Published online Mar 14, 2025. doi: 10.3748/wjg.v31.i10.102393
Revised: January 4, 2025
Accepted: February 8, 2025
Published online: March 14, 2025
Processing time: 132 Days and 21.4 Hours
There are few clinicopathologic characteristics and clinical results for oesophageal gastrointestinal stromal tumours (GISTs). Thus, the objective of this study was to identify the clinicopathologic characteristics and clinical results of oesophageal GISTs.
To investigate endoscopic treatment effective of oesophageal GISTs.
It was retrospective research that collected 32 patients with oesophageal GISTs treated by endoscopic resection (ER) between January 2012 and January 2023 in two Hospital. Clinicopathologic, endoscopic records, and follow-up data were collected and analysed.
Thirty-one patients underwent en bloc resection and 24 (75.0%) lesions underwent R0 resection. The size of GISTs was 2.12 ± 1.88 cm. The overall complication rate was 25.0%, including hydrothorax and post-endoscopic submu
ER is safe and effective for patients with low-risk oesophageal GISTs. Early detection of oesophageal GISTs is essential to achieve a favourable prognosis.
Core Tip: Oesophageal gastrointestinal stromal tumours are extremely rare. Endoscopic resection proves to be both safe and effective for patients with low-risk. Early detection plays a critical role in determining the prognosis of these tumours.
- Citation: Xu EP, Qi ZP, Zhang JW, Li B, Ren Z, Cai MY, Cai SL, Lv ZT, Chen ZH, Liu JY, Zhong YS, Zhou PH, Shi Q. Endoscopic treatment outcome of oesophageal gastrointestinal stromal tumours. World J Gastroenterol 2025; 31(10): 102393
- URL: https://www.wjgnet.com/1007-9327/full/v31/i10/102393.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i10.102393
Gastrointestinal stromal tumours (GISTs) are most commonly found in the stomach, whereas oesophageal GISTs are relatively rare[1]. Given the rarity of oesophageal GISTs, clinicopathologic data pertaining to oesophageal GISTs are restricted and only few case reports are available[2].
Indeed, optimal surgical management recommendations are lacking due to the rarity of oesophageal GISTs[3]. It has been reported that esophagectomy is advised for GISTs larger than 9 cm, while enucleation of oesophageal GISTs is acceptable for smaller tumours (2-5 cm)[2]. The clinical results of these two operations have been identified to be comparable assuming suitable patient selection[3-7]. With the development of endoscopic resection (ER), complete removal can now be achieved via endoscopy[8], but the efficacy still requires further research for confirmation.
The outcomes of GIST patients vary depending on tumour size, tumour location, and mitotic activity[9]. Many GIST risk stratification criteria have been developed based on these factors to assess the prognosis of GISTs[10-13]. However, the efficiency of these models in predicting the prognosis of oesophageal GISTs was unclear, as few oesophageal GISTs were included in their development[3]. Feng et al[9] reported that oesophageal GISTs differ greatly from gastric GIST in terms of clinicopathologic features.
The number of published reports involving ER of oesophageal GISTs have increased in the past decade; however, these studies were generally based on small sample sizes with limited long-term follow-up data[14,15]. The objective of our retrospective study was to investigate the effective of ER for oesophageal GISTs on oncologic outcomes using multidisciplinary treatment.
We collected patients with localized, primary oesophageal GISTs that could be verified histologically. Patients was from Zhongshan Hospital Fudan University and Xuhui District Central Hospital from January 2012 to January 2023. The Committee of Medical Ethics (Zhongshan Hospital Fudan University) approved this study. The criteria for inclusion were as follows: (1) > 18 years of age; (2) Pathologic diagnosis of oesophageal (E-GIST); and (3) No recurrent or metastasis before treatment. The criteria for exclusion were as follows: (1) Patients who were lost to follow-up; and (2) Existence of other malignant tumours.
Finally, thirty-two patients who had regular follow-up were enrolled.
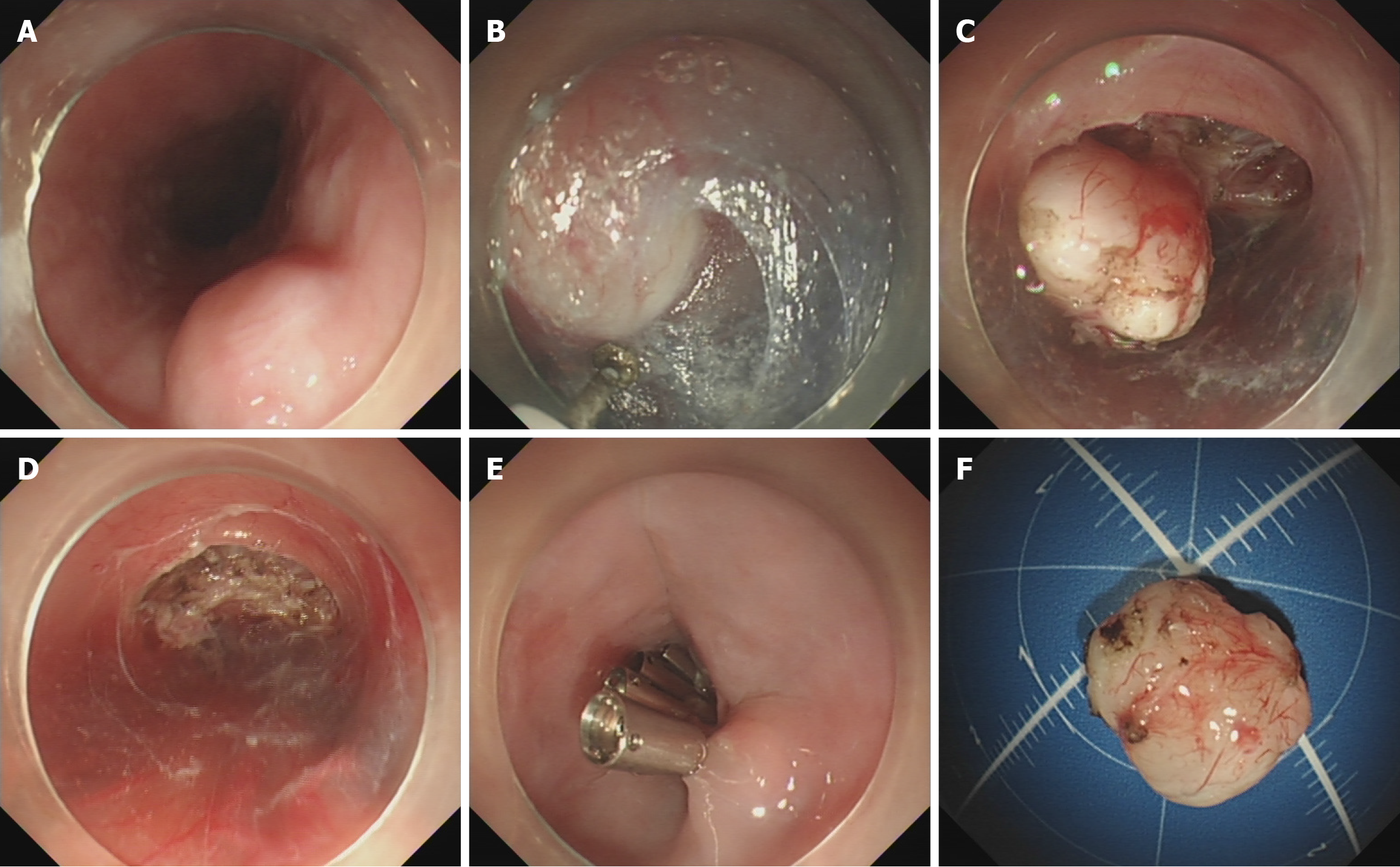
Before the surgery, all patients underwent a 24-hour fast. The patient was under general anaesthesia to the ER. The procedure was conducted using a standard, single-channel gastroscope (GIF-Q260J; Olympus Medical Systems Corp., Tokyo, Japan). The tip of the endoscope was capped with a transparent cap (D-201-11802; Olympus). Firstly, the mixed normal saline was injected at 3-5 cm away from oral orientation of GIST to create a fluid cushion. Then, hook or hybrid knife was used to create a 2 cm longitudinal mucosal incision at the top of the fluid cushion, entering into the submucosa. A submucosal tunnel was carefully created, and the tumour was resected within the tunnel as previously described (Figure 1)[16].
Every oesophageal GIST specimen was routinely subjected to a histologic investigation in our hospital's pathology department. Using the modified National Institutes of Health (NIH) criteria to evaluate the malignancy risk of GIST by calculating maximum tumour size and the number of mitoses per 50 high-power fields (HPF)[12]. To support the diagnosis of a GIST, immunohistochemistry was carried by using a set of antibodies, such as against CD34, CD117 (c-Kit), and so on.
A multidisciplinary team, including endoscopist, pathologist, and surgeon, was assembled to develop a treatment strategy (imatinib adjuvant therapy, scheduled follow-up evaluation, or surgery) for patients with positive surgical margins. Scheduled follow-up was arranged for patients who were not receiving imatinib adjuvant therapy.
Main outcomes were identified as en bloc resection, R0 resection, recurrence, and residual. En bloc resection was termed as the tumour resected in one piece. R0, R1, and R2 resection were termed as negative lateral and vertical margins, microscopic residual tumour, and macroscopic residual tumour basing on en bloc resection, respectively[17]. ER0 was termed as no residual tumour under endoscopic observation, and ER1 was termed as having residual tumour under endoscopic observation[18].
Clinicopathologic data and endoscopic outcomes were collected from electronic medical records in our hospital. Tumour size was reported by pathologist measuring the largest diameter of the tumour specimen. The GISTs were classified into high-risk, intermediate, low, and very low groups according to the modified NIH criteria[12]. Due to nature of retrospec
Patients had scheduled follow-up at 1, 3, 6, and 12 months after resection with endoscopy and/or endoscopic ultra
The statistical analysis was performed by SPSS 21.0 program (IBM Corp., Armonk, NY, United States). We used Fisher's exact test or χ2 test to compare classified variables. We used analysis of variance or Student’s t-test to calculate continuous variables. Kaplan-Meier survival curve was drawn by GraphPad Prism.
Table 1 provided a detailed description of the clinical features. Thirty-two patients with oesophageal GISTs were iden
| Parameter | Variable |
| Age, mean ± SD (year) | 57.78 ± 7.39 |
| < 50 | 4 (12.5) |
| ≥ 50 | 28 (87.5) |
| Sex | |
| Male | 18 (56.3) |
| Female | 14 (43.8) |
| Localization | |
| Upper | 2 (6.3) |
| Middle | 10 (31.3) |
| Lower | 20 (62.5) |
| Tumour size (cm) | 2.12 ± 1.88 (median: 1.50, range: 0.80-2.75) |
| Mitotic rate | |
| Mean (per 50 HPF, ± SD) | 3.34 ± 5.04 |
| Median (per 50 HPF, range) | 1.50 (1.00-4.00) |
| NIH risk category | |
| High | 6 (18.8) |
| Intermediate | 2 (6.3) |
| Low | 6 (18.8) |
| Very Low | 18 (56.3) |
| Symptoms | |
| Asymptomatic (discovered incidentally) | 28 (87.5) |
| Abdominal pain | 3 (9.4) |
| Obstruction | 1 (3.1) |
| Operation time, mean (± SD), minute | 52.63 ± 43.29 |
| Hospital stay duration, mean (± SD), day | 4.22 ± 2.21 |
| Surgical resection | |
| En bloc resection | 31 (96.9) |
| ER0 resection | 32 (100) |
| R0 resection | 24 (75.0) |
| R1 resection | 8 (25.0) |
| Tumour growth pattern | |
| Intraluminal growth | 29 (90.6) |
| Extraluminal growth | 3 (9.4) |
| Complication | 8 (25.0) |
| Hydrothorax | 4 (12.5) |
| PEECS | 5 (15.6) |
In total, thirty-one (96.9%) patients underwent an en bloc resection, of whom 32 (100%) and 24 (75%) underwent ER0 and R0 resections, respectively. The mean operative time was 52.63 ± 43.29 minutes; The mean hospital stay duration was 4.22 ± 2.21 days. Overall, eight (25.0%) patients had complications, including 4 (12.5%) patients with hydrothoraces and 5 (15.6%) patients with post-endoscopic submucosal dissection electrocoagulation syndrome (PEECS). All patients with complications recovered fully following conservative treatment.
The mean tumour size was 2.12 ± 1.88 (median: 1.50, range: 0.80-2.75) cm. The mean mitotic index was 3.34 ± 5.04 (median, 1.50; range, 1.00-4.00). According to the modified NIH criteria, 6 (18.8%), 2 (6.3%), 6 (18.8%), and 18 (56.3%) patients were identified as high, intermediate, low, and very low risk, respectively. Two patients received imatinib the
Oesophageal GIST survival data were analysed and were presented in Table 2. Three patients had recurrences. The mean follow-up was 64.69 ± 33.13 months (median, 65.50; range, 37.75-93.00). Table 3 summarized the detailed features of patients who experienced recurrences. The 5-year overall survival (OS) and disease-free survival (DFS) rate were 100% (32/32) and 90.6% (29/32), respectively. The survival curves were showed in Figure 2.
| Parameter | Variable |
| Treatment | |
| Imatinib | 2 (6.3) |
| Wait and see | 30 (93.7) |
| Follow-up time | |
| Mean (mean ± SD) | 64.69 ± 33.13 |
| Median (month, range) | 65.50 (37.75-93.00) |
| Survival data | |
| Recurrence | 3 |
| GISTs-related deaths | 0 |
| Survival rates (%) | |
| 1-/3-/5-year OS | 100/100/100 |
| 1-/3-/5-year DFS | 100/90.6/90.6 |
| Patient | Location | Size, cm | Mitotic rates | Growth pattern | Margin status | Classification of tumour | Connection to MP layer | Follow-up time, months | Imatinib time, months | Reoperation | Survive |
| 1 | Middle | 7 | 1 | Extraluminal | R1 | High risk | Yes | 33 | No | No | Yes |
| 2 | Lower | 2 | 0 | Intraluminal | R0 | Very low risk | Yes | 107 | No | No | Yes |
| 3 | Lower | 1 | 7 | Intraluminal | R0 | Low risk | Yes | 40 | No | No | Yes |
Oesophageal GIST was a very rare subtype of GIST with little demographic and clinicopathologic characteristic[19], although there are some studies that have focused on oesophageal GISTs based on European and American populations[20,21]. Therefore, we evaluated demographic and clinical outcome of 32 oesophageal GIST patients.
In our study, the median duration of follow-up was 64.69 ± 33.13 months. Overall, three patients had recurrences. The 5-year OS and DFS was 100% and 90.6%, respectively. The mean size of oesophageal GISTs was 2.12 ± 1.88 cm. The lower oesophagus was the most frequently location, followed by the middle oesophagus, then upper oesophagus. It was con
Many researches demonstrated that oesophageal GIST had a higher propensity for malignancy, with an increased risk of metastases and/or tumour recurrence, as well as a high mortality rate[6,9,19]. In contrast, our study showed that pa
We are of the opinion that oesophageal GISTs were shown to be malignant in other studies because oesophageal GISTs were diagnosed late rather than inherent malignancy of the tumour. The oesophagus GIST and g-GIST shared a common basic pathophysiology, which oesophageal GISTs also develop from interstitial pacemaker cells of Cajal. It has been reported that the distribution of mitotic indices between oesophageal and g-GISTs are similar[9,24]. According to previous studies, 75% of patients with oesophageal GISTs present with bleeding (10%), weight loss (20%), and dysphagia (51%)[19]. Prior to the onset of symptoms, oesophageal GISTs grow steadily over time. One case was reported in which the oesophageal GIST grow from 1.8 cm to 10 cm within 2 years, and the oesophageal GIST volume doubling time was assessed to be 3.1 months[25]. When patients present with symptoms, oesophageal GISTs are often large with increased malignant potential. In our study, 87.5% GISTs were detected incidentally and the mean size was 2.12 ± 1.88 cm, which is significantly smaller than reported in the literature[21]. This finding also accounts, at least in part, for the good prognosis in our oesophageal GIST patients. This finding clearly reflects the importance of early diagnosis and treatment.
It was accepted that oesophageal GISTs, like other GISTs, should be managed by surgery[23]; however, the optimal surgical method has not been established[14]. In our research, the total complication rate was 25%, and all patients recovered with conservative treatment. The complication was hydrothorax and PEECS, which are non-fatal complications (Clavien-Dindo < 3b). As for esophagectomy, it was reported Clavien-Dindo ≥ 3b was 10.3%[21]. They also need to take reconstruction. Another biggest advantage of ER is that it preserves the structural integrity of the esophagus, allowing patients to have a higher quality of life. What’s more, the recurrence rate after ER is 9.4% which was lower than the surgery (16.5%)[26]. The generally accepted principle for surgical resection of GIST was that it should be completely resected with negative margins[27]. However, recent studies have suggested that R1 resection may also be considered feasible. A multicenter retrospective study[21] on oesophageal GISTs found that 83.3% of patients who underwent enucleation had R1 resection, and the study indicated that R1 resection was not a prognostic risk factor. Even the recurrence rate of enucleation group was lower than surgical group. Some studies also have found that R1 resection with gastric GIST may not have significant effects on recurrence[28,29]. Our R0 resection rate 75.0%, respectively. The recurrence rate of R1 resection was 12.5% which was lower than the other published oesophageal GIST (18.2%-25.5%)[9,19,21]. We hypothesized that endoscopic R1 resection maybe not the main factor for oesophageal GIST recurrence. We are of the opinion that the margin of endoscopic R1 resection was different from surgical R1 resection. Tumour microstruc
Enucleation was the preferred approach for treating smaller E-GISTs (2-5 cm)[31,32]. Compared to surgical operation, ER focuses on minimizing invasiveness and promoting faster recovery[33]. The selection of the appropriate endoscopic technique was an important topic worthy of exploration (some ER for E-GIST was listed in Supplementary Table 1). We recommended submucosal tunneling ER (STER) as the preferred ER strategy. STER facilitated complete removal of the lesion in the esophagus while preserving the integrity of the esophageal mucosa, effectively reducing postoperative strictures and gas-related complications[34]. Due to the limitations of the tunnel, STER is suitable for the resection of GISTs smaller than 4 cm[32]. Endoscopic full-thickness resection (EFTR) was employed to resect lesions originating from the deep MP, especially large lesions that cannot be excised by STER due to limited tunneling space.
There were also some limitations. Firstly, it was a two-institution design, which restrict the generalizability of findings to a broader population. The sample size and patient demographics may not fully represent the diversity of patients seen in population-based studies. The results may be influenced by institutional factors such as specific surgical techniques, expertise, and patient selection, which may not be applicable to other settings. To address these limitations, future researches should adopt a multi-center design to increase the diversity of patient populations and enhance the generalizability of the findings. Future research could also incorporate a prospective design to minimize biases and enhance the accuracy of data collection. Secondly, the number of oesophageal GIST sample was insufficient, resulting in sampling errors. Thirdly, there was a lack of data on surgical treatment of oesophageal GIST, and it was unable to establish the appropriate criteria for surgical or ER.
ER is safe and effective in patients with low-risk oesophageal GISTs. Early detection of oesophageal GISTs is crucial for prognosis.
| 1. | Wu GX, Ituarte PH, Paz IB, Kim J, Raz DJ, Kim JY. A Population-Based Examination of the Surgical Outcomes for Patients with Esophageal Sarcoma. Ann Surg Oncol. 2015;22 Suppl 3:S1310-S1317. [PubMed] [DOI] [Full Text] |
| 2. | Hihara J, Mukaida H, Hirabayashi N. Gastrointestinal stromal tumor of the esophagus: current issues of diagnosis, surgery and drug therapy. Transl Gastroenterol Hepatol. 2018;3:6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (33)] |
| 3. | Neofytou K, Costa Neves M, Giakoustidis A, Benson C, Mudan S. Effective Downsizing of a Large Oesophageal Gastrointestinal Stromal Tumour with Neoadjuvant Imatinib Enabling an Uncomplicated and without Tumour Rupture Laparoscopic-Assisted Ivor-Lewis Oesophagectomy. Case Rep Oncol Med. 2015;2015:165736. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (33)] |
| 4. | Lee HJ, Park SI, Kim DK, Kim YH. Surgical resection of esophageal gastrointestinal stromal tumors. Ann Thorac Surg. 2009;87:1569-1571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 41] [Article Influence: 2.4] [Reference Citation Analysis (33)] |
| 5. | Blum MG, Bilimoria KY, Wayne JD, de Hoyos AL, Talamonti MS, Adley B. Surgical considerations for the management and resection of esophageal gastrointestinal stromal tumors. Ann Thorac Surg. 2007;84:1717-1723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 70] [Article Influence: 3.7] [Reference Citation Analysis (36)] |
| 6. | Jiang P, Jiao Z, Han B, Zhang X, Sun X, Su J, Wang C, Gao B. Clinical characteristics and surgical treatment of oesophageal gastrointestinal stromal tumours. Eur J Cardiothorac Surg. 2010;38:223-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 1.8] [Reference Citation Analysis (36)] |
| 7. | Robb WB, Bruyere E, Amielh D, Vinatier E, Mabrut JY, Perniceni T, Piessen G, Mariette C; FREGAT Working Group--FRENCH. Esophageal gastrointestinal stromal tumor: is tumoral enucleation a viable therapeutic option? Ann Surg. 2015;261:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (36)] |
| 8. | Reinehr R. [Endoscopic submucosal excavation (ESE) is a safe and useful technique for endoscopic removal of submucosal tumors of the stomach and the esophagus in selected cases]. Z Gastroenterol. 2015;53:573-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 9. | Feng F, Tian Y, Liu Z, Xu G, Liu S, Guo M, Lian X, Fan D, Zhang H. Clinicopathologic Features and Clinical Outcomes of Esophageal Gastrointestinal Stromal Tumor: Evaluation of a Pooled Case Series. Medicine (Baltimore). 2016;95:e2446. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (44)] |
| 10. | Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol. 2006;23:70-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1595] [Cited by in RCA: 1324] [Article Influence: 66.2] [Reference Citation Analysis (1)] |
| 11. | Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130:1466-1478. [PubMed] [DOI] [Full Text] |
| 12. | Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 985] [Cited by in RCA: 900] [Article Influence: 50.0] [Reference Citation Analysis (3)] |
| 13. | Joensuu H, Vehtari A, Riihimäki J, Nishida T, Steigen SE, Brabec P, Plank L, Nilsson B, Cirilli C, Braconi C, Bordoni A, Magnusson MK, Linke Z, Sufliarsky J, Federico M, Jonasson JG, Dei Tos AP, Rutkowski P. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol. 2012;13:265-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 807] [Cited by in RCA: 691] [Article Influence: 49.4] [Reference Citation Analysis (3)] |
| 14. | Kang G, Kang Y, Kim KH, Ha SY, Kim JY, Shim YM, Heinrich MC, Kim KM, Corless CL. Gastrointestinal stromal tumours of the oesophagus: a clinicopathological and molecular analysis of 27 cases. Histopathology. 2017;71:805-812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 15. | Duffaud F, Meeus P, Bertucci F, Delhorme JB, Stoeckle E, Isambert N, Bompas E, Gagniere J, Bouché O, Toulmonde M, Salas S, Blay JY, Bonvalot S; French Sarcoma Group. Patterns of care and clinical outcomes in primary oesophageal gastrointestinal stromal tumours (GIST): A retrospective study of the French Sarcoma Group (FSG). Eur J Surg Oncol. 2017;43:1110-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (4)] |
| 16. | Xu MD, Cai MY, Zhou PH, Qin XY, Zhong YS, Chen WF, Hu JW, Zhang YQ, Ma LL, Qin WZ, Yao LQ. Submucosal tunneling endoscopic resection: a new technique for treating upper GI submucosal tumors originating from the muscularis propria layer (with videos). Gastrointest Endosc. 2012;75:195-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 247] [Article Influence: 17.6] [Reference Citation Analysis (4)] |
| 17. | Hermanek P, Wittekind C. The pathologist and the residual tumor (R) classification. Pathol Res Pract. 1994;190:115-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 173] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 18. | Zhu Y, Xu MD, Xu C, Zhang XC, Chen SY, Zhong YS, Zhang YQ, Chen WF, Chen TY, Xu JX, Yao LQ, Li QL, Zhou PH. Microscopic positive tumor margin does not increase the rate of recurrence in endoscopic resected gastric mesenchymal tumors compared to negative tumor margin. Surg Endosc. 2020;34:159-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 19. | Lott S, Schmieder M, Mayer B, Henne-Bruns D, Knippschild U, Agaimy A, Schwab M, Kramer K. Gastrointestinal stromal tumors of the esophagus: evaluation of a pooled case series regarding clinicopathological features and clinical outcome. Am J Cancer Res. 2015;5:333-343. [PubMed] |
| 20. | Pesenti C, Bories E, Caillol F, Ratone JP, Godat S, Monges G, Poizat F, Raoul JL, Ries P, Giovannini M. Characterization of subepithelial lesions of the stomach and esophagus by contrast-enhanced EUS: A retrospective study. Endosc Ultrasound. 2019;8:43-49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 21. | Mohammadi M, IJzerman NS, Hohenberger P, Rutkowski P, Jones RL, Martin-Broto J, Gronchi A, Schöffski P, Vassos N, Farag S, Baia M, Oosten AW, Steeghs N, Desar IME, Reyners AKL, van Sandick JW, Bastiaannet E, Gelderblom H, Schrage Y. Clinicopathological features and treatment outcome of oesophageal gastrointestinal stromal tumour (GIST): A large, retrospective multicenter European study. Eur J Surg Oncol. 2021;47:2173-2181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 22. | Briggler AM, Graham RP, Westin GF, Folpe AL, Jaroszewski DE, Okuno SH, Halfdanarson TR. Clinicopathologic features and outcomes of gastrointestinal stromal tumors arising from the esophagus and gastroesophageal junction. J Gastrointest Oncol. 2018;9:718-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 23. | Pence K, Correa AM, Chan E, Khaitan P, Hofstetter W, Kim MP. Management of esophageal gastrointestinal stromal tumor: review of one hundred seven patients. Dis Esophagus. 2017;30:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (3)] |
| 24. | Yamada H, Shinohara T, Yokoyama K, Takasu K, Fujimori Y, Yamagishi K. Thoracoscopic enucleation of esophageal gastrointestinal stromal tumor using prone positioning in a patient with severe chronic obstructive lung disease. J Laparoendosc Adv Surg Tech A. 2011;21:635-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 25. | Li B, Chen T, Qi ZP, Yao LQ, Xu MD, Shi Q, Cai SL, Sun D, Zhou PH, Zhong YS. Efficacy and safety of endoscopic resection for small submucosal tumors originating from the muscularis propria layer in the gastric fundus. Surg Endosc. 2019;33:2553-2561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 26. | Nakano A, Akutsu Y, Shuto K, Uesato M, Kono T, Hoshino I, Akanuma N, Maruyama T, Isozaki Y, Matsubara H. Giant esophageal gastrointestinal stromal tumor: report of a case. Surg Today. 2015;45:247-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 27. | Theiss L, Contreras CM. Gastrointestinal Stromal Tumors of the Stomach and Esophagus. Surg Clin North Am. 2019;99:543-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 28. | DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF. Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231:51-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1916] [Cited by in RCA: 1652] [Article Influence: 63.5] [Reference Citation Analysis (5)] |
| 29. | McCarter MD, Antonescu CR, Ballman KV, Maki RG, Pisters PW, Demetri GD, Blanke CD, von Mehren M, Brennan MF, McCall L, Ota DM, DeMatteo RP; American College of Surgeons Oncology Group (ACOSOG) Intergroup Adjuvant Gist Study Team. Microscopically positive margins for primary gastrointestinal stromal tumors: analysis of risk factors and tumor recurrence. J Am Coll Surg. 2012;215:53-59; discussion 59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 137] [Article Influence: 9.8] [Reference Citation Analysis (6)] |
| 30. | Lin SC, Huang MJ, Zeng CY, Wang TI, Liu ZL, Shiay RK. Clinical manifestations and prognostic factors in patients with gastrointestinal stromal tumors. World J Gastroenterol. 2003;9:2809-2812. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 40] [Cited by in RCA: 41] [Article Influence: 1.8] [Reference Citation Analysis (7)] |
| 31. | Zhang FB, Shi HC, Shu YS, Shi WP, Lu SC, Zhang XY, Tu SS. Diagnosis and surgical treatment of esophageal gastrointestinal stromal tumors. World J Gastroenterol. 2015;21:5630-5634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (4)] |
| 32. | Lian J, Ji Y, Chen T, Wang G, Wang M, Li S, Cao J, Shen L, Lu W, Xu M. Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study. Therap Adv Gastroenterol. 2024;17:17562848241255304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 33. | Chai N, Du C, Gao Y, Niu X, Zhai Y, Linghu E, Liu Y, Yang B, Lu Z, Li Z, Wang X, Tang P. Comparison between submucosal tunneling endoscopic resection and video-assisted thoracoscopic enucleation for esophageal submucosal tumors originating from the muscularis propria layer: a randomized controlled trial. Surg Endosc. 2018;32:3364-3372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 34. | Zhou Y, Zheng S, Sun M, Li Q. Diagnosis and Endoscopic Treatment of Gastrointestinal Stromal Tumors Arising from Esophagus. J Laparoendosc Adv Surg Tech A. 2020;30:759-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (6)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/