent lower gastrointestinal bleeding from transverse colonic anastomotic varices in a non-cirrhotic patient who was successfully treated using a minimally invasive transhepatic embolization approach. This case highlights the important diagnostic and therapeutic considerations for this uncommon condition.
A 65-year-old man presented with recurrent episodes of melena and progressive anemia.
He underwent a laparoscopic transverse colectomy for the mid-transverse colon cancer 4 years ago. The roots of the middle colic artery and vein were identified, D3 lymph node dissection was performed, and the vessels were clipped and ligated. Reconstruction was performed using a functional end-to-end anastomosis technique. The postoperative course was uneventful, with no clinical episodes suggestive of bowel obstruction, and he was discharged on postoperative day 8. The postoperative pathological finding was stage I (pT2N0M0). No adjuvant chemotherapy or radiotherapy was ad
Thirty months after the surgery, the patient experienced a sudden episode of melena. Colonoscopy revealed dilated submucosal veins at the anastomotic site with active bleeding, whereas endoscopic clipping provided only temporary hemostasis. At 35 months and 38 months after surgery, the patient experienced second and third episodes of melena. On both occasions, colonoscopy demonstrated active hemorrhage originating from sites other than the previous clipping areas. Despite three endoscopic hemostasis attempts, bleeding recurred shortly after the third procedure. Therefore, different therapeutic approaches were required. The quantitative clinical course for each episode, including blood pressure/heart rate/shock index, hemoglobin and hematocrit levels, use of red blood cell (RBC) transfusions, iron therapy, length of hospital stay, and the absence of anticoagulant/antiplatelet therapy, is summarized in Table 1. During the third hospitalization, the patient received intravenous iron therapy for 5 days and underwent six units of RBC transfusion altogether. Anticoagulant or antiplatelet therapy was not administered.
| Episode (timing after colectomy) | 1st (30 months) | 2nd (35 months) | 3rd (38 months) |
| Blood pressure (mmHg) | 137/78 | 147/94 | 116/82 |
| Heart rate (beats/minute) | 72 | 66 | 93 |
| Shock index (HR/SBP) | 0.53 | 0.45 | 0.80 |
| Hemoglobin (g/dL) | 13.3 | 14.1 | 7.5 |
| Hematocrit (%) | 40.9 | 41.5 | 23.7 |
| Iron therapy | Intravenous iron during hospitalization | None | Intravenous iron between admission and IR |
| Anticoagulant/antiplatelet therapy | None | None | None |
| Transfusion | None | None | RBC 4 units 4 days before IR; RBC 2 units after IR |
| Management | Endoscopic clipping | Endoscopic clipping | Endoscopic clipping and IR |
| Length of hospital stay | 5 days | 7 days | 22 days |
| Outcome | Temporary control | Temporary control | No recurrent bleeding after IR; objective follow-up negative |
Past medical history other than colorectal cancer included prior cholecystectomy, hypertension, and dyslipidemia. The patient had no history of liver disease, portal hypertension, viral hepatitis, or alcohol-related liver injury.
He did not consume excessive alcohol, and had no family history of colorectal cancer, liver disease, or hematological or hereditary bleeding disorders.
At the time of rebleeding after the third endoscopic treatment, the patient’s vital signs were as follows: Temperature
Laboratory data showed: White blood cell 8.8 × 103/µL; hemoglobin 7.5 g/dL; hematocrit 23.7%; platelets 166 × 103/µL; aspartate aminotransferase (AST) 28 U/L; alanine aminotransferase (ALT) 49 U/L; total bilirubin 0.6 mg/dL; albumin 3.9 g/dL; blood urea nitrogen 22 mg/dL; creatinine 0.95 mg/dL; C-reactive protein 0.22 mg/dL; prothrombin time 85.4%; activated partial thromboplastin time 28.8 seconds. Liver function was preserved, and the findings were consistent with progressive anemia. During routine follow-up before these events, the patient’s baseline hemoglobin level was approximately 14-15 g/dL. Platelet counts remained stable over time (145 × 103-209 × 103/µL), without chronic thrombocytopenia. Preoperative and early postoperative laboratory test results showed no evidence of hepatic dysfunction or coagulopathy (Supplementary Table 1).
