Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.117109
Revised: January 27, 2026
Accepted: March 9, 2026
Published online: April 28, 2026
Processing time: 134 Days and 16.2 Hours
Type 2 autoimmune pancreatitis (AIP-2) is a rare immunoglobulin G4 (IgG4)-negative inflammatory disease whose diagnosis is often challenging because it closely mimics pancreaticobiliary malignancies, frequently resulting in misdiagnosis and unnecessary surgery.
A 35-year-old man presented with obstructive jaundice. Imaging revealed pancreatic enlargement and a distal common bile duct stricture, whereas serum IgG4 levels were normal. Given the strong suspicion of malignancy, he underwent pancreaticoduodenectomy. Histopathological examination of the resected spe
AIP-2 must be considered in IgG4-negative obstructive jaundice with biliary stricture.
Core Tip: Type 2 autoimmune pancreatitis is an immunoglobulin G4-negative inflammatory disorder typically confined to the pancreas. In the present case, thickening of the common bile duct wall was observed, which may represent secondary changes resulting from extrinsic compression due to pancreatic inflammation. Mimicking pancreaticobiliary malignancies, type 2 autoimmune pancreatitis should be included in the differential diagnosis of obstructive jaundice to prevent unwarranted surgical intervention.
- Citation: Wang KR, Sha SM, Wang SH, Zhao P, Shi HT, Liu L, Wu J, Zhao G. Biliary stricture as an atypical presentation of type 2 autoimmune pancreatitis: A case report. World J Gastroenterol 2026; 32(16): 117109
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/117109.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.117109
Autoimmune pancreatitis (AIP) is a distinct form of chronic fibro-inflammatory pancreatic disease. Based on histopathological characteristics, AIP is classified into two distinct clinical subtypes: (1) Type 1 AIP; and (2) Type 2 AIP (AIP-2). Type 1 AIP represents the pancreatic manifestation of immunoglobulin G4 (IgG4)-related disease, typically presenting with systemic multi-organ involvement. Its characteristic histopathological features include dense lymphoplasmacytic infiltrates, storiform fibrosis, obliterative phlebitis, and often eosinophilic infiltration; hence, it is also termed lymphoplasmacytic sclerosing pancreatitis. In contrast, AIP-2, also known as idiopathic duct-centric pancreatitis, exhibits distinct clinical and histologic profiles and is comparatively rare. The histopathologic hallmark of AIP-2 is granulocytic epithelial lesions (GELs) affecting medium-to-small ducts and occasionally pancreatic acini[1]. IgG4-positive plasma cells are typically absent or scarce (< 10 cells per high-power field). AIP-2 is not associated with elevated serum IgG4 levels, and no reliable serum biomarker currently exists. While extra-pancreatic involvement is generally absent, AIP-2 may co-occur with inflammatory bowel disease in some patients. Biliary strictures may occur, usually resulting from extrinsic compression by inflamed pancreatic tissue rather than primary biliary inflammation. The lack of specific serologic markers and systemic signs contributes to a high risk of misdiagnosis and inappropriate surgical intervention[2].
We herein report a case of obstructive jaundice with elevated carbohydrate antigen (CA)-199 and distinct common bile duct (CBD) wall thickening, initially suspicious for malignancy. The patient underwent surgical resection, with final histopathology confirming AIP-2. This case underscores the importance of including AIP-2 in the differential diagnosis of IgG4-negative biliary strictures to avoid unnecessary surgery.
A 35-year-old man presented with a 1-week history of progressive jaundice, upper abdominal discomfort, nausea, and dark urine.
Symptoms, including progressive jaundice, upper abdominal discomfort, nausea, and dark urine, began one week prior to presentation. The patient reported no history of alcohol use or a high-fat diet preceding the onset of symptoms. There was no significant weight loss noted.
The patient denied any history of alcohol, tobacco, or illicit drug use. There was no personal or family history of malignancy or pancreatic disease.
Physical examination revealed mild abdominal tenderness.
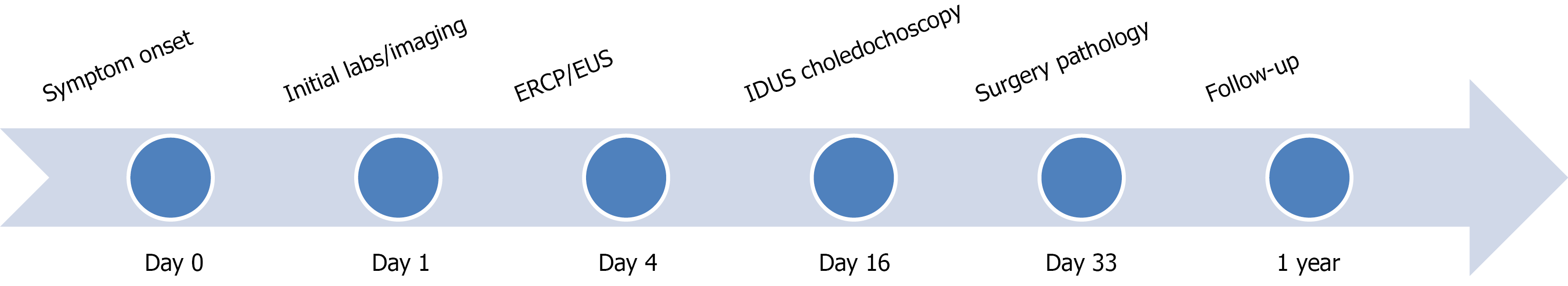
A timeline illustrating the clinical course of this case is provided (Figure 1). Laboratory tests showed marked cholestasis (reference ranges in parentheses): (1) Total bilirubin 68 μmol/L (< 23 μmol/L); (2) Direct bilirubin 34.68 μmol/L (< 4 μmol/L); (3) Indirect bilirubin 33.31 μmol/L (< 19 μmol/L); (4) Gamma-glutamyl transferase 567 U/L (10-60 U/L); and (5) Alkaline phosphatase 483 IU/L (45-125 IU/L). Elevated pancreatic enzymes levels and liver dysfunction were noted: (1) Serum amylase 157 IU/L; (2) Urine amylase 1625 IU/L; (3) Aspartate aminotransferase 165 IU/L (15-40 IU/L); and (4) Alanine aminotransferase 282 IU/L (9-50 IU/L). Furthermore, C-reactive protein was 55.62 mg/L (< 10 mg/L), and CA-199 was 87.3 U/mL (0-30 U/mL). Routine blood and stool tests, and serum IgG and IgG4 levels were within normal limits.
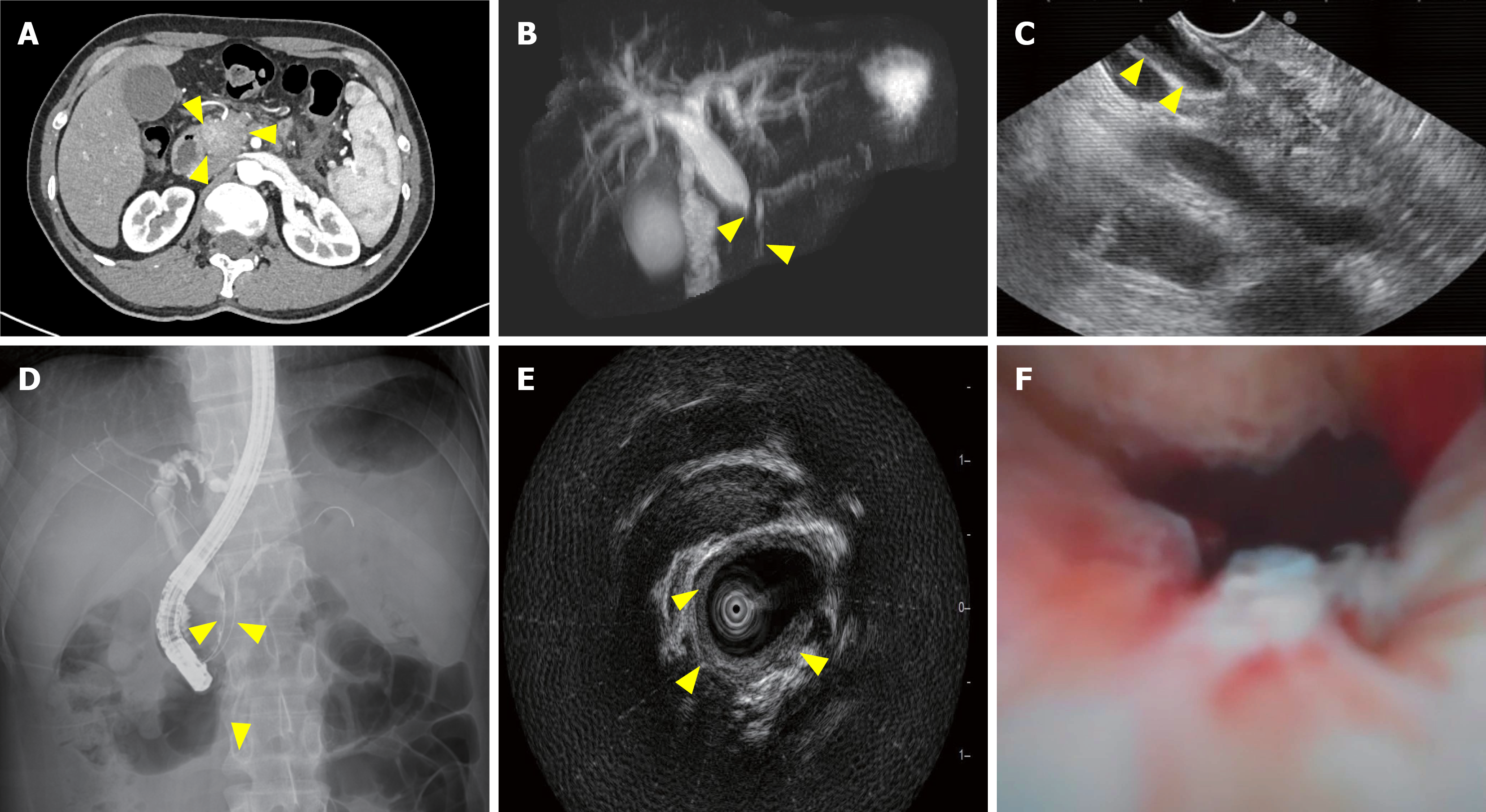
Radiology and endoscopy: Abdominal contrast-enhanced computed tomography revealed enlargement of the pancreatic head, characterized by heterogeneous enhancement, accompanied by concomitant biliary and pancreatic duct dilation (Figure 2A). Magnetic resonance cholangiopancreatography revealed irregular narrowing with marked upstream dilation of the distal CBD (17 mm in diameter), mild pancreatic duct