Published online Mar 28, 2025. doi: 10.3748/wjg.v31.i12.103952
Revised: January 26, 2025
Accepted: March 3, 2025
Published online: March 28, 2025
Processing time: 111 Days and 16.2 Hours
Acute liver failure (ALF) is a loss of liver function due to a severe hepatic insult. Studies utilizing the azoxymethane (AOM) mouse model of ALF, which also generates hepatic encephalopathy, have primarily focused on development of neurological deficits. However, the molecular processes that generate liver damage have not been fully characterized. Therefore, a more comprehensive characterization of the hepatic consequences of AOM toxicity is needed to better understand this disease model.
To identify molecular pathology contributing to hepatic injury during the progression of AOM-induced ALF.
C57BL/6 mice were injected with AOM to produce ALF and hepatic encephalopathy. Tissue was collected at defined stages of neurological decline up to coma. Liver injury, CYP2E1 expression, oxidative stress, inflammation, apoptosis, necroptosis, and hepatocellular senescence were assessed.
Increased hepatic necrosis and exacerbated liver injury were observed after AOM injection as mice progressed towards coma. CYP2E1 expression decreased in AOM-treated mice as liver injury progressed. Malondialdehyde, myeloperoxidase and other measures of oxidative stress were significantly increased during AOM-induced ALF. Hepatic CCL2 and tumor necrosis factor α expression increased as AOM-induced liver injury progressed. Mixed lineage kinase domain-like protein phosphorylation was increased early during the progression of AOM-induced liver injury. Measures of apoptosis and cellular senescence all increased as the time course of AOM progressed.
These data support that necrosis, oxidative stress, inflammation, apoptosis, and senescence were elevated in AOM-treated mice, with inflammation being the earliest significant change.
Core Tip: The azoxymethane (AOM) model of liver injury is used as a toxin-induced model to study acute liver failure and the development of hepatic encephalopathy (HE). After initial characterization of the liver injury in this model, little research has been performed to understand the specific cellular and molecular mechanisms of hepatic injury, with most studies focusing on neurological changes associated with HE. Here, we identify that the AOM model is associated with hepatic inflammation and oxidative stress that progresses to apoptosis, necrosis and hepatocellular senescence. These findings provide additional insight into this model and will help determine which components of hepatic pathology to investigate when therapeutic approaches are employed.
- Citation: Bhattarai SM, Jhawer A, Frampton G, Troyanovskaya E, DeMorrow S, McMillin M. Characterization of hepatic pathology during azoxymethane-induced acute liver failure. World J Gastroenterol 2025; 31(12): 103952
- URL: https://www.wjgnet.com/1007-9327/full/v31/i12/103952.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i12.103952
Acute liver failure (ALF) results from a rapid loss of liver function caused by toxins, viral infections or other injuries that directly impact the liver. In Western Europe and North America, acetaminophen-induced toxicity is the primary cause of ALF[1]. However, drug-induced liver injury (DILI) from other medications is the second most common cause of ALF in the United States as reported by the ALF Study Group Registry[2]. Morbidity and mortality are lower in patients with ALF secondary to acetaminophen toxicity compared to other etiologies due to the availability of N-acetyl cysteine which can function as an antidote if used prior to overt liver injury[3]. In patients with DILI from medications other than ace
Azoxymethane (AOM) is a chemical found in cycad palm nuts from Guam and is both hepatotoxic and carcinogenic[5,6]. Matkowskyj et al[7] identified that intraperitoneal injection of AOM into mice at doses of 50-200 mg/kg was able to induce ALF and hepatic encephalopathy. This study focused primarily on assessing liver injury through elevated markers such as serum transaminases and histologic evaluation of the liver without investigating the cellular processes and molecular mechanisms underlying the development of ALF pathology. Over 20 years later, the AOM model is recommended by the International Society of Hepatic Encephalopathy and Nitrogen Metabolism as a model of toxin-induced ALF and hepatic encephalopathy in mice[8]. While investigators have characterized aspects of neurological dysfunction in this model, there has been limited progress in elucidating how hepatic cellular and molecular pathology develops during AOM-induced ALF[9,10]. Defining and characterizing the underlying liver pathology is essential in this mouse model to further understand the pathogenesis of the neurological complications observed.
Mechanisms of injury in other hepatotoxic models have expanded our understanding of hepatic pathology during ALF. Acetaminophen-induced hepatotoxicity studies have identified that centrilobular necrosis is driven by the accumulation of toxic metabolites which form protein adducts and generate oxidative stress that induces JNK-mediated signaling and cell death[11]. In addition, liver regeneration following acetaminophen-induced liver injury is impaired by an increase in cellular senescence driven by transforming growth factor β1 (TGFβ1) signaling[12]. In the lipopolysaccharide/D-galactosamine model of ALF, inflammation plays a critical role in pathogenesis. Inhibition of tumor necrosis factor α (TNFα) signaling was shown to alleviate liver injury, significantly reduce serum transaminases and decrease M1 macrophage accumulation in the liver[13]. The distinct pathological mechanisms observed in these hepatotoxin models support that ALF pathogenesis is multifactorial.
In the AOM mouse model of ALF, high AOM concentrations lead to hepatocyte necrosis that progresses to liver failure and hepatic coma, typically within 24-48 hours[7]. In the colon, AOM has been shown to disrupt mitochondria function as it reduces mitochondria enzyme levels, such as alpha ketoglutarate[14]. Therefore, there is support that inflammation, oxidative stress, cellular senescence and multiple mechanisms of cell death could contribute to hepatic pathology during AOM-induced ALF. However, the molecular pathology and cell signaling pathways that drive AOM-induced liver pathology have not been characterized to date. Thus, the goal of this study was to identify and examine the molecular and cellular mechanisms that contribute to hepatic injury during the progression of AOM-induced liver injury.
AOM, Sudan Black B, total serum bile acid assay, aspartate aminotransferase (AST) activity assay, alanine aminotransferase (ALT) activity assay, alkaline phosphatase (ALP) activity assay, GSH assay, and the β-actin antibody were purchased from MilliporeSigma (Burlington, MA, United States). The TUNEL assay, H2O2 assay, MDA activity assay, MPO activity assay, albumin ELISA assay, CYP2E1 antibody and cleaved caspase 3 antibody were purchased from Abcam (Boston, MA, United States). Mouse CCL2 and TNFα Quantikine ELISA kits were purchased from R&D Systems (Minneapolis, MN, United States). SYBR green, iScript cDNA Synthesis Kit, and primers recognizing CYP2E1, TNFα, CCL2 and GAPDH were purchased from Bio-Rad (Hercules, CA, United States). The SOD1, and phosphorylated MLKL (pMLKL) antibodies were purchased from Invitrogen (Carlsbad, CA, United States). Santa Cruz Biotechnology (Dallas, TX, United States) was the source of p21 and TGFβ1 antibodies. The GAPDH antibody was bought from GeneTex (Irvine, CA, United States). QS and Vectastain Elite ABC kits were purchased from Vector Laboratories (Newark, CA, United States). Western blot blocking buffer and secondary antibodies were purchased from LI-COR Biosciences (Lincoln, NE, United States). The RNeasy Mini Kit for RNA isolation was purchased from Qiagen (Germantown, MD, United States). Ephredia Cytoseal XYL mountant was purchased from Fisher Scientific (Hampton, NH, United States).
Male C57BL/6 mice (25-30 g; Charles River Laboratories, Wilmington, MA, United States) were used to perform in vivo animal experiments and were acclimated to the animal facility for one week prior to random allocation to control or treatment groups. All animal experiments were approved by the University of Texas at Austin Institutional Animal Care and Use Committee. Experiments were performed in compliance with the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals. ARRIVE guidelines were followed for all animal experiments.
To induce ALF, AOM (100 mg/kg intraperitoneally) was injected in C57BL/6 mice and their cages were placed on 37 °C heating pads to minimize incidence of hypothermia. After 12 hours, mice were observed and monitored every 2 hours for body temperature, weight, and neurologic score as previously described[15,16]. Liver tissue was gathered at four defined timepoints of neurologic status: before the onset of symptoms (pre), at the presence of mild ataxia and weakened reflexes (minor), at the presence of significant ataxia and reflex deficits (major), and at the loss of righting and corneal reflexes (coma) as we have previously described[15].
Histological staining was performed on 4 µm sections of paraffin-embedded liver tissue mounted onto positively-charged slides. After deparaffinization, the slides were stained with Hematoxylin QS for 1 minute and subsequently stained with eosin Y for 1 minute before rinsing in 95% ethanol. Then, the slides were dipped in 100% ethanol followed by 2 xylene washes and mounting of coverslips. The slides were imaged using a Leica Aperio AT2 imaging system and Aperio ImageScope software (Deer Park, IL, United States). Necrosis quantification was conducted on hematoxylin & eosin (H&E) slides using ImageJ software. Cells absent of nuclei were manually identified by a researcher blind to the groups and % area measurement was taken.
Immunohistochemistry and TUNEL staining were performed on liver tissue prepared as described above. Immunohistochemistry staining was performed by following instructions from the Vector Laboratories Vectastain Elite ABC kits with no modifications. Antibodies against CD16 were incubated overnight at 4 °C. Hematoxylin QS was used as the counterstain. For TUNEL staining, instructions from Abcam were followed with no modifications. Slides were imaged using a Leica Aperio AT2 imaging system and Aperio ImageScope software. For TUNEL staining, images were converted to grayscale and quantified by determining the percent area of positive staining using ImageJ software.
ALT, AST, ALP, and total bile acids were measured in serum of all mouse groups using commercially-available kits according to the manufacturer’s instructions. Liver GSH, malondialdehyde (MDA), H2O2, and myeloperoxidase (MPO) were assayed in freshly homogenized liver sections that were homogenized in recommended lysis buffer using a Miltenyi Biotec gentle MACS Octo Dissociator (Gaithersburg, MD, United States). GSH and H2O2 were normalized to liver tissue weight. MDA and MPO were normalized to lysate protein concentrations determined by a ThermoFisher Scientific Pierce BCA protein assay (Waltham, MA, United States). Absorbance or fluorescence was read using a Molecular Devices SpectraMax M5 plate reader (San Jose, CA, United States).
Protein was isolated from frozen liver tissue via homogenization in a MiltenyiBiotec gentle MACS Octo Dissociator. Protein concentration of liver tissue homogenates was measured using a ThermoFisher Scientific Pierce BCA protein assay. Twenty to forty μg of protein in Laemmli buffer was loaded into 10%-15% SDS-PAGE gels and transferred onto nitrocellulose membranes with a Bio-Rad Trans-Blot Turbo apparatus. Antibodies for CYP2E1, β-actin, SOD1, GAPDH, Cleaved caspase 3, pMLKL, p21, and TGFβ1 were incubated overnight. Secondary antibodies were incubated for 1 hour at room temperature. Images were taken using Bio-Rad ChemiDoc MP Imaging System. Fluorescent band intensity of the specified antibody was determined and compared against the loading controls β-actin or GAPDH. The vehicle groups were set to a protein expression value of 1 as a baseline for all other treatment groups. Immunoblotting quantifications of band intensities were completed using ImageJ software and the data were expressed as mean relative protein ± SE. Whole gel images with bands selected for analysis are provided (Supplementary Figure 1).
RNA was isolated from liver homogenized using a Miltenyi Biotec gentleMACS Octo Dissociator and reagents from a Qiagen RNeasy Mini Kit. RNA concentration in each sample was determined using a ThermoFisher Scientific Nanodrop 2000 spectrophotometer. A Bio-Rad iScript cDNA Synthesis Kit was used for synthesis of cDNA. RTPCR was performed using commercially-available primers against mouse CYP2E1, CCL2, TNFα, and GAPDH. SYBR green fluorescence was measured using a Bio-Rad CFX96 Real-Time System. A ΔΔCt analysis was performed as previously described with vehicle-treated liver as the control group[16].
Quantitative protein determination of CCL2 and TNFα was conducted in liver according to instructions from R&D Systems. Frozen liver tissue was homogenized from all treatment groups using a Miltenyi Biotec gentleMACS Octo Dissociator. Protein quantity of homogenates was measured using a ThermoFisher Pierce BCA Protein Assay kit according to the kit’s instructions. Input protein was 100 µg for each sample. A SpectraMax M5 plate reader from Molecular Devices was utilized to measure absorbance values, and data were presented as the picogram concentration of CCL2/TNFα per milligram of protein.
Statistical analyses were conducted utilizing GraphPad Prism software (La Jolla, CA, United States). The Student’s t test was utilized during the analysis of differences between 2 groups and analysis of variance when comparing the differences between 3 or more groups. All results were denoted as mean ± SE. Differences were considered significant when P < 0.05.
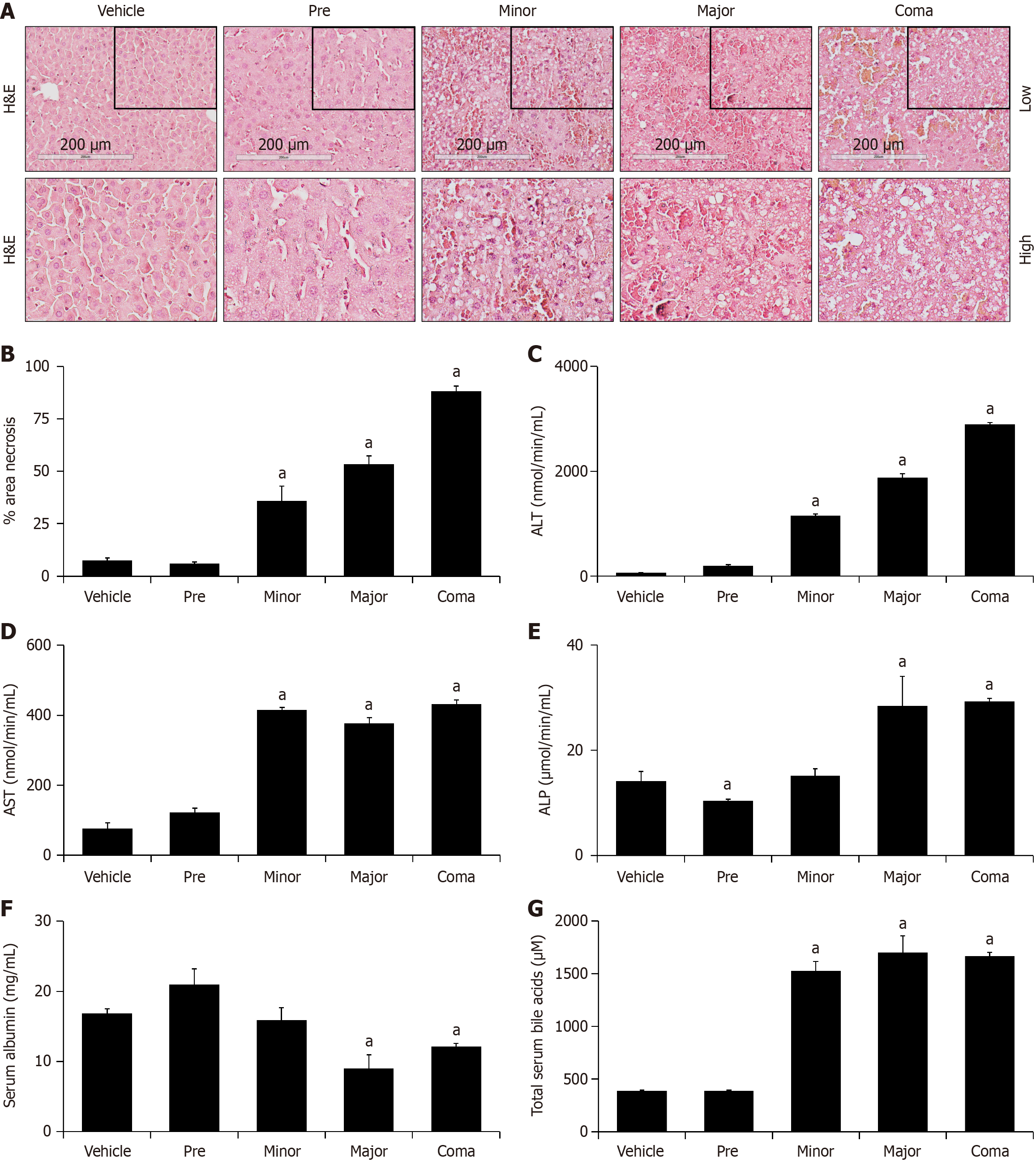
Initially, validation that AOM induced progressive liver injury resulting from duration of toxin exposure was tested. H&E staining of AOM-treated liver sections determined that microvesicular steatosis and hepatocyte necrosis were observable beginning at the minor time point and became more evident throughout the time course of this model (Figure 1A). To characterize liver injury, a quantitative assessment of percent area of necrosis was performed in H&E-stained liver sections. This analysis revealed a significant and progressive increase in necrosis in the minor, major, and coma groups of AOM-treated mice (Figure 1B).
Intrahepatic ammonia concentrations were found to significantly increase in the major and coma groups when compared to vehicle mice as determined with Nessler’s staining area (Supplementary Figure 2). We also analyzed serum ALT, AST, ALP, albumin and total bile acids to determine changes in hepatocyte and cholangiocyte function. Serum ALT was significantly increased in AOM-treated mice in a time-dependent response in the minor, major and coma groups (Figure 1C). Serum AST displayed a similar trend and was significantly increased in AOM-treated mice in the minor, major and coma groups (Figure 1D). Unique to ALP was a significant decrease in the pre group followed by a significant increase at major and coma groups (Figure 1E). Serum albumin was significantly decreased in AOM-treated mice in the major and coma groups, further indicating hepatic dysfunction (Figure 1F). Total bile acids were significantly increased in AOM-treated mice in the minor, major and coma groups (Figure 1G). These findings suggest that AOM induces necrosis, hepatocyte dysfunction, biliary injury and reduced hepatic function that progressively worsen as AOM-treated mice progress towards coma.
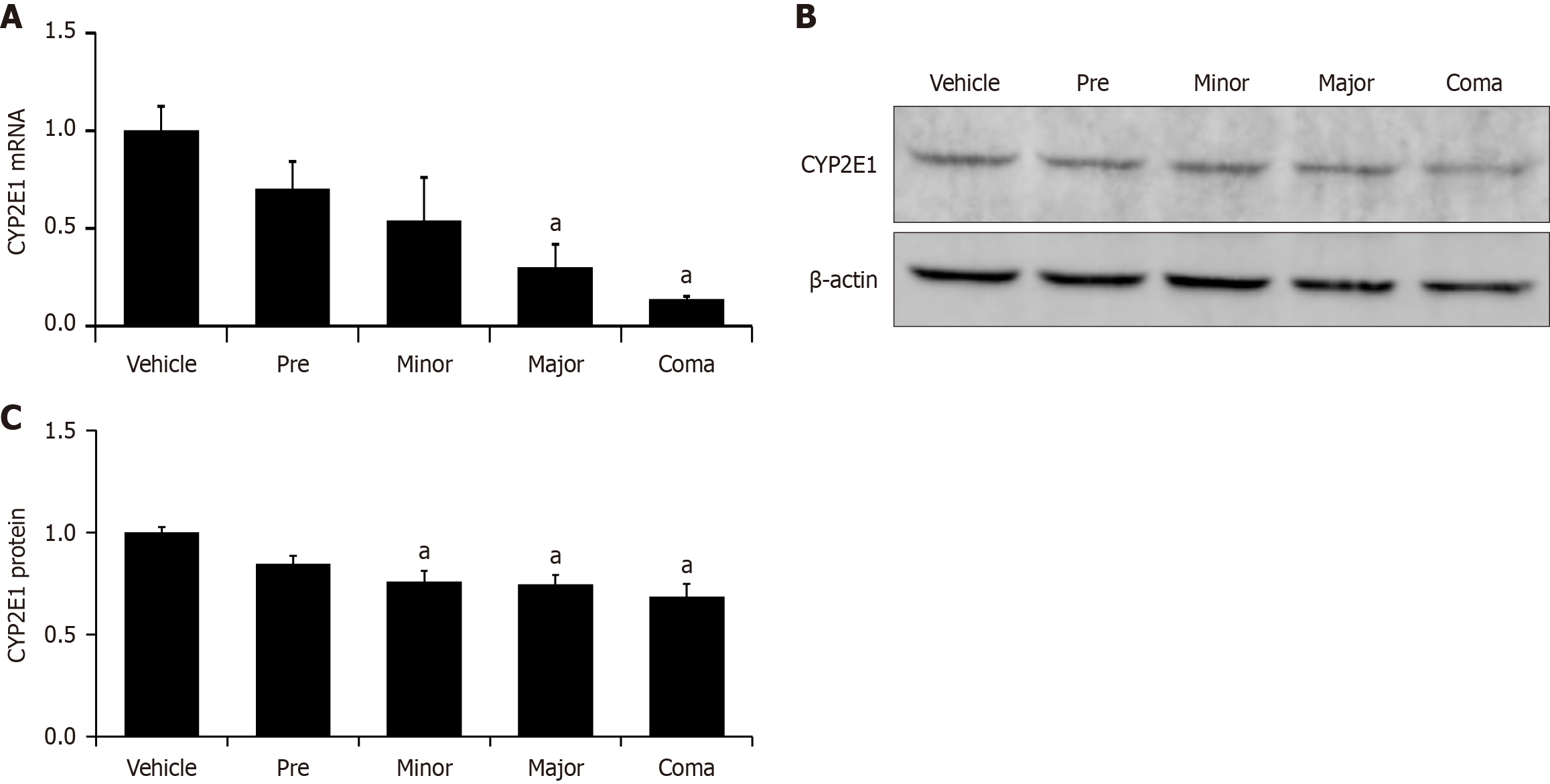
CYP2E1 is the primary cytochrome p450 enzyme responsible for the breakdown of AOM and its metabolites; therefore, its expression during AOM-induced liver injury can offer insights into AOM metabolism[17]. CYP2E1 mRNA expression was significantly decreased in AOM-treated mice in the major and coma groups (Figure 2A). Protein expression of CYP2E1 was significantly decreased in minor, major, and coma AOM-treated mice. However, levels remained detectable and above 65% relative expression compared to vehicle-treated mice (Figure 2B and C). These data support that AOM-induced liver injury reduced the expression of CYP2E1 mRNA and protein. Therefore, AOM metabolism may be impaired as liver injury progresses.
With AOM inducing significant hepatotoxic liver injury in mice models, examining how hepatic cells respond to AOM in vitro before undergoing cell death could elucidate additional details about AOM-induced hepatocyte injury. Initially, HepG2 cells were treated with AOM for up to 24 hours, revealing that concentrations of 100 nM and 1 mmol/L significantly reduced cell viability at both 2 and 12 hours compared to the untreated controls (Supplementary Figure 3A). Going forward, the 12-hour time point was used as it best represented the greatest effect regarding loss of cell viability and more closely represented the in vivo AOM exposure time. Interestingly, 100 nM and 1 mmol/L of AOM was found to significantly reduce HepG2 cells in the G0/G1 phase and significantly increase HepG2 cells in the S phase compared to basal (Supplementary Figure 3B). In addition, there was a significant increase of oxidative stress in HepG2 cells treated with 1 mmol/L of AOM compared to basal (Supplementary Figure 3C). Finally, HepG2 cells administered 1 mmol/L of AOM were found to have metabolic alterations such as increased proton leak (Supplementary Figure 3D) and reduced maximal cellular respiration (Supplementary Figure 3E). Collectively, these findings support that AOM induces cell death of HepG2 cells, likely due to mitochondrial dysfunction and increased oxidative stress, and that these cells increase cell replication in response to AOM-induced cell death.
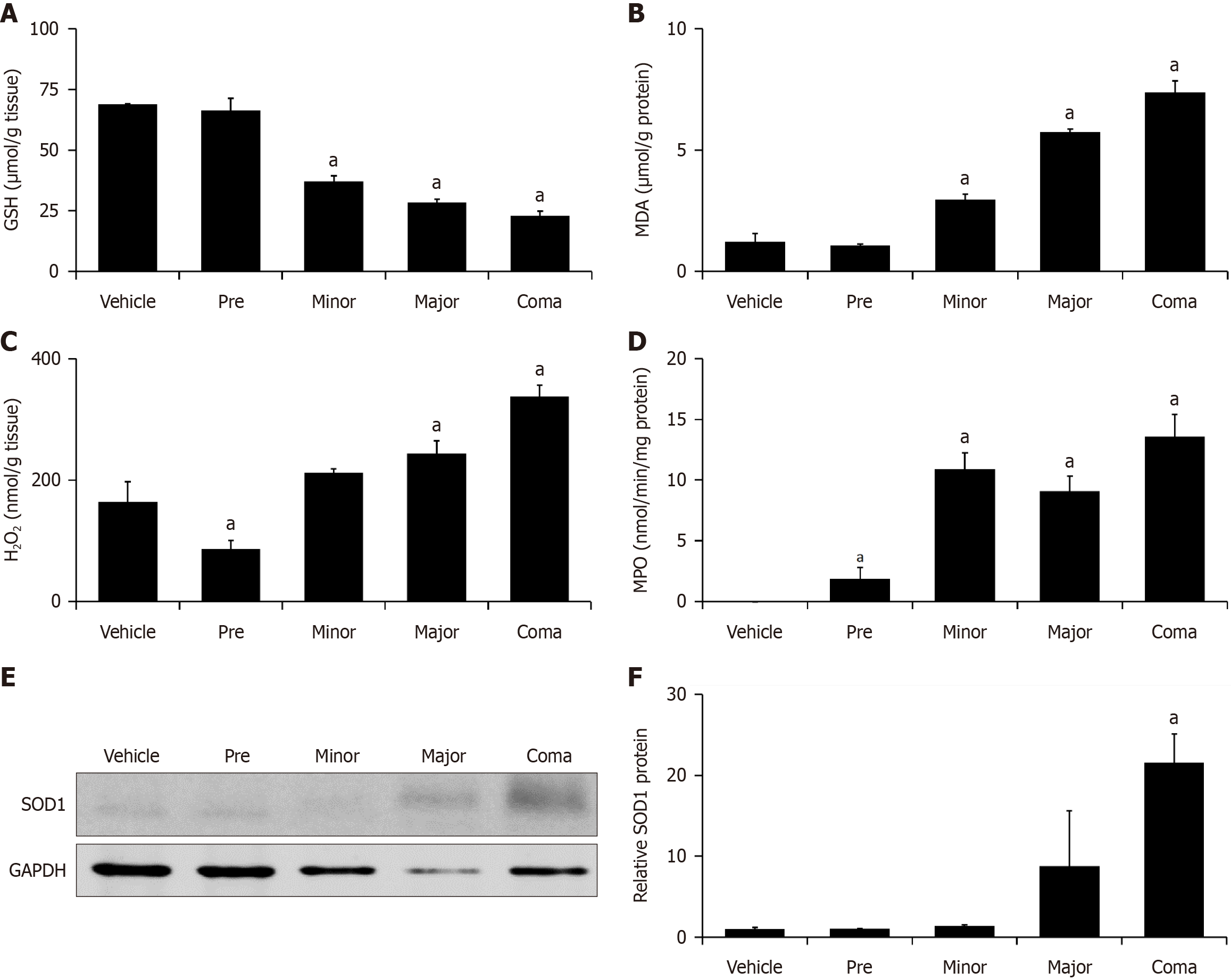
To determine if oxidative stress and compensatory mechanisms are present during AOM-induced ALF in vivo, hepatic GSH, MDA, H2O2, MPO and SOD1 were measured. GSH is the primary antioxidant mechanism in the liver and its expression was significantly decreased in a time-dependent response in the minor, major and coma groups of AOM-treated mice (Figure 3A). MDA is a measure of free radical formation and lipid peroxidation and was found to be significantly increased in the livers of minor, major and coma AOM-treated mice when compared to vehicle (Figure 3B). H2O2 concentrations were unique in that initially there was a significant decrease in the pre group of AOM-treated mice but then a rapid shift to a significant increase at major and coma time points (Figure 3C). MPO activity significantly increased in AOM-treated mice in the pre, minor, major and coma groups (Figure 3D). As MPO is the most prevalent proinflammatory enzyme in neutrophil granules, these data support involvement of neutrophils in hepatic inflammation and oxidative stress in AOM-treated mice[18].
To assess whether oxidative stress in this model resulted from decreased production of antioxidant enzymes, SOD1 expression was assessed in the liver of vehicle and AOM-treated mice. Hepatic SOD1 protein expression significantly increased at time of coma in AOM-treated mice (Figure 3E and F). This demonstrates that even though SOD1 is upregulated in AOM-treated mice, the oxidative stress generated by AOM overcomes this potential compensatory mechanism.
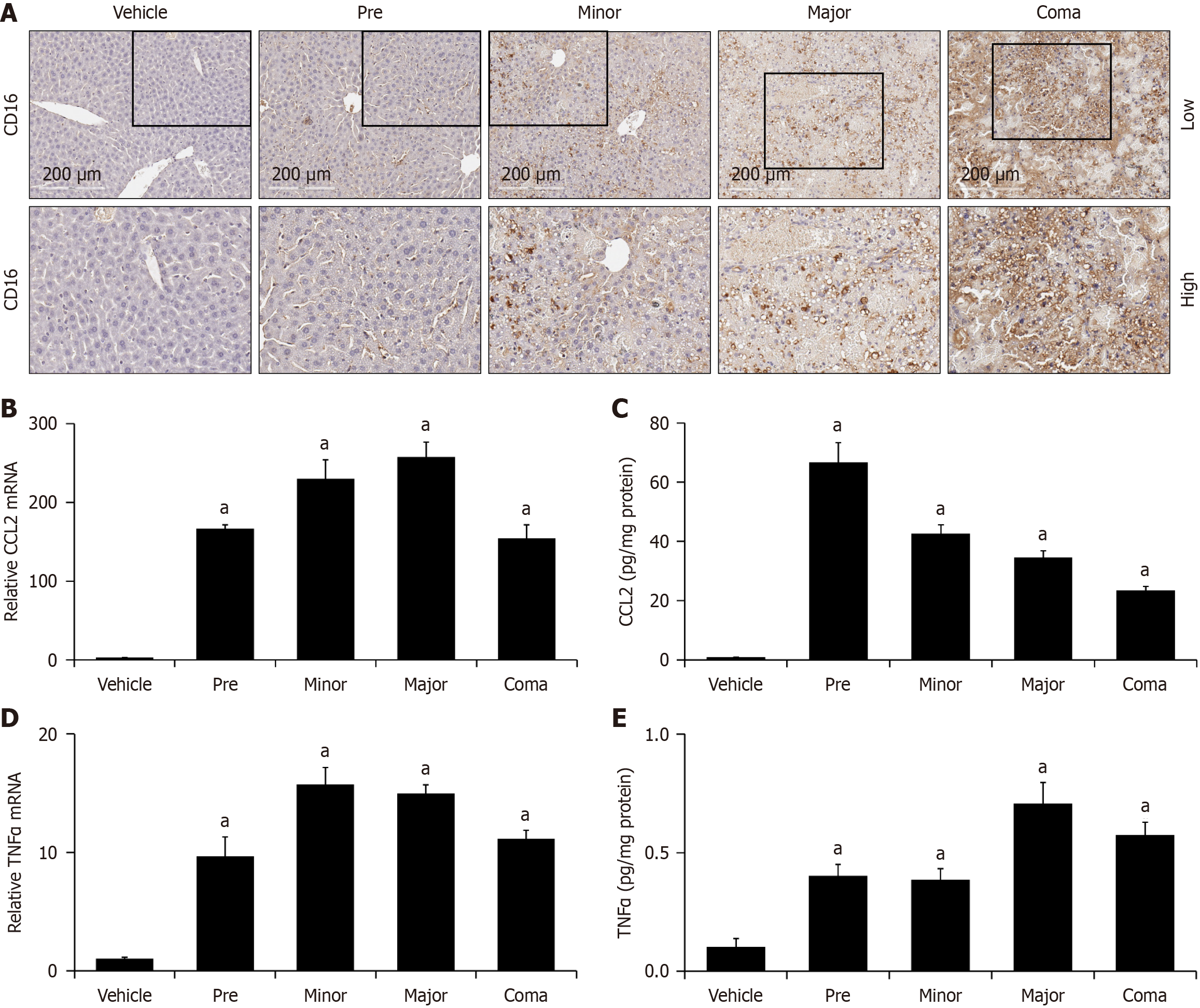
Neuroinflammation and its contribution to the progression of neurological dysfunction have been studied in AOM-treated mice. That being said, the hepatic changes in immune cells and cytokines throughout the AOM time course are not fully defined. Staining for CD16, which stains macrophages, natural killer cells, and neutrophils, was performed and identified a significant increase in all AOM treatment groups (Figure 4A). Staining for other immune cell markers such as F4/80 and CCR2 were performed with no change between the vehicle and AOM-treated groups (data not shown). CCL2 is a proinflammatory chemokine and CCL2 receptor antagonists can slow neurological decline towards coma in AOM-treated mice[19]. CCL2 expression was assessed to characterize hepatic inflammation throughout the time course of AOM-treated mice. Hepatic CCL2 mRNA expression was significantly elevated in all AOM-treated groups (Figure 4B). Interestingly, CCL2 protein is elevated throughout AOM-induced liver injury and showed its largest increase in comparison to vehicle at the pre time point, giving support that this chemokine is elevated as an early event during AOM-induced ALF (Figure 4C). Activation of proinflammatory chemokines leads to an immune response and proinflammatory cytokine production. TNFα mRNA and protein were significantly increased in all AOM groups compared to the vehicle group, though their peak increases occur later than what was observed for CCL2 (Figure 4D and E).
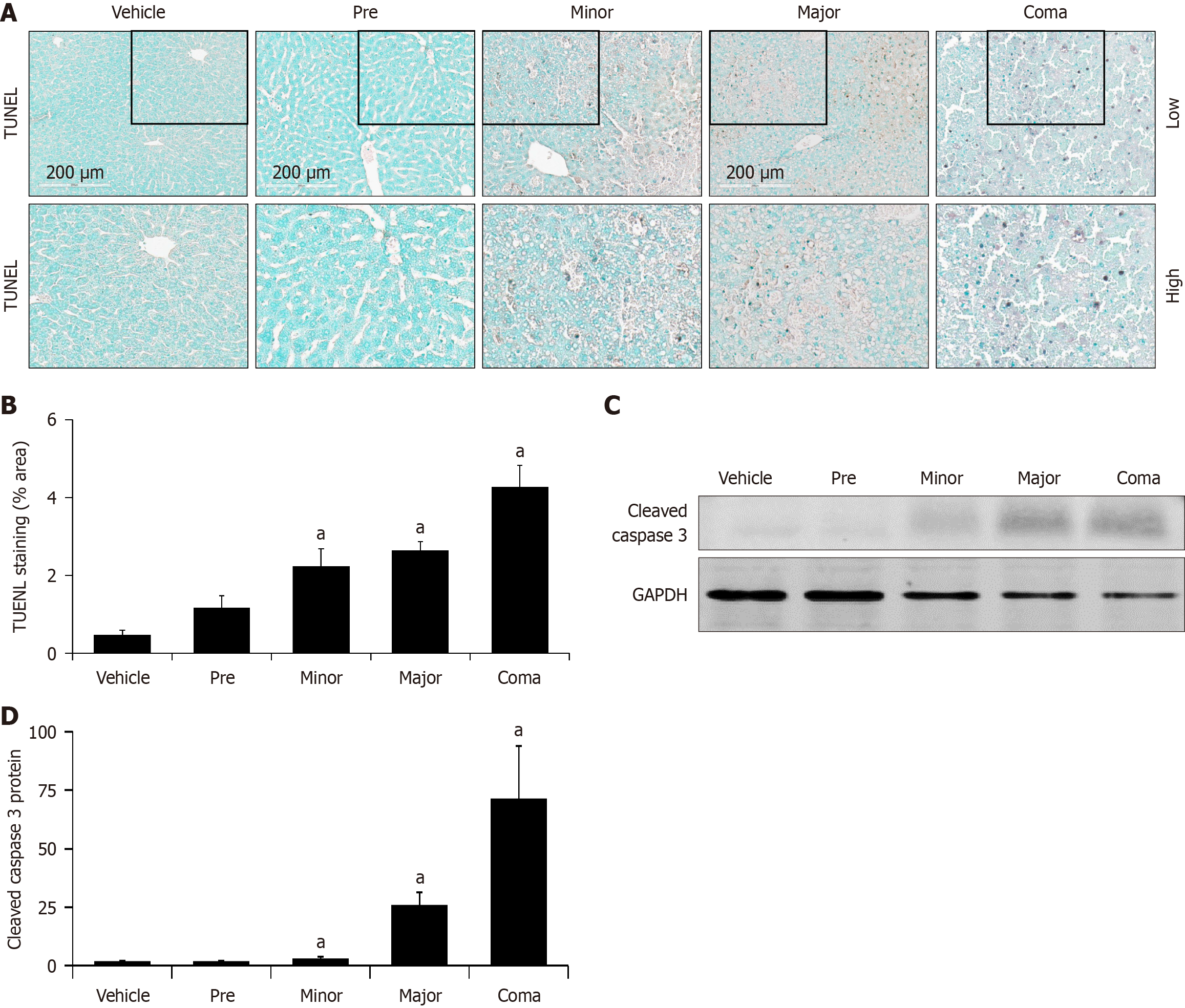
With the significant loss of hepatic function observed during AOM-induced ALF, mechanisms of programmed cell death may be contributing to worse pathology in this model. Apoptosis was assessed with TUNEL staining, which showed a significant increase of stained nuclei in AOM-treated mice at the minor, major and coma time points (Figure 5A and B). The protein expression of the apoptosis activator cleaved caspase 3 was also significantly increased in AOM-treated mice at the minor, major and coma time points (Figure 5C and D). These data suggest that significantly increased cell death and DNA fragmentation were evident throughout the AOM time course, beginning in the minor group.
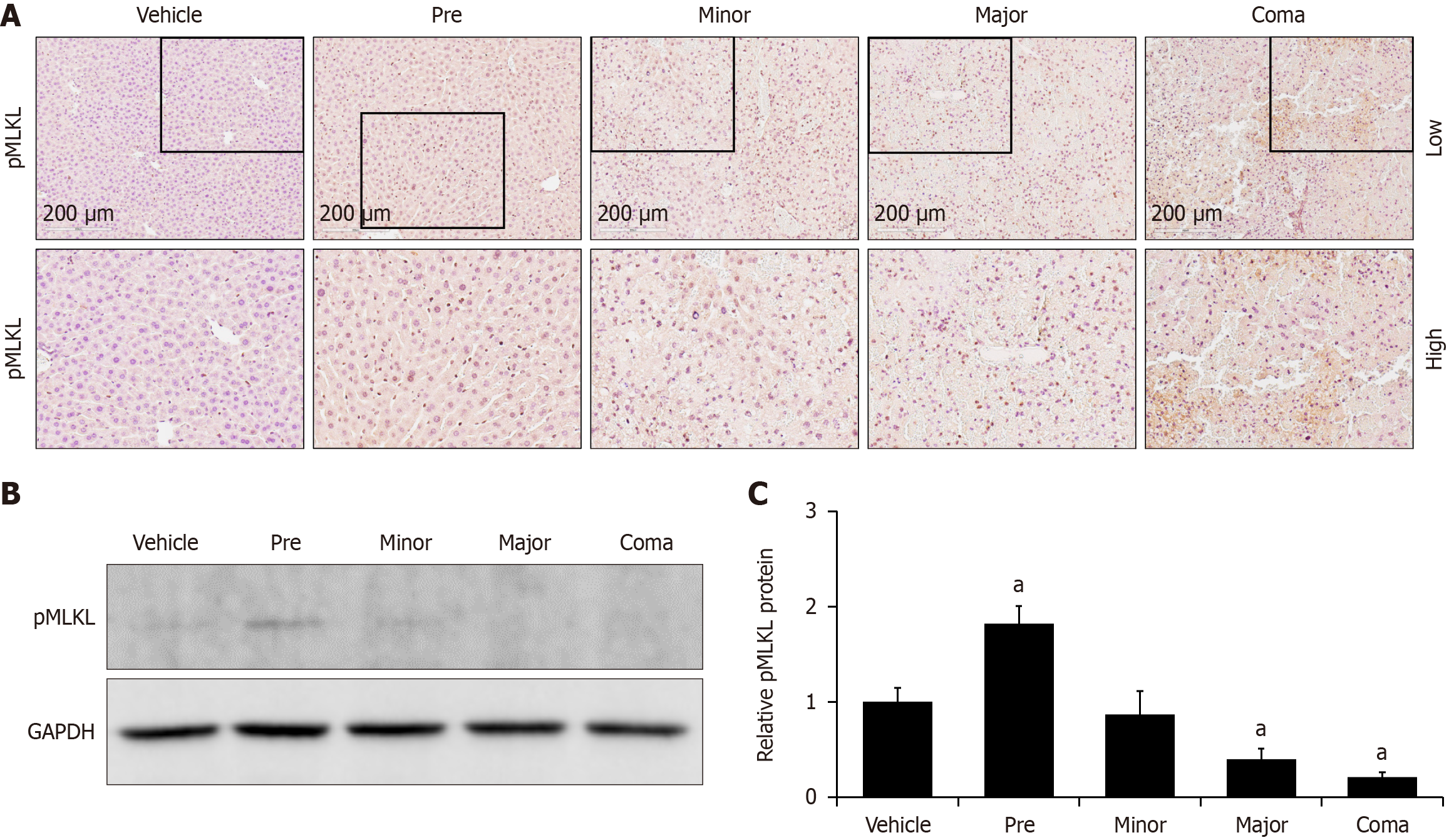
Programmed necrosis, or necroptosis, can be a mechanism of induced cell death and is driven by signaling including phosphorylation of MLKL[20]. Staining for pMLKL was performed and while increased staining was observed, it was not consistent throughout the entire liver lobule (Figure 6A). To understand the changes in expression in a more robust manner, pMLKL protein was assessed by immunoblot and showed a biphasic expression pattern with a significant increase observed at the pre time point and significant decreases observed in the major and coma groups (Figure 6B and C). These data suggest that necroptosis occurs early and decreases as caspases become activated, leading to the onset of apoptosis that induces cell death at the later time points in AOM-treated mice.
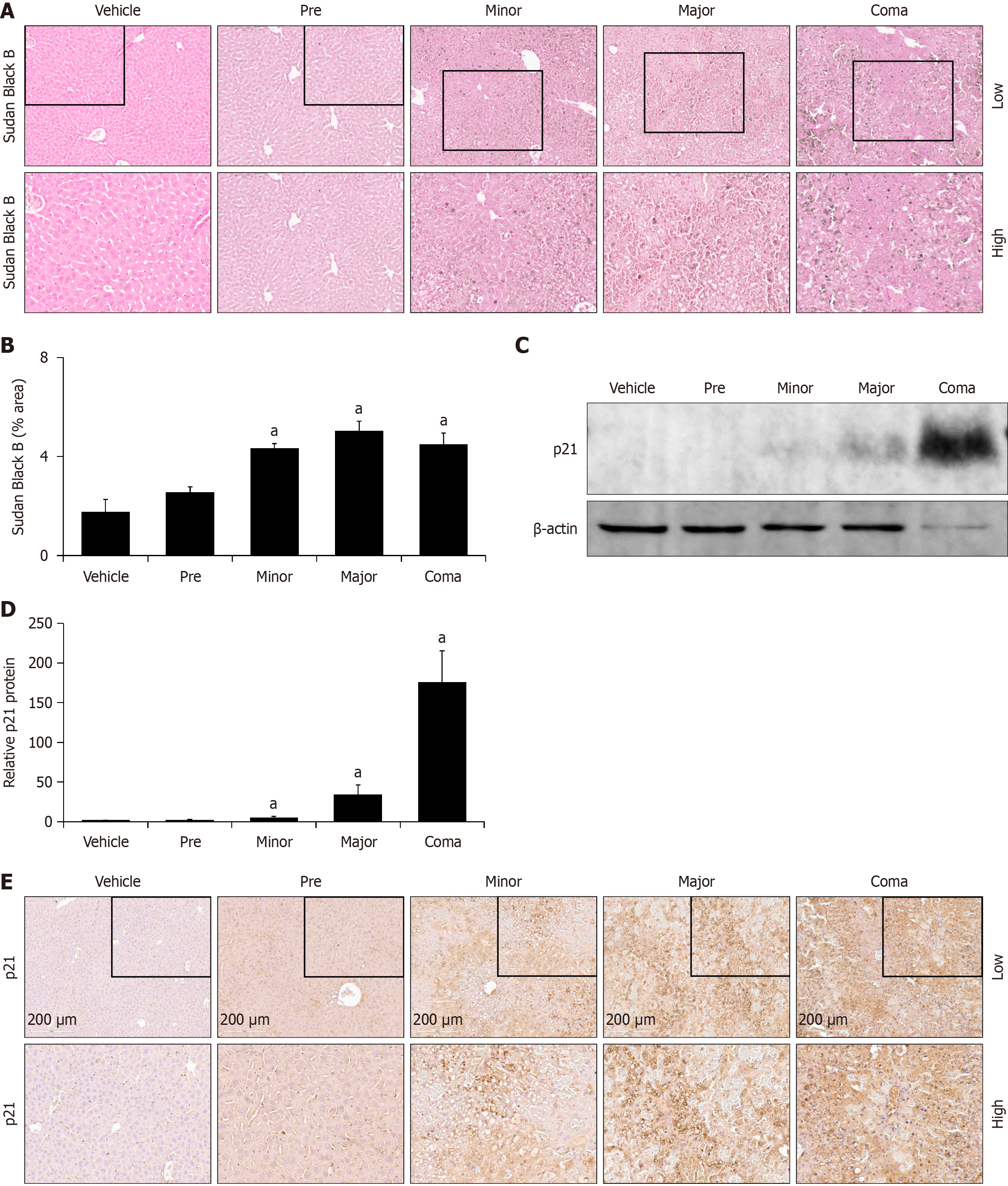
As AOM-induced progressive liver injury worsens with prolonged toxin exposure, it is likely that impaired hepatocellular repair mechanisms, particularly hepatocellular senescence, could contribute to AOM-induced ALF. Sudan Black B staining, which stains for lipofuscin and is a marker of cellular senescence, was significantly increased in minor, major and coma groups of AOM-induced ALF (Figure 7A and B). Increased p21 expression is associated with cell cycle repair or withdrawal to senescent or apoptotic pathways and its protein expression was significantly increased in AOM-injected mice at minor, major and coma time points (Figure 7C and D). These large increases of p21 protein at the later stages of AOM-induced ALF were also observed as p21 staining increased throughout the time course of AOM-induced liver injury (Figure 7E). Collectively, these data support an increase of hepatocellular senescence as AOM-induced ALF progresses towards coma.
The significant findings from this study are that AOM-induced liver injury generates hepatocyte and biliary injury that worsens over time and is associated with increased hepatic necrosis. In addition, there is increased oxidative stress and inflammation in the liver, though the largest increases of inflammation occur prior to coma. Apoptosis and cellular senescence increased as mice progressed towards coma. Compared to previous studies utilizing AOM treatment in mice, this report is the first to investigate hepatic cell death signaling, cellular senescence and oxidative stress throughout this disease state. Together, these data support that AOM hepatotoxicity initially generates inflammation and oxidative stress which progresses towards multiple forms of cell death and impaired liver regeneration via increased cellular senescence.
AOM is primarily metabolized by the cytochrome p450 enzyme CYP2E1, which accounts for approximately 98% of the toxin’s breakdown[17]. The finding that CYP2E1 expression decreases, likely resulting from death of centrilobular hepatocytes as AOM-induced injury progresses, gives support that decreased AOM metabolism likely occurs, increasing its bioavailability to the liver and inducing greater injury. In addition, CYP2E1 metabolizes AOM into methylazoxymethanol (MAM), another very toxic metabolite with a long half-life[17,21]. MAM has been shown to induce liver carcinogenesis and hepatic lipid peroxidation in animal models indicating that this AOM metabolite likely induces hepatic injury as well[22-24]. With the long half-life of MAM and reduced CYP2E1 expression, there is support that the bioavailability of MAM is also increased in AOM-treated mice. Greater characterization of MAM and its role in AOM-induced ALF are needed.
Of the data presented, the hepatic inflammation and inflammation-mediated necroptosis findings showed the earliest elevation in the context of AOM progression. Among the systemic treatments that are protective in this model, therapies aimed at targeting inflammation offer the most benefit. Therapeutic hypothermia can reduce inflammation and oxidative stress and has been shown to confer hepatic protection in multiple models of liver injury including sepsis and acetaminophen-induced acute liver injury[25,26]. In AOM-treated mice, therapeutic hypothermia induced by reducing body temperature from 37 to 35 ˚C led to significantly reduced ALT levels, less hepatocellular injury, reduced neutrophil infiltration and a reduction of circulating inflammatory cytokines including IL-1β[27]. Evidence of direct targeting of specific inflammatory mediators has shown that systemic inhibition of TNFα with etanercept, or CCL2 signaling with INCB 3284 dimesylate or C021 dihydrochloride, led to reduced hepatocellular injury and a significant decrease of serum ALT concentrations when compared to control AOM-treated mice[19,28]. Therefore, it appears that inflammation is a key initiator of AOM-induced liver injury progression.
Oxidative stress occurs when the levels of oxidants exceed antioxidants in a biological system, disrupting redox signaling and causing molecular damage[29]. Mitochondria in healthy states do generate reactive oxygen species but these oxidants are quenched by antioxidants such as GSH[30]. With AOM generating significant hepatic injury and inflammation, it is feasible that hepatic oxidative stress was increased. In our study we found GSH levels significantly decreased during AOM-induced liver injury, though some antioxidant capacity may have been restored by the significant increase of SOD1 protein. This was not enough to reduce oxidative stress as concentrations of MDA, H2O2 and MPO were highest when AOM-treated mice reached coma. No reports that mechanistically assess hepatic oxidative stress during the AOM mouse model of ALF have been performed. Outside of this model, during acetaminophen-induced liver injury oxidative stress increases and induces liver injury by depletion of GSH by the acetaminophen metabolite N-acetyl-p-benzoquinone[31,32]. In addition, oxidative stress increases liver injury during alcoholic liver disease, metabolic dysfunction-associated fatty liver disease, DILI and other hepatic disorders[33-35]. One report found that resveratrol conferred hepatoprotection and reduced hepatic MDA levels, while increasing GSH activity, in rats administered AOM at 5 mg/kg twice a week for 7 weeks in a chronic AOM-induced hepatic injury model[36]. Additional studies are needed in AOM-induced ALF to determine if reducing oxidative stress would be a beneficial therapeutic target in this model.
In the context of ALF, cell death occurs rapidly through two main mechanisms: Necroptosis and apoptosis. The timing of and factors which promote each of these cell death processes can provide insights into the progression of ALF. In AOM-treated mice, we observe early necroptosis followed by significant increases in both apoptosis and necrosis. It is possible that necroptosis is reduced due to increased caspase activity during the later stages of AOM-induced liver injury. In other ALF models, different cell death mechanisms are activated depending upon the toxin. Acetaminophen induces necroptosis and necrosis while D-galactosamine and LPS can induce apoptosis[37,38]. AOM-induced liver injury appears to be unique in that it involves multiple cell death mechanisms depending upon the duration of exposure.
For the liver to repair and regenerate from the cell death that occurs during ALF, hepatocytes must reenter the cell cycle and repopulate the liver with functional hepatocytes. During acetaminophen-induced ALF, measures of hepatocellular senescence and paracrine senescence were shown to be driven by macrophage-derived TGFβ1 signaling[12]. This was similar to what was observed in the current study as there was a significant elevation of senescence markers and proteins, giving support that cellular senescence occurs during AOM-induced ALF. A recent report describes that administration of the senolytics dasatinib and quercetin to mice with acetaminophen-induced liver injury did not confer a significant improvement of liver function[39]. With the reduced regeneration observed in AOM-induced liver injury compared to acetaminophen-induced liver injury, targeting senescence with senolytics could prove efficacious in the AOM model and will need to be investigated in future studies.
One area this study addresses, which is not commonly investigated in animal models of ALF, is biliary injury. A ductular reaction can be observed in the most severe cases of ALF and can often be associated with biliary injury[40]. Increases of both ALT and ALP are associated with a mixed DILI etiology in clinical patients, giving evidence of both hepatocyte and cholangiocyte injury, which is what was observed during AOM toxicity in the current study[41]. Here, we observe a significant increase of ALP serum levels and an increase of serum bile acids. This increase in serum bile acids could further synergize with the hepatotoxic effects of AOM since this has been shown to occur during acetaminophen hepatoxicity[42]. In addition to this, our findings show that hepatic CYP2E1 expression during AOM-induced liver injury may impair AOM metabolism as injury progresses. Cholangiocytes have also been found to express CYP2E1 and its role in the metabolism of AOM as well the exact contribution of biliary injury to AOM-induced ALF is unknown and warrants further investigation[43,44].
AOM has been shown to induce hepatic pathology outside the scope of acute liver injury. For example, AOM can induce chronic liver injury if administered multiple times at lower concentrations. Rachakonda et al[45] demonstrate that 10 mg/kg ip weekly injection of AOM into mice for 6 weeks led to increased fibrosis, as determined by Sirius red staining and activation of hepatic stellate cells, at 20 weeks after the initial AOM dose. The cumulative dose given in these mice was less than the single dose of 100 mg/kg used in the current study; as the injury persists 20 weeks later, this supports our findings regarding reduced AOM metabolism via CYP2E1 and increased hepatocellular senescence. Outside of chronic liver injury, AOM has the potential to induce liver tumorigenesis though this is not common. One study injecting 15 mg/kg ip AOM into 5-week-old normal and T-cell-deficient rats observed that none of the normal rodents developed tumors but all of the T-cell deficient animals did when they were 48 weeks of age[46]. These studies provide evidence that AOM concentration and duration, as well as the genetic background of the animals, have the potential to generate hepatic pathology different from that presented by the data in this report.
Limitations of this report arise from its focus on AOM-induced liver injury, an experimental rodent model that does not have a direct clinical counterpart, despite being a well-documented model for studying Type A hepatic encephalopathy. There are components of the pathology generated that are relevant to other models of liver injury, such as oxidative stress, inflammation and senescence. Therefore, it is likely that many causes of DILI in patients rely on similar pathologic mechanisms. Another limitation is that HepG2 cells were used for in vitro studies though they cannot mimic all aspects of hepatocytes in vivo, such as cell cycle regulation, as they are derived from hepatoblastoma[47].
In conclusion, this report demonstrates that the AOM model generates progressive liver injury driven by increased inflammation, oxidative stress and cell death mechanisms, which was exacerbated by increased senescence. This further demonstrates that the AOM mouse model is unique as it is a progressive liver injury that does not spontaneously recover, making it a useful model of progressive liver injury leading to ALF. Future interventional studies targeting each of these molecular pathology pathways will be necessary to determine their direct roles in AOM-induced hepatotoxicity.
This material is the result of work supported with resources and the use of facilities at the Central Texas Veterans Health Care System, Temple, TX, United States. The content is the responsibility of the author(s) alone and does not necessarily reflect the views or policies of the Department of Veterans Affairs or the United States Government.
| 1. | Bernal W, Wendon J. Acute liver failure. N Engl J Med. 2013;369:2525-2534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1040] [Cited by in RCA: 884] [Article Influence: 68.0] [Reference Citation Analysis (0)] |
| 2. | Stravitz RT, Lee WM. Acute liver failure. Lancet. 2019;394:869-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 723] [Cited by in RCA: 656] [Article Influence: 93.7] [Reference Citation Analysis (3)] |
| 3. | Bunchorntavakul C, Reddy KR. Acetaminophen (APAP or N-Acetyl-p-Aminophenol) and Acute Liver Failure. Clin Liver Dis. 2018;22:325-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 127] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 4. | Reuben A, Koch DG, Lee WM; Acute Liver Failure Study Group. Drug-induced acute liver failure: results of a U.S. multicenter, prospective study. Hepatology. 2010;52:2065-2076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 656] [Cited by in RCA: 538] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 5. | Laqueur GL, Mickelsen O, Whiting MG, Kurland LT. Carcinogenic Properties Of Nuts From Cycas Circinalis L. Indigenous To Guam. J Natl Cancer Inst. 1963;31:919-951. [PubMed] |
| 6. | Ward JM. Dose response to a single injection of azoxymethane in rats. Induction of tumors in the gastrointestinal tract, auditory sebaceous glands, kidney, liver and preputial gland. Vet Pathol. 1975;12:165-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 29] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Matkowskyj KA, Marrero JA, Carroll RE, Danilkovich AV, Green RM, Benya RV. Azoxymethane-induced fulminant hepatic failure in C57BL/6J mice: characterization of a new animal model. Am J Physiol. 1999;277:G455-G462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 54] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | DeMorrow S, Cudalbu C, Davies N, Jayakumar AR, Rose CF. 2021 ISHEN guidelines on animal models of hepatic encephalopathy. Liver Int. 2021;41:1474-1488. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 9. | Bélanger M, Côté J, Butterworth RF. Neurobiological characterization of an azoxymethane mouse model of acute liver failure. Neurochem Int. 2006;48:434-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 10. | Grant S, McMillin M, Frampton G, Petrescu AD, Williams E, Jaeger V, Kain J, DeMorrow S. Direct Comparison of the Thioacetamide and Azoxymethane Models of Type A Hepatic Encephalopathy in Mice. Gene Expr. 2018;18:171-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 11. | Woolbright BL, Jaeschke H. Role of the inflammasome in acetaminophen-induced liver injury and acute liver failure. J Hepatol. 2017;66:836-848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 347] [Cited by in RCA: 313] [Article Influence: 34.8] [Reference Citation Analysis (1)] |
| 12. | Bird TG, Müller M, Boulter L, Vincent DF, Ridgway RA, Lopez-Guadamillas E, Lu WY, Jamieson T, Govaere O, Campbell AD, Ferreira-Gonzalez S, Cole AM, Hay T, Simpson KJ, Clark W, Hedley A, Clarke M, Gentaz P, Nixon C, Bryce S, Kiourtis C, Sprangers J, Nibbs RJB, Van Rooijen N, Bartholin L, McGreal SR, Apte U, Barry ST, Iredale JP, Clarke AR, Serrano M, Roskams TA, Sansom OJ, Forbes SJ. TGFβ inhibition restores a regenerative response in acute liver injury by suppressing paracrine senescence. Sci Transl Med. 2018;10:eaan1230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 207] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 13. | Wang Y, Zhang H, Chen Q, Jiao F, Shi C, Pei M, Lv J, Zhang H, Wang L, Gong Z. TNF-α/HMGB1 inflammation signalling pathway regulates pyroptosis during liver failure and acute kidney injury. Cell Prolif. 2020;53:e12829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 193] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 14. | Pandurangan AK, Ananda Sadagopan SK, Dharmalingam P, Ganapasam S. Luteolin, a bioflavonoid, attenuates azoxymethane-induced effects on mitochondrial enzymes in BALB/c mice. Asian Pac J Cancer Prev. 2014;14:6669-6672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 15. | Jefferson B, Ali M, Grant S, Frampton G, Ploof M, Andry S, DeMorrow S, McMillin M. Thrombospondin-1 Exacerbates Acute Liver Failure and Hepatic Encephalopathy Pathology in Mice by Activating Transforming Growth Factor β1. Am J Pathol. 2020;190:347-357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 16. | Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158539] [Cited by in RCA: 139395] [Article Influence: 5575.8] [Reference Citation Analysis (7)] |
| 17. | Sohn OS, Ishizaki H, Yang CS, Fiala ES. Metabolism of azoxymethane, methylazoxymethanol and N-nitrosodimethylamine by cytochrome P450IIE1. Carcinogenesis. 1991;12:127-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 94] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 18. | Pulli B, Ali M, Forghani R, Schob S, Hsieh KL, Wojtkiewicz G, Linnoila JJ, Chen JW. Measuring myeloperoxidase activity in biological samples. PLoS One. 2013;8:e67976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 313] [Cited by in RCA: 279] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 19. | McMillin M, Frampton G, Thompson M, Galindo C, Standeford H, Whittington E, Alpini G, DeMorrow S. Neuronal CCL2 is upregulated during hepatic encephalopathy and contributes to microglia activation and neurological decline. J Neuroinflammation. 2014;11:121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 20. | Samson AL, Zhang Y, Geoghegan ND, Gavin XJ, Davies KA, Mlodzianoski MJ, Whitehead LW, Frank D, Garnish SE, Fitzgibbon C, Hempel A, Young SN, Jacobsen AV, Cawthorne W, Petrie EJ, Faux MC, Shield-Artin K, Lalaoui N, Hildebrand JM, Silke J, Rogers KL, Lessene G, Hawkins ED, Murphy JM. MLKL trafficking and accumulation at the plasma membrane control the kinetics and threshold for necroptosis. Nat Commun. 2020;11:3151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 281] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 21. | Megaraj V, Ding X, Fang C, Kovalchuk N, Zhu Y, Zhang QY. Role of hepatic and intestinal p450 enzymes in the metabolic activation of the colon carcinogen azoxymethane in mice. Chem Res Toxicol. 2014;27:656-662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 22. | Deschner EE, Zedeck MS. Lipid peroxidation in liver and colon of methylazoxymethanol treated rats. Cancer Biochem Biophys. 1986;9:25-29. [PubMed] |
| 23. | Mori H, Tanaka T, Shima H, Kuniyasu T, Takahashi M. Inhibitory effect of chlorogenic acid on methylazoxymethanol acetate-induced carcinogenesis in large intestine and liver of hamsters. Cancer Lett. 1986;30:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 64] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 24. | Harada T, Hatanaka J, Enomoto M. Liver cell carcinomas in the medaka (Oryzias latipes) induced by methylazoxymethanol-acetate. J Comp Pathol. 1988;98:441-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 9] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 25. | Lee JH, Kim K, Jo YH, Kim MA, Rim KP, Kang KW, Rhee JE, Lee MJ, Lee HS, Kwon WY, Suh GJ. Therapeutic hypothermia attenuates liver injury in polymicrobial sepsis model of rats via Akt survival pathway. J Surg Res. 2013;181:114-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Vaquero J, Bélanger M, James L, Herrero R, Desjardins P, Côté J, Blei AT, Butterworth RF. Mild hypothermia attenuates liver injury and improves survival in mice with acetaminophen toxicity. Gastroenterology. 2007;132:372-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (2)] |
| 27. | Bémeur C, Desjardins P, Butterworth RF. Antioxidant and anti-inflammatory effects of mild hypothermia in the attenuation of liver injury due to azoxymethane toxicity in the mouse. Metab Brain Dis. 2010;25:23-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 28. | Chastre A, Bélanger M, Beauchesne E, Nguyen BN, Desjardins P, Butterworth RF. Inflammatory cascades driven by tumor necrosis factor-alpha play a major role in the progression of acute liver failure and its neurological complications. PLoS One. 2012;7:e49670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 76] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 29. | Sies H. Oxidative stress: a concept in redox biology and medicine. Redox Biol. 2015;4:180-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2494] [Cited by in RCA: 2062] [Article Influence: 187.5] [Reference Citation Analysis (8)] |
| 30. | Bhatti JS, Bhatti GK, Reddy PH. Mitochondrial dysfunction and oxidative stress in metabolic disorders - A step towards mitochondria based therapeutic strategies. Biochim Biophys Acta Mol Basis Dis. 2017;1863:1066-1077. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1414] [Cited by in RCA: 1211] [Article Influence: 134.6] [Reference Citation Analysis (7)] |
| 31. | Jaeschke H, McGill MR, Ramachandran A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity. Drug Metab Rev. 2012;44:88-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 780] [Cited by in RCA: 704] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 32. | Yan M, Huo Y, Yin S, Hu H. Mechanisms of acetaminophen-induced liver injury and its implications for therapeutic interventions. Redox Biol. 2018;17:274-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 571] [Cited by in RCA: 497] [Article Influence: 62.1] [Reference Citation Analysis (1)] |
| 33. | Villanueva-Paz M, Morán L, López-Alcántara N, Freixo C, Andrade RJ, Lucena MI, Cubero FJ. Oxidative Stress in Drug-Induced Liver Injury (DILI): From Mechanisms to Biomarkers for Use in Clinical Practice. Antioxidants (Basel). 2021;10:390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 34. | Kim HG, Huang M, Xin Y, Zhang Y, Zhang X, Wang G, Liu S, Wan J, Ahmadi AR, Sun Z, Liangpunsakul S, Xiong X, Dong XC. The epigenetic regulator SIRT6 protects the liver from alcohol-induced tissue injury by reducing oxidative stress in mice. J Hepatol. 2019;71:960-969. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 119] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 35. | Chen Z, Tian R, She Z, Cai J, Li H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic Biol Med. 2020;152:116-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1110] [Cited by in RCA: 964] [Article Influence: 160.7] [Reference Citation Analysis (6)] |
| 36. | Gurocak S, Karabulut E, Karadag N, Ozgor D, Ozkeles N, Karabulut AB. Preventive effects of resveratrol against azoxymethane induced damage in rat liver. Asian Pac J Cancer Prev. 2013;14:2367-2370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 37. | Jaeschke H, Ramachandran A, Chao X, Ding WX. Emerging and established modes of cell death during acetaminophen-induced liver injury. Arch Toxicol. 2019;93:3491-3502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 110] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 38. | Yu Z, Ding Y, Zeng T, Zhao X, Zhang C. Hepatoprotective effect of diallyl trisulfide against lipopolysaccharide and D-galactosamine induced acute liver failure in mice via suppressing inflammation and apoptosis. Toxicol Res (Camb). 2022;11:263-271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 39. | Umbaugh DS, Nguyen NT, Smith SH, Ramachandran A, Jaeschke H. The p21(+) perinecrotic hepatocytes produce the chemokine CXCL14 after a severe acetaminophen overdose promoting hepatocyte injury and delaying regeneration. Toxicology. 2024;504:153804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 40. | Fyfe B, Zaldana F, Liu C. The Pathology of Acute Liver Failure. Clin Liver Dis. 2018;22:257-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 41. | Leise MD, Poterucha JJ, Talwalkar JA. Drug-induced liver injury. Mayo Clin Proc. 2014;89:95-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 280] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 42. | Ghallab A, Hassan R, Hofmann U, Friebel A, Hobloss Z, Brackhagen L, Begher-Tibbe B, Myllys M, Reinders J, Overbeck N, Sezgin S, Zühlke S, Seddek AL, Murad W, Brecklinghaus T, Kappenberg F, Rahnenführer J, González D, Goldring C, Copple IM, Marchan R, Longerich T, Vucur M, Luedde T, Urban S, Canbay A, Schreiter T, Trauner M, Akakpo JY, Olyaee M, Curry SC, Sowa JP, Jaeschke H, Hoehme S, Hengstler JG. Interruption of bile acid uptake by hepatocytes after acetaminophen overdose ameliorates hepatotoxicity. J Hepatol. 2022;77:71-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 43. | Visentin M, Lenggenhager D, Gai Z, Kullak-Ublick GA. Drug-induced bile duct injury. Biochim Biophys Acta Mol Basis Dis. 2018;1864:1498-1506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 44. | Lakehal F, Wendum D, Barbu V, Becquemont L, Poupon R, Balladur P, Hannoun L, Ballet F, Beaune PH, Housset C. Phase I and phase II drug-metabolizing enzymes are expressed and heterogeneously distributed in the biliary epithelium. Hepatology. 1999;30:1498-1506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 48] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Rachakonda V, Jadeja RN, Urrunaga NH, Shah N, Ahmad D, Cheng K, Twaddell WS, Raufman JP, Khurana S. M1 Muscarinic Receptor Deficiency Attenuates Azoxymethane-Induced Chronic Liver Injury in Mice. Sci Rep. 2015;5:14110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 46. | Wu B, Ootani A, Iwakiri R, Sakata Y, Fujise T, Amemori S, Yokoyama F, Tsunada S, Toda S, Fujimoto K. T cell deficiency leads to liver carcinogenesis in azoxymethane-treated rats. Exp Biol Med (Maywood). 2006;231:91-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 47. | López-Terrada D, Cheung SW, Finegold MJ, Knowles BB. Hep G2 is a hepatoblastoma-derived cell line. Hum Pathol. 2009;40:1512-1515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 267] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/