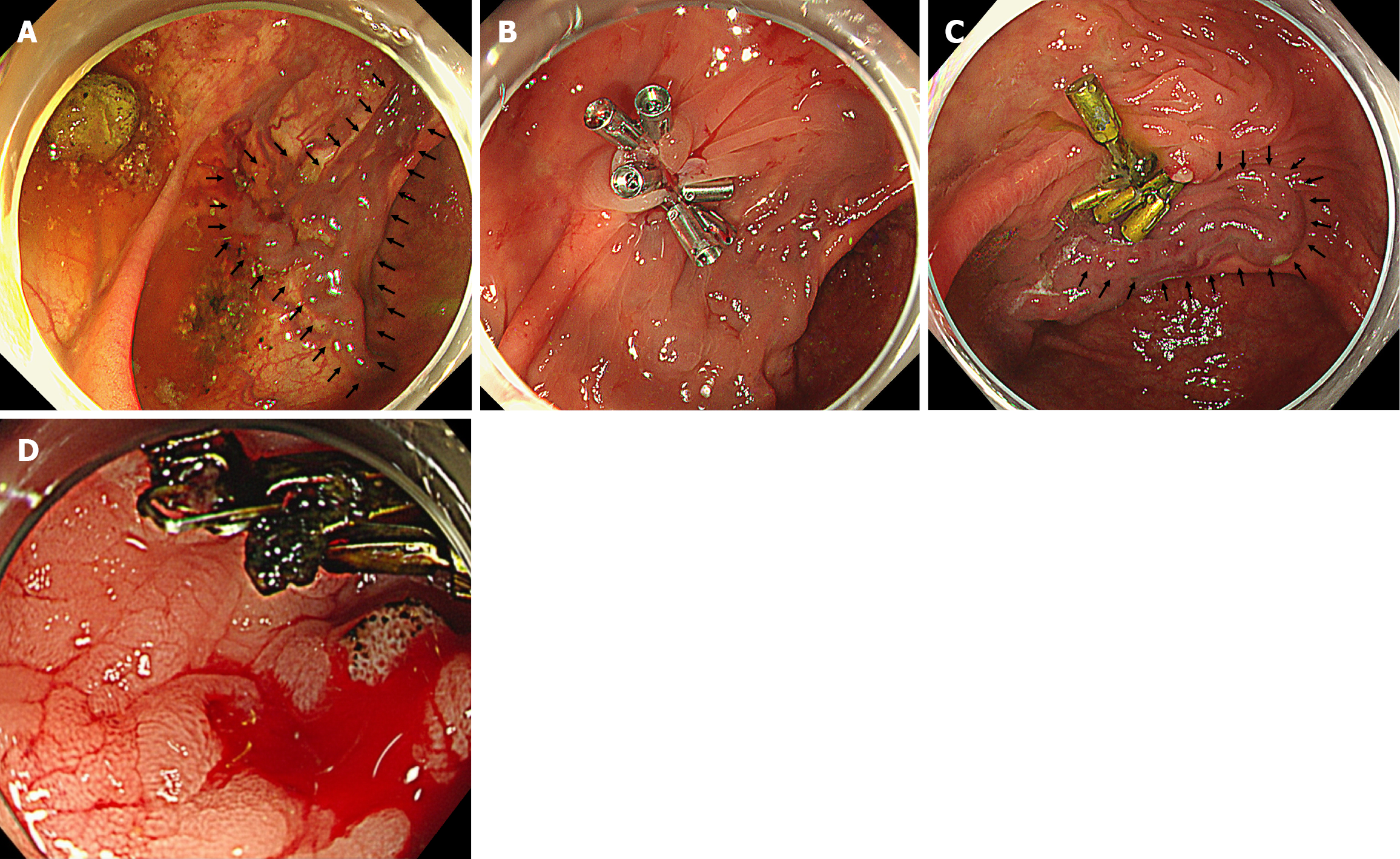
During the first bleeding episode, colonoscopy revealed markedly dilated and tortuous submucosal veins around the anastomotic site with active bleeding from the varix (Figure 1A). Hemostasis was achieved via clipping (Figure 1B). During the second and third episodes, similar venous dilatation was again observed near the anastomotic site. Exudative bleeding was identified at a different site from the previous clip site (Figure 1C and D), and temporary hemostasis was achieved with clipping. A few days after the third hemostatic procedure, the patient experienced recurrent rectal bleeding.