dilatation, and a duct-penetrating sign (Figure 2B). On the day 4 of hospitalization, endoscopic ultrasonography revealed mild pancreatic duct dilation (3.6 mm in diameter) and a short-segment stricture at the distal CBD, accompanied by upstream ductal dilatation (Figure 2C). Endoscopic retrograde cholangiopancreatography (ERCP) revealed a markedly dilated common hepatic duct (maximum diameter, 13 mm) with a tapered 15-mm-long stricture in the distal CBD and associated marked intrahepatic biliary dilation (Figure 2D). It is noteworthy that bile aspiration during ERCP may lead to a measured duct diameter slightly less than that observed on pre-procedural cross-sectional imaging. Plastic pancreatic duct stenting and endoscopic nasobiliary drainage were performed to alleviate jaundice. A recheck 12 days later showed a CA-199 level of 55.7 U/mL. Repeat evaluations via intraductal ultrasound and choledochoscopy revealed circumferential wall thickening of the distal CBD and stenosis in the middle to lower CBD segments (Figure 2E and F). A plastic stent was placed in the bile duct to alleviate symptoms during the second ERCP. Biopsy revealed chronic mucosal inflammation with glandular and focal atypical epithelial hyperplasia. Six days after bile duct stent placement, retesting revealed a CA-199 level of 52 U/mL.
