Published online Mar 28, 2025. doi: 10.3748/wjg.v31.i12.97120
Revised: September 20, 2024
Accepted: October 25, 2024
Published online: March 28, 2025
Processing time: 306 Days and 23.7 Hours
A deficiency of γδ T cells has been described in Crohn's disease (CD).
To analyze the gene expression of interleukin 7 (IL-7) and its receptors in the tissues of patients with CD.
We studied the peripheral blood of 80 patients with CD, comparing them with a group of 80 healthy subjects. The number and apoptosis of αβ and γδ T cells in peripheral blood and the proportion of αβ and γδ T cells in the intestinal tissues of patients with CD (n = 25) were studied. The gene and protein expression of IL-7, IL-2 receptor subunit γ [cluster of differentiation 132 (CD132)], receptor α (CD127), and caspase-3 in tissues was analyzed by quantitative PCR. Serum IL-7 levels were also analyzed.
In patients with CD, a decreased number of γδ T cells and an increase in the apoptosis of CD56+ αβ and γδ T cells in peripheral blood was observed (P < 0.0001 and P < 0.01) respectively, and there was an inverse correlation among T subsets and their apoptosis. In addition, IL-7 gene expression and IL-7 protein in the tissues of these patients were increased. The titers of caspase-3 in tissues were low vs control group (P > 0.01). The percentage of CD8+γδ T cells decreased in tissues (P < 0.01), and was directly related to IL-7 levels in peripheral blood. The expression of IL-2 receptor subunit γ (CD132) was greatly decreased in the tissues of patients with CD (P < 0.05).
There may be a cause-effect relationship between the lower gene expression of the IL-2 receptor subunit γ (CD132) in tissues of patients with CD and γδ T cells immunodeficiency.
Core Tip: A deficiency in T cells, and specifically in γδ T cells, has been described in Crohn's disease (CD). Interleukin 7 (IL-7) is essential for the activation and proliferation of this lymphocyte’s subsets. This work demonstrates a decrease in IL-2 receptor subunit γ [cluster of differentiation 132 (CD132)] in tissues of patients with CD. This could be related to the deficiency of γδ T cells, and could open new works and lines of research for the application of new therapies against this disease.
- Citation: Andreu-Ballester JC, Hurtado-Marcos C, García-Ballesteros C, Pérez-Griera J, Izquierdo F, Ollero D, Jiménez A, Gil-Borrás R, Llombart-Cussac A, López-Chuliá F, Cuéllar C. Decreased gene expression of interleukin 2 receptor subunit γ (CD132) in tissues of patients with Crohn’s disease. World J Gastroenterol 2025; 31(12): 97120
- URL: https://www.wjgnet.com/1007-9327/full/v31/i12/97120.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i12.97120
The exact cause of Crohn's disease (CD) remains unknown due to a lack of knowledge about its etiology. Recent studies suggest that defects affecting innate immune response after disruption of the intestinal epithelial barrier is the primary cause in CD[1,2]. In the immune system, γδ T cells, due to their frequent intraepithelial location in the mucous mem
Interleukin 7 (IL-7) plays an important role in the normal functioning of the immune system, exerting a signal necessary for the proliferation, differentiation, and survival of B, T, natural killer (NK), and other immune cells. It exerts its biological actions by binding to its specific receptor (IL-7R), a heterodimer consisting of two different subunits, IL-7Rα [cluster of differentiation 127 (CD127)] and IL-2 receptor subunit γ (CD132)[6]. Alterations in both IL-7 and its receptors are associated with immunodeficiency and other autoimmune and inflammatory diseases, including cancer. Therefore, IL-7 was proposed as a promising therapeutic approach for many diseases[7]. IL-7 signaling is required for all γδ T cell development. In fact, IL-7R-deficient mice develop few αβ T cells and B cells, but γδ T cells are absent, demonstrating that IL-7R signaling is critical for the development of thymic and extrathymic γδ T cells[8]. IL-7 is produced by the mesenchymal stromal cells of bone marrow and epithelial cells of the thymus, lymph nodes, skin, liver, and intestines. Thus, IL-7 expression by enterocytes is sufficient for the extrathymic development of γδ T cells[9,10]. We observed that levels of IL-7 in peripheral blood of patients with CD were decreased[11]. The role of IL-7 and its receptors in tissues of patients with CD was studied by Belarif et al[12]. In this paper, the authors described that the failure of inflammatory bowel disease (IBD) treatments (CD and ulcerative colitis) is strongly associated with the overexpressed IL-7R signaling pathway of these patients, although only IL-7Rα (CD127) was analyzed[12].
Apoptosis or programmed cell death of T cells is an important mechanism to maintain peripheral immune tolerance. Observations by several laboratories showed that mucosal T cells of patients with IBD were highly resistant to apoptosis[13-18]. Due to these findings, it has been reasoned that the persistent T cell activation in the mucosa would explain that the inflammatory reaction in patients with IBD is resilient to resolution. However, apoptosis in peripheral blood lymphocytes and tissues of patients with CD was hardly studied. Similarly, there are no known studies on IL-7 in tissues and its relationship with apoptosis.
Our objective was to analyze the gene expression of IL-7 and its receptors in tissues of patients with CD, to determine the relationship between the concentration of this cytokine and the deficiency of γδ T cells.
In this prospective case-control study, we studied the peripheral blood of 160 subjects, 80 patients with CD, comparing them with 80 healthy subjects (control group). In addition, we analyzed the tissues of 25 of these patients compared to a group of 25 healthy subjects. The tissues (intestinal biopsies) were obtained from patients with CD who underwent endoscopy and surgery at the Arnau de Vilanova Hospital in Valencia, Spain. The Lennard-Jones criteria were used for the diagnosis of CD[19]. Activity was measured using the CD activity index (CDAI)[20]. Patients with CD were divided according to three clinical scenarios: “New patients” with active CD presenting at diagnosis with no prior CD treatment, or with only treatment within the previous 24 hours, “remission” (CDAI < 150 for at least 12 months), and “active disease” (CDAI > 150 and signs and symptoms of disease). Control healthy subjects were required to have the following characteristics: They were not suffering from immunodeficiency or autoimmune or inflammatory diseases, had not been vaccinated in the last 3 months, and were not undergoing immunosuppressive therapy. Tissues from healthy subjects were biopsied by routine colonoscopies program to detect colon cancer and reported as normal. Cases and controls were matched for sex and age ± 5 years. The study was approved by the Research Ethics Committee of the Arnau de Vilanova-Lliria University Hospital.
Three to five biopsies of ileum and colon were collected at 1-2 cm intervals from each subject. Some samples were fixed through immersion in a solution of 10% buffered formalin. Specimens were routinely processed by paraffin embedding. Tissue was stained by a hematoxylin-eosin staining method. Some samples were frozen without fixing for cellular and molecular analyses. Small biopsy samples of tissues were homogenized and washed in separation medium [phosphate-buffered saline (PBS), 1% fetal bovine serum (FBS), 1 mmol/L DTT, 1 mmol/L EDTA] for 15 minutes at 37 °C.
After centrifugation, the pellet was incubated in digestion medium consisting of 5% FBS, 0.5 mg/mL collagenase type VIII (Sigma-Aldrich, St. Louis, MO, United States) for 30 minutes at 37 °C by gentle shaking. Cells were passed through a mesh, centrifuged, and stained with lineage markers. Human lymphocytes were isolated from the intestinal mucosa of healthy individuals and patients with CD.
Blood cell counts were obtained using a cell counter (LH750; Beckman Coulter, Inc., Fullerton, CA, United States). An enrichment of the sample in mononuclear cells was obtained by density gradient centrifugation from EDTA anticoagulated blood sample using Lymphoprep™ (Palex Medical SA, Barcelona, Spain). After two washes in PBS, the cells obtained were resuspended in 200 mL binding buffer from the ANNEXIN V-FITC/7-AAD Kit (Beckman Coulter, Inc) in the presence of calcium.
To evaluate the functional analysis of γδ and αβ T cells - peripheral blood and intestinal tissue - we performed flow cytometry analysis with the following monoclonal antibodies: Anti-TCR PAN αβ- PE, anti-TCR PAN γδ- PE and FITC, CD19- PC7, CD56- PC7 and PE and CD4-PC7, CD3-PC5 and ECD, CD8-PC7 and FITC, CD5- FITC, CD45-ECD (Beckman Coulter). A total of 100000 events were acquired in a multiparameter Navios flow cytometer (Beckman Coulter) and later analyzed with Kaluza software.
Apoptosis detection in peripheral blood was performed with ANNEXIN V-FITC/7-AAD Kit (Beckman Coulter), based on the binding properties of Annexin V to phosphatidylserine and on the specificity of 7-amino-actinomycin D (7-AAD) for DNA guanine-cytosine base pair, following instructions of the manufacturer. The gating strategy for T cell subsets in healthy controls vs patients with CD is shown in Supplementary Figure 1.
The presence of serum IL-7 was measured by a solid phase sandwich enzyme-linked immunosorbent assay (ELISA) (Quantikine® HS ELISA; R&D Systems, Minneapolis, MN, United States). According to the product information, the detection limit of the human IL-7 was 0.1 pg/mL with an assay range for serum samples of 0.25-16 pg/mL.
The samples that were homogenized with 1 mL Trizol® Reagent (Ambion, Life Technologies, Austin, TX, United States) for RNA isolation following manufacturer’s instructions. RNA concentration was measured using a GE NanoVue Spectrophotometer (GE Healthcare Life Sciences, Coventry, England).
A reverse transcription reaction was performed from 1 mg extracted RNA using the Thermo Scientific RevertAid H Minus First Strand cDNA Synthesis kit (Thermo Fisher Scientific, Waltham, MA, United States). Resulting cDNA was amplified and subjected to quantitative PCR (qPCR) in Gene Amp 5700 (PE Biosystems, Framingham, MA, United States). The primer sequences are shown in Supplementary Table 1. The relative expression of each gene was normalized against GADPH expression, used as reference standards.
Western blotting was performed with protein extracts from intestinal biopsies. Trizol/Guanidine Protein Purification Trizol® Reagent (Ambion, Life Technologies) and guanidine hydrochloride (Sigma) following the manufacturers’ instructions, preserved in TNT buffer (20 mmol/L Tris-HCl, pH 7.5, 0.2 M NaCl, 1% Triton X-100) supplemented immediately before use with protease inhibitor cocktail tablets (10%) (Roche, Indianapolis, IN, United States). Protein concentration was determined using the Pierce BCA Protein Assay Reagent (Pierce, Rockford, IL, United States). The samples (20 µg protein) were subjected to electrophoresis on 12% polyacrylamide gels in the presence of sodium dodecyl sulfate. The proteins were transferred to a nitrocellulose membrane (Bio-Rad, Hercules, CA, United States), using the Bio-Rad Mini Protean II system in accordance with the conditions recommended by the commercial company.
The membranes were incubated with the corresponding primary antibody (anti IL-7 or anti-caspase-3) (Supplementary Table 2) in Tris-buffered saline (TBS) (20 mmol/L Tris-HCl pH 7.5 and 150 mmol/L NaCl) with 0.1% blocking agent, for 12 hours at 4 ºC. Membranes were washed to remove excess primary antibody with TBS supplemented with 0.05% Tween, and then incubated with a 1/1000 dilution of the secondary antibody (Anti-Rabbit Ig-HRP; Sigma). Membranes were again washed, visualizing the positive bands by an image digitizer with the Enhanced Chemiluminescence reagent from Amersham (Buckinghamshire, England), and were quantified by densitometry using ImageJ software.
Mann-Whitney U test was used to compare differences between means of quantitative variables. Correlation studies using Spearman’s correlation coefficient were performed to compare IL-7 gene expression, serum IL-7 levels, and T cell subsets. Data were analyzed using the Statistical Package for Social Sciences (SPSS 19.0; SPSS Inc., Chicago, IL, United States). Figures were performed using GraphPad Prism version 8.0.0 (GraphPad Software, San Diego, CA, United States).
Supplementary Table 3 shows the clinical characteristics of patients with CD (n = 80).
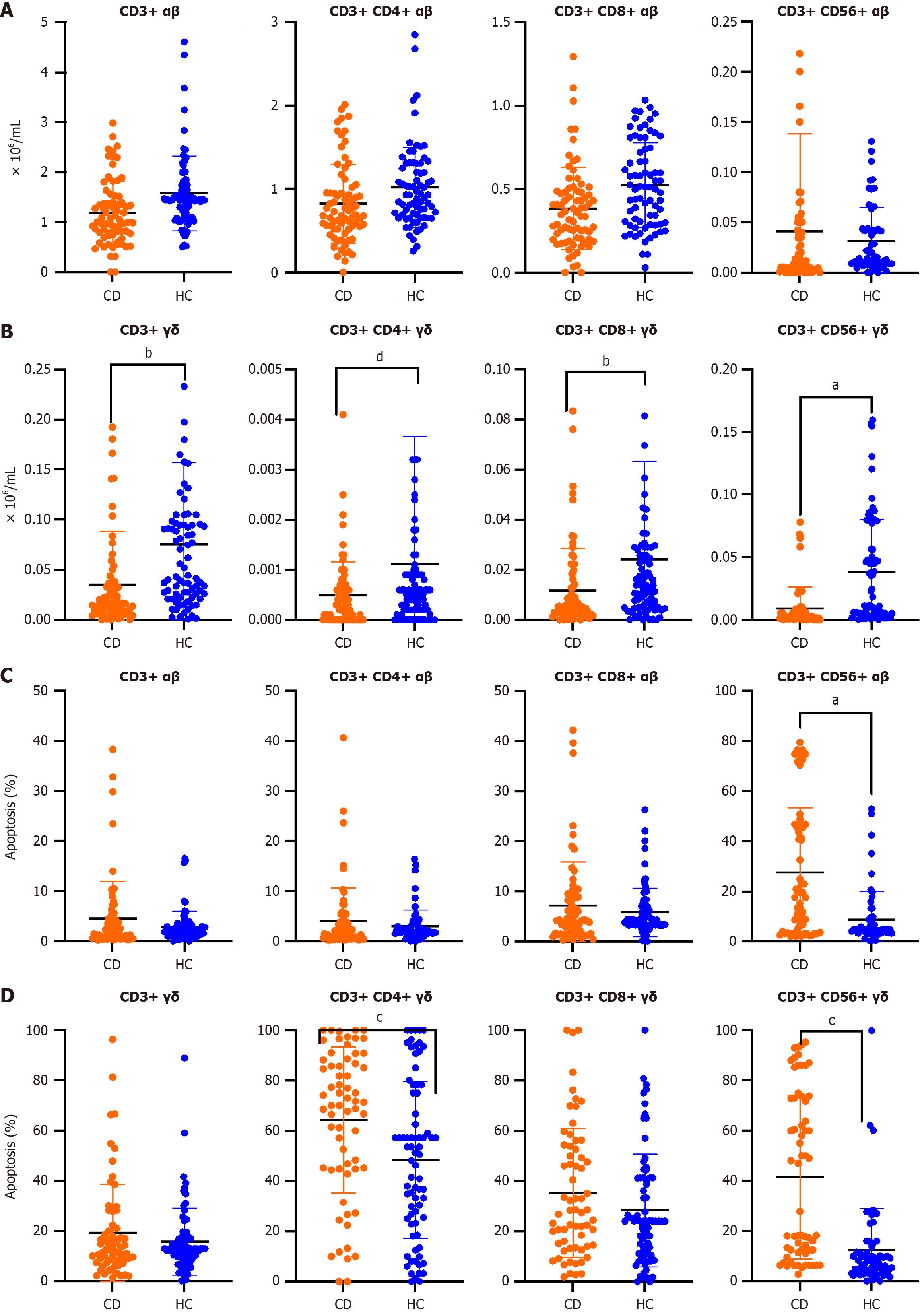
There were no differences in the number of αβ T cells in patients with CD vs healthy subjects (Figure 1A). However, a significant decrease in γδ T cells in peripheral blood of patients with CD compared to control subjects was observed (Figure 1B). Percentages of αβ and γδ T cell subsets in the peripheral blood of patients with CD vs control healthy controls are shown in Supplementary Figure 2. We did not find any differences with respect to the absolute values.
Apoptosis of CD3+ CD56+ αβ, CD3+ CD56+ γδ, and CD3+ CD4+ γδ T cells was increased in patients with CD vs healthy subjects (Figure 1C and D). According to clinical scenarios, we found no significant differences in the number or apoptosis of αβ and γδ T cell subsets, except in CD3+ CD8+ γδ T cell number in active disease (0.0057 ± 0.0056 × 106/mL) vs remission (0.0193 ± 0.01991 × 106/mL) (P = 0.008, Bonferroni test). There were also no differences among T cell subsets according to patients who were receiving treatment (n = 44, 55.0%) or were not treated at the time of the study (Supplementary Table 4).
In Supplementary Figure 3, a significant relationship between γδ T cell subsets and their apoptosis is described in healthy subjects and patients with CD. There was no significant relationship between αβ T cells and their apoptosis in patients with CD.
In patients with CD, there was an inverse correlation (Spearman’s r test) between T cell subsets and their apoptosis.
In healthy controls, there was an inverse correlation among both αβ and γδ T cell subsets and apoptosis except in CD3+CD56+ αβ T cells.
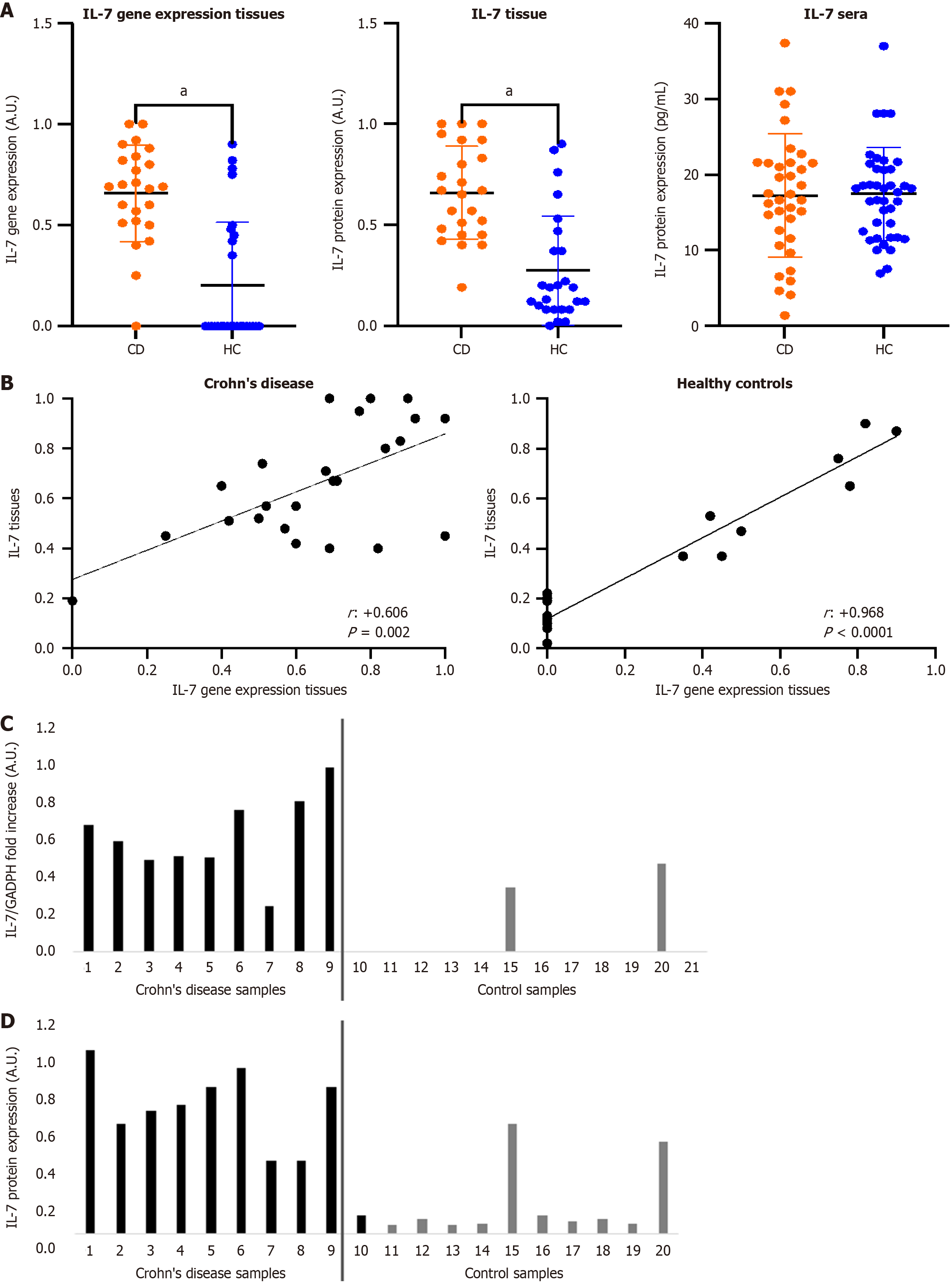
Figure 2A shows IL-7 gene expression and levels of IL-7 cytokine in tissues of patients with CD vs healthy controls. Values were significantly higher in the CD group. In addition, there was a positive correlation between IL-7 gene expression and its tissue production (Figure 2B). However, there were no significant differences in serum IL-7 levels between patients and controls. Panel C shows the IL7 gene expression and Panel D shows the IL-7 protein levels (Figure 2C and D).
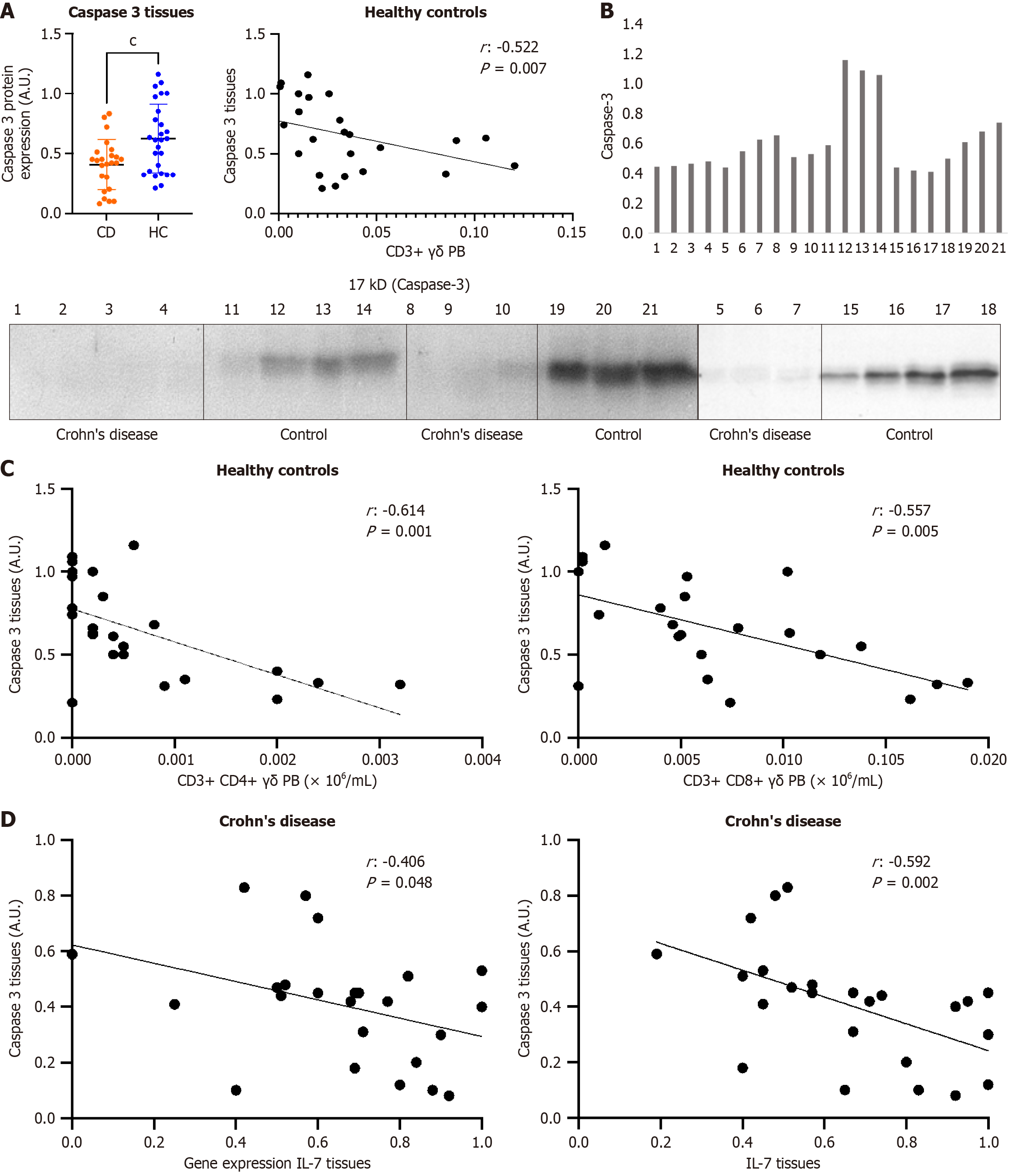
Caspase-3 was significantly less expressed in the tissues of patients with CD when compared to controls (Figure 3A and B). Negative correlations were observed among expression of caspase-3 and frequencies of γδ T cells in the healthy control group (Figure 3C). We found no relationship between caspase-3 in tissues and αβ T cells in peripheral blood in healthy controls. This relationship also did not exist in αβ and γδ T cells in patients with CD.
Moreover, caspase-3 was negatively correlated with both IL-7 cytokine levels and IL-7 gene expression in tissues from patients with CD (Figure 3D).
We did not find significant differences in serum and tissue IL-7 cytokine, IL-7 gene expression and caspase-3 according to clinical scenarios and treatments.
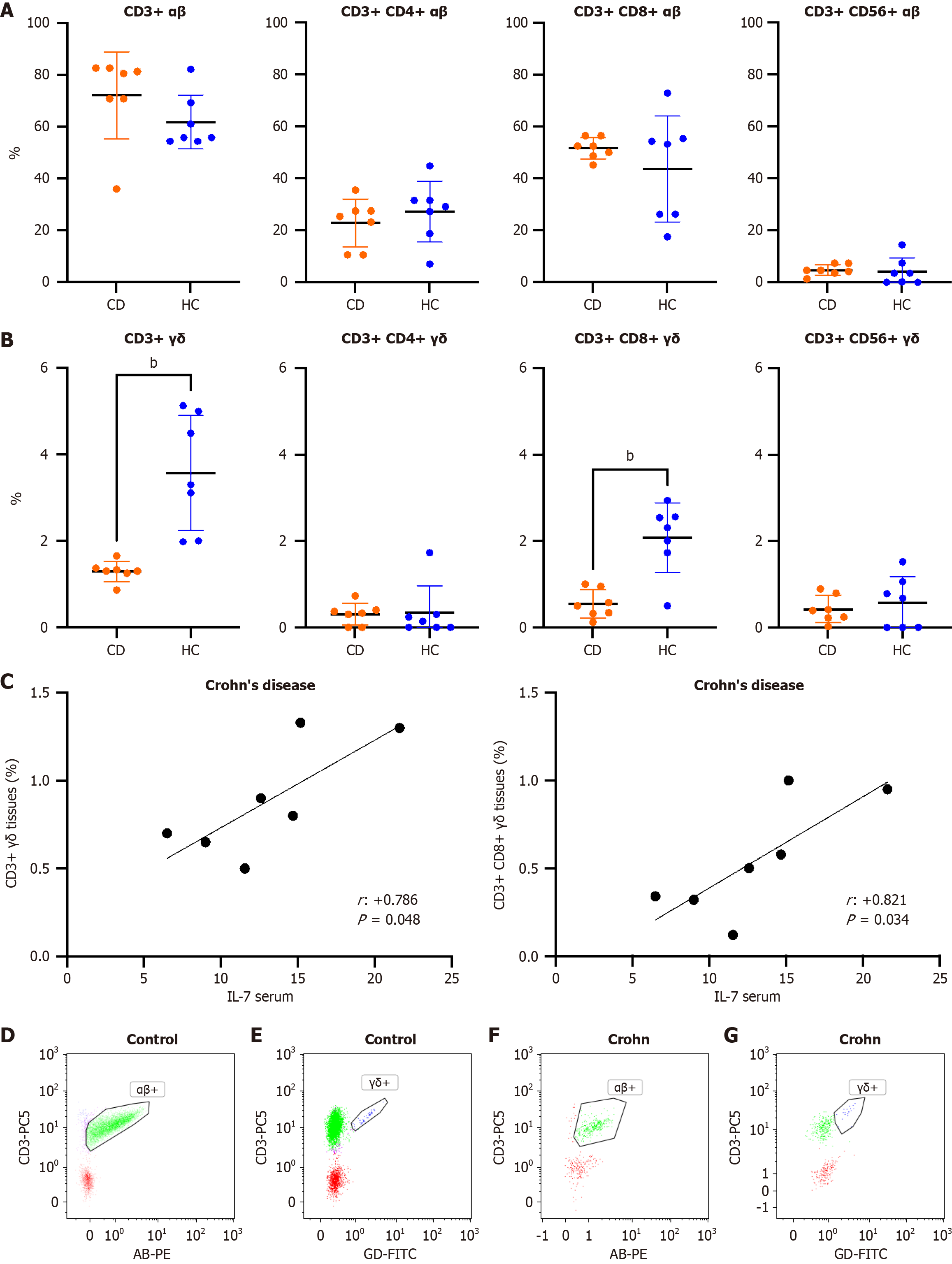
Figure 4A and B show differences between tissue αβ and γδ T cells of patients with CD vs healthy controls, respectively. CD3+ γδ and CD3+CD8+ γδ T cells were significantly reduced in the tissue of patients with CD compared to controls. Serum IL-7 levels positively correlated with CD3+ γδ and CD3+ CD8+ γδ T cells in tissue of patients with CD (Figure 4C). There were no differences in the percentages of tissue T cells between patients with and without treatment. Panels D-G show flow cytometry imaging of αβ and γδ T cells (Figure 4D-G).
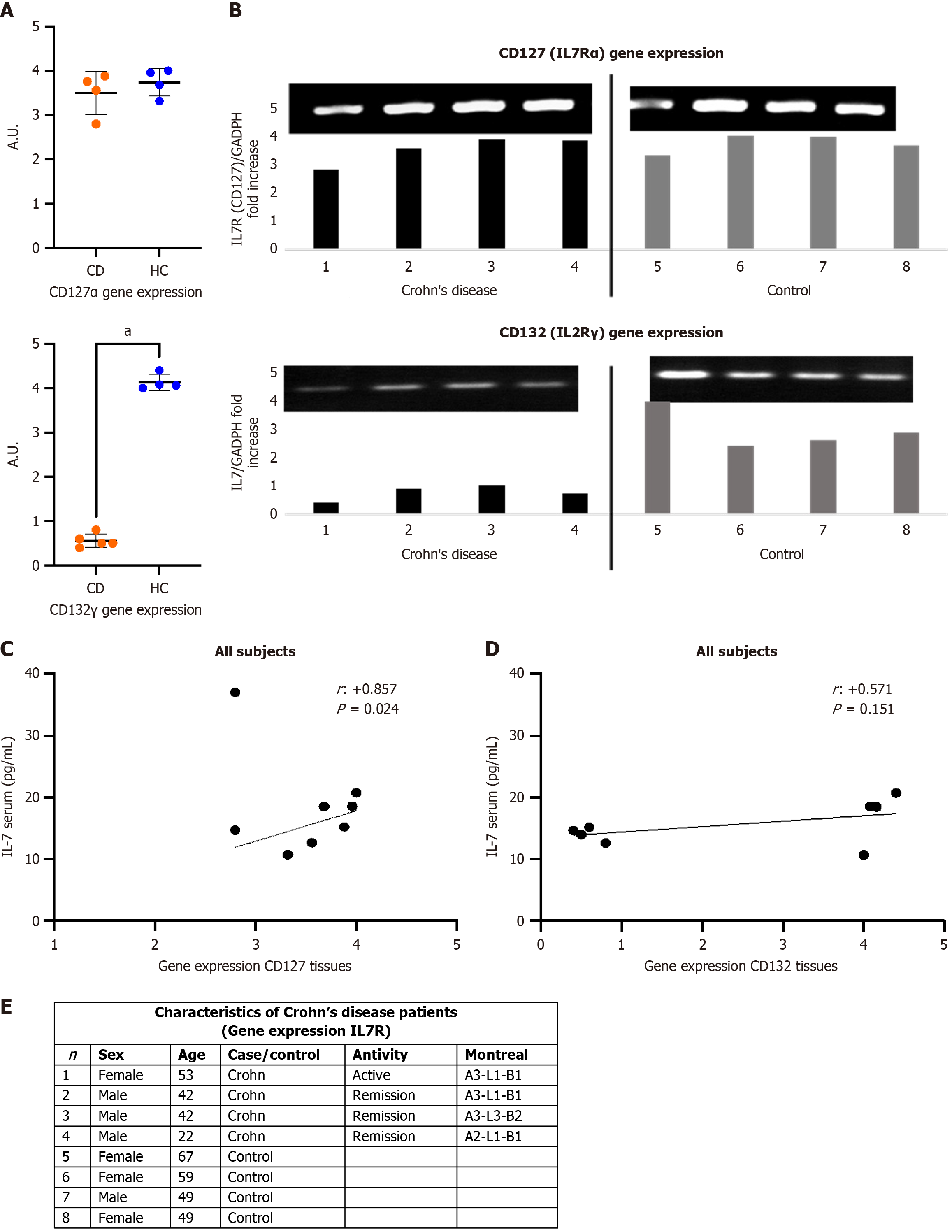
Since there was no decrease in the gene expression of IL-7 and IL-7 cytokine in the tissues of patients with CD, we analyzed the expression of their receptors. As we can see in Figure 5A and B, the expression of IL-2 receptor subunit γ (CD132) was significantly lower in tissues of patients with CD compared to the control group. There was a significant positive correlation between the gene expression of IL7Rα (CD127) and serum IL-7 of all subjects (Figure 5C). In the case of CD132, the correlation, although positive, was not significant (Figure 5D). In four patients with CD, the gene expression of IL7R was studied. Three of the four patients were in remission and one in active disease. The patient with active disease was being treated with anti-TNF-α. No statistically differences were observed in the values of CD127 and CD132 between patients (Figure 5E).
The expression of the IL-7 gene and its receptors and the possible relationship with immunity and apoptosis of conventional and non-conventional αβ and γδ T cells, respectively, were studied in intestinal tissue and peripheral blood of patients with CD. In the present study, we confirmed the previously observed deficiency of γδ T cell subsets in peripheral blood from patients with CD[4]. Likewise, we observed the lowest mean frequency in CD3+ CD56+ γδ T cells. In addition, the apoptosis of CD3+ CD56+ αβ, CD3+ CD4+ γδ and CD3+ CD56+ γδ T cells was found to be higher in the patients with CD compared with controls. CD3+ CD56+ γδ T cells, share receptors with NK cells, have high cytotoxic activity and it was proposed to name them γδ NKT cells[21]. We previously described the importance of this subpopulation in immunity against infections and tumors. It has also been shown that both a deficiency in the number of cells and an increase in their apoptosis were related to a worse prognosis of sepsis and cancer disease[22-24]. Therefore, it is not surprising that, in the present study, patients with CD had lower CD3+ CD56+ γδ (γδ NKT cells) frequencies but with a higher degree of apoptosis.
The importance of IL-7 and its relationship in activation of T cells and specifically in γδ T cells was previously indicated in the introduction section. In the present work, we observed significantly higher tissue IL-7 gene expression in patients with CD compared to healthy controls. This increase was directly correlated to the production of the IL-7 cytokine. In addition, there were lower caspase-3 levels in patients with CD that were inversely correlated to expression of both tissue IL-7 gene and IL-7 protein. IL-7 prevented the spontaneous apoptosis of γδ intestinal intraepithelial lymphocytes in a murine model, although caspase activity was only slightly inhibited. This paper suggested that IL-7, stimulated by some unknown factor, could maintain enough γδ T cells by preventing their apoptosis[25]. These facts are in accordance with our results because the increase in the protein levels and gene expression of IL-7 together with the reduction of caspase-3 in the tissues of patients with CD suggests an attempt to maintain the number of γδ T cells. It has been observed that thymic stromal lymphopoietin may down-regulate caspase-3 expression through activation of the STAT3 pathway, thereby suppressing the apoptosis of γδ T cells[26].
However, this situation is not achieved, generating an immunosuppression (deficiency of γδ T cells) at mucosa-associated lymphoid tissue level that can be used by opportunistic pathogens that try to infect through the mucous membranes.
However, we observed discrepancies between tissue and serum levels of IL-7. The tissue levels of IL-7 increased to try to compensate for the γδ T cell deficit in patients with CD. However, serum IL-7 levels were similar in patients as compared to healthy controls. This lack of increase in serum IL-7 Levels could be explained by a problem in the autocrine/paracrine effects of this cytokine. The cytokine may be being produced in large amounts but is being consumed by binding to the receptor, hence the importance of analyzing the IL-7 receptor.
The common gamma chain (γc or CD132 or IL-2 receptor subunit gamma or IL-2RG) integrates the IL-7 receptor heterodimer together with IL-7 receptor α (CD127). CD132 is a cytokine receptor subunit that, in addition to IL-7, is common to the other IL receptors: IL-2, IL-4, IL-9, IL-15 and IL-21. This receptor, expressed on most lymphocyte subsets, contributes to the proper functioning of the immune system, regulating development, proliferation, survival, and differentiation of immune cells[27]. Mutations in the gene encoding CD132 cause X-linked severe combined immunodeficiency in humans. Mechanisms of signaling and gene regulation of the related cytokines, provide information for immunodeficiency, autoimmunity, allergic diseases, and cancer[28]. Mutations in genes encoding cytokine receptors have been implicated in IBD, such as polymorphisms in IL-2, IL-21, IL-2RA, andIL-15RA[29]. According to the results shown in this study, the expression of the IL-2 receptor subunit γ (CD132) is greatly decreased in tissues of patients with CD. Gene expression levels of the IL-7 receptor α (CD127) were similar for controls and patients with CD. It is necessary to highlight that the integrity of the two subunits is necessary for the correct receptor functioning.
Therefore, we propose that significant increases in IL-7 gene expression, which also produces an increase of IL-7 production, and the low expression of caspase-3, which can lead a decrease in apoptosis, is insufficient to maintain homeostatic levels of γδ T cells in peripheral blood and tissues of patients with CD. It seems that a marked specific downregulation of the CD132 gene would inhibit the right effect of IL-7, leading to an immunodeficiency of γδ T cells that would favor superinfections or lack of elimination of some factors. This would allow the proliferation of etiological agents with consequent chronic inflammation. In a previous study, we described the relationship of an opportunistic fungus (microsporidia) with CD[30]. Recently, the destructive effects of microsporidium infection on epithelial barriers were demonstrated in a murine model by means of an increase of epithelium permeability and dysregulation of intestinal microbiota[31]. Based on the above evidence, we suggest that the infection by microsporidium, (either directly the pathogen or some protein thereof), could interferes with the expression of the receptor (CD132) as a mechanism to evade the host innate and adaptive immune responses.
There is an immunodeficiency of CD56+ αβ and γδ T cells in peripheral blood and tissues of patients with CD with increased apoptosis in peripheral blood. IL-7 gene expression and IL-7 protein levels in tissues of patients with CD were increased while the levels of caspase-3 in tissues were low. The increase in IL-7 and a lower expression of caspase-3 suggests a failure in the attempt to recover normal values of γδ T cells. The expression of IL-2 receptor subunit γ (CD132) is greatly decreased in the tissues of patients with CD. The lower gene expression of CD132 may be the cause of the decrease of γδ T cells. Further research would be necessary to determine the cause-effect of this correlation.
| 1. | Coulombe F, Behr MA. Crohn's disease as an immune deficiency? Lancet. 2009;374:769-770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 2. | Torres J, Mehandru S, Colombel JF, Peyrin-Biroulet L. Crohn's disease. Lancet. 2017;389:1741-1755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2287] [Cited by in RCA: 2054] [Article Influence: 228.2] [Reference Citation Analysis (4)] |
| 3. | Adams EJ, Gu S, Luoma AM. Human gamma delta T cells: Evolution and ligand recognition. Cell Immunol. 2015;296:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 171] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 4. | Andreu-Ballester JC, Amigó-García V, Catalán-Serra I, Gil-Borrás R, Ballester F, Almela-Quilis A, Millan-Scheiding M, Peñarroja-Otero C. Deficit of gammadelta T lymphocytes in the peripheral blood of patients with Crohn's disease. Dig Dis Sci. 2011;56:2613-2622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 5. | Chen R, Li C, Zheng J, Fan Z, Li L, Chen M, Chen B, Zhang S. Lymphocyte subsets for predicting inflammatory bowel disease progression and treatment response: a systematic review. Front Immunol. 2024;15:1403420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 6. | Rochman Y, Spolski R, Leonard WJ. New insights into the regulation of T cells by gamma(c) family cytokines. Nat Rev Immunol. 2009;9:480-490. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 889] [Cited by in RCA: 843] [Article Influence: 49.6] [Reference Citation Analysis (0)] |
| 7. | Chen D, Tang TX, Deng H, Yang XP, Tang ZH. Interleukin-7 Biology and Its Effects on Immune Cells: Mediator of Generation, Differentiation, Survival, and Homeostasis. Front Immunol. 2021;12:747324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 188] [Article Influence: 37.6] [Reference Citation Analysis (2)] |
| 8. | Rodríguez-Caparrós A, Tani-Ichi S, Casal Á, López-Ros J, Suñé C, Ikuta K, Hernández-Munain C. Interleukin-7 receptor signaling is crucial for enhancer-dependent TCRδ germline transcription mediated through STAT5 recruitment. Front Immunol. 2022;13:943510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Li GQ, Xia J, Zeng W, Luo W, Liu L, Zeng X, Cao D. The intestinal γδ T cells: functions in the gut and in the distant organs. Front Immunol. 2023;14:1206299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 31] [Reference Citation Analysis (0)] |
| 10. | Laky K, Lefrançois L, Lingenheld EG, Ishikawa H, Lewis JM, Olson S, Suzuki K, Tigelaar RE, Puddington L. Enterocyte expression of interleukin 7 induces development of gammadelta T cells and Peyer's patches. J Exp Med. 2000;191:1569-1580. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 115] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 11. | Andreu-Ballester JC, Pérez-Griera J, Garcia-Ballesteros C, Amigo V, Catalán-Serra I, Monforte-Albalat A, Bixquert-Jiménez M, Ballester F. Deficit of interleukin-7 in serum of patients with Crohn's disease. Inflamm Bowel Dis. 2013;19:E30-E31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Belarif L, Danger R, Kermarrec L, Nerrière-Daguin V, Pengam S, Durand T, Mary C, Kerdreux E, Gauttier V, Kucik A, Thepenier V, Martin JC, Chang C, Rahman A, Guen NS, Braudeau C, Abidi A, David G, Malard F, Takoudju C, Martinet B, Gérard N, Neveu I, Neunlist M, Coron E, MacDonald TT, Desreumaux P, Mai HL, Le Bas-Bernardet S, Mosnier JF, Merad M, Josien R, Brouard S, Soulillou JP, Blancho G, Bourreille A, Naveilhan P, Vanhove B, Poirier N. IL-7 receptor influences anti-TNF responsiveness and T cell gut homing in inflammatory bowel disease. J Clin Invest. 2019;129:1910-1925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 106] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 13. | Santaolalla R, Mañé J, Pedrosa E, Lorén V, Fernández-Bañares F, Mallolas J, Carrasco A, Salas A, Rosinach M, Forné M, Espinós JC, Loras C, Donovan M, Puig P, Mañosa M, Gassull MA, Viver JM, Esteve M. Apoptosis resistance of mucosal lymphocytes and IL-10 deficiency in patients with steroid-refractory Crohn's disease. Inflamm Bowel Dis. 2011;17:1490-1500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | de Souza HS, West GA, Rebert N, de la Motte C, Drazba J, Fiocchi C. Increased levels of survivin, via association with heat shock protein 90, in mucosal T cells from patients with Crohn's disease. Gastroenterology. 2012;143:1017-26.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Atreya R, Mudter J, Finotto S, Müllberg J, Jostock T, Wirtz S, Schütz M, Bartsch B, Holtmann M, Becker C, Strand D, Czaja J, Schlaak JF, Lehr HA, Autschbach F, Schürmann G, Nishimoto N, Yoshizaki K, Ito H, Kishimoto T, Galle PR, Rose-John S, Neurath MF. Blockade of interleukin 6 trans signaling suppresses T-cell resistance against apoptosis in chronic intestinal inflammation: evidence in crohn disease and experimental colitis in vivo. Nat Med. 2000;6:583-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1053] [Cited by in RCA: 998] [Article Influence: 38.4] [Reference Citation Analysis (3)] |
| 16. | Itoh J, de La Motte C, Strong SA, Levine AD, Fiocchi C. Decreased Bax expression by mucosal T cells favours resistance to apoptosis in Crohn's disease. Gut. 2001;49:35-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 93] [Article Influence: 3.7] [Reference Citation Analysis (4)] |
| 17. | Sturm A, Leite AZ, Danese S, Krivacic KA, West GA, Mohr S, Jacobberger JW, Fiocchi C. Divergent cell cycle kinetics underlie the distinct functional capacity of mucosal T cells in Crohn's disease and ulcerative colitis. Gut. 2004;53:1624-1631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 64] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 18. | Doering J, Begue B, Lentze MJ, Rieux-Laucat F, Goulet O, Schmitz J, Cerf-Bensussan N, Ruemmele FM. Induction of T lymphocyte apoptosis by sulphasalazine in patients with Crohn's disease. Gut. 2004;53:1632-1638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 72] [Article Influence: 3.3] [Reference Citation Analysis (4)] |
| 19. | Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, Hayee B, Lomer MCE, Parkes GC, Selinger C, Barrett KJ, Davies RJ, Bennett C, Gittens S, Dunlop MG, Faiz O, Fraser A, Garrick V, Johnston PD, Parkes M, Sanderson J, Terry H; IBD guidelines eDelphi consensus group, Gaya DR, Iqbal TH, Taylor SA, Smith M, Brookes M, Hansen R, Hawthorne AB. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:s1-s106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1995] [Cited by in RCA: 1775] [Article Influence: 253.6] [Reference Citation Analysis (8)] |
| 20. | Kishi M, Hirai F, Takatsu N, Hisabe T, Takada Y, Beppu T, Takeuchi K, Naganuma M, Ohtsuka K, Watanabe K, Matsumoto T, Esaki M, Koganei K, Sugita A, Hata K, Futami K, Ajioka Y, Tanabe H, Iwashita A, Shimizu H, Arai K, Suzuki Y, Hisamatsu T. A review on the current status and definitions of activity indices in inflammatory bowel disease: how to use indices for precise evaluation. J Gastroenterol. 2022;57:246-266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 49] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 21. | Matsuo Y, Tsujimura T, Drexler HG. Proposal for the designation of the natural killer antigens-positive γδ T-cell subset as γδ NKT-cells: nomenclature based on immunoprofile. Hum Cell. 2021;34:1278-1279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Liao XL, Feng T, Zhang JQ, Cao X, Wu QH, Xie ZC, Kang Y, Li H. Phenotypic Changes and Impaired Function of Peripheral γδ T Cells in Patients With Sepsis. Shock. 2017;48:321-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 23. | Lafont V, Sanchez F, Laprevotte E, Michaud HA, Gros L, Eliaou JF, Bonnefoy N. Plasticity of γδ T Cells: Impact on the Anti-Tumor Response. Front Immunol. 2014;5:622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 24. | Hu Y, Chen D, Hong M, Liu J, Li Y, Hao J, Lu L, Yin Z, Wu Y. Apoptosis, Pyroptosis, and Ferroptosis Conspiringly Induce Immunosuppressive Hepatocellular Carcinoma Microenvironment and γδ T-Cell Imbalance. Front Immunol. 2022;13:845974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 25. | Yada S, Nukina H, Kishihara K, Takamura N, Yoshida H, Inagaki-Ohara K, Nomoto K, Lin T. IL-7 prevents both caspase-dependent and -independent pathways that lead to the spontaneous apoptosis of i-IEL. Cell Immunol. 2001;208:88-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Duan J, Jiang XP, Li MQ, Fan DX, Wang Y, Li DJ, Jin LP. Thymic stromal lymphopoietin suppresses the apoptosis of decidual gamma-delta T cells via regulation of the signal transduction and activation of transcription 3/caspase-3 signaling pathway. Am J Reprod Immunol. 2013;70:464-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Barata JT, Durum SK, Seddon B. Flip the coin: IL-7 and IL-7R in health and disease. Nat Immunol. 2019;20:1584-1593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 276] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 28. | Leonard WJ, Lin JX, O'Shea JJ. The γ(c) Family of Cytokines: Basic Biology to Therapeutic Ramifications. Immunity. 2019;50:832-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 303] [Article Influence: 43.3] [Reference Citation Analysis (2)] |
| 29. | Huang H, Fang M, Jostins L, Umićević Mirkov M, Boucher G, Anderson CA, Andersen V, Cleynen I, Cortes A, Crins F, D'Amato M, Deffontaine V, Dmitrieva J, Docampo E, Elansary M, Farh KK, Franke A, Gori AS, Goyette P, Halfvarson J, Haritunians T, Knight J, Lawrance IC, Lees CW, Louis E, Mariman R, Meuwissen T, Mni M, Momozawa Y, Parkes M, Spain SL, Théâtre E, Trynka G, Satsangi J, van Sommeren S, Vermeire S, Xavier RJ; International Inflammatory Bowel Disease Genetics Consortium, Weersma RK, Duerr RH, Mathew CG, Rioux JD, McGovern DPB, Cho JH, Georges M, Daly MJ, Barrett JC. Fine-mapping inflammatory bowel disease loci to single-variant resolution. Nature. 2017;547:173-178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 564] [Cited by in RCA: 457] [Article Influence: 50.8] [Reference Citation Analysis (4)] |
| 30. | Andreu-Ballester JC, Garcia-Ballesteros C, Amigo V, Ballester F, Gil-Borrás R, Catalán-Serra I, Magnet A, Fenoy S, del Aguila C, Ferrando-Marco J, Cuéllar C. Microsporidia and its relation to Crohn's disease. A retrospective study. PLoS One. 2013;8:e62107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 31. | Jin J, Tang Y, Cao L, Wang X, Chen Y, An G, Zhang H, Pan G, Bao J, Zhou Z. Microsporidia persistence in host impairs epithelial barriers and increases chances of inflammatory bowel disease. Microbiol Spectr. 2024;12:e0361023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/