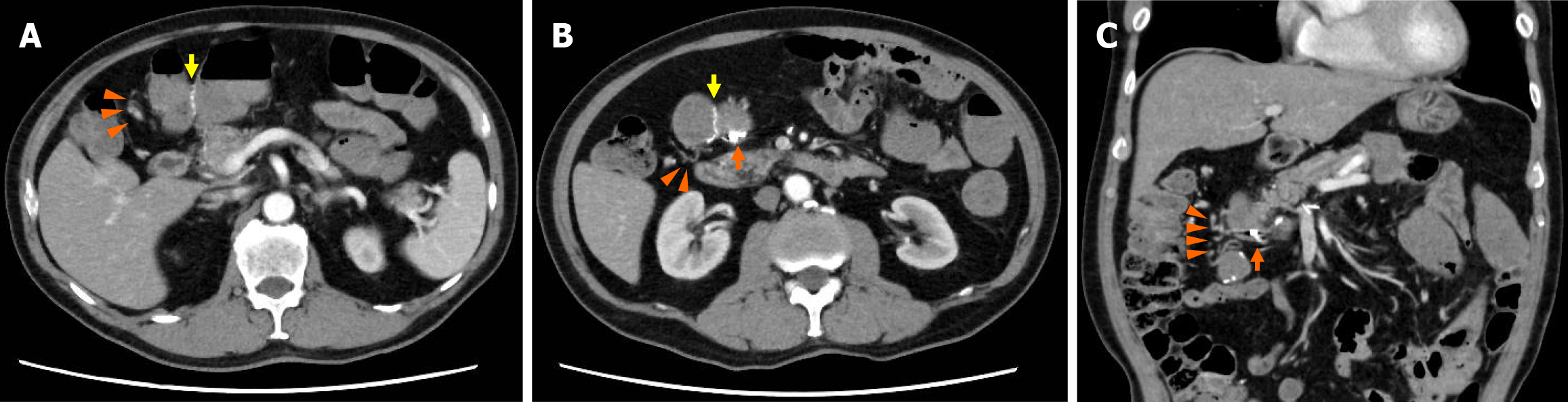
Contrast-enhanced computed tomography (CT) performed before the third hemostatic attempt revealed metallic clips near the anastomosis site (Figure 2). The anastomosis site was at the midpoint of the transverse colon (where the tumor was located), with an anastomosis diameter of 55 mm. Abnormal vascular structures were observed around the anastomotic region. Slightly hyperdense intraluminal fluid collection was noted; however, no active extravasation was observed. No imaging findings suggested liver cirrhosis or portal hypertension, and no evidence of tumor recurrence was identified. Routine postoperative surveillance ultrasonography demonstrated no splenomegaly (approximately 90 mm × 50 mm) and a main portal vein diameter of approximately 10 mm. Liver stiffness measurements, serum fibrosis markers, and Doppler ultrasonography were not performed.
As the patient had refractory gastrointestinal bleeding despite multiple endoscopic treatments, segmental colectomy with re-anastomosis was considered. However, owing to the invasiveness of surgery and uncertain bleeding mechanisms, IR was selected for diagnostic evaluation and potential therapy.