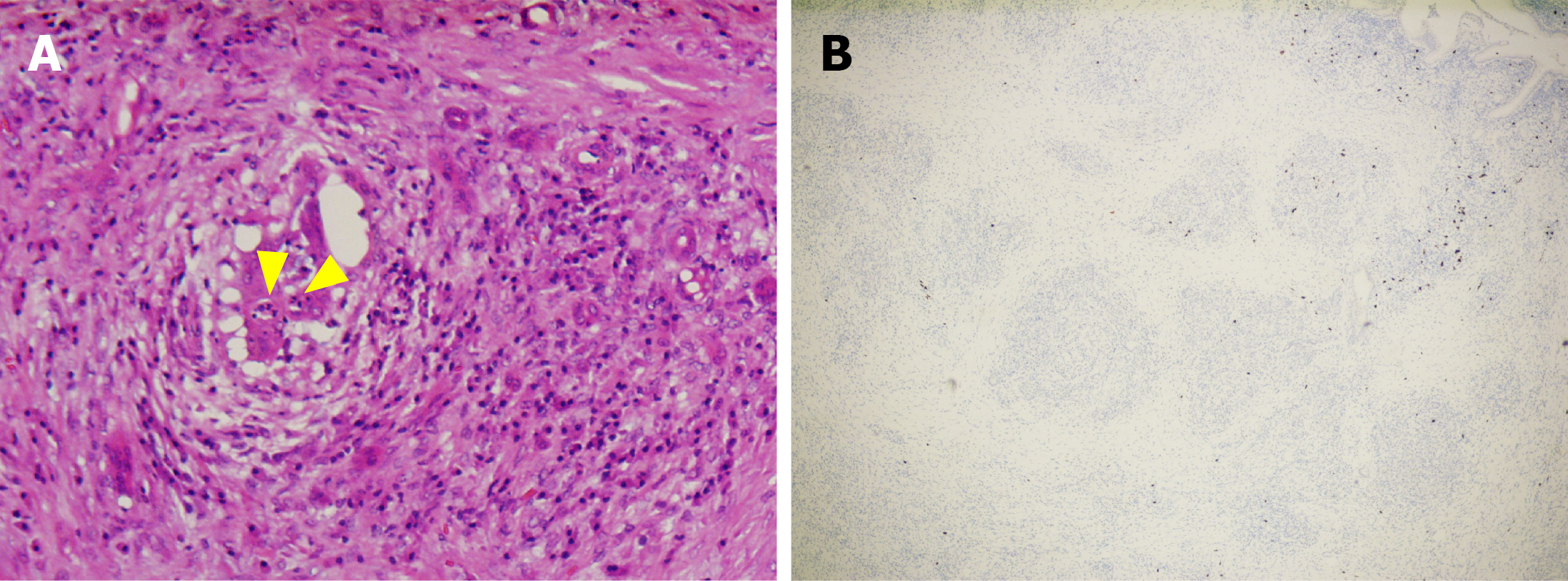
Histopathology: In the present case, the patient presented with jaundice, and radiological findings revealed circumferential wall thickening of the bile duct with obstruction. Additionally, CA-199 levels were consistently elevated. Therefore, the patient underwent surgery at another hospital to differentiate between bile duct malignancies approximately two weeks later. During surgery, a mass was palpated in the distal section of the bile duct and enlarged lymph nodes were identified along the common hepatic artery. Considering the possibility of cholangiocarcinoma, a duodenopancreatectomy was performed. Histopathological analysis confirmed a diagnosis of AIP-2. The findings were characterized by: (1) GELs with neutrophilic destruction and obliteration of the duct lumen; and (2) Periductal and interlobular fibrosis accompanied by a dense inflammatory infiltrate composed of plasma cells and neutrophils (Figure 3A). Notably, IgG4-positive plasma cells were rare (< 10 cells/high-power field) (Figure 3B). No evidence of a tumor was observed. Based on the international consensus diagnostic criteria for AIP, the final diagnosis was AIP-2[3].
The final diagnosis was AIP-2.
The patient underwent surgery at another hospital to differentiate bile duct malignancies. Twelve days after surgery, the patient was discharged with no severe postoperative complications. Postoperatively, steroid therapy was not initiated.
Although an association between AIP-2 and inflammatory bowel disease has been recognized, our patient did not present with typical gastrointestinal symptoms (such as diarrhea or bloody stools) and had normal stool studies. Therefore, a colonoscopy was not performed during the initial diagnostic workup. Besides, steroid therapy was not initiated postoperatively. Over 1 year of follow-up, the patient reported no gastrointestinal symptoms, including abdominal pain or changes in bowel habits with no signs of relapse.
AIP-2, also known as idiopathic duct-centric pancreatitis, is a rare inflammatory disease of the pancreas characterized by GELs in the absence of IgG4 seropositivity or systemic involvement[4]. Biliary strictures and ductal dilation can occur in AIP-2, resulting from extrinsic compression by inflamed pancreatic tissue[5,6]. This case presented with CBD wall thickening in AIP-2, which was verified by histology of the biopsy specimen and choledochoscopy. The lesions exhibited chronic mucosal inflammation with atypical epithelial hyperplasia that closely mimicked primary neoplastic strictures. This observation underscores the importance of including AIP-2 in the differential diagnosis of CBD strictures as it can be easily misdiagnosed as a pancreaticobiliary malignancy, leading to unnecessary surgery[7,8]. To enhance diagnostic precision and avoid unnecessary surgery, a strong clinical suspicion of AIP-2 is essential, particularly in IgG4-negative biliary strictures. It is advisable to undergo positron emission tomography/computed tomography and histology via en
Obstructive jaundice with biliary stricture and wall thickening in AIP-2 often mimics malignancy, leading to unnecessary resection. Therefore, incorporating AIP-2 as a crucial differential diagnosis in IgG4-negative cases is essential to prevent unwarranted surgery. A repeat biopsy of the pancreatic or biliary duct and a closely monitored steroid trial should be considered for diagnostic clarification.
| 1. | Li Y, Song H, Meng X, Li R, Leung PSC, Gershwin ME, Zhang S, Sun S, Song J. Autoimmune pancreatitis type 2 (idiopathic duct-centric pancreatitis): A comprehensive review. J Autoimmun. 2023;140:103121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 2. | Hart PA, Levy MJ, Smyrk TC, Takahashi N, Abu Dayyeh BK, Clain JE, Gleeson FC, Pearson RK, Petersen BT, Topazian MD, Vege SS, Zhang L, Chari ST. Clinical profiles and outcomes in idiopathic duct-centric chronic pancreatitis (type 2 autoimmune pancreatitis): the Mayo Clinic experience. Gut. 2016;65:1702-1709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 3. | Shimosegawa T, Chari ST, Frulloni L, Kamisawa T, Kawa S, Mino-Kenudson M, Kim MH, Klöppel G, Lerch MM, Löhr M, Notohara K, Okazaki K, Schneider A, Zhang L; International Association of Pancreatology. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas. 2011;40:352-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1338] [Cited by in RCA: 1086] [Article Influence: 72.4] [Reference Citation Analysis (3)] |
| 4. | Gallo C, Dispinzieri G, Zucchini N, Invernizzi P, Massironi S. Autoimmune pancreatitis: Cornerstones and future perspectives. World J Gastroenterol. 2024;30:817-832. [PubMed] [DOI] [Full Text] |
| 5. | Patel Z, Patel S, Grendell J, Marciano T. Type 2 autoimmune pancreatitis: case report of a 9-year-old female and a review of the literature. Clin J Gastroenterol. 2015;8:421-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 6. | de Pretis N, Frulloni L. Autoimmune pancreatitis type 2. Curr Opin Gastroenterol. 2020;36:417-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Kamisawa T, Chari ST, Lerch MM, Kim MH, Gress TM, Shimosegawa T. Recent advances in autoimmune pancreatitis: type 1 and type 2. Gut. 2013;62:1373-1380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 123] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 8. | Maeda N, Tanoue S, Fujino Y, Hinokuchi M, Arima S, Sasaki F, Hashimoto S, Kanmura S, Higashi M, Ido A. Focal Type 2 Autoimmune Pancreatitis Mimicking Pancreatic Cancer: Treatment with Neoadjuvant Chemotherapy and Surgery. Intern Med. 2023;62:2499-2505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Christodoulidis G, Kouliou MN, Koumarelas KE. Understanding autoimmune pancreatitis: Clinical features, management challenges, and association with malignancies. World J Gastroenterol. 2024;30:2091-2095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 10. | Zavrtanik H, Tomažič A. Is Surgery in Autoimmune Pancreatitis Always a Failure? Medicina (Kaunas). 2023;59:193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |