INTRODUCTION
Non-alcoholic liver disease (NAFLD) has become the leading cause of liver disease in western countries and has attracted researchers’ and clinicians’ interest in recent years. It affects 20%-30% of the adult population in developed countries and its prevalence rises to 95% among subjects with morbid obesity and 70% among those with type 2 diabetes mellitus (DM2)[1,2]. Close to two-thirds of NAFLD patients have some metabolic factor and one-third have metabolic syndrome (MetS). Although NAFLD has been proposed as the hepatic manifestation of MetS, in actual fact the relationship between these two conditions is complex and reciprocal, mediated by insulin resistance. NAFLD is known to be an independent risk factor for the development of DM2/prediabetes and cardiovascular mortality[2-4].
NAFLD is a dynamic disease that includes an evolving spectrum of anatomo-clinical lesions, including hepatic steatosis (fat deposition in hepatocytes, usually in the form of macrovacuoles), steatohepatitis (NASH; which adds cell death and inflammatory infiltrates) and cirrhosis. Although most patients have a mild form of the disease, up to 20%-30% may have NASH lesions and one-third may have progressive fibrosis[1,5].
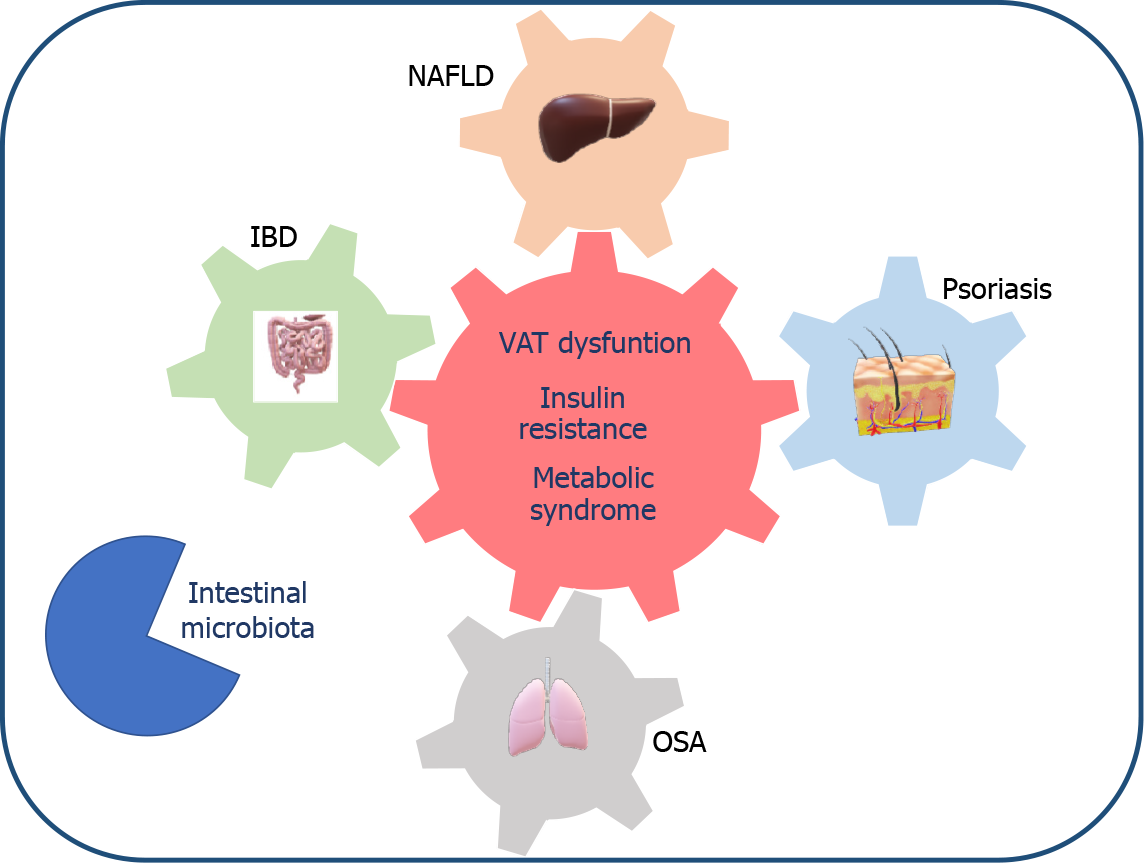
Excluding alcohol consumption has long been the key criterion to diagnosis of NAFLD[6,7]. However, its close association with metabolic factors has recently led some experts to propose replacing the term NAFLD with “MAFLD” (abbreviating for “metabolic dysfunction-associated fatty liver disease”), which would include patients with metabolic conditions, even if they consume alcohol and/or have other causes of fatty liver disease[8,9]. Likewise, “extra-hepatic manifestations” is probably not a suitable designation for the morbidities that coexist with NAFLD in metabolically unhealthy subjects, including inflammatory bowel disease (IBD), obstructive syndrome apnea (OSA) and psoriasis[7,10,11] (Figure 1).
Figure 1 Potential pathogenic factors contributing to the co-existence of non-alcoholic fatty liver disease and extrahepatic comorbidities (i.e. inflammatory bowel disease, obstructive syndrome apnea and psoriasis).
IBD: Inflammatory bowel disease; NAFLD: Non-alcoholic fatty liver disease; OSA: Obstructive syndrome apnea; VAT: Visceral adipose tissue.
The mechanisms that link NAFLD with these extrahepatic comorbidities are complex and multifactorial. It appears that the imbalance in favor of proinflammatory vs anti-inflammatory cytokines produced by the visceral adipose tissue (VAT) of patients with metabolic dysfunction not only plays a role in the onset of an insulin resistance state, hepatic lipotoxicity and NAFLD but also interacts synergistically with the local production of these same inflammatory mediators in the intestine or skin, as occurs in IBD and psoriasis, or is enhanced by the hypoxemia present in OSA[12-15]. Moreover, there is increasing evidence that changes in the intestinal microbiota and their metabolic interaction with the host also play a role in the multi-hit pathogenesis of NAFLD and related extra-hepatic diseases[12,16].
In this review, our aim was to update the available evidence on the link between NAFLD and three increasingly prevalent diseases: IBD, OSA and psoriasis. Ultimately, we aim to draw attention to the impact that NAFLD may have on the management and prognosis of patients with these extra-hepatic comorbidities in clinical practice.
NAFLD AND IBD
IBD is a complex and multifactorial gastrointestinal disease that usually presents in the form of acute outbreaks on a chronic immune-mediated inflammatory substrate. Although its main symptoms relate to the intestine, in both types of IBD — Crohn’s disease and ulcerative colitis — extraintestinal symptoms can appear in organs such as the skin, joints, eyes and liver[15]. Hepatobiliary manifestations have been reported in 3%-50% of patients with IBD and elevated transaminases in up to one-third of them, often being transient and attributable to the immunomodulatory drugs used in IBD. NAFLD is now considered the leading cause of liver disease among IBD patients[17,18].
Since the link between ulcers in the intestine and fatty hepatomegaly was first described in 1873[19], multiple cases, series and observational cohort studies have been published that analyze the prevalence of NAFLD in patients with IBD and the factors that may link the two diseases, both metabolic (e.g., DM2, obesity, arterial hypertension, dyslipemia, MetS) and relating to the IBD itself (e.g., type, duration, inflammatory activity, extension, drugs, intestinal surgery)[20-30].
Zou et al[31] conducted a systematic review and meta-analysis covering 19 observational studies published up to 2018, involving a total of 5620 patients with IBD. Although NAFLD prevalence varied significantly (8%-40%), in the most recent studies they found an increase in the frequency of this liver disease among IBD patients, to the point that it now exceeds the level in the general population (33% vs 25%, respectively). Furthermore, the authors identify age, metabolic factors, methotrexate use, previous bowel surgery, and chronic kidney disease as risk factors for NAFLD. More recent publications confirm most of these data and attribute them to the changed metabolic profile of IBD patients in recent years. Thanks to new biologic drugs, these patients have gone from being malnourished to being obesity/overweight in up to a third of cases[27,32,33].
The liver-intestine interaction in IBD and NAFLD patients could be explained by mechanisms such as the synergism between inflammatory mediators produced by hypertrophic adipocytes in the VAT and the increase of proinflammatory cytokines from the intestine[12,15]. It could also be explained by the involvement of intestinal microbiota, including changes in its diversity, interaction of metabolites produced by intestinal microorganisms with the host’s lipogenesis and host hydrocarbon metabolism, or changes to intestinal permeability. All these mechanisms could favor the development of insulin resistance, MetS and NAFLD[34]. Animal experiments with drug delivery system (DDS)-induced colitis support this interrelationship between intestinal barrier disruption, endotoxemia, metabolic dysfunction in adipose tissue and NAFLD[35].
It has recently been recognized that patients with IBD, mainly those with Crohn’s disease, have a specific type of mesenteric adipose tissue, located in the areas of inflamed bowel, called “creeping fat”. This is an immunologically active tissue that behaves similarly to VAT, promoting inflammation of the intestinal mucosa and perhaps playing a role in the metabolic changes involved in the onset of NAFLD[36,37].
There are some practical issues of particular interest to IBD gastroenterologists, such as: (1) Identifying IBD patients with NAFLD risk; (2) The impact of a NAFLD diagnosis on the treatment and prognosis of patients with IBD; and (3) When to refer the patient with IBD to a hepatologist.
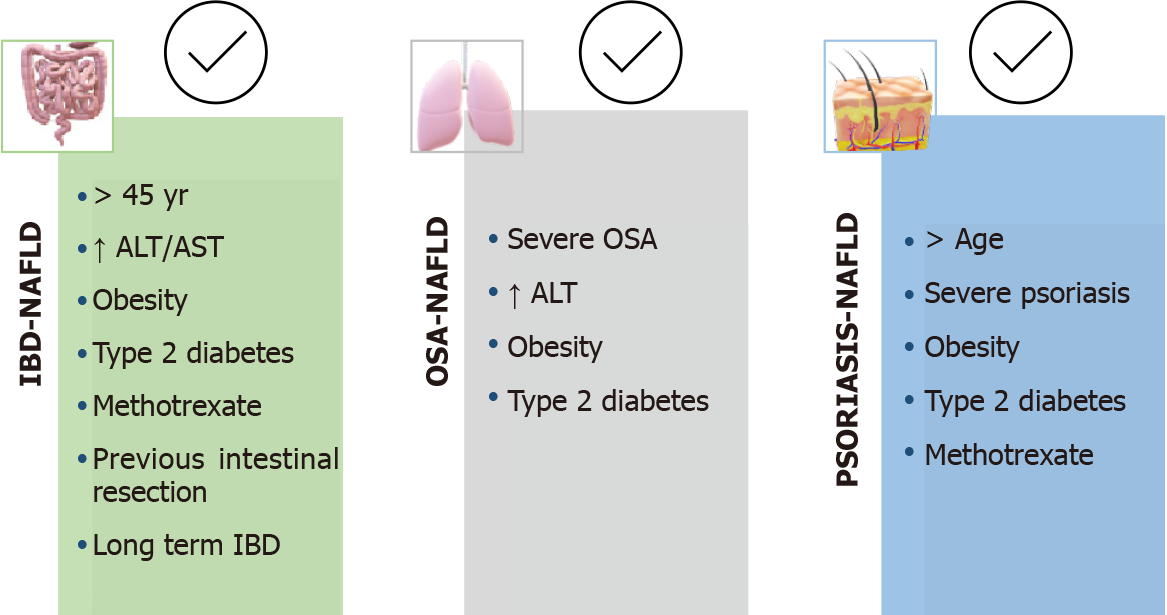
NAFLD should generally be suspected in older IBD patients with metabolic conditions, previous bowel surgery, or long-standing bowel disease[31,36]. However, comparing NAFLD patients with and without IBD, it appears that patients with NAFLD and IBD are younger and have less metabolic factors than those NAFLD patients without bowel disease. This is why some authors have proposed defining two phenotypes of patients with IBD and NAFLD: “classic or metabolic” and “IBD-specific”. The first group included subjects > 45-years-old with elevated transaminases, obesity, DM2 or arterial hypertension, and a later onset of bowel disease, while the second group included younger individuals with normal transaminases and less metabolic factors[23,25] (Figure 2).
Figure 2 Non-alcoholic fatty liver disease and extrahepatic comorbidities (i.e. inflammatory bowel disease, obstructive syndrome apnea and psoriasis) risk phenotypes.
ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; IBD: Inflammatory bowel disease; NAFLD: Non-alcoholic fatty liver disease; OSA: Obstructive syndrome apnea.
We do not have prospective studies analyzing whether there are differences in natural history and prognosis between these two phenotypes. Sartini et al[25] have associated NAFLD severity, measured by the degree of ultrasound steatosis, with fewer metabolic conditions and a “severe” IBD phenotype (i.e. more than one annual inflammatory bowel flare, more extensive IBD and previous intestinal surgery). These data suggest that in addition to metabolic conditions, other IBD-specific factors are likely to be involved in the onset and progression of NAFLD.
Based on animals and in vitro experimental studies, it has been speculated that tumor necrosis factor-alpha inhibitors may protect against developing NAFLD and also that glucocorticoids and immunomodulators increase the risk of liver disease progression[38,39]. However, few clinical studies have adequately collected the time and dose of treatments and thus conclusive information is not available at present. A synergistic effect of NAFLD and methotrexate treatment has been suggested, favoring liver toxicity and progression of NAFLD to more severe forms, especially in patients with obesity or DM2[40,41]. Lapumnuaypol et al[27] found a very high prevalence of NAFLD (54%) in their series of 80 patients with IBD treated only with biologics (i.e. infliximab, adalimumab, certolizumab or goligumab), most of whom were male and obese. In the multivariate analysis, they found an association between NAFLD and the clinical activity of IBD but not with drugs.
Considering that liver fibrosis is the main prognostic marker in NAFLD patients, it is important for gastroenterologists to use non-invasive markers of fibrosis in patients with IBD and NAFLD. These markers consist of mathematical algorithms that include clinical and analytical variables whose result enables the identification and stratification of patients with liver fibrosis. Among them, FIB-4 and NFS (non-alcoholic fatty liver disease score) have been validated in patients with NAFLD[4,6,7]. Retrospective and longitudinal studies that include the calculation of both serological markers of fibrosis found that only 2.2% of subjects with IBD and NAFLD have advanced fibrosis, and that it remains stable during a 3-5 year follow-up in most patients. Age, metabolic factors and duration of IBD appear to increase the risk of fibrosis progression[26,42,43].
Palumbo et al[42] were the authors of the only prospective study designed as part of a screening program for NAFLD and fibrosis involving 384 patients with IBD using control attenuation parameter (CAP) and hepatic elastography available on the FibroScan device probe (Echosens, France). They found any grade NAFLD in 32.8% of patients (CAP ≥ 248 dB/m), severe NAFLD (CAP > 300 dB/m), significant fibrosis in 24.6% (> 7 kPa) and advanced fibrosis in 18% (> 8.7 kPa). These NAFLD prevalence data should be taken with caution as there is evidence that higher CAP cutoffs than those used by these authors improve the diagnostic accuracy of this method[44,45]. The authors compared the presence of significant and advanced fibrosis in patients with NAFLD vs non-NAFLD and found a higher prevalence in the NAFLD group (24.6% vs 6.2% and 18.3% vs 3.1%, respectively; P < 0.001). Age, obesity, plasma triglycerides and methotrexate use were factors associated with liver fibrosis. In this way, they stratified the patients and referred a third of them to the hepatologist. In addition, they highlighted the presence of chronic kidney disease and cardiovascular disease among patients with IBD and NAFLD, in many cases related to subclinical atherosclerosis, information which supports the multidisciplinary approach to at-risk patients.
Although we now have scientific evidence to establish the suspicion of NAFLD in patients with IBD, prospective, longitudinal and control group studies are needed to identify those phenotypes at risk of developing fibrosis and advanced liver disease before we can establish an appropriate algorithm for screening, diagnosis and follow-up of patients with IBD and NAFLD.
NAFLD AND OSA
OSA is a clinical condition usually considered a respiratory disease, which has begun to be recognized as a multisystemic disease in the last two decades. It consists of recurrent nocturnal episodes of complete (apnea) and incomplete (hypopnea) obstruction of the upper airway, leading to hypoxemia and reoxygenation phenomena. It affects 1%-4% of the population, mostly males, with obesity and/or MetS, usually as a consequence of increased fat deposition in the upper airways and surrounding soft tissues[46,47]. Although, from a clinical perspective, OSA patients fundamentally consult due to sleep fragmentation and daytime sleepiness, the most important consequence is the chronic intermittent ischemia that occurs in different organs, such as the liver[46-48].
Over the last decade, at least 20 clinical studies have been published linking NAFLD and OSA, including systematic reviews and meta-analyses[49]. Although most include short series of patients and histological information is usually obtained from liver biopsies during morbid obesity surgery, OSA is considered to be an independent risk factor for the development and progression of NAFLD. In a recent meta-analysis and systematic review, Jin et al[50] found that patients with severe OSA (number of apnea-hypopnea episodes > 30/h) have higher aspartate aminotransferase values and a greater degree of steatosis, inflammation, ballooning degeneration and fibrosis. Trzepizur et al[51] found in their cohort of almost 1300 patients that those with severe OSA had a 2.5-fold increased risk of liver fibrosis; although, this association was not independent of other factors when logistic regression analysis was performed. When determining the epidemiological and pathogenic relationship between NAFLD and OSA, it is difficult to avoid the impact of obesity and other metabolic conditions that are so frequent in both diseases. Evidence from experimental models shows that chronic intermittent ischemia and increased sympathetic tone triggered by nocturnal hypoxemia phenomena are the main causes of cardiometabolic manifestations linked to OSA (i.e. DM2, dyslipemia, arterial hypertension, atherosclerosis) and of the onset and progression of NAFLD lesions[13,46].
The decrease in oxygen tension that occurs during nocturnal apnea-hypopnea episodes primarily affects hepatocytes in zone 3 of the hepatic lobule, where NAFLD lesions predominate, and results in the release of ischemia-induced factors (commonly known as HIF)[52,53]. These HIF favor the expression of genes involved in lipogenesis, with the consequent excess of triglycerides in the hepatocytes (steatosis), free fatty acids and hepatokines. This state of liver lipotoxicity leads to inflammation, mitochondrial dysfunction, oxidative stress, lipoperoxidation, cell damage (steatohepatitis) and fibrosis. In addition to this direct hepatic mechanism, VAT ischemia and sympathetic nervous system stimulation secondary to chronic intermittent ischemia promote a state of insulin resistance, lipolysis and hepatic fatty acid overload, which contribute to the onset of DM2, atherogenic dyslipemia, arterial hypertension and NAFLD[46,51,54]. Finally, different authors have proposed that nocturnal hypoxemia may alter the integrity of the intestinal barrier and favor the hit attributed to dysbiosis and bacterial translocation in the pathogenesis of NAFLD[55,56].
At present, we do not have prospective studies that allow us to determine the prevalence of NAFLD in patients with OSA or vice versa. However, considering that chronic intermittent ischemia can promote and aggravate liver damage, some authors propose screening for OSA in patients with NAFLD[47,49]. Taking into account the available information, hepatologists should always ask NAFLD patients about OSA-related symptoms and consider referring to the pneumologist those with elevated transaminases and/or advanced liver lesions, and/or associated metabolic factors, especially DM2 and obesity (Figure 2).
The use of a continuous positive airway pressure (CPAP) device and lifestyle changes to diet and exercise, especially in obesity/overweight patients, comprise the standard treatment of OSA patients. CPAP increases pharyngeal intraluminal pneumatic pressure, prevents hypoxemic events related to airway collapse, improves quality of life and decreases cardiovascular mortality[57,58]. However, its impact on metabolic factors and NAFLD lesions is controversial. The available evidence in this regard is of low quality because it is based on observational uncontrolled studies in patients with OSA-NAFLD and had a follow-up of only 3 mo of CPAP treatment[59]. Prospective, controlled and randomized studies that assess patient adherence to CPAP for more than 12 mo, could establish whether this measure can improve or prevent the progression of NAFLD.
NAFLD AND PSORIASIS
Psoriasis is one of the diseases that dermatologists are most concerned about because of its prevalence (it affects 2%-3% of the adult population in developed countries), the gaps in our knowledge of its pathogenesis, and its relationship with extracutaneous pathologies[14]. Currently, psoriasis is considered a systemic immune-mediated chronic inflammatory disease, since psoriasis patients not only present skin lesions but also frequently have comorbidities that can condition their prognosis and treatment, such as MetS and its hepatic manifestation, NAFLD[60-63].
There is epidemiological evidence and pathogenic hypotheses linking psoriasis to NAFLD[64,65]. In the last 10 years, multiple controlled cross-sectional observational studies have been published, some in large populations, studying the prevalence of NAFLD in subjects with psoriasis and the specific characteristics of the subpopulation of patients with psoriasis and NAFLD[66-69]. Van der Voort et al[66] analyzed these data in a Dutch cohort of 2292 individuals > 55 years of age and found that the prevalence of NAFLD among the 118 psoriasis patients was significantly higher than among the 2174 healthy controls (46.2% vs 33.3%; P < 0.005). Furthermore, after adjusting for confounders, including MetS, they determined that elderly patients with psoriasis are 70% more likely to have NAFLD than those without psoriasis. Gisondi et al[67] found that 44% of the 124 psoriasis patients included in their study had NAFLD vs 26% of the 79 healthy controls (P < 0.001). Comparing patients with psoriasis and NAFLD vs no-NAFLD, they found that those included in the first group were more frequently male and had a higher body mass index, transaminase values (Alanine aminotransferase and aspartate aminotransferase), and psoriasis severity [measured according to Psoriasis Area Severity Index (PASI) score. It combines the assessment of the severity of the lesion and affected area into a single figure between the values of 0-no disease to 72-maximum disease].
In 2015, Candia et al[68] published the first meta-analysis and systematic review of seven case-control studies evaluating the psoriasis-NAFLD association in populations from different continents (n = 267761). Their combined analysis indicated that psoriasis patients have twice the risk of developing NAFLD [odds ratio (OR): 2.15] and identified male sex (OR: 2.28), obesity (OR: 12.25), DM2 (OR: 2.63), arterial hypertension (OR: 2.7), MetS (OR: 9.03) and arthritis (OR: 2.25) as predisposing factors. They also found that patients with moderate to severe psoriasis had a 2-fold increased risk of NAFLD. These data have recently been confirmed in an updated systematic review and adjusted meta-analysis (n = 3019308 subjects)[69].
Approximately one-third of psoriasis patients have joint involvement and this has been linked to increased inflammatory burden, severity of skin lesions and risk of NAFLD[14,70]. However, not all authors found an association between arthropathy and liver disease[71]. The systematic review and meta-analysis by Candia et al[68] found that patients with psoriatic arthritis had double the risk of NAFLD when compared to those without arthropathy (OR: 2.25, 95%CI: 1.4-3.7; P < 0.05). Although this information would suggest considering patients with psoriasis and joint involvement as a special risk group for NAFLD, a recent meta-analysis raises doubts on the increased risk for NAFLD of such an association[69].
Although most studies found an independent link between psoriasis and NAFLD, those with metabolic factors appear to be particularly at risk[61,62]. The high prevalence of obesity (20%-40%), DM2 (12%) or MetS (20%-50%) among psoriasis patients, and the close relationship between these conditions and insulin resistance and NAFLD, means the same patient having both diseases cannot be seen as totally independent[72-74]. This clinical link has a common pathogenic substrate, the protagonist of which is the imbalance between overproduction of proinflammatory cytokines [e.g., tumor necrosis factor-alpha, interleukin (IL)-1, IL-6, IL-17] and the decrease of anti-inflammatory ones, mainly adiponectin. These mediators act locally by promoting cell growth and differentiation of the epidermis and dermis, the proliferation of keratinocytes and the consequent appearance of psoriatic plaques[63,64]. However, they may also act systemically and contribute to insulin resistance and other inflammatory mediators implicated in the metabolic dysfunction involved in NAFLD[63,65]. Interestingly, it is speculated that modifications in the skin microbiota present in psoriasis and their relationship with gut microbes may play a role in the psoriasis-NAFLD association[75]. These pathogenic hypotheses may explain why patients with psoriasis and NAFLD have more advanced forms of liver disease and severe skin lesions.
Most studies have assessed NAFLD using ultrasound, while few have involved liver biopsy. Roberts et al[76] found NAFLD in half of psoriasis patients (n = 103) and NASH histological lesions in 22% (liver biopsy in 52/103), one-third of them with significant advanced fibrosis. Furthermore, they linked NASH lesions with higher PASI scores, obesity and higher transaminase levels. More authors have analyzed the severity of liver lesions in psoriasis using non-invasive markers of fibrosis, serological or technological (FibroScan®)[67,77,78]. Using these scores it was found that 7%-8% of psoriasis patients have advanced liver fibrosis. This risk is multiplied by 4 when NAFLD is associated with this skin disease. Although fibrosis is more frequent in older subjects with metabolic factors, psoriasis appears to be an independent risk factor for fibrosis when adjusting in the multivariate logistic regression analysis, including metabolic conditions and drugs used to treat psoriasis[78].
From a practical perspective, the psoriasis-NAFLD association not only has an impact on the severity of skin and liver lesions, but may also influence dermatologists’ decisions when selecting systemic treatment for psoriasis[62,79].
Although some studies associate methotrexate liver toxicity with cumulative dose and treatment time, most find that the risk of liver damage is primarily related to the presence of risk conditions such as obesity, DM2, alcohol consumption, and NAFLD[80,81]. Cyclosporine and acitretin, a retinoid derived from vitamin A, are considered to be diabetogenic and to promote atherogenic dyslipemia and arterial hypertension[79,82].
It has been speculated that new biologic drugs could be beneficial in patients with psoriasis and NAFLD by acting on the proinflammatory cytokines involved in both diseases[63]. Although controlled studies with some tumor necrosis factor-alpha inhibitors have demonstrated their ability to decrease insulin resistance and the risk of DM2 (etanercept), others appear to favor weight gain and dyslipemia (infliximab, adalimumab)[79]. Recent research studies in animals have shown that IL-17 is involved in the progression of hepatic steatosis to NASH. There is speculation that the use of anti-IL17 monoclonal antibodies (secukinumab) could be beneficial in patients with psoriasis and NAFLD[83,84].
Considering that being overweight or obesity significantly increases psoriasis risk and severity, recent clinical trials have shown that weight loss, both with a hypocaloric diet and physical exercise or with bariatric surgery, improves psoriasis activity and also favors the response to systemic treatment. These interventions could be of particular importance in patients with psoriasis and risk of NAFLD[61,74].
Aware of the impact of NAFLD on psoriasis patients, experts from the European and American academies of dermatology have recently published recommendations for the management of psoriasis comorbidities, including MetS and NAFLD[82,85]. According to the available evidence, they consider NAFLD screening to be indicated in patients with a risk phenotype (i.e. moderate-severe psoriasis and metabolic factors) by means of transaminase measures and ultrasound, and propose a monitoring and follow-up algorithm that includes evaluation by the hepatologist in patients with suspected liver disease (Figure 2). In addition, they recommend that a diagnosis of NAFLD be taken into account when selecting psoriasis treatment. However, these recommendations do not appear to be implemented universally in dermatologists’ clinical practice, nor has a specific hepatologist referral protocol for patients with psoriasis and NAFLD been established in most hospitals.
CONCLUSION
NAFLD should be considered more than a liver disease and be taken into account not only by hepatologists but also by clinicians caring for patients with other related diseases, such as IBD, OSA and psoriasis. The scientific evidence shows that these comorbidities share an inflammatory background that synergistically impacts the severity and management of these patients. It is essential that screening and referral algorithms for NAFLD subjects are developed from a multidisciplinary perspective in which not only liver, intestinal, respiratory or skin lesions are analyzed but also the risk of morbidity and mortality from metabolic and cardiovascular causes.
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Sociedad Española De Patologia Digestiva.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ferraioli G, Grefhorst A S-Editor: Wang JJ L-Editor: A P-Editor: Yuan YY