Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.116781
Revised: December 31, 2025
Accepted: January 28, 2026
Published online: April 28, 2026
Processing time: 143 Days and 4.1 Hours
Postoperative recurrence (POR) of Crohn’s disease (CD) is a major therapeutic challenge.
To evaluate ustekinumab (UST) for the POR management in CD in a Chinese multicenter cohort.
This retrospective analysis included postoperative CD patients with ≥ 2 high-risk features receiving UST at four tertiary centers. Patients were stratified into a first-line group (biologic-naïve, n = 45) and a second-line group (prior biologic failure, n = 26). The primary outcome was endoscopic improvement (≥ a 1-point reduction in the Rutgeerts score). Multivariate generalized estimating equations (GEE) were used to identify therapeutic response predictors.
Overall, 81.7% achieved endoscopic improvement, with 36.6% achieving complete remission. Clinical remission reached 91.5% with a significant Harvey-Bradshaw Index reduction (5.5 ± 2.8 to 2.1 ± 1.7, P value < 0.001). The GEE multivariate analysis identified first-line UST therapy (OR = 1.727, 95%CI: 1.042-2.862, P = 0.034) and shorter disease duration (OR = 0.979, 95%CI: 0.967-0.992, P = 0.001) as independent predictors of endoscopic improvement. Significant improvements were observed in nutritional parameters (i.e., albumin and hemoglobin) and inflammatory markers (i.e., C-reactive protein and fecal occult blood positivity). Adverse events occurred in 9.9% of patients. These were all mild-to-moderate severity.
UST demonstrated significant effectiveness and a favorable safety profile for the management of POR in CD. These findings support its use as an effective therapeutic option, particularly under a first-line setting, and provide valuable evidence to optimize postoperative management strategies for CD recurrence in Chinese populations.
Core Tip: This multicenter study demonstrated that ustekinumab (UST) effectively addressed the management of postoperative recurrence in patients with Crohn's disease in China. Results showed high rates of endoscopic (81.7%) and clinical remission (91.5%) at 24 weeks. A multivariate analysis identified first-line UST use and shorter disease duration as independent predictors of superior outcomes. The favorable safety profile supports UST as a potent therapeutic option, particularly when initiated early as a first-line biologic strategy for postoperative management.
- Citation: Ma XZ, Han XZ, Zhang WY, Tian F, Zhou XC, Zhou LY, Teng YS, Lei L, Sheng JQ, Jin P, Zhao XM, Jia Y. Effectiveness of ustekinumab in postoperative Crohn's disease management: Evidence from a Chinese multicenter cohort. World J Gastroenterol 2026; 32(16): 116781
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/116781.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.116781
Crohn's disease (CD) is a chronic, relapsing inflammatory bowel disease characterized by transmural inflammation that can affect any segment of the gastrointestinal tract[1]. While its global burden is significant, the rising incidence and prevalence of CD in China present a growing clinical challenge. Additionally, emerging evidence suggests potential distinctions in its etiopathogenesis and presentation compared to those of Western populations[2-4]. Intestinal resection remains a crucial intervention for the management of complications such as strictures or penetrating disease[1]. Population-based studies from Europe have indicated that five-year surgical rates range from 12%-27%, with 30%-50% of patients with CD eventually requiring surgery during their disease course[5]. Consistent with this pattern, a large Chinese cohort study reported that 41.9% of patients with CD underwent at least one CD-related surgery during a median follow-up of 102 months[6].
Surgery, is not curative. Postoperative recurrence (POR) represents a fundamental challenge in long-term CD management[7]. Endoscopic recurrence rates can reach 70%-90% within the first year after ileocolonic resection without prophylactic therapy[7]. The recurrence process typically begins with histological inflammation, progresses to endoscopic lesions, and may eventually manifest as clinical or surgical recurrence. Early ileocolonoscopy within 6-12 months after surgery is therefore crucial to identify endoscopic predictors of clinical recurrence and to adjust treatment strategies accordingly[8,9]. The Rutgeerts scoring system is a valuable risk stratification tool, and even mild ileal lesions predicting future clinical recurrence[7,10]. These remains establish an urgent need for effective prophylactic strategies in the postoperative setting. Anti-TNF-α agents have been a cornerstone for this purpose; but approximately one-fifth of patients with CD still experience primary non-response to anti-TNF therapy. Also, an additional 10%-15% experience treatment failure annually, creating a significant therapeutic gap[11].
Ustekinumab (UST) is a fully human monoclonal antibody that targets the shared p40 subunit of interleukin (IL)-12 and IL-23. It has emerged as a highly effective therapeutic for moderate-to-severe CD[12]. Its efficacy in inducing and maintaining remission in biologic-naïve and biologic-experienced patients has been firmly established in the landmark UNITI/IM-UNITI trials[12-14]. Consequently, UST is a promising candidate for POR mitigation. However, the existing body of literature regarding the application of UST, specifically in the postoperative setting, remains relatively scarce and heterogeneous[15-20]. This evidence gap is particularly pronounced in Chinese populations, where differences in genetic background, gut microbiota, and clinical practice may influence treatment outcomes[9]. Current studies are often limited by small sample sizes, single-center designs, or a lack of comprehensive multivariate analyses that examine the response predictors (Supplementary Table 1).
We therefore conducted this multicenter, retrospective cohort study with the primary objective of evaluating the real-world efficacy and safety of UST for POR management in a Chinese population. We aim to rigorously assess its impact on both clinical and endoscopic outcomes and to identify key patient- and disease-related factors associated with treatment success, thereby informing optimal patient selection and clinical decision-making.
This multicenter, retrospective cohort study was conducted at four geographically diverse tertiary medical centers in China. These consisted of institutions from the north (the Seventh Medical Center of Chinese PLA General Hospital), the south (the Nanfang Hospital of Southern Medical University), the northeastern (the Shengjing Hospital of China Medical University), and the southwestern (the First Hospital of Chongqing Medical University). The study period spanning from October 2024 to January 2025. The study protocol received ethical approval (Approval No. S2024-093-01) and the informed consent was waived.
Eligible patients were adults (≥ 18 years) with CD who had underwent intestinal resection and initiated UST for postoperative management. The inclusion criteria were: (1) At least two high-risk POR factors (prior intestinal surgery, penetrating disease, active smoking, perianal disease, extensive lesions, or residual activities); (2) UST initiation ≥ 2 weeks post-surgery; (3) Completion of ≥ 3 UST doses; and (4) Available follow-up colonoscopy and clinical data approximately week 24 post-UST initiation. Patients were stratified into two groups based on their postoperative biologic treatment history: The first-line group, was defined as patients for whom UST was the first biologic agent initiated after the index surgery (regardless of the pre-operative biologic exposure), and the second-line group, was defined as patients who had received and failed at least one other biologic agent (e.g., anti-TNFs) after the index surgery prior to switching to UST. Patients with incomplete electronic medical records (EMRs) were excluded.