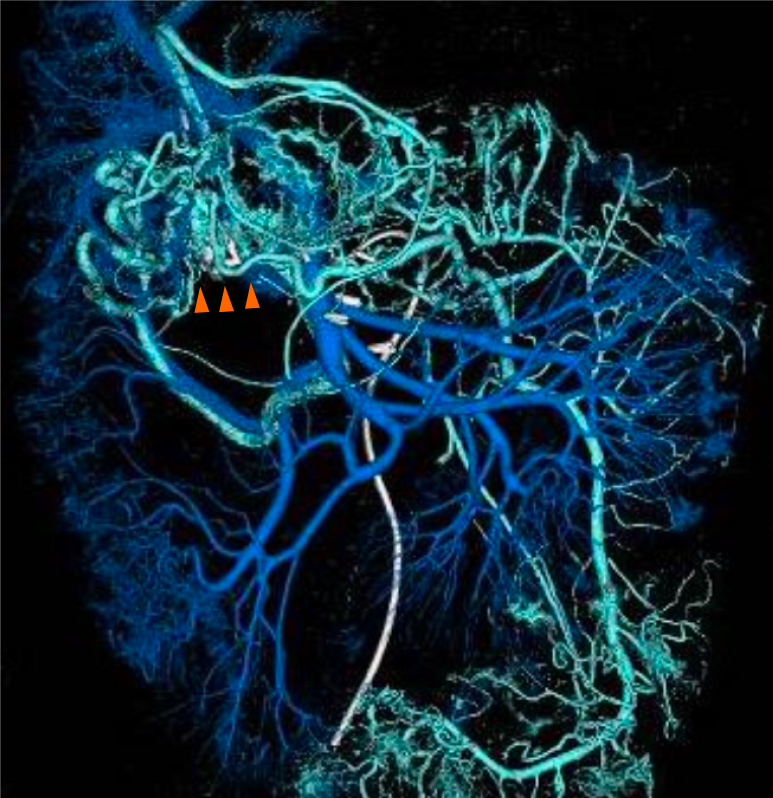
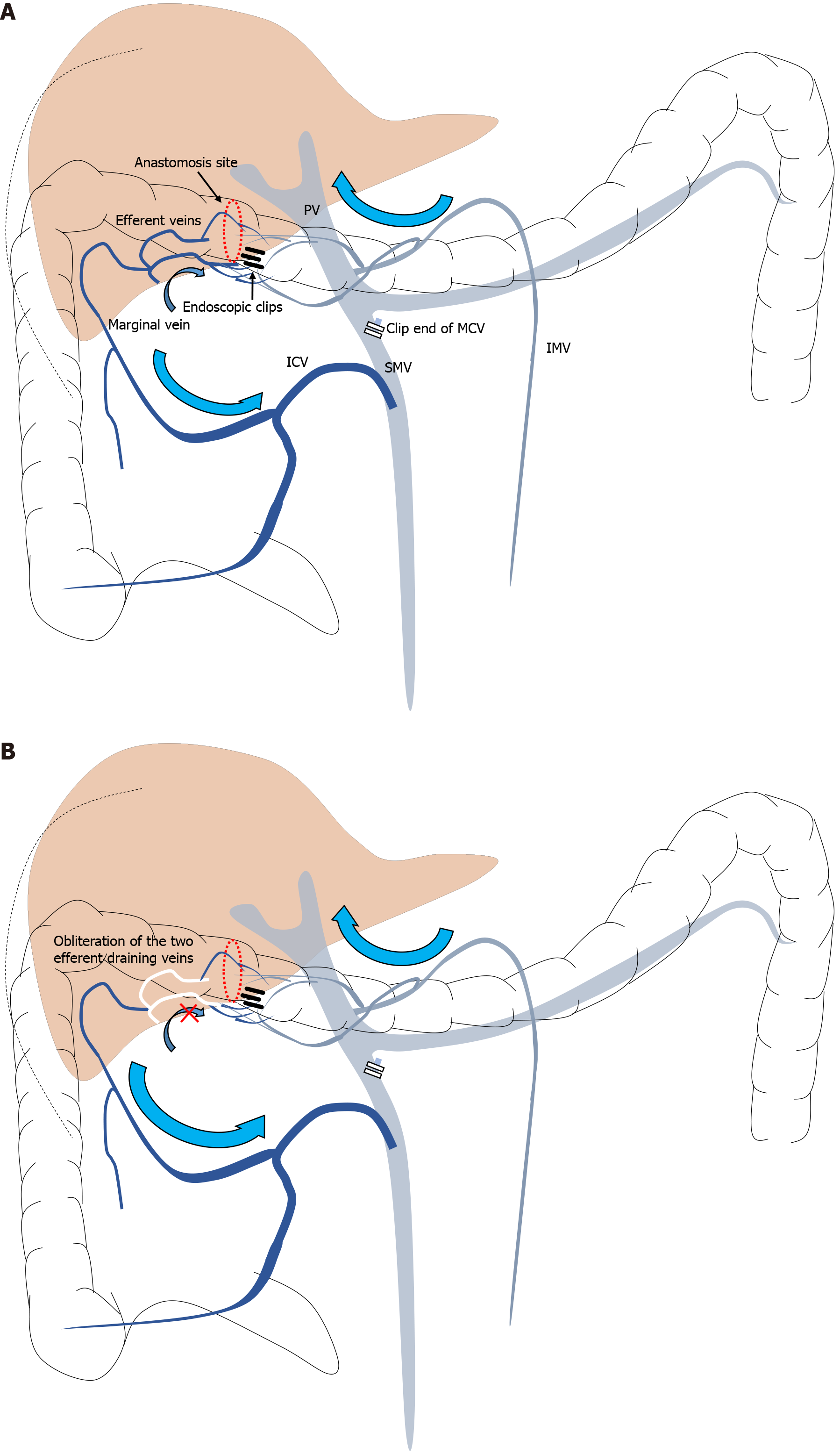
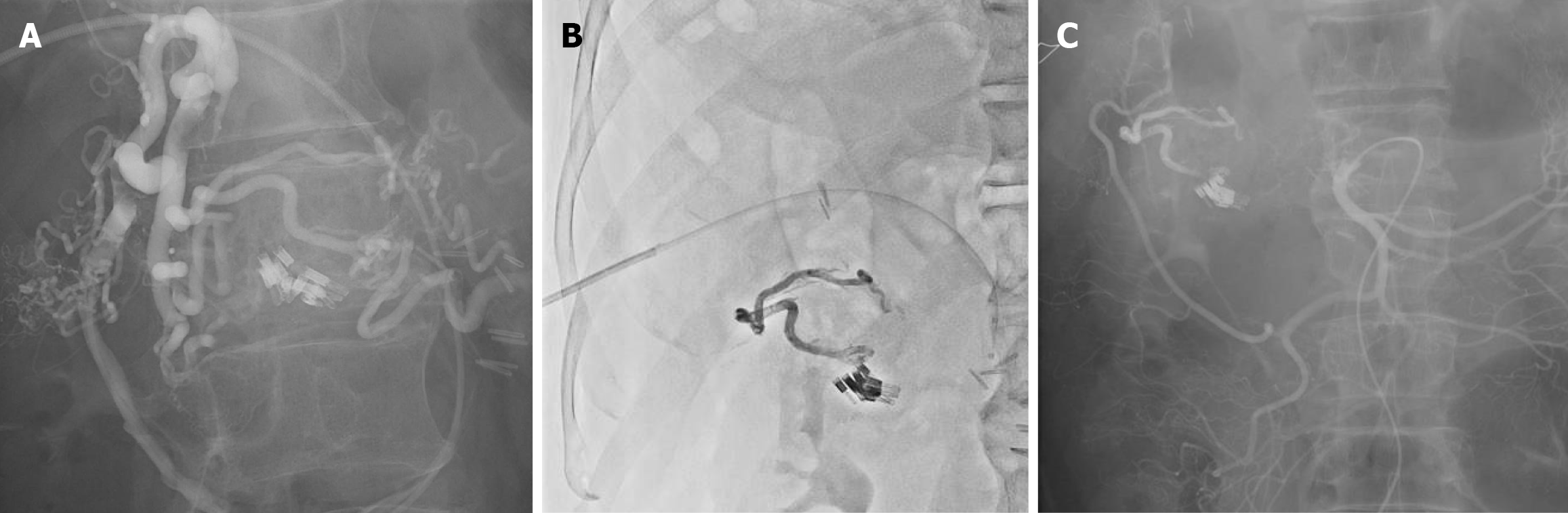
Portal-phase angiography via the superior mesenteric artery (SMA) and inferior mesenteric artery (IMA), along with CT portography, demonstrated convergence of venous drainage from both territories during transverse colon anastomosis (Figure 3). This convergence corresponded precisely to the previously clipped bleeding sites. These findings indicated that the dominant inflow arose from the marginal vein [superior mesenteric vein (SMV) territory] and the dominant outflow drained into the inferior mesenteric vein (IMV) through two efferent veins (Figure 4A). Localized venous congestion at the site of anastomosis was the pathophysiological basis of varix formation and hemorrhage. No stenosis, obstruction, or thrombus was observed in the portal vein.
The final diagnosis is anastomotic varices at the transverse colon anastomosis.
Given the diffuse submucosal variceal plexus and recurrent bleeding, a combined strategy using ethanolamine oleate (EO) and N-butyl-2-cyanoacrylate (NBCA) was selected to achieve extensive thrombosis and durable occlusion. IR-guided embolization was performed to achieve definitive hemostasis. The procedure was performed under local anesthesia in an angiography suite equipped with an interventional CT scanner. Under real-time ultrasonographic guidance, the right anterior branch of the portal vein was accessed using an 18-G needle, and a 5-Fr Arrow-Flex sheath (Teleflex Medical, United States) was inserted into the portal venous system. The portal pressure was 5 mmHg, and there was no clinical or radiological evidence of portal hypertension. A 5-Fr/2.9-Fr MP balloon catheter (B-2; Terumo, Tokyo, Japan) was advanced through the portal, superior mesenteric, ileocolic, and marginal veins near the variceal lesion. Balloon occlusion was performed at the right side of the anastomotic varices. Balloon-occluded venography revealed variceal drainage extending beyond the previously placed endoscopic clips and draining toward the IMV (Figure 5A).
