Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.116287
Revised: December 25, 2025
Accepted: January 16, 2026
Published online: March 28, 2026
Processing time: 132 Days and 18.2 Hours
Chronic hepatitis B (CHB) is a major global health burden, with China being the most affected. Achieving a clinical cure, defined as hepatitis B surface antigen (HBsAg) clearance, is the ideal treatment endpoint. While a 48-week interferon course is standard, extended therapy may improve HBsAg clearance rates. However, there exists a no
To develop a predictive model for identifying patients who require extended interferon therapy (≥ 48 weeks) for HBsAg clearance.
This multicenter retrospective study included CHB patients, including those with compensated cirrhosis, who achieved HBsAg clearance (HBsAg < 0.05 IU/mL) following treatment with pegylated interferon alpha-2b, either alone or in combination with nucleoside analogs. After propensity score matching, we employed least absolute sh
A total of 688 eligible patients with CHB were enrolled in this study. After propensity score matching at a 1:1 ratio, 375 patients remained, including 196 in the training cohort. Among the training cohort, 36 (18.37%) were classified in the extended course (≥ 48 weeks) and 160 (81.63%) in the regular course (< 48 weeks). LASSO and multivariate regression analyses identified baseline HBsAg and cirrhosis as significant risk factors for extended interferon therapy. The model demonstrated strong discriminatory ability, with the area under the curve of 0.83 [95% con
This study successfully constructed and validated a prediction model based on baseline HBsAg and cirrhosis to identify potential populations that may benefit from extended interferon therapy (≥ 48 weeks).
Core Tip: This study’s innovation lies in its exploration of the factors influencing extended interferon therapy (≥ 48 weeks) through multicenter cohort data, thereby avoiding the premature termination of potentially effective therapies and providing an evidence-based foundation for individualized treatment planning. For the first time, it incorporates baseline cirrhosis status and constructs a predictive model that integrates baseline hepatitis B surface antigen levels. The model demonstrated strong discriminatory ability in both the training cohort and the externally validated cohort.
- Citation: Yan F, Xue XL, Guo Y, Zhang QR, You RR, Shang J, Wu XP, Geng JW, Gao XH, Ye Q, Liang J, Wang XY, Zeng JY, Chen J, Lin YC, Chen XY, Du Q, Yin WL, Liu L, Wang F, Xu BG, Zhang WH, Xiang HL. Baseline hepatitis B surface antigen and cirrhosis predict extended interferon therapy in chronic hepatitis B: A retrospective study. World J Gastroenterol 2026; 32(12): 116287
- URL: https://www.wjgnet.com/1007-9327/full/v32/i12/116287.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i12.116287
Chronic hepatitis B (CHB) represents a significant global public health challenge, affecting approximately 257 million individuals worldwide. China bears the highest burden of chronic hepatitis B virus (HBV) infection, with an estimated 86 million cases, which poses substantial challenges to the healthcare system[1-5].
Clinical cure, also referred to as functional cure, is defined by the persistent undetectable serum hepatitis B surface antigen (HBsAg) and HBV DNA, negative hepatitis B envelope antigen (HBeAg), and HBsAg seroconversion following a limited treatment course. Achieving clinical cure markedly reduces the risk of cirrhosis and hepatocellular carcinoma. A review conducted by Jeng and Lok[6] underscores that HBsAg clearance remains the ideal objective of contemporary hepatitis B treatment, and the incorporation of pegylated interferon α-2b (Peg-IFNα-2b) therapy in eligible patients is an effective strategy to attain clinical cure[7-10]. National and international guidelines recommend a routine interferon treatment course of 48 weeks for adult CHB patients. However, studies have indicated that an extended treatment duration may enhance the HBsAg clearance rate[11]. Existing research has predominantly concentrated on identifying populations that benefit from a standard treatment course (48 weeks), characterized by factors such as low HBsAg levels and high alanine aminotransferase (ALT)[12,13]. Nonetheless, there exists a notable gap in predictive modeling studies concerning extended treatment courses (≥ 48 weeks), and the baseline characteristics of patients who may necessitate such extended treatments remain inadequately defined.
This study aims to develop and validate a predictive model to identify populations requiring extended interferon therapy (≥ 48 weeks) for HBsAg clearance. The model seeks to prevent premature discontinuation of effective treatments and provide an evidence-based rationale for individualized treatment regimens through a multicenter retrospective cohort analysis.
This multicenter, retrospective study included patients with CHB who attended hospitals participating in the China Reduction of Hepatocellular Carcinoma Incidence in Hepatitis B Patients Study (OASIS) project across Tianjin, Xiamen (Fujian Province), and Taiyuan (Shanxi Province), China, from January 2018 to March 2024. Part of the data is sourced from the OASIS project, with data cleaning and quality control handled by National Medical Center of Infectious Diseases Liver Diseases Research Group. These patients received PEG-IFNα-2b either as monotherapy or in conjunction with nucleoside analogs (NAs).
Inclusion criteria: (1) Participants must be aged between 18 and 70 years; (2) They should have a diagnosis of CHB or hepatitis B-related compensated cirrhosis, as defined by the Chinese Guidelines for the Prevention and Control of CHB (2022); (3) Participants must be receiving Peg-IFNα-2b and have achieved HBsAg clearance during therapy; and (4) They must not have received any immunosuppressive agents or hormonal drugs.
Exclusion criteria: (1) Individuals with viral hepatitis other than CHB, such as hepatitis A, C, D, or E; (2) Those who have interrupted Peg-IFNα-2b treatment for more than three months; and (3) Participants with incomplete case data.
Baseline information, including gender, age, initiation of Peg-IFNα-2b treatment, and the time of HBsAg clearance (defined as the first clearance of HBsAg), was collected. Baseline laboratory indicators included HBsAg, HBeAg, HBV DNA, white blood cell, platelet count, hemoglobin, ALT, total bilirubin, alkaline phosphatase, gamma-glutamyl trans
Patients were divided into a training cohort (from Tianjin and Xiamen) and a validation cohort (from other sites) based on hospital origin. The propensity score matching (PSM) method was employed to match the cohorts in a 1:1 ratio to minimize heterogeneity regarding age, gender, HBeAg status, and baseline HBV DNA levels.
The assignment to extended interferon treatment was guided primarily by real-world clinical practice. Based on the duration of Peg-IFNα-2b therapy, patients were categorized into two groups: The extended-duration group (≥ 48 weeks) and the standard-duration group (< 48 weeks). In clinical practice, treatment decisions are typically guided by the dynamic change of HBsAg during therapy. Physicians generally consider continuing IFN if a sustained decline in HBsAg is observed. Conversely, treatment is often paused or discontinued if HBsAg plateau is reached or if intolerable adverse events emerge.
HBV markers were assayed using the Abbott ARCHITECT i4000SR system (Abbott Diagnostics, Abbott Park, IL, United States), employing chemiluminescent microparticle immunoassay technology. The following reagent kits were utilized: HBsAg reagent kit (6C36/08P08, Abbott, Ireland), Anti-HBs reagent kit (7C18/07P89, Abbott, Ireland), HBeAg reagent kit (6C32/07P64, Abbott GmbH, Germany), and Anti-HBe reagent kit (6C34/07P63, Abbott GmbH, Germany). According to the manufacturer’s instructions, a result of HBsAg < 0.05 IU/mL was considered negative; Anti-HBs ≥ 10 mIU/mL was also considered negative; HBeAg signal/cutoff (S/CO) value < 1.00 was negative; and Anti-HBe S/CO value > 1.00 was negative.
HBV DNA assays typically utilize two detection techniques. The first technique employs the Anadas9850 platform for automated nucleic acid extraction and real-time polymerase chain reaction system, utilizing the HBV DNA reagent kit (Amplly, China), which has a detection range exceeding 50 IU/mL. According to the manufacturer’s instructions, results indicating HBV DNA levels below 50 IU/mL are considered negative. The second technique involves automated sample processing with an AmpliPrep analyzer, followed by automated nucleic acid amplification and detection using a TaqMan48 analyzer (Roche, Pleasanton, CA, United States), also utilizing the HBV DNA reagent kit (Roche, Pleasanton, CA, United States), which has a detection range of over 20 IU/mL. As per the manufacturer’s guidelines, HBV DNA levels below 20 IU/mL are classified as negative.
Biochemical parameters and peripheral blood counts were analyzed using an automated biochemical analyzer and an automated blood cell analyzer (Wako Pure Chemical Industries, Ltd., Tokyo, Japan) for whole blood testing. The me
Continuous variables were expressed as mean ± SD or median (interquartile range), while categorical variables were presented as n (%). Differences among continuous variables were assessed using t test or Kruskal-Wallis one-way analysis of variance, and categorical variables were compared using the χ2 test or Fisher’s exact test as appropriate. Serum HBsAg and HBV DNA levels were logarithmically transformed for analysis. PSM was conducted based on baseline age, gender, HBeAg, HBV DNA, and ALT levels, utilizing a caliper width of 0.25 times the standard deviation of the propensity score. Variables were screened using least absolute shrinkage and selection operator (LASSO) regression and multivariable logistic regression. Model performance was validated using the area under the receiver operating characteristic curve (AUC) and bootstrap resampling (500 iterations). Subgroup analyses were performed among individuals with varying baseline ALT levels, cirrhosis status, and interferon treatment duration. The robustness of the results was assessed using likelihood ratio tests for interaction. Statistical analyses were conducted using R version 4.4.1 and EmpowerStats soft
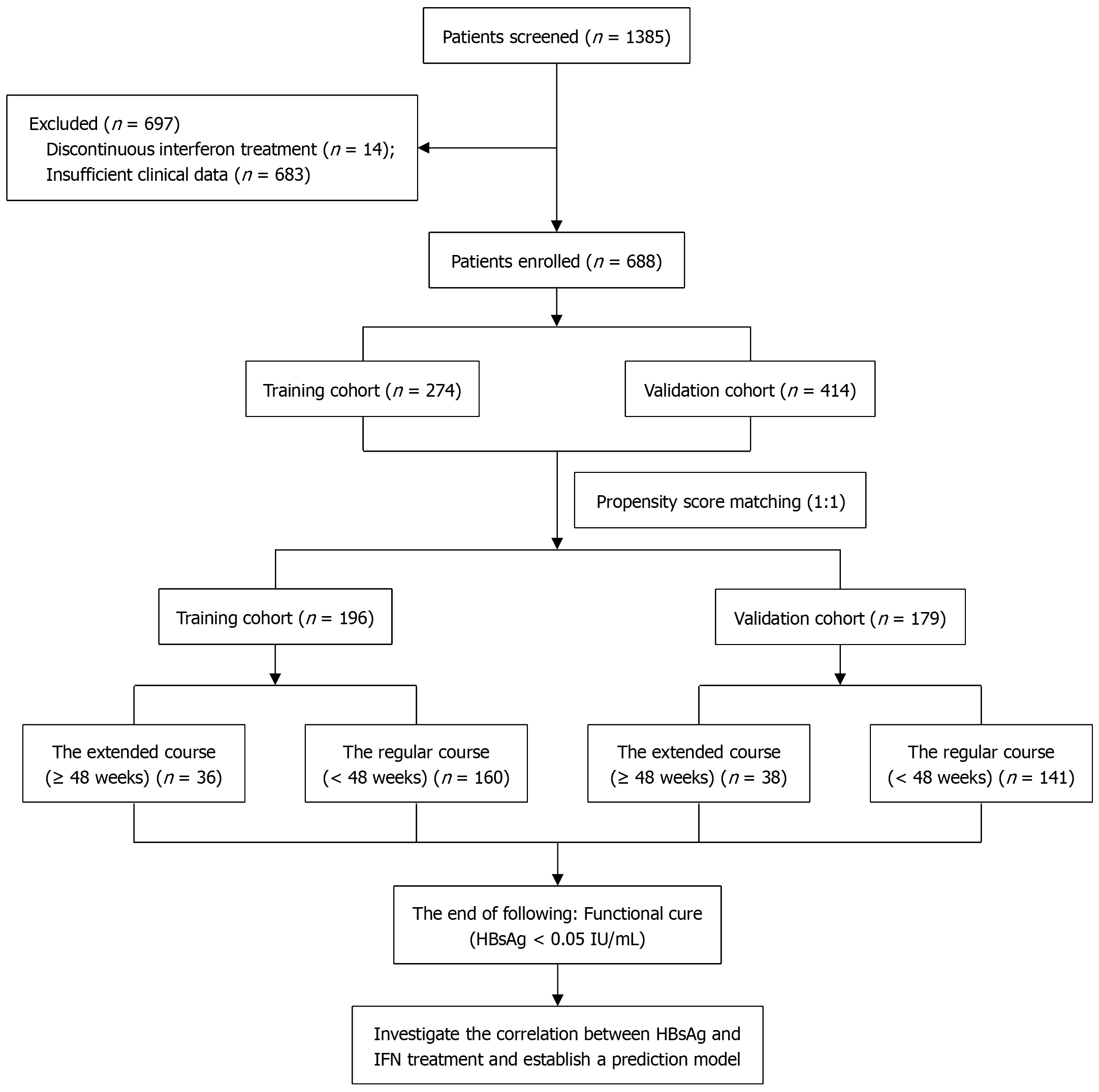
This study included a total of 1385 patients with CHB from multiple hospitals participating in the OASIS project, which encompassed locations such as Tianjin, Xiamen (Fujian Province), and Taiyuan (Shanxi Province). Of these, 697 patients were excluded: 14 due to treatment interruptions during interferon therapy and 683 due to insufficient clinical data. Ul
A total of 688 CHB patients who achieved HBsAg clearance were enrolled in this study. The mean age of the participants was 40.82 ± 9.19 years, with 421 (61.19%) being male. Among them, 91 (13.36%) were HBeAg positive, and 71 (10.68%) had compensated cirrhosis. Following PSM, 375 patients remained in the analysis. The baseline characteristics, including age, gender, HBeAg status, HBV DNA levels, and ALT levels, were balanced between the training cohort (n = 196) and the validation cohort (n = 179) (Table 1).
| Characteristic | Before PSM | After PSM | ||||||
| Overall (n = 688) | Training cohort (n = 274) | Validation cohort (n = 414) | P value | Overall (n = 375) | Training cohort (n = 196) | Validation cohort (n = 179) | P value | |
| Age (year) | 40.82 ± 9.19 | 41.38 ± 8.69 | 40.44 ± 9.49 | 0.189 | 40.91 ± 8.91 | 41.15 ± 7.88 | 40.65 ± 9.94 | 0.588 |
| Gender | 0.790 | 0.805 | ||||||
| Male | 421 (61.19) | 166 (60.58) | 255 (61.59) | 228 (60.80) | 118 (60.20) | 110 (61.45) | ||
| Female | 267 (38.81) | 108 (39.42) | 159 (38.41) | 147 (39.20) | 78 (39.80) | 69 (38.55) | ||
| Cirrhosis | 0.085 | 0.135 | ||||||
| (-) | 594 (89.32) | 238 (86.86) | 356 (91.05) | 333 (89.04) | 170 (86.73) | 163 (91.57) | ||
| (+) | 71 (10.68) | 36 (13.14) | 35 (8.95) | 41 (10.96) | 26 (13.27) | 15 (8.43) | ||
| IFN treatment course (week) | 0.673 | 0.988 | ||||||
| Median (Q1, Q3) | 24.36 (15.00, 40.71) | 24.00 (16.00, 42.00) | 25.00 (15.00, 39.71) | 24.14 (15.00, 40.00) | 24.00 (16.00, 39.25) | 24.86 (14.14, 40.71) | ||
| 32.0 ± 23.15 | 32.54 ± 23.68 | 31.77 ± 22.82 | 31.82 ± 22.35 | 31.83 ± 23.02 | 31.80 ± 21.67 | |||
| IFN group | 0.550 | 0.487 | ||||||
| < 48 | 555 (80.67) | 218 (79.56) | 337 (81.40) | 301 (80.27) | 160 (81.63) | 141 (78.77) | ||
| ≥ 48 | 133 (19.33) | 56 (20.44) | 77 (18.60) | 74 (19.73) | 36 (18.37) | 38 (21.23) | ||
| HBeAg | 0.001 | 0.417 | ||||||
| (-) | 590 (86.64) | 247 (91.82) | 343 (83.25) | 338 (90.13) | 179 (91.33) | 159 (88.83) | ||
| (+) | 91 (13.36) | 22 (8.18) | 69 (16.75) | 37 (9.87) | 17 (8.67) | 20 (11.17) | ||
| HBV DNA (log10 copies/mL) | 1.66 ± 1.80 | 1.39 ± 1.93 | 1.88 ± 1.66 | 0.001 | 1.78 ± 1.69 | 1.65 ± 1.85 | 1.92 ± 1.48 | 0.123 |
| HBsAg (IU/mL), median (Q1, Q3) | 44.25 (3.39, 334.78) | 37.08 (2.91, 401.76) | 52.35 (3.95, 314.81) | 0.755 | 49.54 (4.54, 330.75) | 47.80 (5.56, 469.24) | 51.78 (3.99, 235.90) | 0.943 |
| HBsAg (log10 IU/mL) | 1.53 ± 1.31 | 1.51 ± 1.27 | 1.55 ± 1.34 | 0.687 | 1.56 ± 1.29 | 1.61 ± 1.28 | 1.50 ± 1.30 | 0.412 |
| 12-week HBsAg (log10 IU/mL), median (Q1, Q3) | -0.30 (-1.68, 1.28) | -0.26 (-1.51, 1.37) | -0.30 (-1.96, 1.15) | 0.270 | -0.29 (-1.51, 1.23) | -0.24 (-1.51, 1.37) | -0.30 (-1.82, 0.72) | 0.246 |
| HBsAg decline at 12 weeks (log10 IU/mL), median (Q1, Q3) | 1.46 (0.51, 2.74) | 1.42 (0.51, 2.55) | 1.51 (0.53, 2.87) | 0.156 | 1.46 (0.60, 2.80) | 1.58 (0.59, 2.72) | 1.43 (0.62, 2.90) | 0.485 |
| ALT (U/L), median (Q1, Q3) | 24.00 (16.00, 41.00) | 23.30 (15.00, 36.00) | 24.55 (17.00, 42.85) | 0.236 | 24.00 (16.45, 39.00) | 24.00 (15.93, 37.38) | 23.00 (17.00, 40.50) | 0.298 |
| TBiL (μmol/L) | 16.19 ± 10.87 | 16.70 ± 10.35 | 15.87 ± 11.19 | 0.331 | 16.60 ± 11.46 | 16.79 ± 10.60 | 16.41 ± 12.35 | 0.749 |
| ALP (U/L) | 77.30 ± 31.11 | 72.88 ± 31.03 | 80.67 ± 30.78 | 0.002 | 76.25 ± 32.06 | 74.46 ± 33.45 | 78.21 ± 30.44 | 0.258 |
| GGT (U/L), median (Q1, Q3) | 21.00 (14.00, 34.00) | 20.00 (14.00, 30.00) | 22.00 (15.00, 35.00) | 0.807 | 21.00 (14.00, 34.50) | 21.50 (14.00, 32.25) | 21.00 (15.00, 36.00) | 0.557 |
| ALB | 0.067 | 0.486 | ||||||
| < 40 | 637 (95.50) | 255 (97.33) | 382 (94.32) | 359 (95.73) | 189 (96.43) | 170 (94.97) | ||
| ≥ 40 | 30 (4.50) | 7 (2.67) | 23 (5.68) | 16 (4.27) | 7 (3.57) | 9 (5.03) | ||
| WBC (109/L), median (Q1, Q3) | 5.39 (4.30, 6.55) | 5.51 (4.72, 6.66) | 5.28 (4.20, 6.52) | 0.155 | 5.43 (4.43, 6.50) | 5.51 (4.57, 6.66) | 5.30 (4.40, 6.47) | 0.730 |
| HB (g/L) | 146.18 ± 21.76 | 146.02 ± 25.34 | 146.28 ± 19.13 | 0.880 | 145.50 ± 23.51 | 146.28 ± 26.96 | 144.64 ± 19.07 | 0.502 |
| PLT (109/L) | 208.52 ± 67.10 | 222.42 ± 66.80 | 199.51 ± 65.83 | < 0.001 | 211.83 ± 60.98 | 215.57 ± 57.89 | 207.73 ± 64.10 | 0.214 |
The training cohort consisted of 196 participants, with a mean age of 41.15 ± 7.88 years. Among these, 118 (60.20%) were male, and 26 (13.27%) had cirrhosis. Among the 196 individuals with HBsAg clearance and clearly documented treat
| Characteristic | Overall (n = 196) | The regular course (< 48 weeks) (n = 160) | The extended course (≥ 48 weeks) (n = 36) | P value |
| Age (year) | 41.15 ± 7.88 | 41.69 ± 8.01 | 38.72 ± 6.82 | 0.041 |
| Gender | 0.800 | |||
| Male | 118 (60.20) | 97 (60.62) | 21 (58.33) | |
| Female | 78 (39.80) | 63 (39.38) | 15 (41.67) | |
| Treatment regimen | 0.420 | |||
| IFN monotherapy | 99 (50.51) | 83 (51.88) | 16 (44.44) | |
| Combined with ETV | 97 (49.49) | 77 (48.12) | 20 (55.56) | |
| Cirrhosis | 0.004 | |||
| (-) | 170 (86.73) | 144 (90.00) | 26 (72.22) | |
| (+) | 26 (13.27) | 16 (10.00) | 10 (27.78) | |
| HBeAg | 0.059 | |||
| (-) | 179 (91.33) | 149 (93.12) | 30 (83.33) | |
| (+) | 17 (8.67) | 11 (6.88) | 6 (16.67) | |
| HBV DNA (log10 copies/mL) | 1.65 ± 1.85 | 1.63 ± 1.72 | 1.74 ± 2.39 | 0.765 |
| HBsAg (IU/mL), median (Q1, Q3) | 47.80 (5.56, 469.24) | 27.05 (2.50, 230.15) | 60.19 (60.39, 1878.06) | 0.002 |
| HBsAg (log10 IU/mL) | 1.61 ± 1.28 | 1.35 ± 1.20 | 2.75 ± 0.97 | < 0.001 |
| 12-week HBsAg (log10 IU/mL), median (Q1, Q3) | -0.24 (-1.51, 1.37) | -0.47 (-1.68, 0.83) | 1.94 (0.36, 2.75) | < 0.001 |
| HBsAg decline at 12 weeks (log10 IU/mL), median (Q1, Q3) | 1.58 (0.59, 2.72) | 1.68 (0.72, 2.72) | 0.75 (0.16, 2.60) | 0.240 |
| ALT (U/L), median (Q1, Q3) | 24.00 (15.93 37.38) | 24.00 (15.38, 35.60) | 25.50 (20.35, 44.58) | 0.681 |
| TBiL (μmol/L) | 16.79 ± 10.60 | 17.02 ± 11.25 | 15.75 ± 7.09 | 0.518 |
| ALP (U/L) | 74.46 ± 33.45 | 75.22 ± 35.56 | 71.08 ± 21.78 | 0.504 |
| GGT (U/L), median (Q1, Q3) | 21.50 (14.00, 32.25) | 21.50 (14.00, 33.25) | 21.50 (14.75, 28.25) | 0.682 |
| ALB | 0.776 | |||
| < 40 | 189 (96.43) | 154 (96.25) | 35 (97.22) | |
| ≥ 40 | 7 (3.57) | 6 (3.75) | 1 (2.78) | |
| WBC (109/L), median (Q1, Q3) | 5.51 (4.57, 6.66) | 5.46 (4.36, 6.66) | 5.63 (4.94, 6.48) | 0.918 |
| HB (g/L) | 146.28 ± 26.96 | 146.54 ± 26.68 | 145.09 ± 28.53 | 0.771 |
| PLT (109/L) | 215.57 ± 57.89 | 216.75 ± 60.08 | 210.33 ± 47.33 | 0.549 |
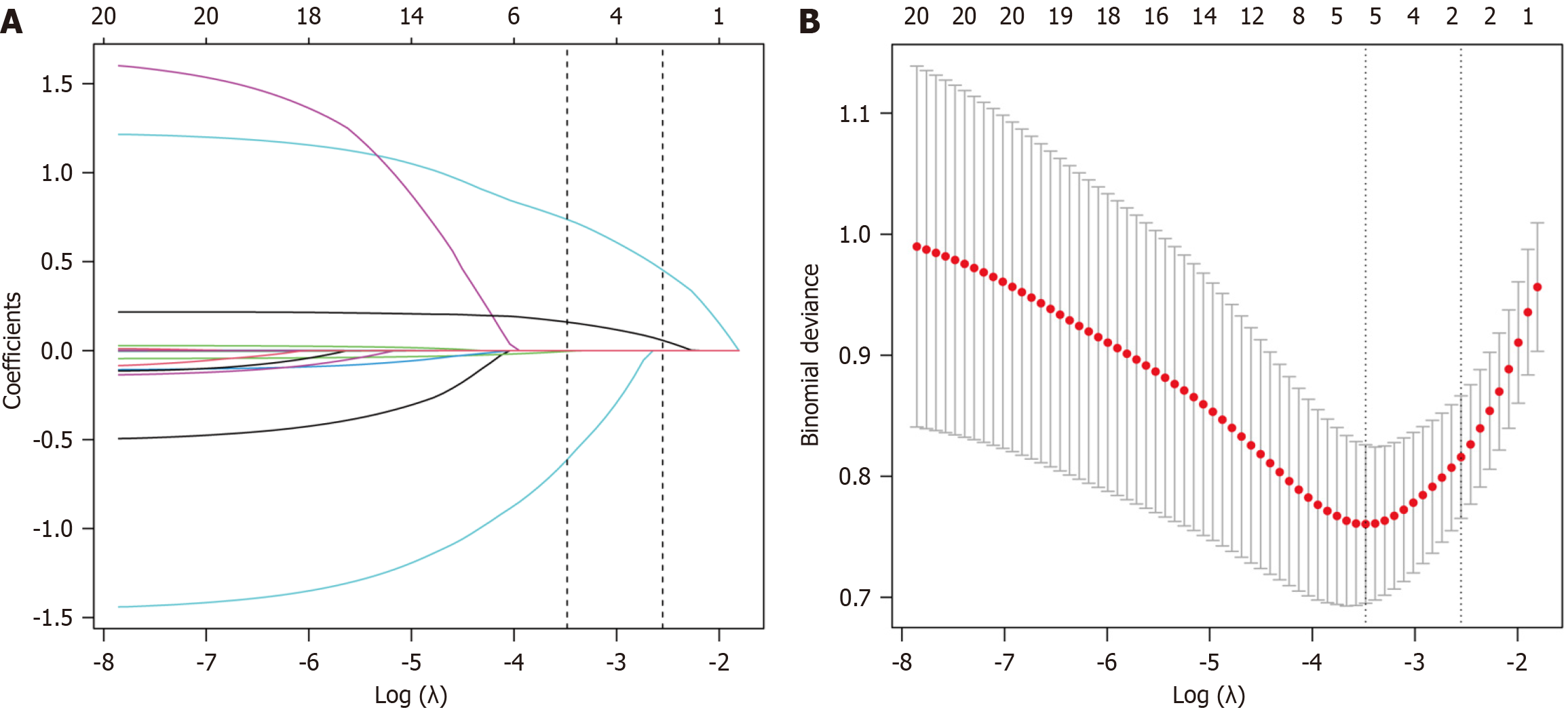
To prevent overfitting, we employed LASSO regression for parameter selection during model construction. The optimal λ value was determined through 10-fold cross-validation, aiming to minimize the cross-validation error using the 1-SE criterion. We found that at λ = 0.0308 (Log λ = -3.4816), LASSO regression identified four significant variables: Age, baseline HBsAg, 12-week HBsAg, and cirrhosis. Multifactorial logistic regression analysis revealed that an elevated baseline HBsAg level [odds ratio (OR) = 2.66, 95% confidence interval (CI): 1.56-4.53] and the presence of cirrhosis (OR = 3.48, 95%CI: 1.10-10.99) were independent risk factors for requiring the extended course of interferon therapy (both P < 0.05). Specifically, for every 1-log unit increase in baseline HBsAg, the odds of necessitating an extended course of interferon therapy increased by 1.66-fold (OR = 2.66, P < 0.05). Furthermore, patients with baseline cirrhosis were 3.48 times more likely to require the extended course of interferon therapy compared to those without cirrhosis (OR = 3.48, P < 0.05) (Figure 2 and Table 3).
| Characteristic | β | SE | Z value | P value | OR (95%CI) |
| Intercept | -2.04 | 1.36 | -1.50 | 0.133 | 0.13 (0.01-1.87) |
| Age (years) | -0.05 | 0.03 | -1.50 | 0.133 | 0.95 (0.90-1.01) |
| Baseline HBsAg (log10 IU/mL) | 0.98 | 0.27 | 3.58 | < 0.001 | 2.66 (1.56-4.53) |
| 12-week HBsAg (log10 IU/mL) | 0.26 | 0.14 | 1.91 | 0.056 | 1.30 (0.99-1.70) |
| Cirrhosis | |||||
| (-) | 1.00 | ||||
| (+) | 1.25 | 0.59 | 2.13 | 0.033 | 3.48 (1.10-10.99) |
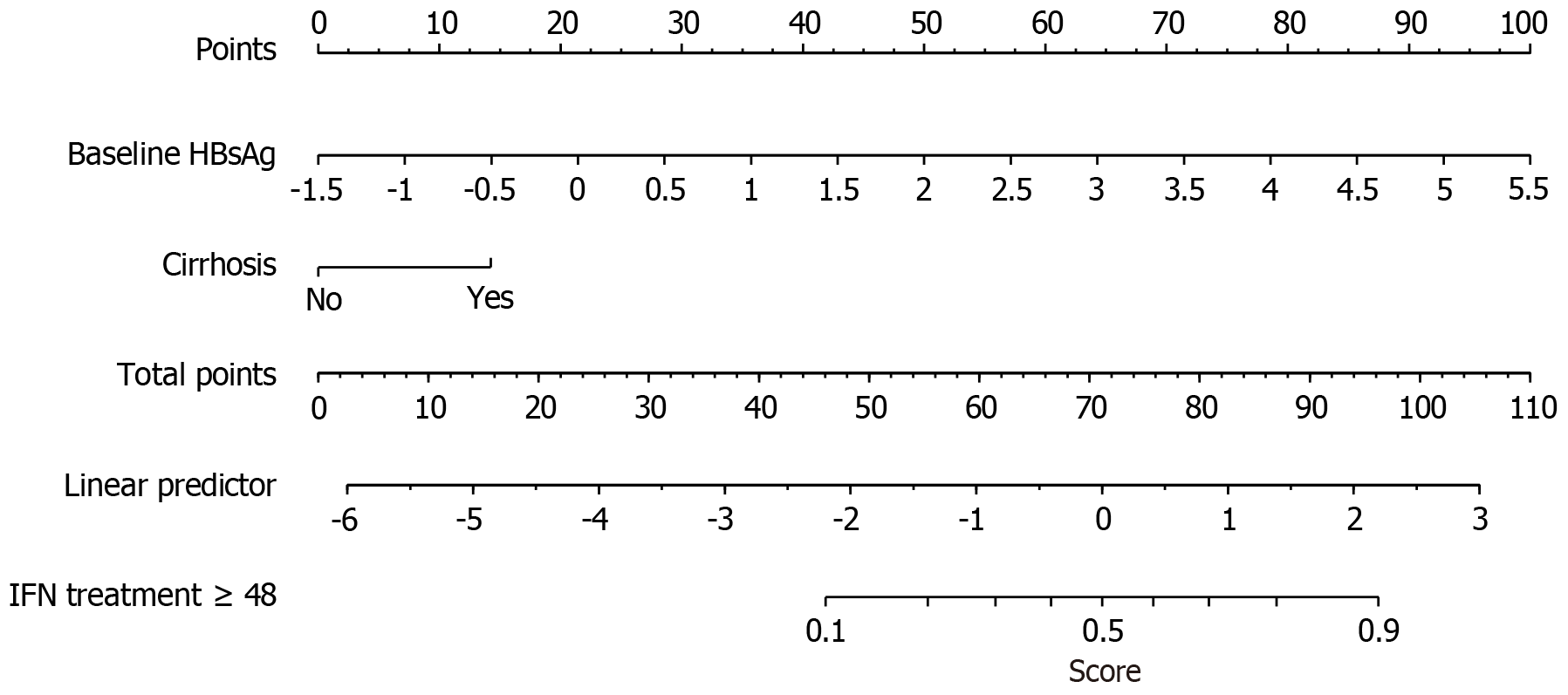
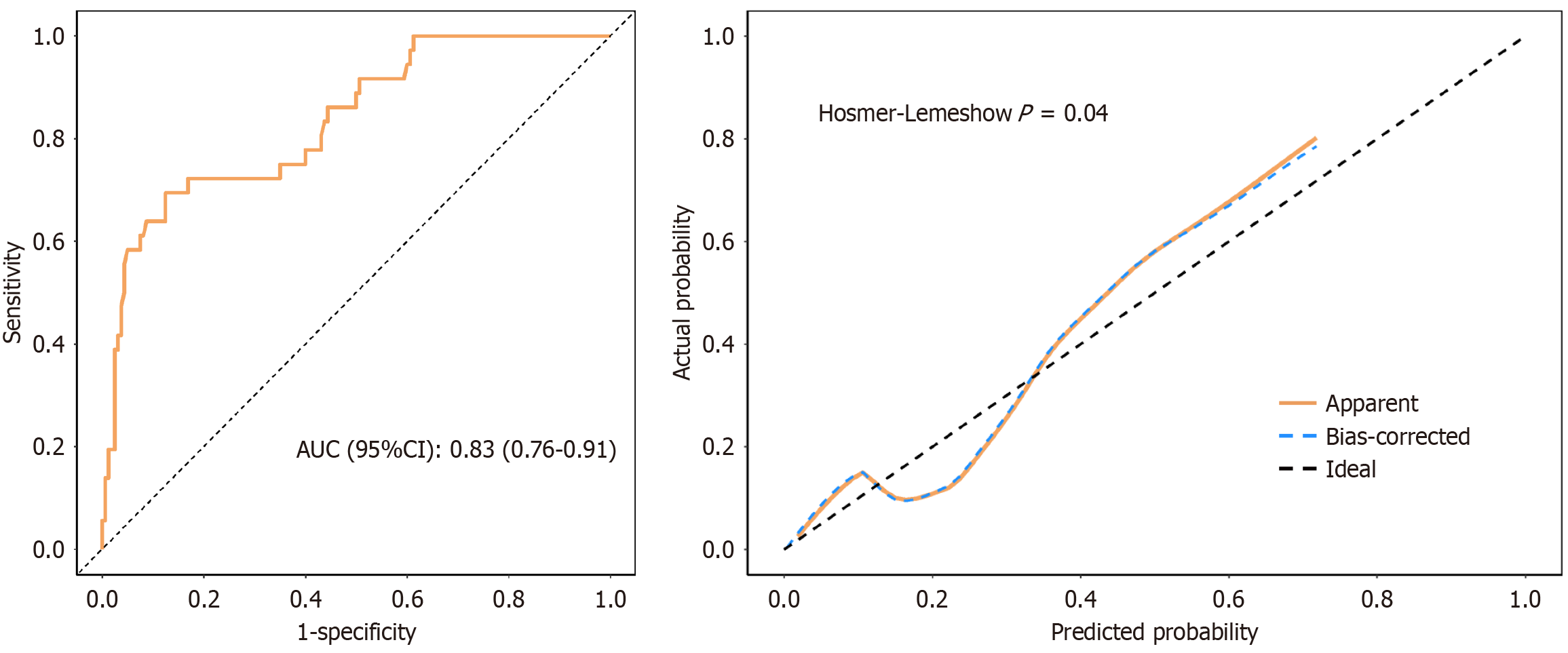
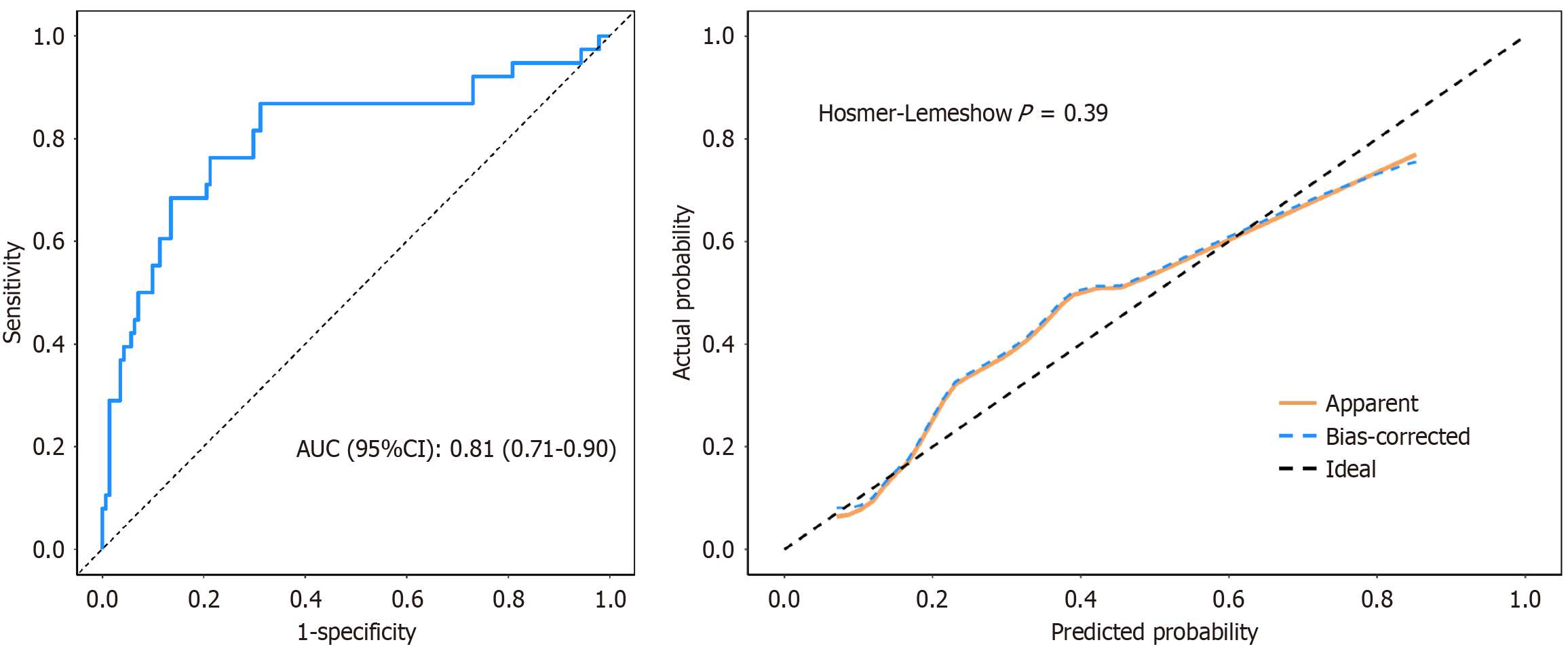
Based on the results of the LASSO-logistic regression analysis, a prediction model for an extended course of interferon therapy (≥ 48 weeks) was developed, incorporating baseline HBsAg levels and the presence of cirrhosis. The model is represented as log (P) = -4.354 + 1.251 × baseline HBSAGLOG + 1.247 × CIRRHOSIS (CIRRHOSIS = 1, 0). In the training cohort, the AUC was 0.83 (95%CI: 0.76-0.91), with a sensitivity of 0.88 and specificity of 0.69. Bootstrap internal validation (conducted 500 times) yielded an AUC of 0.82. The Hosmer-Lemeshow goodness-of-fit test was employed to assess the calibration ability of this prediction model. The result for the training cohort was χ2 = 16.30, P = 0.04 (Figures 3 and 4). The AUC for the external validation cohort was 0.81 (95%CI: 0.71-0.90), and the goodness-of-fit test resulted in χ2 = 8.41 and P = 0.39, indicating that the difference between the predicted and actual values of the predictive model was not statistically significant (P > 0.05). This finding suggests that the calibration of the predictive model was satisfactory (Figure 5).
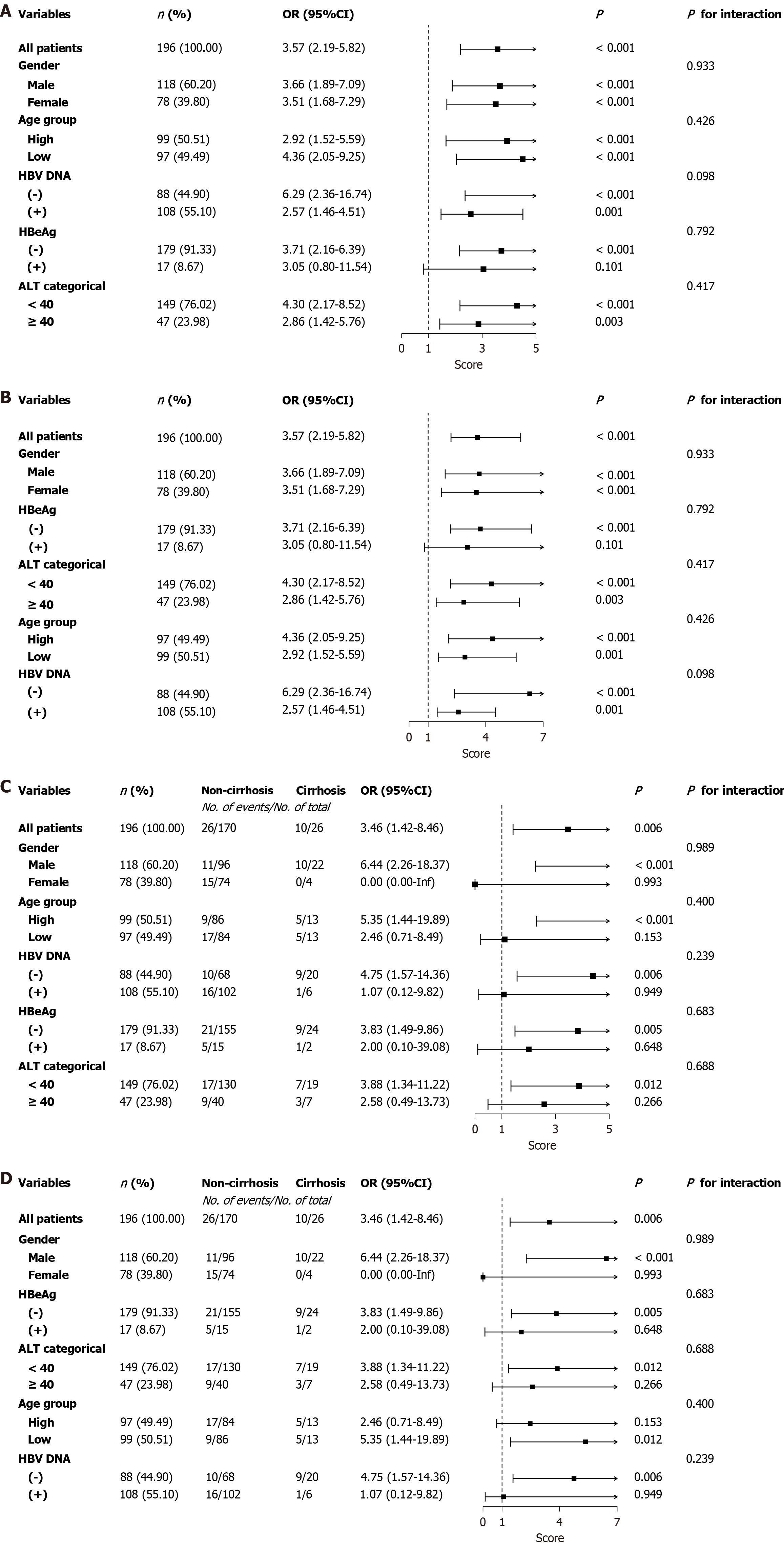
Subgroup analyses of the training and validation cohorts were conducted to evaluate the consistency of the associations between extended course interferon (≥ 48 weeks) and baseline characteristics, including cirrhosis, across different populations. This analysis examined interactions based on sex, age, baseline HBeAg, baseline HBV DNA, and baseline ALT levels. The results indicated that the relationships of both baseline HBsAg and liver cirrhosis with extended course interferon (≥ 48 weeks) were consistent with the primary outcome in most subgroups. Furthermore, no significant interactions were observed concerning the association with extended course interferon (≥ 48 weeks), suggesting that this correlation remains stable and independent of sex, age, baseline HBV DNA, and baseline ALT effects (P > 0.05) (Figure 6).
CHB is a serious liver disease that is widely prevalent worldwide, posing a major threat to human health. An estimated 73% of global deaths due to cancer are associated with HBV infection[4]. Studies have shown that patients who achieve HBsAg clearance have a significantly reduced risk of complications such as cirrhosis and hepatocellular carcinoma[14,15]. Currently, antiviral regimens based on Peg-IFNα remain a crucial strategy for achieving clinical cure. Research indicates that interferon therapy, which demonstrates higher HBsAg clearance rates compared to NAs[16-20], can lead to clinical cure in a greater number of CHB patients, resulting in reductions of 45% and 54% in the overall risk of adverse hepatic events and cirrhosis, respectively. In recent years, several large-scale prospective cohort studies conducted in China, including the Everest program, the OASIS project, and the Starlight Program, have led to the clinical cure of over 8000 patients, with an overall HBsAg clearance rate of 33.2% at 48 weeks. Consequently, several guidelines recommend a routine interferon treatment course of 48 weeks for adult CHB patients. However, optimizing the treatment course while balancing efficacy and tolerability remains a clinical challenge.
A study by Li et al[21] found that the rate of HBsAg clearance was 15.32% at 24 weeks following interferon therapy, which increased to 21.43% when the treatment duration was extended to 48 weeks. A prospective, controlled study reported that after 96 weeks of Peg-IFN-α treatment, HBsAg clearance rates rose to 47.06% and 31.34%[22-24]. Most do
Studies indicate that younger female patients with elevated ALT levels and lower HBsAg levels exhibit the most favo
The mechanisms by which cirrhosis affects interferon efficacy are intricate. Patients with cirrhosis frequently exhibit significant inflammation and fibrosis, which may alter the pharmacokinetics and pharmacodynamics of interferon[36,37]. Studies have demonstrated that the immune microenvironment is modified in cirrhotic patients, potentially diminishing the efficacy of interferon in activating the immune response or inhibiting the interferon signaling pathway[38,39]. A study by Buster et al[27] supports the notion that cirrhosis is an independent predictor of poor response to interferon therapy. In our present study, we observed a higher proportion of baseline cirrhosis in the extended course group compared to the regular course group (10.00% vs 27.78%, P < 0.05). The presence of cirrhosis at baseline independently influenced the need for extended course interferon therapy (≥ 48 weeks).
In addition to the aforementioned baseline characteristics, meta-analyses[40] have demonstrated that the 12-week HBsAg level possesses independent predictive value for IFN treatment. Notably, the 12-week HBsAg levels of certain patients were included in this study. Although the 12-week HBsAg level exhibited potential predictive value in LASSO regressions, it did not achieve statistical significance in the multifactorial model (P = 0.05). The observed statistical signi
Recently research indicate that interferon therapy is generally safe in patients with compensated cirrhosis[41,42]. The most frequently reported adverse events including fever (53/54, 98.15%), fatigue (52/54, 96.30%), weight loss (41/54, 75.93%), and alopecia (7/54, 12.96%) were comparable to those observed in CHB patients without cirrhosis, with very few cases progressing to decompensation. In our present study, 41 patients (10.96% of the cohort) had compensated cirrhosis. Treatment was well tolerated throughout, with only two cases of mild ascites reported; both resolved completely after temporary interferon interruption and symptomatic management. No episodes of severe hepatic failure, death, or hospitalization occurred. Nevertheless, given the distinct clinical profile of cirrhotic patients, we implemented intensified hematologic and hepatic function monitoring at baseline and during therapy for all interferon-treated cirrhotic individuals to facilitate early detection of significant adverse reactions and allow timely adjustment of treatment strategy.
This study’s innovation lies in its exploration of the factors influencing extended interferon therapy (≥ 48 weeks) through multicenter cohort data. For the first time, it incorporates baseline cirrhosis status and constructs a predictive model that integrates baseline HBsAg levels. This model may serve as a valuable reference for clinicians to identify high-risk patients who require extended interferon therapy early in the treatment process, allowing for the development of individualized regimens that consider the patient’s economic status and tolerability. However, as a retrospective study, certain limitations must be acknowledged: First, the small sample size may result in biased outcomes; Second, dynamic indicators such as HBsAg and ALT levels at 12 weeks and 24 weeks during treatment, as well as novel biomarkers (e.g., HBV RNA), were not included, potentially affecting the model’s comprehensiveness. Furthermore, previous studies have indicated that the treatment strategy (monotherapy vs combination) is one of the factors affecting interferon response kinetics[43,44]. It should be noted that the present analysis focused solely on the duration of interferon therapy among patients who achieved a cure. A comparative assessment of cure rates across different treatment strategies was not per
This study successfully constructed and validated a prediction model based on baseline HBsAg levels and cirrhosis status. This model can identify potential patients who may need to extend their interferon regimen (≥ 48 weeks) to achieve HBsAg clearance prior to treatment, thereby avoiding the premature termination of potentially effective therapies and providing an evidence-based foundation for individualized treatment planning.
We thank all those who have been involved in this study, including the patients, the investigators and colleagues from Tianjin University Central Hospital, The First Affiliated Hospital of Xiamen University, The School of Clinical Medicine of Fujian Medical University, The Third people’s Hospital of Taiyuan, National Medical Center for Infectious Diseases, Huashan Hospital, Henan Provincial People’s Hospital, The First Affiliated Hospital of Nanchang University, First People’s Hospital of Yunnan Province, Affiliated Hospital of Kunming University of Science and Technology, Yanan University Affiliated Hospital. Finally, we would like to express our gratitude once again to the China Reduction of Hepatocellular Carcinoma Incidence in Hepatitis B Patients Study project for the data support provided.
| 1. | CDA Foundation. Polaris Observatory. [cited January 14, 2026]. Available from: https://cdafound.org/polaris/. |
| 2. | Chen S, Li J, Wang D, Fung H, Wong LY, Zhao L. The hepatitis B epidemic in China should receive more attention. Lancet. 2018;391:1572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 3. | Hsu YC, Huang DQ, Nguyen MH. Global burden of hepatitis B virus: current status, missed opportunities and a call for action. Nat Rev Gastroenterol Hepatol. 2023;20:524-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 474] [Cited by in RCA: 411] [Article Influence: 137.0] [Reference Citation Analysis (0)] |
| 4. | Ott JJ, Stevens GA, Groeger J, Wiersma ST. Global epidemiology of hepatitis B virus infection: new estimates of age-specific HBsAg seroprevalence and endemicity. Vaccine. 2012;30:2212-2219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1441] [Cited by in RCA: 1311] [Article Influence: 93.6] [Reference Citation Analysis (1)] |
| 5. | GBD 2019 Hepatitis B Collaborators. Global, regional, and national burden of hepatitis B, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Gastroenterol Hepatol. 2022;7:796-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 605] [Cited by in RCA: 558] [Article Influence: 139.5] [Reference Citation Analysis (2)] |
| 6. | Jeng WJ, Lok ASF. What will it take to cure hepatitis B? Hepatol Commun. 2023;7:e0084. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 7. | Chinese Society of Hepatology; Chinese Medical Association; Chinese Society of Infectious Diseases, Chinese Medical Association. [Guidelines for the prevention and treatment of chronic hepatitis B (version 2022)]. Zhonghua Gan Zang Bing Za Zhi. 2022;30:1309-1331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 42] [Reference Citation Analysis (1)] |
| 8. | European Association for the Study of the Liver. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2017;67:370-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4470] [Cited by in RCA: 4069] [Article Influence: 452.1] [Reference Citation Analysis (1)] |
| 9. | Terrault NA, Lok ASF, McMahon BJ, Chang KM, Hwang JP, Jonas MM, Brown RS Jr, Bzowej NH, Wong JB. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology. 2018;67:1560-1599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3558] [Cited by in RCA: 3177] [Article Influence: 397.1] [Reference Citation Analysis (2)] |
| 10. | Chinese Society of Infectious Disease Chinese Society of Hepatology; Chinese Medical Association. [The expert consensus on clinical cure (functional cure) of chronic hepatitis B]. Zhonghua Gan Zang Bing Za Zhi. 2019;27:594-603. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (2)] |
| 11. | Wang Z, Sun L, Wu Y, Xia Q. Extended duration versus standard duration of peginterferon alfa-2a in treatment of chronic hepatitis B: A systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2016;40:195-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | Ye YM, Lin Y, Sun F, Yang WY, Zhou L, Lin C, Pan C. A predictive model for functional cure in chronic HBV patients treated with pegylated interferon alpha: a comparative study of multiple algorithms based on clinical data. Virol J. 2024;21:333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Zhong W, Wang C, Wang J, Chen T. Machine learning models to further identify advantaged populations that can achieve functional cure of chronic hepatitis B virus infection after receiving Peg-IFN alpha treatment. Int J Med Inform. 2025;193:105660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 14. | Cornberg M, Lok AS, Terrault NA, Zoulim F; 2019 EASL-AASLD HBV Treatment Endpoints Conference Faculty. Guidance for design and endpoints of clinical trials in chronic hepatitis B - Report from the 2019 EASL-AASLD HBV Treatment Endpoints Conference(‡). J Hepatol. 2020;72:539-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 268] [Article Influence: 44.7] [Reference Citation Analysis (1)] |
| 15. | Fung S, Choi HSJ, Gehring A, Janssen HLA. Getting to HBV cure: The promising paths forward. Hepatology. 2022;76:233-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 119] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 16. | Tang LSY, Covert E, Wilson E, Kottilil S. Chronic Hepatitis B Infection: A Review. JAMA. 2018;319:1802-1813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 582] [Cited by in RCA: 527] [Article Influence: 65.9] [Reference Citation Analysis (0)] |
| 17. | Ye J, Chen J. Interferon and Hepatitis B: Current and Future Perspectives. Front Immunol. 2021;12:733364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 134] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 18. | Wu F, Liu C, He L, Wang Y, Zhang X, Li M, Lu R, Kang P, Li M, Li Y, Jia X, Dang S. Peripheral Blood CD4(+)/CD8(+) T Cell Ratio Predicts HBsAg Clearance in Inactive HBsAg Carriers Treated with Peginterferon Alpha. J Clin Transl Hepatol. 2025;13:130-142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (2)] |
| 19. | Wong GL, Yiu KK, Wong VW, Tsoi KK, Chan HL. Meta-analysis: reduction in hepatic events following interferon-alfa therapy of chronic hepatitis B. Aliment Pharmacol Ther. 2010;32:1059-1068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 20. | Liang KH, Hsu CW, Chang ML, Chen YC, Lai MW, Yeh CT. Peginterferon Is Superior to Nucleos(t)ide Analogues for Prevention of Hepatocellular Carcinoma in Chronic Hepatitis B. J Infect Dis. 2016;213:966-974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 72] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 21. | Li Y, XU J, Wang YJ, Ding Q. [Retrospective Analysis of Predictors of HBsAg Clearance in the Treatment of Chronic Hepatitis B with Pegylated Interferon Alpha]. Yixue Yanjiu Zazhi. 2022;51:109-113. [DOI] [Full Text] |
| 22. | Jeong J, Shin JW, Jung SW, Park EJ, Park NH. Tenofovir alafenamide treatment may not worsen the lipid profile of chronic hepatitis B patients: A propensity score-matched analysis. Clin Mol Hepatol. 2022;28:254-264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 23. | Mak LY, Hui RW, Fung J, Liu F, Wong DK, Li B, Cheung KS, Yuen MF, Seto WK. Reduced hepatic steatosis is associated with higher risk of hepatocellular carcinoma in chronic hepatitis B infection. Hepatol Int. 2021;15:901-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 24. | Cao Z, Liu Y, Ma L, Lu J, Jin Y, Ren S, He Z, Shen C, Chen X. A potent hepatitis B surface antigen response in subjects with inactive hepatitis B surface antigen carrier treated with pegylated-interferon alpha. Hepatology. 2017;66:1058-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 100] [Article Influence: 11.1] [Reference Citation Analysis (1)] |
| 25. | Yan YJ, Wang XX, Cao ZH, Lu JF, Jin Y, He ZM, Geng N, Ren S, Ma LN, Chen XY. [Low-levels of HBsAg quantification at 48-week in HBeAg-negative chronic hepatitis B patients are the advantageous population for HBsAg clearance]. Zhonghua Gan Zang Bing Za Zhi. 2018;26:813-818. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 26. | Jiang S, Guo S, Huang Y, Yin Y, Feng J, Zhou H, Guo Q, Wang W, Xin H, Xie Q. Predictors of HBsAg seroclearance in patients with chronic HBV infection treated with pegylated interferon-α: a systematic review and meta-analysis. Hepatol Int. 2024;18:892-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (3)] |
| 27. | Buster EH, Hansen BE, Lau GK, Piratvisuth T, Zeuzem S, Steyerberg EW, Janssen HL. Factors that predict response of patients with hepatitis B e antigen-positive chronic hepatitis B to peginterferon-alfa. Gastroenterology. 2009;137:2002-2009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 329] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 28. | Li K, Ning HB, Jin HM, Peng Z, Sang J. [Effect of pegylated interferona-2b on serum HBsAg clearance rate in treatment of patients with chronic hepatitis B]. Linchuang Gandanbing Zazhi. 2023;39:1819-1824. [DOI] [Full Text] |
| 29. | Hu P, Shang J, Zhang WH, Gong GZ, Li YG, Chen XY, Jiang JN, Xie Q, Dou XG, Sun YT, Li YF, Liu YX, Liu GZ, Ma DW, Chi XL, Tang H, Li XO, Xie Y, Chen XP, Jiang JJ, Zha P, Hou JL, Gao ZL, Fan HM, Ding JG, Zhang DZ, Ren H. [HBsAg loss with Pegylated-interferon alfa-2a in hepatitis B patients with partial response to nucleos(t)-ide analog: new switch study]. Zhonghua Gan Zang Bing Za Zhi. 2018;26:756-764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 30. | Ning Q, Han M, Sun Y, Jiang J, Tan D, Hou J, Tang H, Sheng J, Zhao M. Switching from entecavir to PegIFN alfa-2a in patients with HBeAg-positive chronic hepatitis B: a randomised open-label trial (OSST trial). J Hepatol. 2014;61:777-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 223] [Article Influence: 18.6] [Reference Citation Analysis (1)] |
| 31. | Li WY, Fan ZQ, Wu JG, Wang J. [Analysis of virological characteristics of peginterferona-2b in the treatment of advantaged patients with chronic hepatitis]. Linchuang Yixue Yanjiu Yu Shijian. 2023;8:37-40. [DOI] [Full Text] |
| 32. | Zhang PX, Tang QQ, Zhu J, Deng WY, Zhang ZH. Predictive models for functional cure in patients with CHB receiving PEG-IFN therapy based on HBsAg quantification through meta-analysis. Hepatol Int. 2024;18:1110-1121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 22] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 33. | Wang L, Cao X, Wang Z, Gao Y, Deng J, Liu X, Zhuang H. Correlation of HBcrAg with Intrahepatic Hepatitis B Virus Total DNA and Covalently Closed Circular DNA in HBeAg-Positive Chronic Hepatitis B Patients. J Clin Microbiol. 2019;57:e01303-e01318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 34. | Mahajan A, Kharawala S, Desai S, Kendrick S, Das J, Gielen V. Association of Hepatitis B Surface Antigen Levels With Long-Term Complications in Chronic Hepatitis B Virus Infection: A Systematic Literature Review. J Viral Hepat. 2024;31:746-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 35. | Yan F, Tang F, Chen J, Lin Y, Chen X, Du Q, Yin W, Liang J, Liu L, Wang F, Xu B, Ye Q, Xiang H. Exploring using HBsAg to predict interferon treatment course to achieve clinical cure in chronic hepatitis B patients: a clinical study. Front Immunol. 2024;15:1528758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 36. | Li MH, Zhang L, Qu XJ, Lu Y, Shen G, Wu SL, Chang M, Liu RY, Hu LP, Li ZZ, Hua WH, Song SJ, Xie Y. Kinetics of Hepatitis B Surface Antigen Level in Chronic Hepatitis B Patients who Achieved Hepatitis B Surface Antigen Loss during Pegylated Interferon Alpha-2a Treatment. Chin Med J (Engl). 2017;130:559-565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 37. | Zeuzem S, Welsch C, Herrmann E. Pharmacokinetics of peginterferons. Semin Liver Dis. 2003;23 Suppl 1:23-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 65] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 38. | Li Q, Sun B, Zhuo Y, Jiang Z, Li R, Lin C, Jin Y, Gao Y, Wang D. Interferon and interferon-stimulated genes in HBV treatment. Front Immunol. 2022;13:1034968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 39. | Parola M, Pinzani M. Liver fibrosis: Pathophysiology, pathogenetic targets and clinical issues. Mol Aspects Med. 2019;65:37-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 998] [Cited by in RCA: 897] [Article Influence: 128.1] [Reference Citation Analysis (2)] |
| 40. | Yeo YH, Ho HJ, Yang HI, Tseng TC, Hosaka T, Trinh HN, Kwak MS, Park YM, Fung JYY, Buti M, Rodríguez M, Treeprasertsuk S, Preda CM, Ungtrakul T, Charatcharoenwitthaya P, Li X, Li J, Zhang J, Le MH, Wei B, Zou B, Le A, Jeong D, Chien N, Kam L, Lee CC, Riveiro-Barciela M, Istratescu D, Sriprayoon T, Chong Y, Tanwandee T, Kobayashi M, Suzuki F, Yuen MF, Lee HS, Kao JH, Lok AS, Wu CY, Nguyen MH. Factors Associated With Rates of HBsAg Seroclearance in Adults With Chronic HBV Infection: A Systematic Review and Meta-analysis. Gastroenterology. 2019;156:635-646.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 213] [Article Influence: 30.4] [Reference Citation Analysis (1)] |
| 41. | Wang Z, Wang X, Zhou L, Shi S, Hua Y, Feng Y. Safety and efficacy of 48-week pegylated interferon-α-2b therapy in patients with hepatitis B virus-related compensated liver cirrhosis: a pilot observational study. Front Med (Lausanne). 2024;11:1489671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 42. | Mira JA, García-Rey S, Rivero A, de los Santos-Gil I, López-Cortés LF, Girón-González JA, Téllez F, Márquez M, Merino D, Ríos-Villegas MJ, Macías J, Rivero-Juárez A, Pineda JA. Response to pegylated interferon plus ribavirin among HIV/hepatitis C virus-coinfected patients with compensated liver cirrhosis. Clin Infect Dis. 2012;55:1719-1726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 43. | Wen C, Wang Y, Tian H, Lei Y, Wang Z, Cai D, Zhou Z, Shi X. Clinical cure induced by pegylated interferon α-2b in the advantaged population of chronic hepatitis B virus infection: a retrospective cohort study. Front Cell Infect Microbiol. 2023;13:1332232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 44. | Zheng C, Yan H, Zeng J, Cai S, Wu X. Comparison of pegylated interferon monotherapy and de novo pegylated interferon plus tenofovir combination therapy in patients with chronic hepatitis B. Infect Drug Resist. 2019;12:845-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |