Published online Mar 28, 2025. doi: 10.3748/wjg.v31.i12.100155
Revised: November 26, 2024
Accepted: February 26, 2025
Published online: March 28, 2025
Processing time: 229 Days and 23.4 Hours
Evidence on adherence, diagnostic performance and impact on survival to intensive follow-up after surgery for colorectal cancer (CRC) is limited.
To analyze the diagnostic performance of surveillance colonoscopy, computed tomography (CT), and tumor markers (TMs) in detecting CRC recurrence or metastasis during follow-up after CRC resection. Secondary objectives included degree of adherence to clinical practice guidelines surveillance recommendations and factors associated with adherence and all-cause and CRC mortality.
The single-center retrospective cohort study including patients undergoing curative resection of stage I-III CRC during 2010-2015. Follow-up was performed using TMs every 6 months, yearly CT for 5 years, and colonoscopy at years 1 and 4. Demographic, primary tumor data, and results at follow-up were collected.
Of 574 included patients included, 153 had recurrences or metastases. Of this group, 136 (88.9%) were diagnosed by CT, 10 (6.5%) by CT and colonoscopy, and 7 (4.6%) by colonoscopy; only 67.8% showed TMs elevation. Adherence to follow-up recommendations was 68.8% for the first colonoscopy, 74% for the first CT scan, and 96.6% for the first blood test; these values declined over time. Younger age at diagnosis [odds ratio (OR) 0.93; 95%CI: 0.91-0.95], CRC stages I-II (OR 0.38; 95%CI: 0.24-0.61), and adherence to follow-up recommendations (OR 0.30; 95%CI: 0.20-0.46) were independently associated with lower risk for all-cause death at 5 years.
CT scan had the highest diagnostic yield. Adherence to follow-up recommendations was low and decreased during follow-up. Younger age at diagnosis, stage, and follow-up adherence were associated with lower 5-year mortality.
Core Tip: Post-surgical follow-up of colorectal cancer allows detection of local and distant recurrences. The incidence of metastasis or recurrence within the first 3-5 years after surgery is high. However, evidence on diagnostic yield of computed tomography (CT), colonoscopy and tumour markers (TMs) and impact on survival is limited. CT scan is associated with the highest yield for metastasis and recurrence detection. If follow-up is performed with blood test TMs only, an important proportion of recurrences would be missed. Compliance with intensive follow-up is an independent protective factor against mortality.
- Citation: Sala-Miquel N, Carrasco-Muñoz J, Bernabeu-Mira S, Mangas-Sanjuan C, Baile-Maxía S, Madero-Velázquez L, Ausina V, Yuste A, Gómez-González L, Romero Simó M, Zapater P, Jover R. Diagnostic yield of follow-up in patients undergoing surgery for non-metastatic colorectal cancer. World J Gastroenterol 2025; 31(12): 100155
- URL: https://www.wjgnet.com/1007-9327/full/v31/i12/100155.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i12.100155
Colorectal cancer (CRC) is the most frequent gastrointestinal neoplasm worldwide, the third most common in incidence in both sexes, and the tumor type responsible for the second highest number of deaths[1,2]. The incidence of metastasis or recurrence within the first 3-5 years after surgery is high. Some studies estimate that 60%-80% of local and metastatic recurrences are detected within the first 3 years[3-6]. Consequently, most medical associations recommend follow-up with an annual thoracoabdominal computed tomography (CT) scan for the first 3-5 years after surgery, along with blood tumor marker (TMs) testing every 3-6 months for the first 5 years and colonoscopy at 1 and 4 years after surgery, followed by longer intervals[6-8]. After 5 years, the probability of finding recurrences, metastases, or other tumors is very low[6,9,10].
The available evidence does not show, however, which postoperative surveillance modality has the best diagnostic yield. Different studies[11-17] have compared intensive surveillance, defined as follow-up with CT plus colonoscopy and TMs with non-intensive post-surgical follow-up (follow-up with TMs only, with fewer additional tests for assessment or more spaced out over time). These studies have not shown consistent differences between these two strategies in rate or time to recurrence, but intensive surveillance has been associated with a higher number of surgical interventions. Some studies have identified an association of intensive surveillance with a significant increase in survival[13].
Given the heterogeneity in recommendations for post-surgical follow-up of CRC and the limited evidence on adherence and performance, our main objective was to analyze and compare diagnostic performance among surveillance colonoscopies, CT, and TMs in detecting CRC recurrence or metastasis during follow-up. Secondary objectives were determining the degree of adherence to clinical practice guidelines for surveillance colonoscopies, CT, and TMs after CRC resection, and factors associated with adherence. We also analyzed follow-up factors associated with risks of CRC mortality and all-cause mortality.
We conducted a retrospective cohort study in patients who underwent curative resection of CRC between 2010 and 2015 at Hospital General Universitario Dr. Balmis and were followed up at this center for 5 years. Patients with stage I/II/III CRC were included, and those with previous CRC or stage IV CRC were excluded. CRC was staged according to the eighth revision (2017) of the tumor-node-metastasis (TNM) classification[18].
Our protocol followed current Spanish clinical practice guidelines recommendations and consisted of blood tests with carcinoembryonic antigen (CEA) and carbohydrate antigen (CA) 19-9 determination every 6 months for the first 5 years, annual thorax-abdomen-pelvis CT scan for the first 5 years, and colonoscopy at 1 year, 4 years, and every 5 years thereafter.
Baseline colonoscopy was considered as the colonoscopy performed within 6 months prior to surgery or within 6 months thereafter. The first surveillance colonoscopy corresponded to the procedure performed one year after surgery with a variation of ± 6 months (6-18 months). The second surveillance colonoscopy was performed 3 years after the previous colonoscopy, with the same deviation of ± 6 months (30-42 months); and the third surveillance colonoscopy was performed 5 years ± 6 months (54-66 months) after the previous colonoscopy. Unless high-risk lesions were detected that warranted closer surveillance. For calculation of patients who had or had not undergone the first and second surveillance colonoscopies, those who died within one year of surgery were excluded from the first colonoscopy dataset and those who died within 30 months of surgery were excluded from the second colonoscopy dataset.
