Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.119002
Revised: February 5, 2026
Accepted: March 2, 2026
Published online: March 28, 2026
Processing time: 62 Days and 17.2 Hours
The co-occurrence of hereditary hemorrhagic telangiectasia (HHT) and hepatitis C cirrhosis represents an exceptionally rare clinical entity, which can pose a diag
We reported a patient with portal hypertension-induced esophageal and gastric variceal rupture due to HHT-hepatic arterioportal fistula and hepatitis C cirrhosis. Computed tomography confirmed hepatic diffuse arteriovenous fistula. Fol
TIPS is an effective treatment option for esophageal and gastric variceal bleeding secondary to portal hypertension in patients with both HHT and liver cirrhosis.
Core Tip: Interventional embolization remains the first-line treatment for hepatic arterioportal fistula. However, when embolization fails to achieve hemostasis, transjugular intrahepatic portosystemic shunt (TIPS) may serve as an alternative therapeutic strategy. Herein, we report a female patient with liver cirrhosis and hereditary hemorrhagic telangiectasia who presented with recurrent bleeding and was successfully managed with TIPS.
- Citation: Zhang TQ, Zhang L, Yong X, Tian C, Chen BJ, Qin JP, Mu D, Tang SH. Transjugular intrahepatic portosystemic shunt for variceal bleeding due to hereditary hemorrhagic telangiectasia with cirrhosis: A case report. World J Gastroenterol 2026; 32(12): 119002
- URL: https://www.wjgnet.com/1007-9327/full/v32/i12/119002.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i12.119002
Hereditary hemorrhagic telangiectasia (HHT) is an autosomal dominant hereditary vascular disease with a prevalence of approximately 1 in 5000. Small arteriovenous malformations are the hallmark of HHT[1]. Hepatic arterioportal fistula (HAPF), a common type of vascular malformation in present case, leads to variceal bleeding due to portal hypertension. Transcatheter embolization of diffuse arterioportal fistulas is ineffective in treating such conditions, thus necessitating the exploration of more effective treatment options. This case report successfully treated a patient with hepatitis C cirrhosis and HHT complicated by HAPF and variceal bleeding using the transjugular intrahepatic portosystemic shunt (TIPS) method, which has rarely been reported before. The aim of this report is to provide more references and options for the treatment of patients with decompensated liver cirrhosis and HHT HAPF.
A 61-year-old female was admitted, presenting with a three-month history of abdominal distension and a one-month history of melaena.
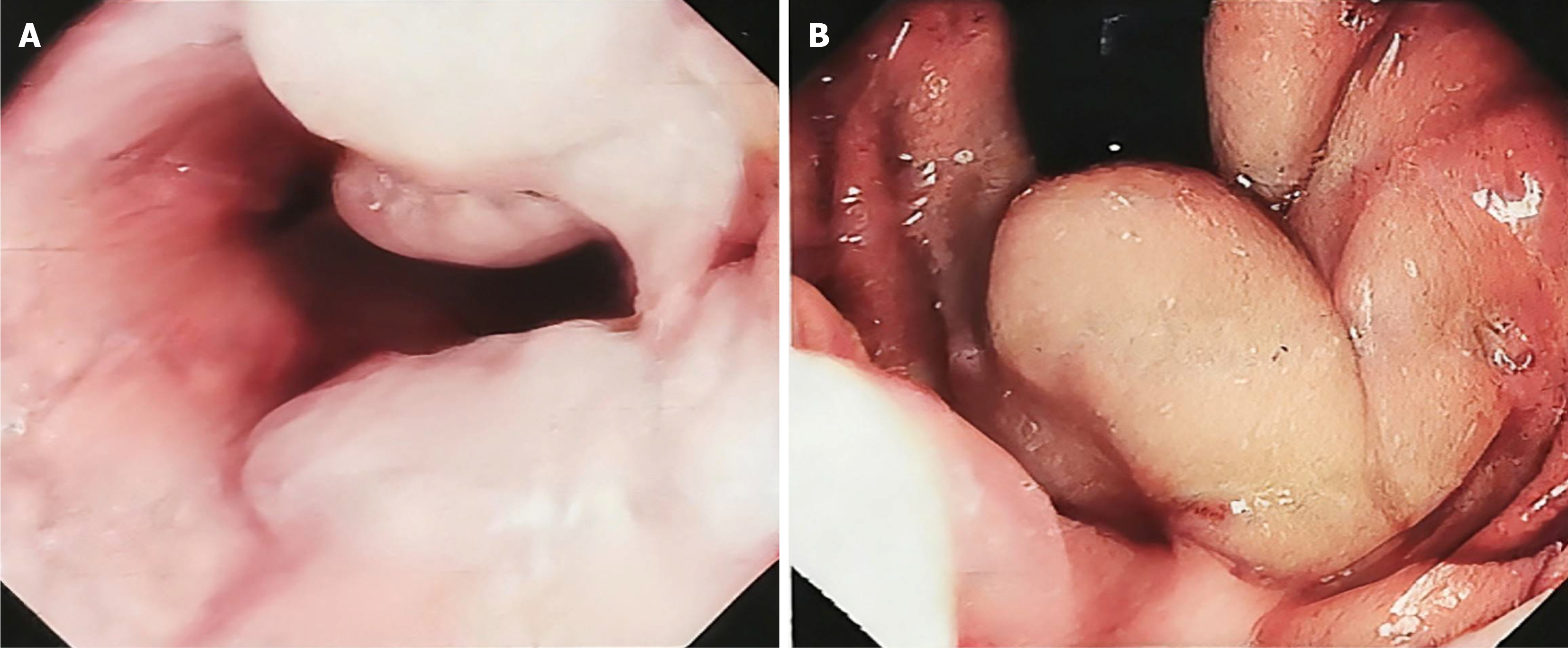
Three months prior to admission, the patient developed unexplained abdominal distension, which persisted despite initial management at a local hospital. One month before admission, her condition evolved to include three episodes of melaena and two episodes of hematemesis with blood clots, accompanied by symptoms of dizziness and fatigue. Subsequent comprehensive evaluation at local hospital revealed a positive hepatitis C cirrhosis antibody result. And the patient received anti hepatitis C virus treatment. Gastroscopy revealed esophagogastric varices (severe RC+), and chronic non-atrophic gastritis with erosion (Figure 1).
The patient had no history of past illness.
No family history of chronic hepatic disease was elicited.
On examination, the patient was conscious with an anemic complexion. Cutaneous telangiectasias were visible on the hands, and the tongue mucosa exhibited similar vascular lesions. Cardiopulmonary auscultation revealed clear lung fields and a regular heart rhythm without murmurs. Abdominal examination revealed no obvious positive signs.
Blood routine revealed platelet count 123 × 109, hemoglobin concentration 99 g/L, red blood cell count: 3.40 × 1012/L. liver function was basically normal with Child-Pugh grade A. Serological testing for hepatitis and autoimmune antibodies yielded negative results. The quantitative detection of hepatitis C virus RNA was negative.
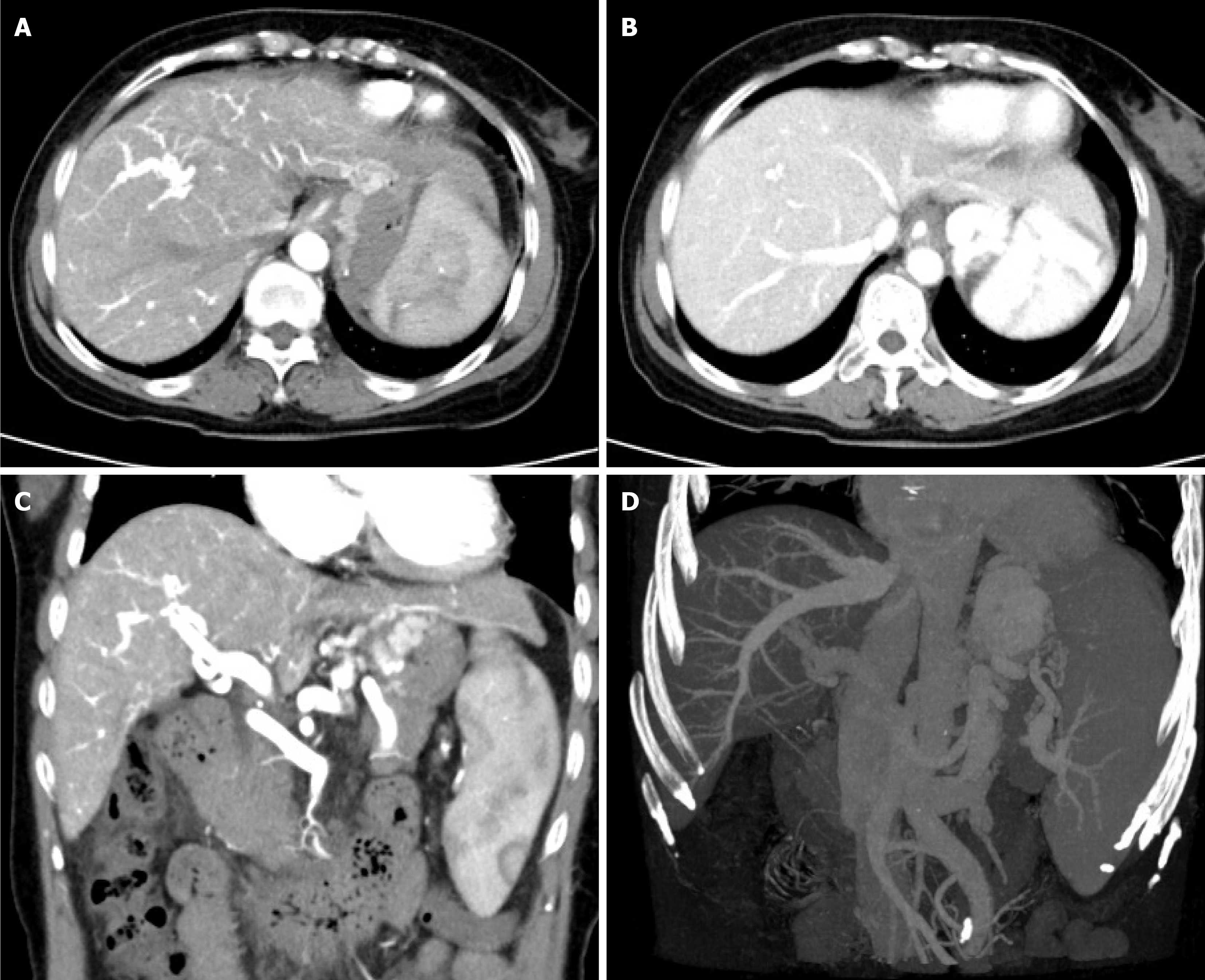
Contrast-enhanced computed tomography (CT) portal angiography revealed: (1) Early enhancement of the main and left/right branches of the portal vein in hepatic arterial-phase, suggesting possible arterioportal fistula; and (2) Liver cirrhosis and splenomegaly with diameter of portal vein being 1.5 cm. Small low-density shadows at the edge of the main portal vein, suspected embolism; left branch portal vein narrowed with spongiform degeneration and multiple collateral circulation formation (Figure 2).
Based on the above results, the patient was diagnosed with diffuse intrahepatic arterioportal fistula, portal hypertension and cavernous transformation of the portal vein. Further history taking revealed a long-standing but previously un
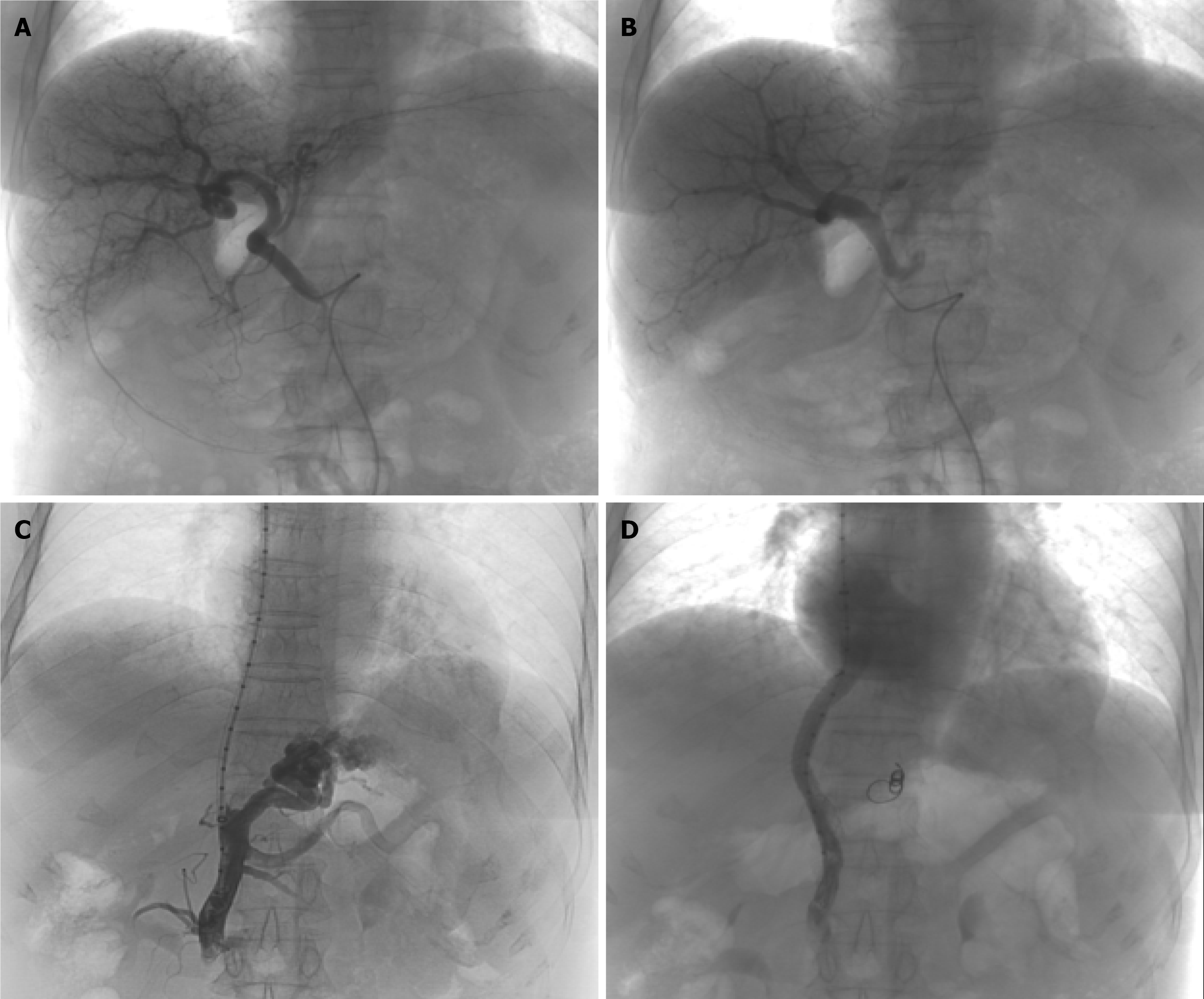
Variceal bleeding was therefore deemed secondary to both liver cirrhosis and high-flow arterioportal fistula. Then, the patient was treated with hepatic partial arterioportal fistula embolization under digital subtraction angiography (Figure 3A and B). However, melaena recurred two days post-operatively, suggesting persistent portal hypertension secondary to multiple fistulas even after embolization. Following comprehensive multidisciplinary evaluation and in
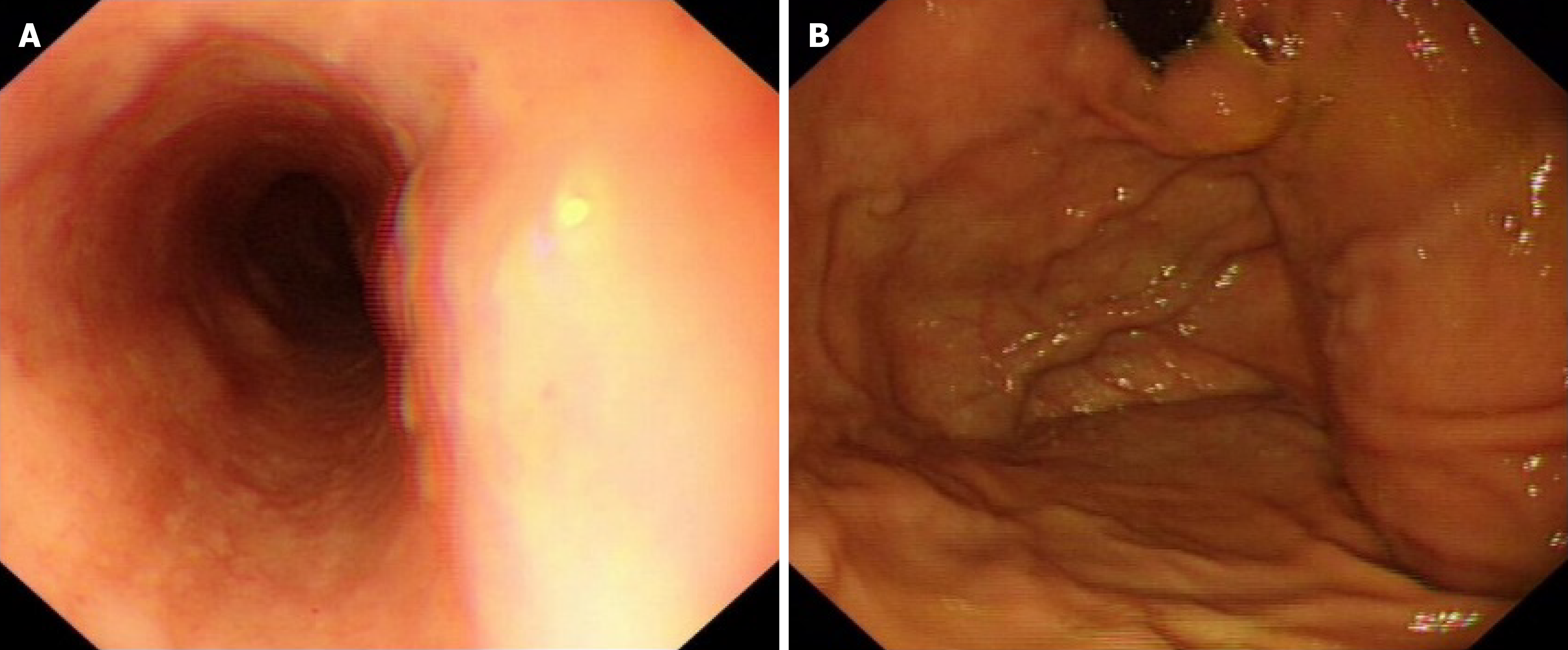
One month postoperatively, endoscopic follow-up revealed complete resolution of esophageal varices (Figure 4). No postoperative complications including elevated blood ammonia levels or hepatic encephalopathy were observed during subsequent approximately 5-month follow-up.
HHT, also known as Osler-Weber-Rendu syndrome, typically presents with arteriovenous malformations in organs such as the brain, lungs, gastrointestinal tract, and liver, manifesting as epistaxis, gastrointestinal bleeding, iron-deficiency anemia, and characteristic mucocutaneous telangiectasias[2,3]. And loss-of-function mutations in bone morphogenetic protein 9-10/ENG/ALK1/SMAD4 pathway were supposed to be the cause[4]. Based on the diagnostic criteria of HHT[5] and clinical manifestations of the patient, a definitive diagnosis of HHT is supported. Studies indicate that complications such as chronic gastrointestinal bleeding, anemia, symptomatic hepatic arteriovenous malformations, and neurological disorders (stroke, brain abscess) significantly increase mortality rates in HHT patients[5,6]. Therefore, proactive ma
In conclusion, TIPS represents a critical and effective intervention for patients with portal hypertension secondary to both liver cirrhosis and HAPF, though ongoing surveillance is warranted to ensure sustained positive outcomes.
We would like to acknowledge our patient for her support.
| 1. | Al Tabosh T, Al Tarrass M, Tourvieilhe L, Guilhem A, Dupuis-Girod S, Bailly S. Hereditary hemorrhagic telangiectasia: from signaling insights to therapeutic advances. J Clin Invest. 2024;134:e176379. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 47] [Reference Citation Analysis (1)] |
| 2. | Faughnan ME, Palda VA, Garcia-Tsao G, Geisthoff UW, McDonald J, Proctor DD, Spears J, Brown DH, Buscarini E, Chesnutt MS, Cottin V, Ganguly A, Gossage JR, Guttmacher AE, Hyland RH, Kennedy SJ, Korzenik J, Mager JJ, Ozanne AP, Piccirillo JF, Picus D, Plauchu H, Porteous ME, Pyeritz RE, Ross DA, Sabba C, Swanson K, Terry P, Wallace MC, Westermann CJ, White RI, Young LH, Zarrabeitia R; HHT Foundation International - Guidelines Working Group. International guidelines for the diagnosis and management of hereditary haemorrhagic telangiectasia. J Med Genet. 2011;48:73-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 859] [Cited by in RCA: 705] [Article Influence: 47.0] [Reference Citation Analysis (1)] |
| 3. | Shovlin CL, Guttmacher AE, Buscarini E, Faughnan ME, Hyland RH, Westermann CJ, Kjeldsen AD, Plauchu H. Diagnostic criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber syndrome). Am J Med Genet. 2000;91:66-67. [PubMed] [DOI] [Full Text] |
| 4. | Desroches-Castan A, Tillet E, Bouvard C, Bailly S. BMP9 and BMP10: Two close vascular quiescence partners that stand out. Dev Dyn. 2022;251:178-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (1)] |
| 5. | Thompson KP, Nelson J, Kim H, Pawlikowska L, Marchuk DA, Lawton MT, Faughnan ME; Brain Vascular Malformation Consortium HHT Investigator Group. Predictors of mortality in patients with hereditary hemorrhagic telangiectasia. Orphanet J Rare Dis. 2021;16:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 6. | Donaldson JW, McKeever TM, Hall IP, Hubbard RB, Fogarty AW. Complications and mortality in hereditary hemorrhagic telangiectasia: A population-based study. Neurology. 2015;84:1886-1893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 84] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 7. | Gryboski JD, Clemett A. Congenital hepatic artery aneurysm with superior mesenteric artery insufficiency: a steal syndrome. Pediatrics. 1967;39:344-347. [PubMed] |
| 8. | Guzman EA, McCahill LE, Rogers FB. Arterioportal fistulas: introduction of a novel classification with therapeutic implications. J Gastrointest Surg. 2006;10:543-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 84] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 9. | Strodel WE, Eckhauser FE, Lemmer JH, Whitehouse WM Jr, Williams DM. Presentation and perioperative management of arterioportal fistulas. Arch Surg. 1987;122:563-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 37] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 10. | Tang S, Zeng W, Qin J, He Q, Wu X, Zhou X, Wang Z, Zeng J, Jin Z, Jiang M. Gastric Variceal Bleeding Caused by an Arterioportal Fistula Formation After TIPS and Related Complications. Dig Dis Sci. 2016;61:2442-2444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 11. | Vauthey JN, Tomczak RJ, Helmberger T, Gertsch P, Forsmark C, Caridi J, Reed A, Langham MR Jr, Lauwers GY, Goffette P, Lerut J. The arterioportal fistula syndrome: clinicopathologic features, diagnosis, and therapy. Gastroenterology. 1997;113:1390-1401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 128] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 12. | Citone M, Annese A, Gabbani G, Pindozzi F, Falcone G, Casamassima E, Santolupo A, Aspite S, Ragozzino L, Falcini M, Biagi F, Rosi M, Adotti V, Dragoni G, Roccarina D, Innocenti T, Messerini L, Gitto S, Mondaini F, Marra F, Fanelli F, Vizzutti F. Hepatic barrage to high-flow, intra-hepatic arteroportal fistulas requiring combined interventional approach. BJR Case Rep. 2025;11:uaaf034. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 13. | Kumar A, Ahuja CK, Vyas S, Kalra N, Khandelwal N, Chawla Y, Dhiman RK. Hepatic arteriovenous fistulae: role of interventional radiology. Dig Dis Sci. 2012;57:2703-2712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 14. | Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: Risk stratification, diagnosis, and management: 2016 practice guidance by the American Association for the study of liver diseases. Hepatology. 2017;65:310-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1775] [Cited by in RCA: 1550] [Article Influence: 172.2] [Reference Citation Analysis (5)] |
| 15. | Fortune BE, Garcia-Tsao G, Ciarleglio M, Deng Y, Fallon MB, Sigal S, Chalasani NP, Lim JK, Reuben A, Vargas HE, Abrams G, Lewis MD, Hassanein T, Trotter JF, Sanyal AJ, Beavers KL, Ganger D, Thuluvath PJ, Grace ND, Groszmann RJ; Vapreotide Study Group. Child-Turcotte-Pugh Class is Best at Stratifying Risk in Variceal Hemorrhage: Analysis of a US Multicenter Prospective Study. J Clin Gastroenterol. 2017;51:446-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 71] [Article Influence: 7.9] [Reference Citation Analysis (2)] |
| 16. | Rajesh S, George T, Philips CA, Ahamed R, Kumbar S, Mohan N, Mohanan M, Augustine P. Transjugular intrahepatic portosystemic shunt in cirrhosis: An exhaustive critical update. World J Gastroenterol. 2020;26:5561-5596. [PubMed] [DOI] [Full Text] |
| 17. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on TIPS. J Hepatol. 2025;83:177-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 50.0] [Reference Citation Analysis (1)] |