Published online Jul 28, 2022. doi: 10.3748/wjg.v28.i28.3573
Peer-review started: January 17, 2022
First decision: April 11, 2022
Revised: April 19, 2022
Accepted: June 26, 2022
Article in press: June 26, 2022
Published online: July 28, 2022
Processing time: 191 Days and 2.2 Hours
The management of hepatocellular carcinoma (HCC) is challenging because most patients have underlying cirrhosis, and the treatment provides, historically, a limited impact on the natural history of patients with advanced-stage disease. Additionally, recurrence rates are high for those patients who receive local and locoregional modalities, such as surgical (resection and transplantation) or image-guided (ablation and intra-arterial) therapies. Translational research has led to new concepts that are reshaping the current clinical practice. Substantial advancements were achieved in the understanding of the hallmarks that drive hepatocarcinogenesis. This has primed a successful incorporation of novel agents with different targets, such as anti-angiogenic drugs, targeted-therapies, and immune-checkpoint inhibitors. Although clinical trials have proven efficacy of systemic agents in advanced stage disease, there is no conclusive evidence to support their use in combination with loco-regional therapy. While novel local modalities are being incorporated (e.g., radioembolization, microwave ablation, and irreversible electroporation), emerging data indicate that locoregional treatments may induce tumor microenvironment changes, such as hyperexpression of growth factors, release of tumor antigens, infiltration of cytotoxic lymphocytes, and modulation of adaptative and innate immune response. Past trials that evaluated the use of antiangiogenic drugs in the adjuvant setting after ablation or chemoembolization fail to demonstrate a substantial improvement. Current efforts are directed to investigate the role of immunotherapy-based regimens in this context. The present review aims to describe the current landscape of systemic and locoregional treatments for HCC, present evidence to support combination approaches, and address future perspectives.
Core Tip: Management of hepatocellular carcinoma (HCC) is based on stages defined by tumor burden, liver function, and performance status. With the advent of more effective systemic treatments, such as immunotherapy and immunotherapy-based combinations, patients with advanced stage disease have better outcomes. The migration of systemic treatment to earlier stages, in combination with locoregional therapies, are expected to improve the outcomes and cure rates. Currently, the research field is moving towards an increasing interest in combining locoregional and systemic treatments for HCC.
- Citation: da Fonseca LG, Araujo RLC. Combination approaches in hepatocellular carcinoma: How systemic treatment can benefit candidates to locoregional modalities. World J Gastroenterol 2022; 28(28): 3573-3585
- URL: https://www.wjgnet.com/1007-9327/full/v28/i28/3573.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i28.3573
Hepatocellular carcinoma (HCC) is a highly fatal disease, representing the fourth cause of cancer-related mortality worldwide[1]. This situation is attributed to the challenging management of patients with HCC per se in concomitance with an underlying liver disease and cirrhosis-related complications. Moreover, a significant proportion of patients are diagnosed with advanced stage disease, not amenable to curative options. Finally, even patients who are treated with local therapies, such as surgery and ablation, present high rates of recurrence, reaching up to 70% in 5 years[2].
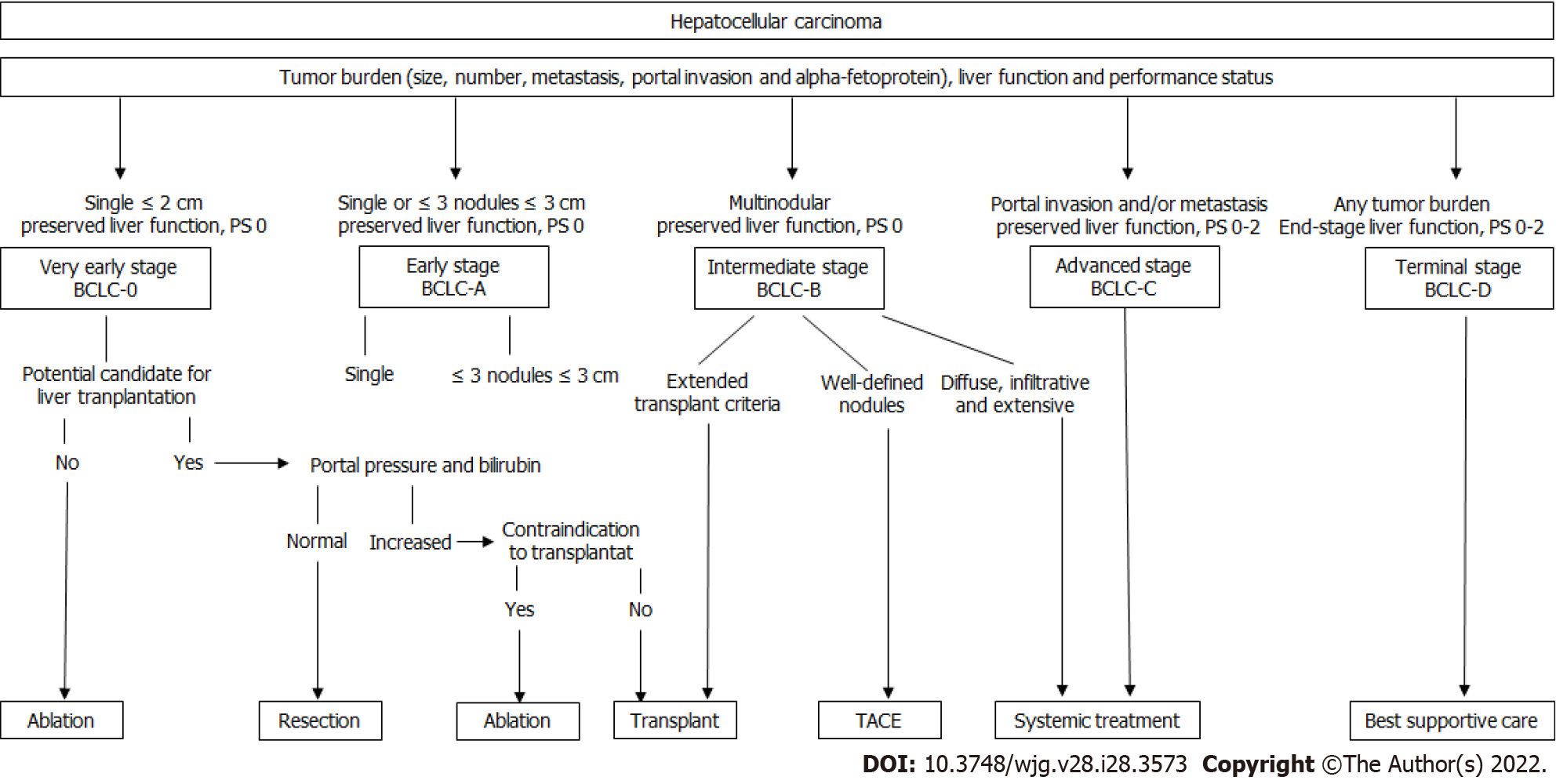
Treatment options for HCC are selected based on liver function, performance status, and tumor burden (size, number of lesions, metastatic spread, and vascular invasion). These patient- and tumor-centered characteristics define the stage of the cancer, each with a different prognosis[3]. Thus, clinical practice guidelines according to both tumor presentation and liver function integrate the available evidence based on well-delineated clinical trials and recommend treatment strategies for each stage[2,4] (Figure 1).
Liver resection, local ablation, and liver transplantation are recommended for very-early and early-stage disease and are considered curative-intent treatments for HCC. Intra-arterial therapies, such as transarterial chemoembolization (TACE), are indicated for cases classified as intermediate stage (multinodular liver disease beyond Milan-criteria with preserved liver function), and systemic treatments are used for patients with advanced stage disease[3]. Therapeutic management of HCC is evolving rapidly with the incorporation of novel locoregional techniques and the increasing number of systemic agents tested in clinical trials, including immunotherapy. Each treatment approach is directed to treat HCC at different stages, and there is a lack of evidence to support the use of combined systemic and locoregional treatments. Therefore, the biological rational behind combination strategies and the urgent need to improve outcomes for this lethal disease prime intense research activity in both basic and clinical fields. This review aims to address the current landscape of systemic and locoregional treatments and the future perspectives regarding combined approaches.
Systemic treatment is recommended for patients with advanced stage disease (preserved liver function, performance status 0-2, metastatic spread, and/or macrovascular invasion) and for patients in earlier stages who have contra-indications or progressed after locoregional modalities according to the concept of treatment stage migration[3].
The natural history of advanced HCC is poor, with a median overall survival of 4-8 mo without active treatments[5]. Since 2008, the use of agents that target hallmarks of hepatocarcinogenesis are gradually improving prognosis and achieving substantial clinical benefit for HCC patients. Sorafenib, a multikinase inhibitor, has been shown to improve overall survival over placebo in the SHARP and Asia-Pacific trials and became the first approved drug for advanced HCC[6,7]. After almost a decade, other drugs of the same class were tested with positive results in phase III trials, such as lenvatinib (non-inferior to sorafenib in first-line)[8], regorafenib (superior over placebo in second-line for patients who tolerated sorafenib)[9], and cabozantinib (superior over placebo after sorafenib failure)[10]. These drugs share common targets, such as vascular endothelial growth factor receptor (VEGFR) and platelet-derived growth factor receptor, while some of them present, individually, more direct actions against specific targets, such as the case of lenvatinib against fibroblast growth factor receptor, and cabozantinib against mesenchymal-epithelial transition (MET) receptor. Additionally, ramucirumab, a monoclonal antibody against VEGFR, was shown to improve overall survival over placebo in patients who present with alpha-fetoprotein (AFP) ≥ 400 ng/mL after sorafenib failure and is also approved for clinical use[11]. Generally, these drugs present antiangiogenic activity and promote clinical benefit by delaying tumor progression rather than inducing a substantial reduction in tumor burden. Trials with these drugs are consistent in showing a statically significant, but modest, increase in overall survival, with survivals around 11-13 mo in the first line, a response rate of less than 10% (except for lenvatinib, with a response rate of 18.8% in the REFLECT trial), and a range of class-related adverse events, such as fatigue, arterial hypertension, skin reaction, and diarrhea[12].
In the past years, the advent of immune-based treatments, such as immune checkpoint inhibitors, brought huge advances in HCC management. The biological background for the use of immunotherapy in HCC is based on the remarkable immunotolerance of the liver due to the high antigenic load derived from the enteral-portal circulation. Additionally, HCC develops in a microenvironment of chronic inflammation and underlying liver disease. Low infiltration of CD8 + T lymphocytes, responsible for the antitumor immune response, and a marked presence of exhausted lymphocytes and regulatory T lymphocytes are described in HCC, contributing to an immunosuppressive and procarcinogenic microenvironment[13,14].
The immune checkpoint inhibitors aimed at boosting anti-tumoral immunity in the priming phase by the recognition of antigens presented by dendritic cells to lymphocytes (anti-CTLA4 agents) and in the effector phase of T-CD8 cytotoxic cells against tumor cells (anti-PD1, anti-PDL1) were tested in phase I/II trials showing durable response rates of around 15%-20%. Therefore, pembrolizumab and nivolumab (anti-PD1 drugs) received approval for use in second-line treatments[15,16]. However, comparative trials showed that immune checkpoint inhibitors as monotherapy did not improve outcomes comparing to the available treatments. In a phase III trial, nivolumab was not proven to improve survival compared to sorafenib in the first line[17] and pembrolizumab did not achieve statistical superiority over placebo in the second-line[18].
The use of combinations of agents with different mechanisms of action can improve results of immunotherapy through additive effect and because VEGF can enhance the immunosuppression of the tumor microenvironment by inhibiting the function of effector T cells, increasing the recruitment of regulatory T cells and myeloid-derived suppressor cells[19]. This combination was explored in the IMBRAVE150 trial, which randomized patients to receive sorafenib vs a combination of bevacizumab (an anti-VEGF antibody) and atezolizumab (an anti-PD-L1 antibody). This trial demonstrated a significant improvement in overall survival (19.2 mo vs 13.4 mo), and also in progression-free survival and response rate[20,21]. IMBRAVE150 trial was a landmark in HCC management and marked the transition towards the use of combined systemic therapies (using dual-immunotherapy or antiangiogenic plus immunotherapy combinations). Several combinations showed encouraging results in phases I and II trials, such as lenvatinib plus pembrolizumab[22], pembrolizumab plus regorafenib[23], nivolumab plus ipilimumab[24], and nivolumab, ipilimumab, and cabozantinib[25]. These preliminary results suggested that combinations may yield response rates of more than 20% according to RECIST criteria, which seems to compare favorably to the rates with multikinase inhibitors. However, it is important to point out some limitations of early phase trials. Firstly, non-comparative trials are not adequate to draw definitive conclusions. Besides, the response rate is not a surrogate marker for survival benefit in HCC, even more considering that RECIST may not capture the spectrum and patterns of progression and response in patients under immunotherapy.
Recently, durvalumab and tremelimumab as a combination treatment was announced to yield survival benefit over sorafenib in a phase III trial and is expected to be incorporated in the first-line setting as an alternative to atezolizumab plus bevacizumab[26,27]. Currently, other phase III trials are awaiting results in the context of combination therapies for advanced HCC (Table 1).
| Study name | Design | Experimental arm | Median overall survival, mo | Response rate- RECIST, % | Grade 3 - 4 treatment-related adverse events |
| IMBRAVE 150[21] | Phase III; First-line | Atezolizumab + Bevacizumab | 19.2 | 30% | 36% |
| KEYNOTE 524/Study 116[22] | Phase Ib; First-line | Pembrolizumab + Lenvatinib | 22 | 36% | 67% |
| REG-PEMBRO-HCC[23] | Phase Ib; First-line | Pembrolizumab + Regorafenib | 26.5 | 32% | 86% |
| CHECKMATE 040[24] | Phase II; Second-line | Nivolumab + Ipilimumab (arm A) | 22.8 | 32% | 53% |
| STUDY 22[27] | Phase II; Second-line | Durvalumab + Tremelimumab | 18.7 | 24% | 35.1% |
| CHECKMATE-040[25] | Phase II; First and second-line | Nivolumab + Ipilimumab + Cabozantinib | Not-reached | 26% | 71% |
Liver resection is indicated in early HCC, regardless of the presence of cirrhosis, since the liver function remains compensated, and is rarely is does the patient have a clinically significant portal hypertension. However, the risk of liver decompensation after resection in patients with restricted hepatic functional reserve is a concern, especially in cases requiring major hepatectomies (3 or more segments)[2,28,29]. On the other side, liver transplantation has the advantage of treating both cirrhosis and offer curative-intent treatment for HCC. Thus, liver transplantation decreases both the risk of recurrence, since the most recurrence is in the liver, and de novo lesions by removing the cirrhotic liver. But its application in clinical practice is hampered by organ shortage, complexity, heterogenous availability in different worldwide regions, and the risk of progression on waiting list[30].
In patients with liver-only disease not amenable to surgical modalities, locoregional interventional procedures play a key role. Locoregional treatment is defined as imaging-guided tumor directed procedures, and it is estimated that 50% of patients with HCC might receive any of these treatments during the course of the disease[31,32]. Basically, there are two groups of locoregional therapies: (1) Ablative therapies; or (2) Intra-arterial therapies. Although both modalities are consolidated with high level evidence, emerging approaches and techniques are being increasingly adopted and are expected to improve clinical outcomes. Besides, the risk of recurrence and progression in patients treated with locoregional therapies has led to intense research activity towards combination with systemic treatment[2,4].
Ablative therapies induce tumor destruction through different mechanisms according to the method: Chemical [percutaneous ethanol injection (PEI)], thermal [radiofrequency ablation (RFA), microwave (MWA), and cryoablation], and short pulses of high voltage (irreversible electroporation). In general, ablative techniques are considered a curative therapy for HCC < 2-3 cm and is associated with complete responses in 70%-90% of the cases, although recurrences occur in approximately 50% of the cases within 5 years[33-35].
The most used and recommended method is RFA, which has shown survival superiority over PEI in randomized trials[33]. Tumor size, number of nodules, and Child-Pugh class are associated with prognosis in patients treated with RFA[36-38]. Comparative studies vs resection demonstrated that RFA seems to be inferior to surgery in tumors > 3 cm[39]. Limitations to RFA include proximity to large vessels (due to heat effect), size, and location.
MWA is another technique that has been increasingly used and has the advantage of achieving a faster heating over a larger volume and being less susceptible to heat sink effect. Most studies that addressed comparisons between RFA and MWA showed similar efficacy, with a trend toward best results with MWA in tumors > 3 cm[40-42].
Alternative techniques, such as IRE and cryoablation, are under active research, with a small series of studies showing encouraging results[43,44]. IRE has the advantage of causing less thermal damage to adjacent tissues. However, more data are still required for IRE and cryoablation in order to fully incorporate these techniques into clinical practice guidelines.
The high risk of local and distant recurrence after ablation indicates the need for adjuvant strategies to improve cure rates. Features, such as large tumors, multinodularity, and vascular invasion (macroscopic or microscopic), are significantly related to higher recurrence rates. Novel markers, such as genetic signatures, circulating microRNA, circulating non-coding RNA, circulating cell-free DNA, gut microbiota, and circulating tumor cells, have also been shown to predict the risk of recurrence[45-47].
To evaluate the impact of multikinase inhibitors with antiangiogenic activity combined with ablation, the STORM trial randomized patients treated with resection (n = 900) or ablation (n = 214) to sorafenib or placebo for a total of 4 years. Unfortunately, there was no difference in recurrence-free survival between groups, with a median to time to recurrence of 33.3 mo in the sorafenib group and 33.7 mo for the placebo group (HR = 0.94, P = 0.26)[48]. Thus, the positive results obtained with sorafenib in advanced disease have not been reproduced in the context of adjuvant post-resection or ablation. Other drugs used in the advanced stage, such as cabozantinib, regorafenib, ramucirumab, and lenvatinib have not been evaluated in patients after ablation. In sum, VEGFR-directed therapies as monotherapy do not seem to improve the results of ablation.
Recently, the immune features of localized HCC and the response to local treatment are being investigated[49-51]. The concentration of intratumor CD3 + and CD8 + T cells and the expression of programmed death ligand 1 (PD-L1) by immune and tumor cells appear to be associated with HCC aggressiveness and risk of recurrence after curative treatments, such as ablation and surgery[13]. The high expression of PD1 can lead a condition of exhausted CD8 + impairing cytotoxicity and decreasing of pro-inflammatory cytokines production and anti-tumor ability[13].
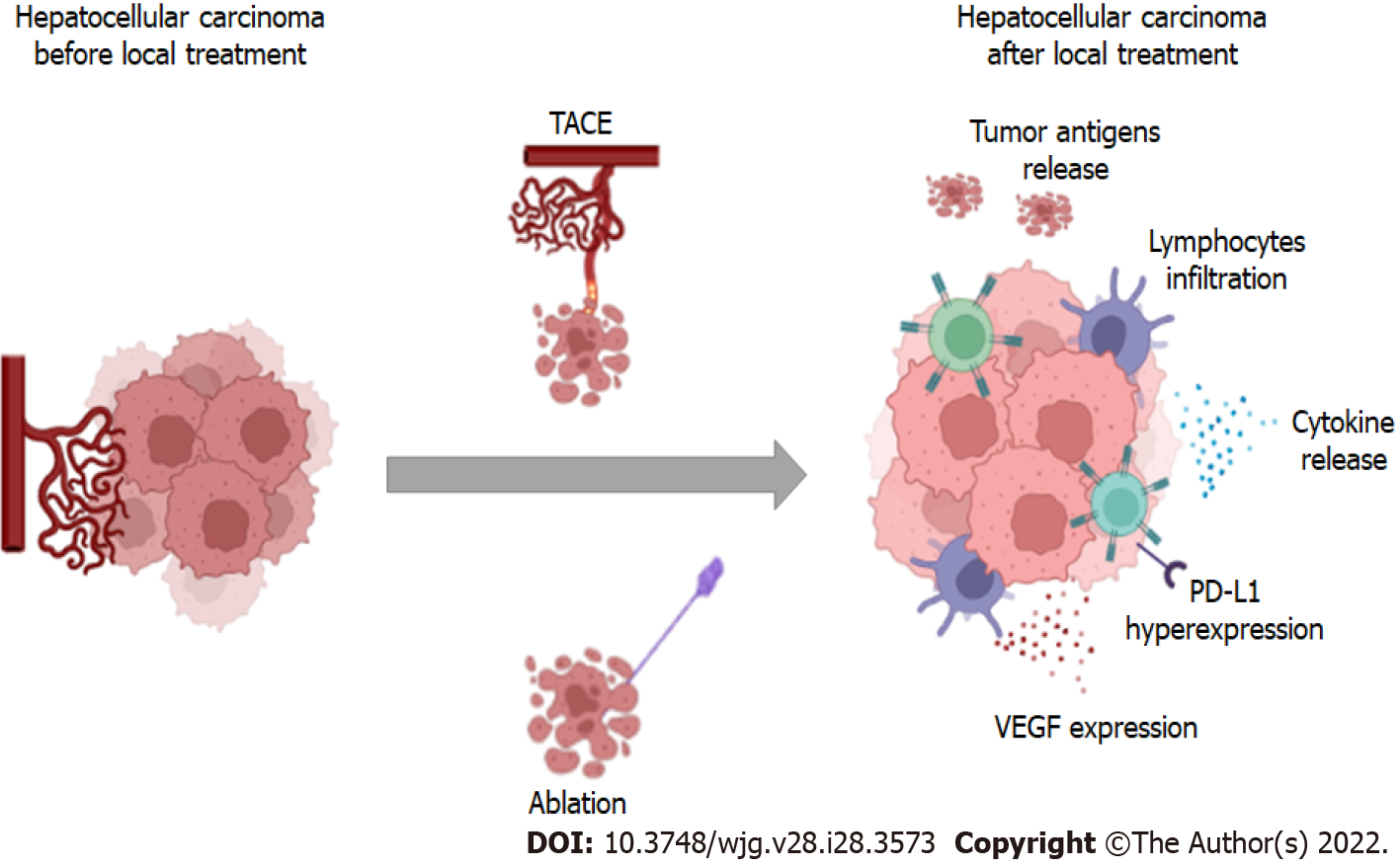
Ablation can induce stimulate inflammatory and cytokine production within the treated site. The tumor debris released upon ablation represent a tumor antigen repertoire that can boost antitumoral immunologic response. Indeed, tumor antigens can be found in dendritic cells in lymph nodes following ablation[52]. This background indicates a potential role for immune checkpoint inhibitors directed to PD1 and PD-L1 in combination with ablative therapies (Figure 2). A phase I trial including 32 patients treated with tremelimumab and ablation showed accumulation of CD8+ cells, suggesting immune system activation[53]. Preliminary results of the NIVOLVE trial were presented in 2021. In this phase II single arm trial, 55 patients after surgery (n = 33) or ablation (n = 22) received nivolumab for 12 mo. The median recurrence-free survival was 26 mo. In this trial, the authors suggested that patients with tumor-infiltrating lymphocytes, positive PD-L1, and negative staining for beta-catenin tend to develop fewer recurrences[54]; however, larger and comparative studies are needed to confirm these findings. Currently, several active trials (recruiting or waiting results) are addressing the role of immune-checkpoint inhibitors in combination with anti-VEGFR agents or alone after curative treatments, such as ablation (Table 2).
| Trial | Trial registration | Drug | Control | n | Disease stage | Local treatment | Expected termination | Primary end-point |
| CHECKMATE 9DX | NCT03383458 | Nivolumab | Placebo | 530 | Early/intermediate | Surgery/Ablation | 2025 | Recurrence-free survival |
| KEYNOTE 937 | NCT03867084 | Pembrolizumab | Placebo | 950 | Early/intermediate | Surgery/Ablation | 2025 | Recurrence-free survival and overall survival |
| IMBRAVE 050 | NCT04102098 | Atezolizumab + Bevacizumab | Surveillance | 662 | Early/intermediate /advanced | Surgery/Ablation | 2027 | Recurrence-free survival |
| EMERALD-2 | NCT03847428 | Durvalumab + Bevacizumab | Placebo | 888 | Early/intermediate | Surgery/Ablation | 2024 | Recurrence-free survival |
Intra-arterial therapies harness the arterial supply of HCC that differs from the liver parenchyma, which receives its blood supply predominantly from the portal vein. This group of therapies are composed of transarterial embolization (TAE), chemoembolization (TACE), and selective internal radiotherapy (SIRT) or radioembolization (TARE).
The mechanism of action of TAE is based on the obstruction of the blood supply causing ischemia, while TACE adds a cytotoxic agent (most commonly doxorubicin) and a carrier agent (commonly lipiodol). Drug-eluting bead (DEB)-TACE uses microspheres carrying cytotoxic agents which are associated with a sustained drug release and, theoretically, a lower systemic exposure to chemotherapy[55].
Based on randomized clinical trials of TACE vs no active treatment and also in a large meta-analysis, this is considered the standard of care for patients with intermediate-stage [Barcelona clinic liver cancer (BCLC)-B: Multinodular disease, preserved liver function, and performance status of 0][56,57]. In published cohorts with selected patients, the median overall survival with TACE reaches 48 mo[58]. There is no definitive evidence for the superiority of TACE over TAE, although data from meta-analysis suggests that the former is associated with better outcomes[57]. A prospective trial demonstrated that DEB-TACE presents lower treatment-related toxicities and a trend towards a better objective response rate compared to conventional TACE[59].
Guidelines recommend that patients who present progression with infiltrative tumors, vascular invasion, metastatic spread, or absence of a significant response after two TACEs should be considered for systemic treatment[2,4]. The risk of liver deterioration should be carefully monitored after TACE due to the prognostic impact of liver decompensation in patients with HCC[60]. The decision to declare TACE failure and switch to systemic treatment is heterogenous in different parts of the world, and many scores have been proposed to help in this decision, although some of them still require further validation in a population treated strictly according to the BCLC algorithm[61-65]. For example, the hepatoma-arterial embolization (HAP) score includes albumin, AFP, bilirubin and tumor diameter and was concepted to predict prognosis before the first TACE in a heterogeneous cohort with around 30% of BCLC-C patients[64]. The Assessment for Retreatment with TACE (ART) score was designed to evaluate benefit of a 2nd TACE and assess Child-Pugh score, AST levels, and tumor response after the first TACE. The ART score was originally developed in a cohort that included around 20% of patients with impaired liver function (Child-Pugh B8 or more)[66].
With the availability of more effective systemic agents, the migration from TACE to systemic treatment is a key decision in the management of HCC and should not be delayed at a point of irreversible deterioration of liver function or performance status.
TARE has been increasingly studied in HCC and has become a common practice in many centers. Data comparing TACE to TARE demonstrated in patients with early or intermediate stage a longer time to progression favoring TARE, but no survival benefit[67]. Although encouraging, there is no solid evidence that TARE should became standard of care in patients eligible to intra-arterial therapies.
A recent single-arm retrospective study including patients with solitary HCC ≤ 8 cm reported objective response rates of 88.3% and a 3-years survival of 86.6%. In this study TARE served as a neoadjuvant therapy for transplantation or resection in 21% and 6.8% of patients, respectively[68].
In uncontrolled trials, TARE was suggested to be safe in patients with portal vein invasion[69]. However, randomized trials in the setting of advanced stage HCC did not prove superiority of TARE over sorafenib in overall survival[70,71]. Therefore, TARE is not recommended as a standard therapy in patients classified as BCLC-C. There is a need to better explore the role of TARE in HCC in prospective and randomized trials, especially for subgroups of patients requiring downstaging, segmental portal vein thrombosis, and using rigorous dosimetry.
Although TACE is the standard treatment for intermediate stage, only around 50% of patients present objective response rate and most of them will eventually present distant and local progression[72]. This has led to studies focused on improving the outcomes for HCC patients treated with TACE by combining multikinase inhibitors.
The rational for testing these combinations comes from the demonstration of increased intratumorally micro vessel density and VEGF expression in residual tumors after TACE, suggesting that TACE may stimulate tumor angiogenesis[73]. However, antiangiogenic drugs, such as sorafenib[74,75], brivanib[76], and bevacizumab[77], failed to demonstrated survival benefit in combination with TACE in controlled trials.
Recently, TACTIS trials randomized patients to TACE alone or combined with sorafenib and showed that sorafenib improved progression-free survival (25.2 mo vs 13.5 mo; P = 0.006), but with no significant impact on overall survival (36.2 mo vs 30.8 mo; P = 0.40)[78,79]. Currently, available evidence fails to encourage the use of multikinase inhibitors in combination with TACE as a standard practice, but there is a growing enthusiasm with the use of immunotherapy in this setting.
Similar to ablative treatments, intra-arterial therapies may modulate innate and adaptative immunity by releasing cellular debris and inflammatory cytokines and inducing T-cell responses, what is suggested to be related to improved prognosis[80,81] (Figure 2). A recent study compared HCC tissue from patients who were previously submitted to TACE to patients not treated with TACE and found that the former had a lower density of immune-exhausted effector cytotoxic and T-regs with an upregulation of pro-inflammatory pathways[82]. Considering this background, strategies aimed at blocking negative regulators such as PD-1 and CTLA4 are promising and are being investigated in phase III clinical trials (Table 3).
| Trial | Trial registration | Drug | Control | n | Disease stage | Local treatment | Expected termination | Primary end-point |
| EMERALD-1 | NCT03778957 | Durvalumab + Bevacizumab plus TACE | TACE plusplacebo | 600 | Intermediate/advanced | TACE | 2024 | Progression-free survival |
| TACE-3 | NCT04268888 | Nivolumab plus DEB- TACE | DEB- TACE | 522 | Intermediate | DEB-TACE | 2026 | Overall survival |
| LEAP-012 | NCT04246177 | Lenvatinib plus Pembrolizumab plus cTACE | cTACE | 950 | Intermediate | Surgery/Ablation | 2029 | Overall survival and progression-free survival |
| CheckMate 74 W | NCT04340193 | Nivolumab plusIpilimumab/placebo plus cTACE | cTACE plusplacebo | 765 | Intermediate | Surgery/Ablation | Non-available | Time-to-progression and overall survival |
TARE also seems to modulate immunological microenvironment in HCC. A study that compared findings before and after TARE showed that tumor-infiltrating lymphocytes after TARE exhibited signs of immune activation, such as higher expression of granzyme-B and infiltration of CD8+Tcells, NK cells, and antigen-presenting cells[83,84]. Initial data on the combination of nivolumab and SIRT showed favorable safety profile, but efficacy data are pending[85,86].
Besides the biological background, the use of systemic treatment in earlier stages (combined with locoregional treatment) may benefit those patients who would present clinical deterioration or liver function worsening during TACE-refractoriness and would not found fit to receive further systemic treatment in that situation.
The treatment landscape of HCC management evolved rapidly in the past years. The most relevant achievements took place in the management of advanced stage disease. The median overall survival in pivotal clinical trials shifted from 5-8 mo in placebo arms[6], to almost 20 mo with immunotherapy combinations[21]. Still, some questions remain to be answer regarding the identification of predictive biomarkers and the optimal sequence in first-, second-, and later lines[87-89].
On the other, the field evolved modestly in the setting of local and locoregional stages, comprising early and intermediate stages. Patients in the early stage are offered surgical resection, liver transplantation, or ablation. Due to organ shortage and strict criteria to transplant, surgery and ablation has been widely used. Unfortunately, their recurrence rates are high and determine results in long-term mortality[33-35]. Although attempts were made, no systemic treatment has improved outcomes in early stage. Robust evidence supports those immunological changes occur after tumor ablation and the use immunotherapy is encouraging.
Patients in the intermediate stage have historically been offered TACE with the aim to achieve tumor control. Similarly, most of them will eventually present local or distant progression and will be considered for systemic treatment. The concomitant use of immunotherapy-based systemic agents and intra-arterial therapies may enhance anti-tumor immune response and impact positively on the outcomes. Several clinical trials are eagerly awaited.
The most promising ongoing trials approach the combination of TACE and immunotherapy, with the aim to significantly delay time-to-progression and, hopefully, increase cure rates for patients who achieve complete response with locoregional treatment. Similarly, trials that test adjuvant immunotherapy after ablation may increase cure rates by reducing the risk of recurrence and -de novo tumors. It is likely that biomarkers and subsets of clinical features will emerge from these trials and will support the selection of patients that will derive more benefit from combination approaches.
Finally, it is key to consider that if active trials succeed in showing a better anti-tumor efficacy with combination of locoregional and systemic treatment, a longer survival of patients with HCC will be expected. Furthermore, this will highlight the burden of cirrhosis-related complications as a competing risk of mortality and the importance of multidisciplinary teams as the standard approach for HCC.
| 1. | Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, Gores G. Hepatocellular carcinoma. Nat Rev Dis Primers. 2016;2:16018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2068] [Cited by in RCA: 1955] [Article Influence: 195.5] [Reference Citation Analysis (2)] |
| 2. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6763] [Cited by in RCA: 6559] [Article Influence: 819.9] [Reference Citation Analysis (2)] |
| 3. | Reig M, Forner A, Rimola J, Ferrer-Fàbrega J, Burrel M, Garcia-Criado Á, Kelley RK, Galle PR, Mazzaferro V, Salem R, Sangro B, Singal AG, Vogel A, Fuster J, Ayuso C, Bruix J. BCLC strategy for prognosis prediction and treatment recommendation: The 2022 update. J Hepatol. 2022;76:681-693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3613] [Cited by in RCA: 3292] [Article Influence: 823.0] [Reference Citation Analysis (3)] |
| 4. | Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR, Zhu AX, Murad MH, Marrero JA. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3374] [Cited by in RCA: 3168] [Article Influence: 396.0] [Reference Citation Analysis (6)] |
| 5. | Llovet JM, Bustamante J, Castells A, Vilana R, Ayuso Mdel C, Sala M, Brú C, Rodés J, Bruix J. Natural history of untreated nonsurgical hepatocellular carcinoma: rationale for the design and evaluation of therapeutic trials. Hepatology. 1999;29:62-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 962] [Cited by in RCA: 906] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 6. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11029] [Cited by in RCA: 10354] [Article Influence: 575.2] [Reference Citation Analysis (8)] |
| 7. | Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS, Xu J, Sun Y, Liang H, Liu J, Wang J, Tak WY, Pan H, Burock K, Zou J, Voliotis D, Guan Z. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4833] [Cited by in RCA: 4629] [Article Influence: 272.3] [Reference Citation Analysis (6)] |
| 8. | Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, Baron A, Park JW, Han G, Jassem J, Blanc JF, Vogel A, Komov D, Evans TRJ, Lopez C, Dutcus C, Guo M, Saito K, Kraljevic S, Tamai T, Ren M, Cheng AL. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4538] [Cited by in RCA: 4213] [Article Influence: 526.6] [Reference Citation Analysis (3)] |
| 9. | Bruix J, Qin S, Merle P, Granito A, Huang YH, Bodoky G, Pracht M, Yokosuka O, Rosmorduc O, Breder V, Gerolami R, Masi G, Ross PJ, Song T, Bronowicki JP, Ollivier-Hourmand I, Kudo M, Cheng AL, Llovet JM, Finn RS, LeBerre MA, Baumhauer A, Meinhardt G, Han G; RESORCE Investigators. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389:56-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3029] [Cited by in RCA: 2859] [Article Influence: 317.7] [Reference Citation Analysis (1)] |
| 10. | Abou-Alfa GK, Meyer T, Cheng AL, El-Khoueiry AB, Rimassa L, Ryoo BY, Cicin I, Merle P, Chen Y, Park JW, Blanc JF, Bolondi L, Klümpen HJ, Chan SL, Zagonel V, Pressiani T, Ryu MH, Venook AP, Hessel C, Borgman-Hagey AE, Schwab G, Kelley RK. Cabozantinib in Patients with Advanced and Progressing Hepatocellular Carcinoma. N Engl J Med. 2018;379:54-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1967] [Cited by in RCA: 1877] [Article Influence: 234.6] [Reference Citation Analysis (1)] |
| 11. | Zhu AX, Park JO, Ryoo BY, Yen CJ, Poon R, Pastorelli D, Blanc JF, Chung HC, Baron AD, Pfiffer TE, Okusaka T, Kubackova K, Trojan J, Sastre J, Chau I, Chang SC, Abada PB, Yang L, Schwartz JD, Kudo M; REACH Trial Investigators. Ramucirumab versus placebo as second-line treatment in patients with advanced hepatocellular carcinoma following first-line therapy with sorafenib (REACH): a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2015;16:859-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 709] [Cited by in RCA: 674] [Article Influence: 61.3] [Reference Citation Analysis (0)] |
| 12. | da Fonseca LG, Reig M, Bruix J. Tyrosine Kinase Inhibitors and Hepatocellular Carcinoma. Clin Liver Dis. 2020;24:719-737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 45] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 13. | Ma J, Zheng B, Goswami S, Meng L, Zhang D, Cao C, Li T, Zhu F, Ma L, Zhang Z, Zhang S, Duan M, Chen Q, Gao Q, Zhang X. PD1Hi CD8+ T cells correlate with exhausted signature and poor clinical outcome in hepatocellular carcinoma. J Immunother Cancer. 2019;7:331. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 283] [Cited by in RCA: 260] [Article Influence: 37.1] [Reference Citation Analysis (0)] |
| 14. | Makarova-Rusher OV, Medina-Echeverz J, Duffy AG, Greten TF. The yin and yang of evasion and immune activation in HCC. J Hepatol. 2015;62:1420-1429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 253] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 15. | Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A, Sarker D, Verset G, Chan SL, Knox J, Daniele B, Webber AL, Ebbinghaus SW, Ma J, Siegel AB, Cheng AL, Kudo M; KEYNOTE-224 investigators. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19:940-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2080] [Cited by in RCA: 1986] [Article Influence: 248.3] [Reference Citation Analysis (1)] |
| 16. | El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Dela Cruz C, Lang L, Neely J, Tang H, Dastani HB, Melero I. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389:2492-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3553] [Cited by in RCA: 3460] [Article Influence: 384.4] [Reference Citation Analysis (1)] |
| 17. | Yau T, Park JW, Finn RS, Cheng AL, Mathurin P, Edeline J, Kudo M, Harding JJ, Merle P, Rosmorduc O, Wyrwicz L, Schott E, Choo SP, Kelley RK, Sieghart W, Assenat E, Zaucha R, Furuse J, Abou-Alfa GK, El-Khoueiry AB, Melero I, Begic D, Chen G, Neely J, Wisniewski T, Tschaika M, Sangro B. Nivolumab versus sorafenib in advanced hepatocellular carcinoma (CheckMate 459): a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2022;23:77-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 980] [Cited by in RCA: 894] [Article Influence: 223.5] [Reference Citation Analysis (2)] |
| 18. | Finn RS, Ryoo BY, Merle P, Kudo M, Bouattour M, Lim HY, Breder V, Edeline J, Chao Y, Ogasawara S, Yau T, Garrido M, Chan SL, Knox J, Daniele B, Ebbinghaus SW, Chen E, Siegel AB, Zhu AX, Cheng AL; KEYNOTE-240 investigators. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J Clin Oncol. 2020;38:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1511] [Cited by in RCA: 1401] [Article Influence: 233.5] [Reference Citation Analysis (1)] |
| 19. | Hegde PS, Wallin JJ, Mancao C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin Cancer Biol. 2018;52:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 355] [Article Influence: 44.4] [Reference Citation Analysis (1)] |
| 20. | Cheng AL, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Lim HY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Ma N, Nicholas A, Wang Y, Li L, Zhu AX, Finn RS. Updated efficacy and safety data from IMbrave150: Atezolizumab plus bevacizumab vs. sorafenib for unresectable hepatocellular carcinoma. J Hepatol. 2022;76:862-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1370] [Cited by in RCA: 1275] [Article Influence: 318.8] [Reference Citation Analysis (3)] |
| 21. | Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL; IMbrave150 Investigators. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382:1894-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6170] [Cited by in RCA: 5560] [Article Influence: 926.7] [Reference Citation Analysis (2)] |
| 22. | Finn RS, Ikeda M, Zhu AX, Sung MW, Baron AD, Kudo M, Okusaka T, Kobayashi M, Kumada H, Kaneko S, Pracht M, Mamontov K, Meyer T, Kubota T, Dutcus CE, Saito K, Siegel AB, Dubrovsky L, Mody K, Llovet JM. Phase Ib Study of Lenvatinib Plus Pembrolizumab in Patients With Unresectable Hepatocellular Carcinoma. J Clin Oncol. 2020;38:2960-2970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 958] [Cited by in RCA: 939] [Article Influence: 156.5] [Reference Citation Analysis (1)] |
| 23. | El-Khoueiry AB, Kim RD, Harris WP, Sung MW, Galle PR. Phase Ib study of regorafenib (REG) plus pembrolizumab (PEMBRO) for first-line treatment of advanced hepatocellular carcinoma (HCC). J Clin Oncol. 2020;38:564-564. [DOI] [Full Text] |
| 24. | Yau T, Kang YK, Kim TY, El-Khoueiry AB, Santoro A, Sangro B, Melero I, Kudo M, Hou MM, Matilla A, Tovoli F, Knox JJ, Ruth He A, El-Rayes BF, Acosta-Rivera M, Lim HY, Neely J, Shen Y, Wisniewski T, Anderson J, Hsu C. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients With Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020;6:e204564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1069] [Cited by in RCA: 1072] [Article Influence: 178.7] [Reference Citation Analysis (0)] |
| 25. | Yau T, Zagonel V, Santoro A, Acosta-Rivera M, Choo SP, Matilla A, He AR, Gracián AC, El-Khoueiry AB, Sangro B, Eldawy T, Bruix J, Frassineti G, Vaccaro GM, Tschaika M, Scheffold C, Shen Y, Neely J, Piscaglia F. Nivolumab (NIVO) + ipilimumab (IPI) + cabozantinib (CABO) combination therapy in patients (pts) with advanced hepatocellular carcinoma (aHCC): Results from CheckMate 040. J Clin Oncol. 2020;38:478-478. [DOI] [Full Text] |
| 26. | AstraZeneca. Imfinzi plus tremelimumab significantly improved overall survival in HIMALAYA Phase III trial in 1st-line unresectable liver cancer. [cited January 2, 2022] Available from: https://www.astrazeneca.com/media-centre/press-releases/2021/imfinzi-and-tremelimumab-improved-os-in-liver-cancer.html. |
| 27. | Kelley RK, Sangro B, Harris W, Ikeda M, Okusaka T, Kang YK, Qin S, Tai DW, Lim HY, Yau T, Yong WP, Cheng AL, Gasbarrini A, Damian S, Bruix J, Borad M, Bendell J, Kim TY, Standifer N, He P, Makowsky M, Negro A, Kudo M, Abou-Alfa GK. Safety, Efficacy, and Pharmacodynamics of Tremelimumab Plus Durvalumab for Patients With Unresectable Hepatocellular Carcinoma: Randomized Expansion of a Phase I/II Study. J Clin Oncol. 2021;39:2991-3001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 369] [Article Influence: 73.8] [Reference Citation Analysis (1)] |
| 28. | Makary MS, Ramsell S, Miller E, Beal EW, Dowell JD. Hepatocellular carcinoma locoregional therapies: Outcomes and future horizons. World J Gastroenterol. 2021;27:7462-7479. [PubMed] [DOI] [Full Text] |
| 29. | Kong FH, Miao XY, Zou H, Xiong L, Wen Y, Chen B, Liu X, Zhou JJ. End-stage liver disease score and future liver remnant volume predict post-hepatectomy liver failure in hepatocellular carcinoma. World J Clin Cases. 2019;7:3734-3741. [PubMed] [DOI] [Full Text] |
| 30. | Bhoori S, Mazzaferro V. Current challenges in liver transplantation for hepatocellular carcinoma. Best Pract Res Clin Gastroenterol. 2014;28:867-879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 31. | Park JW, Chen M, Colombo M, Roberts LR, Schwartz M, Chen PJ, Kudo M, Johnson P, Wagner S, Orsini LS, Sherman M. Global patterns of hepatocellular carcinoma management from diagnosis to death: the BRIDGE Study. Liver Int. 2015;35:2155-2166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1057] [Cited by in RCA: 1039] [Article Influence: 94.5] [Reference Citation Analysis (6)] |
| 32. | Llovet JM, Kelley RK, Villanueva A, Singal AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J, Finn RS. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7:6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1323] [Reference Citation Analysis (0)] |
| 33. | Cho YK, Kim JK, Kim MY, Rhim H, Han JK. Systematic review of randomized trials for hepatocellular carcinoma treated with percutaneous ablation therapies. Hepatology. 2009;49:453-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 353] [Cited by in RCA: 323] [Article Influence: 19.0] [Reference Citation Analysis (2)] |
| 34. | Breen DJ, Lencioni R. Image-guided ablation of primary liver and renal tumours. Nat Rev Clin Oncol. 2015;12:175-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 35. | Kloeckner R, Galle PR, Bruix J. Local and Regional Therapies for Hepatocellular Carcinoma. Hepatology. 2021;73 Suppl 1:137-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 99] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 36. | Yao XS, Yan D, Jiang XX, Li X, Zeng HY, Li H. Short-term outcomes of radiofrequency ablation for hepatocellular carcinoma using cone-beam computed tomography for planning and image guidance. World J Clin Cases. 2021;9:1580-1591. [PubMed] [DOI] [Full Text] |
| 37. | Lee DH, Lee JM, Lee JY, Kim SH, Yoon JH, Kim YJ, Han JK, Choi BI. Radiofrequency ablation of hepatocellular carcinoma as first-line treatment: long-term results and prognostic factors in 162 patients with cirrhosis. Radiology. 2014;270:900-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 260] [Article Influence: 21.7] [Reference Citation Analysis (1)] |
| 38. | N'Kontchou G, Mahamoudi A, Aout M, Ganne-Carrié N, Grando V, Coderc E, Vicaut E, Trinchet JC, Sellier N, Beaugrand M, Seror O. Radiofrequency ablation of hepatocellular carcinoma: long-term results and prognostic factors in 235 Western patients with cirrhosis. Hepatology. 2009;50:1475-1483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 335] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 39. | Xia Y, Li J, Liu G, Wang K, Qian G, Lu Z, Yang T, Yan Z, Lei Z, Si A, Wan X, Zhang H, Gao C, Cheng Z, Pawlik TM, Wang H, Lau WY, Wu M, Shen F. Long-term Effects of Repeat Hepatectomy vs Percutaneous Radiofrequency Ablation Among Patients With Recurrent Hepatocellular Carcinoma: A Randomized Clinical Trial. JAMA Oncol. 2020;6:255-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 160] [Article Influence: 26.7] [Reference Citation Analysis (1)] |
| 40. | Glassberg MB, Ghosh S, Clymer JW, Qadeer RA, Ferko NC, Sadeghirad B, Wright GW, Amaral JF. Microwave ablation compared with radiofrequency ablation for treatment of hepatocellular carcinoma and liver metastases: a systematic review and meta-analysis. Onco Targets Ther. 2019;12:6407-6438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 84] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 41. | Tan W, Deng Q, Lin S, Wang Y, Xu G. Comparison of microwave ablation and radiofrequency ablation for hepatocellular carcinoma: a systematic review and meta-analysis. Int J Hyperthermia. 2019;36:264-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 84] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 42. | Harari CM, Magagna M, Bedoya M, Lee FT Jr, Lubner MG, Hinshaw JL, Ziemlewicz T, Brace CL. Microwave Ablation: Comparison of Simultaneous and Sequential Activation of Multiple Antennas in Liver Model Systems. Radiology. 2016;278:95-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 43. | Sutter O, Calvo J, N'Kontchou G, Nault JC, Ourabia R, Nahon P, Ganne-Carrié N, Bourcier V, Zentar N, Bouhafs F, Sellier N, Diallo A, Seror O. Safety and Efficacy of Irreversible Electroporation for the Treatment of Hepatocellular Carcinoma Not Amenable to Thermal Ablation Techniques: A Retrospective Single-Center Case Series. Radiology. 2017;284:877-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 113] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 44. | Wang C, Wang H, Yang W, Hu K, Xie H, Hu KQ, Bai W, Dong Z, Lu Y, Zeng Z, Lou M, Gao X, Chang X, An L, Qu J, Li J, Yang Y. Multicenter randomized controlled trial of percutaneous cryoablation versus radiofrequency ablation in hepatocellular carcinoma. Hepatology. 2015;61:1579-1590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 183] [Article Influence: 16.6] [Reference Citation Analysis (2)] |
| 45. | Dapito DH, Mencin A, Gwak GY, Pradere JP, Jang MK, Mederacke I, Caviglia JM, Khiabanian H, Adeyemi A, Bataller R, Lefkowitch JH, Bower M, Friedman R, Sartor RB, Rabadan R, Schwabe RF. Promotion of hepatocellular carcinoma by the intestinal microbiota and TLR4. Cancer Cell. 2012;21:504-516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1155] [Cited by in RCA: 1078] [Article Influence: 77.0] [Reference Citation Analysis (0)] |
| 46. | Portolani N, Coniglio A, Ghidoni S, Giovanelli M, Benetti A, Tiberio GA, Giulini SM. Early and late recurrence after liver resection for hepatocellular carcinoma: prognostic and therapeutic implications. Ann Surg. 2006;243:229-235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 752] [Cited by in RCA: 757] [Article Influence: 37.9] [Reference Citation Analysis (0)] |
| 47. | Okajima W, Komatsu S, Ichikawa D, Miyamae M, Ohashi T, Imamura T, Kiuchi J, Nishibeppu K, Arita T, Konishi H, Shiozaki A, Morimura R, Ikoma H, Okamoto K, Otsuji E. Liquid biopsy in patients with hepatocellular carcinoma: Circulating tumor cells and cell-free nucleic acids. World J Gastroenterol. 2017;23:5650-5668. [PubMed] [DOI] [Full Text] |
| 48. | Bruix J, Takayama T, Mazzaferro V, Chau GY, Yang J, Kudo M, Cai J, Poon RT, Han KH, Tak WY, Lee HC, Song T, Roayaie S, Bolondi L, Lee KS, Makuuchi M, Souza F, Berre MA, Meinhardt G, Llovet JM; STORM investigators. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015;16:1344-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 896] [Cited by in RCA: 833] [Article Influence: 75.7] [Reference Citation Analysis (3)] |
| 49. | Pu T, Li ZH, Jiang D, Chen JM, Guo Q, Cai M, Chen ZX, Xie K, Zhao YJ, Liu FB. Nomogram based on inflammation-related markers for predicting survival of patients undergoing hepatectomy for hepatocellular carcinoma. World J Clin Cases. 2021;9:11193-11207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 2] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 50. | Liu P, Tan XZ, Zhang T, Gu QB, Mao XH, Li YC, He YQ. Prediction of microvascular invasion in solitary hepatocellular carcinoma ≤ 5 cm based on computed tomography radiomics. World J Gastroenterol. 2021;27:2015-2024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 22] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 51. | Tian YL, Ji JJ, Chen LN, Cui XL, Liu ST, Mao L, Qiu YD, Li BB. Risk factors for long-term prognosis of hepatocellular carcinoma patients after anatomic hepatectomy. World J Clin Cases. 2020;8:713-722. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 52. | den Brok MH, Sutmuller RP, Nierkens S, Bennink EJ, Toonen LW, Figdor CG, Ruers TJ, Adema GJ. Synergy between in situ cryoablation and TLR9 stimulation results in a highly effective in vivo dendritic cell vaccine. Cancer Res. 2006;66:7285-7292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 146] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 53. | Duffy AG, Ulahannan SV, Makorova-Rusher O, Rahma O, Wedemeyer H, Pratt D, Davis JL, Hughes MS, Heller T, ElGindi M, Uppala A, Korangy F, Kleiner DE, Figg WD, Venzon D, Steinberg SM, Venkatesan AM, Krishnasamy V, Abi-Jaoudeh N, Levy E, Wood BJ, Greten TF. Tremelimumab in combination with ablation in patients with advanced hepatocellular carcinoma. J Hepatol. 2017;66:545-551. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 687] [Cited by in RCA: 668] [Article Influence: 74.2] [Reference Citation Analysis (2)] |
| 54. | Kudo M, Ueshima K, Nakahira S, Nishida N, Ida H, Minami Y, Nakai T, Wada H, Kubo S, Ohkawa K, Morishita A, Nomi T, Ishida K, Kobayashi S, Umeda M, Tsurusaki M, Chiba Y, Yoshimura K, Sakai K, Nishio K. Adjuvant nivolumab for hepatocellular carcinoma (HCC) after surgical resection (SR) or radiofrequency ablation (RFA) (NIVOLVE): A phase 2 prospective multicenter single-arm trial and exploratory biomarker analysis. J Clin Oncol. 2021;39:4070-4070. [DOI] [Full Text] |
| 55. | Salem R, Lewandowski RJ. Chemoembolization and radioembolization for hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2013;11:604-11; quiz e43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 56. | Llovet JM, Real MI, Montaña X, Planas R, Coll S, Aponte J, Ayuso C, Sala M, Muchart J, Solà R, Rodés J, Bruix J; Barcelona Liver Cancer Group. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359:1734-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2830] [Cited by in RCA: 2589] [Article Influence: 107.9] [Reference Citation Analysis (3)] |
| 57. | Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37:429-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2440] [Cited by in RCA: 2238] [Article Influence: 97.3] [Reference Citation Analysis (3)] |
| 58. | Burrel M, Reig M, Forner A, Barrufet M, de Lope CR, Tremosini S, Ayuso C, Llovet JM, Real MI, Bruix J. Survival of patients with hepatocellular carcinoma treated by transarterial chemoembolisation (TACE) using Drug Eluting Beads. Implications for clinical practice and trial design. J Hepatol. 2012;56:1330-1335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 394] [Cited by in RCA: 376] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 59. | Lammer J, Malagari K, Vogl T, Pilleul F, Denys A, Watkinson A, Pitton M, Sergent G, Pfammatter T, Terraz S, Benhamou Y, Avajon Y, Gruenberger T, Pomoni M, Langenberger H, Schuchmann M, Dumortier J, Mueller C, Chevallier P, Lencioni R; PRECISION V Investigators. Prospective randomized study of doxorubicin-eluting-bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Intervent Radiol. 2010;33:41-52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1269] [Cited by in RCA: 1182] [Article Influence: 73.9] [Reference Citation Analysis (2)] |
| 60. | Pipa-Muñiz M, Sanmartino S, Mesa A, Álvarez-Navascués C, González-Diéguez ML, Cadahía V, Rodríguez JE, Vega F, Rodríguez M, Costilla-García SM, Varela M. The development of early ascites is associated with shorter overall survival in patients with hepatocellular carcinoma treated with drug-eluting embolic chemoembolization. BMC Gastroenterol. 2020;20:166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 61. | Teyateeti A, Mahvash A, Long J, Abdelsalam M, Avritscher R, Kaseb A, Odisio B, Ravizzini G, Surasi D, Teyateeti A, Macapinlac H, Kappadath SC. Disease control and failure patterns of unresectable hepatocellular carcinoma following transarterial radioembolization with yttrium-90 microspheres and with/without sorafenib. World J Gastroenterol. 2021;27:8166-8181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 62. | Romano F, Chiarelli M, Garancini M, Scotti M, Zago M, Cioffi G, De Simone M, Cioffi U. Rethinking the Barcelona clinic liver cancer guidelines: Intermediate stage and Child-Pugh B patients are suitable for surgery? World J Gastroenterol. 2021;27:2784-2794. [PubMed] [DOI] [Full Text] |
| 63. | Inchingolo R, Acquafredda F, Tedeschi M, Laera L, Surico G, Surgo A, Fiorentino A, Spiliopoulos S, de'Angelis N, Memeo R. Worldwide management of hepatocellular carcinoma during the COVID-19 pandemic. World J Gastroenterol. 2021;27:3780-3789. [PubMed] [DOI] [Full Text] |
| 64. | Kadalayil L, Benini R, Pallan L, O'Beirne J, Marelli L, Yu D, Hackshaw A, Fox R, Johnson P, Burroughs AK, Palmer DH, Meyer T. A simple prognostic scoring system for patients receiving transarterial embolisation for hepatocellular cancer. Ann Oncol. 2013;24:2565-2570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 313] [Cited by in RCA: 301] [Article Influence: 23.2] [Reference Citation Analysis (1)] |
| 65. | Raoul JL, Forner A, Bolondi L, Cheung TT, Kloeckner R, de Baere T. Updated use of TACE for hepatocellular carcinoma treatment: How and when to use it based on clinical evidence. Cancer Treat Rev. 2019;72:28-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 529] [Cited by in RCA: 470] [Article Influence: 67.1] [Reference Citation Analysis (0)] |
| 66. | Sieghart W, Hucke F, Pinter M, Graziadei I, Vogel W, Müller C, Heinzl H, Trauner M, Peck-Radosavljevic M. The ART of decision making: retreatment with transarterial chemoembolization in patients with hepatocellular carcinoma. Hepatology. 2013;57:2261-2273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 275] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 67. | Salem R, Gordon AC, Mouli S, Hickey R, Kallini J, Gabr A, Mulcahy MF, Baker T, Abecassis M, Miller FH, Yaghmai V, Sato K, Desai K, Thornburg B, Benson AB, Rademaker A, Ganger D, Kulik L, Lewandowski RJ. Y90 Radioembolization Significantly Prolongs Time to Progression Compared With Chemoembolization in Patients With Hepatocellular Carcinoma. Gastroenterology. 2016;151:1155-1163.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 564] [Cited by in RCA: 511] [Article Influence: 51.1] [Reference Citation Analysis (2)] |
| 68. | Salem R, Johnson GE, Kim E, Riaz A, Bishay V, Boucher E, Fowers K, Lewandowski R, Padia SA. Yttrium-90 Radioembolization for the Treatment of Solitary, Unresectable HCC: The LEGACY Study. Hepatology. 2021;74:2342-2352. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 441] [Cited by in RCA: 395] [Article Influence: 79.0] [Reference Citation Analysis (0)] |
| 69. | Vilgrain V, Pereira H, Assenat E, Guiu B, Ilonca AD, Pageaux GP, Sibert A, Bouattour M, Lebtahi R, Allaham W, Barraud H, Laurent V, Mathias E, Bronowicki JP, Tasu JP, Perdrisot R, Silvain C, Gerolami R, Mundler O, Seitz JF, Vidal V, Aubé C, Oberti F, Couturier O, Brenot-Rossi I, Raoul JL, Sarran A, Costentin C, Itti E, Luciani A, Adam R, Lewin M, Samuel D, Ronot M, Dinut A, Castera L, Chatellier G; SARAH Trial Group. Efficacy and safety of selective internal radiotherapy with yttrium-90 resin microspheres compared with sorafenib in locally advanced and inoperable hepatocellular carcinoma (SARAH): an open-label randomised controlled phase 3 trial. Lancet Oncol. 2017;18:1624-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 682] [Cited by in RCA: 624] [Article Influence: 69.3] [Reference Citation Analysis (2)] |
| 70. | Ricke J, Klümpen HJ, Amthauer H, Bargellini I, Bartenstein P, de Toni EN, Gasbarrini A, Pech M, Peck-Radosavljevic M, Popovič P, Rosmorduc O, Schott E, Seidensticker M, Verslype C, Sangro B, Malfertheiner P. Impact of combined selective internal radiation therapy and sorafenib on survival in advanced hepatocellular carcinoma. J Hepatol. 2019;71:1164-1174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 279] [Article Influence: 39.9] [Reference Citation Analysis (1)] |
| 71. | Chow PKH, Gandhi M, Tan SB, Khin MW, Khasbazar A, Ong J, Choo SP, Cheow PC, Chotipanich C, Lim K, Lesmana LA, Manuaba TW, Yoong BK, Raj A, Law CS, Cua IHY, Lobo RR, Teh CSC, Kim YH, Jong YW, Han HS, Bae SH, Yoon HK, Lee RC, Hung CF, Peng CY, Liang PC, Bartlett A, Kok KYY, Thng CH, Low AS, Goh ASW, Tay KH, Lo RHG, Goh BKP, Ng DCE, Lekurwale G, Liew WM, Gebski V, Mak KSW, Soo KC; Asia-Pacific Hepatocellular Carcinoma Trials Group. SIRveNIB: Selective Internal Radiation Therapy Versus Sorafenib in Asia-Pacific Patients With Hepatocellular Carcinoma. J Clin Oncol. 2018;36:1913-1921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 544] [Cited by in RCA: 488] [Article Influence: 61.0] [Reference Citation Analysis (0)] |
| 72. | Kwak MS, Lee JH, Yoon JH, Yu SJ, Cho EJ, Jang ES, Kim YJ, Lee HS. Risk factors, clinical features, and prognosis of the hepatocellular carcinoma with peritoneal metastasis. Dig Dis Sci. 2012;57:813-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 73. | Wang B, Xu H, Gao ZQ, Ning HF, Sun YQ, Cao GW. Increased expression of vascular endothelial growth factor in hepatocellular carcinoma after transcatheter arterial chemoembolization. Acta Radiol. 2008;49:523-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 230] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 74. | Meyer T, Fox R, Ma YT, Ross PJ, James MW, Sturgess R, Stubbs C, Stocken DD, Wall L, Watkinson A, Hacking N, Evans TRJ, Collins P, Hubner RA, Cunningham D, Primrose JN, Johnson PJ, Palmer DH. Sorafenib in combination with transarterial chemoembolisation in patients with unresectable hepatocellular carcinoma (TACE 2): a randomised placebo-controlled, double-blind, phase 3 trial. Lancet Gastroenterol Hepatol. 2017;2:565-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 389] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 75. | Lencioni R, Llovet JM, Han G, Tak WY, Yang J, Guglielmi A, Paik SW, Reig M, Kim DY, Chau GY, Luca A, Del Arbol LR, Leberre MA, Niu W, Nicholson K, Meinhardt G, Bruix J. Sorafenib or placebo plus TACE with doxorubicin-eluting beads for intermediate stage HCC: The SPACE trial. J Hepatol. 2016;64:1090-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 572] [Cited by in RCA: 552] [Article Influence: 55.2] [Reference Citation Analysis (0)] |
| 76. | Kudo M, Han G, Finn RS, Poon RT, Blanc JF, Yan L, Yang J, Lu L, Tak WY, Yu X, Lee JH, Lin SM, Wu C, Tanwandee T, Shao G, Walters IB, Dela Cruz C, Poulart V, Wang JH. Brivanib as adjuvant therapy to transarterial chemoembolization in patients with hepatocellular carcinoma: A randomized phase III trial. Hepatology. 2014;60:1697-1707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 284] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 77. | Buijs M, Reyes DK, Pawlik TM, Blackford AL, Salem R, Messersmith WA, Weekes CD, Mulcahy M, Kamel IR, Geschwind JF. Phase 2 trial of concurrent bevacizumab and transhepatic arterial chemoembolization in patients with unresectable hepatocellular carcinoma. Cancer. 2013;119:1042-1049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 78. | Kudo M, Ueshima K, Ikeda M, Torimura T, Aikata H, Izumi N, Yamasaki T, Hino K, Kuzuya T, Isoda N, Yasui K, Aino H, Ido A, Kawabe N, Nakao K, Wada Y, Yoshimura K, Okusaka T, Furuse J, Arai Y. TACTICS: Final overall survival (OS) data from a randomized, open label, multicenter, phase II trial of transcatheter arterial chemoembolization (TACE) therapy in combination with sorafenib as compared with TACE alone in patients (pts) with hepatocellular carcinoma (HCC). J Clin Oncol. 2021;39:270-270. [DOI] [Full Text] |
| 79. | Kudo M, Ueshima K, Ikeda M, Torimura T, Tanabe N, Aikata H, Izumi N, Yamasaki T, Nojiri S, Hino K, Tsumura H, Kuzuya T, Isoda N, Yasui K, Aino H, Ido A, Kawabe N, Nakao K, Wada Y, Yokosuka O, Yoshimura K, Okusaka T, Furuse J, Kokudo N, Okita K, Johnson PJ, Arai Y; TACTICS study group. Randomised, multicentre prospective trial of transarterial chemoembolisation (TACE) plus sorafenib as compared with TACE alone in patients with hepatocellular carcinoma: TACTICS trial. Gut. 2020;69:1492-1501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 568] [Cited by in RCA: 554] [Article Influence: 92.3] [Reference Citation Analysis (0)] |
| 80. | Tampaki M, Ionas E, Hadziyannis E, Deutsch M, Malagari K, Koskinas J. Association of TIM-3 with BCLC Stage, Serum PD-L1 Detection, and Response to Transarterial Chemoembolization in Patients with Hepatocellular Carcinoma. Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 81. | Singh P, Toom S, Avula A, Kumar V, Rahma OE. The Immune Modulation Effect of Locoregional Therapies and Its Potential Synergy with Immunotherapy in Hepatocellular Carcinoma. J Hepatocell Carcinoma. 2020;7:11-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 82. | Pinato DJ, Murray SM, Forner A, Kaneko T, Fessas P, Toniutto P, Mínguez B, Cacciato V, Avellini C, Diaz A, Boyton RJ, Altmann DM, Goldin RD, Akarca AU, Marafioti T, Mauri FA, Casagrande E, Grillo F, Giannini E, Bhoori S, Mazzaferro V. Trans-arterial chemoembolization as a loco-regional inducer of immunogenic cell death in hepatocellular carcinoma: implications for immunotherapy. J Immunother Cancer. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 167] [Article Influence: 33.4] [Reference Citation Analysis (6)] |
| 83. | Craciun L, de Wind R, Demetter P, Lucidi V, Bohlok A, Michiels S, Bouazza F, Vouche M, Tancredi I, Verset G, Garaud S, Naveaux C, Galdon MG, Gallo KW, Hendlisz A, Derijckere ID, Flamen P, Larsimont D, Donckier V. Retrospective analysis of the immunogenic effects of intra-arterial locoregional therapies in hepatocellular carcinoma: a rationale for combining selective internal radiation therapy (SIRT) and immunotherapy. BMC Cancer. 2020;20:135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 84. | Chew V, Lee YH, Pan L, Nasir NJM, Lim CJ, Chua C, Lai L, Hazirah SN, Lim TKH, Goh BKP, Chung A, Lo RHG, Ng D, Filarca RLF, Albani S, Chow PKH. Immune activation underlies a sustained clinical response to Yttrium-90 radioembolisation in hepatocellular carcinoma. Gut. 2019;68:335-346. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 179] [Article Influence: 25.6] [Reference Citation Analysis (0)] |
| 85. | Tai D, Loke K, Gogna A, Kaya NA, Tan SH, Hennedige T, Ng D, Irani F, Lee J, Lim JQ, Too CW, Ng MCH, Tham CK, Lam J, Koo SL, Chong HS, Goh GB, Huang HL, Venkatanarasimha N, Lo R, Chow PKH, Goh BKP, Chung A, Toh HC, Thng CH, Lim TKH, Yeong J, Zhai W, Chan CY, Choo SP. Radioembolisation with Y90-resin microspheres followed by nivolumab for advanced hepatocellular carcinoma (CA 209-678): a single arm, single centre, phase 2 trial. Lancet Gastroenterol Hepatol. 2021;6:1025-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (1)] |
| 86. | Berberabe T. SIRT Administered With Nivolumab Demonstrates Positive Safety, Tolerability Results in HCC. Targeted Therapies Oncol. 2020;9:24. |
| 87. | Han Y, Zhi WH, Xu F, Zhang CB, Huang XQ, Luo JF. Selection of first-line systemic therapies for advanced hepatocellular carcinoma: A network meta-analysis of randomized controlled trials. World J Gastroenterol. 2021;27:2415-2433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 19] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 88. | Long HY, Huang TY, Xie XY, Long JT, Liu BX. Treatment strategies for hepatocellular carcinoma with extrahepatic metastasis. World J Clin Cases. 2021;9:5754-5768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 89. | Piñero F, da Fonseca LG. Trial eligibility in advanced hepatocellular carcinoma: Does it support clinical practice in underrepresented subgroups? World J Gastroenterol. 2021;27:3429-3439. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Noncommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: Brazil
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Abdalla AN, Saudi Arabia; Li S, China; Liu B, China S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Ma YJ