Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.117396
Revised: December 29, 2025
Accepted: January 27, 2026
Published online: April 14, 2026
Processing time: 118 Days and 17.3 Hours
Metabolic dysfunction-associated steatotic liver disease (MASLD) is the most common type of chronic liver disease, encompassing a broad spectrum of pa
Core Tip: Metabolic dysfunction-associated steatotic liver disease (MASLD) encompasses a broad spectrum of pathology ranging from hepatic steatosis to metabolic dysfunction-associated steatohepatitis (MASH). Effective treatments for MASLD remain limited. Hepatic innate immune cells play an essential role in maintaining liver physiological homeostasis and contributing to MASLD pathogenesis and progression by interacting with liver parenchymal cells and adaptive immune cells in the progression of MASLD and MASH. Treatments targeting innate immune cell manipulation and metabolic modulation, such as fibroblast growth factor 21 analogues, farnesoid X receptor agonists, and chemokine receptor antago
- Citation: Yang M, Olaoba OT, Chinwo SC, LeVasseur H, Zhou B, Kimchi ET, Staveley-O’Carroll KF, Li G. Roles of hepatic immunity in metabolic dysfunction-associated steatotic liver disease: Cellular and molecular mechanisms and clinical trials. World J Gastroenterol 2026; 32(14): 117396
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/117396.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.117396
Metabolic dysfunction-associated steatotic liver disease (MASLD) is the most common type of chronic liver disease and is closely associated with obesity and type 2 diabetes (T2D)[1,2]. Its progressive form is metabolic dysfunction-associated steatohepatitis (MASH), characterized by advanced liver inflammation and cell death, as well as varying degrees of fibrosis[3,4]. Both MASLD and MASH can lead to liver cirrhosis and hepatocellular carcinoma (HCC). However, treat
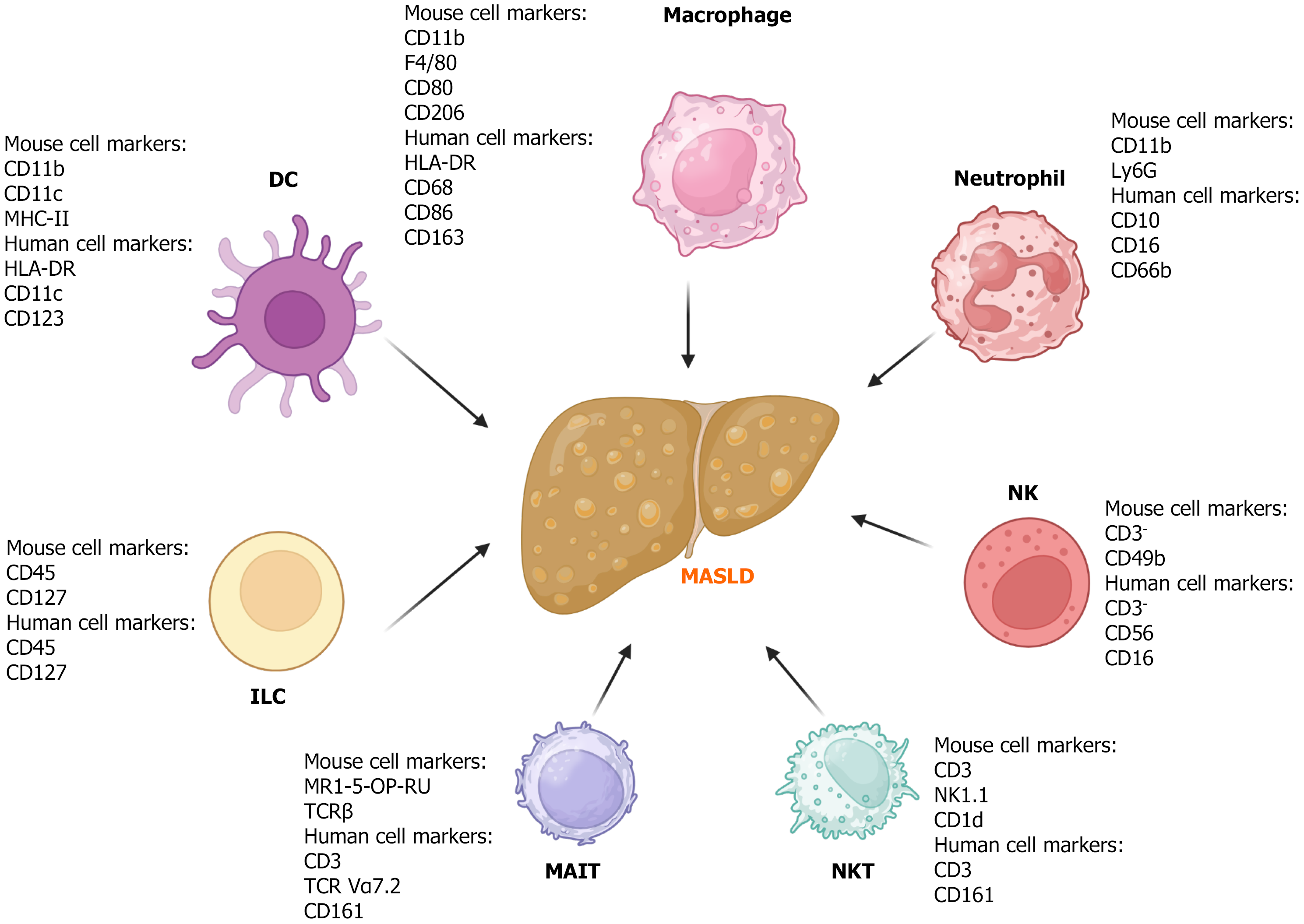
In the liver, innate immune cells primarily consist of dendritic cells (DCs), monocyte-derived macrophages (MDMs), liver-resident macrophages [Kupffer cells (KCs)], neutrophils, natural killer (NK) cells, NK T (NKT) cells, mucosal-associated invariant T (MAIT) cells, and innate lymphoid cells (ILCs). Hepatic innate immune cells comprise a large proportion of hepatic non-parenchymal cells and reciprocally communicate with injured hepatocytes to mediate the progression of liver inflammation and fibrosis during liver injury[8]. Both injured hepatocytes and activated innate immune cells (e.g., KCs) can induce the activation and proliferation of hepatic stellate cells (HSCs) and promote the recruitment of adaptive immune cells, thereby advancing liver fibrosis and cirrhosis.
Studies have used single-cell RNA sequencing (scRNA-seq) to unveil novel innate immune cell functions and effects on hepatocytes in the environment of MASH liver. For example, MASH-associated macrophages exhibit higher expression levels of triggering receptors expressed on myeloid cells 2 (Trem2) compared to KCs[9]. Another study demonstrates that lipid droplets released from damaged hepatocytes in MASH livers can substantially induce Trem2-expressing macrophages to exacerbate MASH[10]. As the most metabolically active organ in the human body, the functions of hepatic innate immune cells contribute to and are also influenced by both intrahepatic and extrahepatic factors. The activation and differentiation of innate immune cells happen in the development and progression of MASLD. In sum
Among the complex, intertwined processes involved in the progression of MASLD, hepatic lobular inflammation repre
Although elevated frequencies of conventional DCs (cDCs) and plasmacytoid DCs in the liver are the hallmarks of MASH, the role of DCs in the pathogenesis and pathophysiology of MASH is dichotomous[12,13]. Over a decade ago, Henning et al[14] reported that intrahepatic DCs expand, mature, and assume an activated phenotype in MASH[14]. Depletion of DCs using diphtheria toxin injection in CD11c-DTR mice exacerbated intrahepatic inflammation, apoptosis, and fibrosis rather than assuaging the pathological phenotype of MASH, suggesting that DCs play a protective role in MASH[14]. In another study, the CD103+ cDC1 subset was identified as a protective DC subtype in a murine model, and adoptive transfer of CD103+ cDC1s to Batf3-deficient animals attenuated liver damage and inflammation[15]. On the contrary, single-cell transcriptomics analysis revealed that X-C motif chemokine receptor 1+ cDC1-T cell pairs in liver-draining lymph nodes showed that cDCs influence inflammatory T cell reprogramming and exacerbate MASH development. However, depletion of cDC1 in XCR1DTA mice abolished liver pathology[16]. These findings suggest that distinct DC populations within this heterogeneous population may play discrete roles in MASH development.
In MASLD or MASH livers, infiltrated and liver-resident macrophages, including MDMs, KCs, and other macrophages, are a focal point in this field due to the supposed specific communication and functionality between cells. Moreover, macrophages are a heterogeneous population, with distinct transcriptional profiles and functions. For example, hepatic macrophages play different roles in HSC activation and collagen degradation[17]. Modern technologies, including scRNA-seq and spatial proteomics, have provided deep interrogation of hepatic macrophages in both diseased and healthy livers and have shifted the paradigm of macrophage function, which was previously implicated as a driver in the progression of steatosis toward end-stage disease, to being heterogeneous with a multitude of phenotypes, activation states, and functions. This new principle that resident and recruited cells display immense heterogeneity in MASLD and MASH could lead to viable therapy strategies as these complex populations are unraveled.
Clustering analysis from scRNA-seq data shows expansion of KCs and MDMs in diet-induced MASH liver, both of which exhibit a shift towards a proinflammatory phenotype, contributing to a proinflammatory environment in MASH liver[9]. Transcriptomic profiling of liver macrophages shows that inflammatory markers are significantly upregulated in MDMs compared to KCs in mice fed with a high-fat diet (HFD)[18]. This suggests that the lineage of macrophages in the liver may define their role in the development of MASH. For instance, Tran et al[19] observed that while MDMs main
While subsets of macrophages and their high inflammatory status in the liver may advance the development of MASH, their activities are under tight regulation by a variety of proteins. MER proto-oncogene tyrosine kinase (MerTK) is a Tyro-Axl-MerTK family protein that is highly upregulated in macrophages. MERTK signaling in macrophages has been shown to induce the expression of transforming growth factor beta 1 (TGF-β1) via extracellular signal-regulated kinase (ERK) 1/2 activation and the subsequent activation of HSCs, leading to the progression of fibrosis[22]. Ablation of MERTK signaling by disintegrin and metalloproteinase domain-containing protein 17 decreased MASH progression[23]. Further, the NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome is an intracellular multimeric protein complex critical to caspase-1-dependent interleukin (IL)-1β secretion[24]. NLRP3 is richly expressed in MDMs, KCs, and other immune cells. Studies have shown that hepatic expression of NLRP3 increases in MASLD mice[25,26]. All components of the NLRP3 inflammasome are reportedly upregulated in macrophages isolated from MASH liver. In NLRP3-deficient mice, protection against HFD-induced fatty liver disease was observed[27], and treatment with NLRP3 inhibitors, such as obeticholic acid, improved MASH pathology[26,28]. Other molecules, such as pyruvate kinase M2[29], complement factor C5a receptor 1[30], yes-associated protein[31], and receptor for advanced glycation end products[32], have been shown to promote macrophage inflammatory status and ultimately advance MASH development. Ablation of these signaling molecules showed protective outcomes. In summary, targeting components of pervasive inflammasomes in liver macrophages may represent important therapeutic strategies in the treatment of MASH disease.
Recent studies have characterized MDMs to account for their heterogeneous roles in innate and adaptive immunity in both infiltrating and resident cells. Specifically, classically activated macrophages or M1 macrophages, which result from stimulation by type 1 stimuli, exhibit inflammatory phenotypes; whilst their counterparts, known as alternatively activated macrophages (AAMs) or M2 macrophages, which were induced by type 2 cytokines such as IL-4 and IL-13, are crucial for phagocytosis and anti-inflammatory function in the liver. Studies have demonstrated that AAMs can promote tissue repair through TGF-β and other factors, fibrosis restoration, and general protection against liver injury[33,34]. Interplays and counterbalance between M1 and M2 macrophages through cell-cell interaction or mediators such as IL-10 directly influence the overall liver homeostasis and the pathogenesis of MASLD. In an acute liver injury mouse model, Starkey et al[35] showed that AAMs exert positive effects in reversing the inflammatory environment of the injury via means of resolution and repair. Furthermore, this study observed a 60% reduction in necrotic liver area and an 8.4-fold increase in bromodeoxyuridine levels, indicative of liver regeneration following injections of AAMs[35]. This phenotype of macrophages is multi-faceted in its ability to reduce inflammatory cytokines [e.g., interferon-γ (IFN-γ) and IL-6], reduce necrotic liver tissue, and stimulate hepatocyte and endothelial cell proliferation[33,34]. During MASH, hepatocyte death is the primary trigger that activates the innate immune system, releasing damage-associated molecular patterns and proinflammatory cytokines and accelerating fibrosis, thus worsening disease severity. AAMs are formidable in the potential of reversing MASLD before MASH or end-stage disease; their highly phagocytic phenotype has been observed to effectively clear necrotic material, reduce pro-inflammatory cytokines, and aid in the proliferation of the major cell populations of the liver[34].
Neutrophil infiltration is a typical histological feature of MASH in animals and humans[36]. Chemokines such as C-X-C motif chemokine ligand 1 (CXCL1) play a crucial role in neutrophil infiltration. In patients with MASH, CXCL1 was highly elevated but not in obese individuals or HFD-fed mice. Hwang and colleagues demonstrated that ectopic expre
Neutrophil extracellular trap (NET) accumulation has been observed in MASH conditions, and these NET reservoirs may indeed be promoted via MASH-specific mechanisms[39]. In MASH liver, transcriptomic profiling revealed that NETs impacted gene expression profile in naïve CD4+ T cells, which metabolically reprogrammed these T cells to differentiate to regulatory T cells, all occurring in the presence of Toll-like receptor (TLR) 4[40]. This study robustly supports the ability of neutrophils to shape adaptive immune response during MASH progression. Notably, neutrophil count is crucial for the computation of systemic immune-inflammation index (SII). Specifically, (platelet count × neutrophil count/Lymphocyte count) has been used to compute SII. In a cross-sectional investigation involving 10505 participants (5937 of whom were diagnosed with hepatic steatosis), multivariable logistic regression showed that high SII level was an independent risk factor for hepatic steatohepatitis[41]. Therefore, SII may represent an affordable way to identify hepatic steatosis, although this needs to be validated by additional studies. On the other hand, neutrophil-specific microRNA production enriched in lipotoxic hepatocytes has been shown to hamper MASH progression via low-density lipoprotein receptor (LDLR)-dependent clearance of miR-223-enriched apolipoprotein E (APOE)-expressing extracellular vesicles (EVs), absence of LDLR and APOE-dependent uptake of miR-223-rich EVs amplify MASH progression[42]. This study suggests that neutrophils may play a beneficial role in MASH; however, this advantageous type of functional role is understudied. Overall, evidence from several studies has shown that neutrophil infiltration can contribute to the progres
The role of NK cells in MASH is complex and environment-dependent. In mice fed with a methionine- and choline-deficient (MCD) diet, MASH development was associated with high expression of NK cell activation markers, NK group 2D and CD107a (lysosomal-associated membrane protein 1 or LAMP-1), while NK cell-deficient Nfil3-/- mice reversed diet-induced MASH development[43]. Further, NK cells isolated from MASH livers have been reported to secrete high levels of pro-inflammatory cytokines, including IFN-γ, IL-1β, IL-12, C-C motif chemokine ligand 4 (CCL4), CCL5, and granulocyte-macrophage colony-stimulating factor (GM-CSF). These cytokines could activate the hepatic Janus kinase (JAK)/signal transducers and activators of transcription (STAT)/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) shunt and induce hepatic damage[43]. This study illustrated the damaging effects of NK cells during MASH progression via the secretion of pro-inflammatory cytokines. Contrary to these findings, other reports have shown that NK cells may play a protective role in MCD diet-induced MASH in mice. Specifically, MCD-induced MASH liver exhibited upregulated expression of CXCL10, resulting in the recruitment of C-X-C motif chemokine receptor 3 (CXCR3)+ NK cells. NK cells recruited via the CXCL10-CXCR3 interaction inhibited hepatic inflammation but not steatohepatitis compared to NK cell-deficient Nfil3-/- mice[44]. Thus, tissue-specific characteristics may impact NK functions and their protective or damaging contribution to MASH. In the adipocytes of mice fed with a HFD, NK cell receptor 1 (NKp46)-activating ligands were upregulated on the adipocytes, leading to increased release of IFN-γ in NK cells. IFN-γ release influenced the infiltration and activation of macrophages. This NK cell-macrophage crosstalk is important to maintain an inflammatory status in MASH[45]. Overall, the role of NK cells in MASH development is paradoxical as it has both protective antifibrotic and deleterious proinflammatory effects during MASH progression.
Like NK cells, NKT cells function as innate immune cells, as they can be rapidly activated to secrete a variety of cytokines such as IL-2, IL-10, and IFN-γ to regulate both innate and adaptive immune cells[46]. Moreover, NKT cells share important features of other T cells, including expression of a semi-invariant T cell receptor (TCR) in type I NKT cells (type II or variant NKT cells lack expression of the invariant TCR-alpha chain) that interacts with CD1d-bound glycolipid antigens, and expression of T cell markers such as CD25, CD44, and CD69[47]. NKT cells showed a protective effect in male mice with MASH induced by a choline-deficient HFD, such that male CD1d-/- Balb/c mice displayed severe liver inflammation and fibrosis compared to wild-type Balb/c mice. However, this protective effect was absent in female mice counterparts, perhaps caused by the low number of hepatic type I NKT cells in female mice compared to male mice[48]. Another study showed that Akkermansia muciniphila (AM06) isolated from breast milk can ameliorate the severity of MASH and suppress MASH-associated HCC progression by increasing the infiltration of liver CXCR6-expressing NKT cells and reducing macrophage recruitment[49]. The role of NKT cells in MASH is also contradictory. For example, it has been demonstrated that NKT cells in MCD-induced MASH liver increased the secretion of osteopontin to promote liver fibrosis[50], whereas in another study, type I or invariant NKT (iNKT)-deficient Jα18-/- gave rise to less severe hepatic steatosis and fibrosis following a choline-deficient L-amino acid-defined diet[51]. In addition, the number IL-17A-expressing iNKT cells was increased in peripheral blood mononuclear cells from patients with MASH compared to that in patients with MASLD or healthy controls[51]. These studies suggest that the subtypes of liver NKT cells play different roles in MASLD.
MAIT cells are innate-like T cells in the liver and are known for their dual roles in tissue repair, inhibition of infection, and resolution of fibrosis under inflammatory conditions. The frequency of MAIT cells (TCRVa7.2+ CD161high CD3+ T cells) in circulating blood in patients with MASH decreased compared to that in the group with steatosis or healthy controls[52]. Activation (CD69) and exhaustion (PD-1) markers were highly expressed on MAIT cells in MASLD patients compared to control groups. In addition, these cells highly expressed CXCR6 with a high tendency to migrate into MASLD liver upon injury. Functional study showed that activated MAIT cells could induce macrophage polarization to an M2-like phenotype by producing cytokines such as IL-4[53]. In contrast, mice deficient in MAIT cells under a MCD displayed severe hepatic inflammation and steatosis with an increased M1 macrophage polarization and a decreased M2 macrophage polarization in the livers[53]. Vα19 mice with high numbers of MAIT cells displayed low levels of serum triglyceride and non-esterified fatty acids compared to wild-type C57BL/6 mice after feeding with a HFD, which may be associated with a reduction of lipid accumulation in the liver or hepatic steatosis[54]. A recent study showed that MASLD-associated polyunsaturated fatty acids can selectively suppress the function of MAIT cells but not CD8+ T and NK cells to reduce MAIT cell expression of anti-tumor cytokines, such as tumor necrosis factor (TNF)-α, IFN-γ, and granzyme B[55]. The molecular mechanism of MAIT cells in fatty liver remains elusive, yet their dual roles in MASH pathogenesis underpin their potential for regulation within the tissue and their communication abilities to infiltrated innate immune cells.
ILCs are derived from common lymphoid progenitors and can be classified into different groups based on their expression of transcription factors and cytokines[56]. This section excludes NK cells that belong to a group of ILCs. Increased production of IL-22 in intestinal type 3 ILCs with depletion of vasoactive intestinal peptide receptor 2 significantly reduced hepatic steatosis in mice with a HFD[57]. Gut microbiota transplantation from healthy donors suppressed HFD-induced hepatic steatosis and type 1 ILC (ILC1) cell population reduction in MASLD in male C57BL/6 mice. Similarly, oral administration of microbial metabolite indole-3-carbinol also improved hepatic steatosis and increased ILC1 cell population by activating aryl hydrocarbon receptor in the livers of MASLD mice[58].
Overall, hepatic innate immune cells are critical in liver inflammation, fibrosis, and lipid metabolism (Table 1), through their interactions with hepatocytes and adaptive immune cells and serving as cellular targets for MASLD and MASH therapy. Their plasticity creates a dynamic landscape throughout MASLD and MASH pathogenesis, which is particularly appealing for functional interrogation and/or drug targeting. Some typical markers can be utilized to differentiate these cell types in mouse and human tissues (Figure 1), which can further be applied to investigate their roles in MASLD.
| Cells | Markers | Functions | Ref. |
| cDCs | CD103 | Adoptive transfer of CD103-expressing cDCs protected liver inflammation and damage in Batf3-deficient animals with a high sucrose diet or MCD diet during MASH progression | Heier et al[15] |
| cDCs | XCR1+ | XCR1+ cDC1 cells influence inflammatory T cell reprogramming and exacerbate MASH development | Deczkowska et al[16] |
| Macrophages | MERTK | Ablation of MERTK signaling by disintegrin and metalloproteinase domain-containing protein 17 decreased MASH progression | Cai et al[23] |
| Macrophages | Ly6C+OPN+ | Infiltration of Ly6C+OPN+ lipid-associated macrophages promoted MASH progression | Kui et al[38] |
| Neutrophils | Ly6G+H3Cit+ | Infiltration of Ly6G+H3Cit+ neutrophils promoted MASH progression | Kui et al[38] |
| NK cells | CXCR3+ | CXCR3+ NK cells decreased liver inflammation in MCD-induced MASH in mice | Fan et al[44] |
| NKT cells | IL-17A+ | IL-17A-expressing invariant NKT cells in peripheral blood mononuclear cells were associated with MASH progression in patients | Maricic et al[51] |
| MAIT cells | TCRVa7.2+ CD161high CD3+ | The frequency of MAIT cells in circulating blood in patients with MASH decreased compared to that in the group with steatosis or healthy controls | Waller et al[52] |
| ILCs | CD45+ TCRβ- B220-; NK1.1+ CD49a+ CD49b- | Increasing the frequency of ILC1 frequency by fecal microbiota transplantation and indole-3-carbinol inhibited MASLD | Hou et al[58] |
Innate immunity plays a pivotal role in the progression of MASLD by regulating liver inflammation and fibrosis[59-61]. Treatments, such as bariatric surgery and anti-inflammatory and antifibrotic medicines, can modulate the function and profiles of innate immune cells to suppress MASLD progression[62,63]. In this section, we review the many molecular regulators in innate immune cell activation and dysfunction in MASLD and MASH pathogenesis, some of which may serve as therapeutic targets for the treatment of liver disease.
Adenosine 5’-monophosphate-activated protein kinase (AMPK) serves as a therapeutic target for MASLD and MASH. The AMPK signaling pathway is implicated in lipid and glucose metabolism by interacting with downstream and upstream genes, such as sterol regulatory element-binding protein 1 (SREBP1)[64] and carnitine palmitoyl transferase 1 A[65]. Cre-lox system knock-out AMPK catalytic subunit alpha 1 or 2 in bone marrow-derived and tissue-resident myeloid cells can accelerate HFD-induced liver fibrosis in both female and male mice[66]. In addition, AMPK activation upregulates peroxisome proliferator-activated receptor gamma (PPARγ) coactivator-1α expression and decreases SREBP1 expression to suppress lipid accumulation in hepatocytes[67].
Glycolysis and mitochondrial respiration were dramatically increased in monocytes of patients with MASH compared to healthy controls, which was associated with proton leak and increased expression of proinflammatory cytokines (IL-1β and TNF-α) in serum. A reduction of AMPK phosphorylation was associated with the metabolic adaptation of monocytes in patients with MASH, which favored the activation of peroxisome-proliferator-activated receptor-gamma coactivator-1α, a transcription regulator of mitochondrial biogenesis and oxidative metabolism[68].
Inhibiting hepatic farnesoid X receptor (FXR) expression can ameliorate HFD-induced lipid accumulation, oxidative stress, inflammation, and insulin resistance in mice[69,70]. In vitro and in vivo mechanistic investigations demonstrated that HFD treatment can inhibit hepatic expression of zinc finger and BTB domain containing 18a, which regulates hepatic lipid accumulation and inhibits macrophage activation by upregulating FXR-mediated clathrin heavy chain expression to suppress NLRP3 inflammasome activation[71]. Therefore, a semi-synthetic bile acid analog that binds to FXR has therapeutic potential in the treatment of MASH.
Additionally, obeticholic acid can directly suppress the activation of macrophage NLRP3 inflammasome to result in a reduction of hepatic lipid accumulation[28]. Treatment with a dual agonist (INT-767) of FXR and Takeda G protein-coupled receptor 5 increased the proportion of anti-inflammatory liver monocytes and the expression of M2-like macrophage marker genes such as CD206, Fizz1 (resistin-like molecule alpha 1), and IL-10[72].
Granulocyte colony-stimulating factor (GCSF)-deficient mice had decreased lipid accumulation in hepatic and adipose tissues and insulin resistance compared to wild-type mice[73]. Mechanistically, GCSF can regulate hepatic infiltration of neutrophils and macrophages during MASLD progression, and disruption of GCSF/GCSF receptor interaction can suppress liver lipid accumulation by increasing phosphorylation of JAK1/2 and STAT3 and their activation through inhibition of suppressor of cytokine signaling 3 expression[73]. Activated NK cells in mice with diet-induced MASH secreted pro-inflammatory cytokines and chemokines such as IL-β, IL-12, GM-CSF, IFN-γ, CCL4, and CCL5, which activated hepatic JAK-STAT1/3 and NF-κB signaling pathways to increase the production of ROS and induce apoptosis, thereby fostering liver damage[43].
Mitogen-activated protein kinases, including ERK, c-Jun N-terminal kinase (JNK), and p38 mitogen-activated protein kinase, play an essential role in modulating liver inflammation via regulation of proinflammatory cytokine products, immune cell recruitment, and the expression of key enzymes in inflammation (e.g., iNOS or inducible nitric oxide synthase)[74,75]. Blocking JNK/NF-κB signaling pathway in macrophages can inhibit liver inflammation and suppress macrophage-hepatocyte interaction to reduce liver injury[76].
Oxysterol binding protein like 8 is encoded by gene Osbpl8, which is highly expressed in EVs derived from bone-marrow-derived macrophages. Osbpl8-enriched EVs display anti-inflammatory and reduce lipotoxicity in MASH livers by regulating lipid metabolism and ameliorating endoplasmic reticulum (ER) stress through suppression of inositol-requiring kinase 1α/X-box binding protein-1 signaling pathway[77]. Hepatic expression of Osbpl3 was increased in patients with advanced MASLD compared to those with early MASLD, which was also observed in mice with MASLD. Further studies illustrated that the expression of Osbpl3 can be regulated by PPARγ[78].
Overexpression of PPARγ can dampen palmitic acid-induced inflammatory cytokine gene expression in macrophages, whereas knockout of PPARg in macrophages worsens liver injury and accelerates MASLD progression in mice[79]. The beneficial function of PPARγ in MASLD is associated with the activation of Keap1/nuclear factor, erythroid 2-like transcription factor 2 signaling pathway[79]. Activation of PPAR signaling pathway was associated with macrophage-mediated inflammation in human MASH and murine MASH models[80]. Another study showed that activation of the PPARγ signaling pathway significantly inhibited M1 macrophage polarization and reduced lipid accumulation in vitro and in vivo[81]. Macrophage-specific knockout of PPARg can exacerbate liver inflammation and damage in mice with MASLD[79]. In addition, PPARγ can upregulate the expression of matrix metallopeptidase 10 to regulate IL-4-stimulated M2 macrophage polarization[82].
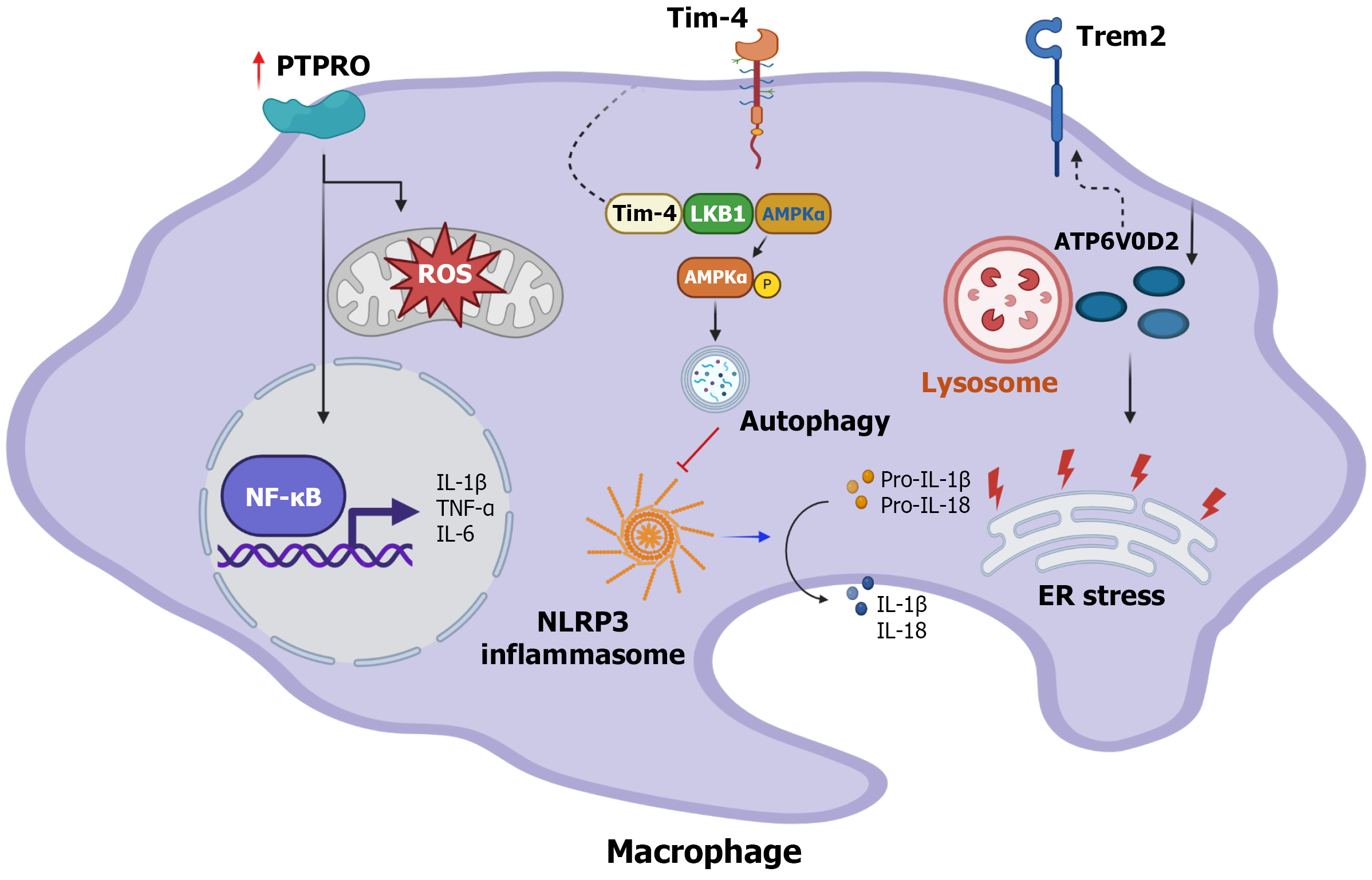
During MASH progression in both mice and humans, protein tyrosine phosphatase receptor type o truncated isoform (PTPROt) expression has been observed to be increased in liver macrophages[83]. Overexpression of PTPROt in macrophages has been demonstrated to be associated with increased inflammation and mitochondrial damage via the activation of the NF-κB signaling pathway, along with increased expression of proinflammatory cytokines (IL-1β, TNF-α, and IL-6) and the production of ROS. In contrast, PTPROt deficiency in primary liver macrophages decreased the expression of NLRP1, NLRP3, NLRC4, and absent in melanoma 2, reducing ROS production[83].
T-cell membrane protein 4 (Tim-4), encoded by gene T-cell immunoglobulin and mucin domain containing 4, is highly expressed in liver macrophages. In macrophages of MASLD liver, Tim-4 interacts with liver kinase B1 and AMPKα to suppress NLRP3 inflammasome activation through phosphorylated AMPKα-regulated autophagy. Therefore, Tim-4 activation inhibits macrophage expression of IL-1β and IL-18 to ameliorate liver inflammation[84]. A recent study also demonstrates that impairing Tim-4 expression in KCs, either through neutralization antibody or genetic depletion, can decrease macrophage efferocytosis of apoptotic hepatocytes and increase the activation of HSCs, thereby promoting the progression of liver fibrosis[85].
A brief two-week treatment with HFD in mice can significantly increase the frequency and cell number of ILC1 cells in MASLD livers[86], which is associated with an increase in the production of proinflammatory cytokines and chemokines, such as TNF-α and CXCR3. RNA-sequencing results demonstrate that TLR9 plays an important role in ILC1 differentiation by activating T-bet (T-box transcription factor) expression[86]. Another study shows that long-term treatment of TLR4 activator lipopolysaccharides (LPS) and HFD induces a more severe form of MASLD by activating NF-κB kinase subunit epsilon/NF-κB signaling, compared to single treatment of a HFD or LPS alone[87].
Liver resident macrophages (KCs) are decreased during MASH progression, whereas the population of MDMs is increased. The expression of the scavenger receptor Trem2 in phagocytes is important for their phagocytic function as well as the clearance of apoptotic cells. Trem2hi phagocytes have been implicated in hepatic injury[88,89]. In MASH liver, over 93% of KCs have been shown to harbor high Trem2 expression. This cell population, also termed MASH-associated macrophages, is responsible for the clearance of apoptotic cells and ECM during liver injury, which alludes to the functional reprogramming of macrophages during MASH pathogenesis[9]. Similarly, Chan and colleagues deemed a similar phenotype in macrophages, calling them LAMs that express Trem2, demonstrating that these LAMs aggregate into hepatic crownlike structures, believed to form around dying hepatocytes and localize them to areas rich in HSCs. These hepatic crownlike structures were observed in human MASH and seen to correlate with fibrosis severity. Based on this information, this phenotype of macrophages is seemingly involved in profibrotic pathways. Yet, the same researchers observed in C-C motif chemokine receptor 2 (CCR2) knockout mice with a HFD, meaning those with reduced LAMs and hepatic crownlike structures, liver fibrosis was increased[17]. This contradiction supports the notion that MDMs and even KCs, altogether, have dynamic phenotypes, altering their crosstalk, fibrotic effects, and overall impact on MASH liver tissue remodeling.
ATP6V0D2, a lysosomal gene in macrophages, regulates efferocytosis in Trem2-expressing macrophages, which supports maintenance of lipid metabolism and evades MASH progression. ATP6V0D2 deficiency in macrophages induces ER stress and promotes lipid accumulation and apoptosis of hepatocytes[90].
In summary, the several signaling pathways described above are uniquely involved with innate immune cell functionality, such as macrophages (Figure 2), thus, consideration of these cells as targets for therapeutic strategies to treat MASLD or MASH is imperative.
Biomarkers expressed in innate immune cells can be applied for the diagnosis and treatment of MASLD. For example, the frequency of sialic acid-binding immunoglobulin-like lectin 7+ CD56dim NK cells was decreased in patients with MASLD[91]. These NK cells highly expressed CD57 and PD-1, which could be targeted for MASLD therapy. Inflammatory markers or obesity-associated biomarkers, such as leptin, adiponectin, leukocyte cell-derived chemotaxin-2, chemerin, and circulating full-length and caspase-cleaved cytokeratin 18, can serve as non-invasive biomarkers in MASLD screening and treatment evaluation[92]. The expression level of IL-1β in circulating mononuclear cells was positively associated with body mass index, hemoglobin A1C, visceral adipose tissue, and MASLD extent or degree in obese patients with prediabetes or T2D[93]. Furthermore, weight loss achieved by treatment of glucagon-like peptide receptor agonist liraglutide or lifestyle management reduced the expression level of IL-1β and MASLD degree[93].
THR-β agonist HSK31679 can ameliorate diet-induced MASH by modulating gut microbiota metabolism and decreasing peripheral DCs and macrophages[94]. Treatment of resmetirom at a dose of 80 mg or 100 mg, once daily for 52 weeks, can improve liver fibrosis and reduce low-density lipoprotein cholesterol (LDL-C) level from the baseline[95]. Compared to placebo, THR-β agonist TERN-501 significantly reduced liver fat content in a dose-dependent manner, which was measured using magnetic resonance imaging proton density fat fraction[96].
CCL24 (eotaxin-2) plays multiple roles in diseases, including the recruitment of immune cells (e.g., eosinophils and neutrophils), macrophage polarization, and tumorigenesis[97]. CCL24 contributes to liver inflammation and fibrosis in MASLD and MASH[98]. Patients with MASLD who received intravenous or subcutaneous injection of CM-101, an anti-human CCL24 monoclonal antibody, every 3 weeks for 12 weeks, had decreased serum levels of inflammatory and fibrotic biomarkers compared with those in the placebo group[99].
Tang and Borlak[100] reported that the expression levels of CD163 and CD206 were negatively correlated with the expression of fibroblast growth factor 21 (FGF21) in patients with MASLD, indicating that FGF21 inhibits macrophage M2 polarization[100]. A phase 2 clinical trial has also demonstrated the beneficial effects of an engineered long-acting FGF21 analogue efimosfermin alfa (BOS-580) in reducing hepatic lipid accumulation[101].
A meta-analysis study shows that FXR agonists can mitigate liver injury by reducing alanine aminotransferase and gamma-glutamyltransferase (GGT) levels, as well as liver fat content in total, and they also can ameliorate liver fibrosis in patients with MASLD compared to placebo[102]. Another meta-analysis also shows that FXR agonists can reduce hepatic steatosis, which is evaluated by non-invasive imaging technology, magnetic resonance imaging-derived proton density fat fraction[103]. Treatment with FXR agonist vonafexor reduced liver fat content and liver enzymes, resulting in a reduction in body weight[104].
As previously described, PPARs play important roles in liver metabolism, inflammation, and fibrosis[105-107]. Treatment with lanifibranor, a PPAR agonist, suppressed the infiltration of MDMs in MASLD livers, which also inhibited palmitic acid-induced activation of bone-marrow-derived macrophages from mice and monocytes from patients with MASLD[108]. Lanifibranor significantly ameliorated hepatic insulin resistance and reduced levels of fasting glucose, glycosylated hemoglobin, and HDL-C[109].
Pemafibrate, a PPARα agonist, can dramatically reduce levels of LDL-C, non-HDL-C, and apolipoprotein B from the baseline in patients with MASLD[110]. Studies have shown that pemafibrate can promote M2 polarization of both murine and human macrophages by increasing the expression of CD163[111]. In addition, it can also inhibit IFN-γ-induced human THP-1 cell inflammation by suppressing the activation of NLRP3/caspase-1 signaling pathway[111].
A randomized, placebo-controlled, phase 2 clinical trial (ClinicalTrials.gov, trial number: No. NCT04667377) showed that treatment with survodutide, a dual agonist of glucagon receptor and glucagon-like peptide-1 receptor, decreased body weight in a dose-dependent manner compared to placebo[112]. Another phage 2 trial showed that survodutide can significantly reduce liver fat content and improve liver fibrosis in patients with MASH compared to placebo treatment[113].
Treatment with icosabutate, a free fatty acid receptor 1 and 4 agonist, at a dose of 600 mg for 52 weeks, reduced liver injury biomarkers (aspartate aminotransferase, GGT, and alkaline phosphatase) compared to placebo, which attained a stage improvement of liver fibrosis by artificial intelligence-assisted digital pathology analysis[114]. Overall, innate immune cells are therapeutic targets for MASLD treatment (Table 2).
| Treatments | Drugs | Trials | Phases | Functions | Ref. |
| THR-β agonist | HSK31679 | NCT05531097 | 1 | Ameliorate diet-induced MASH by modulating gut microbiota and peripheral dendritic cells and macrophages | Zhang et al[94] |
| THR-β agonist | MGL-3196 (resmetirom) | NCT03900429 | 3 | Treatment of resmetirom at a dose of 80 mg or 100 mg, once daily for 52 weeks, can improve liver fibrosis and reduce low-density lipoprotein cholesterol level from the baseline | Harrison et al[95] |
| THR-β agonist | TERN-501 | NCT05415722 | 2 | Reduce liver fat content compared to placebo in a dose-dependent manner | Noureddin et al[96] |
| Anti-human CCL24 monoclonal antibody | CM-101 | NCT06044467 | 1 | It can reduce serum levels of inflammatory and fibrotic biomarkers in patients with MASLD | Mor et al[99] |
| NCT06037577 | 1 | ||||
| NCT06025851 | 1 | ||||
| A long-acting fibroblast growth factor 21 analogue | Efimosfermin alfa | NCT04880031 | 2 | Reduce hepatic glycogen storage, attenuate lipid accumulation, ameliorate fibrosis, and suppress immune response | Tang and Borlak[100]; Loomba et al[101] |
| Farnesoid X receptor agonist | Vonafexor (EYP001a) | NCT03812029 | 2 | Reduce body weight, liver fat, and biomarker enzymes in patients with suspected fibrotic MASH | Ratziu et al[104] |
| PPAR agonist | Lanifibranor | NCT03459079 | 2 | It significantly improved hepatic insulin sensitivity and reduced intrahepatic triglyceride content | Barb et al[109] |
| PPARα agonist | Pemafibrate | NCT03350165 | 2 | Decrease levels of low-density lipoprotein cholesterol, non-HDL-C, and apolipoprotein B from the baselines in patients with MASLD | le Roux et al[112] |
| NCT05923281 | 3 | ||||
| A dual agonist of glucagon receptor and glucagon-like peptide-1 receptor | Survodutide | NCT04771273 | 2 | Reduce liver fat content and improve liver fibrosis in patients with MASH | Sanyal et al[113] |
| Free fatty acid receptor 1 and 4 agonist | Icosabutate (NST-4016) | NCT04052516 | 2 | Reduce liver injury biomarkers and decrease liver inflammation and fibrosis in patients with MASH and mild to severe fibrosis | Harrison et al[114] |
In addition, clinical trials have also demonstrated the beneficial effects of treatments, such as low-dose aspirin (No. NCT04031729)[115], vitamin D[116] or E[117] supplementation, wheat amylase trypsin inhibitors or gluten-free diet (No. NCT04066400)[118], in reducing liver fat accumulation, serum levels of fibrogenic factors, and homeostatic model assessment of insulin resistance in patients with MASLD and MASH.
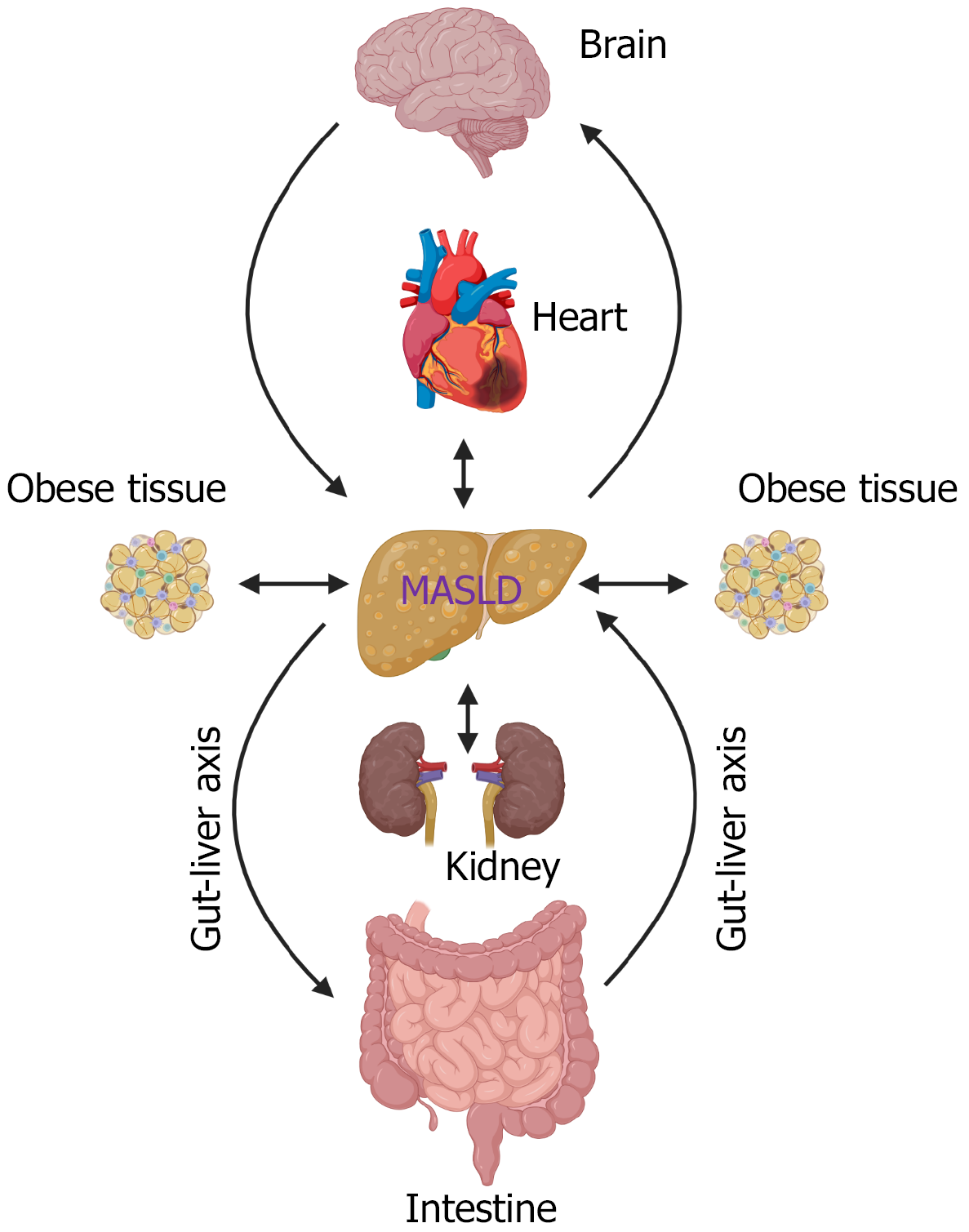
MASLD is a comorbidity associated with many other metabolic disorders and infections, such as T2D and cardiovascular disease[119]. It impacts the progression and treatment of these diseases (Figure 3), such as the gut-liver axis[120]. Accumulating evidence indicates that innate immune cells play essential roles in the development and progression of MASLD. Therefore, it is critically important to investigate it at the cellular and molecular levels to pinpoint the mechanistic shifts in pathogenic hallmarks, such as fibrotic deposits or lipid handling. The regulation of hepatic lipid accumulation and inflammation can alter the proportions of these immune cells, thereby ameliorating liver injury and MASLD progression. Several strategies in the clinic show beneficial effects in preventing MASLD or MASH progression. Physical exercise is beneficial for maintaining body weight and preventing obesity before the onset of MASLD[121]. It is also considered a non-pharmacological treatment option for MASLD or MASH. Interactions of diet-gut microbiota and their associated products, such as metabolites, bile acids, and microbial components, collectively modulate the profile and activation of intrahepatic immune cells. This occurs through the regulation of lipid content, insulin signaling pathway, and various other pathways discussed in this paper, ultimately influencing MASLD progression. Numerous studies have characterized changes in the gut microbiome of patients with MASLD or MASH[112]. For example, the relative abundance of Phocaeicola dorei (P. dorei) has been linked to changes in MASLD severity[122]. Supplementation with P. dorei was shown to reduce Western diet-induced MASLD severity in mice by reducing the expression of liver inflammatory cytokines and chemokines and increasing gene expression involved in lipid β-oxidation. Furthermore, the cell-free supernatant of P. dorei was found to inhibit LPS-induced macrophage inflammation by modulating p38 phosphorylation[123]. Current studies also validate that dietary intervention[123], calorie restriction[124], or weight loss programs[125] can improve liver fat composition, insulin sensitivity, and reduce the risk of hypertension, cardiovascular disease, and T2D[126,127]. However, it remains to be determined whether the combination therapies, including physical activity, diet modifications, and pharmacological treatments, can reverse liver injury in patients with MASH and MASH-associated advanced liver diseases. Several therapies, such as FGF21 analogues, FXR agonists, CCR2 and CCR5 dual antagonists, calorie restriction, bariatric surgery, and exercise, show promise for the prevention and treatment of MASLD in clinical trials. Models like organoids and human-like animal models are important for evaluating the effectiveness of pharmaceutical treatments. Serum biomarkers linked to hepatic immune cell activation are non-invasive methods for MASLD diagnosis and prognosis, as well as for evaluating the treatment efficacy in MASLD or MASH. Emerging biomarkers, such as microRNAs, require extensive evaluation. Additionally, many of the above-mentioned molecular targets identified in pre-clinical models await clinical evaluation.
| 1. | Huttasch M, Roden M, Kahl S. Obesity and MASLD: Is weight loss the (only) key to treat metabolic liver disease? Metabolism. 2024;157:155937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 102] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 2. | Bendixen SM, Jakobsgaard PR, Hansen D, Hejn KH, Terkelsen MK, Bjerre FA, Thulesen AP, Eriksen NG, Hallenborg P, Geng Y, Dam TV, Larsen FT, Wernberg CW, Vijayathurai J, Scott EAH, Marcher AB, Detlefsen S, Grøntved L, Dimke H, Berdeaux R, de Aguiar Vallim TQ, Olinga P, Lauridsen MM, Krag A, Blagoev B, Ravnskjaer K. Single cell-resolved study of advanced murine MASH reveals a homeostatic pericyte signaling module. J Hepatol. 2024;80:467-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 3. | Wang S, Friedman SL. Found in translation-Fibrosis in metabolic dysfunction-associated steatohepatitis (MASH). Sci Transl Med. 2023;15:eadi0759. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 114] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 4. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78:1966-1986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2198] [Cited by in RCA: 2047] [Article Influence: 682.3] [Reference Citation Analysis (8)] |
| 5. | Keam SJ. Resmetirom: First Approval. Drugs. 2024;84:729-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 244] [Article Influence: 122.0] [Reference Citation Analysis (3)] |
| 6. | Shakeel L, Shaukat A, Akilimali A. Resmetirom: A Breakthrough in the Treatment of Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Health Sci Rep. 2025;8:e70920. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 7. | Pfister D, Núñez NG, Pinyol R, Govaere O, Pinter M, Szydlowska M, Gupta R, Qiu M, Deczkowska A, Weiner A, Müller F, Sinha A, Friebel E, Engleitner T, Lenggenhager D, Moncsek A, Heide D, Stirm K, Kosla J, Kotsiliti E, Leone V, Dudek M, Yousuf S, Inverso D, Singh I, Teijeiro A, Castet F, Montironi C, Haber PK, Tiniakos D, Bedossa P, Cockell S, Younes R, Vacca M, Marra F, Schattenberg JM, Allison M, Bugianesi E, Ratziu V, Pressiani T, D'Alessio A, Personeni N, Rimassa L, Daly AK, Scheiner B, Pomej K, Kirstein MM, Vogel A, Peck-Radosavljevic M, Hucke F, Finkelmeier F, Waidmann O, Trojan J, Schulze K, Wege H, Koch S, Weinmann A, Bueter M, Rössler F, Siebenhüner A, De Dosso S, Mallm JP, Umansky V, Jugold M, Luedde T, Schietinger A, Schirmacher P, Emu B, Augustin HG, Billeter A, Müller-Stich B, Kikuchi H, Duda DG, Kütting F, Waldschmidt DT, Ebert MP, Rahbari N, Mei HE, Schulz AR, Ringelhan M, Malek N, Spahn S, Bitzer M, Ruiz de Galarreta M, Lujambio A, Dufour JF, Marron TU, Kaseb A, Kudo M, Huang YH, Djouder N, Wolter K, Zender L, Marche PN, Decaens T, Pinato DJ, Rad R, Mertens JC, Weber A, Unger K, Meissner F, Roth S, Jilkova ZM, Claassen M, Anstee QM, Amit I, Knolle P, Becher B, Llovet JM, Heikenwalder M. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature. 2021;592:450-456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1051] [Cited by in RCA: 1022] [Article Influence: 204.4] [Reference Citation Analysis (5)] |
| 8. | Magee N, Zou A, Zhang Y. Pathogenesis of Nonalcoholic Steatohepatitis: Interactions between Liver Parenchymal and Nonparenchymal Cells. Biomed Res Int. 2016;2016:5170402. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 88] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 9. | Xiong X, Kuang H, Ansari S, Liu T, Gong J, Wang S, Zhao XY, Ji Y, Li C, Guo L, Zhou L, Chen Z, Leon-Mimila P, Chung MT, Kurabayashi K, Opp J, Campos-Pérez F, Villamil-Ramírez H, Canizales-Quinteros S, Lyons R, Lumeng CN, Zhou B, Qi L, Huertas-Vazquez A, Lusis AJ, Xu XZS, Li S, Yu Y, Li JZ, Lin JD. Landscape of Intercellular Crosstalk in Healthy and NASH Liver Revealed by Single-Cell Secretome Gene Analysis. Mol Cell. 2019;75:644-660.e5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 705] [Cited by in RCA: 638] [Article Influence: 91.1] [Reference Citation Analysis (5)] |
| 10. | Zhou L, Qiu X, Meng Z, Liu T, Chen Z, Zhang P, Kuang H, Pan T, Lu Y, Qi L, Olson DP, Xu XZS, Chen YE, Li S, Lin JD. Hepatic danger signaling triggers TREM2(+) macrophage induction and drives steatohepatitis via MS4A7-dependent inflammasome activation. Sci Transl Med. 2024;16:eadk1866. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 29.0] [Reference Citation Analysis (2)] |
| 11. | Ibrahim SH, Hirsova P, Gores GJ. Non-alcoholic steatohepatitis pathogenesis: sublethal hepatocyte injury as a driver of liver inflammation. Gut. 2018;67:963-972. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 239] [Cited by in RCA: 229] [Article Influence: 28.6] [Reference Citation Analysis (1)] |
| 12. | Haas JT, Vonghia L, Mogilenko DA, Verrijken A, Molendi-Coste O, Fleury S, Deprince A, Nikitin A, Woitrain E, Ducrocq-Geoffroy L, Pic S, Derudas B, Dehondt H, Gheeraert C, Van Gaal L, Driessen A, Lefebvre P, Staels B, Francque S, Dombrowicz D. Transcriptional Network Analysis Implicates Altered Hepatic Immune Function in NASH development and resolution. Nat Metab. 2019;1:604-614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 130] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 13. | Kelly A, Fahey R, Fletcher JM, Keogh C, Carroll AG, Siddachari R, Geoghegan J, Hegarty JE, Ryan EJ, O'Farrelly C. CD141⁺ myeloid dendritic cells are enriched in healthy human liver. J Hepatol. 2014;60:135-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 79] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 14. | Henning JR, Graffeo CS, Rehman A, Fallon NC, Zambirinis CP, Ochi A, Barilla R, Jamal M, Deutsch M, Greco S, Ego-Osuala M, Bin-Saeed U, Rao RS, Badar S, Quesada JP, Acehan D, Miller G. Dendritic cells limit fibroinflammatory injury in nonalcoholic steatohepatitis in mice. Hepatology. 2013;58:589-602. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 158] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 15. | Heier EC, Meier A, Julich-Haertel H, Djudjaj S, Rau M, Tschernig T, Geier A, Boor P, Lammert F, Lukacs-Kornek V. Murine CD103(+) dendritic cells protect against steatosis progression towards steatohepatitis. J Hepatol. 2017;66:1241-1250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 16. | Deczkowska A, David E, Ramadori P, Pfister D, Safran M, Li B, Giladi A, Jaitin DA, Barboy O, Cohen M, Yofe I, Gur C, Shlomi-Loubaton S, Henri S, Suhail Y, Qiu M, Kam S, Hermon H, Lahat E, Ben Yakov G, Cohen-Ezra O, Davidov Y, Likhter M, Goitein D, Roth S, Weber A, Malissen B, Weiner A, Ben-Ari Z, Heikenwälder M, Elinav E, Amit I. XCR1(+) type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis. Nat Med. 2021;27:1043-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 152] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 17. | Chan MM, He L, Finck BN, Schilling JD, Daemen S. Cutting Edge: Hepatic Stellate Cells Drive the Phenotype of Monocyte-derived Macrophages to Regulate Liver Fibrosis in Metabolic Dysfunction-associated Steatohepatitis. J Immunol. 2024;213:251-256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 18. | Morinaga H, Mayoral R, Heinrichsdorff J, Osborn O, Franck N, Hah N, Walenta E, Bandyopadhyay G, Pessentheiner AR, Chi TJ, Chung H, Bogner-Strauss JG, Evans RM, Olefsky JM, Oh DY. Characterization of distinct subpopulations of hepatic macrophages in HFD/obese mice. Diabetes. 2015;64:1120-1130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 143] [Article Influence: 13.0] [Reference Citation Analysis (5)] |
| 19. | Tran S, Baba I, Poupel L, Dussaud S, Moreau M, Gélineau A, Marcelin G, Magréau-Davy E, Ouhachi M, Lesnik P, Boissonnas A, Le Goff W, Clausen BE, Yvan-Charvet L, Sennlaub F, Huby T, Gautier EL. Impaired Kupffer Cell Self-Renewal Alters the Liver Response to Lipid Overload during Non-alcoholic Steatohepatitis. Immunity. 2020;53:627-640.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 310] [Article Influence: 51.7] [Reference Citation Analysis (4)] |
| 20. | Huang W, Metlakunta A, Dedousis N, Zhang P, Sipula I, Dube JJ, Scott DK, O'Doherty RM. Depletion of liver Kupffer cells prevents the development of diet-induced hepatic steatosis and insulin resistance. Diabetes. 2010;59:347-357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 446] [Cited by in RCA: 431] [Article Influence: 26.9] [Reference Citation Analysis (1)] |
| 21. | Neyrinck AM, Cani PD, Dewulf EM, De Backer F, Bindels LB, Delzenne NM. Critical role of Kupffer cells in the management of diet-induced diabetes and obesity. Biochem Biophys Res Commun. 2009;385:351-356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 87] [Article Influence: 5.1] [Reference Citation Analysis (3)] |
| 22. | Cai B, Thorp EB, Doran AC, Subramanian M, Sansbury BE, Lin CS, Spite M, Fredman G, Tabas I. MerTK cleavage limits proresolving mediator biosynthesis and exacerbates tissue inflammation. Proc Natl Acad Sci U S A. 2016;113:6526-6531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 219] [Cited by in RCA: 209] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 23. | Thorp E, Vaisar T, Subramanian M, Mautner L, Blobel C, Tabas I. Shedding of the Mer tyrosine kinase receptor is mediated by ADAM17 protein through a pathway involving reactive oxygen species, protein kinase Cδ, and p38 mitogen-activated protein kinase (MAPK). J Biol Chem. 2011;286:33335-33344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 264] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 24. | Dixon LJ, Flask CA, Papouchado BG, Feldstein AE, Nagy LE. Caspase-1 as a central regulator of high fat diet-induced non-alcoholic steatohepatitis. PLoS One. 2013;8:e56100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 148] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 25. | Schuster S, Cabrera D, Arrese M, Feldstein AE. Triggering and resolution of inflammation in NASH. Nat Rev Gastroenterol Hepatol. 2018;15:349-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 818] [Cited by in RCA: 752] [Article Influence: 94.0] [Reference Citation Analysis (5)] |
| 26. | Mridha AR, Wree A, Robertson AAB, Yeh MM, Johnson CD, Van Rooyen DM, Haczeyni F, Teoh NC, Savard C, Ioannou GN, Masters SL, Schroder K, Cooper MA, Feldstein AE, Farrell GC. NLRP3 inflammasome blockade reduces liver inflammation and fibrosis in experimental NASH in mice. J Hepatol. 2017;66:1037-1046. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 966] [Cited by in RCA: 932] [Article Influence: 103.6] [Reference Citation Analysis (2)] |
| 27. | Wree A, Eguchi A, McGeough MD, Pena CA, Johnson CD, Canbay A, Hoffman HM, Feldstein AE. NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology. 2014;59:898-910. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 820] [Cited by in RCA: 796] [Article Influence: 66.3] [Reference Citation Analysis (5)] |
| 28. | Huang S, Wu Y, Zhao Z, Wu B, Sun K, Wang H, Qin L, Bai F, Leng Y, Tang W. A new mechanism of obeticholic acid on NASH treatment by inhibiting NLRP3 inflammasome activation in macrophage. Metabolism. 2021;120:154797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 29. | Xu F, Guo M, Huang W, Feng L, Zhu J, Luo K, Gao J, Zheng B, Kong LD, Pang T, Wu X, Xu Q. Annexin A5 regulates hepatic macrophage polarization via directly targeting PKM2 and ameliorates NASH. Redox Biol. 2020;36:101634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 145] [Article Influence: 24.2] [Reference Citation Analysis (1)] |
