Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.116238
Revised: December 18, 2025
Accepted: February 4, 2026
Published online: April 28, 2026
Processing time: 162 Days and 20.1 Hours
Postoperative pancreatic fistula (POPF) is a major complication of distal pancrea
To examine the association between incidence of POPF following DP and preoperative apoA2-i.
This retrospective cohort study included 51 patients who underwent DP at Nippon Medical School Hospital between November 2021 and December 2024. Preoperative plasma apoA2-i (ATQ/ATQ and AT/AT) levels were measured using an enzyme-linked immunosorbent assay. The primary outcome was the occurrence of POPF, classified as grade BL or grade B/C according to the 2016 International Study Group of Pancreatic Surgery criteria.
Preoperative apoA2-AT/AT levels were significantly higher in patients who developed POPF (n = 30) than in those who did not (n = 21) (P < 0.001). Receiver operating characteristic curve analysis yielded an area under the curve of 0.802 [95% confidence interval (CI): 0.681-0.924] for predicting POPF. The optimal cutoff value calculated by Youden’s index was 58.17 μg/mL. Plasma apoA2-AT/AT levels ≥ 58.17 μg/mL were significantly associated with POPF (P < 0.001) and remained an independent risk factor for POPF following DP in both univariate (odds ratio = 55.7; 95%CI: 6.58-7319.4; P < 0.001) and multivariate (odds ratio = 163.9; 95%CI: 10.50-35710.29; P < 0.001) logistic regression analyses.
Preoperative measurement of apoA2-i may serve as a useful predictor of POPF following DP. These findings support its use for preoperative risk stratification and individualized perioperative management.
Core Tip: Postoperative pancreatic fistula (POPF) is a major complication after distal pancreatectomy. This retrospective cohort study of 51 patients investigated the association between POPF and preoperative plasma apolipoprotein A2 isoforms, as a marker of pancreatic exocrine function. Patients with POPF had significantly higher apoA2-AT/AT levels than those without. Receiver operating characteristic analysis yielded an area under the curve of 0.802, with optimal cutoff value of 58.17 μg/mL calculated by Youden’s index. Levels above this threshold were independently associated with POPF. Thus, preoperative apolipoprotein A2 isoforms levels may help predict POPF risk, enabling better perioperative planning and personalized care for patients undergoing distal pancreatectomy.
- Citation: Ono T, Matsushita A, Yoshimori D, Hamaguchi A, Murokawa T, Ueda J, Shimizu T, Kawano Y, Kawashima M, Nakamura Y, Nagashima K, Kashiro A, Honda K, Yoshida H. Plasma apolipoprotein A2 isoforms predict the development of pancreatic fistula following distal pancreatectomy. World J Gastroenterol 2026; 32(16): 116238
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/116238.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.116238
Distal pancreatectomy (DP) is a commonly performed surgical procedure for the treatment of benign and malignant pancreatic tumors[1,2]. Despite advances in surgical techniques and perioperative care, postoperative pancreatic fistula (POPF) remains a major complication following DP, leading to inflammation, infection, hemorrhage, and prolonged hospitalization, all of which can significantly compromise patient outcomes[3-5]. Recent studies have suggested that routine prophylactic drainage following DP may be unnecessary in selected patients at low risk of developing POPF[6-8]. These findings highlight the importance of precise preoperative risk stratification to facilitate tailored surgical approaches and minimize unnecessary procedures.
The known risk factors for POPF include patient-specific pancreatic parenchymal characteristics such as pancreatic texture and parenchymal thickness[9-11]. A soft pancreas has been consistently associated with an increased risk of POPF, possibly due to the preserved acinar cell density and secretory activity in non-fibrotic pancreatic tissue. Although such features are commonly considered surrogate markers of pancreatic exocrine function, the relationship between POPF after DP and preoperative pancreatic exocrine function has not yet been investigated.
Fecal elastase-1 testing is frequently employed as an indirect approach to evaluate pancreatic exocrine function, primarily due to its ease of use, noninvasiveness, and affordability[12]. Using this test, Speicher and Traverso[13] reported that most patients undergoing DP showed normal fecal elastase-1 levels both preoperatively and postoperatively, indicating preserved exocrine function. This finding suggests that preoperative pancreatic exocrine function is maintained even after DP and may play a significant role in the development of POPF. However, the collection of stool samples is less practical than blood testing and can be physically burdensome for patients. Moreover, since the fecal elastase test lacks approval as an in vitro diagnostic method in Japan, there has been growing interest in establishing blood-based biomarkers as a more practical alternative for assessing pancreatic exocrine function. The identification of reliable and versatile biomarkers reflecting pancreatic exocrine function is urgently required.
We recently identified blood biomarkers known as apolipoprotein A2 isoforms (apoA2-i) to detect patients with pancreatic cancer through their ability to reflect alterations in the pancreatic microenvironment predisposed to carcinogenesis[14]. In the bloodstream, apoA2-i appears in three dimeric configurations: The heavy isoform (ATQ/ATQ), the light isoform (AT/AT), and the intermediate isoform (ATQ/AT), each reflecting distinct proteolytic processing. The pancreatic enzyme carboxypeptidase A modifies apoA2 by cleaving its C-terminal residues in an atypical manner. In individuals with pancreatic exocrine insufficiency, the apoA2-AT/AT light isoform is significantly decreased compared to that in individuals with normal exocrine function[15,16].
We hypothesized that apoA2-AT/AT, the light isoform of apoA2-i, could serve as a noninvasive biomarker for assessing pancreatic exocrine function and preoperative risk of POPF following DP. To our knowledge, this is the first study to report a significant association between preoperative pancreatic exocrine function and the risk of POPF following DP using a blood-based biomarker. The study aimed to evaluate the relationship between preoperative plasma apoA2-AT/AT levels and the development of POPF following DP.
We conducted a retrospective cohort study at a single tertiary referral center to investigate the risk of developing POPF following DP. The study population comprised consecutive patients who underwent DP with informed consent between November 2021 and December 2024 at Nippon Medical School Hospital. The study was approved by the Ethics Committee of Nippon Medical School, No. A-2020-044[17]. Two independent investigators reviewed the medical records of all patients who underwent DP. The demographic and perioperative variables extracted from the institutional database and chart reviews included age, sex, body mass index, history of tobacco use, alcohol intake, diagnosis, surgical approach, and laboratory data. The pancreatic thickness (measured in a straight anterior-posterior line) and pancreatic duct diameter were determined on the last preoperative computed tomography scan at the level of the pancreatic transection line by two independent investigators.
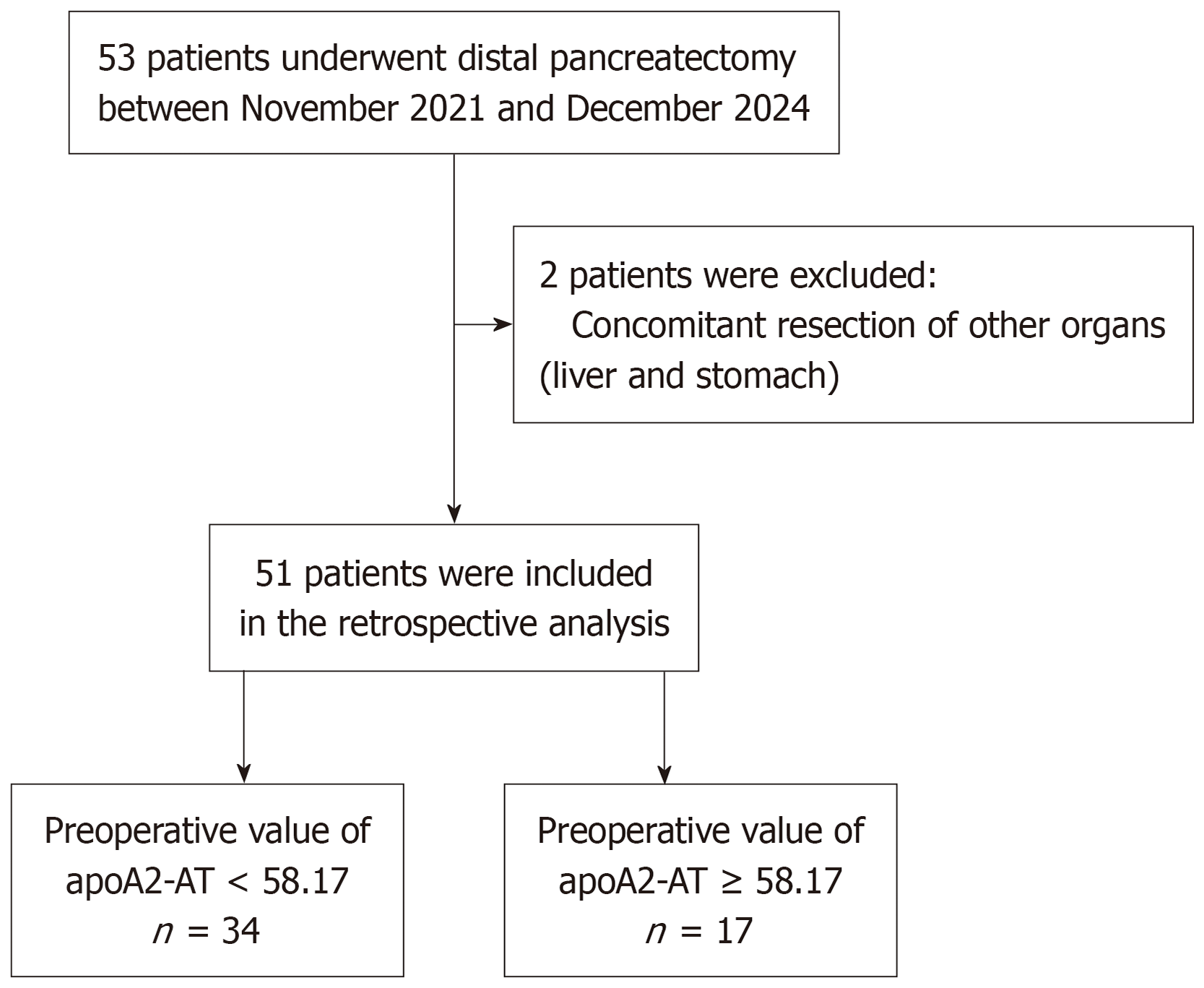
All patients who underwent DP during the study period were eligible for inclusion in this study. Written informed consent was obtained from all participants. Patients who had undergone concomitant resection of other organs were excluded from the study. The study was conducted in accordance with the principles of the Declaration of Helsinki (revised in 2013).
Preoperative plasma samples were collected as part of routine blood testing before surgery. Plasma levels of apoA2-i, including the heavier homodimer (ATQ/ATQ) and the lighter homodimer (AT/AT), were assessed using the APOA2-iTQ ELISA kit (Toray Inc., Tokyo, Japan), designed for in vitro diagnostics[14]. The amount of apoA2-ATQ/AT was derived based on established calculation methods reported in earlier studies[14].
All patients underwent DP using a stapler with a bioabsorbable reinforcement material after adequate peri-firing compression[18]. A closed silicone drain (8-mm round, multi-channel type) was placed close to the pancreatic transection margin at the end of the operation. Drain fluid amylase levels were measured on postoperative days 1, 3, and 4. The drain was removed on postoperative day 4 if the amylase levels showed a decreasing trend and there were no clinical signs of complications. Perioperative antibiotic prophylaxis was administered to all patients, consisting of intravenous cefazolin (1 g) within 30 minutes before skin incision, followed by administration every 12 hours for up to 48 hours postoperatively.
The primary outcome was the occurrence of POPF, including both grade BL and grade B/C fistulas, according to the 2016 criteria of the International Study Group of Pancreatic Surgery[19].
To evaluate the overall performance of apoA2-AT/AT, apoA2-ATQ/ATQ, and apoA2-ATQ/AT in classifying POPF, a receiver operating characteristic (ROC) curve analysis was performed, and the area under the ROC (AUC) was calculated for each isoform. Furthermore, differences in AUCs between apoA2-AT/AT, apoA2-ATQ/ATQ, and apoA2-ATQ/AT were compared using Venkatraman’s test to evaluate differences in the overall classification performance of each isoform.
The cutoff value for apoA2-AT/AT was determined using Youden’s index. Patients were divided into two groups based on this value: High (≥ 58.17 μg/mL) and low (< 58.17 μg/mL). Baseline variables were summarized as n (%) for categorical data, and mean ± SD for continuous data. Categorical variables were compared using the χ2 test or Fisher’s exact test, while continuous variables were analyzed using Welch’s t-test or the Wilcoxon rank-sum test, as appropriate. Univariate and multivariate logistic regression models were used to assess the relationship between apoA2-AT/AT levels and the occurrence of POPF. In the multivariate logistic regression model, additional factors, such as age, sex, body mass index, main pancreatic duct diameter, and parenchymal thickness, were included to adjust for potential confounders. The numbers of missing values for patient characteristics were presented; as other analyses had no missing values, imputation was not required. A two-sided P < 0.05 was considered statistically significant. All statistical analyses were performed using R software (version 4.4.3; R Foundation for Statistical Computing, Vienna, Austria).
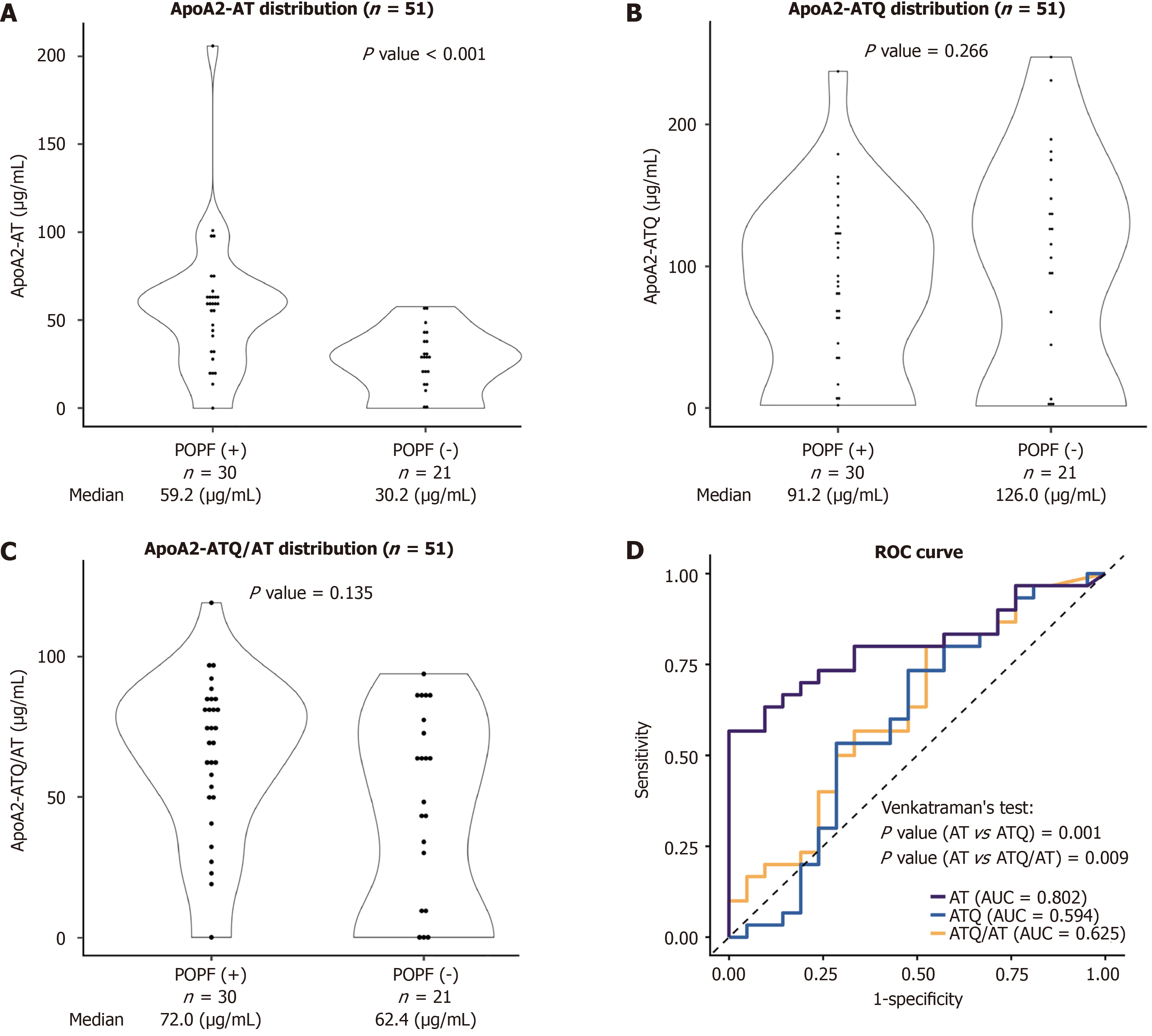
A total of 53 patients underwent DP during the study period. Among these, 2 patients were excluded from the analysis owing to combined resection of other organs, including the liver; thus, 51 patients were included in the final analysis (Figure 1). Perioperative levels of apoA2-AT/AT, apoA2-ATQ/AT, and apoA2-ATQ/ATQ were compared between patients who developed POPF (n = 30) and those who did not (n = 21). The median apoA2-AT/AT level was significantly higher in the POPF group than in the non-POPF group (median 59.2 μg/mL vs 30.2 μg/mL, Wilcoxon rank-sum test; P < 0.001; Figure 2A). By comparison, patients in the POPF group exhibited a lower median apoA2-ATQ/ATQ concentration (91.2 μg/mL) than those without POPF (126.0 μg/mL), although this difference was not statistically significant (Wilcoxon rank-sum test; P = 0.26; Figure 2B). The POPF group showed a slightly higher median apoA2-ATQ/AT level (72.0 μg/mL) compared to the non-POPF group (62.4 μg/mL), though this difference was not statistically significant (Wilcoxon rank-sum test; P = 0.135; Figure 2C). Neither apoA2-ATQ/ATQ nor apoA2-ATQ/AT levels differed significantly between the two groups. Among the isoforms, apoA2-AT/AT demonstrated the strongest predictive value for POPF, with an AUC of 0.802, outperforming apoA2-ATQ/ATQ (0.594) and apoA2-ATQ/AT (0.625; Figure 2D). Statistical comparisons revealed that apoA2-AT/AT differed significantly from apoA2-ATQ/ATQ (P = 0.001) and apoA2-ATQ/AT (P = 0.009) in its ability to predict POPF. These findings suggest that among the three isoforms, apoA2-AT/AT offers the highest predictive accuracy for POPF following DP.
ROC curve analysis of apoA2-AT/AT showed an AUC of 0.802 [95% confidence interval (CI): 0.681-0.924] (Figure 2D). Based on Youden’s index, the most effective threshold for distinguishing POPF risk was determined to be 58.17 μg/mL (Figure 2D). Based on this cutoff, patients who underwent DP were classified into two groups: A high-expression group (≥ 58.17 μg/mL; n = 17) and a low-expression group (< 58.17 μg/mL; n = 34). The baseline characteristics of the two groups are shown in Table 1. Significant differences were observed in surgical time (P = 0.011), blood loss (P = 0.028), and receipt of preoperative chemotherapy (P = 0.009) between the high and low apoA2-AT/AT expression groups (P < 0.05; Table 1).
| Variable | n | ApoA2-AT < 58.17 µg/mL, | ApoA2-AT ≥ 58.17 µg/mL, | P value1 |
| Age, years (continuous) | 51 | 66.3 ± 14.6 | 65.5 ± 16.6 | 0.873 |
| Age, years (categorical) | 51 | > 0.999 | ||
| < 65 | 13 (38.2) | 6 (35.3) | ||
| ≥ 65 | 21 (61.8) | 11 (64.7) | ||
| Sex | 51 | > 0.999 | ||
| Female | 15 (44.1) | 8 (47.1) | ||
| Male | 19 (55.9) | 9 (52.9) | ||
| BMI, kg/m2 | 51 | 21.9 ± 4.1 | 21.7 ± 3.9 | 0.907 |
| Diabetes | 51 | 10 (29.4) | 5 (29.4) | > 0.999 |
| History of epigastric laparotomy | 51 | 4 (11.8) | 4 (23.5) | 0.416 |
| History of cancer | 51 | 9 (26.5) | 6 (35.3) | 0.532 |
| Smoking | 51 | > 0.999 | ||
| No | 16 (47.1) | 8 (47.1) | ||
| Yes | 15 (44.1) | 7 (41.2) | ||
| Unknown | 3 (8.8) | 2 (11.8) | ||
| Alcohol | 51 | 0.229 | ||
| No | 18 (52.9) | 10 (58.8) | ||
| Yes | 11 (32.4) | 2 (11.8) | ||
| Unknown | 5 (14.7) | 5 (29.4) | ||
| CA19-9, U/mL | 50 | 73.8 ± 174.3 | 49.0 ± 149.0 | 0.602 |
| Missing | 1 | 0 | ||
| CEA, ng/mL | 50 | 3.1 ± 2.0 | 3.3 ± 2.0 | 0.749 |
| Missing | 1 | 0 | ||
| Total cholesterol | 49 | 178.6 ± 37.1 | 200.4 ± 42.3 | 0.090 |
| Missing | 1 | 1 | ||
| Amylase | 51 | 104.0 ± 96.3 | 101.5 ± 33.4 | 0.890 |
| Alb, g/dL | 51 | 4.0 ± 0.6 | 4.3 ± 0.7 | 0.249 |
| Splenectomy | 51 | 28 (82.4) | 12 (70.6) | 0.472 |
| ASA | 51 | 0.687 | ||
| 1 | 1 (2.9) | 1 (5.9) | ||
| 2 | 28 (82.4) | 15 (88.2) | ||
| 3 | 5 (14.7) | 1 (5.9) | ||
| Pancreatic parenchymal thickness, mm (continuous) | 51 | 10.8 ± 3.4 | 11.6 ± 4.0 | 0.482 |
| Pancreatic parenchymal thickness, mm (categorical) | 51 | > 0.999 | ||
| < 12 | 23 (67.6) | 11 (64.7) | ||
| ≥ 12 | 11 (32.4) | 6 (35.3) | ||
| MPD diameter, mm (continuous) | 51 | 2.4 (1.7) | 2.6 (1.4) | 0.645 |
| MPD diameter, mm (categorical) | 51 | 0.545 | ||
| < 3 | 22 (64.7) | 9 (52.9) | ||
| ≥ 3 | 12 (35.3) | 8 (47.1) | ||
| Surgical method | 51 | 0.129 | ||
| Laparoscopic approach | 18 (52.9) | 13 (76.5) | ||
| Open approach | 6 (17.6) | 0 (0.0) | ||
| Robot approach | 10 (29.4) | 4 (23.5) | ||
| Surgical time, minutes | 51 | 376.8 ± 220.9 | 272.0 ± 36.9 | 0.011 |
| Blood loss, mL | 51 | 443.0 ± 698.1 | 144.5 ± 221.2 | 0.028 |
| Blood transfusion | 51 | 5 (14.7) | 1 (5.9) | 0.650 |
| Preoperative chemotherapy | 51 | 11 (32.4) | 0 (0.0) | 0.009 |
All patients in the high apoA2-AT/AT group (≥ 58.17 μg/mL) developed POPF, a rate markedly greater than the 38.2% observed in the low-expression group, with statistical significance confirmed by Pearson’s χ2 test (P < 0.001; Table 2).
| Feature | n | ApoA2-AT < 58.17 µg/mL, n = 34 | ApoA2-AT ≥ 58.17 µg/mL, n = 17 | P value |
| 51 | < 0.001 | |||
| POPF (+) | 13 (38.2) | 17 (100.0) | ||
| POPF (-) | 21 (61.8) | 0 (0.0) |
As presented in Tables 3 and 4, a preoperative apoA2-AT/AT level of ≥ 58.17 μg/mL emerged as a significant predictor of POPF in the univariate model. In the multivariate model, both elevated apoA2-AT/AT levels and male sex remained independent risk factors. The corresponding odds ratios for apoA2-AT/AT ≥ 58.17 μg/mL were 55.7 (95%CI: 6.58-7319.4) in the univariate analysis and 163.39 (95%CI: 10.50-35710.29) in the multivariate analysis.
| Variable | n | OR | 95%CI | P value |
| ApoA2-AT, μg/mL | 51 | |||
| < 58.17 | Reference | |||
| ≥ 58.17 | 55.7 | 6.58-7319.4 | < 0.001 | |
| Age, years | 51 | |||
| < 65 | Reference | |||
| ≥ 65 | 0.52 | 0.15-1.68 | 0.286 | |
| Sex | 51 | |||
| Female | Reference | |||
| Male | 2.30 | 0.75-7.41 | 0.151 | |
| BMI, kg/m2 | 51 | |||
| < 25 | Reference | |||
| ≥ 25 | 1.83 | 0.44-9.40 | 0.427 | |
| Pancreatic parenchymal thickness, mm | 51 | |||
| < 12 | Reference | |||
| ≥ 12 | 0.70 | 0.21-2.29 | 0.547 | |
| MPD diameter, mm | 51 | |||
| < 3 | Reference | |||
| ≥ 3 | 1.08 | 0.35-3.48 | 0.891 |
| Variable | n | OR | 95%CI | P value |
| ApoA2-AT, μg/mL | 51 | |||
| < 58.17 | Reference | |||
| ≥ 58.17 | 163.39 | 10.50-35710.29 | < 0.001 | |
| Age, years | 51 | |||
| < 65 | Reference | |||
| ≥ 65 | 0.29 | 0.05-1.38 | 0.122 | |
| Sex | 51 | |||
| Female | Reference | |||
| Male | 7.10 | 1.38-57.93 | 0.017 | |
| BMI, kg/m2 | 51 | |||
| < 25 | Reference | |||
| ≥ 25 | 2.96 | 0.37-37.30 | 0.309 | |
| Pancreatic parenchymal thickness, mm | 51 | |||
| < 12 | Reference | |||
| ≥ 12 | 0.19 | 0.02-1.07 | 0.060 | |
| MPD diameter, mm | 51 | |||
| < 3 | Reference | |||
| ≥ 3 | 1.07 | 0.21-5.74 | 0.932 |
To the best of our knowledge, this study is the first to show that high plasma levels of apoA2-AT/AT are significantly correlated with the development of POPF in patients following DP, by measuring plasma apoA2-i levels to assess pancreatic exocrine function. Recently, several fistula risk scores for predicting POPF following DP were reported[10,11,20]. Risk stratification using preoperative risk factors for POPF following DP is necessary for optimizing postoperative management, including decisions regarding prophylactic drainage[7]. Risk stratification for POPF following DP has conventionally relied on a combination of preoperative imaging findings, clinical factors, and intraoperative assessments[9,21]. Among these, preoperative indicators such as pancreatic duct diameter and parenchymal thickness are commonly used. However, the intraoperative assessment of pancreatic texture is subjective and operator-dependent, thus limiting its reproducibility[22,23]. Furthermore, although histological evaluation of resected specimens has shown that pancreatic fibrosis is inversely correlated with the incidence of POPF[24], such information becomes available only postoperatively and, therefore, is of limited utility for preoperative decision-making.
ApoA2 is a liver-derived apolipoprotein, and its AT/AT isoform has been recognized as a blood-based biomarker of pancreatic exocrine function[14]. Previous studies have reported that patients with pancreatic exocrine insufficiency exhibit reduced carboxypeptidase A activity, leading to decreased circulating apoA2-AT/AT levels[20]. These findings underscore the need for objective and noninvasive biomarkers to assess pancreatic exocrine function before surgery. The AT/AT isoform of apoA2 likely serves as a surrogate marker for the exocrine capacity of the remaining pancreatic tissue, which is often preserved following DP. Elevated apoA2-AT/AT levels may indicate robust acinar cell activity, potentially leading to increased pancreatic secretion at the transection site, thereby contributing to the development of POPF. In patients with high apoA2-AT/AT levels, the function of pancreatic acinar cells is preserved, which may increase pancreatic juice production in the pancreatic stump; we consider this one of the important causative factors of POPF. Our findings demonstrated that patients who developed POPF had significantly elevated preoperative apoA2-AT/AT levels compared to those without POPF. ROC curve analysis identified 58.17 μg/mL as the optimal threshold to differentiate between high and low expression groups. POPF occurred more frequently in the high-expression group. Furthermore, multivariate logistic regression confirmed that elevated apoA2-AT/AT levels independently predicted POPF, even when accounting for male sex, a known contributing factor[9]. These results support the utility of apoA2-AT/AT as a blood-based biomarker for anticipating POPF risk following DP. Notably, the universal occurrence of POPF in the high-expression group underscores the potential of apoA2-AT/AT as a marker for identifying individuals at heightened risk. Therefore, these findings prompt reconsideration of both drain placement and removal strategies as part of perioperative management. Because apoA2-AT/AT reflects a biological mechanism distinct from imaging- and surgery-based parameters, it may complement existing risk models rather than replace them. In particular, apoA2-AT/AT may provide additional information regarding pancreatic functional characteristics that are not directly captured by conventional imaging-based risk scores. For example, among patients classified as having intermediate risk by the DP fistula risk score or moderate risk by the distal pancreatectomy fistula risk score, apoA2-AT/AT may help further refine risk stratification by providing information on pancreatic exocrine function.
This study has several limitations. First, although POPF was defined according to the 2016 International Study Group of Pancreatic Surgery criteria[19], both grade BL and grade B cases were included in the analysis. Although grade BL is generally regarded as a biochemical leak without clinical significance, emerging evidence suggests that some grade BL cases may represent an early stage of clinically relevant POPF, particularly depending on postoperative drain management strategies[25]. In our cohort, 6 of the 7 patients who developed grade B POPF initially met the criteria for grade BL during the early postoperative period. Including such cases may have enhanced the sensitivity of this exploratory biomarker-based analysis. Nonetheless, future investigations should aim to clearly distinguish between clinically relevant POPF and non-clinically relevant POPF to enhance the specificity and clinical applicability of predictive models.
This study was a retrospective, single-center investigation with a small sample size. The high odds ratio with wide CIs suggest the possibility of some degree of imprecision and overfitting. While the results should be interpreted with caution, they offer a compelling rationale for further research. Prospective, external multicenter studies with larger cohorts are warranted to validate the clinical utility and generalizability of apoA2-AT/AT as a preoperative biomarker for POPF risk stratification.
In conclusion, preoperative plasma apoA2-AT/AT may serve as a blood-based and objective biomarker of pancreatic exocrine function and a useful predictor of POPF following DP. These findings provide a strong rationale for further investigation, and prospective multicenter studies are warranted to validate the clinical utility of apoA2-AT/AT and facilitate its integration into perioperative risk assessment and management strategies.
| 1. | Lof S, van der Heijde N, Abuawwad M, Al-Sarireh B, Boggi U, Butturini G, Capretti G, Coratti A, Casadei R, D'Hondt M, Esposito A, Ferrari G, Fusai G, Giardino A, Groot Koerkamp B, Hackert T, Kamarajah S, Kauffmann EF, Keck T, Marudanayagam R, Nickel F, Manzoni A, Pessaux P, Pietrabissa A, Rosso E, Salvia R, Soonawalla Z, White S, Zerbi A, Besselink MG, Abu Hilal M; European Consortium on Minimally Invasive Pancreatic Surgery (E-MIPS). Robotic versus laparoscopic distal pancreatectomy: multicentre analysis. Br J Surg. 2021;108:188-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 2. | Nakamura T, Okada KI, Ohtsuka M, Higuchi R, Takahashi H, Nagai K, Unno M, Murakami Y, Oba A, Tomikawa M, Kato A, Horiguchi A, Nakamura M, Yagi S, Satoi S, Endo I, Amano R, Matsumoto I, Ito YM, Nagakawa T, Hirano S. Insights from managing clinical issues in distal pancreatectomy with en bloc coeliac axis resection: experiences from 626 patients. Br J Surg. 2023;110:1387-1394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | van der Heijde N, Lof S, Busch OR, de Hingh I, de Kleine RH, Molenaar IQ, Mungroop TH, Stommel MW, Besselink MG, van Eijck C; Dutch Pancreatic Cancer Group. Incidence and impact of postoperative pancreatic fistula after minimally invasive and open distal pancreatectomy. Surgery. 2022;171:1658-1664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 4. | Wennerblom J, Ateeb Z, Jönsson C, Björnsson B, Tingstedt B, Williamsson C, Sandström P, Ansorge C, Blomberg J, Del Chiaro M. Reinforced versus standard stapler transection on postoperative pancreatic fistula in distal pancreatectomy: multicentre randomized clinical trial. Br J Surg. 2021;108:265-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 5. | Kawai M, Hirono S, Okada K, Sho M, Nakajima Y, Eguchi H, Nagano H, Ikoma H, Morimura R, Takeda Y, Nakahira S, Suzumura K, Fujimoto J, Yamaue H. Randomized Controlled Trial of Pancreaticojejunostomy versus Stapler Closure of the Pancreatic Stump During Distal Pancreatectomy to Reduce Pancreatic Fistula. Ann Surg. 2016;264:180-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 100] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 6. | Van Buren G 2nd, Bloomston M, Schmidt CR, Behrman SW, Zyromski NJ, Ball CG, Morgan KA, Hughes SJ, Karanicolas PJ, Allendorf JD, Vollmer CM Jr, Ly Q, Brown KM, Velanovich V, Winter JM, McElhany AL, Muscarella P 2nd, Schmidt CM, House MG, Dixon E, Dillhoff ME, Trevino JG, Hallet J, Coburn NSG, Nakeeb A, Behrns KE, Sasson AR, Ceppa EP, Abdel-Misih SRZ, Riall TS, Silberfein EJ, Ellison EC, Adams DB, Hsu C, Tran Cao HS, Mohammed S, Villafañe-Ferriol N, Barakat O, Massarweh NN, Chai C, Mendez-Reyes JE, Fang A, Jo E, Mo Q, Fisher WE. A Prospective Randomized Multicenter Trial of Distal Pancreatectomy With and Without Routine Intraperitoneal Drainage. Ann Surg. 2017;266:421-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 114] [Article Influence: 12.7] [Reference Citation Analysis (1)] |
| 7. | van Bodegraven EA, Balduzzi A, van Ramshorst TME, Malleo G, Vissers FL, van Hilst J, Festen S, Abu Hilal M, Asbun HJ, Michiels N, Koerkamp BG, Busch ORC, Daams F, Luyer MDP, Ramera M, Marchegiani G, Klaase JM, Molenaar IQ, de Pastena M, Lionetto G, Vacca PG, van Santvoort HC, Stommel MWJ, Lips DJ, Coolsen MME, Mieog JSD, Salvia R, van Eijck CHJ, Besselink MG; Dutch Pancreatic Cancer Group. Prophylactic abdominal drainage after distal pancreatectomy (PANDORINA): an international, multicentre, open-label, randomised controlled, non-inferiority trial. Lancet Gastroenterol Hepatol. 2024;9:438-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 8. | Kaiser J, Niesen W, Hinz U, Diener MK, Pianka F, Klotz R, Strobel O, Mehrabi A, Berchtold C, Müller B, Schneider M, Loos M, Michalski C, Büchler MW, Hackert T, Probst P. Reconsidering Abdominal Drainage After Left Pancreatectomy: The Randomized Controlled PANDRA II Trial. Ann Surg. 2025;282:203-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 9. | Miyasaka Y, Mori Y, Nakata K, Ohtsuka T, Nakamura M. Attempts to prevent postoperative pancreatic fistula after distal pancreatectomy. Surg Today. 2017;47:416-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 10. | Bonsdorff A, Ghorbani P, Helanterä I, Tarvainen T, Kontio T, Belfrage H, Sirén J, Kokkola A, Sparrelid E, Sallinen V. Development and external validation of DISPAIR fistula risk score for clinically relevant postoperative pancreatic fistula risk after distal pancreatectomy. Br J Surg. 2022;109:1131-1139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 11. | De Pastena M, van Bodegraven EA, Mungroop TH, Vissers FL, Jones LR, Marchegiani G, Balduzzi A, Klompmaker S, Paiella S, Tavakoli Rad S, Groot Koerkamp B, van Eijck C, Busch OR, de Hingh I, Luyer M, Barnhill C, Seykora T, Maxwell T T, de Rooij T, Tuveri M, Malleo G, Esposito A, Landoni L, Casetti L, Alseidi A, Salvia R, Steyerberg EW, Abu Hilal M, Vollmer CM, Besselink MG, Bassi C. Distal Pancreatectomy Fistula Risk Score (D-FRS): Development and International Validation. Ann Surg. 2023;277:e1099-e1105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 76] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 12. | Whitcomb DC, Buchner AM, Forsmark CE. AGA Clinical Practice Update on the Epidemiology, Evaluation, and Management of Exocrine Pancreatic Insufficiency: Expert Review. Gastroenterology. 2023;165:1292-1301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 81] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 13. | Speicher JE, Traverso LW. Pancreatic exocrine function is preserved after distal pancreatectomy. J Gastrointest Surg. 2010;14:1006-1011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (3)] |
| 14. | Honda K, Kobayashi M, Okusaka T, Rinaudo JA, Huang Y, Marsh T, Sanada M, Sasajima Y, Nakamori S, Shimahara M, Ueno T, Tsuchida A, Sata N, Ioka T, Yasunami Y, Kosuge T, Miura N, Kamita M, Sakamoto T, Shoji H, Jung G, Srivastava S, Yamada T. Plasma biomarker for detection of early stage pancreatic cancer and risk factors for pancreatic malignancy using antibodies for apolipoprotein-AII isoforms. Sci Rep. 2015;5:15921. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 15. | Hayasaki A, Murata Y, Usui M, Hibi T, Fujii T, Iizawa Y, Kato H, Tanemura A, Azumi Y, Kuriyama N, Kishiwada M, Mizuno S, Sakurai H, Uchida K, Isaji S. Clinical Significance of Plasma Apolipoprotein-AII Isoforms as a Marker of Pancreatic Exocrine Disorder for Patients with Pancreatic Adenocarcinoma Undergoing Chemoradiotherapy, Paying Attention to Pancreatic Morphological Changes. Biomed Res Int. 2019;2019:5738614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 16. | Futagami S, Agawa S, Nakamura K, Watanabe Y, Habiro M, Kawawa R, Yamawaki H, Tsushima R, Kirita K, Akimoto T, Ueki N, Tomohide T, Itokawa N, Suzuki N, Naito Y, Takeuchi K, Kashiro A, Ohta R, Mizutani S, Taniai N, Yoshida H, Iwakiri K, Honda K. Apolipoprotein A2 isoforms associated with exocrine pancreatic insufficiency in early chronic pancreatitis. J Gastroenterol Hepatol. 2023;38:1949-1957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Otsuka T, Matsuyama K. Nippon Medical School's Ethical Review Processes for Studies Involving Human Subjects. J Nippon Med Sch. 2024;91:136-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 18. | Nakamura M, Ueda J, Kohno H, Aly MY, Takahata S, Shimizu S, Tanaka M. Prolonged peri-firing compression with a linear stapler prevents pancreatic fistula in laparoscopic distal pancreatectomy. Surg Endosc. 2011;25:867-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 71] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 19. | Bassi C, Marchegiani G, Dervenis C, Sarr M, Abu Hilal M, Adham M, Allen P, Andersson R, Asbun HJ, Besselink MG, Conlon K, Del Chiaro M, Falconi M, Fernandez-Cruz L, Fernandez-Del Castillo C, Fingerhut A, Friess H, Gouma DJ, Hackert T, Izbicki J, Lillemoe KD, Neoptolemos JP, Olah A, Schulick R, Shrikhande SV, Takada T, Takaori K, Traverso W, Vollmer CM, Wolfgang CL, Yeo CJ, Salvia R, Buchler M; International Study Group on Pancreatic Surgery (ISGPS). The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 Years After. Surgery. 2017;161:584-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3570] [Cited by in RCA: 3311] [Article Influence: 367.9] [Reference Citation Analysis (4)] |
| 20. | Bonsdorff A, Kjeseth T, Kirkegård J, de Ponthaud C, Ghorbani P, Wennerblom J, Williamson C, Acher AW, Thillai M, Tarvainen T, Helanterä I, Uutela A, Sirén J, Kokkola A, Sahakyan M, Kleive D, Hagen R, Lund A, Nielsen MF, Vaillant JC, Fristedt R, Biörserud C, Bratlie SO, Tingstedt B, Labori KJ, Gaujoux S, Wigmore SJ, Hallet J, Sparrelid E, Sallinen V. International multicentre validation of the left pancreatectomy pancreatic fistula prediction models and development and validation of the combined DISPAIR-FRS prediction model. Br J Surg. 2025;112:znae313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 21. | Peng YP, Zhu XL, Yin LD, Zhu Y, Wei JS, Wu JL, Miao Y. Risk factors of postoperative pancreatic fistula in patients after distal pancreatectomy: a systematic review and meta-analysis. Sci Rep. 2017;7:185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 91] [Article Influence: 10.1] [Reference Citation Analysis (5)] |
| 22. | Wellner UF, Kayser G, Lapshyn H, Sick O, Makowiec F, Höppner J, Hopt UT, Keck T. A simple scoring system based on clinical factors related to pancreatic texture predicts postoperative pancreatic fistula preoperatively. HPB (Oxford). 2010;12:696-702. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 164] [Cited by in RCA: 161] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 23. | Timme S, Kayser G, Werner M, Litkevych S, Màlyi AG, Keck T, Bronsert P, Wellner UF, Petrova E, Honselmann KC; RECOPANC Review Group. Surgeon vs Pathologist for Prediction of Pancreatic Fistula: Results from the Randomized Multicenter RECOPANC Study. J Am Coll Surg. 2021;232:935-945.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 24. | Nahm CB, Alzaabi S, Sahni S, Gill AJ, Samra JS, Mittal A. Increased postoperative pancreatic fistula rate after distal pancreatectomy compared with pancreatoduodenectomy is attributable to a difference in acinar scores. J Hepatobiliary Pancreat Sci. 2021;28:533-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Ciprani D, Bannone E, Marchegiani G, Nessi C, Salvia R, Bassi C. Progression from biochemical leak to pancreatic fistula after distal pancreatectomy. Don't cry over spilt amylase. Pancreatology. 2022;22:817-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |