Published online Mar 28, 2025. doi: 10.3748/wjg.v31.i12.100855
Revised: January 10, 2025
Accepted: February 18, 2025
Published online: March 28, 2025
Processing time: 209 Days and 23.8 Hours
Occult pancreaticobiliary reflux (OPBR) is characterized by the absence of congenital anomalies at the pancreaticobiliary junction yet leads to altered bile composition and an increased incidence of gallbladder stones.
To explore the computed tomography (CT) imaging characteristics of gallbladder stones in patients diagnosed with OPBR.
We analyzed 362 patients undergoing cholecystectomy (November 2020 to January 2022). Intraoperative bile samples were assayed for amylase (> 110 U/L indicated OPBR). CT features, including stone density and visibility, were compared between 54 OPBR and 308 controls. Stone attenuation (HU) was measured under standardized conditions (uCT-780, 120 kVp, 160 mAs). Logistic regression and receiver operating characteristic curve analysis identified independent OPBR predictors, forming a validated nomogram.
OPBR patients exhibited significantly higher rates of CT-invisible stones (35.2% vs 12.3%) and uniform stones (87% vs 73.1%) along with lower overall stone density
CT imaging distinctly identifies gallbladder stone density, indicating a heightened risk of OPBR in patients with uniform and CT-invisible stones. Such imaging is crucial for preoperative assessments to evaluate potential recurrent biliary pathologies post-cholecystectomy.
Core Tip: This study presented a novel approach to diagnosing occult pancreaticobiliary reflux in patients with gallbladder stones using non-invasive computed tomography (CT) imaging. The findings highlighted that uniform and CT-invisible stones, along with lower stone density, are significant predictors of occult pancreaticobiliary reflux. The development of a predictive nomogram based on these imaging features offers clinicians an effective tool for early identification and intervention, potentially reducing the risk of recurrent biliary pathologies post-cholecystectomy. This innovative diagnostic model may enhance patient management and improve clinical outcomes in gallbladder disease.
- Citation: Qiu C, Xiang YK, Hu H, Da XB, Li G, Zhang YY, Zhang HL, Zhang C, Yang YL. Characterization of gallbladder stones associated with occult pancreaticobiliary reflux using computed tomography. World J Gastroenterol 2025; 31(12): 100855
- URL: https://www.wjgnet.com/1007-9327/full/v31/i12/100855.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i12.100855
Pancreaticobiliary reflux (PBR) refers to a pathological phenomenon characterized by the abnormal directional flow of bile and pancreatic juice predominantly observed in patients with pancreaticobiliary maljunction (PBM). PBM represents a congenital anatomical anomaly wherein the pancreatic duct and bile duct are interconnected outside the duodenal wall, typically resulting in the formation of an elongated common channel. Consequently, the sphincter of Oddi fails to exert a direct and effective influence over the pancreaticobiliary junction[1,2]. In such aberrant conditions, the existence of a pressure differential between the bile duct and pancreatic duct frequently results in the reflux of pancreatic juice into the biliary tract, consequently leading to markedly elevated levels of pancreatic enzymes in the bile. For diagnosing PBM, the presence of an abnormally long total channel or an anomalous conjunction between the pancreatic and bile ducts can be confirmed through direct cholangiography or magnetic resonance cholangiopancreatography (MRCP)[3,4].
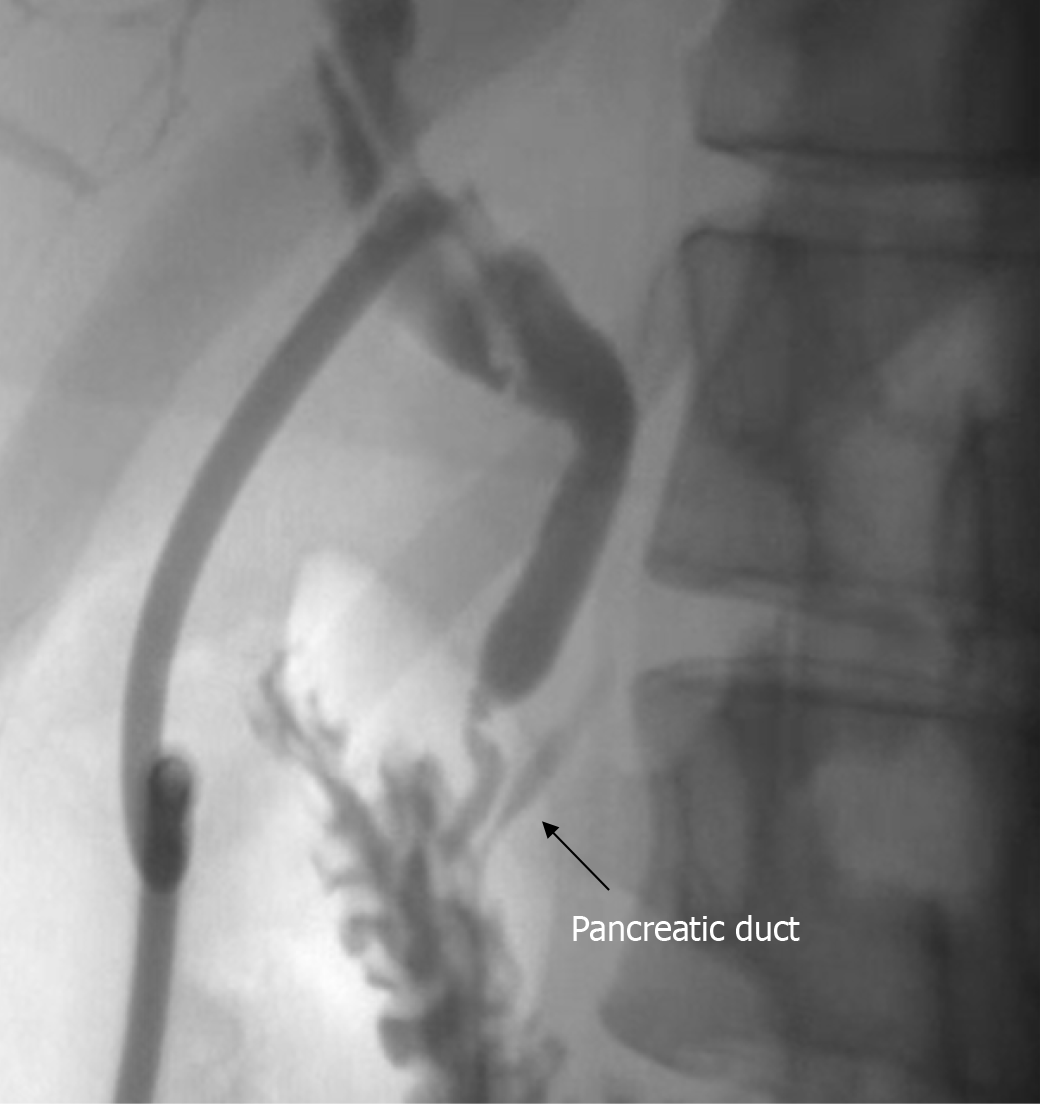
However, pancreatic juice-bile reflux can also be detected under conditions of a normal pancreaticobiliary junction (NPBJ). This phenomenon is referred to as occult pancreaticobiliary reflux (OPBR)[5-7]. A plausible explanation for the occurrence of OPBR is attributed to dysfunction of the sphincter of Oddi, bile duct, and pancreatic duct sphincter mechanisms[8-10]. The main diagnostic basis for OPBR includes: (1) The anatomical structure of the confluence of the bile and pancreatic ducts is normal; (2) The pancreatic duct can be visualized by antegrade cholangiography (Figure 1); and (3) Bile amylase level > the upper limit of normal for serum amylase. The author has elaborated on this part in previous articles[11].
Despite the distinct anatomical differences between PBM and OPBR, they share a similar pathological basis: The components of pancreatic juice entering the biliary tract and their interaction with bile may stimulate the gallbladder and bile duct mucosa, leading to chronic inflammation. This affects the reabsorption function of the gallbladder, resulting in a reduced capacity for gallbladder emptying, which ultimately leads to the formation of gallbladder and bile duct stones. Concurrently, long-term stimulation of the biliary epithelium can cause biliary epithelial cell dysplasia, metaplasia, atypical hyperplasia, and ultimately cancer[12-14].
While PBM can be detected through imaging due to congenital anatomical defects, predicting OPBR through non-invasive examinations is challenging, posing a significant challenge to current diagnostic imaging techniques. PBR constitutes one of the significant risk factors for gallbladder stones, with studies indicating that the prevalence of gallbladder stones in the Asian population ranges from 5% to 20%[15]. In a prospective study involving cholecystectomy, preoperative imaging confirmed the presence of PBR in 59% of 76 patients with gallbladder stones under conditions of NPBJ[9]. Utilizing a similar approach, Beltrán et al[8] detected PBR in 83.5% of patients with gallbladder stones. For this demographic even after undergoing cholecystectomy, the persistent existence of reflux maintains a high risk of developing common bile duct stones.
Currently, there are several methods to demonstrate the occurrence of PBR in patients with NPBJ: (1) Intravenous administration of secretin, which allows for pancreatic duct imaging via MRCP anterograde cholangiography. However, this indirect method may not be deemed necessary for benign gallbladder diseases; (2) Direct bile sampling from the gallbladder during cholecystectomy; and (3) Bile sampling from the gallbladder bile duct through endoscopic retrograde cholangiopancreatography, though this method lacks precision as the sphincter of Oddi is inevitably compromised during the procedure leading to pancreatic juice reflux and rendering the results inaccurate[16,17]. Current diagnostic tools for OPBR are invasive and costly, highlighting the need for non-invasive and reliable alternatives.
This study evaluated computed tomography (CT) imaging as a tool to identify OPBR-related gallbladder stone characteristics, aiming to provide clinicians with a practical diagnostic model. Identifying OPBR non-invasively could have profound implications for treatment strategies and patient management. Early diagnosis could help prevent complications such as recurrent common bile duct stones and biliary tract malignancies. Furthermore, it could guide clinicians in tailoring surgical approaches, optimizing follow-up plans, and implementing preventive measures for high-risk patients.
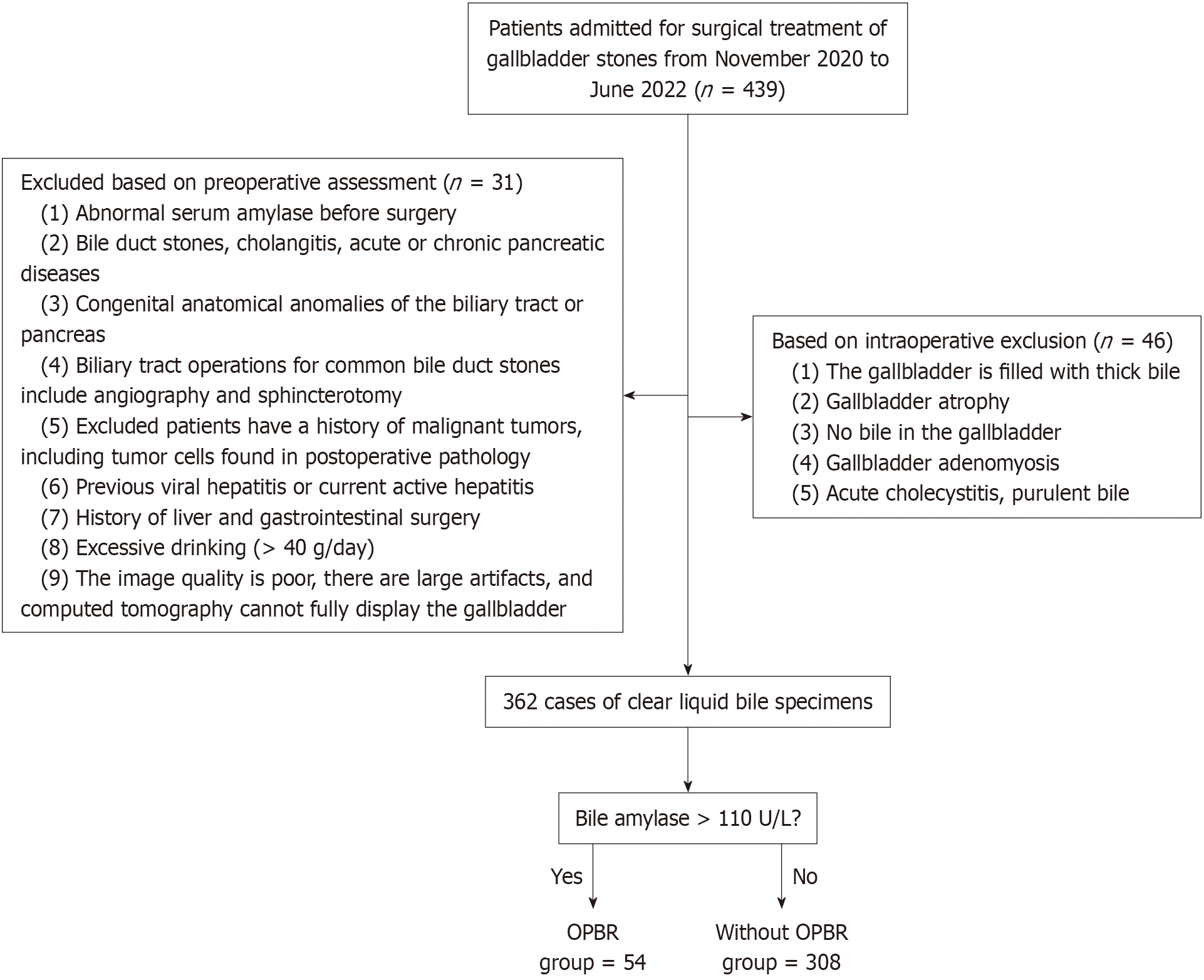
This study was authorized by the local ethics committee of East Hospital Affiliated to Tongji University (number: 2022-107). Patients consented to the extraction of bile samples during surgery and signed informed consent forms. All methods were carried out in accordance with relevant guidelines and regulations. The exclusion criteria for patients are depicted in Figure 2. The study included a total of 362 patients who underwent gallbladder stone surgery between November 2020 and January 2022. All patients were verified to have NPBJ through MRCP examination prior to surgery.
CT scanning was conducted using an 80-channel scanner (Uct-780; UNITED IMAGING, Shanghai). Prior to scanning, patients were trained to hold their breath at the end of inhalation to obtain optimal scan results. Patients were positioned supine with arms raised to minimize scanning artifacts. Beam collimation was set at 80 mm × 0.5 mm, with a helical pitch of 0.984, tube voltage at 120 kVp, and tube current at 160 mAs, utilizing automatic exposure control (iDose; Philips Healthcare). Images were reconstructed with a slice thickness and interval of 2 mm each. Following the scan, raw data were uploaded to a processing workstation for further extraction of imaging features.
All patient data were anonymized prior to analysis to ensure confidentiality and ethical compliance. Patient identifiers were replaced with unique codes, and all imaging and clinical data were stored in a secure, password-protected imaging database, with radiologists recording features using SOFT Windows. Image feature analysis was conducted through quantitative assessment by two certified thoracic radiologists, boasting 6 and 12 years of experience in abdominal CT imaging, respectively, who were blinded to clinical information and treatment plans.
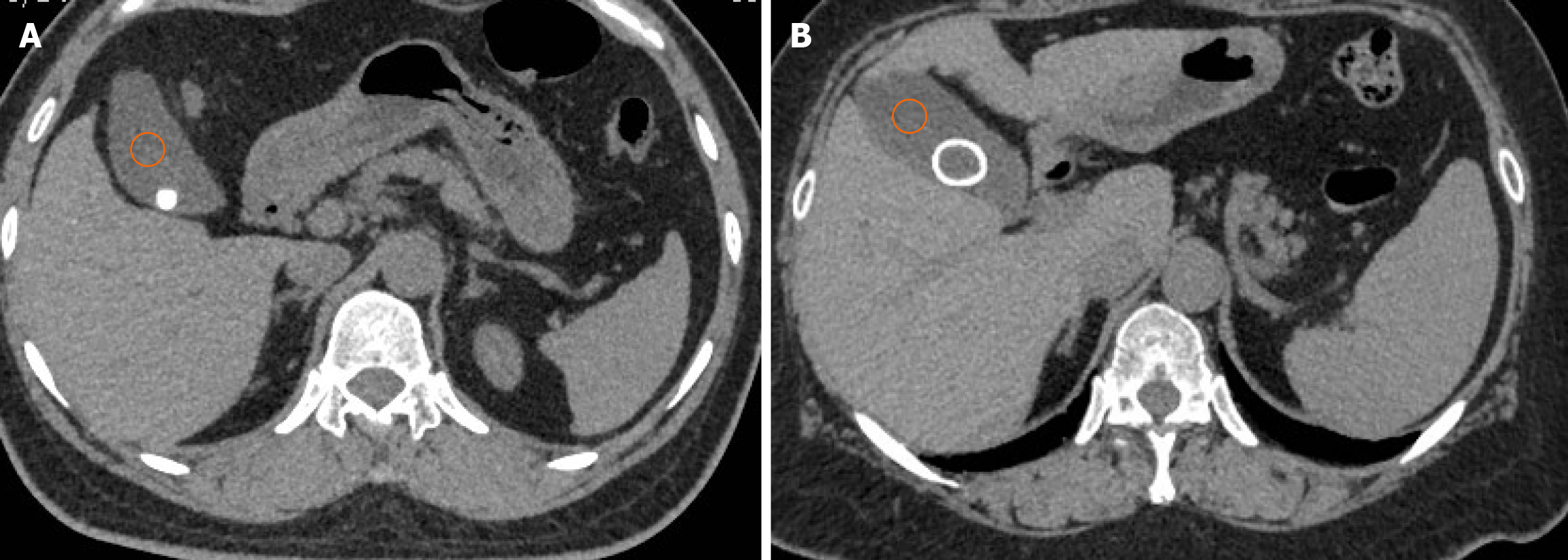
CT images were reviewed anonymously and randomly, with features recorded using SOFT Windows. The method for recording CT image features involved manually drawing three 2 cm2 regions of interest (ROIs) at two different locations within the right lobe of the liver (according to the Couinaud system, segments VII and VIII) to measure hepatic attenuation[18,19]. Splenic attenuation was measured by averaging the HU values of three 2 cm2 circular ROIs placed at three different locations within the middle third of the spleen. Placement of ROIs was designed to exclude visually identifiable hepatic vessels, biliary structures, and any focal liver lesions as well as maintaining a distance of at least 5-10 mm from the liver periphery[20]. The same method was employed to measure the HU values of bile, categorizing stones based on their characteristics into uniform stones (those with a uniformly consistent density, as shown in Figure 3A) and not uniform stones (those with uneven density, such as ring-like stones, as shown in Figure 3B), measuring the core, periphery, and overall HU values. CT-negative stones refer to stones that were not detected during imaging and were invisible to the naked eye. All CT image features were independently recorded by the two radiologists, with any discrepancies resolved through consensus.
We employed the most accurate method currently recognized for measuring PBR, which involves directly sampling bile from the gallbladder during cholecystectomy before any manipulation of Calot’s triangle or the common bile duct[21]. Serum samples were extracted within 48 h prior to surgery to assess related biochemical parameters such as amylase and lipase, ensuring that the patients’ serum amylase levels were within the normal range. Under aseptic conditions during surgery, 5 mL of bile was extracted and immediately stored at 4 °C for analysis the following day. Correspondingly, we did not disclose study details to the laboratory personnel and processed samples with Roche Cobas c702 (Roche Diagnostics, Basel, Switzerland); the normal range for serum amylase is 30-110 U/L. Calibration and quality control were performed daily according to the manufacturer’s guidelines to ensure precision and accuracy. To date, studies have shown that bile amylase levels remain at a lower range. Following the methodologies adopted by other research, our study considered that once bile amylase levels exceeded the normal plasma standards, it was indicative of the presence of PBR[17,21-23].
Statistical analyses were performed using SPSS version 25 (IBM, Armonk, NY, United States). Given the skewed distribution of the data, we chose to summarize continuous variables using the median and interquartile range, which better reflects the central tendency and spread of data in non-normally distributed variables. Comparisons were made using the Mann-Whitney U test. The Pearson χ2 test was utilized to compare categorical variables, which were described as percentages. To evaluate correlations between continuous variables, Spearman’s rank correlation analysis was used due to the non-normal distribution of data. Univariate logistic regression was employed to assess the association between clinical and imaging parameters and the presence of OPBR. Variables with a P value < 0.1 in univariate analysis were included in the multivariate logistic regression model. Receiver operating characteristic (ROC) curve analysis was undertaken to determine the optimal efficacy of two CT models in diagnosing OPBR. Analysis of nomograms, calibration curves, and decision curve analysis (DCA) was performed using R version 4.3.3 (http://www.rproject.org/).
In this study, the median age of the patients was 50 years. The median age of the OPBR group was significantly higher at 55 years compared with the control group (P = 0.03). Compared with the control group, the OPBR group showed significant increases in bile amylase, lipase, and free fatty acids. In terms of imaging features, the external density of stones and overall stone HU were significantly lower in the OPBR group compared with the control group (P = 0.002 and P = 0.01, respectively). Additionally, the OPBR group had a higher proportion of CT-negative stones, with 19 cases accounting for 35.2%. Uniform stones were also more prevalent in the OPBR group, with 47 cases accounting for 87.0% (Table 1). No significant differences were observed in gender, cholesterol, triglycerides, stone core, bile, liver, spleen HU, and the presence of fatty liver in relation to the occurrence of OPBR.
| All subjects | Without OPBR | With OPBR | P value | |
| Patients | 362 | 308 | 54 | - |
| Age (years) | 50 (38-60) | 50 (37-60) | 55 (45-62) | 0.030 |
| Gender | 0.370 | |||
| Male | 128 (35.40) | 106 (34.40) | 22 (40.70) | |
| Female | 234 (64.60) | 202 (65.60) | 32 (59.30) | |
| Bile amylase (U/L) | 10.00 (0-20.00) | 10.00 (0-20.00) | 525.00 (335.00-1367.50) | < 0.001 |
| Bile lipase (mmol/L) | 94.00 (48.00-198.00) | 83.00 (47.00-161.50) | 2264.30 (1258.00-5357.25) | < 0.001 |
| Bile free fatty acids (mmol/L) | 0.20 (0-0.50) | 0.20 (0-0.40) | 0.99 (0.40-2.04) | < 0.001 |
| Bile cholesterol (mmol/L) | 11.60 (8.40-15.40) | 11.70 (8.60-15.50) | 10.20 (5.68-15.33) | 0.200 |
| Bile triglycerides (mmol/L) | 3.40 (2.40-4.20) | 3.40 (2.40-4.20) | 3.20 (2.58-5.73) | 0.600 |
| Core density of stones (HU) | 130.33 (27.21-450.40) | 130.33 (25.79-451.55) | 130.18 (46.41-430.23) | 0.130 |
| External density of stones (HU) | 244.00 (114.13-503.70) | 254.63 (112.90-508.28) | 162.90 (114.64-519.69) | 0.002 |
| Overall density of stone (HU) | 167.55 (71.71-439.63) | 174.45 (70.81-450.85) | 139.60 (102.90-394.60) | 0.010 |
| Visibility of stones | < 0.001 | |||
| Yes | 305 (84.30) | 270 (87.70) | 35 (64.80) | |
| No | 57 (15.70) | 38 (12.30) | 19 (35.20) | |
| Characteristics of stones | 0.030 | |||
| Uniform | 272 (75.10) | 225 (73.10) | 47 (87.00) | |
| Not uniform | 90 (24.90) | 83 (26.90) | 7 (13.00) | |
| Bile density (HU) | 19.69 (13.16-25.88) | 20 (13.18-25.81) | 19.07 (11.46-29.46) | 0.770 |
| Liver density (HU) | 62.38 (57.38-65.82) | 62.24 (57.44-65.81) | 62.68 (54.87-66.41) | 0.960 |
| Spleen density (HU) | 51.91 (49.12-54.1) | 52.04 (49.80-54.18) | 51.45 (49.03-54.08) | 0.220 |
| Hepatic steatosis | 0.880 | |||
| No | 346 (95.60) | 284 (92.21) | 52 (96.30) | |
| Yes | 15 (4.14) | 13 (4.22) | 2 (3.70) |
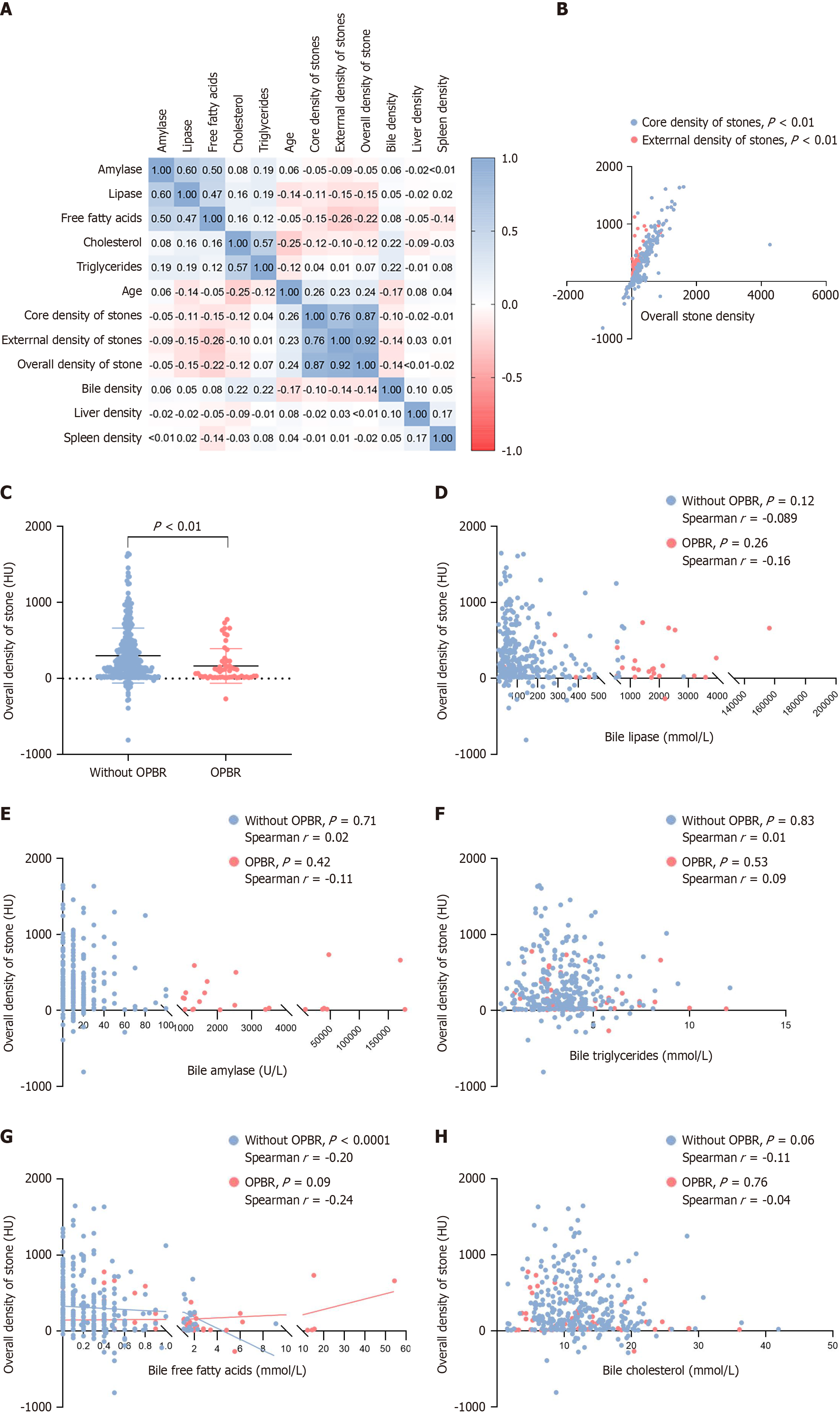
As depicted in Figure 4A, a clear correlation was observed among the stone periphery HU, stone core HU, and overall stone HU, with the Spearman correlation test confirming an absolute positive correlation between the stone periphery, core HU, and overall values (P < 0.01; Figure 4B). To eliminate the interference of collinearity, we selected the overall stone as the subject of study and found that in the OPBR group, the overall HU values of gallbladder stones were lower (P < 0.01; Figure 4C). Our research indicated that in the group analysis no correlation was observed between other components in the bile and the overall stone HU (Figure 4D-F). Furthermore, in the group of gallbladder stones without accompanying OPBR, there was a statistically significant negative correlation between the overall stone HU and the level of free fatty acids in the bile (P < 0.0001; Figure 4G). This trend suggests that the content of free fatty acids in the gallbladder luminal bile affects the density of the stone. Similarly, no significant correlation was found between bile cholesterol levels and overall stone HU (Figure 4H).
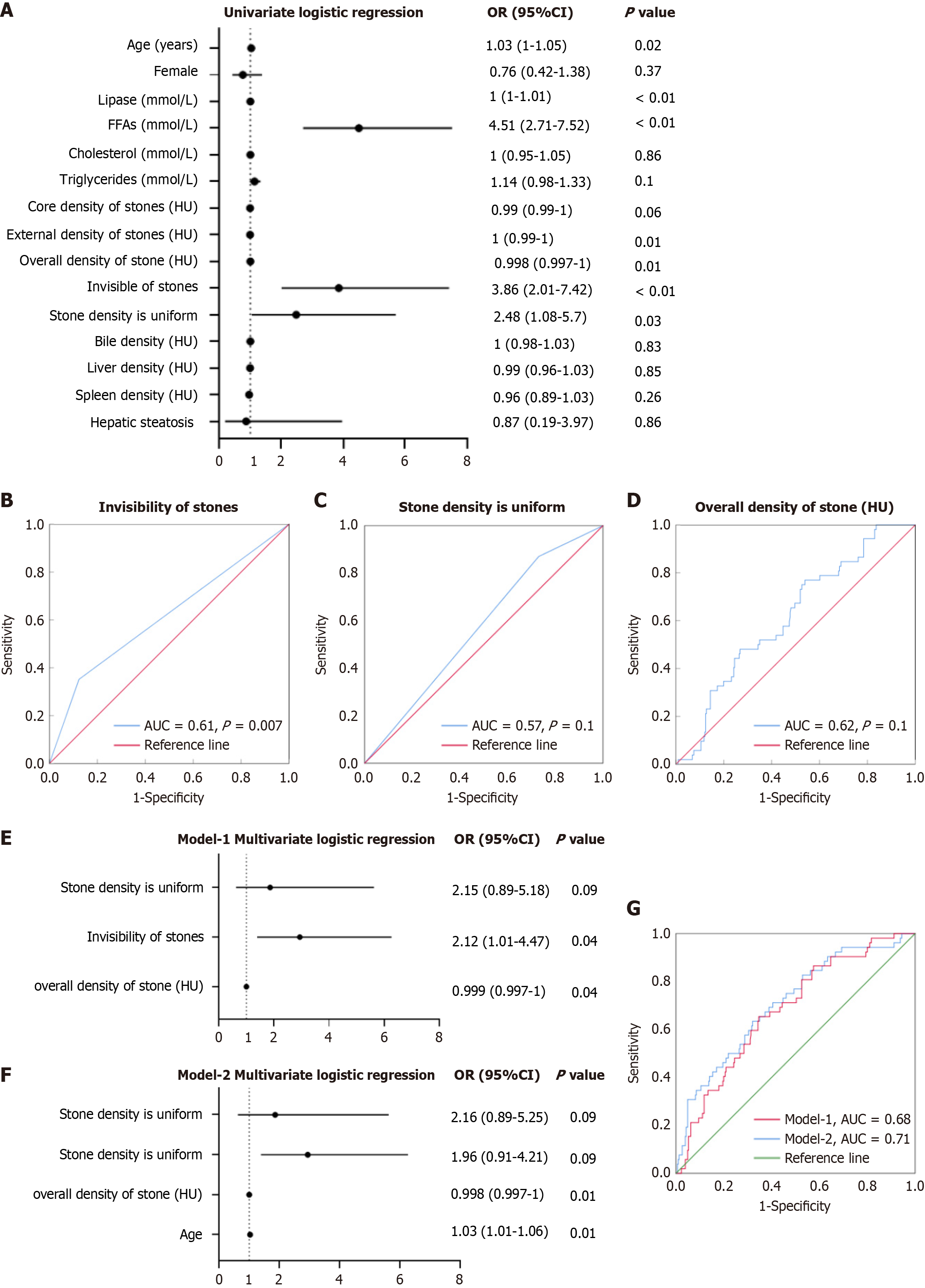
In the univariate logistic regression analysis aimed at assessing the correlation between clinical and imaging parameters and OPBR, age emerged as a strong predictive factor for OPBR [odds ratio (OR) = 1.03, 95% confidence interval (CI): 1.00-1.05, P = 0.02]. Lipase and free fatty acids were also significant indicators of reflux. Among imaging parameters, the HU value of stones indicated that lower HU values serve as risk factors for diagnosing OPBR. Non-contrast-enhanced stones (OR = 3.86, 95%CI: 2.01-7.42, P < 0.01) and uniform stones (OR = 2.48, 95%CI: 1.08-5.7, P = 0.03) also demonstrated a positive correlation with the risk of OPBR.
However, other factors such as gender, bile, liver, spleen, and the presence of a fatty liver did not show significant correlations with the risk of OPBR (Figure 5A). Subsequent evaluations of the efficacy of three different diagnostic indicators for discriminating OPBR were conducted. ROC analysis revealed that the first diagnostic factor had a certain predictive value [area under the curve (AUC) = 0.61, P = 0.007] (Figure 5B), the predictive efficacy of the second factor was not pronounced (AUC = 0.57, P = 0.1; Figure 5C), and the AUC value of the third factor was 0.62 (P = 0.01). The ROC curve revealed that the overall density of stones has a certain degree of predictive power in predicting the sensitivity and specificity of OPBR (Figure 5D).
Variables with a P value < 0.1 in univariate analysis were included in the multivariate logistic regression model. This threshold was chosen to ensure that potentially significant predictors were not excluded prematurely. Additionally, variables with high collinearity were excluded to avoid overfitting. Upon evaluating the multivariate logistic regression model to determine predictors of OPBR, we found that stone visibility and stone density demonstrated statistical predictive value in Model-1 (Figure 5E and F). When age was incorporated into Model-2, the overall diagnostic accuracy of the model improved (AUC increased from 0.68 to 0.71; Figure 5G).
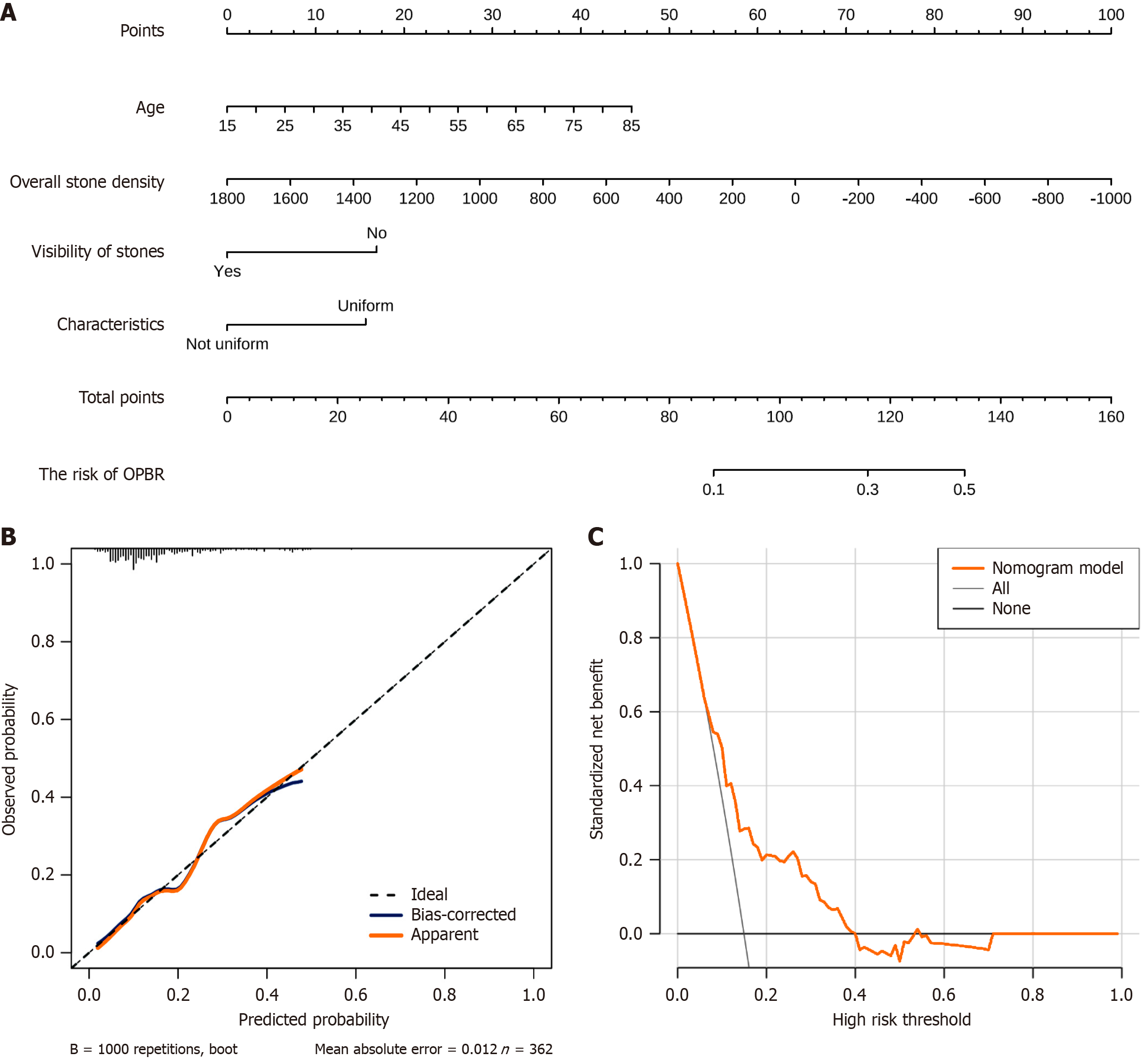
A multivariate analysis was utilized to construct a predictive nomogram for OPBR, with subsequent calibration and DCA conducted to evaluate the effectiveness of the model. The nomogram, based on patient age, overall stone density, visibility, and stone characteristics, provides a quantified risk assessment tool (Figure 6A). The calibration curve demonstrated a high degree of consistency between the predictions of the model after bias correction and the observed data, indicating good predictive accuracy of the model (Figure 6B). DCA further confirmed the standardized net benefit for patients across different clinical decision thresholds (Figure 6C).
It is acknowledged that under normal circumstances the sphincter of Oddi, which encircles the pancreaticobiliary junction at the end of the bile duct, regulates the flow of bile while simultaneously preventing bile reflux at the duodenal papilla. The pathological mechanism of PBM has been demonstrated to be due to the reflux of pancreatic enzymes (not controlled by the sphincter of Oddi) back into the bile duct and gallbladder. However, elevated levels of bile duct amylase can occur in patients with NPBJ. Current research indicates that the pathological phenomenon of PBR is caused by dysfunction of the sphincter of Oddi (abnormal sphincter tension and retrograde contractions)[21,24,25], leading to impaired bile flow and consequently reflux of pancreatic juice components.
Furthermore, studies by Sugiyama and Atomi[26] have identified a significant increase in amylase concentration around the bile duct in patients with periampullary diverticula. Endoscopic sphincterotomy temporarily induces the phenomenon of PBR, but the function gradually recovers after 7 days post-surgery[27]. Kamisawa et al[28] found that PBR can be detected in patients with a common channel > 5 mm (and the confluence of the pancreaticobiliary duct is within the duodenal wall).
A prospective study showed that endoscopic papillary balloon dilation temporarily diminishes sphincter function, potentially leading to PBR occurrence, but does not increase the risk of cancer[29]. The elevation of bile amylase levels due to PBR is highly associated with gallbladder cancer[6,9,30]. Phospholipase A2 in pancreatic juice can hydrolyze bile lecithin, producing lysophosphatidylcholine and free fatty acids. The increased activity of these substances in bile can cause direct and indirect damage to the bile duct epithelium, thus affecting the stability and function of the entire biliary system[10,23,31]. These findings comprehensively demonstrate that OPBR represents a functional disorder of pancreatic juice reflux under anatomically NPBJ conditions, indicating that dysfunction of the pancreaticobiliary system can lead to clinically significant consequences even in the absence of evident anatomical defects. Hence, the necessity for early diagnosis of OPBR needs to be emphasized. Although utilizing endoscopic retrograde cholangiopancreatography or MRCP to assess NPBJ is crucial for diagnosing OPBR, these costly and somewhat invasive imaging methods pose challenges when diagnosing patients with benign gallbladder diseases.
Non-enhanced CT is the preferred method for abdominal screening. Hence, in our analysis of 362 patients with gallbladder stones, the levels of bile amylase and CT characteristics of stones were examined, revealing that uniform stones and CT-negative gallbladder stones were more prevalent in the OPBR group, with the overall attenuation values of stones also being lower. The correlation between OPBR and age observed in our previous studies has been elucidated[32]. During the imaging review process radiologists calculated the core, periphery, and overall HU values of stones to categorize the majority of stone types observable by CT. The results indicated a strong positive correlation between the overall HU values of stones and their cores and peripheries. This suggests that factors influencing stone density, such as cholesterol content and calcium salt deposition, are relatively evenly distributed throughout the stone. Additionally, HU values measured by CT serve as a reliable and accurate method for assessing the characteristics of gallbladder stones.
To uncover the potential impact of OPBR on the density of gallstones, an interesting phenomenon was observed. Within the gallbladder high levels of free fatty acids were associated with lower stone density. This phenomenon aligns with the research, which suggests that an increase in the concentration of free fatty acids, acting as nucleating agents, can lead to a decrease in the solubility of cholesterol, thereby facilitating the formation of cholesterol stones[33,34]. This also reflects the abnormality in bile composition, suggesting an independent mechanism for stone formation that does not involve pancreatic juice reflux.
Further analysis was conducted to determine which variables independently predict the presence of PBR after controlling for other variables. The CT density of gallbladder stones, uniform stones, and CT-negative stones served as effective indicators for predicting the existence of OPBR. Multifactorial logistic regression analysis indicated that the overall density of stones is a risk factor for OPBR, while the HU values of the liver, spleen, and bile show no difference in relation to the presence of OPBR, indicating no direct correlation between OPBR and the density of these organs.
Based on these observations we constructed a predictive model. The diagnostic efficacy of two models (with and without considering age) for predicting OPBR was compared using the ROC curve. Model 2, which included age as a factor, had a higher AUC value than Model 1, indicating an improved diagnostic capability with the inclusion of age. The calibration curve evaluated the accuracy of the probability predictions of the model for OPBR. The blue line (bias-corrected) demonstrated the performance of the corrected model, which approaches the ideal diagonal line, indicating good prediction accuracy after correction. The DCA curve showed that the nomogram model (red line) offered a standardized net benefit that was higher than no intervention (gray line) across most thresholds, suggesting that our model will be valuable in clinical applications.
The integration of imaging-based parameters into predictive models represents a growing trend in personalized medicine. Cai et al[35] demonstrated the utility of a machine learning-based web calculator for predicting fractures post-vertebroplasty. Li et al[36] highlighted the potential of deep learning systems in improving decision-making for osteosarcoma patients. Building on these advancements our study introduced a nomogram that incorporates clinical and imaging data to improve risk stratification for OPBR by offering a practical and non-invasive solution for personalized patient care.
This study had multiple clinical implications including early detection and risk stratification. The preoperative identification of OPBR enables personalized management, such as closer postoperative monitoring and additional imaging to prevent recurrent biliary pathologies. Another clinical application is surgical planning. Recognizing OPBR may guide surgical decisions, including bile duct exploration or intraoperative cholangiography, to minimize postoperative complications. The preventive management and early diagnosis of OPBR helps mitigate risks of chronic inflammation and malignant transformation through targeted therapies and closer postoperative surveillance.
The predictive nomogram developed in this study could be a valuable tool for clinical practice. By incorporating CT imaging characteristics and clinical parameters, it enables non-invasive preoperative risk stratification, allowing clinicians to identify high-risk OPBR patients and plan targeted interventions. Postoperatively, the nomogram supports tailored surveillance strategies, helping to monitor high-risk patients for recurrent stones and preventing complications such as chronic inflammation or malignancy.
Although a robust model for predicting OPBR using clinical and imaging parameters exists, limitations remain. These include a small sample size and the potential undetected risk factors. CT resolution limitations also affect statistical power and generalizability. Large multicenter studies are needed to validate the model in different populations and healthcare settings. In addition, stone HU values may vary between devices and protocols. The model requires external validation in different settings. Future studies should focus on understanding the pathophysiology of OPBR and endoscopic treatment of OPBR in the clinic and validate the model in a larger cohort. In addition, exploring advanced imaging techniques could improve diagnostic accuracy.
Our study introduced a novel method for assessing the presence of OPBR in patients with gallbladder stones through CT imaging features, providing valuable clinical information for the early identification and intervention of PBR. It underscored the importance of considering OPBR as a diagnosis and its impact on the risk of recurrence of gallbladder stones when managing patients with gallbladder stones. Particularly for patients with low stone density and CT-negative stones, the potential risk of recurrent common bile duct stones post-cholecystectomy warrants vigilance. Therefore, the management strategy for this specific group requires more meticulous and individualized approaches, potentially including further evaluation and monitoring of the pancreaticobiliary junction.
| 1. | Kamisawa T, Kuruma S, Chiba K, Tabata T, Koizumi S, Kikuyama M. Biliary carcinogenesis in pancreaticobiliary maljunction. J Gastroenterol. 2017;52:158-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 61] [Article Influence: 6.8] [Reference Citation Analysis (5)] |
| 2. | Kamisawa T, Kaneko K, Itoi T, Ando H. Pancreaticobiliary maljunction and congenital biliary dilatation. Lancet Gastroenterol Hepatol. 2017;2:610-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 92] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 3. | Ono A, Arizono S, Isoda H, Togashi K. Imaging of Pancreaticobiliary Maljunction. Radiographics. 2020;40:378-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 4. | Kamisawa T, Ando H, Hamada Y, Fujii H, Koshinaga T, Urushihara N, Itoi T, Shimada H; Japanese Study Group on Pancreaticobiliary Maljunction. Diagnostic criteria for pancreaticobiliary maljunction 2013. J Hepatobiliary Pancreat Sci. 2014;21:159-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 89] [Article Influence: 7.4] [Reference Citation Analysis (2)] |
| 5. | Fujimoto T, Ohtsuka T, Nakashima Y, Gotoh Y, Date K, Mori Y, Sadakari Y, Takahata S, Oda Y, Nakamura M. Elevated bile amylase level without pancreaticobiliary maljunction is a risk factor for gallbladder carcinoma. J Hepatobiliary Pancreat Sci. 2017;24:103-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Horaguchi J, Fujita N, Noda Y, Kobayashi G, Ito K, Takasawa O, Obana T, Endo T, Nakahara K, Ishida K, Yonechi M, Hirasawa D, Suzuki T, Sugawara T, Ohhira T, Onochi K, Harada Y. Amylase levels in bile in patients with a morphologically normal pancreaticobiliary ductal arrangement. J Gastroenterol. 2008;43:305-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Yun SP, Lee JY, Jo HJ, Kim HS, Kim DH, Kim JH, Park SJ, Park DY, Seo HI. Long-term follow-up may be needed for pancreaticobiliary reflux in healthy adults. J Korean Surg Soc. 2013;84:101-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 8. | Beltrán MA, Contreras MA, Cruces KS. Pancreaticobiliary reflux in patients with and without cholelithiasis: is it a normal phenomenon? World J Surg. 2010;34:2915-2921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Beltrán MA, Vracko J, Cumsille MA, Cruces KS, Almonacid J, Danilova T. Occult pancreaticobiliary reflux in gallbladder cancer and benign gallbladder diseases. J Surg Oncol. 2007;96:26-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 10. | Beltrán MA. Pancreaticobiliary reflux in patients with a normal pancreaticobiliary junction: Pathologic implications. World J Gastroenterol. 2011;17:953-962. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 11. | Gallbladder-Preserving Surgery Committee; Endoscopy Specialist Branch of Chinese Medical Doctor Association; Expert Consensus Collaborative Group on Endoscopic Diagnosis and Treatment of the Benign Pancreaticobiliary Junction Disease. [Expert consensus on endoscopic diagnosis and treatment of the benign pancreaticobiliary junction disease (2023 edition)]. Zhonghua Yi Xue Za Zhi. 2023;103:3174-3179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 12. | Sai JK, Suyama M, Nobukawa B, Kubokawa Y, Yokomizo K, Sato N. Precancerous mucosal changes in the gallbladder of patients with occult pancreatobiliary reflux. Gastrointest Endosc. 2005;61:264-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Kamisawa T, Takuma K, Anjiki H, Egawa N, Kurata M, Honda G, Tsuruta K, Sasaki T. Pancreaticobiliary maljunction. Clin Gastroenterol Hepatol. 2009;7:S84-S88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 88] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 14. | Sakamoto H, Mutoh H, Ido K, Satoh S, Kumagai M, Hayakawa H, Tamada K, Sugano K. Intestinal metaplasia in gallbladder correlates with high amylase levels in bile in patients with a morphologically normal pancreaticobiliary duct. Hum Pathol. 2009;40:1762-1767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Lammert F, Gurusamy K, Ko CW, Miquel JF, Méndez-Sánchez N, Portincasa P, van Erpecum KJ, van Laarhoven CJ, Wang DQ. Gallstones. Nat Rev Dis Primers. 2016;2:16024. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 737] [Cited by in RCA: 595] [Article Influence: 59.5] [Reference Citation Analysis (3)] |
| 16. | Sai JK, Suyama M, Kubokawa Y, Tadokoro H, Sato N, Maehara T, Iida Y, Kojima K. Occult pancreatobiliary reflux in patients with a normal pancreaticobiliary junction. Gastrointest Endosc. 2003;57:364-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 17. | Itokawa F, Itoi T, Nakamura K, Sofuni A, Kakimi K, Moriyasu F, Tsuchida A, Aoki T. Assessment of occult pancreatobiliary reflux in patients with pancreaticobiliary disease by ERCP. J Gastroenterol. 2004;39:988-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 18. | Chung J, Park HS, Kim YJ, Yu MH, Park S, Jung SI. Association of Hepatic Steatosis Index with Nonalcoholic Fatty Liver Disease Diagnosed by Non-Enhanced CT in a Screening Population. Diagnostics (Basel). 2021;11:2168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 19. | Kim MG, Lee SS, Jun MJ, Byun J, Sung YS, Shin Y, Lee MG. Feasibility of non-enhanced CT for assessing longitudinal changes in hepatic steatosis. Medicine (Baltimore). 2019;98:e15606. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Jawahar A, Gonzalez B, Balasubramanian N, Adams W, Goldberg A. Comparison of correlations between lipid profile and different computed tomography fatty liver criteria in the setting of incidentally noted fatty liver on computed tomography examinations. Eur J Gastroenterol Hepatol. 2017;29:1389-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Beltrán MA. Current knowledge on pancreaticobiliary reflux in normal pancreaticobiliary junction. Int J Surg. 2012;10:190-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Donaldson LA, Joffe SN, McIntosh W, Brodie MJ. Amylase activity in human bile. Gut. 1979;20:216-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Xiang Y, Kong X, Zhang C, He C, Cai J, Lu R, Zhang B, Lu L, Yang Y. Free fatty acids and triglyceride change in the gallbladder bile of gallstone patients with pancreaticobiliary reflux. Lipids Health Dis. 2021;20:97. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 24. | Cicala M, Habib FI, Fiocca F, Pallotta N, Corazziari E. Increased sphincter of Oddi basal pressure in patients affected by gall stone disease: a role for biliary stasis and colicky pain? Gut. 2001;48:414-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Løtveit T, Osnes M, Aune S, Larsen S. Studies of the choledocho-duodenal sphincter in patients with and without juxta-papillary duodenal diverticula. Scand J Gastroenterol. 1980;15:875-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 26. | Sugiyama M, Atomi Y. Periampullary diverticula cause pancreatobiliary reflux. Scand J Gastroenterol. 2001;36:994-997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Sugiyama M, Atomi Y. Does endoscopic sphincterotomy cause prolonged pancreatobiliary reflux? Am J Gastroenterol. 1999;94:795-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 28. | Kamisawa T, Suyama M, Fujita N, Maguchi H, Hanada K, Ikeda S, Igarashi Y, Itoi T, Kida M, Honda G, Sai J, Horaguchi J, Takahashi K, Sasaki T, Takuma K, Itokawa F, Ando H, Takehara H; Committee of Diagnostic Criteria of The Japanese Study Group on Pancreaticobiliary. Pancreatobiliary reflux and the length of a common channel. J Hepatobiliary Pancreat Sci. 2010;17:865-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Sugiyama M, Atomi Y. Endoscopic papillary balloon dilation causes transient pancreatobiliary and duodenobiliary reflux. Gastrointest Endosc. 2004;60:186-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 30. | Sai JK, Suyama M, Nobukawa B, Kubokawa Y, Sato N. Severe dysplasia of the gallbladder associated with occult pancreatobiliary reflux. J Gastroenterol. 2005;40:756-760. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 31. | Qiu C, Xiang YK, Da XB, Zhang HL, Kong XY, Hou NZ, Zhang C, Tian FZ, Yang YL. Phospholipase A2 enzymes PLA2G2A and PLA2G12B as potential diagnostic and prognostic biomarkers in cholangiocarcinoma. World J Gastrointest Surg. 2024;16:289-306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 32. | Xiang Y, Qiu C, Hu H, Cai J, Da X, Kong X, Wang Y, He C, Zhang C, Yang Y. Age and incidence of occult pancreaticobiliary reflux in patients with benign gallbladder diseases. Scand J Gastroenterol. 2024;59:584-591. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 33. | Mingrone G, Greco AV, Passi S. The possible role of free fatty acids in the pathogenesis of cholesterol gallstones in man. Biochim Biophys Acta. 1983;751:138-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 34. | Halpern Z, Goldman G, Peled Y, Konikoff F, Rattan J, Lichtenberg D, Gilat T. Free fatty acids have nucleating effects in model biles. Liver. 1992;12:107-111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 35. | Cai S, Liu W, Cai X, Xu C, Hu Z, Quan X, Deng Y, Yao H, Chen B, Li W, Yin C, Xu Q. Predicting osteoporotic fractures post-vertebroplasty: a machine learning approach with a web-based calculator. BMC Surg. 2024;24:142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 36. | Li W, Dong Y, Liu W, Tang Z, Sun C, Lowe S, Chen S, Bentley R, Zhou Q, Xu C, Li W, Wang B, Wang H, Dong S, Hu Z, Liu Q, Cai X, Feng X, Zhao W, Yin C. A deep belief network-based clinical decision system for patients with osteosarcoma. Front Immunol. 2022;13:1003347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/