| 30. | Jiang K, Lu S, Li D, Liu M, Jin H, Lei B, Wang S, Long K, He S, Zhong F. Blockade of C5aR1 alleviates liver inflammation and fibrosis in a mouse model of NASH by regulating TLR4 signaling and macrophage polarization. J Gastroenterol. 2023;58:894-907. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (2)] |
| 31. | Qing J, Ren Y, Zhang Y, Yan M, Zhang H, Wu D, Ma Y, Chen Y, Huang X, Wu Q, Mazhar M, Wang L, Liu J, Ding BS, Cao Z. Dopamine receptor D2 antagonism normalizes profibrotic macrophage-endothelial crosstalk in non-alcoholic steatohepatitis. J Hepatol. 2022;76:394-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 89] [Article Influence: 22.3] [Reference Citation Analysis (1)] |
| 32. | Parthasarathy G, Mauer AS, Golla N, Daniel PV, Kim LH, Sidhu GS, Marek GW 3rd, Loeuillard E, Krishnan A, Lee HSK, Pavelko KD, Charlton M, Hirsova P, Ilyas SI, Malhi H. Macrophage RAGE activation is proinflammatory in NASH. JCI Insight. 2024;9:e169138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 33. | Humphries C, Addison ML, Dear JW, Forbes SJ. The emerging role of alternatively activated macrophages to treat acute liver injury. Arch Toxicol. 2025;99:103-114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 34. | Candela ME, Addison M, Aird R, Man TY, Cartwright JA, Ashmore-Harris C, Kilpatrick AM, Starkey Lewis PJ, Drape A, Barnett M, Mitchell D, McLean C, McGowan N, Turner M, Dear JW, Forbes SJ. Cryopreserved human alternatively activated macrophages promote resolution of acetaminophen-induced liver injury in mouse. NPJ Regen Med. 2025;10:5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 35. | Starkey Lewis P, Campana L, Aleksieva N, Cartwright JA, Mackinnon A, O'Duibhir E, Kendall T, Vermeren M, Thomson A, Gadd V, Dwyer B, Aird R, Man TY, Rossi AG, Forrester L, Park BK, Forbes SJ. Alternatively activated macrophages promote resolution of necrosis following acute liver injury. J Hepatol. 2020;73:349-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 156] [Article Influence: 26.0] [Reference Citation Analysis (1)] |
| 36. | Zang S, Wang L, Ma X, Zhu G, Zhuang Z, Xun Y, Zhao F, Yang W, Liu J, Luo Y, Liu Y, Ye D, Shi J. Neutrophils Play a Crucial Role in the Early Stage of Nonalcoholic Steatohepatitis via Neutrophil Elastase in Mice. Cell Biochem Biophys. 2015;73:479-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 37. | Hwang S, He Y, Xiang X, Seo W, Kim SJ, Ma J, Ren T, Park SH, Zhou Z, Feng D, Kunos G, Gao B. Interleukin-22 Ameliorates Neutrophil-Driven Nonalcoholic Steatohepatitis Through Multiple Targets. Hepatology. 2020;72:412-429. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 157] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 38. | Kui L, Kim AD, Onyuru J, Hoffman HM, Feldstein AE. BRP39 Regulates Neutrophil Recruitment in NLRP3 Inflammasome-Induced Liver Inflammation. Cell Mol Gastroenterol Hepatol. 2024;17:481-497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 39. | Lv T, Xiong X, Yan W, Liu M, Xu H, He Q. Mitochondrial general control of amino acid synthesis 5 like 1 promotes nonalcoholic steatohepatitis development through ferroptosis-induced formation of neutrophil extracellular traps. Clin Transl Med. 2023;13:e1325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 40. | Wang H, Zhang H, Wang Y, Brown ZJ, Xia Y, Huang Z, Shen C, Hu Z, Beane J, Ansa-Addo EA, Huang H, Tian D, Tsung A. Regulatory T-cell and neutrophil extracellular trap interaction contributes to carcinogenesis in non-alcoholic steatohepatitis. J Hepatol. 2021;75:1271-1283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 316] [Article Influence: 63.2] [Reference Citation Analysis (1)] |
| 41. | Song Y, Guo W, Li Z, Guo D, Li Z, Li Y. Systemic immune-inflammation index is associated with hepatic steatosis: Evidence from NHANES 2015-2018. Front Immunol. 2022;13:1058779. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 95] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 42. | He Y, Rodrigues RM, Wang X, Seo W, Ma J, Hwang S, Fu Y, Trojnár E, Mátyás C, Zhao S, Ren R, Feng D, Pacher P, Kunos G, Gao B. Neutrophil-to-hepatocyte communication via LDLR-dependent miR-223-enriched extracellular vesicle transfer ameliorates nonalcoholic steatohepatitis. J Clin Invest. 2021;131:e141513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 147] [Article Influence: 29.4] [Reference Citation Analysis (1)] |
| 43. | Wang F, Zhang X, Liu W, Zhou Y, Wei W, Liu D, Wong CC, Sung JJY, Yu J. Activated Natural Killer Cell Promotes Nonalcoholic Steatohepatitis Through Mediating JAK/STAT Pathway. Cell Mol Gastroenterol Hepatol. 2022;13:257-274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 44. | Fan Y, Zhang W, Wei H, Sun R, Tian Z, Chen Y. Hepatic NK cells attenuate fibrosis progression of non-alcoholic steatohepatitis in dependent of CXCL10-mediated recruitment. Liver Int. 2020;40:598-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 45] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 45. | Wensveen FM, Jelenčić V, Valentić S, Šestan M, Wensveen TT, Theurich S, Glasner A, Mendrila D, Štimac D, Wunderlich FT, Brüning JC, Mandelboim O, Polić B. NK cells link obesity-induced adipose stress to inflammation and insulin resistance. Nat Immunol. 2015;16:376-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 387] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 46. | Khan MA, Khan A. Role of NKT Cells during Viral Infection and the Development of NKT Cell-Based Nanovaccines. Vaccines (Basel). 2021;9:949. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 47. | Wu L, Van Kaer L. Natural killer T cells in health and disease. Front Biosci (Schol Ed). 2011;3:236-251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 48. | Cuño-Gómiz C, de Gregorio E, Tutusaus A, Rider P, Andrés-Sánchez N, Colell A, Morales A, Marí M. Sex-based differences in natural killer T cell-mediated protection against diet-induced steatohepatitis in Balb/c mice. Biol Sex Differ. 2023;14:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 49. | Li T, Lin X, Shen B, Zhang W, Liu Y, Liu H, Wang Y, Zheng L, Zhi F. Akkermansia muciniphila suppressing nonalcoholic steatohepatitis associated tumorigenesis through CXCR6(+) natural killer T cells. Front Immunol. 2022;13:1047570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 50. | Syn WK, Agboola KM, Swiderska M, Michelotti GA, Liaskou E, Pang H, Xie G, Philips G, Chan IS, Karaca GF, Pereira Tde A, Chen Y, Mi Z, Kuo PC, Choi SS, Guy CD, Abdelmalek MF, Diehl AM. NKT-associated hedgehog and osteopontin drive fibrogenesis in non-alcoholic fatty liver disease. Gut. 2012;61:1323-1329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 236] [Cited by in RCA: 227] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 51. | Maricic I, Marrero I, Eguchi A, Nakamura R, Johnson CD, Dasgupta S, Hernandez CD, Nguyen PS, Swafford AD, Knight R, Feldstein AE, Loomba R, Kumar V. Differential Activation of Hepatic Invariant NKT Cell Subsets Plays a Key Role in Progression of Nonalcoholic Steatohepatitis. J Immunol. 2018;201:3017-3035. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 85] [Article Influence: 10.6] [Reference Citation Analysis (2)] |
| 52. | Waller KJ, Saihi H, Li W, Brindley JH, De Jong A, Syn WK, Bessant C, Alazawi W. Single-cell phenotypes of peripheral blood immune cells in early and late stages of non-alcoholic fatty liver disease. Clin Mol Hepatol. 2023;29:417-432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 53. | Li Y, Huang B, Jiang X, Chen W, Zhang J, Wei Y, Chen Y, Lian M, Bian Z, Miao Q, Peng Y, Fang J, Wang Q, Tang R, Gershwin ME, Ma X. Mucosal-Associated Invariant T Cells Improve Nonalcoholic Fatty Liver Disease Through Regulating Macrophage Polarization. Front Immunol. 2018;9:1994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 87] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 54. | Kishi H, Usui I, Jojima T, Fujisaka S, Wakamatsu S, Mizunuma-Inoue Y, Niitani T, Sakurai S, Iijima T, Tomaru T, Tobe K, Aso Y. Increased Number of Mucosal-Associated Invariant T Cells Is Associated with the Inhibition of Nonalcoholic Fatty Liver Disease in High Fat Diet-Fed Mice. Int J Mol Sci. 2022;23:15309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 55. | Deschler S, Pohl-Topcu J, Ramsauer L, Meiser P, Erlacher S, Schenk RP, Maurer HC, Shen P, Kager J, Zink J, Pistrenko K, Monte ER, Weber J, Wasmaier L, Laschinger M, Hüser N, Geisler F, Thorburn D, Nieß H, Wiedemann GM, Zischka H, Heikenwälder M, Kleigrewe K, Mogler C, Böttcher JP, Knolle PA, Schmid RM, Böttcher K. Polyunsaturated fatty acid-induced metabolic exhaustion and ferroptosis impair the anti-tumour function of MAIT cells in MASLD. J Hepatol. 2025;83:1364-1378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 56. | Ebbo M, Crinier A, Vély F, Vivier E. Innate lymphoid cells: major players in inflammatory diseases. Nat Rev Immunol. 2017;17:665-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 277] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 57. | Nguyen HH, Talbot J, Li D, Raghavan V, Littman DR. Modulating intestinal neuroimmune VIPergic signaling attenuates the reduction in ILC3-derived IL-22 and hepatic steatosis in MASLD. Hepatol Commun. 2024;8:e0528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 58. | Hou PF, Yao Y, Wu Y, Yu HT, Qin Y, Yi L, Mi MT. Fecal microbiota transplantation improves hepatic steatosis induced by HFD in a mouse model associated with liver ILC1 regulation and indole-3-carbinol level. Front Nutr. 2025;12:1500293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 59. | Yang M, Qi X, Li N, Kaifi JT, Chen S, Wheeler AA, Kimchi ET, Ericsson AC, Rector RS, Staveley-O'Carroll KF, Li G. Western diet contributes to the pathogenesis of non-alcoholic steatohepatitis in male mice via remodeling gut microbiota and increasing production of 2-oleoylglycerol. Nat Commun. 2023;14:228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 82] [Article Influence: 27.3] [Reference Citation Analysis (1)] |
| 60. | Shrestha S, Jeon JH, Hong CW. Neutrophils in MASLD and MASH. BMB Rep. 2025;58:116-123. [PubMed] [DOI] [Full Text] |
| 61. | Sawada K, Chung H, Softic S, Moreno-Fernandez ME, Divanovic S. The bidirectional immune crosstalk in metabolic dysfunction-associated steatotic liver disease. Cell Metab. 2023;35:1852-1871. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 94] [Article Influence: 31.3] [Reference Citation Analysis (1)] |
| 62. | Shera S, Katzka W, Yang JC, Chang C, Arias-Jayo N, Lagishetty V, Balioukova A, Chen Y, Dutson E, Li Z, Mayer EA, Pisegna JR, Sanmiguel C, Pawar S, Zhang D, Leitman M, Hernandez L, Jacobs JP, Dong TS. Bariatric-induced microbiome changes alter MASLD development in association with changes in the innate immune system. Front Microbiol. 2024;15:1407555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 63. | Yang M, Kimchi ET, Staveley-O'Carroll KF, Li G. Astaxanthin Prevents Diet-Induced NASH Progression by Shaping Intrahepatic Immunity. Int J Mol Sci. 2021;22:11037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 64. | Dai W, Hou Q, Ye J. Rhein alleviates hepatic steatosis in NAFLD mice by activating the AMPK/ACC/SREBP1 pathway to enhance lipid metabolism. Mol Med. 2025;31:255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 65. | Wang D, Liu S, Wan J, Chen S, Feng K, Hou J, Yang Y, Wang P. Activation of TRPA1 prevents metabolic dysfunction-associated steatotic liver disease in diet-induced obese mice through stimulating the AMPK/CPT1A signaling pathway. J Physiol Biochem. 2025;81:359-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 66. | Nunes JRC, O'Dwyer C, Ghorbani P, Smith TKT, Chauhan S, Robert-Gostlin V, Girouard MD, Viollet B, Foretz M, Fullerton MD. Myeloid AMPK signaling restricts fibrosis but is not required for metformin improvements during CDAHFD-induced NASH in mice. J Lipid Res. 2024;65:100564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 67. | Patil NY, Rus I, Ampadu F, Abu Shukair HM, Bonvicino S, Brush RS, Eaton E, Agbaga MP, Oh TG, Friedman JE, Joshi AD. Cinnabarinic acid protects against metabolic dysfunction-associated steatohepatitis by activating aryl hydrocarbon receptor-dependent AMPK signaling. Am J Physiol Gastrointest Liver Physiol. 2025;328:G433-G447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 68. | Sangineto M, Ciarnelli M, Colangelo T, Moola A, Bukke VN, Duda L, Villani R, Romano A, Giandomenico S, Kanwal H, Serviddio G. Monocyte bioenergetics: An immunometabolic perspective in metabolic dysfunction-associated steatohepatitis. Cell Rep Med. 2024;5:101564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 69. | Liu M, Zhang G, Wu S, Song M, Wang J, Cai W, Mi S, Liu C. Schaftoside alleviates HFD-induced hepatic lipid accumulation in mice via upregulating farnesoid X receptor. J Ethnopharmacol. 2020;255:112776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 70. | Shen C, Pan Z, Wu S, Zheng M, Zhong C, Xin X, Lan S, Zhu Z, Liu M, Wu H, Huang Q, Zhang J, Liu Z, Si Y, Tu H, Deng Z, Yu Y, Liu H, Zhong Y, Guo J, Cai J, Xian S. Emodin palliates high-fat diet-induced nonalcoholic fatty liver disease in mice via activating the farnesoid X receptor pathway. J Ethnopharmacol. 2021;279:114340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 71. | Zhang L, Chen J, Yang X, Shen C, Huang J, Zhang D, Liu N, Liu C, Zhong Y, Chen Y, Tang K, Guo J, Cui T, Duan S, Li J, Huang S, Pan H, Zhang H, Tang X, Chang Y, Gao Y. Hepatic Zbtb18 (Zinc Finger and BTB Domain Containing 18) alleviates hepatic steatohepatitis via FXR (Farnesoid X Receptor). Signal Transduct Target Ther. 2024;9:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 72. | McMahan RH, Wang XX, Cheng LL, Krisko T, Smith M, El Kasmi K, Pruzanski M, Adorini L, Golden-Mason L, Levi M, Rosen HR. Bile acid receptor activation modulates hepatic monocyte activity and improves nonalcoholic fatty liver disease. J Biol Chem. 2013;288:11761-11770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 180] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 73. | Zhang Y, Zhou X, Liu P, Chen X, Zhang J, Zhang H, Li S, Chen Y, Song X, Wang J, Zeng H, Zhang X, Tang C, Yu C, Li Y, Xu C. GCSF deficiency attenuates nonalcoholic fatty liver disease through regulating GCSFR-SOCS3-JAK-STAT3 pathway and immune cells infiltration. Am J Physiol Gastrointest Liver Physiol. 2021;320:G531-G542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 74. | Heo YJ, Lee N, Choi SE, Jeon JY, Han SJ, Kim DJ, Kang Y, Lee KW, Kim HJ. Amphiregulin Induces iNOS and COX-2 Expression through NF-κB and MAPK Signaling in Hepatic Inflammation. Mediators Inflamm. 2023;2023:2364121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 75. | Salvoza N, Giraudi P, Gazzin S, Bonazza D, Palmisano S, de Manzini N, Zanconati F, Raseni A, Sirianni F, Tiribelli C, Rosso N. The potential role of omentin-1 in obesity-related metabolic dysfunction-associated steatotic liver disease: evidence from translational studies. J Transl Med. 2023;21:906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 76. | Ye J, Zhu W, Cui Y, Zhang Q, Xiong Y, Jin L, Wang A, Lin M, Dong H, Liang G, Hu X, Luo W. Compound J27 alleviates high-fat diet-induced metabolic dysfunction-associated steatotic liver disease by targeting JNK. Int Immunopharmacol. 2025;154:114570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 77. | Chen Y, Xie K, Chen C, Wang X, Ma C, Huang Z, Jiao Y, Yu W. BMDM-derived ORP8 suppresses lipotoxicity and inflammation by relieving endoplasmic reticulum stress in mice with MASH. Mol Med. 2025;31:213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 78. | Aibara D, Sakaguchi A, Matsusue K. Oxysterol-binding protein-like 3 is a novel target gene of peroxisome proliferator-activated receptor γ in fatty liver disease. Mol Cell Endocrinol. 2023;565:111887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 79. | Chen YX, Wu YP, Zhang Y, Ji PX, Hua J. Modulation of Lipid Metabolism and Keap1-Nrf2 Pathway Activation in Macrophages by Targeting PPARγ Affects NAFLD Progression. J Gastroenterol Hepatol. 2025;40:2119-2133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 80. | Ning M, Lu D, Teng B, Liang D, Ren PG. Comprehensive study of the murine MASH models' applicability by comparing human liver transcriptomes. Life Sci. 2025;376:123723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 81. | Xia J, Xiong W, Yang C, Tan Y, Peng X, Wang W. Swertianin Suppresses M1 Macrophage Polarization and Inflammation in Metabolic Dysfunction-Associated Fatty Liver Disease via PPARG Activation. Genes (Basel). 2025;16:693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 82. | Chang L, Gao J, Yu Y, Liao B, Zhou Y, Zhang J, Ma X, Hou W, Zhou T, Xu Q. MMP10 alleviates non-alcoholic steatohepatitis by regulating macrophage M2 polarization. Int Immunopharmacol. 2023;124:111045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 83. | Jin K, Liu Y, Shi Y, Zhang H, Sun Y, Zhangyuan G, Wang F, Yu W, Wang J, Tao X, Chen X, Zhang W, Sun B. PTPROt aggravates inflammation by enhancing NF-κB activation in liver macrophages during nonalcoholic steatohepatitis. Theranostics. 2020;10:5290-5304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 84. | Liu W, Bai F, Wang H, Liang Y, Du X, Liu C, Cai D, Peng J, Zhong G, Liang X, Ma C, Gao L. Tim-4 Inhibits NLRP3 Inflammasome via the LKB1/AMPKα Pathway in Macrophages. J Immunol. 2019;203:990-1000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 85. | Shi H, Wang X, Sloas C, Gerlach B, Yurdagul A Jr, Moore MP, Jung EJ, Mirshahi F, Ronzoni L, Sanyal AJ, Valenti L, Lin CS, Montgomery J, Zinker B, Klichinsky M, Tabas I. Impaired TIM4-mediated efferocytosis by liver macrophages contributes to fibrosis in metabolic dysfunction-associated steatohepatitis. Sci Transl Med. 2025;17:eadv2106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 86. | Hou PF, Yao Y, Bai Q, Lang HD, Qin Y, Zhu JD, Zhang QY, Yi L, Mi MT. Short term high-fat diet induced liver ILC1 differentiation associated with the TLR9 activation. J Nutr Biochem. 2025;136:109810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 87. | He Q, Zeng J, Yao K, Wang W, Wu Q, Tang R, Xia X, Zou X. Long-term subcutaneous injection of lipopolysaccharides and high-fat diet induced non-alcoholic fatty liver disease through IKKε/ NF-κB signaling. Biochem Biophys Res Commun. 2020;532:362-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 88. | Ganguly S, Rosenthal SB, Ishizuka K, Troutman TD, Rohm TV, Khader N, Aleman-Muench G, Sano Y, Archilei S, Soroosh P, Olefsky JM, Feldstein AE, Kisseleva T, Loomba R, Glass CK, Brenner DA, Dhar D. Lipid-associated macrophages' promotion of fibrosis resolution during MASH regression requires TREM2. Proc Natl Acad Sci U S A. 2024;121:e2405746121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 40.0] [Reference Citation Analysis (0)] |
| 89. | Perugorria MJ, Esparza-Baquer A, Oakley F, Labiano I, Korosec A, Jais A, Mann J, Tiniakos D, Santos-Laso A, Arbelaiz A, Gawish R, Sampedro A, Fontanellas A, Hijona E, Jimenez-Agüero R, Esterbauer H, Stoiber D, Bujanda L, Banales JM, Knapp S, Sharif O, Mann DA. Non-parenchymal TREM-2 protects the liver from immune-mediated hepatocellular damage. Gut. 2019;68:533-546. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 131] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 90. | Wang Z, Chen X, Li J, Chen M, Zhu H, Li X, Yu W, Xia N, Zhang Y, Sun L, Xiao Y, Han S, Pu L, Wang X. Macrophages Atp6v0d2 regulates XBP1-mediated cholesterol metabolism to suppress metabolic dysfunction-associated steatohepatitis progression. Int Immunopharmacol. 2025;161:115088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 91. | Sakamoto Y, Yoshio S, Doi H, Mori T, Matsuda M, Kawai H, Shimagaki T, Yoshikawa S, Aoki Y, Osawa Y, Yoshida Y, Arai T, Itokawa N, Atsukawa M, Ito T, Honda T, Mise Y, Ono Y, Takahashi Y, Saiura A, Taketomi A, Kanto T. Increased Frequency of Dysfunctional Siglec-7(-)CD57(+)PD-1(+) Natural Killer Cells in Patients With Non-alcoholic Fatty Liver Disease. Front Immunol. 2021;12:603133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 92. | Mogna-Peláez P, Riezu-Boj JI, Milagro FI, Herrero JI, Elorz M, Benito-Boillos A, Tobaruela-Resola AL, Tur JA, Martínez JA, Abete I, Zulet MA. Inflammatory markers as diagnostic and precision nutrition tools for metabolic dysfunction-associated steatotic liver disease: Results from the Fatty Liver in Obesity trial. Clin Nutr. 2024;43:1770-1781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 93. | Simeone PG, Costantino S, Liani R, Tripaldi R, Di Castelnuovo A, Tartaro A, Mengozzi A, Cosentino F, Cipollone F, Consoli A, Paneni F, Santilli F. Interleukin-1β in circulating mononuclear cells predicts steatotic liver disease improvement after weight loss in subjects with obesity and prediabetes or type 2 diabetes. Cardiovasc Diabetol. 2025;24:247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (4)] |
| 94. | Zhang YH, Xie R, Dai CS, Gao HW, Zhou G, Qi TT, Wang WY, Wang H, Cui YM. Thyroid hormone receptor-beta agonist HSK31679 alleviates MASLD by modulating gut microbial sphingolipids. J Hepatol. 2025;82:189-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 95. | Harrison SA, Bedossa P, Guy CD, Schattenberg JM, Loomba R, Taub R, Labriola D, Moussa SE, Neff GW, Rinella ME, Anstee QM, Abdelmalek MF, Younossi Z, Baum SJ, Francque S, Charlton MR, Newsome PN, Lanthier N, Schiefke I, Mangia A, Pericàs JM, Patil R, Sanyal AJ, Noureddin M, Bansal MB, Alkhouri N, Castera L, Rudraraju M, Ratziu V; MAESTRO-NASH Investigators. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N Engl J Med. 2024;390:497-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1457] [Cited by in RCA: 1329] [Article Influence: 664.5] [Reference Citation Analysis (2)] |
| 96. | Noureddin M, Alkhouri N, Lawitz EJ, Kowdley KV, Loomba R, Lee L, Jones C, Schlegel A, Marmon T, Anderson K, Li Y, Quirk E, Harrison SA. TERN-501 monotherapy and combination therapy with TERN-101 in metabolic dysfunction-associated steatohepatitis: the randomized phase 2a DUET trial. Nat Med. 2025;31:2297-2305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 97. | Lim SJ. CCL24 Signaling in the Tumor Microenvironment. Adv Exp Med Biol. 2021;1302:91-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 98. | Segal-Salto M, Barashi N, Katav A, Edelshtein V, Aharon A, Hashmueli S, George J, Maor Y, Pinzani M, Haberman D, Hall A, Friedman S, Mor A. A blocking monoclonal antibody to CCL24 alleviates liver fibrosis and inflammation in experimental models of liver damage. JHEP Rep. 2020;2:100064. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (1)] |
| 99. | Mor A, Friedman S, Hashmueli S, Peled A, Pinzani M, Frankel M, Safadi R. Targeting CCL24 in Inflammatory and Fibrotic Diseases: Rationale and Results from Three CM-101 Phase 1 Studies. Drug Saf. 2024;47:869-881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 100. | Tang S, Borlak J. A comparative genomic study across 396 liver biopsies provides deep insight into FGF21 mode of action as a therapeutic agent in metabolic dysfunction-associated steatotic liver disease. Clin Transl Med. 2025;15:e70218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (2)] |
| 101. | Loomba R, Kowdley KV, Rodriguez J, Kim NJ, Alvarez AM, Morrow L, Jeglinski B, Clawson A, Chowdhury S, Bain G, Odrljin T. Efimosfermin alfa (BOS-580), a long-acting FGF21 analogue, in participants with phenotypic metabolic dysfunction-associated steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2a trial. Lancet Gastroenterol Hepatol. 2025;10:734-745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 102. | Mekontso JGK, Nnang JYB, Tembi TBT, Kortim AB, Nguefang GL, Wagner J, Bernstein M. Efficacy, Safety, and Tolerability of Farnesoid X Receptor Agonists in the Treatment of Metabolic Dysfunction-associated Steatotic Liver Disease: A Systematic Review and Meta-analysis. J Clin Exp Hepatol. 2025;15:102563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 103. | Kumar J, Hasan M, Mohsin S, Alzaher MH, Nagar T, Jamil A, Ahmed A, Lavu VK, Kumar S. Assessing the efficacy of farnesoid X receptor agonists in the management of metabolic dysfunction-associated steatotic liver disease: A systematic review and meta-analysis: Efficacy of Farnesoid X Receptor Agonists in Metabolic Dysfunction-associated Steatotic Liver Disease: Systematic Review and Meta-analysis. Clin Res Hepatol Gastroenterol. 2025;49:102530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 104. | Ratziu V, Harrison SA, Loustaud-Ratti V, Bureau C, Lawitz E, Abdelmalek M, Alkhouri N, Francque S, Girma H, Darteil R, Couchoux H, Wolf M, Sanyal A, Vonderscher J, Scalfaro P. Hepatic and renal improvements with FXR agonist vonafexor in individuals with suspected fibrotic NASH. J Hepatol. 2023;78:479-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 82] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 105. | Nagasawa T, Inada Y, Nakano S, Tamura T, Takahashi T, Maruyama K, Yamazaki Y, Kuroda J, Shibata N. Effects of bezafibrate, PPAR pan-agonist, and GW501516, PPARdelta agonist, on development of steatohepatitis in mice fed a methionine- and choline-deficient diet. Eur J Pharmacol. 2006;536:182-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 179] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 106. | Peyrou M, Ramadori P, Bourgoin L, Foti M. PPARs in Liver Diseases and Cancer: Epigenetic Regulation by MicroRNAs. PPAR Res. 2012;2012:757803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 107. | Liu H, Yin G, Kohlhepp MS, Schumacher F, Hundertmark J, Hassan MIA, Heymann F, Puengel T, Kleuser B, Mosig AS, Tacke F, Guillot A. Dissecting Acute Drug-Induced Hepatotoxicity and Therapeutic Responses of Steatotic Liver Disease Using Primary Mouse Liver and Blood Cells in a Liver-On-A-Chip Model. Adv Sci (Weinh). 2024;11:e2403516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 108. | Lefere S, Puengel T, Hundertmark J, Penners C, Frank AK, Guillot A, de Muynck K, Heymann F, Adarbes V, Defrêne E, Estivalet C, Geerts A, Devisscher L, Wettstein G, Tacke F. Differential effects of selective- and pan-PPAR agonists on experimental steatohepatitis and hepatic macrophages(☆). J Hepatol. 2020;73:757-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 241] [Article Influence: 40.2] [Reference Citation Analysis (0)] |
| 109. | Barb D, Kalavalapalli S, Godinez Leiva E, Bril F, Huot-Marchand P, Dzen L, Rosenberg JT, Junien JL, Broqua P, Rocha AO, Lomonaco R, Abitbol JL, Cooreman MP, Cusi K. Pan-PPAR agonist lanifibranor improves insulin resistance and hepatic steatosis in patients with T2D and MASLD. J Hepatol. 2025;82:979-991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 51] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 110. | Tanigawa R, Nakajima A, Eguchi Y, Takahashi H, Loomba R, Suganami H, Tanahashi M, Saito A, Iida Y, Yamashita S. Effects of Pemafibrate on LDL-C and Related Lipid Markers in Patients with MASLD: A Sub-Analysis of the PEMA-FL Study. J Atheroscler Thromb. 2025;32:823-839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 111. | Shinkai Y, Sasaki K, Tamura R, Ike T, Takahashi A, Osaki Y, Ishiuchi N, Maeoka Y, Nakashima A, Masaki T. Selective activation of PPARα by pemafibrate mitigates peritoneal inflammation and fibrosis through suppression of NLRP3 inflammasome and modulation of inflammation. Sci Rep. 2024;14:23816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 112. | le Roux CW, Steen O, Lucas KJ, Startseva E, Unseld A, Hennige AM. Glucagon and GLP-1 receptor dual agonist survodutide for obesity: a randomised, double-blind, placebo-controlled, dose-finding phase 2 trial. Lancet Diabetes Endocrinol. 2024;12:162-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 141] [Article Influence: 70.5] [Reference Citation Analysis (4)] |
| 113. | Sanyal AJ, Bedossa P, Fraessdorf M, Neff GW, Lawitz E, Bugianesi E, Anstee QM, Hussain SA, Newsome PN, Ratziu V, Hosseini-Tabatabaei A, Schattenberg JM, Noureddin M, Alkhouri N, Younes R; 1404-0043 Trial Investigators. A Phase 2 Randomized Trial of Survodutide in MASH and Fibrosis. N Engl J Med. 2024;391:311-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 322] [Article Influence: 161.0] [Reference Citation Analysis (1)] |
| 114. | Harrison SA, Alkhouri N, Ortiz-Lasanta G, Rudraraju M, Tai D, Wack K, Shah A, Besuyen R, Steineger HH, Fraser DA, Sanyal AJ; ICONA Study Investigators. A phase IIb randomised-controlled trial of the FFAR1/FFAR4 agonist icosabutate in MASH. J Hepatol. 2025;83:293-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 115. | Simon TG, Wilechansky RM, Stoyanova S, Grossman A, Dichtel LE, Lauer GM, Miller KK, Hoshida Y, Corey KE, Loomba R, Chung RT, Chan AT. Aspirin for Metabolic Dysfunction-Associated Steatotic Liver Disease Without Cirrhosis: A Randomized Clinical Trial. JAMA. 2024;331:920-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 64] [Article Influence: 32.0] [Reference Citation Analysis (1)] |
| 116. | Ebrahimpour-Koujan S, Sohrabpour AA, Giovannucci E, Vatannejad A, Esmaillzadeh A. Effects of vitamin D supplementation on liver fibrogenic factors, vitamin D receptor and liver fibrogenic microRNAs in metabolic dysfunction-associated steatotic liver disease (MASLD) patients: an exploratory randomized clinical trial. Nutr J. 2024;23:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 117. | Podszun MC, Alawad AS, Lingala S, Morris N, Huang WCA, Rotman Y. Development of Subtle Iron Deficiency During Vitamin E Treatment For Metabolic Dysfunction-Associated Steatotic Liver Disease. J Diet Suppl. 2025;22:284-299. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 118. | Armandi A, Bespaljko H, Mang A, Huber Y, Michel M, Labenz C, Galle PR, Neerukonda M, Bugianesi E, Schuppan D, Schattenberg JM. Short-term reduction of dietary gluten improves metabolic-dysfunction associated steatotic liver disease: A randomised, controlled proof-of-concept study. Aliment Pharmacol Ther. 2024;59:1212-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 119. | Xiang L, Wang G, Zhuang Y, Luo L, Yan J, Zhang H, Li X, Xie C, He Q, Peng Y, Chen H, Li Q, Li X, Guo L, Lv G, Ding Y. Safety and efficacy of GLP-1/FGF21 dual agonist HEC88473 in MASLD and T2DM: A randomized, double-blind, placebo-controlled study. J Hepatol. 2025;82:967-978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 120. | Qi X, Yang M, Stenberg J, Dey R, Fogwe L, Alam MS, Kimchi ET, Staveley-O'Carroll KF, Li G. Gut microbiota mediated molecular events and therapy in liver diseases. World J Gastroenterol. 2020;26:7603-7618. [PubMed] [DOI] [Full Text] |
| 121. | Zhang C, Yang M. Current Options and Future Directions for NAFLD and NASH Treatment. Int J Mol Sci. 2021;22:7571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (1)] |
| 122. | Choi J, Choi YR, Jeong MK, Song HH, Yu JS, Song SH, Park JH, Kim MJ, Park H, Ham YL, Han SH, Kim DJ, Lee DY, Suk KT. Phocaeicola dorei ameliorates progression of steatotic liver disease by regulating bile acid, lipid, inflammation and proliferation. Gut Microbes. 2025;17:2539448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 123. | Khodadadi N, Sadeghi A, Poustchi H, Abbasi B, Nilghaz M, Melekoglu E, Yari Z, Hekmatdoost A. Effectiveness of flaxseed consumption and fasting mimicking diet on anthropometric measures, biochemical parameters, and hepatic features in patients with Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): a randomized controlled clinical trial. Nutr Diabetes. 2024;14:93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 124. | Sun X, Li F, Yan H, Chang X, Yao X, Yang X, Wu S, Suo Y, Zhu X, Wang C, Gao J, Wang H, Chen Y, Xia M, Bian H, Gao X. Intermittent compared with continuous calorie restriction for treatment of metabolic dysfunction-associated steatotic liver disease: a randomized clinical trial. Am J Clin Nutr. 2025;121:158-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 125. | Lambert JE, Ramos-Roman MA, Valdez MJ, Browning JD, Rogers T, Parks EJ. Weight loss in MASLD restores the balance of liver fatty acid sources. J Clin Invest. 2025;135:e174233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 126. | Tsitsou S, Bali T, Adamantou M, Saridaki A, Poulia KA, Karagiannakis DS, Papakonstantinou E, Cholongitas E. Effects of a 12-Week Mediterranean-Type Time-Restricted Feeding Protocol in Patients With Metabolic Dysfunction-Associated Steatotic Liver Disease: A Randomised Controlled Trial-The 'CHRONO-NAFLD Project'. Aliment Pharmacol Ther. 2025;61:1290-1309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 127. | van den Burg EL, Schoonakker MP, van Peet PG, le Cessie S, Numans ME, Pijl H, Lamb HJ. A fasting-mimicking diet programme reduces liver fat and liver inflammation/fibrosis measured by magnetic resonance imaging in patients with type 2 diabetes. Clin Nutr. 2025;47:136-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |