Published online Jun 7, 2025. doi: 10.3748/wjg.v31.i21.106939
Revised: April 23, 2025
Accepted: May 23, 2025
Published online: June 7, 2025
Processing time: 87 Days and 9.3 Hours
Patients with microsatellite stable (MSS) metastatic colorectal cancer (mCRC) typically exhibit an immunosuppressive tumor microenvironment and demon
To evaluate the effectiveness and safety of chemotherapy combined with beva
A retrospective analysis was conducted on patients with MSS mCRC diagnosed at Peking University First Hospital and Jilin Cancer Hospital from January 2020 to December 2024. The patients were divided into two groups: The experimental group receiving second-line chemotherapy combined with bevacizumab and anti-PD-1 immunotherapy, and the control group receiving chemotherapy combined with bevacizumab. Propensity score matching was applied to balance potential prognostic factors, including age, gender, Eastern Cooperative Oncology Group score, number of metastases, and primary tumor site. The progression-free sur
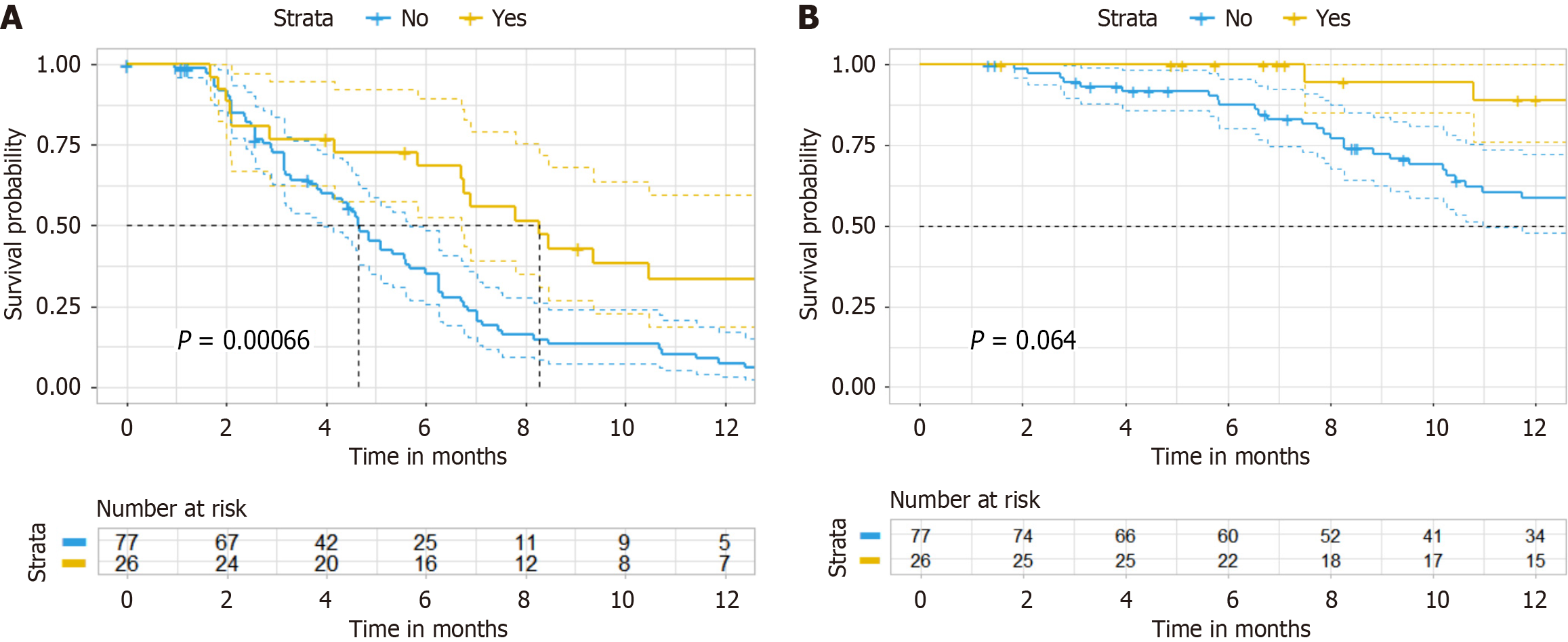
Propensity score matching resulted in 103 matched eligible patients. The median follow-up period was 13.9 months in the matched cohort. The objective response rate was 11.5% and 9% for the experimental and control groups, respectively (P = 0.710), while the disease control rate was 76.9% and 53.2%, respectively (P = 0.058). The median progression-free survival in the experimental group was 8.27 months [95% confidence interval (CI): 6.7-14.7 months], significantly higher than that in the control group, which was 4.63 months (95%CI: 3.9-5.67 months) (hazard ratio = 0.4143, 95%CI: 0.2462-0.6972, P = 0.00066). There was a trend towards the higher median overall survival in the experimental group compared to the control group (hazard ratio = 0.4504, 95%CI: 0.1897-1.07, P = 0.064). The incidences of adverse events were similar between the two groups.
Compared with the standard second-line chemotherapy combined with bevacizumab regimen, second-line therapy that combines chemotherapy with bevacizumab and anti-PD-1 immunotherapy has demonstrated promising efficacy in the treatment of MSS mCRC, while exhibiting a similar safety profile.
Core Tip: This manuscript addresses to evaluate the effectiveness and safety of chemotherapy combined with bevacizumab with or without anti-programmed death 1 immunotherapy as the second-line regimen for microsatellite stable metastatic colorectal cancer. As there is currently no clinical data on the second-line treatment of advanced colorectal cancer with the combination of immunotherapy, anti-angiogenic drugs, and anti-programmed death 1 immunotherapy, we conducted a multicenter retrospective cohort clinical study to explore the safety and efficacy of this triplet therapy in second-line treat
- Citation: Gao Z, Wang XY, Shen ZG, Liu JH, Wang XY, Wu SK, Jin X. Chemotherapy plus bevacizumab with or without anti-programmed death 1 immunotherapy as the second-line therapy in colorectal cancer. World J Gastroenterol 2025; 31(21): 106939
- URL: https://www.wjgnet.com/1007-9327/full/v31/i21/106939.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i21.106939
The latest statistical data show that both the incidence and mortality rates of colorectal cancer are on the rise. In 2022, colorectal cancer ranked third in terms of new cases and second in terms of deaths among all malignant tumors worldwide[1]. For metastatic colorectal cancer (mCRC), the standard first-line recommended regimen currently involves chemotherapy combined with targeted therapy, which can improve tumor control rates in advanced patients to some extent. The first- and second-line chemotherapy options mainly involve sequencing folinic acid, 5-fluorouracil, oxaliplatin (FOLFOX) with folinic acid, 5-fluorouracil, irinotecan (FOLFIRI) or vice versa, with no impact on treatment efficacy[2]. In terms of targeted drug selection, for patients with wild-type RAS/BRAF genes, bevacizumab is the standard choice after cetuximab resistance; however, for those with primary resistance to bevacizumab in the first line, current clinical studies suggest that switching to cetuximab does not provide additional benefit compared to continuing bevacizumab[3,4]. Therefore, for second-line treatment of mCRC, regardless of gene status and primary tumor location, bevacizumab combined with second-line chemotherapy is the standard treatment option recommended by current guidelines[5]. Nevertheless, after first-line treatment failure, the overall response rates (ORR) of second-line chemotherapy combined with bevacizumab (doublet regimen) was only 5%-36%, with the median progression-free survival (PFS) of just 4-7 months[6-12]. Therefore, improving the efficacy of second-line treatment for mCRC is a major challenge in current clinical practice.
Immune checkpoint inhibitors have made significant breakthroughs in the treatment of multiple tumors, particularly in microsatellite instability-high mCRC, where anti-programmed death 1 (PD-1) immunotherapy has demonstrated outstanding efficacy[13,14]. However, this patient population accounts for less than 5% of all mCRC cases. For the majority of patients with microsatellite stable (MSS) mCRC, immunotherapy is largely ineffective, limiting their options for chemotherapy and targeted therapy. Therefore, new combination therapy regimens are needed to improve the response to immunotherapy for this subtype. Previous studies have shown that anti-angiogenic therapy can reverse the immunosuppressive tumor microenvironment by normalizing blood vessels and inducing T-cell infiltration and activation[15]. The phase II CheckMate 9X8 study compared the efficacy of nivolumab combined with modified FOLFOX6 (mFOLFOX6) (plus bevacizumab vs standard treatment (mFOLFOX6 plus bevacizumab) in first-line treatment of mCRC patients[16]. Subgroup analysis revealed that patients with consensus molecular subtype (CMS)3[17] could benefit from the addition of nivolumab to mFOLFOX6 plus bevacizumab. The BBCAPX study demonstrated that sinti
Based on these findings, chemotherapy combined with bevacizumab and anti-PD-1 immunotherapy (triplet regimen) has a solid theoretical foundation and has shown preliminary efficacy and safety in MSS mCRC patients. Therefore, the optimal combination of these three therapeutic agents - chemotherapy, anti-angiogenic drugs, and anti-PD-1 immunotherapy - should be an important research direction for changing the current treatment landscape of mCRC. As there is currently no clinical data on the second-line treatment of advanced colorectal cancer with the combination of che
This study employed a multicenter retrospective cohort research design. Patients with advanced colorectal cancer who were treated at Peking University First Hospital and Jilin Cancer Hospital between January 1, 2020 and December 30, 2024 were enrolled. The experimental group consisted of patients who received second-line treatment with chemotherapy combined with bevacizumab and anti-PD-1 immunotherapy, while the control group comprised patients who received conventional treatment (chemotherapy combined with bevacizumab).
The inclusion criteria were: (1) Histologically or cytologically confirmed unresectable mCRC (stage IV according to the American Joint Committee on Cancer Staging Manual 8th edition) with measurable lesions based on the Response Evaluation Criteria In Solid Tumors (RECIST) 1.1 criteria; (2) Progression after prior first-line standard two-drug che
The exclusion criteria were: (1) Patients with deficient DNA mismatch repair or microsatellite instability-high, or BRAF mutations; (2) Presence of symptomatic brain metastases; (3) Uncontrolled active infection; (4) Dysphagia, intractable vomiting, or known drug absorption disorders; and (5) Patients with symptomatic or high-risk obstruction, bleeding, or perforation, or those who have undergone intestinal stent placement to relieve intestinal obstruction.
In most cases, the chemotherapy regimen comprised an oxaliplatin-based doublet (FOLFOX, CAPEOX, or raltitrexed and oxaliplatin) or a topoisomerase inhibitor-based (FOLFIRI or CAPEOX: Capecitabine and irinotecan (IRI), or raltitrexed and IRI). anti-PD-1 immunotherapy included penpulimab, pembrolizumab, sintilimab, tislelizumab, and toripalimab. The anti-angiogenic agent was bevacizumab.
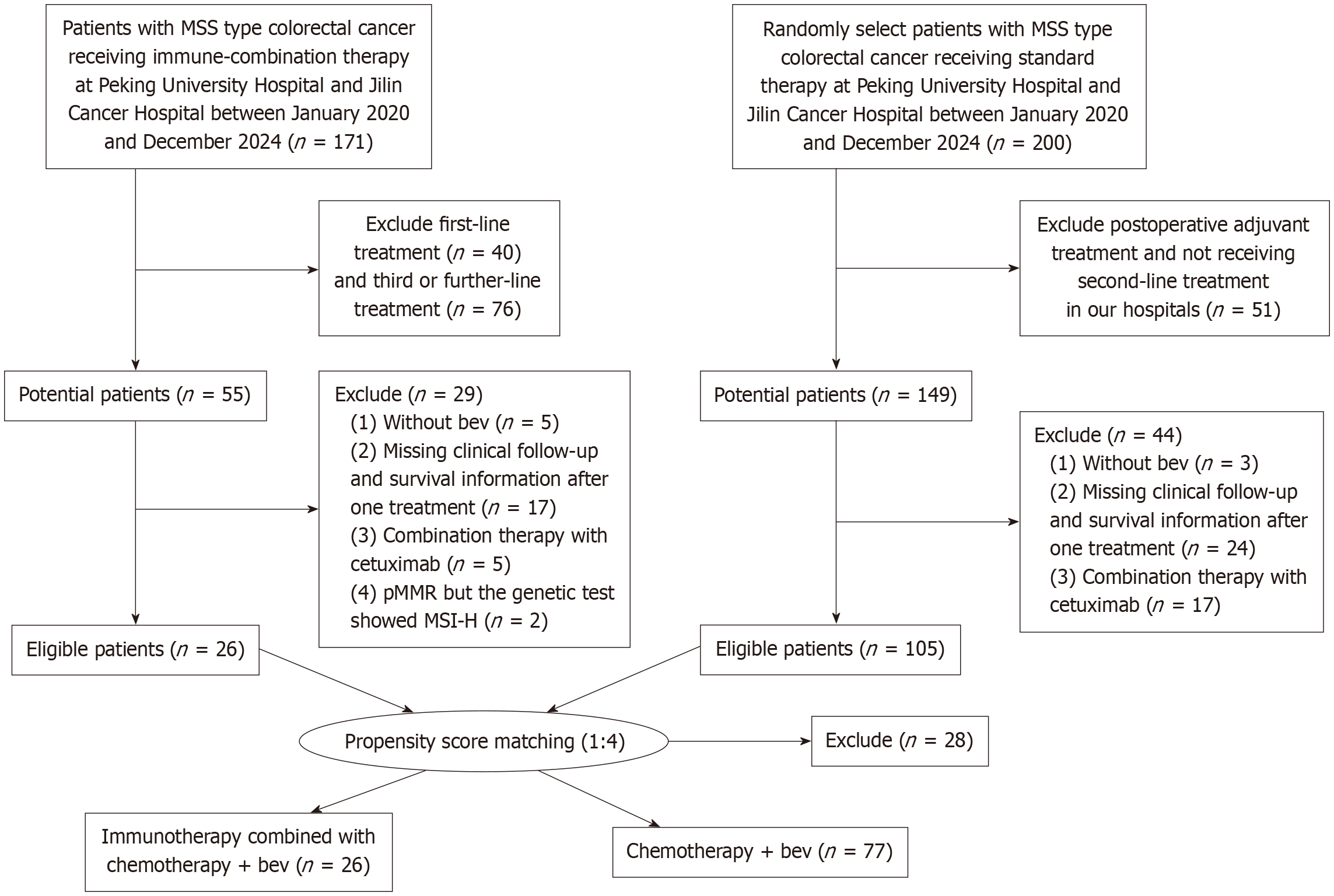
This study was conducted in compliance with the postulates of Declaration of Helsinki and approved by the Ethics Committee of Peking University First Hospital and Jilin Cancer Hospital. The requirement for patient approval or informed consent was waived by the Human Ethics Committee of Peking University First Hospital and Jilin Cancer Hospital, owing to the retrospective nature of the study and because the analysis used anonymous clinical data. The flowchart of patient selection is shown in Figure 1.
Follow-up data were collected through hospital records, telephone interviews, outpatient visits, and rehospitalizations. The data included age, sex, height, weight, ECOG status, primary tumor location, number of metastatic sites, tumor differentiation grade, percentage reduction in tumor volume, PFS, overall survival (OS), follow-up duration, and survival status. Additionally, peripheral blood indicators within 7 days prior to the initial triplet regimen were collected, encompassing absolute leukocyte count, absolute neutrophil count (ANC), absolute lymphocyte count (ALC), platelet (PLT) count, absolute monocyte count, absolute eosinophil count, albumin (ALB), lactate dehydrogenase (LDH), car
The last follow-up date was in January 2025. PFS was the primary outcome, defined as the time from enrollment to the first documented disease progression according to RECIST version 1.1, or death from any cause, whichever occurred first. Secondary outcomes included OS, ORR, disease control rate (DCR), and safety evaluation. OS was calculated from the date of enrollment to the date of death from any cause, with censored cases defined by the last available follow-up. ORR was defined as the proportion of patients with a best objective response of complete response or partial response according to RECIST criteria (version 1.1). DCR was defined as the proportion of patients with complete response, partial response, or stable disease according to RECIST criteria (version 1.1). Treatment-related adverse events were evaluated according to the Common Terminology Criteria for Adverse Events version 4.0.
The PSM method was used to eliminate potential confounding factors that could influence the therapeutic effect between the experimental and control groups. PSM analysis was conducted using the nearest-neighbor method with a caliper of 0.018 and a 1:4 matching ratio to balance characteristics such as age, sex, ECOG status, metastasis, and tumor location using the MatchIt package[20]. Categorical variables were compared using the χ2 test or Fisher’s exact test, while continuous variables were assessed using the Mann-Whitney test. Kaplan-Meier estimates were obtained to compare the actuarial survival and the two treatment efficacy endpoints between the two groups. Independent prognostic factors, hazard ratios (HRs), and 95% confidence intervals (CIs) were evaluated using the Cox proportional hazards (PH) model. The cutoff values for continuous variable hematological indicators in predicting patient survival were set at the median. Based on this cutoff value, patients were categorized into high-expression and low-expression groups. The predictive value of hematological indicators for the triplet regimen in MSS mCRC was assessed using receiver operating characteristic curve analysis. A two-sided P value of < 0.050 was considered statistically significant. All statistical analyses in our study were performed using R software (version 4.4.2).
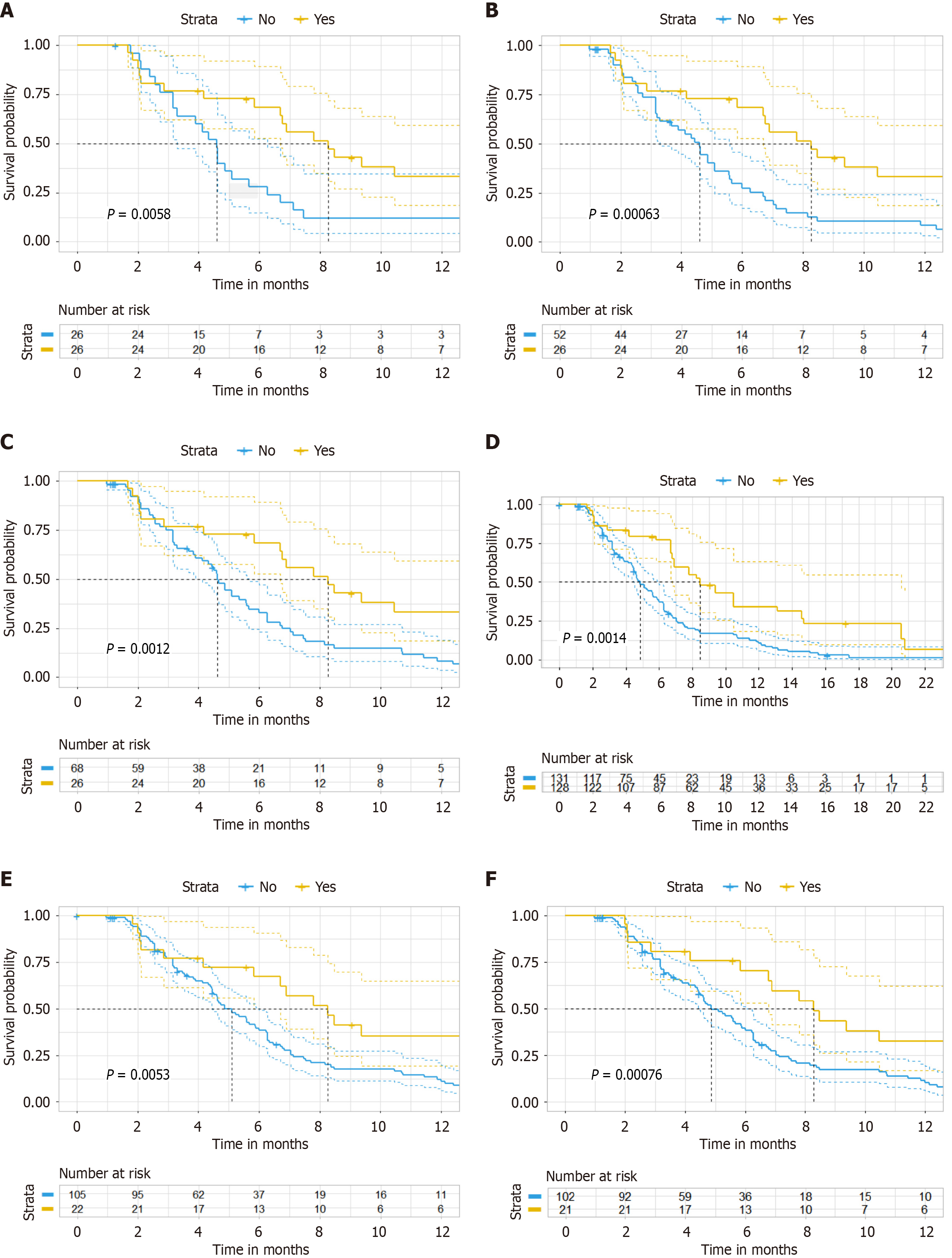
To address potential biases, three sensitivity analyses were conducted. First, we performed the Schoenfeld residual test on the original data to assess whether the covariates satisfied the PHs assumption, using the cox. zph function from the survival package[21]. If the PH assumption was met, we proceeded with univariate and multivariate regression analyses. Second, PSM analysis with varying matching ratios and inverse probability of treatment weighting are employed to adjust for baseline characteristics and evaluate treatment outcomes of PFS and OS. Third, to evaluate the robustness of the results, we both excluded four patients who received single-agent chemotherapy combined with bevacizumab and immunotherapy, and focused our analysis on patients treated with IRI-based regimens.
After one-to-four lines of PSM, the experimental group receiving triplet regimen included 26 patients, while the control group comprised 77 patients. Among the 103 patients (73 males and 30 females), 45 patients were over 60 years old. There were 29 cases (28.2%) of right-sided colon cancer, 85 patients underwent primary tumor surgery, and 69 patients (67%) had metastases in two or more organs. Sixty-eight patients had RAS mutations, and all cases were pMMR (Table 1). The main baseline characteristics of eligible patients were well-balanced between the two groups (Supplementary Figure 1A).
| Characteristics | Levels | Control group (n = 77) | Experiment group (n = 26) | P value |
| Age | > 60 | 33 (42.9) | 12 (46.2) | 0.949 |
| ≤ 60 | 44 (57.1) | 14 (53.8) | ||
| Gender | Male | 55 (71.4) | 18 (69.2) | 1.000 |
| Female | 22 (28.6) | 8 (30.8) | ||
| ECOG | 0-1 | 69 (89.6) | 22 (84.6) | 0.739 |
| 2 | 8 (10.4) | 4 (15.4) | ||
| Primary tumor location | Right colon | 20 (26) | 9 (34.6) | 0.552 |
| Left colon and rectum | 57 (74) | 17 (65.4) | ||
| Primary tumor surgery | No | 15 (19.5) | 3 (11.5) | 0.533 |
| Yes | 62 (80.5) | 23 (88.5) | ||
| Number of metastatic organs | 1 | 26 (33.8) | 8 (30.8) | 0.968 |
| ≥ 2 | 51 (66.2) | 18 (69.2) | ||
| Liver metastasis | No | 19 (24.7) | 8 (30.8) | 0.724 |
| Yes | 58 (75.3) | 18 (69.2) | ||
| Lung metastasis | No | 41 (53.2) | 11 (42.3) | 0.461 |
| Yes | 36 (46.8) | 15 (57.7) | ||
| RAS mutation type | Unknown | 15 (19.5) | 5 (19.2) | 0.872 |
| Wild-type | 12 (15.6) | 3 (11.5) | ||
| Mutation | 50 (64.9) | 18 (69.2) |
As of December 2024, the median follow-up period was 13.9 months in the matched cohort. The median PFS was 5.33 months, (95%CI: 4.6-6.33 months), and the median OS was 23 months. In the experimental group, the median PFS was 8.27 months (95%CI: 6.7-14.7 months), and the median OS was 8.6 months. In the control group, the median PFS was 4.63 months (95%CI: 3.9-5.67 months). The PFS in the experimental group was superior to that in the control group (HR = 0.414, 95%CI: 0.2462-0.6972, P = 0.00066) (Figure 2A). There was a trend towards a higher median OS in the experimental group compared to the control group (HR = 0.4504, 95%CI: 0.1897-1.07, P = 0.064) (Figure 2B). The ORR was 11.5% in the experimental group and 9% in the control group (P = 0.710), while the DCR was 76.9% and 53.2%, respectively (P = 0.058) (Table 2).
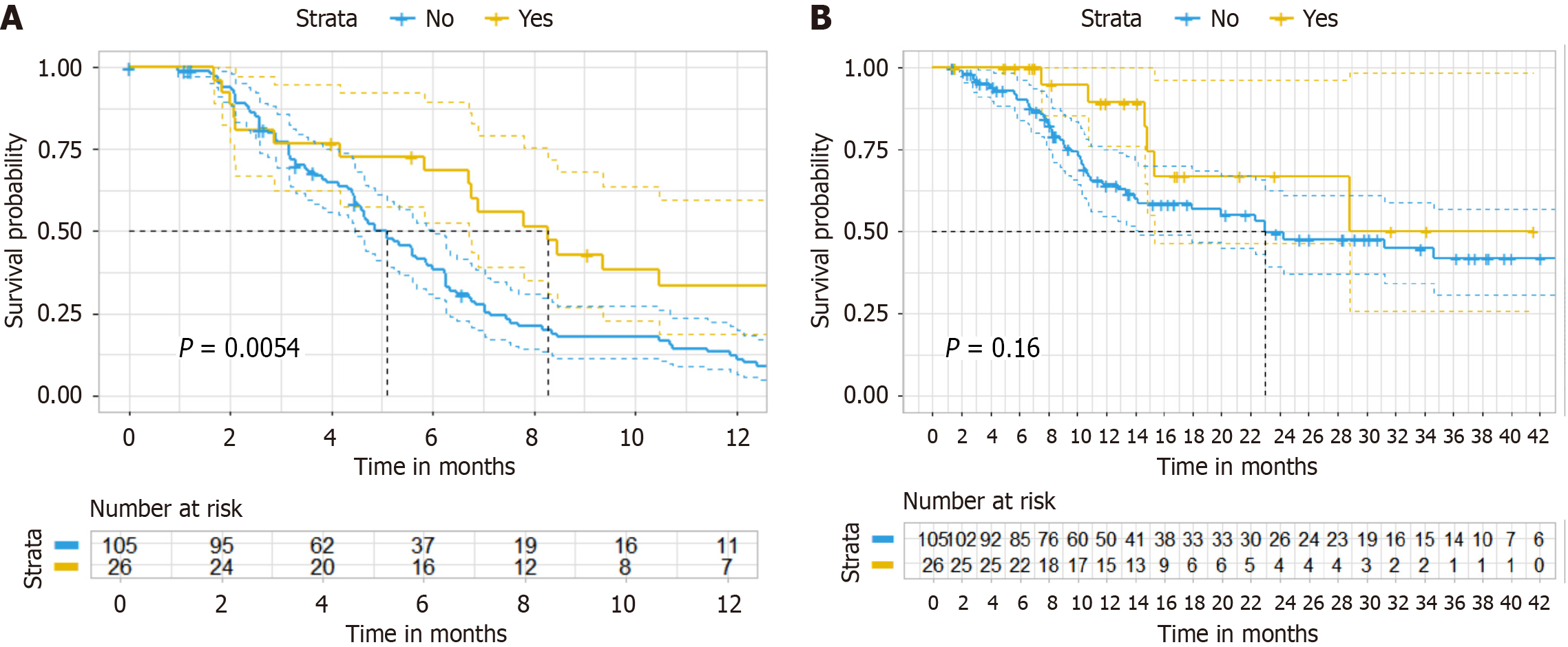
For the original cohort, the PFS in the experimental group was superior to that in the control group (HR = 0.414, 95%CI: 0.2462-0.6972, P = 0.0054) (Figure 3A) and the OS did not show a statistically significant difference between the two groups (HR = 0.547, 95%CI: 0.2328-1.287, P = 0.167) (Figure 3B). The independent variables conformed to the PHs assum
| Dependent: Survival (PFS/30, status) | All patients (n = 131) | HR (univariable) | HR (multivariable) | |
| Gender | Male | 76 (58.0) | - | - |
| Female | 55 (42.0) | 0.87 (0.60-1.26, P = 0.458) | - | |
| Age | > 60 | 66 (50.4) | - | - |
| ≤ 60 | 65 (49.6) | 0.73 (0.50-1.07, P = 0.110) | - | |
| ECOG | 0-1 | 116 (88.5) | - | - |
| 2 | 15 (11.5) | 1.34 (0.73-2.47, P = 0.340) | - | |
| Number of metastatic organs | 1 | 55 (42.0) | - | - |
| ≥ 2 | 76 (58.0) | 1.96 (1.32-2.90, P < 0.001) | 1.90 (1.27-2.84, P = 0.002) | |
| Primary tumor location | Right colon | 32 (24.4) | - | - |
| Left colon and rectum | 99 (75.6) | 0.92 (0.61-1.40, P = 0.697) | - | |
| Liver metastasis | No | 46 (35.1) | - | - |
| Yes | 85 (64.9) | 1.57 (1.04-2.36, P = 0.032) | 1.28 (0.84-1.96, P = 0.254) | |
| Lung metastasis | No | 70 (53.4) | - | - |
| Yes | 61 (46.6) | 1.23 (0.85-1.78, P = 0.274) | - | |
| RAS mutation type | Unknown | 25 (19.1) | - | - |
| Wild type | 20 (15.3) | 0.97 (0.51-1.85, P = 0.930) | - | |
| Mutation | 86 (65.6) | 1.37 (0.84-2.24, P = 0.202) | - | |
| Group | Control group | 105 (80.2) | - | - |
| Experimental group | 26 (19.8) | 0.51 (0.31-0.82, P = 0.006) | 0.49 (0.30-0.80, P = 0.004) | |
No substantial differences in adverse events were observed between the two groups. The incidence of grade 1-2 fatigue was slightly higher in the experimental group compared to the control group (30.8% vs 11.7%, P = 0.0332), but the incidence of grade 3-4 adverse events was similar between the two groups (Table 4).
| Toxicities | Experimental group (n = 26) | Control group (n = 77) | P value for grade 1-2 | P value for grade 3-4 | ||||
| Grade 0 | Grade 1-2 | Grade 3-4 | Grade 0 | Grade 1-2 | Grade 3-4 | |||
| Anemia | 8 (30.8) | 15 (57.7) | 3 (11.5) | 47 (61.0) | 29 (37.7) | 1 (1.3) | 0.1197a | 0.2451b |
| Neutropenia | 18 (69.2) | 7 (26.9) | 1 (3.8) | 58 (75.3) | 14 (18.2) | 5 (6.5) | 0.4996a | 1b |
| Leukocytopenia | 14 (53.8) | 11 (42.3) | 1 (3.8) | 55 (71.4) | 18 (23.4) | 4 (5.2) | 0.1088a | 0.3667b |
| Thrombocytopenia | 17 (65.4) | 9 (34.6) | 0 (0.0) | 62 (80.5) | 15 (19.5) | 0 (0.0) | 0.1902a | 1b |
| Proteinuria | 21 (80.8) | 5 (19.2) | 0 (0.0) | 70 (90.9) | 7 (9.1) | 0 (0.0) | 0.2984a | 1b |
| Aspartate transaminase increased | 18 (69.2) | 7 (26.9) | 1 (3.8) | 56 (72.7) | 20 (26.0) | 1 (1.3) | 1a | 1b |
| Alanine transaminase increased | 20 (76.9) | 5 (19.2) | 1 (3.8) | 65 (84.4) | 12 (15.6) | 0 (0.0) | 0.8985a | 1b |
| Alkaline phosphatase increased | 21 (80.8) | 5 (19.2) | 0 (0.0) | 69 (89.6) | 7 (9.1) | 1 (1.3) | 0.1732b | 1b |
| Blood bilirubin increased | 20 (76.9) | 5 (19.2) | 1 (3.8) | 61 (79.2) | 16 (20.8) | 0 (0.0) | 1a | 1b |
| Triglycerides increased | 20 (76.9) | 4 (15.4) | 2 (7.7) | 67 (87.0) | 10 (13.0) | 0 (0.0) | 0.7475b | 0.4967 b |
| Nausea | 18 (69.2) | 8 (30.8) | 0 (0.0) | 47 (61.0) | 30 (39.0) | 0 (0.0) | 0.6076a | 1b |
| Vomiting | 19 (73.1) | 7 (26.9) | 0 (0.0) | 61 (79.2) | 16 (20.8) | 0 (0.0) | 0.7054a | 1b |
| Fatigue | 18 (69.2) | 8 (30.8) | 0 (0.0) | 68 (88.3) | 9 (11.7) | 0 (0.0) | 0.0332b | 1b |
| Fever | 24 (92.3) | 2 (7.7) | 0 (0.0) | 73 (94.8) | 4 (5.2) | 0 (0.0) | 0.6407b | 1b |
| Diarrhea | 24 (92.3) | 1 (3.8) | 1 (3.8) | 71 (92.2) | 6 (7.8) | 0 (0.0) | 0.6759b | 1b |
| Peripheral neurotoxicity | 25 (96.2) | 1 (3.8) | 0 (0.0) | 77 (100.0) | 0 (0.0) | 0 (0.0) | 0.2524b | 1b |
| Hand-foot syndrome | 23 (88.5) | 3 (11.5) | 0 (0.0) | 73 (94.8) | 4 (5.2) | 0 (0.0) | 0.3648b | 1b |
| Hypertension | 24 (92.3) | 1 (3.8) | 1 (3.8) | 72 (93.5) | 4 (5.2) | 1 (1.3) | 1b | 1b |
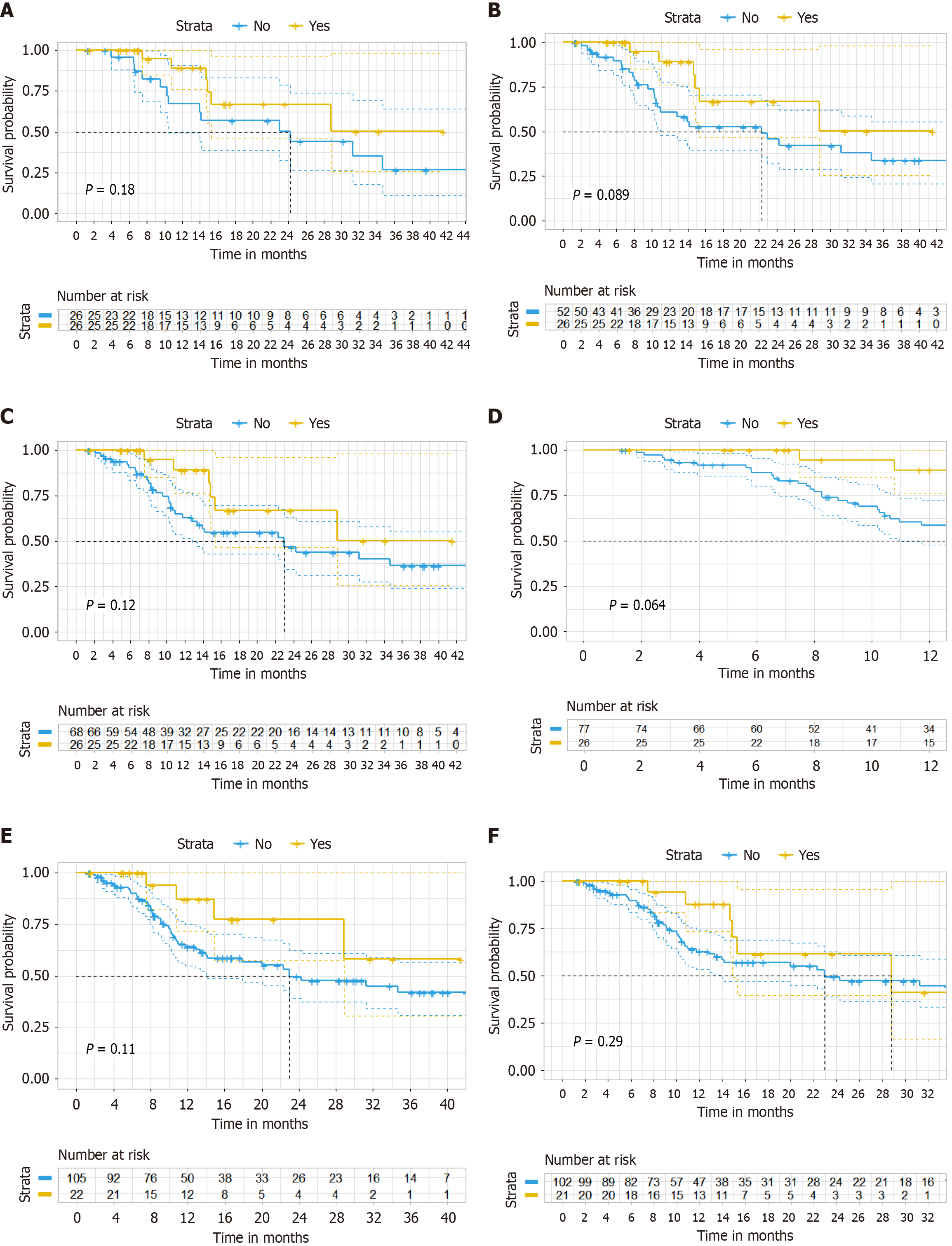
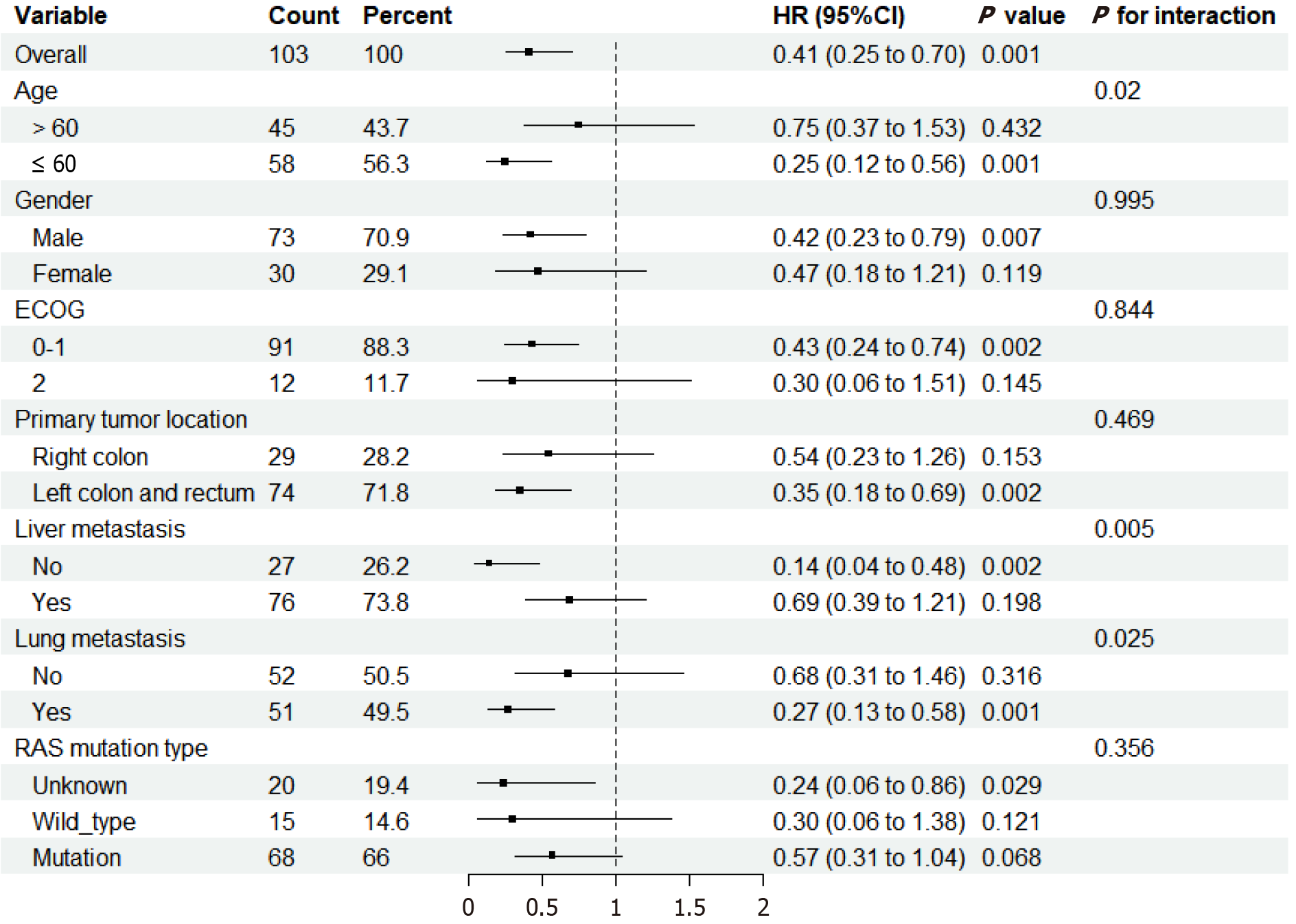
Subgroup analysis showed that patients younger than 60 years old (HR = 0.25, 95%CI: 0.12-0.56, P = 0.001), those without liver metastases (HR = 0.14, 95%CI: 0.04-0.48, P = 0.002), and those with lung metastases (HR = 0.27, 95%CI: 0.13-0.58, P = 0.001) are more likely to benefit from triplet regimen (Figure 6).
We analyzed the baseline hematological indicators associated with PFS in patients receiving triplet regimen (Table 5). Univariate analysis revealed that patients with advanced age, right colorectal cancer (CRC), normal carbohydrate antigen 199 levels, high advanced lung cancer inflammation index, and low LDH could benefit from triplet regimen. Multivariate analysis further identified liver metastasis (HR = 8.15, 95%CI: 1.39-47.83, P = 0.020) and LDH (HR = 4.11, 95%CI: 1.02-16.55, P = 0.046) as independent prognostic risk factors (Table 6).
| Characteristics | Stats | Normal range |
| Height (cm) | 168.3 ± 8.0 | 140-190 |
| Weight (kg) | 67.7 ± 12.0 | 40-100 |
| ALLC (109/L) | 6.0 ± 1.9 | 3.5-9.5 |
| RDW (%) | 15.2 ± 3.8 | 11.6-14.8 |
| PLT (109/L) | 178.2 ± 63.7 | 125-350 |
| ANC (109/L) | 4.1 ± 1.4 | 1.8-6.3 |
| ALC (109/L) | 1.4 ± 0.5 | 1.1-3.2 |
| AMC (109/L) | 0.4 ± 0.1 | 0.1-0.6 |
| AEC (109/L) | 0.2 ± 0.2 | 0.02-0.52 |
| ALB (g/L) | 41.4 ± 4.0 | 40-55 |
| LDH (IU/L) | 240.0 ± 154.9 | 109-245 |
| FIB (g/L) | 3.6 ± 0.7 | 2-4 |
| Dimer (ng/ml) | 257.3 ± 647.3 | 0-500 |
| CEA (ng/mL), n (%) | 6 (24.0%) | 0-5 |
| 19 (76.0%) | - | |
| CA199 (IU/mL), n (%) | 13 (52.0%) | 0-37 |
| 12 (48.0%) | - |
| Dependent: Survival (PFS/30, status) | All | HR (univariable) | HR (multivariable) | |
| Age | > 60 | 11 (44.0) | - | - |
| ≤ 60 | 14 (56.0) | 0.20 (0.07-0.62, P = 0.005) | 0.27 (0.05-1.48, P = 0.133) | |
| Gender | Male | 18 (72.0) | - | - |
| Female | 7 (28.0) | 1.01 (0.40-2.54, P = 0.979) | - | |
| ECOG | 1 | 21 (84.0) | - | - |
| 2 | 4 (16.0) | 1.01 (0.23-4.55, P = 0.985) | - | |
| Location | Right colon | 9 (36.0) | - | - |
| Left colon and rectum | 16 (64.0) | 0.73 (0.31-1.74, P = 0.477) | - | |
| Liver metastasis | No | 7 (28.0) | - | - |
| Yes | 18 (72.0) | 5.23 (1.48-18.50, P = 0.010) | 8.15 (1.39-47.83, P = 0.020) | |
| Lung metastasis | No | 10 (40.0) | - | - |
| Yes | 15 (60.0) | 0.62 (0.25-1.51, P = 0.291) | - | |
| RAS mutation type | Unknown | 5 (20.0) | - | - |
| Wild type | 3 (12.0) | 0.75 (0.13-4.20, P = 0.742) | - | |
| Mutation | 17 (68.0) | 2.30 (0.74-7.14, P = 0.151) | - | |
| CEA | Normal | 6 (24.0) | - | - |
| Abnormal | 19 (76.0) | 0.59 (0.21-1.67, P = 0.316) | - | |
| CA199 | Normal | 13 (52.0) | - | - |
| Abnormal | 12 (48.0) | 3.16 (1.12-8.88, P = 0.029) | 1.06 (0.23-4.90, P = 0.942) | |
| NLR | ≤ 2.9 | 13 (52.0) | - | - |
| > 2.9 | 12 (48.0) | 1.45 (0.60-3.55, P = 0.410) | - | |
| LMR | ≤ 3.63 | 13 (52.0) | - | - |
| > 3.63 | 12 (48.0) | 0.57 (0.23-1.39, P = 0.216) | - | |
| PLR | ≤ 121 | 13 (52.0) | - | - |
| > 121 | 12 (48.0) | 1.43 (0.58-3.48, P = 0.436) | - | |
| BMI | ≤ 24.9 | 12 (48.0) | - | - |
| > 24.9 | 13 (52.0) | 1.02 (0.43-2.42, P = 0.963) | - | |
| ALI | ≤ 260.9 | 13 (52.0) | - | - |
| > 260.9 | 12 (48.0) | 0.30 (0.10-0.85, P = 0.023) | 0.50 (0.15-1.64, P = 0.252) | |
| SII | ≤ 426.8 | 13 (52.0) | - | - |
| > 426.8 | 12 (48.0) | 1.74 (0.72-4.25, P = 0.220) | - | |
| ALLC | ≤ 6.0 | 13 (52.0) | - | - |
| > 6.0 | 12 (48.0) | 1.31 (0.53-3.20, P = 0.559) | - | |
| ANC | ≤ 4.1 | 13 (52.0) | - | - |
| > 4.1 | 12 (48.0) | 1.31 (0.53-3.20, P = 0.559) | - | |
| ALC | ≤ 1.4 | 13 (52.0) | - | - |
| > 1.4 | 12 (48.0) | 0.61 (0.25-1.50, P = 0.285) | - | |
| AMC | ≤ 0.4 | 13 (52.0) | - | - |
| > 0.4 | 12 (48.0) | 0.71 (0.28-1.82, P = 0.473) | - | |
| AEC | ≤ 0.2 | 13 (52.0) | - | - |
| > 0.2 | 12 (48.0) | 1.59 (0.65-3.89, P = 0.309) | - | |
| RDW | ≤ 15.2 | 13 (52.0) | - | - |
| > 15.2 | 12 (48.0) | 1.04 (0.42-2.57, P = 0.938) | - | |
| LDH | ≤ 240.0 | 13 (52.0) | - | - |
| > 240.0 | 12 (48.0) | 3.09 (1.16-8.22, P = 0.024) | 5.72 (1.58-20.72, P = 0.008) | |
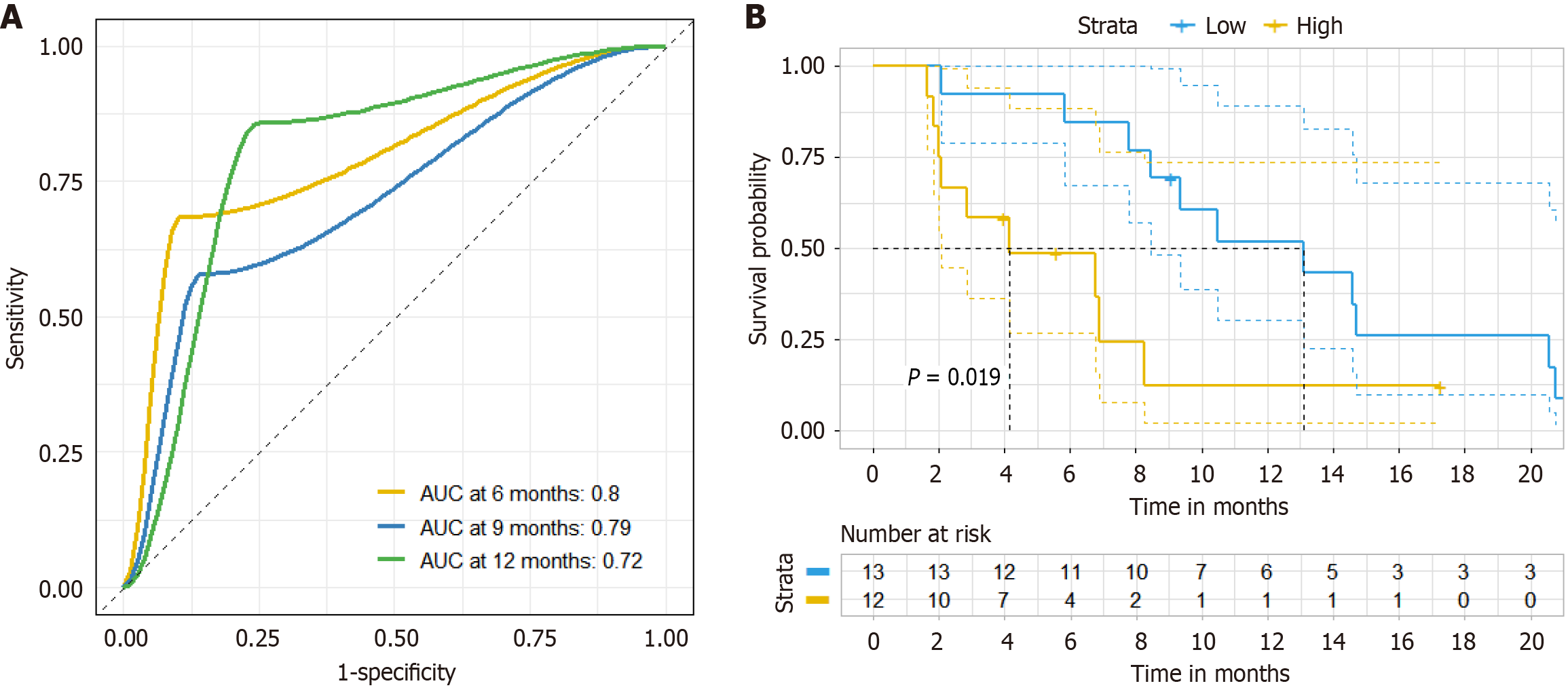
The area under curve values of LDH in predicting the efficacy of second-line triplet regimen for MSS mCRC patients at 6 months, 9 months, and 12 months were 0.80, 0.79, and 0.72 respectively (Figure 7A). Based on the median value of LDH, MSS mCRC patients were divided into high groups and low groups. Survival analysis demonstrated that patients in the high-value group had significantly prolonged PFS compared to those in the low-value group (P = 0.019) (Figure 7B).
Multiple Phase III clinical studies have demonstrated that, following the failure of first-line chemotherapy combined with bevacizumab in advanced colorectal cancer, switching the chemotherapy regimen while continuing anti-angiogenic therapy provides additional survival benefits compared to chemotherapy alone[7,8,22]. However, the role of immunotherapy in second-line sequential treatment for mCRC remains unestablished. Previous studies have shown that IRI-based chemotherapy for second-line treatment of advanced CRC yields an ORR of 4%-18.8%, with the median PFS of 2.5-5.8 months and the median OS of 9.9-19.5 months[8,22-25]. When IRI-based chemotherapy is combined with anti-angiogenic targeted therapy for second-line treatment of advanced CRC, the ORR ranges from 14%-19.8%, with median PFS and OS ranging from 3.5-6.9 months and 11.9-13.5 months, respectively[8,22,24]. This study revealed an ORR of 11.5% and a DCR of 76.9% with triplet regimen. The triplet regimen for second-line treatment of advanced CRC showed superior PFS compared to the doublet regimen (P = 0.00066). Although our analysis demonstrated a trend toward OS benefit (P = 0.064), this finding requires cautious interpretation. Whether PFS improvement and depth of response will ultimately translate into definitive OS gains remains uncertain and necessitates validation through larger sample sizes and prospective cohort studies.
The results of chemotherapy combined with anti-PD-1 immunotherapy for first-line treatment of patients with MSS mCRC are unsatisfactory, with limited overall efficacy improvement. Further consideration is needed regarding the combination and value of this treatment modality. The CHECKMATE-9X8[16] study explored the efficacy of nivolumab plus mFOLFOX6/bevacizumab [nivolumab + standard-of-care (SOC)] vs mFOLFOX6/bevacizumab (SOC) alone for first-line treatment of mCRC. The ORR was 60% in the nivolumab group vs 46% in the SOC group, with a median PFS of 11.9 months in both groups. The median OS was 29.2 months in the nivolumab group, and not reached in the SOC group. Exploratory subgroup analysis of this study indicated that a higher proportion of patients with CMS1 and CMS3 tumor types remained progression-free at 12 months with nivolumab treatment. The AtezoTRIBE randomized Phase II trial, comparing FOLFOXIRI and bevacizumab with or without atezolizumab, showed that adding atezolizumab prolonged PFS in the overall population[26]. However, in the MSS patient subgroup, the addition of atezolizumab did not significantly improve PFS. In multivariate analysis, high tumor mutational burden and high immune score were inde
Basic studies have shown that liver metastases from CRC are in an immunosuppressive environment[29]. Subgroup analysis of our study found that patients with liver metastases had worse prognoses than those without liver metastases, while patients with lung metastases had better prognoses with triplet regimen. This is similar to the findings of the REGONIVO study[30], which reported better prognoses for patients with lung metastases compared to those with liver metastases. In contrast, subgroup analysis of the bevacizumab and CAPEOX study found that patients with liver metastases had better prognoses in the triplet regimen[18]. Further basic research is needed to explore the efficacy of anti-PD1 immunotherapy in mCRC with different metastatic sites.
The results of this study indicate that baseline LDH level is an independent prognostic factor for PFS following the triplet regimen in MSS mCRC. From a metabolic perspective, tumor cells tend to prioritize glycolysis for energy production, where glucose is metabolized to pyruvate and then converted to lactate via LDH instead of undergoing aerobic oxidation through the mitochondrial tricarboxylic acid cycle, a phenomenon known as the “Warburg effect”[31]. Research has shown that in metastatic cervical cancer patients receiving combination immunotherapy, higher LDH levels are associated with lower survival rates[32]. Additionally, patients with normal LDH levels before receiving camrelizumab treatment for esophageal squamous cell carcinoma exhibit longer OS[33].
We acknowledge that our multicenter retrospective cohort study has several limitations. First, as a retrospective analysis with a relatively small sample size in the experimental group, data was collected from only two participating centers, and the findings require validation in larger cohorts and more research institutions. Second, the heterogeneity in treatment regimens - including diverse chemotherapy protocols and five different anti-PD-1 agents - may have con
In the second-line treatment of MSS mCRC, chemotherapy combined with bevacizumab and anti-PD-1 immunotherapy is superior to the traditional regimen of chemotherapy with bevacizumab. This indicates that the triplet regimen is a promising therapeutic strategy, which is expected to provide more clinical benefits. Subgroup analysis shows that patients younger than 60 years old, those without liver metastases, and those with lung metastases may benefit more significantly.
We are grateful to the patients and their families for supporting the study.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14770] [Article Influence: 7385.0] [Reference Citation Analysis (21)] |
| 2. | Tournigand C, André T, Achille E, Lledo G, Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G, Landi B, Colin P, Louvet C, de Gramont A. FOLFIRI Followed by FOLFOX6 or the Reverse Sequence in Advanced Colorectal Cancer: A Randomized GERCOR Study. J Clin Oncol. 2023;41:3469-3477. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (6)] |
| 3. | Bennouna J, Hiret S, Bertaut A, Bouché O, Deplanque G, Borel C, François E, Conroy T, Ghiringhelli F, des Guetz G, Seitz JF, Artru P, Hebbar M, Stanbury T, Denis MG, Adenis A, Borg C. Continuation of Bevacizumab vs Cetuximab Plus Chemotherapy After First Progression in KRAS Wild-Type Metastatic Colorectal Cancer: The UNICANCER PRODIGE18 Randomized Clinical Trial. JAMA Oncol. 2019;5:83-90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (7)] |
| 4. | Hecht JR, Cohn A, Dakhil S, Saleh M, Piperdi B, Cline-Burkhardt M, Tian Y, Go WY. SPIRITT: A Randomized, Multicenter, Phase II Study of Panitumumab with FOLFIRI and Bevacizumab with FOLFIRI as Second-Line Treatment in Patients with Unresectable Wild Type KRAS Metastatic Colorectal Cancer. Clin Colorectal Cancer. 2015;14:72-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 91] [Article Influence: 8.3] [Reference Citation Analysis (6)] |
| 5. | Cervantes A, Adam R, Roselló S, Arnold D, Normanno N, Taïeb J, Seligmann J, De Baere T, Osterlund P, Yoshino T, Martinelli E; ESMO Guidelines Committee. Electronic address: clinicalguidelines@esmo.org. Metastatic colorectal cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34:10-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1384] [Cited by in RCA: 1223] [Article Influence: 407.7] [Reference Citation Analysis (11)] |
| 6. | Peeters M, Price TJ, Cervantes A, Sobrero AF, Ducreux M, Hotko Y, André T, Chan E, Lordick F, Punt CJ, Strickland AH, Wilson G, Ciuleanu TE, Roman L, Van Cutsem E, Tzekova V, Collins S, Oliner KS, Rong A, Gansert J. Randomized phase III study of panitumumab with fluorouracil, leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line treatment in patients with metastatic colorectal cancer. J Clin Oncol. 2010;28:4706-4713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 811] [Cited by in RCA: 744] [Article Influence: 46.5] [Reference Citation Analysis (6)] |
| 7. | Bennouna J, Sastre J, Arnold D, Österlund P, Greil R, Van Cutsem E, von Moos R, Viéitez JM, Bouché O, Borg C, Steffens CC, Alonso-Orduña V, Schlichting C, Reyes-Rivera I, Bendahmane B, André T, Kubicka S; ML18147 Study Investigators. Continuation of bevacizumab after first progression in metastatic colorectal cancer (ML18147): a randomised phase 3 trial. Lancet Oncol. 2013;14:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 963] [Cited by in RCA: 910] [Article Influence: 70.0] [Reference Citation Analysis (6)] |
| 8. | Van Cutsem E, Tabernero J, Lakomy R, Prenen H, Prausová J, Macarulla T, Ruff P, van Hazel GA, Moiseyenko V, Ferry D, McKendrick J, Polikoff J, Tellier A, Castan R, Allegra C. Addition of aflibercept to fluorouracil, leucovorin, and irinotecan improves survival in a phase III randomized trial in patients with metastatic colorectal cancer previously treated with an oxaliplatin-based regimen. J Clin Oncol. 2012;30:3499-3506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1173] [Cited by in RCA: 1045] [Article Influence: 74.6] [Reference Citation Analysis (4)] |
| 9. | Giantonio BJ, Catalano PJ, Meropol NJ, O'Dwyer PJ, Mitchell EP, Alberts SR, Schwartz MA, Benson AB 3rd. Bevacizumab in Combination With Oxaliplatin, Fluorouracil, and Leucovorin (FOLFOX4) for Previously Treated Metastatic Colorectal Cancer: Results From the Eastern Cooperative Oncology Group Study E3200. J Clin Oncol. 2023;41:3670-3675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (6)] |
| 10. | Sobrero AF, Maurel J, Fehrenbacher L, Scheithauer W, Abubakr YA, Lutz MP, Vega-Villegas ME, Eng C, Steinhauer EU, Prausova J, Lenz HJ, Borg C, Middleton G, Kröning H, Luppi G, Kisker O, Zubel A, Langer C, Kopit J, Burris HA 3rd. EPIC: phase III trial of cetuximab plus irinotecan after fluoropyrimidine and oxaliplatin failure in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26:2311-2319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 749] [Cited by in RCA: 681] [Article Influence: 37.8] [Reference Citation Analysis (6)] |
| 11. | Seymour MT, Brown SR, Middleton G, Maughan T, Richman S, Gwyther S, Lowe C, Seligmann JF, Wadsley J, Maisey N, Chau I, Hill M, Dawson L, Falk S, O'Callaghan A, Benstead K, Chambers P, Oliver A, Marshall H, Napp V, Quirke P. Panitumumab and irinotecan versus irinotecan alone for patients with KRAS wild-type, fluorouracil-resistant advanced colorectal cancer (PICCOLO): a prospectively stratified randomised trial. Lancet Oncol. 2013;14:749-759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 307] [Article Influence: 23.6] [Reference Citation Analysis (6)] |
| 12. | Peeters M, Price TJ, Cervantes A, Sobrero AF, Ducreux M, Hotko Y, André T, Chan E, Lordick F, Punt CJ, Strickland AH, Wilson G, Ciuleanu TE, Roman L, Van Cutsem E, Tian Y, Sidhu R. Final results from a randomized phase 3 study of FOLFIRI {+/-} panitumumab for second-line treatment of metastatic colorectal cancer. Ann Oncol. 2014;25:107-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 155] [Article Influence: 12.9] [Reference Citation Analysis (6)] |
| 13. | Casak SJ, Marcus L, Fashoyin-Aje L, Mushti SL, Cheng J, Shen YL, Pierce WF, Her L, Goldberg KB, Theoret MR, Kluetz PG, Pazdur R, Lemery SJ. FDA Approval Summary: Pembrolizumab for the First-line Treatment of Patients with MSI-H/dMMR Advanced Unresectable or Metastatic Colorectal Carcinoma. Clin Cancer Res. 2021;27:4680-4684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 128] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 14. | Overman MJ, Lonardi S, Wong KYM, Lenz HJ, Gelsomino F, Aglietta M, Morse MA, Van Cutsem E, McDermott R, Hill A, Sawyer MB, Hendlisz A, Neyns B, Svrcek M, Moss RA, Ledeine JM, Cao ZA, Kamble S, Kopetz S, André T. Durable Clinical Benefit With Nivolumab Plus Ipilimumab in DNA Mismatch Repair-Deficient/Microsatellite Instability-High Metastatic Colorectal Cancer. J Clin Oncol. 2018;36:773-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1757] [Cited by in RCA: 1581] [Article Influence: 197.6] [Reference Citation Analysis (6)] |
| 15. | Qian C, Liu C, Liu W, Zhou R, Zhao L. Targeting vascular normalization: a promising strategy to improve immune-vascular crosstalk in cancer immunotherapy. Front Immunol. 2023;14:1291530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 16. | Lenz HJ, Parikh A, Spigel DR, Cohn AL, Yoshino T, Kochenderfer M, Elez E, Shao SH, Deming D, Holdridge R, Larson T, Chen E, Mahipal A, Ucar A, Cullen D, Baskin-Bey E, Kang T, Hammell AB, Yao J, Tabernero J. Modified FOLFOX6 plus bevacizumab with and without nivolumab for first-line treatment of metastatic colorectal cancer: phase 2 results from the CheckMate 9X8 randomized clinical trial. J Immunother Cancer. 2024;12:e008409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 61] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 17. | Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, Marisa L, Roepman P, Nyamundanda G, Angelino P, Bot BM, Morris JS, Simon IM, Gerster S, Fessler E, De Sousa E Melo F, Missiaglia E, Ramay H, Barras D, Homicsko K, Maru D, Manyam GC, Broom B, Boige V, Perez-Villamil B, Laderas T, Salazar R, Gray JW, Hanahan D, Tabernero J, Bernards R, Friend SH, Laurent-Puig P, Medema JP, Sadanandam A, Wessels L, Delorenzi M, Kopetz S, Vermeulen L, Tejpar S. The consensus molecular subtypes of colorectal cancer. Nat Med. 2015;21:1350-1356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4335] [Cited by in RCA: 3896] [Article Influence: 354.2] [Reference Citation Analysis (12)] |
| 18. | Fang X, Zhong C, Weng S, Hu H, Wang J, Xiao Q, Wang J, Sun L, Xu D, Liao X, Dong C, Zhang S, Li J, Ding K, Yuan Y. Sintilimab plus bevacizumab and CapeOx (BBCAPX) on first-line treatment in patients with RAS mutant, microsatellite stable, metastatic colorectal cancer: study protocol of a randomized, open-label, multicentric study. BMC Cancer. 2023;23:676. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (7)] |
| 19. | Ree AH, Šaltytė Benth J, Hamre HM, Kersten C, Hofsli E, Guren MG, Sorbye H, Johansen C, Negård A, Bjørnetrø T, Nilsen HL, Berg JP, Flatmark K, Meltzer S. First-line oxaliplatin-based chemotherapy and nivolumab for metastatic microsatellite-stable colorectal cancer-the randomised METIMMOX trial. Br J Cancer. 2024;130:1921-1928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 20. | Wang X, Zhang B, Chen X, Mo H, Wu D, Lan B, Li Q, Xu B, Huang J. Lactate dehydrogenase and baseline markers associated with clinical outcomes of advanced esophageal squamous cell carcinoma patients treated with camrelizumab (SHR-1210), a novel anti-PD-1 antibody. Thorac Cancer. 2019;10:1395-1401. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (7)] |
| 21. | Ho DE, Imai K, King G, Stuart EA. MatchIt: Nonparametric Preprocessing for Parametric Causal Inference. J Stat Soft. 2011;42:1-28. [DOI] [Full Text] |
| 22. | Lin H, Zelterman D. Modeling Survival Data: Extending the Cox Model. Technometrics. 2002;44:85-86. [RCA] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 93] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 23. | Tabernero J, Yoshino T, Cohn AL, Obermannova R, Bodoky G, Garcia-Carbonero R, Ciuleanu TE, Portnoy DC, Van Cutsem E, Grothey A, Prausová J, Garcia-Alfonso P, Yamazaki K, Clingan PR, Lonardi S, Kim TW, Simms L, Chang SC, Nasroulah F; RAISE Study Investigators. Ramucirumab versus placebo in combination with second-line FOLFIRI in patients with metastatic colorectal carcinoma that progressed during or after first-line therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine (RAISE): a randomised, double-blind, multicentre, phase 3 study. Lancet Oncol. 2015;16:499-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 805] [Cited by in RCA: 694] [Article Influence: 63.1] [Reference Citation Analysis (8)] |
| 24. | André T, Louvet C, Maindrault-Goebel F, Couteau C, Mabro M, Lotz JP, Gilles-Amar V, Krulik M, Carola E, Izrael V, de Gramont A. CPT-11 (irinotecan) addition to bimonthly, high-dose leucovorin and bolus and continuous-infusion 5-fluorouracil (FOLFIRI) for pretreated metastatic colorectal cancer. GERCOR. Eur J Cancer. 1999;35:1343-1347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 197] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 25. | Peeters M, Strickland AH, Lichinitser M, Suresh AV, Manikhas G, Shapiro J, Rogowski W, Huang X, Wu B, Warner D, Jain R, Tebbutt NC. A randomised, double-blind, placebo-controlled phase 2 study of trebananib (AMG 386) in combination with FOLFIRI in patients with previously treated metastatic colorectal carcinoma. Br J Cancer. 2013;108:503-511. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 26. | Muro K, Boku N, Shimada Y, Tsuji A, Sameshima S, Baba H, Satoh T, Denda T, Ina K, Nishina T, Yamaguchi K, Takiuchi H, Esaki T, Tokunaga S, Kuwano H, Komatsu Y, Watanabe M, Hyodo I, Morita S, Sugihara K. Irinotecan plus S-1 (IRIS) versus fluorouracil and folinic acid plus irinotecan (FOLFIRI) as second-line chemotherapy for metastatic colorectal cancer: a randomised phase 2/3 non-inferiority study (FIRIS study). Lancet Oncol. 2010;11:853-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 186] [Article Influence: 11.6] [Reference Citation Analysis (6)] |
| 27. | Antoniotti C, Rossini D, Pietrantonio F, Salvatore L, Lonardi S, Tamberi S, Marmorino F, Moretto R, Prisciandaro M, Tamburini E, Tortora G, Passardi A, Bergamo F, Raimondi A, Ritorto G, Borelli B, Conca V, Ugolini C, Aprile G, Antonuzzo L, Gelsomino F, Martinelli E, Pella N, Masi G, Boni L, Galon J, Cremolini C. Upfront Fluorouracil, Leucovorin, Oxaliplatin, and Irinotecan Plus Bevacizumab With or Without Atezolizumab for Patients With Metastatic Colorectal Cancer: Updated and Overall Survival Results of the ATEZOTRIBE Study. J Clin Oncol. 2024;42:2637-2644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 28. | Chen EX, Kavan P, Tehfe M, Kortmansky JS, Sawyer MB, Chiorean EG, Lieu CH, Polite B, Wong L, Fakih M, Spencer K, Chaves J, Li C, Leconte P, Adelberg D, Kim R. Pembrolizumab Plus Binimetinib With or Without Chemotherapy for MSS/pMMR Metastatic Colorectal Cancer: Outcomes From KEYNOTE-651 Cohorts A, C, and E. Clin Colorectal Cancer. 2024;23:183-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 29. | Damato A, Bergamo F, Antonuzzo L, Nasti G, Iachetta F, Romagnani A, Gervasi E, Larocca M, Pinto C. FOLFOXIRI/Bevacizumab Plus Nivolumab as First-Line Treatment in Metastatic Colorectal Cancer RAS/BRAF Mutated: Safety Run-In of Phase II NIVACOR Trial. Front Oncol. 2021;11:766500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (7)] |
| 30. | Wang F, Long J, Li L, Wu ZX, Da TT, Wang XQ, Huang C, Jiang YH, Yao XQ, Ma HQ, Lian ZX, Zhao ZB, Cao J. Single-cell and spatial transcriptome analysis reveals the cellular heterogeneity of liver metastatic colorectal cancer. Sci Adv. 2023;9:eadf5464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 147] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 31. | Fakih M, Raghav KPS, Chang DZ, Larson T, Cohn AL, Huyck TK, Cosgrove D, Fiorillo JA, Tam R, D'Adamo D, Sharma N, Brennan BJ, Wang YA, Coppieters S, Zebger-Gong H, Weispfenning A, Seidel H, Ploeger BA, Mueller U, Oliveira CSV, Paulson AS. Regorafenib plus nivolumab in patients with mismatch repair-proficient/microsatellite stable metastatic colorectal cancer: a single-arm, open-label, multicentre phase 2 study. EClinicalMedicine. 2023;58:101917. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 90] [Article Influence: 30.0] [Reference Citation Analysis (7)] |
| 32. | Gatenby RA, Gillies RJ. Why do cancers have high aerobic glycolysis? Nat Rev Cancer. 2004;4:891-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4045] [Cited by in RCA: 3592] [Article Influence: 163.3] [Reference Citation Analysis (6)] |
| 33. | Cheng M, Li G, Liu Z, Yang Q, Jiang Y. Pretreatment Neutrophil-to-Lymphocyte Ratio and Lactate Dehydrogenase Predict the Prognosis of Metastatic Cervical Cancer Treated with Combination Immunotherapy. J Oncol. 2022;2022:1828473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (7)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/