Published online Mar 14, 2025. doi: 10.3748/wjg.v31.i10.100194
Revised: December 16, 2024
Accepted: January 21, 2025
Published online: March 14, 2025
Processing time: 200 Days and 19.1 Hours
Liver cirrhosis and portal hypertension (PHT) can lead to lymphatic abnormalities and coagulation dysfunction. Because lymphangiogenesis may relieve liver cir
To investigate the role of lymphangiogenesis in preclinical PHT models.
Immunohistochemistry and transcriptome sequencing of bile duct ligation (BDL) and control lymphatic samples were conducted to reveal the indicated signaling pathways. Functional enrichment analyses were performed on the differentially expressed genes and hub genes. Adenoviral infection of vascular endothelial growth factor C (VEGF-C), platelet-rich plasma (PRP), and VEGF3 receptor (VEGFR) inhibitor MAZ-51 was used as an intervention for the lymphatic system in PHT models. Histology, hemodynamic tests and western blot analyses were performed to demonstrate the effects of lymphatic intervention in PHT patients.
Lymphangiogenesis was increased in the BDL rat model. Transcriptome sequencing analysis of the extrahepatic lymphatic system revealed its close association with platelet adherence, aggregation, and activation. The role of PHT in the rat model was investigated by activating (PRP) and inhibiting (MAZ-51) the lymphatic system. PRP promoted lymphangiogenesis, which increased lymphatic drainage, alleviated portal pressure, reduced liver fibrosis, inhibited inflammation, inhibited angiogenesis, and suppressed mesenteric artery remodeling. MAZ-51 reversed the above improvements.
Via VEGF-C/VEGFR-3, platelets impede fibrosis, angiogenesis, and mesenteric artery remodeling, ultimately alleviating PHT. Thus, platelet intervention is a therapeutic approach for cirrhosis and PHT.
Core Tip: In the sequencing of the classic bile duct ligation model of portal hypertension in rats, platelet activation plays a crucial role in lymphagiogenesis. Effective lymphatic drainage may alleviate the high flow fluid environment of the hepatic portal vein caused by various etiologies. Based on this research, platelet-rich plasma enhanced intrahepatic and extrahepatic proliferation of lymphatics and simultaneously alleviated portal hypertension.
- Citation: Chen M, Zhao JB, Wu GB, Wu ZH, Luo GQ, Zhao ZF, Zhang CH, Lin JY, Li HJ, Qi XL, Huo HZ, Tuersun A, Fan Q, Zheng L, Luo M. Platelet activation relieves liver portal hypertension via the lymphatic system though the classical vascular endothelial growth factor receptor 3 signaling pathway. World J Gastroenterol 2025; 31(10): 100194
- URL: https://www.wjgnet.com/1007-9327/full/v31/i10/100194.htm
- DOI: https://dx.doi.org/10.3748/wjg.v31.i10.100194
The National Bureau of Statistics has indicated that the number of people with chronic liver disease in China has reached nearly 400 million, and various end-stage diseases, such as cirrhosis (including portal hypertension [PHT]) and liver cancer, have greatly increased social costs[1,2]. PHT is a deleterious syndrome characterized by chronically elevated portal pressure (PP), which is the main consequence of liver cirrhosis[3]. Liver cirrhosis leads to PHT due to increased intrahepatic vascular resistance and heightened blood flow through the portal vein[1]. During liver cirrhosis, hepatic inflammatory factors increase, which promotes the secretion of hepatic stellate cells and the deposition of the extracellular matrix. This process culminates in compression of the hepatic sinuses, resulting in heightened resistance to hepatic blood flow and the subsequent onset of PHT. The exacerbation of PHT can lead to a range of complications, including gastrointestinal bleeding, ascites, hepatorenal syndrome and hepatic encephalopathy, which can potentially be life-threatening.
In liver fibrosis, the lymphatic system undergoes various structural and functional alterations that exhibit a compen
Platelets possess a diverse array of bioactive molecules within their granules and exhibit distinct receptors on their surfaces; therefore, these receptors play a significant role in the processes of inflammation, cancer progression, and metastasis[7]. As an important component of the anticoagulant system, platelet production decreases during cirrhosis because of reduced production of thrombopoietin, bone marrow suppression, hypersplenism, and autoantibody-mediated platelet destruction[8,9]. Platelets contain a diverse array of cytokines and have the capacity to modulate the process of lymphatic vessel formation, thereby exerting potential therapeutic effects in the context of liver cirrhosis[10]. Previous studies have suggested that platelets have the potential to stimulate lymphangiogenesis through the release of vascular endothelial growth factor C (VEGF-C) and subsequent activation of the VEGF-C/VEGF receptor 3 (VEGFR-3) pathway[11]. In the present study, the quantity and quality of platelets within the livers of cirrhotic rats were increased via treatment with platelet-rich plasma (PRP) to induce lymphangiogenesis to investigate the therapeutic impact of platelet-induced lymphangiogenesis on liver cirrhosis and PHT.
Six- to eight-week old male Sprague Dawley (SD) rats, weighing 200-250 g, were purchased from Shanghai Laboratory Animal Centre (Shanghai, Center). The rats were kept under controlled conditions (20-22 °C, 60% humidity, and a 12:12 hour light/dark cycle) with free access to food and water. All SD rats were divided into the following groups: Sham-operated (control), common bile duct ligation (BDL-vehicle [VEH]/BDL), adeno-associated virus-VEGF-C (AAV-VEGF-C) combined with BDL (BDL-AAV-VEGF-C), PRP combined with BDL (BDL-PRP), and MAZ-51 + PRP combined with BDL (BDL-MAZ-51) groups. The specific protocol of BDL was previously described[12]. At the beginning of the surgery and drug treatments, the number of rats in each group was 10, and shortly before sacrificing, the number of live rats in each group was 10 (control), 7 (BDL-VEH/BDL), 8 (BDL-AAV-VEGF-C), 8 (BDL-PRP), and 6 (BDL-MAZ-51).
A rat-VEGFC gene-overexpressing AAV vector serotype 8 (AAV8) was constructed by Jiman Company (Shanghai, China). The characteristics of the AAV8-VEGF-C vector were as follows: PGMAAV-4892: GPAAV-CMV-MCS-3 × Flag-T2A-eGFP-WPRE. At 1 day after BDL, the rats in the BDL-AAV-VEGF-C group were given 100 μL of an AAV-VEGF-C solution (1 × 1013 vg/mL) via tail vein injection. At 1 day after BDL, the rats in the BDL-PRP group received an intraperitoneal injection of PRP in phosphate-buffered saline (PBS) twice a week for 4 weeks at a rate of 0.5 mL/kg after activation with calcium chloride. At 1 day after BDL, the rats in the BDL-MAZ-51 group were given MAZ-51 (10 mg/mL, 1:10 dissolved in PBS; GMBIO) once a week for 4 weeks on the basis of exposure to PRP simultaneously. Ethical approval was granted by the Ethical Committee of Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medi
PRP was obtained from the blood of donor rats. Approximately 10 mL blood was collected from each rat. The ratio of blood to sodium citrate used in the sodium citrate anticoagulant solution was 10:1. PRP was obtained through the following two-step centrifugation method: (1) PRP was centrifuged at 1600 rpm for 10 minutes, and the top layer of plasma was extracted after stratification; and (2) PRP was centrifuged at 2000 rpm for 10 minutes, and the lower part of the plasma, known as PRP, was obtained. PRP was mixed with PBS at a 1:1 ratio, resulting in a PRP concentration of 3.8 × 106/μL. PRP was activated by mixing PRP with 10% calcium chloride (10:1) before use.
The right hepatic lobe and mesenteric samples from each rat were fixed in 10% formalin neutral buffered solution (potential of hydrogen = 7.4) and embedded in paraffin blocks. The liver and mesenteric samples were stained with hematoxylin and eosin (H&E). Masson’s trichrome staining and Sirius red staining were employed to evaluate liver fibrosis and collagen deposition regions, respectively. Deparaffinized samples were rehydrated and then microwaved for 20 minutes to extract antigens for immunohistochemistry. After the washing and blocking procedures, the slides were incubated with the following primary antibodies: lymphatic vessel endothelial hyaluronan receptor-1 (LYVE-1) (1:100; Affinity, Inc., Milwaukee, WI, United States), VEGF-C (1:50; Novus Biologicals, Centennial, CO, United States), VEGFR-3 (1:800; Boster Bio, Pleasanton, CA, United States), transforming growth factor beta (TGF-β) (1:2000; Proteintech Group, Wuhan, China), fibronectin (FN) (1:4000; Abcam, Cambridge, United Kingdom), cluster of differentiation 41 (CD41, 1:200; Abcam), CD31 (1:100; Abcam), matrix metalloproteinase 2 (MMP2, 1:250; Abcam), von Willebrand factor (vWF) (1:600; Abcam), VEGF-A (1:200; Abcam), and CD68 (1:200; Novus Biologicals). The slides were subsequently incubated with a secondary antibody (D-3004; Long Island Biotech, Shanghai, China).
A polyethylene 50 tube was inserted into the portal vein of each rat following anesthesia with 2% isoflurane, as previously described[13]. After the polyethylene 50 was connected to a pressure transducer and analyzed automatically via the ALC-MPA multichannel biological information analysis system (Shanghai Alcott Biotech Co., Ltd., Shanghai, China). A P value was obtained.
The present study focused on lymphatic vessels associated with the superior mesenteric artery. Prior to sampling after 4 weeks of BDL, the rats were fasted for 8-12 hours to ensure an empty stomach. The rats subsequently received an intragastric administration of olive oil in accordance with the standard dosage of 5 mL per 100 g of body weight. Following a 1-hour waiting period, the abdomen was surgically opened, revealing the milky white appearance of the lymphatic vessels that accompany the superior mesenteric artery. The lymphatic vessels were isolated from control rats (n = 6) and BDL rats (n = 6). RNA sequencing analysis was conducted by TIANGEN Biotech (Beijing, China). Total RNA was extracted via the TRNzol reagent (TIANGEN). A NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, United States) and the Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, United States) were used to ensure the concentration, purity, and integrity of the total RNA. One microgram of RNA per sample was used for RNA transcriptome sequencing library construction. After trimming the reads, adapters and low-quality regions were removed to ensure that the data were clean for subsequent analyses.
According to the comprehensive mRNA expression profiles of each sample, principal component analysis was conducted to assess the similarity or dissimilarity between the control and BDL groups. EdgeR software (http://www.r-project.org) was used for the identification of DEGs. The read counts for each sequenced library were adjusted via the EdgeR program package, which applies a scaling-normalized factor. The screening standards for DEGs were |log2 fold change|> 1 and P < 0.05. Heatmaps and volcano plots were generated via the pheatmap and ggplot2 R packages, respectively.
The protein-protein interaction network was constructed via the STRING database (http://string-db.org/), which is renowned for its ability to predict protein-protein interactions. KEGG and GO enrichment analyses of the DEGs were conducted via the cluster Profiler R package. P < 0.05 was set as the cutoff value for the GO and KEGG analyses. Gene set enrichment analysis (GSEA) was performed via GSEA software (http://www.broadinstitute.org/gsea).
Proteins were extracted using radio immunoprecipitation assay buffer (Beyotime, Shanghai, China) and phenylmethylsulfonyl fluoride (Beyotime). The protein concentrations were measured with a bicinchoninic acid assay™ protein assay kit (Pierce, Appleton, WI, United States). The proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred to polyvinylidene difluoride membranes. Primary antibodies against alpha smooth muscle actin (SMA) (1:2000; Abcam) and glyceraldehyde-3-phosphate dehydrogenase (1:8000; Sungene Biotech, China) were used as protein loading controls.
All data are presented as the mean ± SD. The Student’s t test was used to evaluate differences between two groups, and P < 0.05 was considered statistically significant. The statistical methods of this study were reviewed by Fan Q, Zheng L, and Luo M from Department of General Surgery, Shanghai Ninth People’s Hospital Affiliated with Shanghai Jiao Tong University School of Medicine (Shanghai, China).
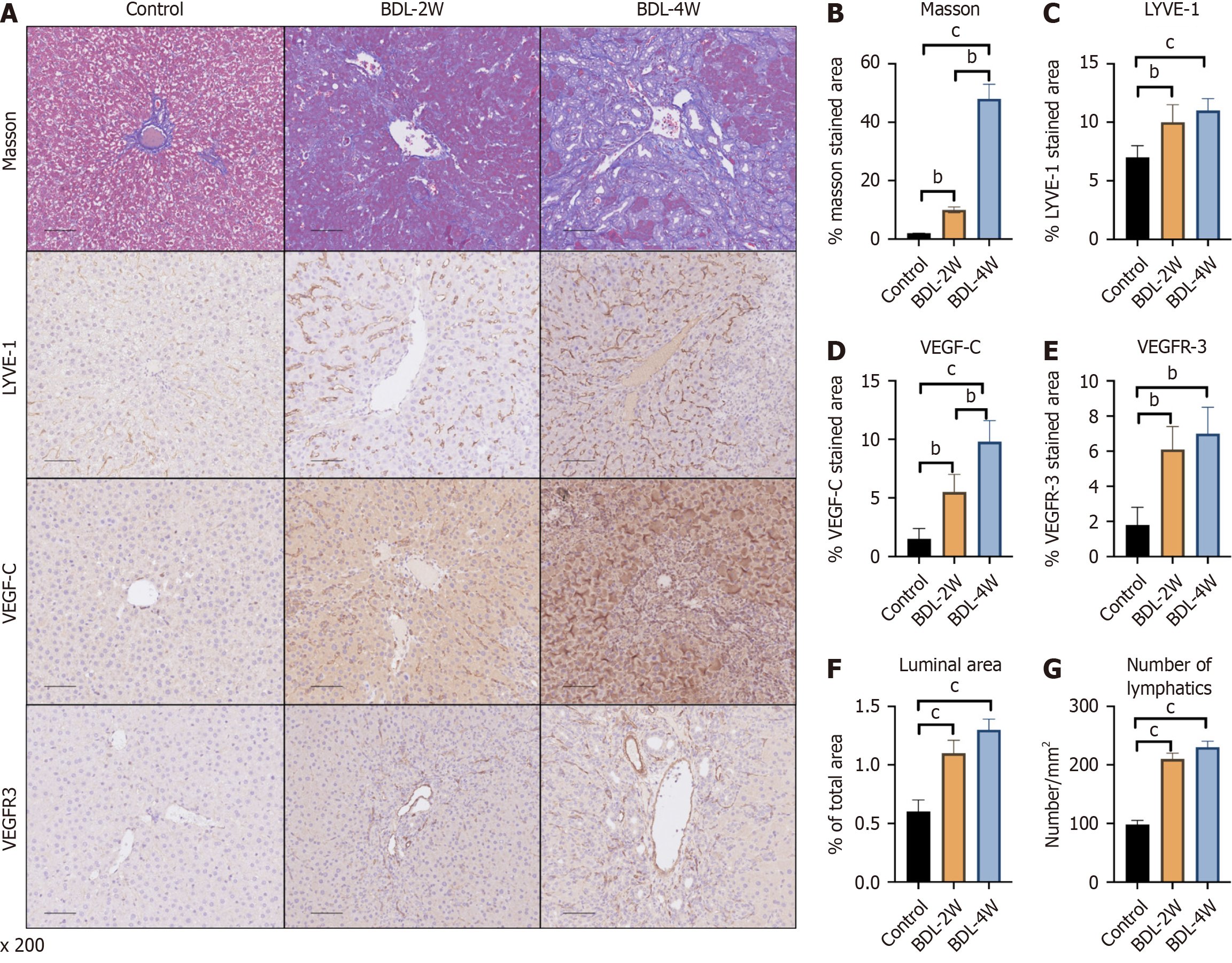
The number and area of lymphatic vessels in the liver were evaluated after BDL, a method for inducing PHT in rats. Masson’s trichrome staining identified liver fibrosis 2 weeks after BDL, and liver cirrhosis was successfully induced after 4 weeks (Figure 1A and B). Compared with that of the sham-operated-VEH group, the area of LYVE-1 was increased at 2 weeks after surgery, and no changes were observed at 4 weeks compared with 2 weeks (Figure 1A and C). However, VEGF-C increased at 2 weeks and was further upregulated at 4 weeks compared with 2 weeks (Figure 1A and D). A similar trend was observed for VEGFR-3 (Figure 1A and E). In addition, the expression of genes related to the lymphatic system, was increased at 2 weeks but did not change at 4 weeks according to the luminal area and number (Figure 1F and G).
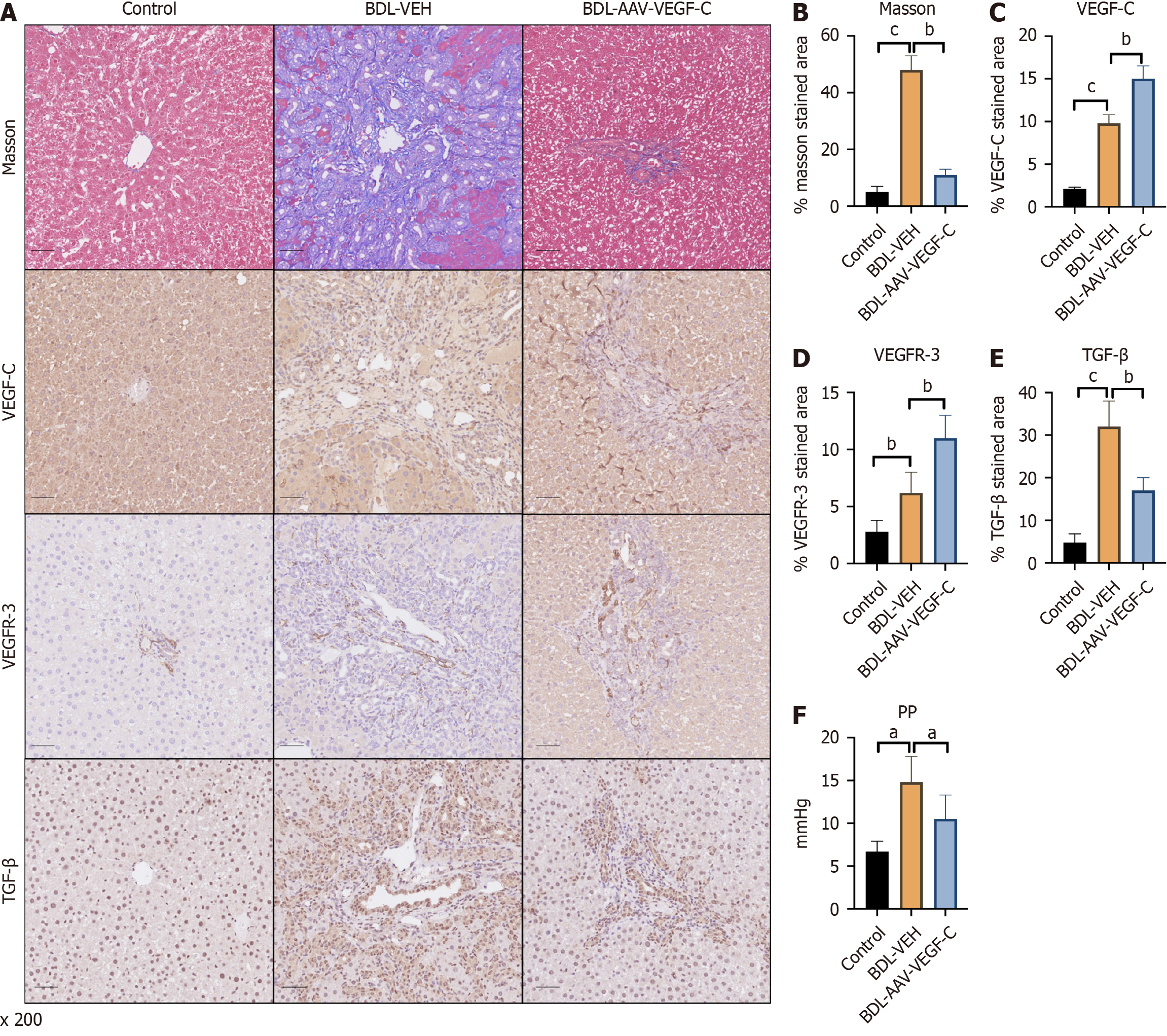
To investigate the function of the lymphatic system in rats with liver fibrosis and PHT, VEGF-C was combined with VEGFR-3 to increase the lymphatic system. Collagen deposition was significantly reduced after VEGF-C intervention (Figure 2A and B). Along with the increase in VEGF-C, the expression of VEGFR-3 also increased compared with that in the BDL-VEH group at the same time (Figure 2A, C and D). These results indicated that the proliferation of intrahepatic lymphatic vessels was strongly enhanced after treatment with AAV-VEGF-C. In addition, the expression of TGF-β, a marker that promotes liver fibrosis, was lower in the AAV-VEGF-C group than in the BDL-VEH group (Figure 2A and E), which indirectly confirmed the high drainage of the lymphatic system due to AAV infection. Moreover, PP was reversed after treatment with AAV-VEGF-C compared with that in the BDL-VEH group (Figure 2F).
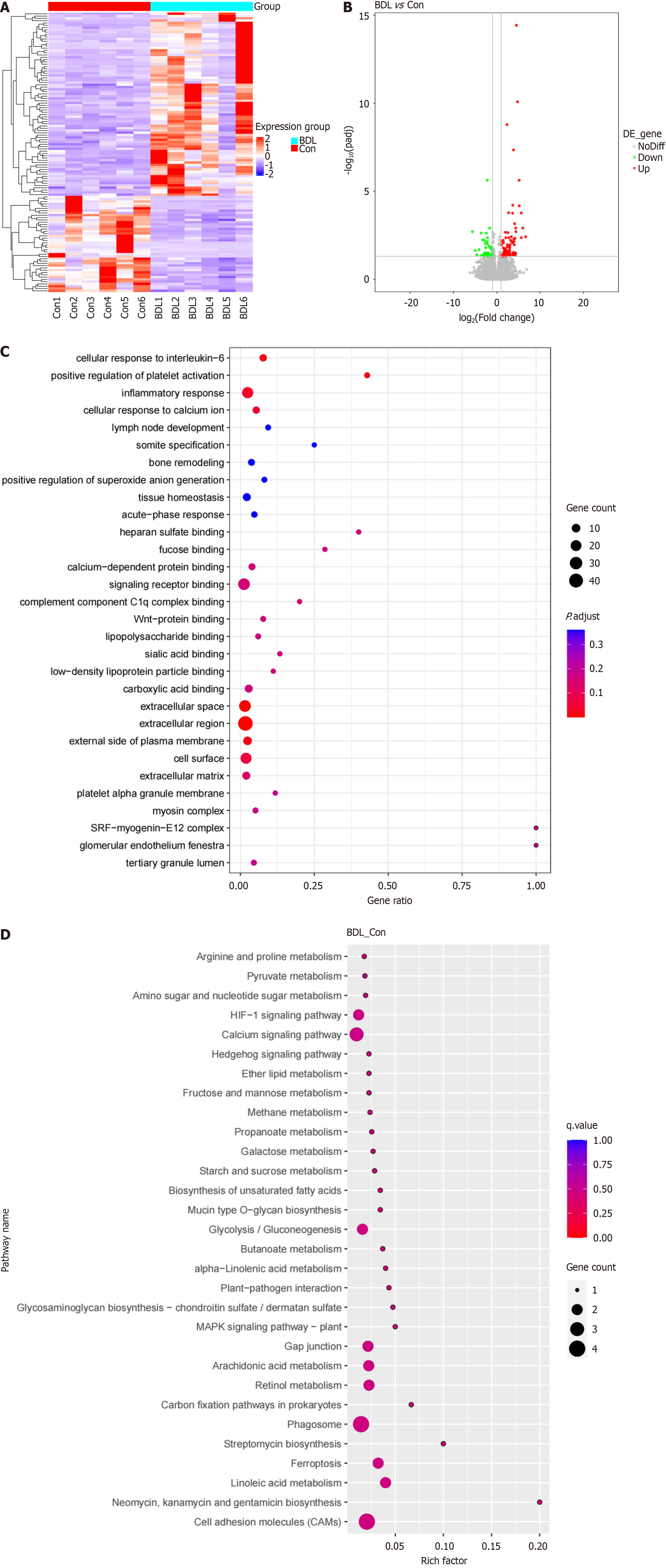
Considering the difficulties in obtaining the lymphatic system in the liver, the lymphatic vessels accompanied by the superior mesenteric artery were collected for whole-transcriptome sequencing. A total of 122 DEGs were screened out after whole-transcriptome sequencing. Between the BDL group and the control group, approximately 42 DEGs were downregulated and 80 DEGs were upregulated (Figure 3A and B). Furthermore, GO and KEGG analyses were per
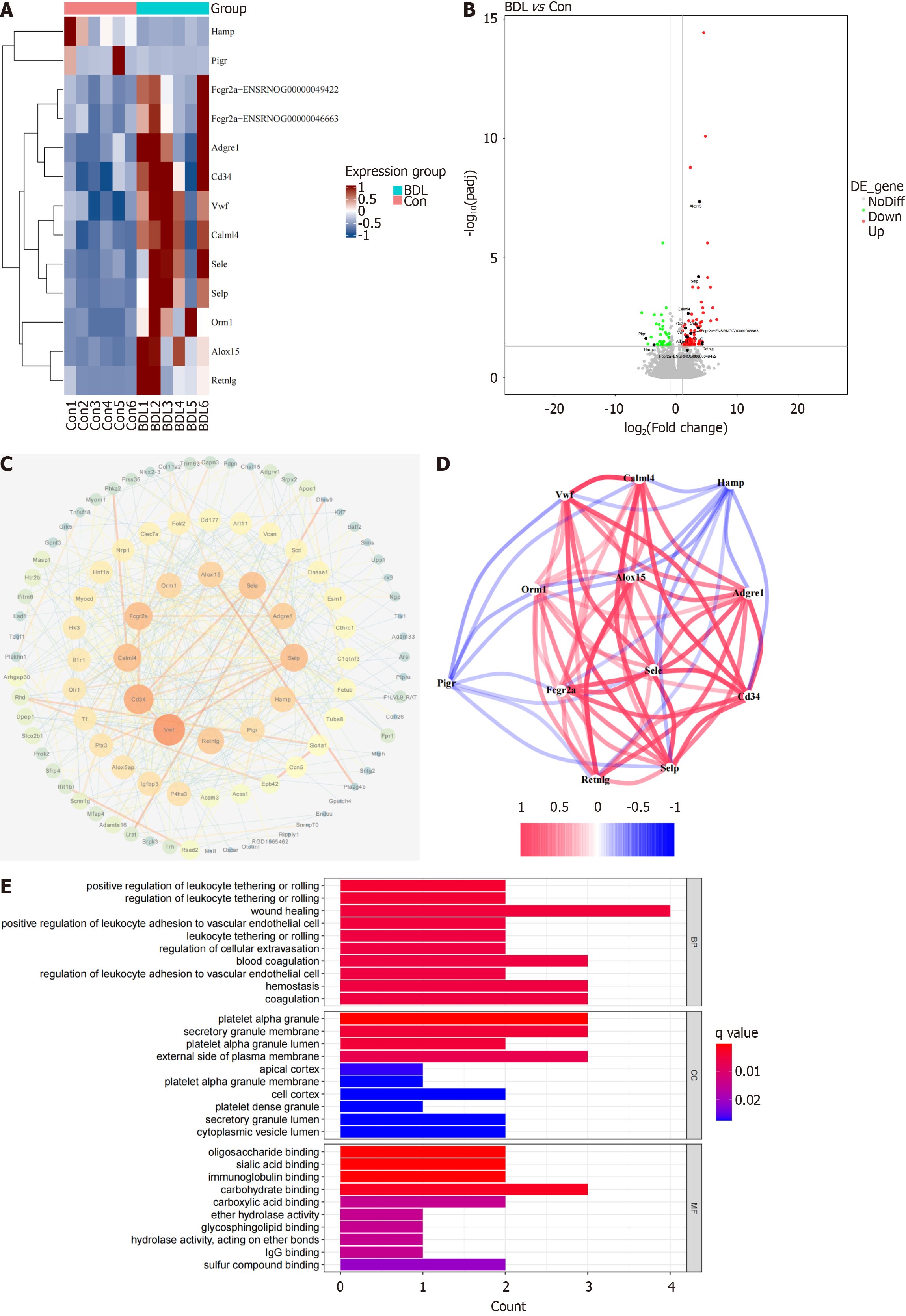
Twelve core DEGs were filtered out according to the degree of gene expression via Cytoscape (Figure 4A). Compared with the control group, the volcano map revealed that HAMP and polymeric immunoglobulin receptor (PIGR) were downregulated in the BDL group, while Fc gamma receptor IIa, adhesion G protein-coupled receptor E1, CD34, vWF, calmodulin like 4, E-selectin, selectin P, orosomucoid 1, arachidonate 15-lipoxygenase, and resistin like gamma were upregulated in the BDL group (Figure 4B). Twelve core genes closely interacted with each other (Figure 4C and D). To further investigate the biological functions of the 12 core genes, GO and KEGG analyses were conducted. GO analysis identified the enrichment of wound healing, platelet alpha granules, platelet alpha granule lumen, platelet alpha granule membrane, and platelet dense granules (Figure 4E). GO analysis also identified the enrichment of cell adhesion mole
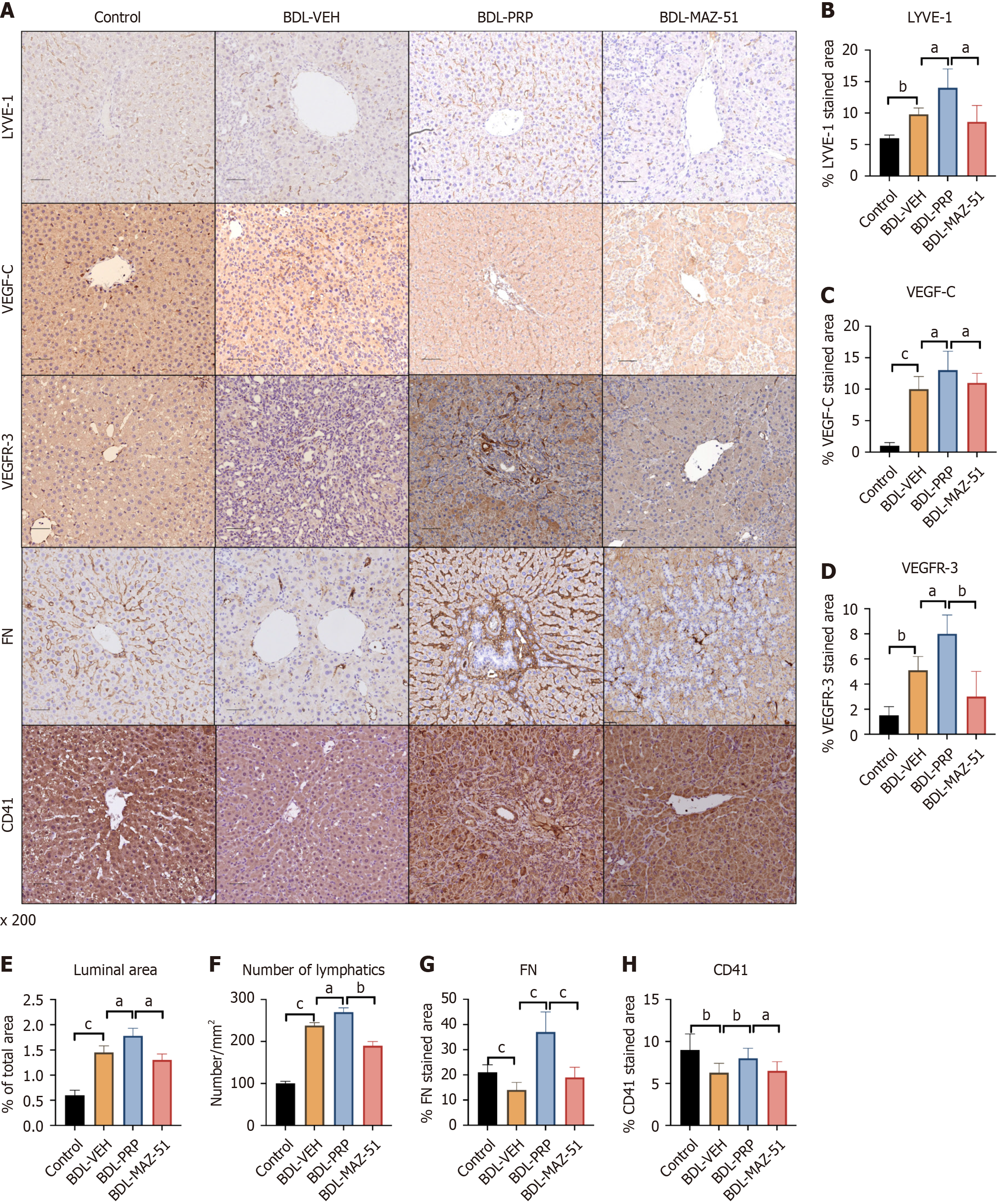
Significantly greater expression of LYVE-1, VEGF-C, and VEGFR-3 was detected in the BDL-PRP group than in the BDL-VEH group, which was reversed after treatment with MAZ-51 (Figure 5A-D). Quantitative analysis of intrahepatic lymphatic vessels revealed a significant increase in both the area and number of lymphatic vessels following treatment with PRP compared with the BDL-VEH group. Conversely, compared with the BDL-PRP group, the MAZ-51 group presented a significant decrease in the number of lymphatic vessels (Figure 5E and F). In addition, the expression of CD41 and FN was increased in the BDL-PRP group compared to the BDL-VEH group. After MAZ-51 intervention, the expression of both platelet markers decreased (Figure 5A, G and H). Due to the enhanced proliferation of lymphatic vessels, the potential phenotypic changes in PHT need to be further assessed.
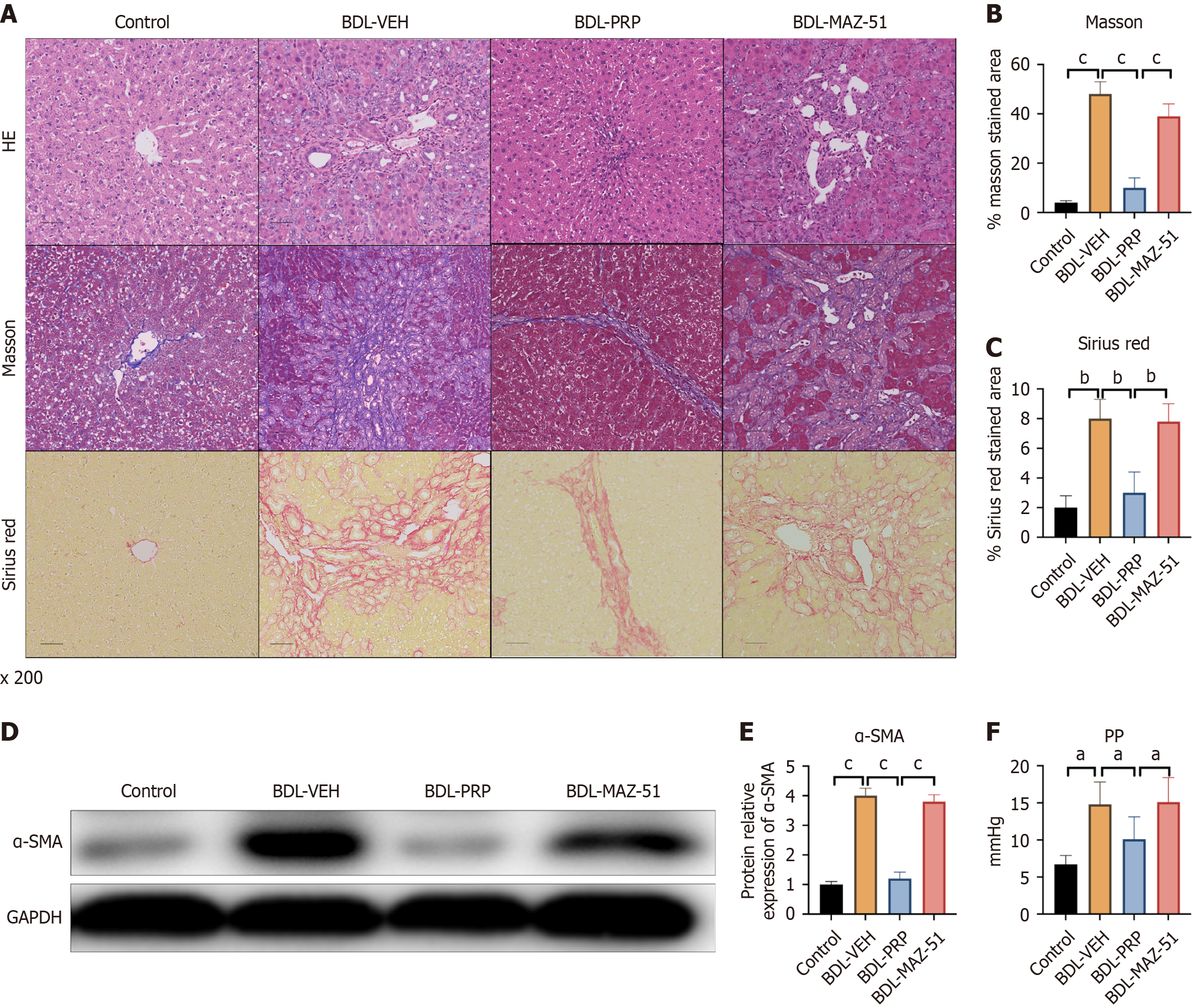
The BDL-VEH group exhibited notable liver fibrosis after BDL surgery. Following PRP intervention, a significant decrease in collagen deposition was evidenced by a decrease in Masson’s trichrome, H&E, and Sirius red staining (Figure 6A-C). However, MAZ-51 treatment induced collagen deposition in the liver, as indicated by increased H&E, Masson’s trichrome, and Sirius red staining compared with that in the BDL-PRP group (Figure 6A-C). The protein expression of α-SMA, a marker of liver fibrosis, showed a similar trend in the BDL-PRP and BDL-MAZ-51 groups (Figure 6D and E). PRP significantly decreased the BDL-induced increase in PP, and this phenomenon was reversed by MAZ-51 (Figure 6F).
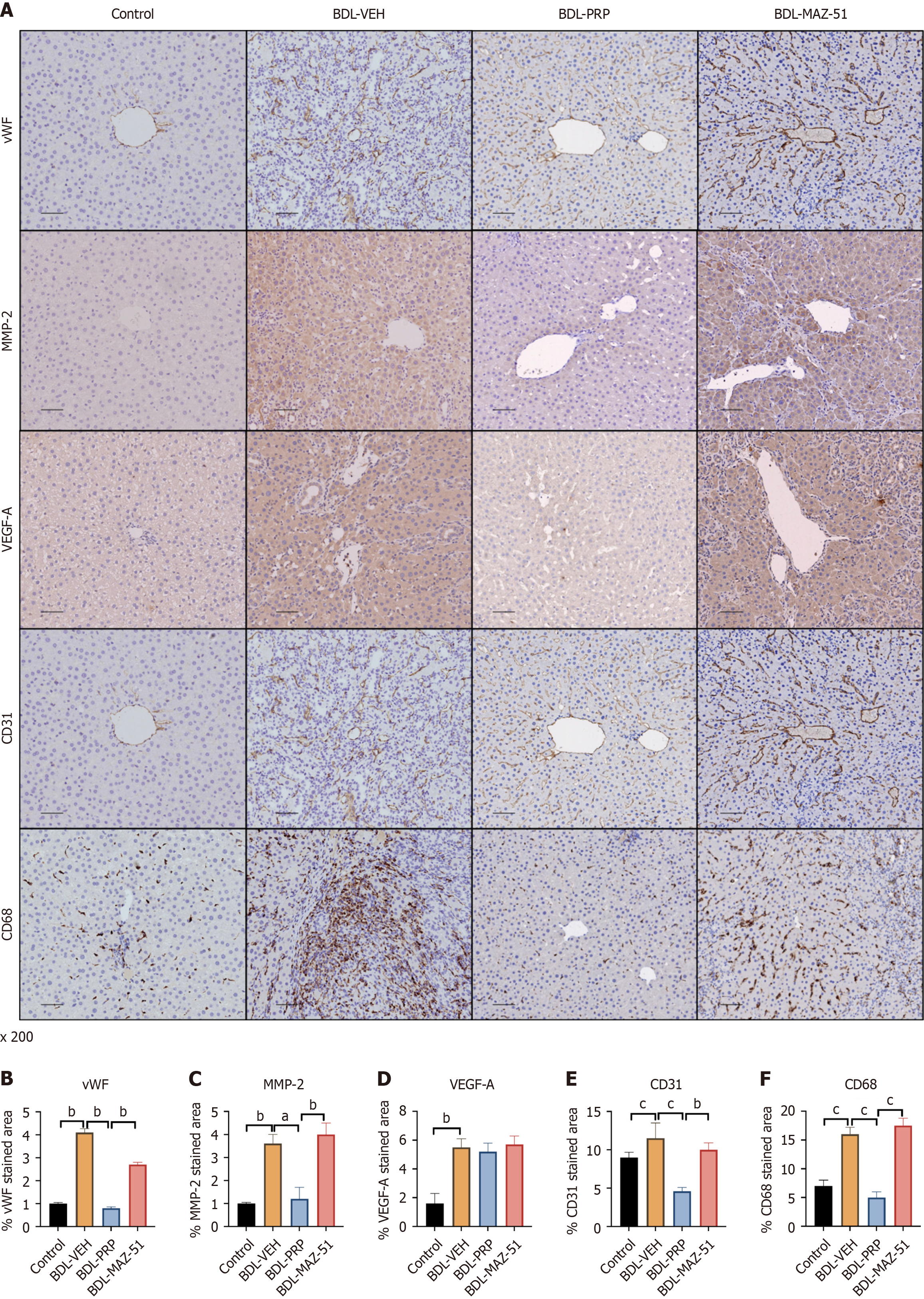
Immunohistochemistry analysis revealed a significant reduction in the elevated levels of angiogenesis- and remodeling-related proteins, namely, vWF, MMP2, VEGF-A, and CD31, in the BDL-PRP group compared with those in the BDL-VEH group. Furthermore, MAZ-51 treatment reversed this phenomenon (Figure 7A-E). In addition, hepatic expression of CD68 was elevated in the BDL-VEH group. However, upon intervention with PRP, the hepatic expression of CD68 was subsequently downregulated. Conversely, the PRP + MAZ-51 intervention, which inhibited lymphangiogenesis, in
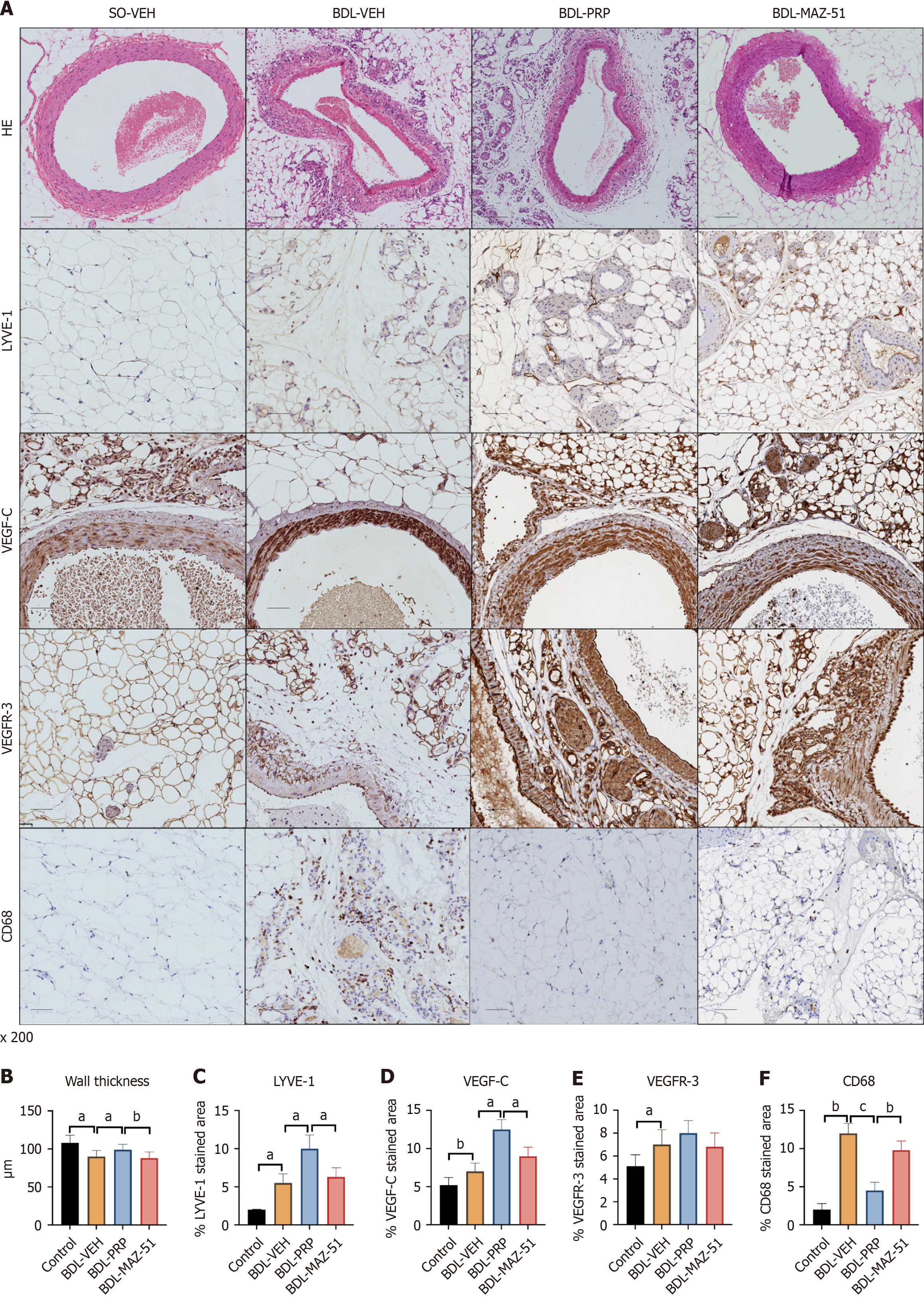
Compared with the control group, the BDL-VEH group presented a reduction in the SMA lumen thickness and disruption of the vascular structure. After PRP intervention, the SMA of the BDL-PRP group was restored towards a relatively normal state, as characterized by a thicker wall. Conversely, the utilization of PRP + MAZ-51 to inhibit lymphatic vessel formation led to a subsequent decrease in wall thickness (Figure 8A and B). Similarly, the levels of the LYVE-1, VEGF-C, and VEGFR-3 lymphatic system markers were increased in the BDL-VEH group and further increased in the BDL-PRP group; these effects were inhibited by MAZ-51 treatment (Figure 8A, C-E). In addition, the levels of the CD68 macrophage marker decreased after PRP intervention, while MAZ-51 treatment increased the CD68 levels in the mesentery (Figure 8A and F).
Cirrhosis and its associated complications contribute significantly to global mortality rates on an annual basis. Appro
The present study employed a method to augment the platelet count in rats, namely, PRP infusion. PRP effectively improves various liver diseases, such as ischemia-reperfusion injury, cirrhosis, cholestatic liver injury, viral hepatitis, and the generation of liver tissue[23-27]. Because platelets directly interact with liver cells through the hepatic sinuses, the growth factors carried by platelets in PRP effectively promote liver cell proliferation and prevent liver cell apoptosis[28,29]. PRP has positive effects on ameliorating carbon tetrachloride (CCl4)-induced liver injury, thioacetamide (TAA)-related liver injury, and liver fibrosis. However, the potential impact of PRP on PHT in advanced cirrhosis remains unexplored[30]. In addition to improving liver tissue, PRP may also promote lymphangiogenesis. Platelets in PRP have been reported to facilitate local lymphangiogenesis during wound healing through the release of VEGF-C[11]. VEGF-C is released upon activation by peripheral blood platelets, thereby contributing to the processes of wound healing and lymphangiogenesis[31]. In the present study, functional enrichment analysis revealed that the wound healing signaling pathway was enriched. The present study demonstrated that PRP-induced platelets led to intrahepatic lymphangio
The present study had several limitations. BDL, a classic PHT model, represents acute cholestatic cirrhosis, but the causes of PHT vary greatly. Poison-induced (CCl4, TAA, and alcohol), chronic cholestasis and genetic defect-induced PHT models need to be investigated. Due to time constraints, the present study lacked a comprehensive investigation into the impact of PRP on lymphangiogenesis and liver fibrosis at various stages.
In summary, lymphangiogenesis is enhanced in PHT models. In addition, the upregulation of the lymphatic system by AAV-VEGF-C alleviates fibrosis and inflammation in PHT. Sequencing analysis of extrahepatic lymphatics revealed a close association between lymphangiogenesis and platelet adherence, aggregation and activation. PRP-induced platelets have the following capabilities: Stimulate lymphangiogenesis; Effectively suppress liver fibrosis, intrahepatic angioge
| 1. | Gracia-Sancho J, Marrone G, Fernández-Iglesias A. Hepatic microcirculation and mechanisms of portal hypertension. Nat Rev Gastroenterol Hepatol. 2019;16:221-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 206] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 2. | Tsochatzis EA, Bosch J, Burroughs AK. Liver cirrhosis. Lancet. 2014;383:1749-1761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1536] [Cited by in RCA: 1354] [Article Influence: 112.8] [Reference Citation Analysis (1)] |
| 3. | Bosch J, Groszmann RJ, Shah VH. Evolution in the understanding of the pathophysiological basis of portal hypertension: How changes in paradigm are leading to successful new treatments. J Hepatol. 2015;62:S121-S130. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 210] [Cited by in RCA: 181] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 4. | Tanaka M, Iwakiri Y. The Hepatic Lymphatic Vascular System: Structure, Function, Markers, and Lymphangiogenesis. Cell Mol Gastroenterol Hepatol. 2016;2:733-749. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 108] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 5. | Maby-El Hajjami H, Petrova TV. Developmental and pathological lymphangiogenesis: from models to human disease. Histochem Cell Biol. 2008;130:1063-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 79] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 6. | van Dijk EL, Jaszczyszyn Y, Naquin D, Thermes C. The Third Revolution in Sequencing Technology. Trends Genet. 2018;34:666-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 967] [Cited by in RCA: 711] [Article Influence: 88.9] [Reference Citation Analysis (0)] |
| 7. | Schlesinger M. Role of platelets and platelet receptors in cancer metastasis. J Hematol Oncol. 2018;11:125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 578] [Cited by in RCA: 508] [Article Influence: 63.5] [Reference Citation Analysis (0)] |
| 8. | Anstee QM, Dhar A, Thursz MR. The role of hypercoagulability in liver fibrogenesis. Clin Res Hepatol Gastroenterol. 2011;35:526-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 9. | Zermatten MG, Fraga M, Moradpour D, Bertaggia Calderara D, Aliotta A, Stirnimann G, De Gottardi A, Alberio L. Hemostatic Alterations in Patients With Cirrhosis: From Primary Hemostasis to Fibrinolysis. Hepatology. 2020;71:2135-2148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 10. | Shen CL, Wang TF, Liu CZ, Wu YF. Platelet Activation and Cytokine Release of Interleukin-8 and Interferon-Gamma-Induced Protein 10 after ChAdOx1 nCoV-19 Coronavirus Vaccine Injection. Vaccines (Basel). 2023;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 11. | Lim L, Bui H, Farrelly O, Yang J, Li L, Enis D, Ma W, Chen M, Oliver G, Welsh JD, Kahn ML. Hemostasis stimulates lymphangiogenesis through release and activation of VEGFC. Blood. 2019;134:1764-1775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 12. | Chang T, Ho HL, Hsu SJ, Chang CC, Tsai MH, Huo TI, Huang HC, Lee FY, Hou MC, Lee SD. Glucobrassicin Metabolites Ameliorate the Development of Portal Hypertension and Cirrhosis in Bile Duct-Ligated Rats. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Wu G, Chen M, Fan Q, Li H, Zhao Z, Zhang C, Luo M. Transcriptome analysis of mesenteric arterioles changes and its mechanisms in cirrhotic rats with portal hypertension. BMC Genomics. 2023;24:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Vollmar B, Wolf B, Siegmund S, Katsen AD, Menger MD. Lymph vessel expansion and function in the development of hepatic fibrosis and cirrhosis. Am J Pathol. 1997;151:169-175. [PubMed] |
| 15. | Lukacs-Kornek V. The Role of Lymphatic Endothelial Cells in Liver Injury and Tumor Development. Front Immunol. 2016;7:548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 16. | Yokomori H, Oda M, Kaneko F, Kawachi S, Tanabe M, Yoshimura K, Kitagawa Y, Hibi T. Lymphatic marker podoplanin/D2-40 in human advanced cirrhotic liver--re-evaluations of microlymphatic abnormalities. BMC Gastroenterol. 2010;10:131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Henriksen JH. Estimation of lymphatic conductance. A model based on protein-kinetic studies and haemodynamic measurements in patients with cirrhosis of the liver and in pigs. Scand J Clin Lab Invest. 1985;45:123-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Juneja P, Ruhina Rahman SN, Jakhar D, Mourya AK, Tripathi DM, Kaur I, Tiwari V, Rohilla S, Gupta A, Rawal P, Baweja S, Rastogi A, Naidu VGM, Sarin SK, Banerjee S, Kaur S. Recombinant VEGF-C (Cys156Ser) improves mesenteric lymphatic drainage and gut immune surveillance in experimental cirrhosis. JHEP Rep. 2023;5:100816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 19. | Chen M, Wu GB, Xie ZW, Shi DL, Luo M. A novel diagnostic four-gene signature for hepatocellular carcinoma based on artificial neural network: Development, validation, and drug screening. Front Genet. 2022;13:942166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 20. | Ho-Tin-Noé B, Demers M, Wagner DD. How platelets safeguard vascular integrity. J Thromb Haemost. 2011;9 Suppl 1:56-65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 229] [Cited by in RCA: 202] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 21. | Sato H, Higashiyama M, Hozumi H, Sato S, Furuhashi H, Takajo T, Maruta K, Yasutake Y, Narimatsu K, Yoshikawa K, Kurihara C, Okada Y, Watanabe C, Komoto S, Tomita K, Nagao S, Miura S, Hokari R. Platelet interaction with lymphatics aggravates intestinal inflammation by suppressing lymphangiogenesis. Am J Physiol Gastrointest Liver Physiol. 2016;311:G276-G285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Suzuki-Inoue K. Essential in vivo roles of the platelet activation receptor CLEC-2 in tumour metastasis, lymphangiogenesis and thrombus formation. J Biochem. 2011;150:127-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 23. | Pak S, Kondo T, Nakano Y, Murata S, Fukunaga K, Oda T, Sasaki R, Ohkohchi N. Platelet adhesion in the sinusoid caused hepatic injury by neutrophils after hepatic ischemia reperfusion. Platelets. 2010;21:282-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 24. | Zaldivar MM, Pauels K, von Hundelshausen P, Berres ML, Schmitz P, Bornemann J, Kowalska MA, Gassler N, Streetz KL, Weiskirchen R, Trautwein C, Weber C, Wasmuth HE. CXC chemokine ligand 4 (Cxcl4) is a platelet-derived mediator of experimental liver fibrosis. Hepatology. 2010;51:1345-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 132] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 25. | Laschke MW, Dold S, Menger MD, Jeppsson B, Thorlacius H. Platelet-dependent accumulation of leukocytes in sinusoids mediates hepatocellular damage in bile duct ligation-induced cholestasis. Br J Pharmacol. 2008;153:148-156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 74] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 26. | Lang PA, Contaldo C, Georgiev P, El-Badry AM, Recher M, Kurrer M, Cervantes-Barragan L, Ludewig B, Calzascia T, Bolinger B, Merkler D, Odermatt B, Bader M, Graf R, Clavien PA, Hegazy AN, Löhning M, Harris NL, Ohashi PS, Hengartner H, Zinkernagel RM, Lang KS. Aggravation of viral hepatitis by platelet-derived serotonin. Nat Med. 2008;14:756-761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 187] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 27. | Myronovych A, Murata S, Chiba M, Matsuo R, Ikeda O, Watanabe M, Hisakura K, Nakano Y, Kohno K, Kawasaki T, Hashimoto I, Shibasaki Y, Yasue H, Ohkohchi N. Role of platelets on liver regeneration after 90% hepatectomy in mice. J Hepatol. 2008;49:363-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 101] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 28. | Watanabe M, Murata S, Hashimoto I, Nakano Y, Ikeda O, Aoyagi Y, Matsuo R, Fukunaga K, Yasue H, Ohkohchi N. Platelets contribute to the reduction of liver fibrosis in mice. J Gastroenterol Hepatol. 2009;24:78-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 84] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 29. | Hisakura K, Murata S, Fukunaga K, Myronovych A, Tadano S, Kawasaki T, Kohno K, Ikeda O, Pak S, Ikeda N, Nakano Y, Matsuo R, Konno K, Kobayashi E, Saito T, Yasue H, Ohkohchi N. Platelets prevent acute liver damage after extended hepatectomy in pigs. J Hepatobiliary Pancreat Sci. 2010;17:855-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 30. | Hesami Z, Jamshidzadeh A, Ayatollahi M, Geramizadeh B, Farshad O, Vahdati A. Effect of Platelet-Rich Plasma on CCl4-Induced Chronic Liver Injury in Male Rats. Int J Hepatol. 2014;2014:932930. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Wartiovaara U, Salven P, Mikkola H, Lassila R, Kaukonen J, Joukov V, Orpana A, Ristimäki A, Heikinheimo M, Joensuu H, Alitalo K, Palotie A. Peripheral blood platelets express VEGF-C and VEGF which are released during platelet activation. Thromb Haemost. 1998;80:171-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 186] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 32. | Maruyama T, Murata S, Takahashi K, Tamura T, Nozaki R, Ikeda N, Fukunaga K, Oda T, Sasaki R, Ohkohchi N. Platelet transfusion improves liver function in patients with chronic liver disease and cirrhosis. Tohoku J Exp Med. 2013;229:213-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 33. | Fernandez M. Molecular pathophysiology of portal hypertension. Hepatology. 2015;61:1406-1415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 34. | Kostallari E, Shah VH. Pericytes in the Liver. Adv Exp Med Biol. 2019;1122:153-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 35. | Corpechot C, Barbu V, Wendum D, Kinnman N, Rey C, Poupon R, Housset C, Rosmorduc O. Hypoxia-induced VEGF and collagen I expressions are associated with angiogenesis and fibrogenesis in experimental cirrhosis. Hepatology. 2002;35:1010-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 368] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 36. | Uschner FE, Schueller F, Nikolova I, Klein S, Schierwagen R, Magdaleno F, Gröschl S, Loosen S, Ritz T, Roderburg C, Vucur M, Kristiansen G, Lammers T, Luedde T, Trebicka J. The multikinase inhibitor regorafenib decreases angiogenesis and improves portal hypertension. Oncotarget. 2018;9:36220-36237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (3)] |
| 37. | Tripodi A, Primignani M, Chantarangkul V, Lemma L, Jovani M, Rebulla P, Mannucci PM. Global hemostasis tests in patients with cirrhosis before and after prophylactic platelet transfusion. Liver Int. 2013;33:362-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 88] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/