Subsequently, a 2.9-Fr microballoon catheter (Logos Grandmaster, PIOLAX, Yokohama, Japan) was advanced to the right side of the feeding veins supplying the variceal component. Through this catheter, 3 mL of 5% EO (Oldamin®, ASKA Pharmaceutical, Tokyo, Japan) was injected with balloon inflation, followed by a 5-minute dwell time, ensuring adequate sclerosant distribution within the varices. After confirming stasis, 1 mL of NBCA mixed with ethiodized oil in a 1:2 ratio (33% NBCA) was injected to selectively occlude the two efferent draining veins, thereby achieving unilateral flow control and directing the sclerosing agent into the targeted variceal complex (Figures 4B and 5B). Throughout the procedure, test injections and continuous fluoroscopic monitoring were used to avoid reflux or migration, and embolization was limited to the variceal complex and its efferent veins to minimize the risks of non-target embolization and bowel ischemia.
Final angiography from both the SMA and IMA demonstrated complete disappearance of the anastomotic varices and elimination of the previously visualized outflow tract, confirming successful obliteration (Figure 5C). The hepatic puncture tract was embolized using a 10 mm × 20 cm coil (Nester Coil; Cook Medical Inc., Bloomington, IN, United States) to prevent postprocedural bleeding. No immediate complications occurred.
Prophylactic cefazolin (1 g, intravenous) was administered immediately before the procedure and again on the morning after the procedure. No postprocedural anticoagulation or antiplatelet therapy was administered.
Preparations were made to proceed with emergency surgery should postprocedure intestinal ischemia occur immedia
The patient was discharged on the 16th day after the procedure. Follow-up colonoscopy on post-procedure day 5 and at 4 months confirmed the absence of active bleeding and no endoscopic evidence of bowel ischemia. Follow-up contrast-enhanced CT on postprocedural day 5 and at 9 months showed no abnormal findings. No late complications, including portal vein thrombosis, ischemic colitis, or segmental colitis, were observed during the 10-month follow-up period. No additional IR-related imaging (e.g., repeat portography or CT portography) was performed after discharge.
Colonic varices are dilated portosystemic venous collaterals within the colonic submucosa and are an uncommon cause of gastrointestinal bleeding, with an estimated prevalence of 0.07% in the general population[10]. They are most frequently associated with portal hypertension; however, noncirrhotic etiologies such as congestive heart failure, portomesenteric venous obstruction, splenic vein thrombosis, and congenital vascular anomalies have also been reported[1,10,11]. Ana
The mechanism underlying anastomotic variceal formation in noncirrhotic patients remains unclear. One proposed explanation is that postoperative inflammation and tissue remodeling around the anastomosis promote abnormal collateralization, resulting in fragile vessels that are prone to progressive dilatation. Moncure et al[16] reported postoperative ectopic/anastomotic varices; however, they occurred in a setting of portal hypertension, where local postoperative changes, such as adhesions, may facilitate collateral development. Although this patient had no evidence of systemic portal hypertension or clinical episodes suggestive of severe adhesive disease, such as bowel obstruction, postoperative local changes (e.g., subtle inflammation, scarring, or adhesions) could still have served as a permissive background for collateral recruitment[17]. Nonetheless, these factors alone may not fully explain the marked, site-limited variceal formation observed in this case.
In our patient, surgical anatomical changes and subsequent hemodynamic alterations after transverse colectomy likely contributed to the variceal development. Transverse colonic anastomosis lies at the interface between the venous territories draining into the SMV and IMV systems. CT portography and balloon-occluded venography suggested a dominant inflow from the marginal vein (SMV territory) and drainage into the IMV system through the two efferent veins. Radical D3 lymph node dissection required the central ligation of the middle colic vein, which may have reduced the primary outflow pathway for the SMV-drained segment and promoted venous rerouting via the marginal venous arcade toward the anastomotic region (Figure 4A). Such convergence may increase the local flow and relative outflow resistance, predisposing the submucosal venous plexus at the anastomosis to progressive dilatation and variceal formation even in the absence of portal hypertension.
Several therapeutic approaches have been reported for the management of colonic and anastomotic varices, including endoscopic therapy, TIPS, balloon-occluded retrograde transvenous obliteration, angiographic embolization, and surgical resection[1,18]. Owing to the rarity of these lesions, no standardized treatment strategy has been established. In our case, during the first and second bleeding episodes, we selected endoscopic clipping as an initial, readily available hemostatic measure to control active bleeding and to monitor the subsequent clinical course. Although clipping achieved temporary hemostasis, bleeding recurred from sites adjacent to, but distinct from, the previous clip placements, suggesting that a definitive strategy beyond repeated endoscopic measures would be required. Argon plasma coagulation was considered unsuitable because the varices were located in the submucosal region. Endoscopic variceal ligation and injection sclerotherapy have been reported for selected colonic varices[19,20]; however, in our patient, the varices were broad-based and located at a prior anastomosis, raising concern about post-treatment ulceration and transmural injury and making controlled band deployment or targeted injection technically challenging. While performing clipping as a bridging measure, we concurrently pursued multidisciplinary consultation and ultimately selected a percutaneous transhepatic IR approach. TIPS was not indicated because portal hypertension was absent. Although colectomy was considered, reoperation around a prior anastomosis was technically difficult, with a prolonged operative time and the possibility of reformation of collateral pathways.
Given the diffuse submucosal variceal network and multifocal drainage, coil embolization alone may require multiple deployments and may not sufficiently address the entire plexus. EO under balloon occlusion can induce thrombosis within the variceal channels in a controlled manner, whereas NBCA provides immediate and durable occlusion of key draining channels and the transhepatic tract. The combined EO + NBCA strategy, together with flow control (temporary balloon occlusion of the inflow channel and selective embolization of the efferent veins), was used to achieve reliable hemostasis while minimizing non-target embolization and bowel ischemia.
As the number of colorectal surgeries continues to increase worldwide, clinicians may encounter these rare complications more frequently. Gastrointestinal surgeons should consider anastomotic varices in the differential diagnosis of painless hematochezia after colectomy, particularly when colonoscopy reveals unexplained submucosal venous dilation near the anastomosis.
This study had several limitations. First, as with all single-case descriptions, the generalizability of this therapeutic approach remains uncertain. Second, long-term outcomes beyond ten months remain unknown.
In summary, we describe an exceptionally rare case of bleeding colonic-to-colonic anastomotic varices that occurred in the absence of portal hypertension and was successfully treated using percutaneous transhepatic embolization. This case demonstrates that anastomotic varices may arise as a delayed postoperative complication even in patients without cirrhosis and should be considered in the differential diagnosis of unexplained lower gastrointestinal bleeding after colorectal surgery. Because evidence guiding the management of colonic or anastomotic varices is limited, treatment must be individualized considering the anatomical features, local hemodynamics, and severity of bleeding. A collaborative evaluation involving gastroenterologists, hepatologists, gastrointestinal surgeons, and interventional radiologists is essential for selecting a minimally invasive but definitive therapy and avoiding unnecessary surgical resections, including repeat colectomy or segmental resection around a prior anastomosis. As shown in this case, IR can be an effective, minimally invasive and surgery-sparing option when endoscopic techniques fail or are unsuitable.
| 1. | Tranah TH, Nayagam JS, Gregory S, Hughes S, Patch D, Tripathi D, Shawcross DL, Joshi D. Diagnosis and management of ectopic varices in portal hypertension. Lancet Gastroenterol Hepatol. 2023;8:1046-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 2. | Bresci G, Gambardella L, Parisi G, Federici G, Bertini M, Rindi G, Metrangolo S, Tumino E, Bertoni M, Cagno MC, Capria A. Colonic disease in cirrhotic patients with portal hypertension: an endoscopic and clinical evaluation. J Clin Gastroenterol. 1998;26:222-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 3. | Watanabe N, Toyonaga A, Kojima S, Takashimizu S, Oho K, Kokubu S, Nakamura K, Hasumi A, Murashima N, Tajiri T. Current status of ectopic varices in Japan: Results of a survey by the Japan Society for Portal Hypertension. Hepatol Res. 2010;40:763-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 98] [Article Influence: 6.1] [Reference Citation Analysis (12)] |
| 4. | Sasson G, Yong E, Steinhart H. A61 Colorectal anastomotic varices in a patient with portal hypertension: a case report and review of the literature. J Can Assoc Gastroenterol. 2018;1 Suppl 2:96-97. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Braun DS, Schneider K, Mantry P. S4300 The First Documented Case of Ileocolic Anastomotic Varices. Am J Gastroenterol. 2024;119:S2765. [DOI] [Full Text] |
| 6. | Schafer TW, Binmoeller KF. Argon plasma coagulation for the treatment of colonic varices. Endoscopy. 2002;34:661-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 7. | Zamora CA, Sugimoto K, Tsurusaki M, Izaki K, Fukuda T, Matsumoto S, Kuwata Y, Kawasaki R, Taniguchi T, Hirota S, Sugimura K. Endovascular obliteration of bleeding duodenal varices in patients with liver cirrhosis. Eur Radiol. 2006;16:73-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 8. | Watanabe K, Imai Y, Takaya H, Nakazawa M, Chikayama T, Ando S, Mizuno Y, Sugawara K, Nakamura Y, Saitoh E, Hamaoka K, Motoya D, Fujimori K, Inao M, Nakayama N, Nagoshi S, Mochida S. A case of liver cirrhosis due to hepatits C virus infection complicating giant anorectal varices treated with balloon-occluded retrograde transvenous obliteration. Clin J Gastroenterol. 2011;4:19-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 9. | Takano M, Imai Y, Nakazawa M, Chikayama T, Ando S, Sugawara K, Nakayama N, Mochida S. A case of liver cirrhosis with bleeding from stomal varices successfully treated using balloon-occluded retrograde transvenous obliteration. Clin J Gastroenterol. 2016;9:145-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Iredale JP, Ridings P, McGinn FP, Arthur MJ. Familial and idiopathic colonic varices: an unusual cause of lower gastrointestinal haemorrhage. Gut. 1992;33:1285-1288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 11. | Henry Z, Uppal D, Saad W, Caldwell S. Gastric and ectopic varices. Clin Liver Dis. 2014;18:371-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 12. | Tai Y, Tong H, Wei B, Wu H. Ectopic variceal bleeding after hepatobiliary surgery: A case report. Medicine (Baltimore). 2021;100:e24975. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Saeki Y, Ide K, Kakizawa H, Ishikawa M, Tashiro H, Ohdan H. Controlling the bleeding of jejunal varices formed at the site of choledochojejunostomy: report of 2 cases and a review of the literature. Surg Today. 2013;43:550-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Garbuzenko DV. Current approaches to the management of jejunal variceal bleeding at the site of hepaticojejunostomy after pancreaticoduodenectomy. World J Gastroenterol. 2024;30:4083-4086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Liu J, Wang P, Wang LM, Guo J, Zhong N. Outcomes of endoscopic sclerotherapy for jejunal varices at the site of choledochojejunostomy (with video): Three case reports. World J Gastroenterol. 2024;30:2059-2067. [PubMed] [DOI] [Full Text] |
| 16. | Moncure AC, Waltman AC, Vandersalm TJ, Linton RR, Levine FH, Abbott WM. Gastrointestinal hemorrhage from adhesion-related mesenteric varices. Ann Surg. 1976;183:24-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 77] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Park RW, Watkins JB. Mesenteric vascular occlusion and varices complicating midgut malrotation. Gastroenterology. 1979;77:565-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Lee EW, Eghtesad B, Garcia-Tsao G, Haskal ZJ, Hernandez-Gea V, Jalaeian H, Kalva SP, Mohanty A, Thabut D, Abraldes JG. AASLD Practice Guidance on the use of TIPS, variceal embolization, and retrograde transvenous obliteration in the management of variceal hemorrhage. Hepatology. 2024;79:224-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 113] [Article Influence: 56.5] [Reference Citation Analysis (0)] |
| 19. | Xie S, Ruan MF, Wang J, Li MB. Endoscopic band ligation for transverse colonic variceal bleeding: case report and review of the literature. Ann Saudi Med. 2020;40:255-258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 20. | Sato T, Yamazaki K, Akaike J, Toyota J, Karino Y, Ohmura T, Nishioka H. Transverse colonic varices successfully treated with endoscopic procedure. Clin J Gastroenterol. 2008;1:52-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |