Published online Mar 28, 2025. doi: 10.3748/wjg.v31.i12.98174
Revised: October 29, 2024
Accepted: December 23, 2024
Published online: March 28, 2025
Processing time: 279 Days and 19.1 Hours
The interleukin-17 (IL-17) mediated aberrant immune-inflammatory response plays a paramount role in ulcerative colitis (UC). γδT17 cells are one of the critical sources of IL-17, but the role they play in UC remains under debate.
To clarify the role of γδT17 cells in patients with mild-to-moderate UC.
A single-centre observational pragmatic study was conducted on patients with UC who attended the outpatient and inpatient departments of Xiyuan Hospital of the China Academy of Traditional Chinese Medicine from September 2020 to December 2022. The research population consisted of two groups of adult patients. The first group consisted of healthy volunteers with no significant abnormalities on colonoscopy, and the other group consisted of patients with mild-to-moderate ulcerative colitis. Serum samples from healthy volunteers and patients with UC were collected for the detection of relevant inflammatory factors. Moreover, five colon mucosa samples were randomly selected from each group for testing and analyses.
An increased number of γδT17 cells and hyperactivation of the NLR family pyrin domain containing 3/IL-1β signaling pathway were observed in colonic mucosal tissues from patients with UC.
Hyperactivation of the NLR family pyrin domain containing 3/IL-1β signaling pathway promotes the activation of γδT17 cells in colonic mucosal tissues of patients with UC.
Core Tip: The contribution of γδT17 cells in patients with ulcerative colitis (UC) is controversial. In this study, serum samples from healthy volunteers and UC patients were analysed for associated inflammatory factors, and five randomly selected colonic mucosal tissue biopsy samples from each group were analysed by immunohistochemistry and immunofluorescence to clarify the role of γδT17 cells in UC patients. The serum levels of inflammatory factors secreted after γδT17 cell activation were significantly elevated in patients with UC, the expression level of γδT17 cells in colonic mucosal tissues was significantly elevated, and the NLR family pyrin domain containing 3/interleukin-1β signaling pathway was hyperactivated.
- Citation: Ma J, Wang FY, Tang XD. Involvement of the NLRP3/IL-1β pathway in activation and effector functions of γδT17 cells in patients with ulcerative colitis. World J Gastroenterol 2025; 31(12): 98174
- URL: https://www.wjgnet.com/1007-9327/full/v31/i12/98174.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i12.98174
Inflammatory bowel disease (IBD) encompasses a series of chronic conditions impacting the gastrointestinal tract and extraintestinal organs[1]. As a global disorder, with more than 6 million cases diagnosed per year worldwide and a prevalence in developed countries approaching 5%, IBD may become a critical health and policy issues around the world[2]. Ulcerative colitis (UC) and Crohn’s disease are the two clinical-histological variants of IBD, the former presents as continuous-type of inflammation of the colorectal or total colonic mucosa and submucosa, and the latter can involve the entire gastrointestinal tract in a discontinuous fashion, most often involving the terminal ileum, colon and perianal area. In addition to protracted course and recurrent attacks[3], up to 24% of IBD patients experience extraintestinal manifestations, such as peripheral arthritis, ankylosing spondylitis and anterior uveitis[4], which can severely impact their quality of life. Furthermore, the risk of colorectal cancer in patients with UC is 2.4 times greater than that in the general population[5]. Despite the pathogenesis of UC is still incompletely understood, abnormal innate and adaptive immune responses are pivotal factors in recurrences and exacerbation of the disease[6].
γδT17 cells are a highly heterogeneous subpopulation of T lymphocytes with surface receptors consisting of γ and δ chains and are mainly distributed in the skin, intestine and respiratory tract[7]. As one of the most abundant T-cell subsets in the intestine, γδT17 cells can interact with a variety of intestinal immune cells, and can participate in abnormal immune-inflammatory responses in the intestinal epithelium[8,9]. Relevant studies have shown elevated levels of γδ T cells in the peripheral blood and intestinal mucosa of patients with IBD[10]. However, other studies have shown a decrease in the number of circulating γδ T cells in patients with Crohn’s disease compared with the normal patients[11,12]. In spontaneous colitis in mice, a special class of γδ T cells with pro-colitis effects has been identified[13], which express two key intestinal homing integrin molecules, CD103 and α4β7, and produce interleukin-17 (IL-17) in response to tretinoin. The mechanism of action of the NLR family pyrin domain containing 3 (NLRP3) inflammasome in intestinal homeostasis has attracted much attention, and as an important part of innate defence, the NLRP3 inflammasome is involved in mediating the pathological process of UC[14]. IL-1β produced by aberrant activation of the NLRP3 inflammasome can induce γδT17 cell activation and thus promote IL-17 secretion. However, in different inflammatory microenvironments, γδT17 cells are plastic and have different functions[15]. Thus, their exact role in IBD remains controversial[16-18].
All participants were enrolled from Xiyuan Hospital, China Academy of Chinese Medical Sciences (Beijing, China) from September 2020 to December 2022. The diagnostic criteria for UC were in accordance with the 2019 American College of Gastroenterology Clinical Guidelines[19]. This clinical protocol was approved by the ethics committee of Xiyuan Hospital (No. 2019XLC003-2), and all the subjects voluntarily participated and signed an informed consent form. The quantities of cytokines such as IL-1β, IL-23, IL-21, tumor necrosis factor α (TNF-α), granulocyte-macrophage colony stimulating factor (GM-CSF), IL-22, IL-17, IL-10 and transforming growth factor β (TGF-β), were analysed with the appropriate ELISA kits. Type-17 immune response mediated by IL-17 plays a crucial role in the pathology of UC and γδT17 cells are one of the important sources of IL-17. Therefore, we measured the expression levels of IL-17 and γδT17 cells in the colonic mucosal tissues of UC patients by immunohistochemistry and immunofluorescence staining.
The study population consisted of an UC group and a normal group, and the ages of all the participants were between 18 and 70 years. Patients with mild-to-moderate UC in the active phase of the disease were eligible for inclusion (active phase: Total modified Mayo index score ≥ 2; mild to moderate defined by modified Truelove and Witts disease severity staging: Stool frequency < 4, light or no blood in the stool, normal pulse, temperature and hemoglobin, and ESR < 20 mm/hour) and rectal biopsy was necessary. Adults with no abnormal clinical manifestations, no significant organic changes on colonoscopy and who were willing to undergo colon biopsy were included in the normal group. Patients with severe heart, liver, kidney or other major organ disease; haematopoietic, neurological, psychiatric disease or combined with other organic lesions of the digestive system (e.g., peptic ulcer), or with systemic diseases affecting the dynamics of the digestive tract (e.g., hyperthyroidism or diabetes mellitus) were excluded.
This study is a clinical observational study. Blood serum and biopsy tissue samples were obtained from eligible parti
During each colonoscopy, at least two biopsies were obtained from different lesion sites. All biopsies were fixed in 10% buffered formalin and subsequently paraffin embedded, cut into 4-μm-thick sections and then stained with haematoxylin and eosin (HE).
Absolute concentrations of cytokines in blood serum were quantified with ELISA kits, including IL-21 (Lot number: 10354), IL-22 (Lot number: 10356), IL-23 (Lot number: 10357), TNF-α (Lot number: 11776), GM-CSF (Lot number: 11005), IL-17 (Lot number: 10344), IL-18 (Lot number: 10349), IL-1β (Lot number: 10351), TGF-β (Lot number: 11800), and IL-10 (Lot number: 13626), according to the manufacturer’s instructions.
Four-micrometer-thick slices of colonic tissue were deparaffinized in xylene and stained with anti-NLRP3 antibody (1:500, Lot number: 19771-1-AP), anti-apoptosis-associated speck-like protein (ASC) antibody (1:100, Lot number: ab180799), anti-caspase-1 antibody (1:100, Lot number: 22915-1-AP), anti-IL-β antibody (1:500, Lot number: ab283818), and anti-IL-17A antibody (1:200, Lot number: ab189377). For immunofluorescence, sections were incubated with in anti-T cell receptor γ/δ antibody (1:50, Lot number: TCR-1061) and anti-IL-17A antibody (1:100, Lot number: 17-7177-81).
The primary aim of this study was to measure the expression levels of γδT17 cells in intestinal mucosal tissues from UC patients. The secondary aim was to evaluate the status of the NLRP3/IL-1β pathway in intestinal mucosal tissues from UC patients.
Statistical data were analysed with GraphPad Prism 8.0 software. The calculation data were tested by χ2-test. The data are expressed as mean ± SDs. Data with normal distribution and homogeneity of variances were analysed by t-test; Mann-Whitney test was used for data without normal distribution or homogeneity of variances. P < 0.05 (two-tailed) was considered statistically significant.
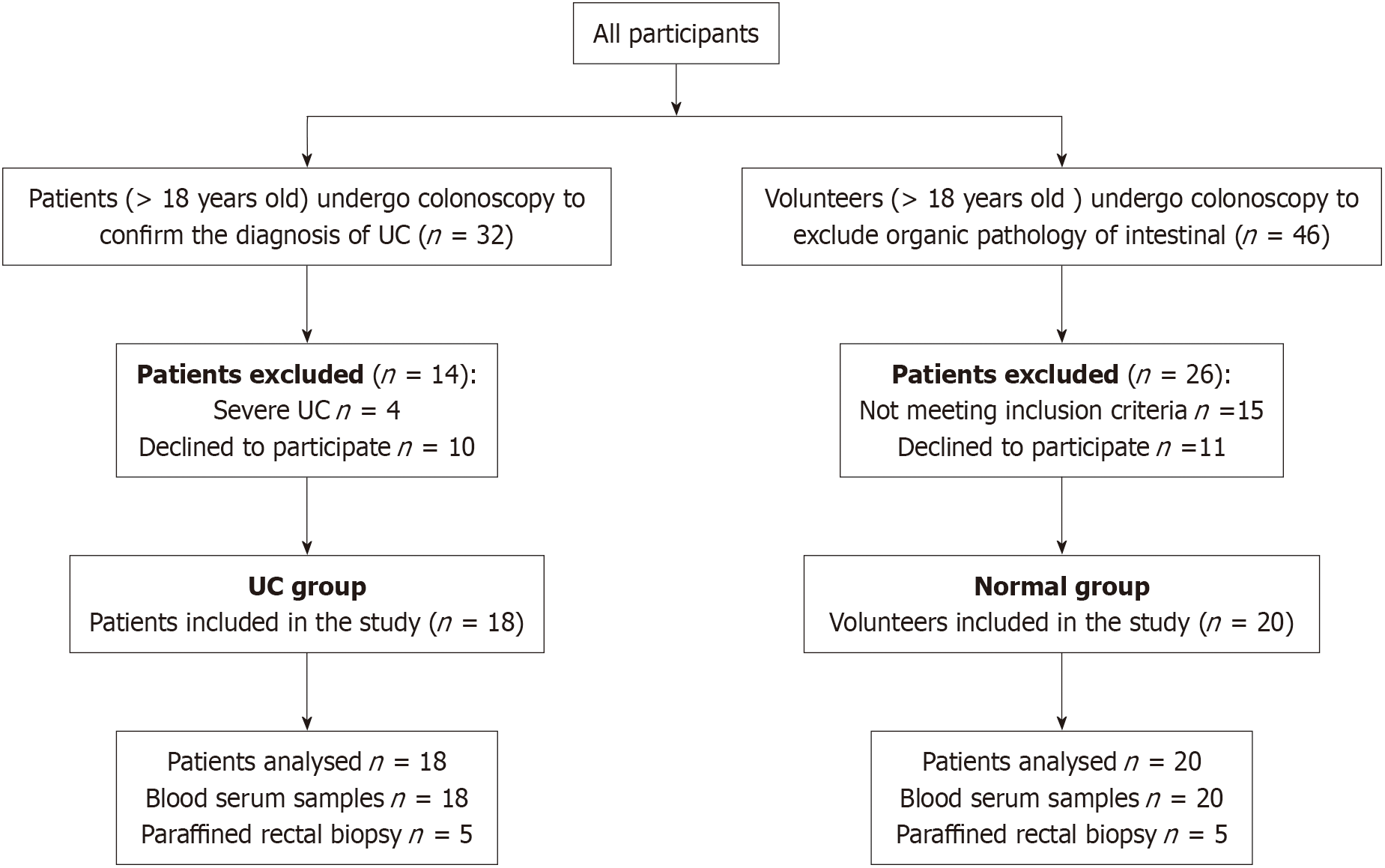
From September 2020 to December 2022, 32 patients with UC and 46 volunteers were screened. Four patients with severe UC and 10 patients who declined to participate were excluded, and volunteers who did not meet the inclusion criteria and declined to participate were also excluded, ultimately 18 patients with UC and 20 healthy volunteers completed our observational study. Figure 1 shows the screening process for the participants in this study. The clinical characteristics such as age, genders and ethnological of the participants in both groups are displayed in Table 1.
| Characteristics | UC group | Normal group | P value |
| Number | 18 | 20 | |
| Age | 42.17 ± 10.04 | 44.45 ± 10.13 | 0.49 |
| Male/female, % | 55.6/44.4 | 40/60 | 0.34 |
| Race | |||
| Han, n (%) | 17 (94.4) | 19 (95) | 0.94 |
| Other, n (%) | 1 (5.6) | 1 (5) |
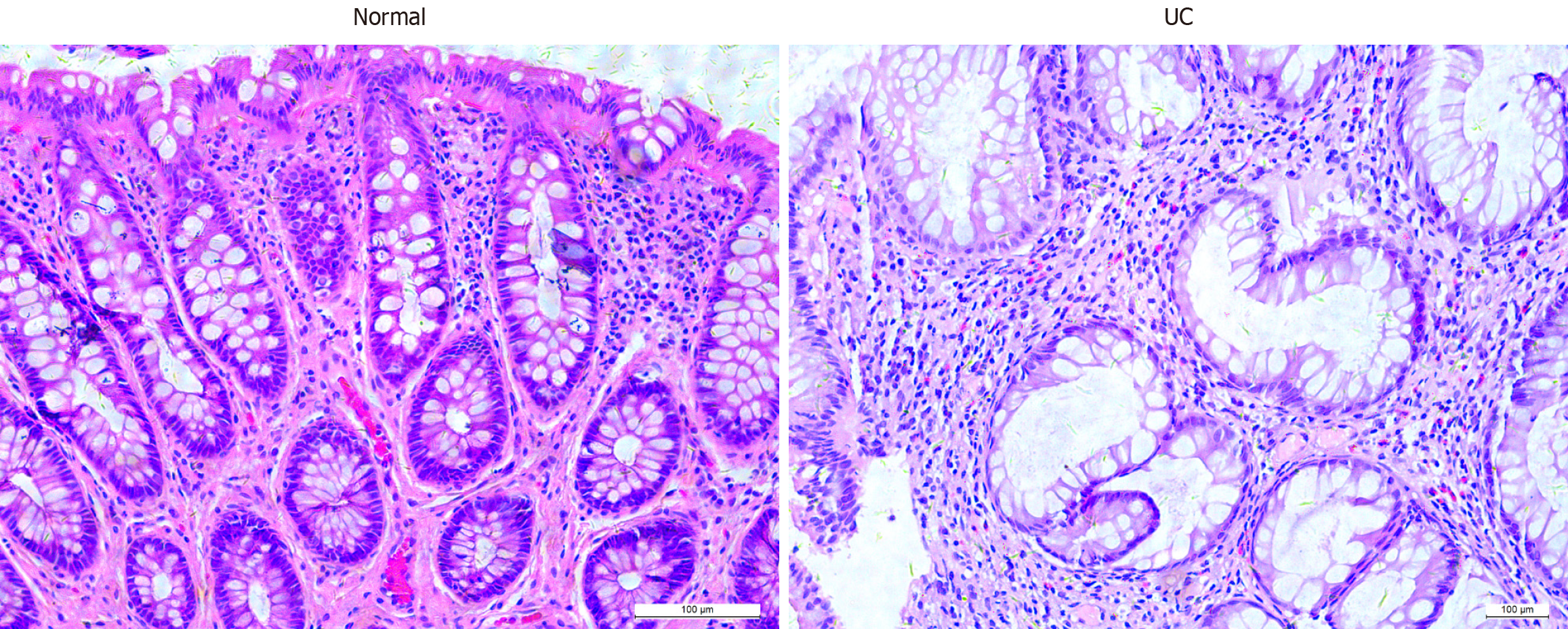
Microscopic images of HE stained sections of colonic mucosal tissues are shown in Figure 2, compared with healthy controls, patients with UC showed a decreased number of glands, deformation, distortion, branching of crypt structures, and an increase in population of inflammatory cells in the lamina propria of the colonic mucosal tissues.
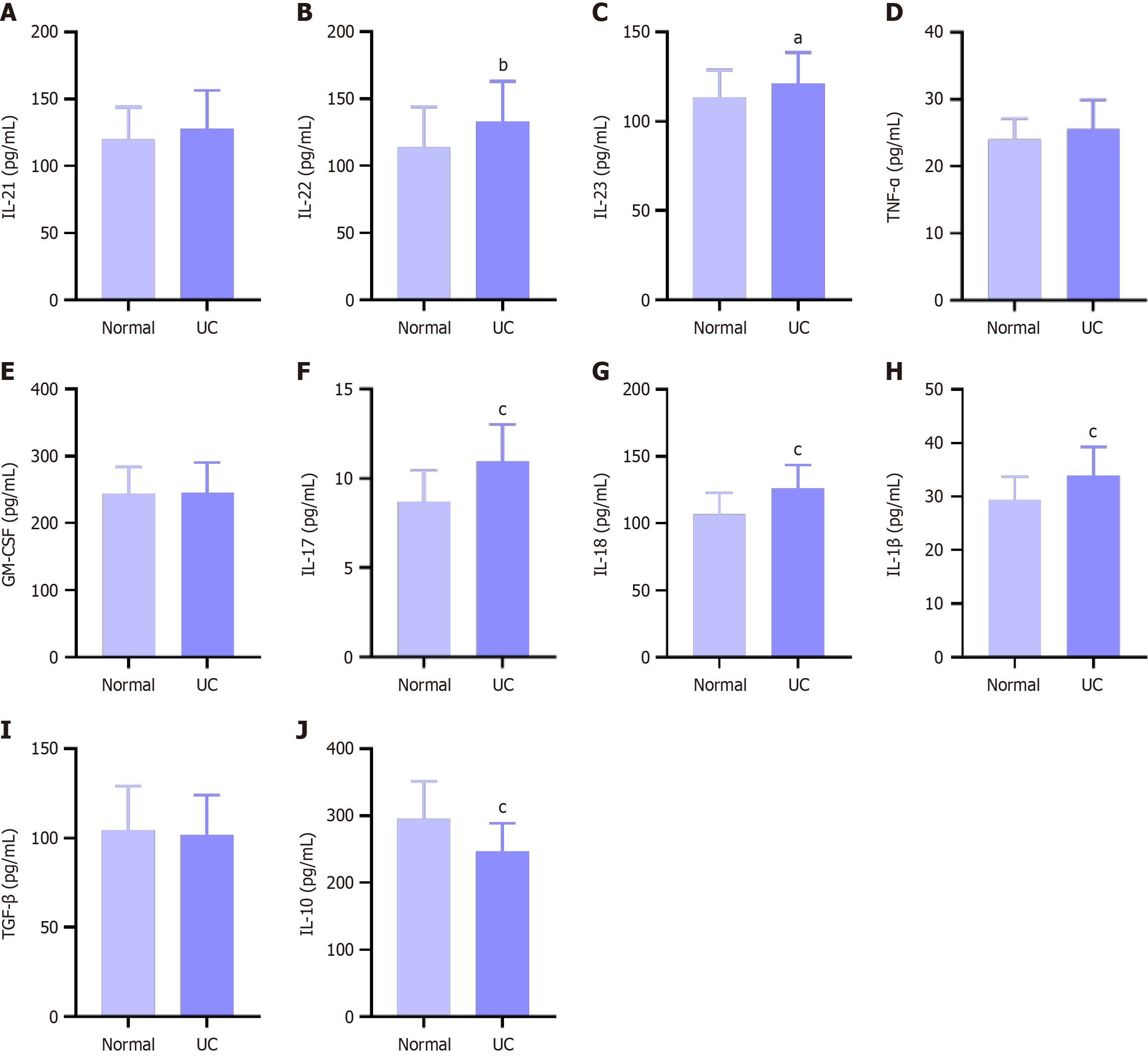
The quantities of cytokines such as IL-1β, IL-23, IL-21, TNF-α, GM-CSF, IL-22, IL-17, IL-10 and TGF-β were analysed with the appropriate ELISA kits. As shown in Figure 3, the levels of cytokines that can induce γδT17 cell activation, such as IL-1β and IL-23, were significantly elevated, and cytokines secreted by γδT17 cells such as IL-21, TNF-α, GM-CSF, IL-22 and IL-17 were elevated to different degrees, all of which indicated abnormal activation of γδT17 cells in patients with UC, while concomitant anti-inflammatory cytokines IL-10 and TGF-β were decreased.
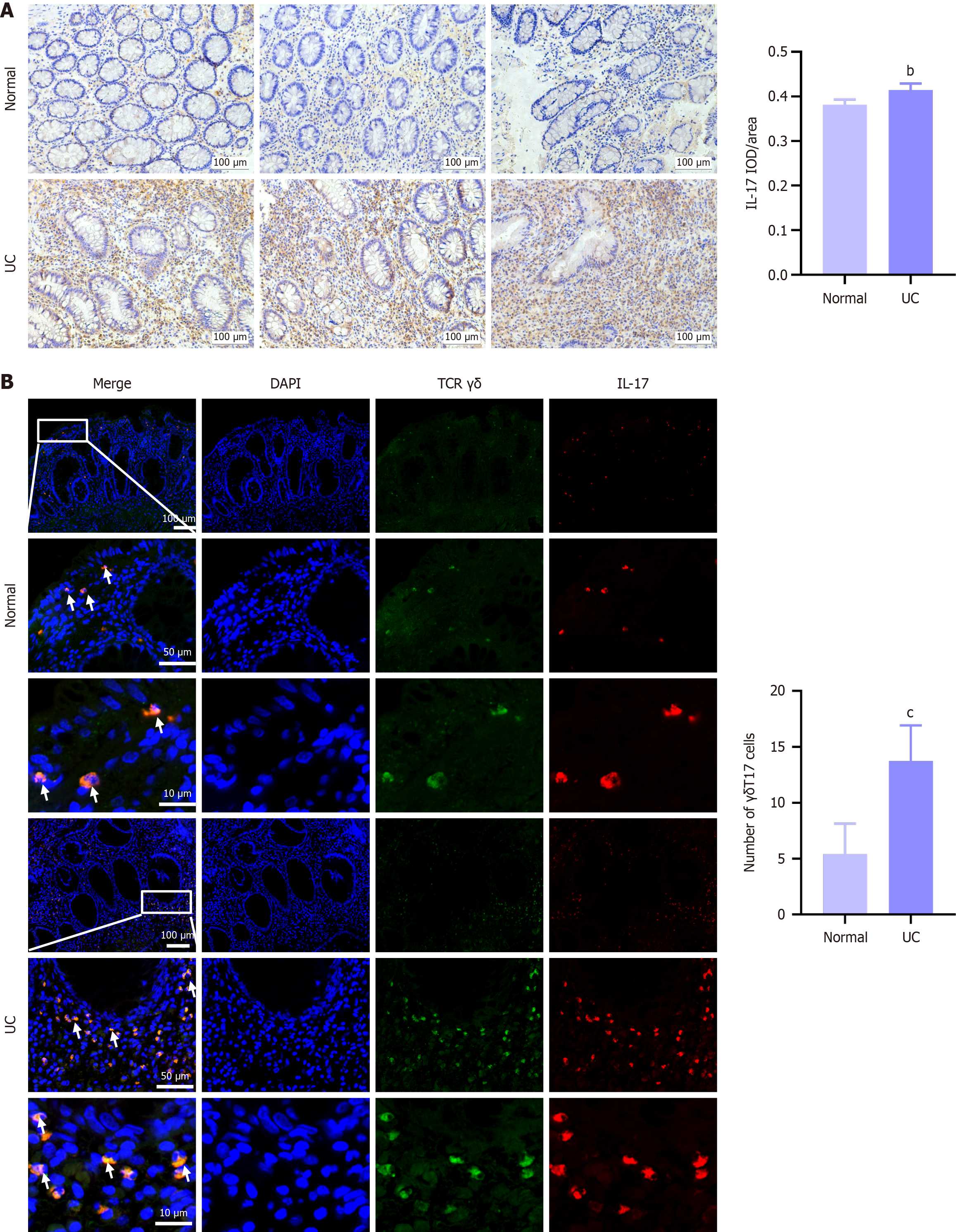
As shown in Figure 4, compared with expression levels measured in the normal group, the expression levels of IL-17 and γδT17 cells were significantly elevated in UC patients, and our results also showed γδT17 cells scattered in the epithelium and lamina propria of the colon, which indicated that the elevated IL-17 in UC patients was significantly correlated with abnormal activation of γδT17 cells.
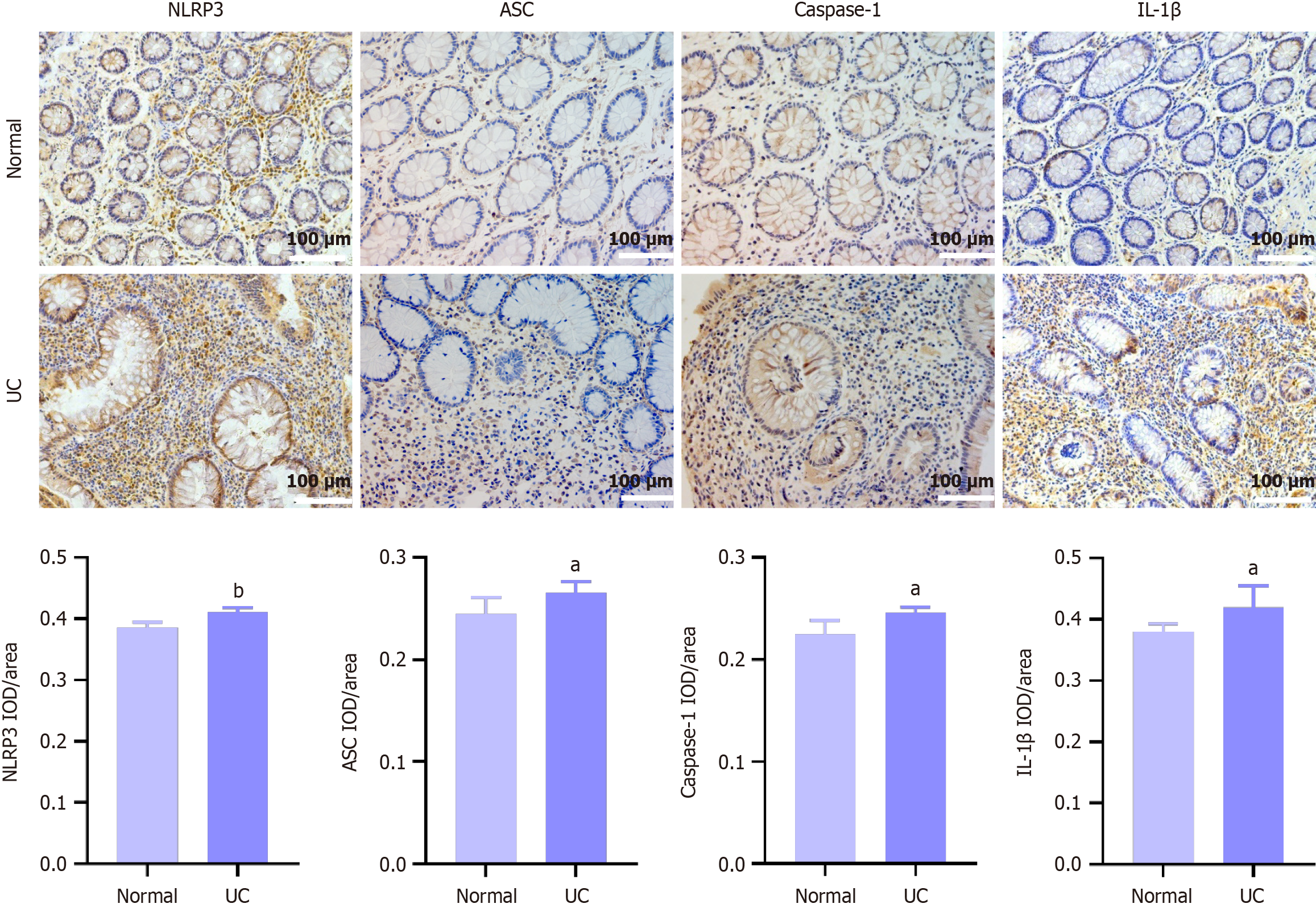
As shown in Figure 5, the expression levels of NLRP3, ASC, caspase-1 and IL-1β in colon mucosal tissue were all elevated in different degrees in UC patients compared with healthy control. The above results indicated that hyperactivation of the NLRP3 inflammasome in colonic tissues of UC patients leads to aberrant activation of γδT17 cells, which can be one of the important pathological mechanisms of UC.
UC is a lifelong inflammatory disease characterized by chronic inflammation of the colorectal mucosa and ulcer formation, with the lesions mainly involving the mucosa or submucosa[7]. IL-17 is a T-cell-derived cytokine with multiple functions such as defence against pathogenic microorganisms, recruitment of inflammatory cells, and induction of proinflammatory cytokine secretion[20]. Some studies have shown that elevated levels of IL-17 gene expression are a prominent feature in the pathogenesis of UC[21]. Our findings also showed that the expression level of IL-17 was dramatically elevated in both serum and colonic mucosal tissues from UC patients. However, what are the sources of IL-17 in UC patients? It is widely known that αβ T cells, γδ T cells, and innate lymphoid cells all have the ability to secrete IL-17, among which Th17 cells are one of the main sources of IL-17 and play a pivotal role in the pathological process of UC[22]. At present, a growing body of clinical evidence has demonstrated that the content of γδT17 cells are significantly elevated in colonic tissues form patients with IBD and colorectal tumours[23,24]. Our results showed that expression levels of γδT17 cells were significantly elevated in colonic mucosal tissues from UC patients compared to expression levels in colonic mucosal tissues from healthy volunteers, which is consistent with existing clinical research. Based on the properties of the δ chain, human γδ T cells can be classified into two main subtypes: Vδ1 cells are mainly expressed in mucosal tissues, whereas infiltrating Vδ2 T cells are mainly expressed in peripheral blood and secondary lymphoid organs, have a distinct memory effect and terminal differentiation phenotype, and produce high levels of TNF-α and IL-17, which correlate with inflammatory responses and the severity of IBD[20], and could be another point of in-depth study for our group.
The inflammatory response triggered by innate immunity is a primary host response against foreign invasion, removing pathogens and attracting adaptive immune cells to the site of injury. NLRP3 is an NLR-family protein that responds to extensive stimulation by forming inflammasomes. The role of inflammasomes is to activate caspases to produce inflammatory mediators such as IL-1β and IL-18. Studies have shown that NLRP3, IL-1β, caspase-1, and ASC expression levels are significantly elevated in colonic mucosal biopsy tissues from patients with in-remission or active IBD[25], and recent investigations have shown that nucleotide polymorphisms in the IL-18 and IL-1β genes of patients with IBD are correlated with poor response to treatment with anti-TNF-α[26]. The regulatory role of the NLRP3 inflammasome in intestinal immune-inflammatory homeostasis is receiving increasing attention, and its continuous activation leads to overproduction of IL-1β, which can bind to IL-1 receptors on the surface of γδT17 cells and promote abnormal activation, resulting in production of large amounts of IL-17 and exacerbating intestinal inflammatory responses. Therefore, we further measured the expression levels of NLRP3, ASC, caspase-1 and IL-1β in the colonic mucosal tissues of UC patients. The results of our study revealed that the expression levels of NLRP3, ASC, caspase-1, and IL-1β in patients with UC were significantly elevated, and that overexpressed IL-1β leads to abnormal activation of γδT17 cells, which exacerbates the abnormal immune-inflammatory response. The results of this simple, observational study will be used as the basis for a multicenter, randomized, double-blind, placebo-controlled study to provide high-level, evidence-based clinical findings, which can address the limitations, such as small sample sizes, regional limitations and variability in patient populations of this current study.
In conclusion, our observational study provides new ideas for clinical research by demonstrating that overactivation of the NLRP3/IL-1β signaling pathway mediated abnormal activation of γδT17 cells, which is an important pathological mechanism in UC patients (Video 1).
| 1. | Federici S, Kviatcovsky D, Valdés-Mas R, Elinav E. Microbiome-phage interactions in inflammatory bowel disease. Clin Microbiol Infect. 2023;29:682-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 41] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 2. | Kaplan GG, Windsor JW. The four epidemiological stages in the global evolution of inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2021;18:56-66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1095] [Cited by in RCA: 987] [Article Influence: 197.4] [Reference Citation Analysis (5)] |
| 3. | Zhang Y, Wu Z, Liu J, Zheng Z, Li Q, Wang H, Chen Z, Wang K. Identification of the core active structure of a Dendrobium officinale polysaccharide and its protective effect against dextran sulfate sodium-induced colitis via alleviating gut microbiota dysbiosis. Food Res Int. 2020;137:109641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 86] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 4. | Rogler G, Singh A, Kavanaugh A, Rubin DT. Extraintestinal Manifestations of Inflammatory Bowel Disease: Current Concepts, Treatment, and Implications for Disease Management. Gastroenterology. 2021;161:1118-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 722] [Cited by in RCA: 621] [Article Influence: 124.2] [Reference Citation Analysis (1)] |
| 5. | Lopez A, Pouillon L, Beaugerie L, Danese S, Peyrin-Biroulet L. Colorectal cancer prevention in patients with ulcerative colitis. Best Pract Res Clin Gastroenterol. 2018;32-33:103-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (3)] |
| 6. | Dhuppar S, Murugaiyan G. miRNA effects on gut homeostasis: therapeutic implications for inflammatory bowel disease. Trends Immunol. 2022;43:917-931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 7. | Pan X, Wang H, Zheng Z, Huang X, Yang L, Liu J, Wang K, Zhang Y. Pectic polysaccharide from Smilax china L. ameliorated ulcerative colitis by inhibiting the galectin-3/NLRP3 inflammasome pathway. Carbohydr Polym. 2022;277:118864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 62] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 8. | Ribot JC, Lopes N, Silva-Santos B. γδ T cells in tissue physiology and surveillance. Nat Rev Immunol. 2021;21:221-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 456] [Cited by in RCA: 392] [Article Influence: 78.4] [Reference Citation Analysis (0)] |
| 9. | Papotto PH, Yilmaz B, Silva-Santos B. Crosstalk between γδ T cells and the microbiota. Nat Microbiol. 2021;6:1110-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 10. | Holtmeier W, Hennemann A, May E, Duchmann R, Caspary WF. T cell receptor delta repertoire in inflamed and noninflamed colon of patients with IBD analyzed by CDR3 spectratyping. Am J Physiol Gastrointest Liver Physiol. 2002;282:G1024-G1034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 11. | McCarthy NE, Hedin CR, Sanders TJ, Amon P, Hoti I, Ayada I, Baji V, Giles EM, Wildemann M, Bashir Z, Whelan K, Sanderson I, Lindsay JO, Stagg AJ. Azathioprine therapy selectively ablates human Vδ2⁺ T cells in Crohn's disease. J Clin Invest. 2015;125:3215-3225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (3)] |
| 12. | Andreu-Ballester JC, Amigó-García V, Catalán-Serra I, Gil-Borrás R, Ballester F, Almela-Quilis A, Millan-Scheiding M, Peñarroja-Otero C. Deficit of gammadelta T lymphocytes in the peripheral blood of patients with Crohn's disease. Dig Dis Sci. 2011;56:2613-2622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 13. | Tyler CJ, McCarthy NE, Lindsay JO, Stagg AJ, Moser B, Eberl M. Antigen-Presenting Human γδ T Cells Promote Intestinal CD4(+) T Cell Expression of IL-22 and Mucosal Release of Calprotectin. J Immunol. 2017;198:3417-3425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 52] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 14. | Wagatsuma K, Nakase H. Contradictory Effects of NLRP3 Inflammasome Regulatory Mechanisms in Colitis. Int J Mol Sci. 2020;21:8145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 15. | Sun X, Cai Y, Fleming C, Tong Z, Wang Z, Ding C, Qu M, Zhang HG, Suo J, Yan J. Innate γδT17 cells play a protective role in DSS-induced colitis via recruitment of Gr-1(+)CD11b(+) myeloid suppressor cells. Oncoimmunology. 2017;6:e1313369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 16. | Heilig JS, Tonegawa S. Diversity of murine gamma genes and expression in fetal and adult T lymphocytes. Nature. 1986;322:836-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 373] [Cited by in RCA: 380] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 17. | Silva-Santos B, Mensurado S, Coffelt SB. γδ T cells: pleiotropic immune effectors with therapeutic potential in cancer. Nat Rev Cancer. 2019;19:392-404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 390] [Cited by in RCA: 352] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 18. | Di Marco Barros R, Roberts NA, Dart RJ, Vantourout P, Jandke A, Nussbaumer O, Deban L, Cipolat S, Hart R, Iannitto ML, Laing A, Spencer-Dene B, East P, Gibbons D, Irving PM, Pereira P, Steinhoff U, Hayday A. Epithelia Use Butyrophilin-like Molecules to Shape Organ-Specific γδ T Cell Compartments. Cell. 2016;167:203-218.e17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 331] [Cited by in RCA: 296] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 19. | Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD. ACG Clinical Guideline: Ulcerative Colitis in Adults. Am J Gastroenterol. 2019;114:384-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1361] [Cited by in RCA: 1174] [Article Influence: 167.7] [Reference Citation Analysis (7)] |
| 20. | Lo Presti E, Di Mitri R, Mocciaro F, Di Stefano AB, Scibetta N, Unti E, Cicero G, Pecoraro G, Conte E, Dieli F, Meraviglia S. Characterization of γδ T Cells in Intestinal Mucosa From Patients With Early-Onset or Long-Standing Inflammatory Bowel Disease and Their Correlation With Clinical Status. J Crohns Colitis. 2019;13:873-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 21. | Gu C, Wu L, Li X. IL-17 family: cytokines, receptors and signaling. Cytokine. 2013;64:477-485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 517] [Cited by in RCA: 467] [Article Influence: 35.9] [Reference Citation Analysis (0)] |
| 22. | Butera A, Di Paola M, Vitali F, De Nitto D, Covotta F, Borrini F, Pica R, De Filippo C, Cavalieri D, Giuliani A, Pronio A, Boirivant M. IL-13 mRNA Tissue Content Identifies Two Subsets of Adult Ulcerative Colitis Patients With Different Clinical and Mucosa-Associated Microbiota Profiles. J Crohns Colitis. 2020;14:369-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 23. | Neurath MF. Targeting immune cell circuits and trafficking in inflammatory bowel disease. Nat Immunol. 2019;20:970-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 622] [Cited by in RCA: 545] [Article Influence: 77.9] [Reference Citation Analysis (3)] |
| 24. | Wu P, Wu D, Ni C, Ye J, Chen W, Hu G, Wang Z, Wang C, Zhang Z, Xia W, Chen Z, Wang K, Zhang T, Xu J, Han Y, Zhang T, Wu X, Wang J, Gong W, Zheng S, Qiu F, Yan J, Huang J. γδT17 cells promote the accumulation and expansion of myeloid-derived suppressor cells in human colorectal cancer. Immunity. 2014;40:785-800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 548] [Cited by in RCA: 511] [Article Influence: 42.6] [Reference Citation Analysis (11)] |
| 25. | Ranson N, Veldhuis M, Mitchell B, Fanning S, Cook AL, Kunde D, Eri R. NLRP3-Dependent and -Independent Processing of Interleukin (IL)-1β in Active Ulcerative Colitis. Int J Mol Sci. 2018;20:57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 73] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 26. | Bank S, Julsgaard M, Abed OK, Burisch J, Broder Brodersen J, Pedersen NK, Gouliaev A, Ajan R, Nytoft Rasmussen D, Honore Grauslund C, Roug S, Galsgaard J, Sprogøe Høyer Finsen D, Lindby K; Danish IBD Genetics Working Group, Sørensen J, Larsen L, Rohr Andersen M, Brandslund I, Thomassen M, Green A, Bo Bojesen A, Bek Sørensen S, Vogel U, Andersen V. Polymorphisms in the NFkB, TNF-alpha, IL-1beta, and IL-18 pathways are associated with response to anti-TNF therapy in Danish patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2019;49:890-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/