Annual CT scans were considered as having been performed on an appropriate date if done within a window of ± 6 months (i.e., first CT at 6-18 months after surgery). For blood test analyses, the same time window was used. For calculation of adherence to CT scan or blood test follow-up, those who died within one year of surgery were excluded from the first CT scan or blood test data. Those who died before the subsequent tests also were excluded.
For the TMs diagnostic validity study, the gold standard for the detection of relapses was considered to be the combination of CT and colonoscopy. TMs results were considered to be pathological if one or both markers were elevated (CEA > 5 ng/mL or CA19.9 > 39 U/mL). For comparison of the results of both techniques, TMs performed < 6 months after a CT scan were considered.
The pathology department drew up a list including patients undergoing CRC resection between 2010 and 2015 together with the reports of the respective anatomopathological studies of the surgical specimen. For data collection, medical records were reviewed using the medical software available at our center. Sociodemographic and primary tumor data, technical and anatomopathological data from baseline, and surveillance colonoscopy data were collected. The research protocol was approved by the Ethics Committee of the Hospital General Universitario Dr. Balmis in 2020, which made it possible to collect data anonymously.
For descriptive frequency analysis, categorical variables were described as proportions and quantitative variables with mean and SD. Comparisons were performed using Pearson's χ2 test for the relationship between qualitative variables and Student’s t-test for the comparison of quantitative variables. To determine the effect of the different independent variables, we conducted multivariate binary logistic regression with variables associated with the outcome in the univariate analysis (P < 0.07). Kaplan-Meier survival analysis was used to calculate mean time to metastasis/relapse and outcome. For the estimates, significance was set at 0.05, and statistical precision was established using 95%CI. Data were analyzed by intention to treat, and all calculations were performed using IBM SPSS Statistics (ver. 25.0).
Data for a total of 574 patients were reviewed and finally included in the study. CRC was more frequent in men (60.5%), and the mean age for the overall cohort was 70 years (SD: 11.36). Most CRCs were diagnosed at TNM stage II (224 cases, 38.5%) and stage III (221 cases, 39%), and the most common location was the left colon (226 cases, 39.4%), followed by the rectum (160 cases, 27.9%) and right colon (153 cases, 26.7%). Laparoscopic surgery was performed in 133 (23.2%) cases. Preoperative complications (intestinal obstruction or perforation) occurred in 70 patients (12.2%), and postoperative complications classified as Dindo et al[19] ≥ 2 occurred in 191 (33.3%) cases. A total of 207 patients (36.1%) received adjuvant chemotherapy, and 71 (12.4%) received neoadjuvant chemotherapy and radiotherapy (Table 1).
| Parameters | Value |
| Age, years, mean ± SD | 70.1 ± 11.36 |
| Sex | |
| Male | 347 (60.5) |
| Female | 227 (39.5) |
| TNM stage | |
| I | 125 (21.8) |
| II | 227 (39.5) |
| III | 222 (38.7) |
| Location | |
| Right colon | 153 (26.7) |
| Transverse colon | 35 (6.1) |
| Left colon | 226 (39.4) |
| Rectum | 160 (27.8) |
| Type of surgery | |
| Right hemicolectomy | 165 (28.8) |
| Left hemicolectomy/sigmoidectomy | 204 (35.6) |
| Anterior rectal resection | 140 (24.4) |
| Abdomino-peritoneal amputation | 26 (4.5) |
| Other | 39 (6.8) |
| Laparoscopic surgery | |
| Yes | 133 (23.2) |
| No | 441 (76.8) |
| Pre-operative complications | |
| Intestinal perforation | 22 (3.8) |
| Intestinal occlusion | 48 (8.4) |
| Postoperative complications | |
| Suture dehiscence | 87 (15.2) |
| Paralytic ileus | 28 (4.9) |
| Eventration | 14 (2.4) |
| Intra-abdominal infection | 20 (3.5) |
| Other | 42 (7.3) |
| Adjuvant chemotherapy | |
| Yes | 207 (36.1) |
| No | 367 (63.9) |
| Neoadjuvant radio-chemotherapy | |
| Yes | 71 (12.4) |
| No | 503 (87.6) |
A total of 153 recurrences or metastases were detected in the 574 (26.6%) patients analyzed. Of these, 136 (88.9%) recurrences or metastases were detected exclusively by CT, 10 (6.5%) by CT and colonoscopy, and 7 (4.6%) only by colonoscopy. A total of 25 (16.3%) cases were local recurrences, while 128 (83.7%) were cases of distant metastases. Table 2 shows the rate of detection of metastasis and recurrences by the different CT scans, colonoscopies, and blood tests performed during follow-up. Most of these recurrences or metastases were found in the first 3 years of follow-up (143 cases, 89.4%). The most metastases or recurrences were detected on the first CT scan (86 cases, 56.2%). Rates were similar for colonoscopy, with 14 cases (82.4%) of recurrence or metachronous CRC detected in the first 3 years of follow-up, 9 of which (52.9%) were detected at the first colonoscopy.
| Liver | Lung | Peritoneal | Anastomotic recurrence | Metachronous CRC | Other | Several locations | Total | |
| CT diagnostic | ||||||||
| CT 1 | 34 | 16 | 6 | 7 | 3 | 3 | 13 | 86 |
| CT 2 | 8 | 10 | 4 | 4 | 1 | 5 | 7 | 42 |
| CT 3 | 2 | 3 | 0 | 2 | 1 | 2 | 5 | 15 |
| CT 4 | 0 | 4 | 0 | 0 | 0 | 2 | 2 | 8 |
| CT 5 | 0 | 1 | 0 | 0 | 0 | 1 | 0 | 2 |
| Total | 44 | 34 | 10 | 13 | 5 | 13 | 27 | 146 |
| Median detection time (months) | 12 (8.8-15.2) | 22 (16.3-27.7) | 13 (6.8-19.2) | 20 (16.8-23.2) | 32 (13.2-50.8) | 26 (16.6-21.4) | ||
| Colonoscopy diagnosis | ||||||||
| Colonoscopy 1 | 7 | 3 | ||||||
| Colonoscopy 2 | 4 | 0 | ||||||
| Colonoscopy 3 | 0 | 3 | ||||||
| Total | 11 | 6 | ||||||
| Median detection time (months) | 22 (18-39.5) | |||||||
| TMs elevation | ||||||||
| Yes | 37 | 14 | 6 | 16 | 7 | 21 | ||
| No | 5 | 20 | 4 | 9 | 5 | 5 | ||
| % Yes (P = 0.01) | 88.1 | 41.2 | 60.0 | 64.0 | 58.3 | 80.8 | ||
Regarding the diagnostic yield of TMs, TMs were elevated in 67.8% of patients with recurrence/metastasis. This rate yielded a sensitivity of 67.8%, a specificity of 96.4%, a positive predictive value of 89.3%, and a negative predictive value of 86.4% (Table 1 and Supplementary Table 1). We also observed significant differences based on the location of metastases. TMs were elevated in 88% of cases of liver metastases but in only 41% of lung metastases (Table 2).
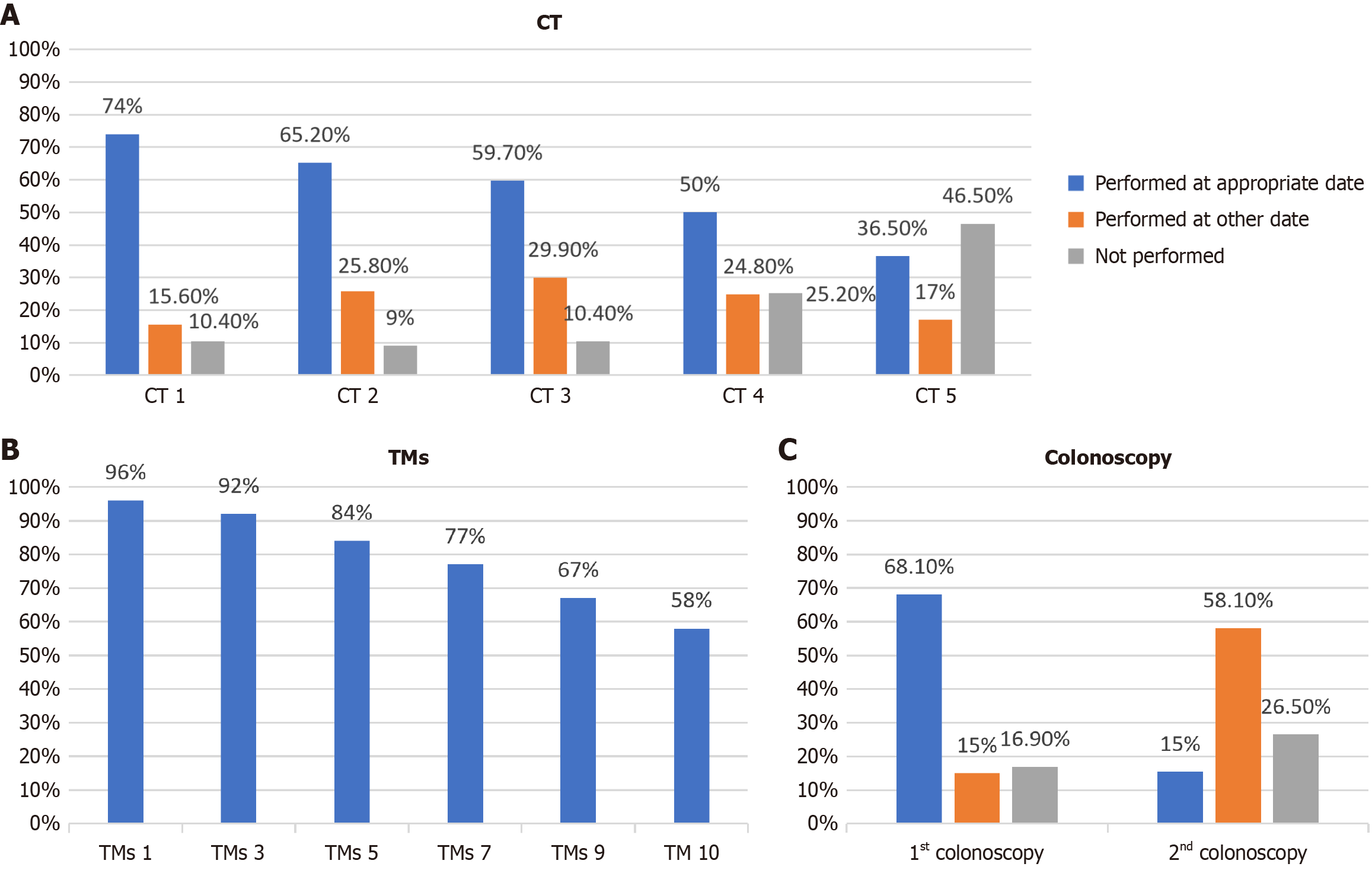
A total of 49 patients died or lost follow-up in the first 12 months after surgery and were not included in the follow-up. Adherence to CT scan, colonoscopy, and TMs guidance in the remaining 525 patients with follow-up is shown in Figure 1. A decline in adherence to surveillance was observed for all modalities. Adherence to CT scanning was 89.6% for the first examination, although only 74% were performed during 6-18 months after surgery (Figure 1A). Adherence decreased progressively, with only 36.5% fulfilling the CT guidance at year 5 (Figure 1A). Regarding TMs, a similar decrease was observed over time, from 96.6% at the first blood test to 58.3% by the tenth (Figure 1B). For colonoscopy, adherence for the first examination was 67.3%, of which 15% were performed after 18 months. A total of 16.9% of patients did not receive any surveillance with colonoscopy. Only 15.0% underwent the second surveillance colonoscopy at the time recommended by guidelines (Figure 1C).
The factors related with not having a first CT scan were older age, type of surgery, and not use of adjuvant chemotherapy or neoadjuvant radio-chemotherapy. Factors related to lack of adherence to the first surveillance colonoscopy were older age, TNM stage III, laparotomy, postoperative complications, and not requiring use of adjuvant chemotherapy or neoadjuvant radio-chemotherapy (Table 3). In multivariate analysis, older age at diagnosis and the use of adjuvant chemotherapy were independent factors for not having a first CT scan or a first surveillance colonoscopy. In addition, stage III CRC and the use of laparotomy also were independently associated with not having a first colonoscopy (Table 3).
| The 1st surveillance colonoscopy | P value | Multivariate analysis OR (95%CI) | P value | CT 1 | P value | Multivariate analysis OR (95%CI) | P value | ||||
| Yes | No | Yes | No | ||||||||
| Average age (Number of patients) | 66.97 (394) | 76.69 (131) | < 0.001 | 0.91 (0.89-0.94) | < 0.001 | 68.7 (483) | 76.6 (56) | < 0.001 | 1.06 (1.03-1.10) | < 0.001 | |
| Sex | Male | 245 (76.6) | 75 (23.4) | 0.32 | 299 (91.2) | 29 (8.8) | 0.14 | ||||
| Female | 149 (72.7) | 56 (27.3) | 184 (87.2) | 27 (12.8) | |||||||
| TNM stage | I-II | 248 (77.3) | 71 (22.3) | 0.05 | 1 | 298 (90.0) | 33 (10.0) | 0.69 | |||
| III | 141 (70.1) | 60 (29.9) | 2.47 (1.46-4.18) | < 0.001 | 185 (88.9) | 23 (11.1) | |||||
| Location | Right colon | 119 (73.0) | 44 (27.0) | 0.47 | 146 (85.9) | 24 (14.1) | 0.05 | 1 | |||
| Left colon | 275 (76.0) | 87 (24.0) | 337 (91.3) | 32 (8.7) | 0.80 (0.44-0.15) | 0.46 | |||||
| Surgery | Laparoscopy | 111 (87.4) | 16 (12.6) | < 0.001 | 1 | 122 (94.6) | 7 (5.4) | 0.03 | 1 | ||
| Laparotomy | 283 (71.1) | 115 (28.9) | 2.59 (1.43-4.78) | 0.002 | 361 (88.0) | 49 (12.0) | 2.03 (0.87-4.71) | 0.10 | |||
| Perioperative complications | Yes | 42 (65.6) | 22 (34.4) | 0.06 | 1 | 59 (90.8) | 6 (9.2) | 0.68 | |||
| No | 352 (76.4) | 109 (23.6) | 0.99 (0.52-1.88) | 0.98 | 424 (89.5) | 50 (10.5) | |||||
| Postoperative complications | Yes | 130 (68.8) | 59 (31.2) | 0.01 | 1 | 173 (89.2) | 21 (10.8) | 0.46 | |||
| No | 264 (78.6) | 72 (21.4) | 0.76 (0.48-1.20) | 0.24 | 310 (89.9) | 35 (10.1) | |||||
| Adjuvant chemotherapy | Yes | 173 (84.8) | 31 (15.2) | < 0.001 | 2.11 (1.18-3.78) | 0.01 | 197 (96.1) | 8 (3.9) | < 0.001 | 2.46 (1.09-5.54) | 0.03 |
| No | 221 (68.8) | 100 (31.2) | 1 | 286 (85.6) | 48 (14.4) | 1 | |||||
| Neoadjuvant radio-chemotherapy | Yes | 63 (91.3) | 6 (8.7) | < 0.001 | 2.18 (0.87-5.50) | 0.01 | 69 (97.2) | 2 (2.8) | 0.03 | 2.29 (0.51-10.16) | 0.28 |
| No | 331 (72.6) | 125 (27.4) | 1 | 414 (88.5) | 54 (11.5) | 1 | |||||
During follow-up, 237 patients (41.3%) died, 98 of them (17.1%) because of the CRC. Factors related to CRC mortality were age, TNM stage, perioperative complications, and lack of adherence to the follow-up recommendations. In multivariate analysis, age at diagnosis (71.5 years vs 66.3 years, OR: 0.97, 95%CI: 0.94-0.99), stage (13.0% of deaths with stages I-II vs 37.0% with stage III, OR: 0.26, 95%CI: 0.15-0.46), and lack of adherence to the recommendations (12.5% of deaths with adherence vs 36.7% without, OR: 0.35, 95%CI: 0.21-0.59) persisted as independent factors (Table 4).
| All-cause mortality | P value | Multivariate analysis, OR (95%CI) | P value | CCR mortality | P value | Multivariate analysis, OR (95%CI) | P value | |||
| Yes | No | Yes | No | |||||||
| Sex | ||||||||||
| Male | 144 (41.5) | 203 (58.5) | 0.9 | 61 (17.6) | 286 (82.4) | 0.7 | ||||
| Female | 93 (41.0) | 134 (59.0) | 37 (16.3) | 190 (83.7) | ||||||
| Average age | 75.3 | 66.3 | < 0.001 | 0.93 (0.91-0.95) | < 0.001 | 71.5 | 66.3 | < 0.001 | 0.97 (0.94-0.99) | 0.05 |
| TNM stage | ||||||||||
| I-II | 125 (35.4) | 228 (64.6) | < 0.001 | 0.38 (0.24-0.61) | < 0.001 | 34 (9.6) | 319 (90.4) | < 0.001 | 0.26 (0.15-0.46) | < 0.001 |
| III | 112 (50.7) | 109 (49.3) | 1 | 64 (29.0) | 157 (71.0) | 1 | ||||
| Location | ||||||||||
| Right hemicolon | 84 (44.7) | 104 (55.3) | 0.25 | 29 (15.4) | 159 (84.6) | 0.46 | ||||
| Left hemicolon | 153 (39.6) | 233 (60.4) | 69 (17.9) | 317 (82.1) | ||||||
| Surgery | ||||||||||
| Laparoscopy | 42 (31.6) | 91 (68.4) | 0.01 | 0.72 (0.44-1.17) | 0.19 | 17 (12.8) | 116 (87.2) | 0.13 | 0.68 (0.36-1.27) | 0.22 |
| Laparotomy | 195 (44.2) | 246 (55.8) | 1 | 81 (18.4) | 360 (81.6) | 1 | ||||
| Perioperative complications | ||||||||||
| Yes | 36 (51.4) | 34 (48.6) | 0.07 | 1.14 (0.62-2.09) | 0.68 | 20 (28.6) | 50 (71.4) | 0.01 | 1.72 (0.91-3.27) | 0.1 |
| No | 201 (39.9) | 303 (60.1) | 1 | 78 (15.5) | 426 (84.5) | 1 | ||||
| Postoperative complications | ||||||||||
| Yes | 113 (51.6) | 106 (48.4) | < 0.001 | 1.41 (0.93-2.14) | 0.1 | 42 (19.2) | 117 (80.8) | 0.29 | 1.25 (0.72-2.04) | 0.37 |
| No | 124 (34.9) | 231 (65.1) | 1 | 56 (15.8) | 299 (84.2) | 1 | ||||
| Adjuvant chemotherapy | ||||||||||
| Yes | 71 (34.3) | 136 (65.7) | 0.01 | 1.04 (0.64-1.74) | 0.84 | 50 (26.9) | 136 (73.1) | 0.06 | 1.60 (0.95-2.72) | 0.08 |
| No | 166 (45.2) | 201 (54.8) | 1 | 48 (19.3) | 201 (80.7) | 1 | ||||
| Neoadjuvant chemo-radiotherapy | ||||||||||
| Yes | 21 (29.6) | 50 (70.4) | 0.03 | 1.14 (0.61-2.31) | 0.69 | 12 (19.4) | 50 (80.6) | 0.518 | ||
| No | 216 (42.9) | 287 (57.1) | 1 | 86 (23.1) | 287 (76.9) | |||||
| Adherence to follow-up recommendations | ||||||||||
| Yes | 62 (21.6) | 225 (78.4) | < 0.001 | 0.30 (0.20-0.46) | < 0.001 | 32 (12.5) | 225 (87.5) | < 0.001 | 0.35 (0.21-0.59) | < 0.001 |
| No | 141 (55.7) | 112 (44.3) | 1 | 65 (36.7) | 112 (63.3) | 1 | ||||
Factors associated with all-cause mortality were age, TNM stage, type of surgery, postoperative complications, use of adjuvant chemotherapy or neoadjuvant radio-chemotherapy, and lack of adherence to the follow-up recommendations. In multivariate analysis, persistent independent risk factors were age at diagnosis (75.3 years vs 66.3 years, OR: 0.93, 95%CI: 0.91-0.95), stage (35.4% of deaths with stages I-II vs 50.7% with stage III, OR 0.38, 95%CI: 0.24-0.61), and lack of adherence to recommendations (21.6% of deaths with adherence vs 55.7% without, OR 0.30, 95%CI: 0.20-0.46; Table 4).
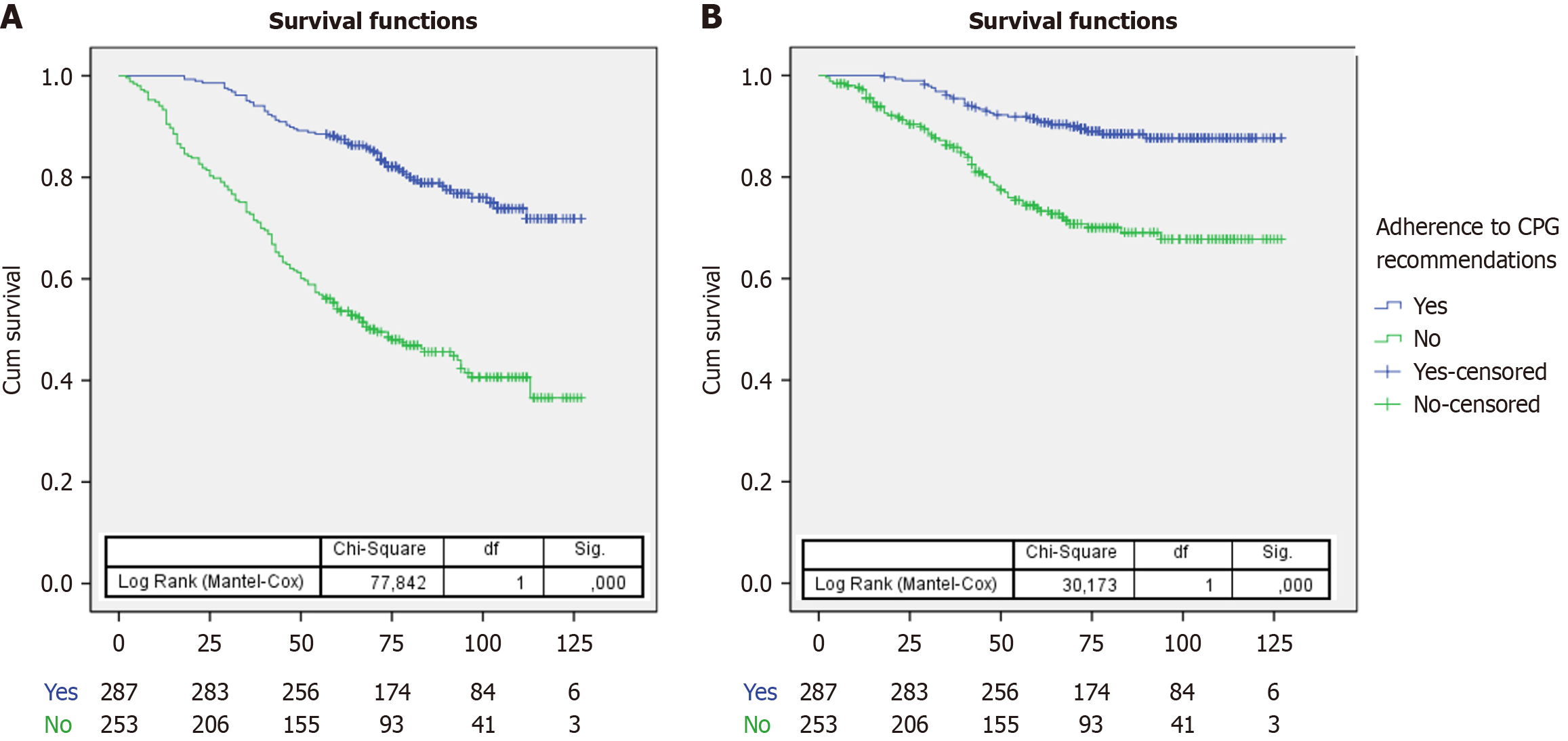
In the Kaplan-Meier survival analysis representing the influence of follow-up recommendations adherence on all-cause mortality, patients whose follow-up was not adherent died earlier (mean 75.8 months, 95%CI: 70.0-81.6) than those whose follow-up was adherent (mean 109.5 months, 95%CI: 105.7-113.4, log-rank 77.84, P < 0.0001). Similar results were obtained in the analysis of the influence of recommendations adherence on CRC deaths, with a higher number appearing earlier and consistently in patients without adherence (mean 99.1 months, 95%CI: 93.4-104.9) compared to patients with adherence (mean 117.3 months, 95%CI: 114.1-120.5, log-rank 30.17, P < 0.0001; Figure 2).
Our findings show the superior diagnostic efficacy of CT scans, detecting more than 90% of recurrences or metastases after CRC resection. Colonoscopy plays a secondary role, focusing on preventing metachronous disease through adenoma resection. Regarding TMs, this follow-up strategy failed to detect around 40% of recurrences or metastases, especially lung metastases. Suboptimal adherence to follow-up, decreasing over time, was observed. Notably, adhering to recommended follow-up significantly reduced the risk of CRC and all-cause mortality independently. Our results support the use of intensive follow-up with CT and colonoscopy after resection of non-metastatic CRC.
After definitive treatment of CRC[20,21], surveillance is recommended with the aim of improving disease-specific and overall survival by early detection of disease recurrence or a second primary cancer to allow for potentially curative surgery. However, the optimal surveillance strategy is unknown, and data from randomized trials have failed to demonstrate a significant survival benefit from intensive surveillance[12,14-16]. Systematic reviews are contradictory regarding the benefits of more intensive surveillance[11,13,17]. Data on which tests offer the most benefit and how often they should be performed are limited, and the survival benefit of surveillance in contemporary practice is unknown. National and international consensus guidelines vary, although many still recommend frequent testing, and guidelines adherence is inconsistent, resulting in both overtesting and undertesting[9,22]. The lack of consensus regarding surveillance intervals and diagnostic modalities has resulted in variations in surveillance policies across different guidelines, institutions, and individual healthcare providers. Although the frequency at which intensive testing could detect recurrence earlier and lead to successful intervention is unknown, recent evidence suggests that an accelerated cadence would have little clinical impact[12,14-16].
Our results support the use of colonoscopy and CT in surveillance, especially in the first 3 years after surgery, with CT having the highest diagnostic yield. In this cohort, if only TMs were to have been performed, 37.2% of recurrences would have gone undetected. This figure would have been much higher for pulmonary sites, with almost 60% of cases potentially missed. According to the present data, TMs are insufficient for follow-up in these patients. Lipská et al[23] evaluated CEA and CA19.9 levels at the time of recurrence detection by CT and found that in almost one-third of cases, these marker levels did not increase. Jones et al[24] obtained similar results, finding that 92.3% of recurrences were detected by CT but that only 57.7% of these cases involved increased CEA. Ongoing and already published studies[25,26] with circulant tumor cells and other biomarkers will probably improve the performance in detection of recurrences and distant metastasis.
The yield of colonoscopy alone for detecting CRC recurrences was low, at only 17 cases with recurrence or metachronous CRC, most of them also detected by CT scan. These recurrences were mainly found at the first colonoscopy at one year after surgery and were predominantly synchronous CRCs missed at the baseline colonoscopy. This rate is consistent with that previously described in the literature[6], with all studies highlighting the role of one-year surveillance in detecting previously missed cancers. The finding that more than 60% of non-anastomotic CRC recurrences were detected at stage I/II further supports the value of endoscopic surveillance, especially in the early high-risk period. The vast majority of anastomotic recurrences were found with colonoscopy shortly after surgery, in the first 24 months of follow-up[6].
Regarding adherence to follow-up, our results show low levels of adherence to recommendations. Adherence to surveillance guidelines after surgical treatment of CRC is quite variable (12%-87%)[27], lower for colonoscopy than for CT scan or blood tests, and declines over time. Surveillance may pose risks for overdiagnosis and the need for invasive procedures. On the other hand, post-treatment CRC surveillance monitors patients for recurrence, promotes prevention and health maintenance, and provides important emotional and psychosocial support. Globally, from a patient point of view, follow-up provides emotional reassurance of being cancer-free. Patients have reported high satisfaction with follow-up and believed that continued follow-up was important for detecting recurrence[28]. Regarding CT surveillance, 74% of patients in our cohort underwent the first CT scan following established recommendations, but this percentage decreased with later scans. A similar pattern was observed in the analytical follow-up of TMs, declining from 96% adherence to the first blood test recommendation to 58% at the end of follow-up. These results also are concordant with previously reported findings that adherence to the different guidelines ranges from 60% to 90% for blood tests with TMSs and it is 50% to 70% for CT[24,27,29].
It is also possible that difficulty in adhering to follow-up recommendations may relate to patient factors that contribute to a variable likelihood of success, such as increased comorbidities that can sometimes be a limitation to accessing tests such as colonoscopy. Stephens and Fahy[30] found that patients with more comorbidities or poorer nutritional status had less adherence to surveillance colonoscopy. Other factors associated with non-adherence to recommendations in the current study included older age at diagnosis, no adjuvant chemotherapy, stage III disease, or undergoing laparotomy. Stage III involves higher morbidity, which could be a contraindication to colonoscopy[31], and laparoscopy facilitates better adherence to surveillance recommendations because it entails a lower number of postoperative complications as well as better recovery after CRC surgery[32]. In addition, adherence to follow-up recommendations is an independent factor in lower CRC and all-cause mortality. This association may be explained by earlier detection of metastasis or recurrence and by other benefits such as better care and follow-up that may improve the management of other comorbidities or early detection of other conditions. In addition, it is known that patients whose follow-up is not adherent to recommendations more often have factors linked to a less healthy lifestyle. Innovative follow-up management tools or educational programs can increase patient understanding of the significance of follow-up visits. Additionally, offering personalized follow-up plans can boost patient engagement and compliance[33,34].
Limitations of this study are that it was retrospective and single center, with access limited to clinical information collected in only one hospital, and we lacked complete information on the diagnosis of recurrence based on symptoms or because of follow-up. Another limitation is the lack of a comparison group. However, its strengths lie in the sizable patient cohort, providing precise results on potential risk factors. In addition, the long follow-up period yielded interesting information about late complications or events. Finally, the fact that the results were obtained in routine clinical practice in patients undergoing CRC surgery adds great value to the conclusions drawn from this study, which can be easily extrapolated to daily care in other hospitals.
In summary, we found that in non-metastatic CRC, adherence to follow-up recommendations after surgery is independently associated with lower CRC mortality as well as lower all-cause mortality. CT scan was associated with the highest yield for metastasis and recurrence detection, and if follow-up were performed with blood test TMs only, an important proportion of recurrences would be missed. However, adherence to this follow-up schedule was low and decreased over time. Therefore, patients who benefit the most from intensive oncological follow-up should be adequately defined to avoid over-surveillance. Our results offer further real-life evidence for this specific population.
| 1. | Asociación Española Contra El Cáncer. Incidencia y Mortalidad de Cáncer Colorrectal en España en la población entre 50 y 69 años. 2022. [cited 13 February 2025]. Available from: https://observatorio.contraelcancer.es/sites/default/files/informes/Informe%20cribado.pdf. |
| 2. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15061] [Article Influence: 7530.5] [Reference Citation Analysis (23)] |
| 3. | Gómez-España MA, Gallego J, González-Flores E, Maurel J, Páez D, Sastre J, Aparicio J, Benavides M, Feliu J, Vera R. SEOM clinical guidelines for diagnosis and treatment of metastatic colorectal cancer (2018). Clin Transl Oncol. 2019;21:46-54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 4. | Cubiella J, Marzo-Castillejo M, Mascort-Roca JJ, Amador-Romero FJ, Bellas-Beceiro B, Clofent-Vilaplana J, Carballal S, Ferrándiz-Santos J, Gimeno-García AZ, Jover R, Mangas-Sanjuán C, Moreira L, Pellisè M, Quintero E, Rodríguez-Camacho E, Vega-Villaamil P; Sociedad Española de Medicina de Familia y Comunitaria y Asociación Española de Gastroenterología. Clinical practice guideline. Diagnosis and prevention of colorectal cancer. 2018 Update. Gastroenterol Hepatol. 2018;41:585-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 5. | Hassan C, Wysocki PT, Fuccio L, Seufferlein T, Dinis-Ribeiro M, Brandão C, Regula J, Frazzoni L, Pellise M, Alfieri S, Dekker E, Jover R, Rosati G, Senore C, Spada C, Gralnek I, Dumonceau JM, van Hooft JE, van Cutsem E, Ponchon T. Endoscopic surveillance after surgical or endoscopic resection for colorectal cancer: European Society of Gastrointestinal Endoscopy (ESGE) and European Society of Digestive Oncology (ESDO) Guideline. Endoscopy. 2019;51:266-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 6. | Fuccio L, Rex D, Ponchon T, Frazzoni L, Dinis-Ribeiro M, Bhandari P, Dekker E, Pellisè M, Correale L, van Hooft J, Jover R, Libanio D, Radaelli F, Alfieri S, Bazzoli F, Senore C, Regula J, Seufferlein T, Rösch T, Sharma P, Repici A, Hassan C. New and Recurrent Colorectal Cancers After Resection: a Systematic Review and Meta-analysis of Endoscopic Surveillance Studies. Gastroenterology. 2019;156:1309-1323.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 7. | van Lanschot MCJ, van Leerdam ME, Lansdorp-Vogelaar I, Doets S, Nagtegaal ID, Schreurs HW, van der Hulst RWM, Carvalho B, Dekker E, van Berkel AM. Yield of Surveillance Colonoscopies 1 Year After Curative Surgical Colorectal Cancer Resections. Clin Gastroenterol Hepatol. 2019;17:2285-2293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 8. | Argilés G, Tabernero J, Labianca R, Hochhauser D, Salazar R, Iveson T, Laurent-Puig P, Quirke P, Yoshino T, Taieb J, Martinelli E, Arnold D; ESMO Guidelines Committee. Localised colon cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2020;31:1291-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1145] [Cited by in RCA: 972] [Article Influence: 162.0] [Reference Citation Analysis (4)] |
| 9. | Meyerhardt JA, Mangu PB, Flynn PJ, Korde L, Loprinzi CL, Minsky BD, Petrelli NJ, Ryan K, Schrag DH, Wong SL, Benson AB 3rd; American Society of Clinical Oncology. Follow-up care, surveillance protocol, and secondary prevention measures for survivors of colorectal cancer: American Society of Clinical Oncology clinical practice guideline endorsement. J Clin Oncol. 2013;31:4465-4470. [PubMed] [DOI] [Full Text] |
| 10. | Pita-Fernández S, Alhayek-Aí M, González-Martín C, López-Calviño B, Seoane-Pillado T, Pértega-Díaz S. Intensive follow-up strategies improve outcomes in nonmetastatic colorectal cancer patients after curative surgery: a systematic review and meta-analysis. Ann Oncol. 2015;26:644-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 192] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 11. | Jeffery M, Hickey BE, Hider PN. Follow-up strategies for patients treated for non-metastatic colorectal cancer. Cochrane Database Syst Rev. 2019;9:CD002200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 12. | Snyder RA, Hu CY, Cuddy A, Francescatti AB, Schumacher JR, Van Loon K, You YN, Kozower BD, Greenberg CC, Schrag D, Venook A, McKellar D, Winchester DP, Chang GJ; Alliance for Clinical Trials in Oncology Network Cancer Surveillance Optimization Working Group. Association Between Intensity of Posttreatment Surveillance Testing and Detection of Recurrence in Patients With Colorectal Cancer. JAMA. 2018;319:2104-2115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 98] [Article Influence: 12.3] [Reference Citation Analysis (4)] |
| 13. | Zhao Y, Yi C, Zhang Y, Fang F, Faramand A. Intensive follow-up strategies after radical surgery for nonmetastatic colorectal cancer: A systematic review and meta-analysis of randomized controlled trials. PLoS One. 2019;14:e0220533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Primrose JN, Perera R, Gray A, Rose P, Fuller A, Corkhill A, George S, Mant D; FACS Trial Investigators. Effect of 3 to 5 years of scheduled CEA and CT follow-up to detect recurrence of colorectal cancer: the FACS randomized clinical trial. JAMA. 2014;311:263-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 381] [Cited by in RCA: 355] [Article Influence: 29.6] [Reference Citation Analysis (3)] |
| 15. | Rosati G, Ambrosini G, Barni S, Andreoni B, Corradini G, Luchena G, Daniele B, Gaion F, Oliverio G, Duro M, Martignoni G, Pinna N, Sozzi P, Pancera G, Solina G, Pavia G, Pignata S, Johnson F, Labianca R, Apolone G, Zaniboni A, Monteforte M, Negri E, Torri V, Mosconi P, Fossati R; GILDA working group. A randomized trial of intensive versus minimal surveillance of patients with resected Dukes B2-C colorectal carcinoma. Ann Oncol. 2016;27:274-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 100] [Article Influence: 10.0] [Reference Citation Analysis (2)] |
| 16. | Wille-Jørgensen P, Syk I, Smedh K, Laurberg S, Nielsen DT, Petersen SH, Renehan AG, Horváth-Puhó E, Påhlman L, Sørensen HT; COLOFOL Study Group. Effect of More vs Less Frequent Follow-up Testing on Overall and Colorectal Cancer-Specific Mortality in Patients With Stage II or III Colorectal Cancer: The COLOFOL Randomized Clinical Trial. JAMA. 2018;319:2095-2103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 194] [Cited by in RCA: 172] [Article Influence: 21.5] [Reference Citation Analysis (4)] |
| 17. | Mokhles S, Macbeth F, Farewell V, Fiorentino F, Williams NR, Younes RN, Takkenberg JJ, Treasure T. Meta-analysis of colorectal cancer follow-up after potentially curative resection. Br J Surg. 2016;103:1259-1268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 87] [Article Influence: 8.7] [Reference Citation Analysis (4)] |
| 18. | Weiser MR. AJCC 8th Edition: Colorectal Cancer. Ann Surg Oncol. 2018;25:1454-1455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 834] [Cited by in RCA: 745] [Article Influence: 93.1] [Reference Citation Analysis (5)] |
| 19. | Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18949] [Reference Citation Analysis (0)] |
| 20. | Li J, Gu A, Nong XM, Zhai S, Yue ZY, Li MY, Liu Y. Six-Membered Aromatic Nitrogen Heterocyclic Anti-Tumor Agents: Synthesis and Applications. Chem Rec. 2023;23:e202300293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 21. | Pericay C, Montagut C, Reina JJ, Melian M, Alcaide J, Tarazona N, Ruiz-Casado A, González-Flores E, Graña B, Grávalos C. SEOM-GEMCAD-TTD clinical guidelines for the adjuvant treatment of colon cancer (2023). Clin Transl Oncol. 2024;26:2812-2825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, Arnold D; ESMO Guidelines Committee. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv22-iv40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1481] [Cited by in RCA: 1337] [Article Influence: 148.6] [Reference Citation Analysis (6)] |
| 23. | Lipská L, Visokai V, Levý M, Svobodová S, Kormunda S, Fínek J. Tumor markers in patients with relapse of colorectal carcinoma. Anticancer Res. 2007;27:1901-1905. [PubMed] |
| 24. | Jones RP, McWhirter D, Fretwell VL, McAvoy A, Hardman JG. Clinical follow-up does not improve survival after resection of stage I-III colorectal cancer: A cohort study. Int J Surg. 2015;17:67-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 25. | Zhang Y, Wang Y, Zhang B, Li P, Zhao Y. Methods and biomarkers for early detection, prediction, and diagnosis of colorectal cancer. Biomed Pharmacother. 2023;163:114786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 101] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 26. | Henriksen TV, Tarazona N, Frydendahl A, Reinert T, Gimeno-Valiente F, Carbonell-Asins JA, Sharma S, Renner D, Hafez D, Roda D, Huerta M, Roselló S, Madsen AH, Løve US, Andersen PV, Thorlacius-Ussing O, Iversen LH, Gotschalck KA, Sethi H, Aleshin A, Cervantes A, Andersen CL. Circulating Tumor DNA in Stage III Colorectal Cancer, beyond Minimal Residual Disease Detection, toward Assessment of Adjuvant Therapy Efficacy and Clinical Behavior of Recurrences. Clin Cancer Res. 2022;28:507-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 197] [Article Influence: 49.3] [Reference Citation Analysis (0)] |
| 27. | Carpentier MY, Vernon SW, Bartholomew LK, Murphy CC, Bluethmann SM. Receipt of recommended surveillance among colorectal cancer survivors: a systematic review. J Cancer Surviv. 2013;7:464-483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 28. | Berian JR, Cuddy A, Francescatti AB, O'Dwyer L, Nancy You Y, Volk RJ, Chang GJ. A systematic review of patient perspectives on surveillance after colorectal cancer treatment. J Cancer Surviv. 2017;11:542-552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 29. | Kupfer SS, Lubner S, Coronel E, Pickhardt PJ, Tipping M, Graffy P, Keenan E, Ross E, Li T, Weinberg DS. Adherence to postresection colorectal cancer surveillance at National Cancer Institute-designated Comprehensive Cancer Centers. Cancer Med. 2018;7:5351-5358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (4)] |
| 30. | Stephens K, Fahy BN. Adherence to surveillance colonoscopy guidelines in patients following curative-intent colorectal cancer resection. Cancer Treat Res Commun. 2021;29:100479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 31. | Pineda MM, Fernández ZR, Tuero JHG, Ramírez RR. Relación diagnóstico y supervivencia en el cáncer de colon. Medisan. 2011;15:1566-1576. |
| 32. | Reza MM, Blasco JA, Andradas E, Cantero R, Mayol J. Systematic review of laparoscopic versus open surgery for colorectal cancer. Br J Surg. 2006;93:921-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 253] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 33. | Heynsbergh N, Heckel L, Botti M, Livingston PM. Feasibility, useability and acceptability of technology-based interventions for informal cancer carers: a systematic review. BMC Cancer. 2018;18:244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 83] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 34. | Schinköthe T. Individualized eHealth Support for Oncological Therapy Management. Breast Care (Basel). 2019;14:130-134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/