UST was administered according to the following standard regimen: An intravenous induction dose of approximately 6 mg/kg, followed by a 90 mg subcutaneous injection at week 8. Subsequent maintenance therapy involved subcutaneous 90 mg injections, with all patients in this cohort receiving their third dose at week 16, aligning with an eight-week interval. Treatment decisions, included the choice of UST as first- or second-line therapy, and they were made at the discretion of the treating gastroenterologist based on individual patient profiles.
Data were systematically extracted from the EMRs of the participating centers using a standardized case report form. The collected variables included the following: (1) Demographics and history: Age, sex, body mass index (BMI), smoking status, and comorbidities; (2) Disease characteristics: Disease duration, Montreal classification (i.e., age at diagnosis: A1 ≤ 16, A2 17-40, A3 > 40; location: L1/L2/L3/L4; and behavior: B1/B2/B3), history of perianal disease, and extraintestinal manifestations; (3) Treatment history: Number and timing of previous CD-related surgeries, prior exposure to biologics or immunomodulators, and the interval from the most recent surgery to the initiation of UST; and (4) Laboratory parameters: C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), albumin, hemoglobin, and fecal occult blood test (FOBT) results at baseline and follow-up.
The primary outcome was endoscopic improvement, that was defined as a reduction of at least one point in the Rutgeerts score from baseline to the week 24 follow-up colonoscopy. A centralized consensus review was conducted to minimize the inter-observer variability inherent to multicenter studies. De-identified endoscopic images and relevant clinical records were collated and reviewed jointly by a panel of senior inflammatory bowel disease specialists from the four participating centers (Tian F, Zhou XC, Jia Y, and Zhao XM) via a secure online conferencing platform. The panel verified the Rutgeerts scores according to standard definitions. In instances where the retrospective EMR description was ambiguous or differed from the image review, the final score was determined by the panel’s consensus. Secondary outcomes encompassed multiple dimensions of treatment responses: (1) Clinical efficacy, assessed through changes in the Harvey-Bradshaw Index (HBI) score, with clinical remission defined as HBI ≤ 4 and active disease as an HBI > 4; (2) Laboratory parameters that included changes in inflammatory markers (i.e., CRP and ESR), nutritional indicators (i.e., albumin and hemoglobin), and FOBT positivity; and (3) A safety profile, that was evaluated through the adverse events (AEs) incidence and characteristics documented during the follow-up period.
Statistical analyses were performed using SPSS (version 26.0, IBM Corp., Armonk, NY, United States) for data management and analyses, and R (version 4.2.0, R Foundation for Statistical Computing, Vienna, Austria) for visua
A multivariate analysis was performed using generalized estimating equations (GEE) to identify factors independently associated with the primary outcome of endoscopic improvement. The GEE model was selected specifically to account for the potential clustering of outcomes within patients from the same medical center. This is a crucial consideration for multicenter cohort studies[21]. The model utilized a binomial distribution, a logit link function, and an exchangeable working correlation structure. Variable selection was based on a combination of clinical relevance and statistical screening. Specifically, the model was adjusted for the following: (1) The full Montreal classification (i.e., age, location, behavior, and perianal disease) to characterize the disease phenotype; (2) Potential confounders that showed baseline imbalances between groups, namely, the disease duration and extraintestinal manifestations; and (3) The baseline endoscopic severity (i.e., the pre-treatment Rutgeerts score). The treatment line (first-line vs. second-line) was the primary exposure of interest. While variables such as time from surgery were considered, they were excluded from the final multivariate model to avoid multicollinearity with the treatment line. Results are presented as ORs with 95%CIs. A two-sided P value of < 0.05 defined statistical significance.
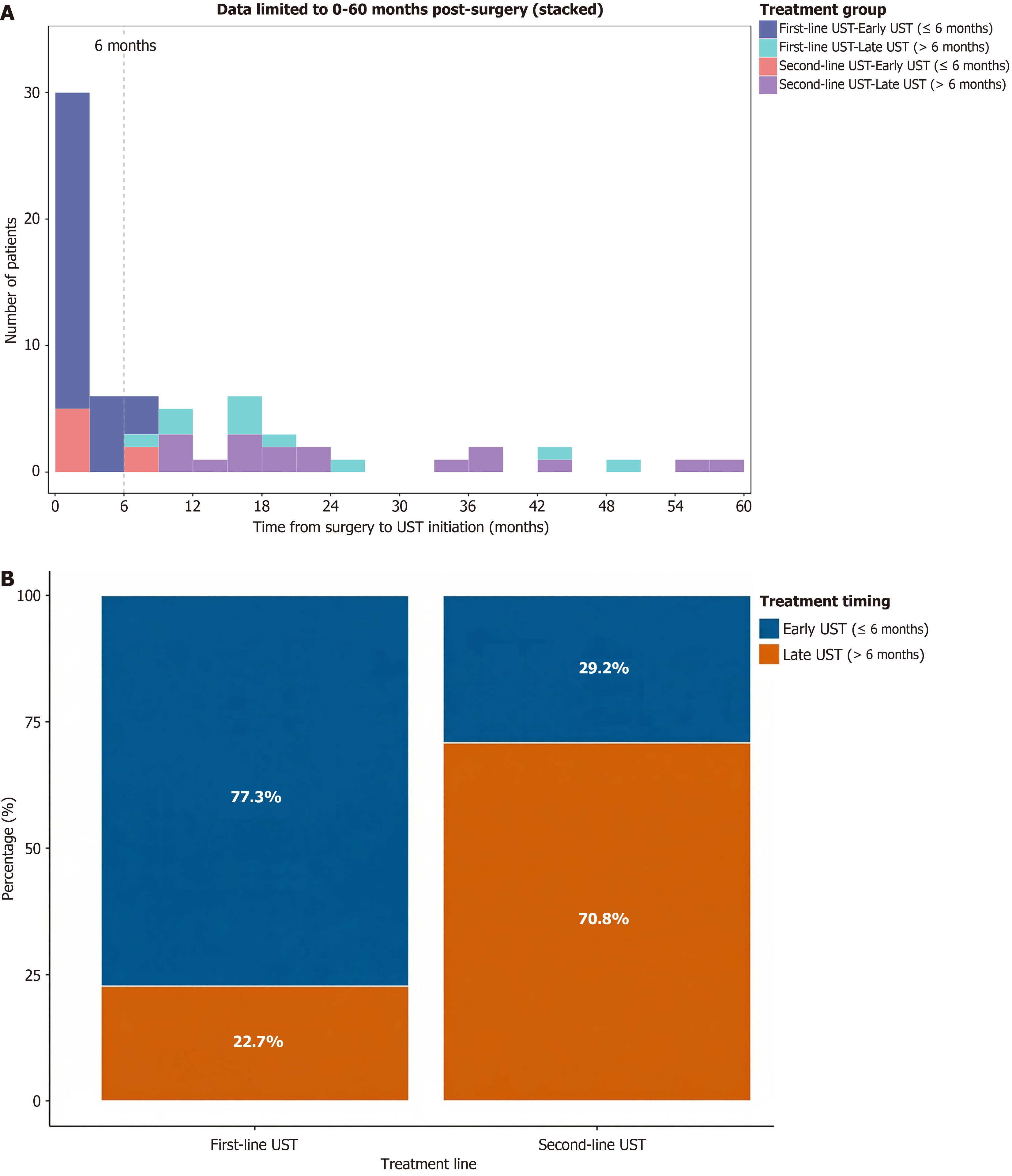
A total of 71 postoperative CD patients treated with UST were included in this multicenter retrospective analysis. Baseline characteristics stratified by treatment lines are detailed in Table 1. The cohort had a mean age of 35.4 ± 11.8 years with male predominance (74.6%). Significant differences emerged between first-line (63.4%, 45/71) and second-line (36.6%, 26/71) groups. The disease durations was longer in second-line patients (median 9.5 years vs 4.0 years, P < 0.001). Ileal disease (L1) was more common in second-line patients (46.2% vs 20.0%), while ileocolonic disease (L3) predominated in first-line patients (71.1% vs 42.3%, P = 0.045). Extraintestinal manifestations were more frequent with second-line therapy (23.1% vs 4.4%, P = 0.024). All of the second-line patients had prior biologic exposure vs 11.1% in the first-line patients (P < 0.001). Notably, the vast majority of patients received UST as biologic monotherapy during the post-operative follow-up period. Concomitant corticosteroids, immunomodulators, and 5-ASA were generally discontinued upon UST initiation to strictly evaluate its therapeutic efficacy, and thus were not analyzed as confounding variables. The surgery-to-UST interval was significantly shorter in first-line patients (median 2.0 months vs 16.5 months, P < 0.001, Figure 1).
| Characteristic | Overall (n = 71) | First-line UST (n = 45) | Second-line UST (n = 26) | P value |
| Demographics | ||||
| Age (years) | 35.4 ± 11.8 | 34.2 ± 11.0 | 37.5 ± 12.9 | 0.260 |
| Male sex | 53 (74.6) | 35 (77.8) | 18 (69.2) | 0.425 |
| Occupation | 0.763 | |||
| Employee | 35 (49.3) | 22 (48.9) | 13 (50.0) | |
| Student | 11 (15.5) | 8 (17.8) | 3 (11.5) | |
| Unemployed | 25 (35.2) | 15 (33.3) | 10 (38.5) | |
| Comorbidities | ||||
| Tuberculosis history | 10 (14.1) | 8 (17.8) | 2 (7.7) | 0.209 |
| Hepatitis B history | 3 (4.2) | 3 (6.7) | 0 (0.0) | 0.248 |
| Cancer history | 2 (2.8) | 1 (2.2) | 1 (3.8) | 0.690 |
| Disease characteristics | ||||
| Disease duration (years) | 5.0 (2.0-10.0) | 4.0 (2.0-6.0) | 9.5 (5.3-17.0) | < 0.001 |
| Montreal age classification | 0.916 | |||
| A1 (≤ 16 years)1 | 7 (9.9) | 4 (8.9) | 3 (11.5) | |
| A2 (17-40 years) | 51 (71.8) | 33 (73.3) | 18 (69.2) | |
| A3 (> 40 years) | 13 (18.3) | 8 (17.8) | 5 (19.2) | |
| Montreal location | 0.045 | |||
| L1 (ileal) | 21 (29.6) | 9 (20.0) | 12 (46.2) | |
| L2 (colonic) | 7 (9.9) | 4 (8.9) | 3 (11.5) | |
| L3 (ileocolonic) | 43 (60.6) | 32 (71.1) | 11 (42.3) | |
| L4 (upper GI) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |
| Montreal behavior | 0.727 | |||
| B1 (non-stricturing, non-penetrating) | 6 (8.5) | 3 (6.7) | 11 (24.4) | |
| B2 (stricturing) | 47 (66.2) | 31 (68.9) | 16 (61.5) | |
| B3 (penetrating) | 18 (25.4) | 11 (24.4) | 7 (26.9) | |
| Perianal disease | 24 (33.8) | 16 (35.6) | 8 (30.8) | 0.681 |
| Extraintestinal manifestations | 8 (11.3) | 2 (4.4) | 6 (23.1) | 0.024 |
| Smoking history | 9 (12.7) | 8 (17.8) | 1 (3.8) | 0.087 |
| Alcohol history | 4 (5.6) | 4 (8.9) | 0 (0.0) | 0.153 |
| History of biologic use | ||||
| Previous biologic exposure | < 0.001 | |||
| None | 41 (57.7) | 40 (88.9) | 1 (3.8) | |
| Infliximab only | 20 (28.2) | 4 (8.9)2 | 16 (61.5) | |
| Adalimumab only | 3 (4.2) | 0 (0.0) | 3 (11.5) | |
| Infliximab and adalimumab | 4 (5.6) | 1 (2.2)2 | 3 (11.5) | |
| Infliximab and vedolizumab | 2 (2.8) | 0 (0) | 2 (7.7) | |
| Infliximab, adalimumab and vedolizumab | 1 (1.4) | 0 (0.0) | 1 (2.8) | |
| Treatment timing | < 0.001 | |||
| Time from surgery to UST (months) | 1.0 (1.0-17.0) | 2.0 (1.0-6.0) | 16.5 (6.0-36.0) |
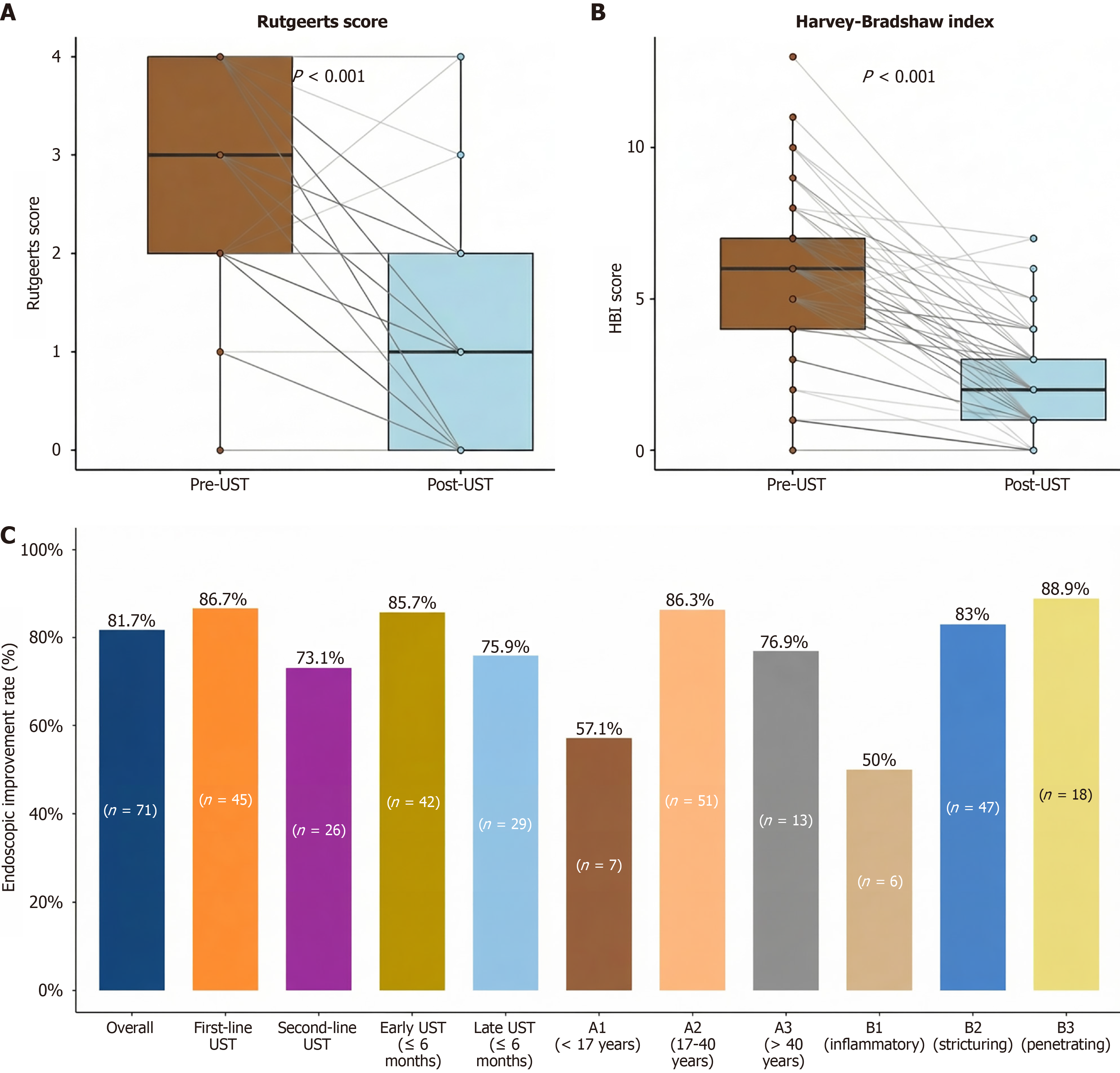
UST treatment resulted in significant improvements for the total cohort (n = 71). Their endoscopic evaluations demonstrated substantial improvements, with the mean Rutgeerts score decreasing from 2.7 ± 1.1 to 1.1 ± 1.1 (P < 0.001). Notably, 81.7% of patients achieved endoscopic improvement, including 36.6% who attained complete endoscopic remission (Rutgeerts i0; Figure 2A). Clinically, the UST treatment induced a robust response. This was evidenced by a pronounced reduction in the mean HBI score from 5.5 ± 2.8 to 2.1 ± 1.7 (P < 0.001, Figure 2B).
Efficacy outcomes were further stratified by treatment lines according to the baseline differences (Table 2). In the first-line group (postoperative biologic-naïve), the UST treatment induced profound improvements across all clinical, endoscopic, and laboratory domains. The mean Rutgeerts score decreased significantly from 2.7 ± 1.1 to 0.9 ± 1.1 (P < 0.001), with an endoscopic improvement rate of 86.7% (39/45). Notably, 77.8% (35/45) of patients in this group achieved or maintained endoscopic remission (Rutgeerts < i2) at week 24. Clinically, the mean HBI score dropped from 5.6 ± 3.0 to 1.8 ± 1.5 (P < 0.001), and the proportion of patients with active disease fell dramatically from 71.1% to 4.4% (P < 0.001). Furthermore, all of the laboratory parameters, including inflammatory markers (i.e., CRP and ESR) and nutritional indices (i.e., albumin, hemoglobin and BMI), showed statistically significant normalization (all P < 0.01). In the second-line group (rescue therapy), UST also demonstrated effectiveness, particularly in endoscopic and clinical disease control. This result was despite a more refractory disease phenotype. The mean Rutgeerts score decreased from 2.7 ± 1.0 to 1.4 ± 1.1 (P < 0.001), and 73.1% (19/26) of patients achieved endoscopic improvement. The HBI score significantly improved from 5.2 ± 2.5 to 2.8 ± 2.0 (P < 0.001), with active disease rates reducing from 73.1% to 15.4% (P < 0.001). However, unlike the first-line group, improvements in systemic inflammatory and nutritional biomarkers (i.e., CRP, ESR, albumin and emoglobin) did not reach statistical significance (P > 0.05) in this subgroup. This result likely reflected the higher inflammatory burden and chronicity associated with second-line patients. The outcomes at week 24 were then compared, and the first-line group achieved significantly lower absolute HBI scores (1.8 vs 2.8, P = 0.019) and Rutgeerts scores (0.9 vs 1.4, P = 0.039) compared to the second-line group. The endoscopic improvement rate was numerically higher in the first-line group (86.7% vs 73.1%), but this difference was not statistically significant (P = 0.154).
| Parameter | First-line group (n = 45) | Second-line group (n = 26) | P value4 | ||
| Before UST | After UST | Before UST | After UST | ||
| Clinical outcomes | |||||
| HBI score | 5.6 ± 3.0 | 1.8 ± 1.5 | 5.2 ± 2.5 | 2.8 ± 2.0 | 0.019 |
| P value1 | < 0.001 | < 0.001 | |||
| Active disease2 | 32 (71.1) | 2 (4.4) | 19 (73.1) | 4 (15.4) | 0.110 |
| P value1 | < 0.001 | < 0.001 | |||
| Endoscopic outcomes | |||||
| Rutgeerts score | 2.7 ± 1.1 | 0.9 ± 1.1 | 2.7 ± 1.0 | 1.4 ± 1.1 | 0.039 |
| P value1 | < 0.001 | < 0.001 | |||
| Endoscopic improvement3 | N/A | 39 (86.7) | N/A | 19 (73.1) | 0.154 |
| Endoscopic remission (< i2) | N/A | 35 (77.8) | N/A | 15 (57.7) | 0.074 |
| BMI (kg/m2) | 18.7 ± 2.8 | 20.1 ± 3.0 | 20.7 ± 4.2 | 21.8 ± 3.7 | 0.037 |
| P value1 | < 0.001 | 0.003 | |||
| Laboratory parameters | |||||
| CRP (mg/L) | 7.1 ± 9.2 | 3.7 ± 3.8 | 7.8 ± 11.1 | 4.6 ± 4.0 | 0.378 |
| P value1 | 0.008 | 0.152 | |||
| ESR (mm/hour) | 20.2 ± 16.7 | 12.3 ± 13.7 | 19.1 ± 18.1 | 13.3 ± 17.2 | 0.558 |
| P value1 | 0.005 | 0.124 | |||
| Albumin (g/L) | 40.2 ± 14.7 | 43.7 ± 14.6 | 40.7 ± 4.0 | 41.2 ± 3.6 | 0.438 |
| P value1 | < 0.001 | 0.615 | |||
| Hemoglobin (g/L) | 110.5 ± 21.8 | 125.2 ± 21.9 | 119.1 ± 21.1 | 125.2 ± 25.8 | 0.734 |
| P value1 | < 0.001 | 0.088 | |||
| Fecal occult blood positive | 29 (64.4) | 9 (20.0) | 14 (53.8) | 8 (30.8) | 0.306 |
| P value1 | < 0.001 | 0.092 | |||
A GEE model was constructed to identify the factors independently associated with endoscopic improvement (i.e., decrease in Rutgeerts score), adjusting for confounders such as disease duration and extraintestinal manifestations (Table 3). Patients who received the first-line UST therapy had significantly higher odds of achieving endoscopic improvement compared to those who received it as a second-line agent (OR = 1.727, 95%CI: 1.042-2.862, P = 0.034). Furthermore, several Montreal classification components were significantly associated with the outcome. Patients in the A2 (OR = 11.149, 95%CI: 1.237-100.482, P = 0.032) and A3 (OR = 2.953, 95%CI: 1.637-5.329, P < 0.001) age groups had significantly higher of improvement odds compared to that of the A1 group. Regarding disease behavior, both stricturing (B2; OR = 3.764, 95%CI: 1.031-13.742, P = 0.045) and penetrating (B3; OR = 11.692, 95%CI: 1.107-123.521, P = 0.041) phenotypes were positive predictors compared to the non-stricturing, non-penetrating (B1) phenotype. Conversely, a longer disease duration was negatively associated with the likelihood of endoscopic improvement (OR = 0.979 per year, 95%CI: 0.967-0.992, P = 0.001). The disease location (L1-L3), perianal disease presence, extraintestinal manifestations, and the pre-treatment Rutgeerts score were not independent predictors in this model. The endoscopic improvement rates across these key subgroups are further illustrated in Figure 2C.
| Variable | β | Robust SE | Wald χ² | OR (95%CI) | P value |
| Intercept | -3.709 | 1.351 | 7.532 | 0.025 (0.002-0.346) | 0.006 |
| Treatment line | |||||
| First-line UST therapy (vs second-line) | 0.546 | 0.258 | 4.498 | 1.727 (1.042-2.862) | 0.034 |
| Montreal classification | |||||
| Age A2 (17-40 years) vs A1 (≤ 16 years) | 2.411 | 1.122 | 4.621 | 11.149 (1.237-100.482) | 0.032 |
| Age A3 (> 40 years) vs A1 (≤ 16 years) | 1.083 | 0.301 | 12.927 | 2.953 (1.637-5.329) | < 0.001 |
| Location L2 (colonic) vs L1 (ileal) | 1.171 | 1.014 | 1.334 | 3.225 (0.442-23.527) | 0.248 |
| Location L3 (ileocolonic) vs L1 (ileal) | 1.342 | 1.022 | 1.725 | 3.827 (0.517-28.348) | 0.189 |
| Behavior B2 (stricturing) vs B1 (non-stricturing, non-penetrating) | 1.325 | 0.661 | 4.023 | 3.764 (1.031-13.742) | 0.045 |
| Behavior B3 (penetrating) vs B1 (non-stricturing, non-penetrating) | 2.459 | 1.203 | 4.179 | 11.692 (1.107-123.521) | 0.041 |
| Perianal disease (yes vs no) | -1.208 | 1.165 | 1.074 | 0.299 (0.030-2.934) | 0.300 |
| Other clinical factors | |||||
| Extraintestinal manifestations (yes vs no) | 0.613 | 0.459 | 1.783 | 1.847 (0.750-4.544) | 0.182 |
| Disease duration (years) | -0.021 | 0.007 | 10.431 | 0.979 (0.967-0.992) | 0.001 |
| Pre-UST Rutgeerts score | 0.607 | 0.331 | 3.369 | 1.835 (0.960-3.509) | 0.066 |
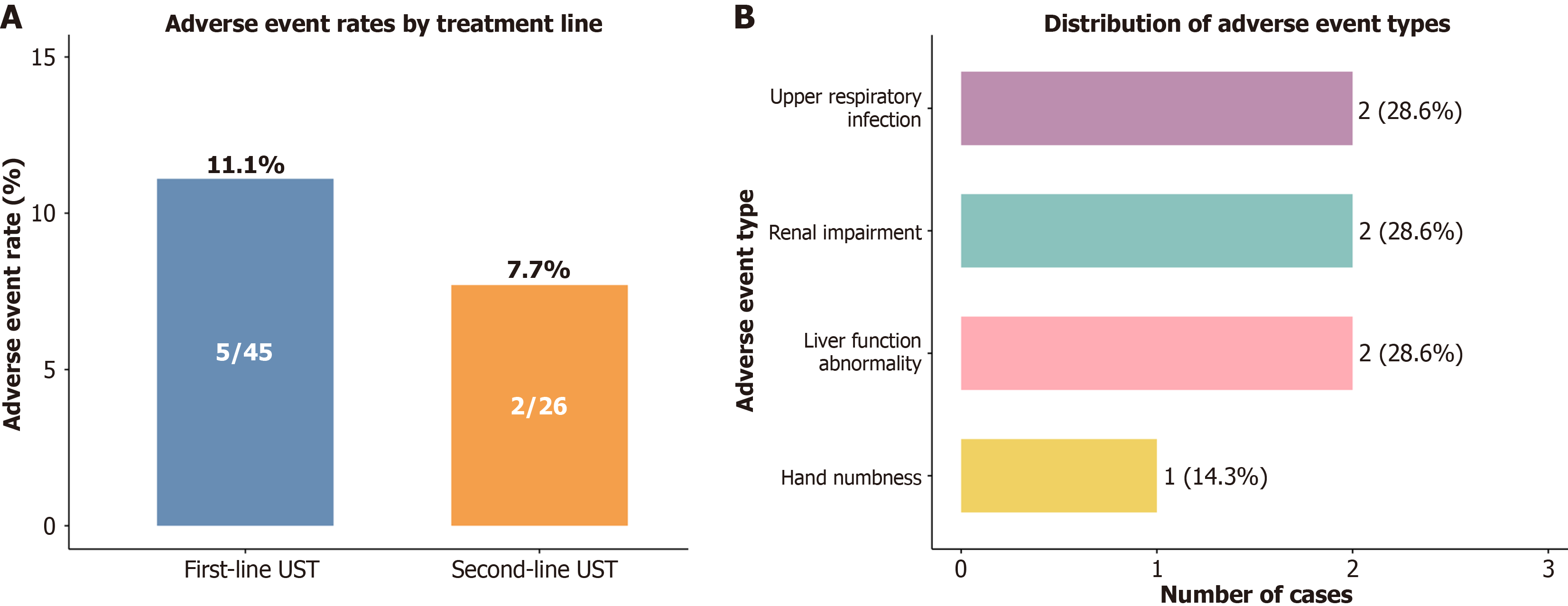
The UST safety profile in this postoperative CD population was favorable. AEs over the study period were reported in 7 out of 71 patients (9.9%). The incidence was comparable between the first-line and second-line treatment groups (11.1% vs 7.7%; Figure 3A). The AEs spectrum included upper respiratory infection (2.8%, 2/71), renal impairment (2.8%, 2/71), liver function abnormality (2.8%, 2/71), and hand numbness (1.4%, 1/71; Figure 3B). All of the reported AEs were graded as mild to moderate in severity. Critically, no serious AEs, opportunistic infections, hepatitis B reactivations, malignancies, or treatment discontinuations attributable to AEs were observed.
POR in CD remains a paramount challenge in clinical gastroenterology[7]. UST has established its efficacy in moderate-to-severe CD[12-14], but robust data on its application in the postoperative setting, particularly among Chinese populations, have been notably limited. Our multicenter, retrospective cohort study provided compelling evidence that UST is a highly effective and safe therapeutic option for POR management in Chinese patients. The significant improvements observed in endoscopic activity, clinical symptoms, and laboratory parameters, coupled with a favorable safety profile, position UST as a pivotal agent in the postoperative management arsenal. Notably, the vast majority of our patients received UST as biologic monotherapy without concomitant corticosteroids or immunomodulators during follow-up, the observed efficacy can be directly attributed to UST. This approach minimized the confounding effects of co-medication. Furthermore, our multivariate analysis delineated specific patient profiles that were most likely to benefit from this therapy, offering valuable insights for treatment personalization.
The demographic and disease characteristics of our cohort aligned with the recognized phenotype of CD that requires surgical intervention[17-20]. Our cohort existed predominantly of young males with a high prevalence of ileocolonic involvement (L3, 60.6%) and stricturing behavior (B2, 66.2%). The significant difference in disease duration and prior biologic exposure between the first-line and second-line groups accurately reflected real-world clinical practice, where patients progressed to second-line biologics often have a longer, more treatment-refractory disease course[18,19,22]. Our findings confirmed and extended the observations from smaller European studies regarding postoperative UST use[15-19]. The dramatic reduction in the median Rutgeerts score from 2.7 to 1.1, with 81.7% of patients achieving endoscopic improvement, is a cornerstone finding. This objective endoscopic efficacy is powerfully corroborated by parallel, statistically significant improvements in clinical activity (HBI score reduction from 5.5 to 2.1) and key systemic biomarkers (i.e., CRP, ESR, albumin and hemoglobin). This multi-dimensional response underscored the capacity of UST to fundamentally alter the postoperative disease course by addressing the underlying inflammatory factors.
A critical contribution of our multivariate GEE analysis was the identification of clinically relevant factors that can predict of endoscopic improvement following UST treatment in postoperative CD. Our data revealed that first-line UST therapy was an independent positive predictor of the endoscopic response. This supports a “top-down” therapeutic strategy[23] in the postoperative setting, where early intervention with high-efficacy biologics may fundamentally alter disease progression and reduce complications. It is important to interpret this finding in the context of clinical practice, where the “first-line” status is intrinsically linked to earlier intervention. The first-line patients in our cohort had significantly shorter disease durations and shorter intervals from surgery to treatment initiation compared to these of the second-line patients. There was the inherent collinearity between biologic-naïve status and early timing, the superior outcomes observed in the first-line group likely reflected the synergistic benefit of an early, first-line intervention strategy, rather than the biologic-naïve status alone. The observed effect size was consistent with the emerging real-world evidence that suggests that UST demonstrated robust efficacy in biologic-naïve patients[23,24]. This result was potentially due to an unaltered immune landscape that responded more effectively to the IL-12/23 pathway inhibition. Conversely, we found a negative association between a longer disease duration and endoscopic improvement. This reinforces the concept of a “therapeutic window of opportunity” early in the disease course[22], underscoring the importance of timely intervention after surgery[7,19]. Each additional year of disease duration was associated with a 2.1% reduction in the odds of endoscopic improvement. This highlighted how cumulative bowel damage and disease chronicity may progressively diminish treatment responsiveness[7]. This finding strongly advocated for the early UST introduction during the postoperative management algorithm to maximize the therapeutic benefit.
One of the most intriguing findings was the superior endoscopic response observed in patients with stricturing (B2) and penetrating (B3) disease behaviors compared to the purely inflammatory (B1) phenotype. This result is seemingly counterintuitive, but this may be explained by several factors. First, patients with complicated phenotypes often undergo more definitive surgical resections, which, when combined with subsequent biologic prophylaxis, may lead to more pronounced endoscopic improvements[7,23]. Second, it is plausible that UST is particularly effective at suppressing specific inflammatory pathways (e.g., IL-12/23 mediated Th1/Th17 responses) that drive these complicated phenotypes. Some studies have suggested that UST may promote transmural healing, and this could be particularly relevant in phenotypes characterized by deeper tissue involvement[15,16,18]. These findings challenged conventional perceptions and warrants further investigation into the phenotype-specific efficacy of UST.
Regarding patient age, our analysis indicated that adult patients (particularly A2, 17-40 years) demonstrated better endoscopic outcomes compared to the pediatric-onset (A1) group. This result may reflect differences in disease biology, medication compliance, or the distinct immune profiles of pediatric-onset CD[25-27]. However, the exceptionally wide confidence interval for the A2 group necessitates a cautious interpretation and likely reflects the limited subgroup sample size. Future studies with larger pediatric and A1 populations are required to validate this relationship. Notably, factors such as disease location, perianal disease, and extraintestinal manifestations did not significantly influence endoscopic outcomes in our cohort. This suggested that the efficacy of UST may be broadly applicable across various disease localizations. This would reinforce its utility in a heterogeneous patient population. The lack of a negative impact from perianal disease is particularly encouraging, as these patients often present a therapeutic challenge[28].
The favorable safety profile observed in our cohort was characterized by a 9.9% incidence of predominantly mild AEs. Crucially, all of the reported AEs were classified as mild to moderate (grade 1 or 2) according to the Common Terminology Criteria for Adverse Events, and none required therapy discontinuation. The absence of serious safety signals aligned with the established UST long-term safety data[15]. This reassuring risk-benefit profile is particularly relevant for a prophylactic or treatment strategy intended for long-term use in a postoperative population that is, by definition, in a state of clinical remission.
Our study has several limitations inherent to its retrospective design, including potential selection bias and unmeasured confounding factors, despite adjustment using the GEE. While this multicenter cohort is a significant real-world dataset in the context of postoperative UST use in China, the sample size remains relatively modest. Consequently, the statistical power to detect differences within smaller subgroups was limited, and the findings regarding specific phenotypes with small numbers (i.e., reflected by wide confidence intervals in the multivariate analysis) should be interpreted as exploratory. Second, detailed surgical variables, such as the specific type of anastomosis (e.g., side-to-side vs end-to-end), were not consistently available in the retrospective records. However, all of the surgeries were performed at tertiary referral centers following the standard bowel-sparing principles, minimizing the surgical technique as a major confounder. Additionally, as noted above, the first-line therapy group represented an early intervention strategy, and most patients had established endoscopic recurrence at baseline (mean Rutgeerts score of 2.7). This distinguished this study from a pure primary prophylaxis setting beginning at i0. Finally, the 24-week follow-up was sufficient to demonstrate the early endoscopic efficacy, but it preclude an assessment of long-term durability and impact on surgical recurrence rates.
Based on these findings, we propose the following for clinical practice. First, UST could be considered as a first-line biologic option for high-risk postoperative CD patients, given its superior efficacy in this position and excellent safety profile. Second, treatment decisions can be individualized based on the predictors of response. Biologic-naïve patients with shorter disease durations and those with stricturing or penetrating disease behavior appeared to derive the greatest benefit from UST. Third, the high efficacy rate supported a proactive postoperative management strategy, that initiates UST early after surgery and confirms the endoscopic response thereafter. Future research should prioritize prospective, randomized controlled trials that directly compare UST with anti-TNF agents under a postoperative setting. Additionally, efforts should focus on the development of integrated clinical-serological predictive models to further optimize patient selection and validate the predictors identified in this study.
In this multicenter retrospective study of Chinese patients with postoperative CD, UST demonstrated promising efficacy for POR management, as evidenced by significant improvements in endoscopic Rutgeerts scores, clinical activity, and relevant laboratory parameters, along with a favorable safety profile. These findings support its consideration as a valuable therapeutic option in the postoperative setting, particularly as a first-line biologic agent. While the retrospective design and sample size limitations require cautious interpretation, this study provides valuable real-world evidence for the application UST in this clinical context. This warrants further validation through larger prospective studies.
We extend our special gratitude to Professor Shi-Rong Li, Chief Physician of the Department of Gastroenterology at the Seventh Medical Center of the Chinese PLA General Hospital, for his guidance and assistance throughout this study. We would like to extend our special thanks to Yu-Jie Wang (Assistant Researcher, Medical Data Science Center, Beijing Tsinghua Changgung Hospital) for providing professional guidance and reviewing the statistical analysis of this study.
| 1. | Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn's disease. Lancet. 2017;389:1741-1755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2287] [Cited by in RCA: 2052] [Article Influence: 228.0] [Reference Citation Analysis (4)] |
| 2. | Yu Z, Ruan G, Bai X, Sun Y, Yang H, Qian J. Growing burden of inflammatory bowel disease in China: Findings from the Global Burden of Disease Study 2021 and predictions to 2035. Chin Med J (Engl). 2024;137:2851-2859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 3. | Gao H, Liu Z. The latest breakthrough on genetic characteristics of inflammatory bowel disease in Chinese and other East Asian ancestries. Precis Clin Med. 2023;6:pbad017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 4. | Park SB, Yoon JY, Cha JM. What Are the Different Phenotypes of Inflammatory Bowel Disease in Asia? Gut Liver. 2022;16:676-685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 5. | Aratari A, Scribano ML, Pugliese D, Baccolini V, De Biasio F, Verna S, Morretta C, Festa S, Armuzzi A, Papi C. Crohn's disease after surgery: Changes in post-operative management strategies over time and their impact on long-term re-operation rate-A retrospective multicentre real-world study. Aliment Pharmacol Ther. 2024;59:1579-1588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 6. | Bai X, Zhang H, Ruan G, Lv H, Li Y, Li J, Tan B, Zheng W, Jin M, Xu H, Yang H, Qian J. Long-term Disease Behavior and Surgical Intervention Analysis in Hospitalized Patients With Crohn's Disease in China: A Retrospective Cohort Study. Inflamm Bowel Dis. 2022;28:S35-S41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Pal P, Reddy DN, Rao GV. Endoscopic Assessment of Postoperative Recurrence in Crohn's Disease: Evolving Concepts. Gastrointest Endosc Clin N Am. 2025;35:121-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 8. | Joustra V, Duijvestein M, Mookhoek A, Bemelman W, Buskens C, Koželj M, Novak G, Hindryckx P, Mostafavi N, D'Haens G. Natural History and Risk Stratification of Recurrent Crohn's Disease After Ileocolonic Resection: A Multicenter Retrospective Cohort Study. Inflamm Bowel Dis. 2022;28:1-8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 9. | Bak MTJ, Demers K, Hammoudi N, Allez M, Silverberg MS, Fuhler GM, Parikh K, Pierik MJ, Stassen LPS, van der Woude CJ, Doukas M, van Ruler O, de Vries AC. Systematic review: Patient-related, microbial, surgical, and histopathological risk factors for endoscopic post-operative recurrence in patients with Crohn's disease. Aliment Pharmacol Ther. 2024;60:310-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 10. | Hernández-Rocha C, Walshe M, Birch S, Sabic K, Korie U, Chasteau C, Miladinova VM, Sabol WB, Mengesha E, Hanna M, Pozdnyakova V, Datta L, Kohen R, Milgrom R, Stempak JM, Bitton A, Brant SR, Rioux JD, McGovern DPB, Duerr RH, Cho JH, Schumm PL, Silverberg MS, Lazarev M. Clinical Predictors of Early and Late Endoscopic Recurrence Following Ileocolonic Resection in Crohn's Disease. J Crohns Colitis. 2024;18:615-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 11. | Barber GE, Yajnik V, Khalili H, Giallourakis C, Garber J, Xavier R, Ananthakrishnan AN. Genetic Markers Predict Primary Non-Response and Durable Response To Anti-TNF Biologic Therapies in Crohn's Disease. Am J Gastroenterol. 2016;111:1816-1822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 84] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 12. | Sandborn WJ, Rebuck R, Wang Y, Zou B, Adedokun OJ, Gasink C, Sands BE, Hanauer SB, Targan S, Ghosh S, de Villiers WJS, Colombel JF, Feagan BG, Lynch JP. Five-Year Efficacy and Safety of Ustekinumab Treatment in Crohn's Disease: The IM-UNITI Trial. Clin Gastroenterol Hepatol. 2022;20:578-590.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 182] [Article Influence: 45.5] [Reference Citation Analysis (0)] |
| 13. | Hanauer SB, Sandborn WJ, Feagan BG, Gasink C, Jacobstein D, Zou B, Johanns J, Adedokun OJ, Sands BE, Rutgeerts P, de Villiers WJS, Colombel JF, Ghosh S. IM-UNITI: Three-year Efficacy, Safety, and Immunogenicity of Ustekinumab Treatment of Crohn's Disease. J Crohns Colitis. 2020;14:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 170] [Article Influence: 28.3] [Reference Citation Analysis (3)] |
| 14. | Narula N, Wong ECL, Dulai PS, Marshall JK, Colombel JF, Reinisch W. Week 6 Calprotectin Best Predicts Likelihood of Long-term Endoscopic Healing in Crohn's Disease: A Post-hoc Analysis of the UNITI/IM-UNITI Trials. J Crohns Colitis. 2021;15:462-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 15. | Buisson A, Nancey S, Manlay L, Rubin DT, Hebuterne X, Pariente B, Fumery M, Laharie D, Roblin X, Bommelaer G, Pereira B, Peyrin-Biroulet L, Vuitton L; USTEK Post-Op study group. Ustekinumab is more effective than azathioprine to prevent endoscopic postoperative recurrence in Crohn's disease. United European Gastroenterol J. 2021;9:552-560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 16. | Yanai H, Kagramanova A, Knyazev O, Sabino J, Haenen S, Mantzaris GJ, Mountaki K, Armuzzi A, Pugliese D, Furfaro F, Fiorino G, Drobne D, Kurent T, Yassin S, Maharshak N, Castiglione F, de Sire R, Nardone OM, Farkas K, Molnar T, Krznaric Z, Brinar M, Chashkova E, Livne Margolin M, Kopylov U, Bezzio C, Bar-Gil Shitrit A, Lukas M, Chaparro M, Truyens M, Nancey S, Lobaton T, Gisbert JP, Saibeni S, Bacsúr P, Bossuyt P, Schulberg J, Hoentjen F, Viganò C, Palermo A, Torres J, Revés J, Karmiris K, Velegraki M, Savarino E, Markopoulos P, Tsironi E, Ellul P, Calviño Suárez C, Weisshof R, Ben-Hur D, Naftali T, Eriksson C, Koutroubakis IE, Foteinogiannopoulou K, Limdi JK, Liu E, Surís G, Calabrese E, Zorzi F, Filip R, Ribaldone DG, Snir Y, Goren I, Banai-Eran H, Broytman Y, Amir Barak H, Avni-Biron I, Ollech JE, Dotan I, Aharoni Golan M. Endoscopic Postoperative Recurrence in Crohn's Disease After Curative Ileocecal Resection with Early Prophylaxis by Anti-TNF, Vedolizumab or Ustekinumab: A Real-World Multicentre European Study. J Crohns Colitis. 2022;16:1882-1892. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 17. | Mañosa M, Fernández-Clotet A, Nos P, Martín-Arranz MD, Manceñido N, Carbajo A, Hinojosa E, Hernández-Camba A, Muñoz-Pérez R, Boscá-Watts M, Calvo M, Sierra-Ausín M, Sánchez-Rodríguez E, Barreiro-de Acosta M, Núñez-Alonso A, Zabana Y, Márquez L, Gisbert JP, Guardiola J, Sáinz E, Delgado-Guillena P, Busquets D, van Domselaar M, Girona E, Lorente R, Casas-Deza D, Huguet JM, Maestro S, Cabello MJ, Castro J, Iborra M, Cañete F, Calafat M, Domènech E; ENEIDA registry by GETECCU. Ustekinumab and vedolizumab for the prevention of postoperative recurrence of Crohn's disease: Results from the ENEIDA registry. Dig Liver Dis. 2023;55:46-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 18. | Macaluso FS, Grova M, Mocciaro F, Di Mitri R, Privitera AC, Distefano ME, Vitello A, Camilleri S, Ferracane C, Pluchino D, Belluardo N, Giangreco E, Fries W, Viola A, Cappello M, D'Amato L, Bertolami C, Ventimiglia M, Renna S, Casà A, D'Antonio E, De Vivo S, Orlando A; Sicilian Network for Inflammatory Bowel Disease (SN-IBD). Ustekinumab is a promising option for the treatment of postoperative recurrence of Crohn's disease. J Gastroenterol Hepatol. 2023;38:1503-1509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 19. | D'Amico F, Tasopoulou O, Fiorino G, Zilli A, Furfaro F, Allocca M, Sileri P, Spinelli A, Peyrin-Biroulet L, Danese S. Early Biological Therapy in Operated Crohn's Disease Patients Is Associated With a Lower Rate of Endoscopic Recurrence and Improved Long-term Outcomes: A Single-center Experience. Inflamm Bowel Dis. 2023;29:539-547. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 20. | Ertem FU, Rivers CR, Ghaffari AA, Watson AR, Tang G, Schwartz M, Johnston E, Barrie A, Harrison J, Dueker JM, Hartman D, Binion DG. Efficacy of Ustekinumab and Vedolizumab Among Postoperative Crohn's Disease Patients as Postoperative Prophylaxis and Rescue Therapy: Real-world Data. Inflamm Bowel Dis. 2025;31:461-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 21. | Kahan BC, Harhay MO. Many multicenter trials had few events per center, requiring analysis via random-effects models or GEEs. J Clin Epidemiol. 2015;68:1504-1511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 22. | Dolinger M, Torres J, Vermeire S. Crohn's disease. Lancet. 2024;403:1177-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 366] [Article Influence: 183.0] [Reference Citation Analysis (0)] |
| 23. | Yao L, Lin X, Tang J, Gao H, Gao X, Cao Q, Chen M. Effectiveness, treatment pattern, and safety of ustekinumab in treating bio-naïve patients with Crohn's disease in the real-world clinical setting in China. Chin Med J (Engl). 2025;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Huang Z, Diao N, Guo Q, Li M, Cheng W, Yang Q, Yang H, Huang Z, Shi L, Tang J, Gao X, Chao K. Comparative Effectiveness of Infliximab vs Ustekinumab for Endoscopic and Transmural Remission in Biologic Naïve Crohn's Disease. Clin Gastroenterol Hepatol. 2026;24:172-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 25. | Rinawi F, Zevit N, Eliakim R, Niv Y, Shamir R, Assa A. Long-Term Outcomes After Primary Bowel Resection in Pediatric-Onset Crohn's Disease. Inflamm Bowel Dis. 2017;24:149-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 26. | Sassine S, Savoie Robichaud M, Lin YF, Djani L, Cambron-Asselin C, Qaddouri M, Zekhnine S, Grzywacz K, Groleau V, Dirks M, Drouin É, Halac U, Marchand V, Girard C, Courbette O, Patey N, Dal Soglio D, Deslandres C, Jantchou P. Changes in the clinical phenotype and behavior of pediatric luminal Crohn's disease at diagnosis in the last decade. Dig Liver Dis. 2022;54:343-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 27. | Lindoso L, Mondal K, Venkateswaran S, Somineni HK, Ballengee C, Walters TD, Griffiths A, Noe JD, Crandall W, Snapper S, Rabizadeh S, Rosh JR, LeLeiko N, Guthery S, Mack D, Kellermayer R, Gulati AS, Pfefferkorn MD, Moulton DE, Keljo D, Cohen S, Oliva-Hemker M, Heyman MB, Otley A, Baker SS, Evans JS, Kirschner BS, Patel AS, Ziring D, Stephens MC, Baldassano R, Dubinsky MC, Markowitz J, Denson LA, Hyams J, Kugathasan S, Ananthakrishnan AN. The Effect of Early-Life Environmental Exposures on Disease Phenotype and Clinical Course of Crohn's Disease in Children. Am J Gastroenterol. 2018;113:1524-1529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 28. | Chapuis-Biron C, Kirchgesner J, Pariente B, Bouhnik Y, Amiot A, Viennot S, Serrero M, Fumery M, Allez M, Siproudhis L, Buisson A, Pineton de Chambrun G, Abitbol V, Nancey S, Caillo L, Plastaras L, Savoye G, Chanteloup E, Simon M, Dib N, Rajca S, Amil M, Parmentier AL, Peyrin-Biroulet L, Vuitton L; GETAID BioLAP Study Group. Ustekinumab for Perianal Crohn's Disease: The BioLAP Multicenter Study From the GETAID. Am J Gastroenterol. 2020;115:1812-1820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 76] [Article Influence: 12.7] [Reference Citation Analysis (0)] |