Published online May 21, 2020. doi: 10.3748/wjg.v26.i19.2416
Peer-review started: January 18, 2020
First decision: February 28, 2020
Revised: April 26, 2020
Accepted: April 29, 2020
Article in press: April 29, 2020
Published online: May 21, 2020
Processing time: 124 Days and 0.9 Hours
Gamma-glutamyl transferase (GGT) is associated with the risk of cardiovascular disease (CVD) in the general population.
To identify the association of baseline GGT level and QRISK2 score among patients with biopsy-proven nonalcoholic fatty liver disease (NAFLD).
This was a retrospective study involving 1535 biopsy-proven NAFLD patients from 10 Asian centers in 8 countries using data collected by the Gut and Obesity in Asia (referred to as “GO ASIA”) workgroup. All patients with available baseline GGT levels and all 16 variables for the QRISK2 calculation (QRISK2-2017; developed by researchers at the United Kingdom National Health Service; https://qrisk.org/2017/; 10-year cardiovascular risk estimation) were included and compared to healthy controls with the same age, sex, and ethnicity. Relative risk was reported. QRISK2 score > 10% was defined as the high-CVD-risk group. Fibrosis stages 3 and 4 (F3 and F4) were considered advanced fibrosis.
A total of 1122 patients (73%) had complete data and were included in the final analysis; 314 (28%) had advanced fibrosis. The median age (interquartile range [IQR]) of the study population was 53 (44-60) years, 532 (47.4%) were females, and 492 (43.9%) were of Chinese ethnicity. The median 10-year CVD risk (IQR) was 5.9% (2.6-10.9), and the median relative risk of CVD over 10 years (IQR) was 1.65 (1.13-2.2) compared to healthy individuals with the same age, sex, and ethnicity. The high-CVD-risk group was significantly older than the low-risk group (median [IQR]: 63 [59-67] vs 49 [41-55] years; P < 0.001). Higher fibrosis stages in biopsy-proven NAFLD patients brought a significantly higher CVD risk (P < 0.001). Median GGT level was not different between the two groups (GGT [U/L]: Median [IQR], high risk 60 [37-113] vs low risk 66 [38-103], P = 0.56). There was no correlation between baseline GGT level and 10-year CVD risk based on the QRISK2 score (r = 0.02).
The CVD risk of NAFLD patients is higher than that of healthy individuals. Baseline GGT level cannot predict CVD risk in NAFLD patients. However, advanced fibrosis is a predictor of a high CVD risk.
Core tip: The median 10-year cardiovascular disease risk of nonalcoholic fatty liver disease (NAFLD) patients according to QRISK2 score was higher than that of healthy individuals of the same age, sex, and ethnicity. Apart from traditional risk factors, advanced fibrosis is a predictor of high-risk cardiovascular disease in NAFLD patients. However, the baseline gamma-glutamyl transferase level and high gamma-glutamyl transferase quartile cannot be used to predict cardiovascular risk in patients with established NAFLD.
- Citation: Pitisuttithum P, Chan WK, Goh GBB, Fan JG, Song MJ, Charatcharoenwitthaya P, Duseja A, Dan YY, Imajo K, Nakajima A, Ho KY, Goh KL, Wong VWS, Treeprasertsuk S. Gamma-glutamyl transferase and cardiovascular risk in nonalcoholic fatty liver disease: The Gut and Obesity Asia initiative. World J Gastroenterol 2020; 26(19): 2416-2426
- URL: https://www.wjgnet.com/1007-9327/full/v26/i19/2416.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i19.2416
Core tip: The median 10-year cardiovascular disease risk of nonalcoholic fatty liver disease (NAFLD) patients according to QRISK2 score was higher than that of healthy individuals of the same age, sex, and ethnicity. Apart from traditional risk factors, advanced fibrosis is a predictor of high-risk cardiovascular disease in NAFLD patients. However, the baseline gamma-glutamyl transferase level and high gamma-glutamyl transferase quartile cannot be used to predict cardiovascular risk in patients with established NAFLD.
Nonalcoholic fatty liver disease (NAFLD) is increasing in prevalence worldwide and has become a major chronic liver disease[1]. Growing clinical evidence shows that NAFLD is not confined to the liver but is also associated with extrahepatic diseases, and the majority of deaths among patients with NAFLD are from cardiovascular disease (CVD)[2]. Most studies have shown that NAFLD is an independent risk factor for CVD after adjustment for other common risk factors[3,4].
The potential pathogenesis of CVD in NAFLD patients is chronic activation of the inflammatory pathway[5]. One of the mechanisms is the alteration in the production, modification, and clearance of circulating lipoproteins in NAFLD, which play a major role in the atherosclerotic process. Previous studies have shown that nonalcoholic steatohepatitis patients have increased in small dense, low-density lipoprotein (LDL) 3, and LDL4 levels, which are related to atherogenesis. Levels of oxidized LDL level, a circulating product of lipid peroxidation, also increase and trigger inflammatory responses[6-8].
Currently, there is no consensus statement related to the evaluation of CVD risk in patients with NAFLD. In the general population, according to recent guidelines on the primary prevention of CVD, adults who are 40-75 years of age and are being evaluated for CVD prevention should undergo race- and sex-specific 10-year atherosclerotic CVD risk estimation that aids in guiding preventive intervention[9]. QRISK2, for estimating the 10-year risk of CVD, is now used across England’s health service (NHS England) with guidance from the National Institute of Health and Care Excellence and NHS Health Check. In addition, QRISK2 has a variety of ethnic specifications in its risk calculators, including for Asians[10-12].
Apart from the risk prediction model, gamma-glutamyl transferase (GGT) is a potential serum marker that has shown promise for the prediction of CVD in the general population. GGT is a membrane-bound enzyme involved in maintaining the metabolism of glutathione, the most important cellular antioxidant in humans. GGT functions by degrading glutathione, as a result, GGT elevation is an indirect measurement of oxidative stress[13-15] and antioxidant inadequacy[16]. A recent large observational study in Korea showed an independent association and dose-response relationship between serum GGT level and atherosclerotic CVD risk[17]. A meta-analysis in 2019 included patients with coronary artery disease and suggested that elevated serum GGT levels were an independent predictor of CVD[18]. A recent study in the general population showed that the highest risk of all-cause mortality and cardiovascular event was in the highest baseline GGT quartile group regardless of the degree of fatty liver[19].
In this study, we identified the association of baseline GGT level and QRISK2-2017, and determined the 10-year cardiovascular risk estimation among patients with biopsy-proven NAFLD using data collected by the Gut and Obesity in Asia (GO ASIA) workgroup.
This is a cross-sectional study involving 1535 biopsy-proven NAFLD patients from 10 Asian centers in 8 countries in the GO ASIA workgroup. The GO ASIA workgroup was formed in November 2014 with the aim of studying the relationship between obesity and gastrointestinal and liver diseases in the Asia-Pacific region through multinational collaborative studies[20]. A database consisting of biopsy-proven NAFLD patients was formed through contributions from participating members of the GO ASIA workgroup.
The centers that participated in this study were as follows: Chulalongkorn University, Thailand; University of Malaya, Malaysia; Singapore General Hospital, Singapore; Shanghai Jiaotong University School of Medicine, China; The Catholic University Korea, Korea; Mahidol University, Thailand; Post Graduate Institute of Medical Education and Research, India; National University of Singapore, Singapore; Yokohama City University Graduate School of Medicine, Japan; and The Chinese University of Hong Kong, Hong Kong, China. Ethics approval was obtained from each center prior to the commencement of the study.
NAFLD was diagnosed based on ultrasonography findings of fatty liver with exclusion of viral hepatitis B and C infection, significant alcohol intake, and the use of medications that can cause hepatic steatosis. Persistent elevated serum aminotransferase levels were the main indication for liver biopsy in order to rule out other possible etiologies. Only patients with biopsy confirmed NAFLD were included. Patients with other causes of chronic liver disease, incomplete histological data, and without significant hepatic steatosis were excluded. Only patients aged 25-84 years were included, according to the age range for which QRISK2-2017 has been validated[10].
Demographic, anthropometric, clinical, and laboratory data were collected using a standard protocol. A patient was considered to have diabetes mellitus if there was a self-reported history of diabetes mellitus or if fasting blood sugar was ≥ 7.0 mmol/L. A patient was considered to have dyslipidemia if there was a self-reported history of dyslipidemia, if the serum total cholesterol (TC) was ≥ 5.2 mmol/L, if the serum triglycerides were ≥ 1.7 mmol/L, if the serum high-density lipoprotein (HDL) was < 1.0 mmol/L for men or < 1.3 mmol/L for women, or if the serum LDL was ≥ 3.4 mmol/L. All laboratory parameters included in the study were measured within 3 mo of liver biopsy. Hypertension was based on self-reported history. If patients were using any drugs for diabetes mellitus, dyslipidemia or hypertension, they were also considered to the corresponding metabolic disorder.
Histopathological findings were reported according to the Non-Alcoholic Steatohepatitis Clinical Research Network Scoring System. The NAFLD Activity Score (NAS) is defined as the unweighted sum of the scores for steatosis, which is given for the presence of significant hepatic steatosis (0-3), lobular inflammation (0-3), and ballooning (0-2), for a total score ranging from 0 to 8. Nonalcoholic steatohepatitis is defined as steatosis with hepatocyte ballooning and inflammation with or without fibrosis[21]. The slides were reviewed by a pathologist at each center. Fibrosis stages 3 and 4 were considered advanced fibrosis (F3-4).
Liver stiffness (LS) was measured by transient elastography (TE) and was expressed in kPa. In this study, only LS values with at least 10 validated measurements and an interquartile range (IQR) to median value ratio (IQR/M) of < 30 were considered reliable.
All patients with available baseline GGT levels and all 16 variables used for QRISK2 calculation (The QRISK®2/2017; developed by researchers at the UK National Health Service; https://qrisk.org/2017/) were included. Absolute 10-year cardiovascular risk was calculated and compared to that of the healthy controls with the same age, sex, and ethnicity using QRISK2 calculator which was well validated from QRESEARCH database[10-12]. The comparison was reported as relative risk[22]. A QRISK2 score of ≥ 10% was defined as the high-CVD-risk group[10]. Any missing data for the QRISK calculation were inputted as missing in the calculator tool.
Normality of continuous variables was verified with the Kolmogorov–Smirnov test. Nonnormally distributed data were reported as the median and IQR and were analyzed by the Mann-Whitney U test. Categorical variables were reported as percentages and were analyzed using the chi-square test. Factors associated with high CVD were identified using binary logistic regression analysis. A two-tailed P value of < 0.05 was considered statistically significant. Analyses were performed using SPSS 22.0 (SPSS Inc., Chicago, IL, United States).
Abnormal GGT levels were different in each center. We analyzed both the absolute GGT level and GGT ratio (absolute GGT level divided by the normal cutoff at each center). GGT level was also divided into 4 quartiles of absolute GGT level: Quartile 1 (Q1), percentile (p) < 25; Q2, p25-p49; Q3, p50-p74; and Q4, p ≥ 75. The correlation between GGT and cardiovascular risk was assessed by Pearson correlation. Comparisons between GGT quartiles were performed by using the Kruskal–Wallis test.
A total of 1122 patients (73%) had complete data and were included in the final analysis. A flow diagram of patient selection is shown in Supplemental Figure 1. The median age (IQR) of the study population was 53 (44-60) years, and 532 (47.4%) were females. The median body mass index (kg/m2) was 28.36 (25.8-31.47). The number of participants of Chinese ethnicity was 492 (43.9%). The median baseline GGT level (U/L) was 64 (41-105). According to liver biopsy results, the median of NAS was 4 (3-5), and advanced fibrosis was found in 314 (28%) patients. The median 10-year CVD risk according to the QRISK2 (IQR) was 5.9% (2.6-10.9), and the median relative risk of 10-year CVD risk according to the QRISK2 (IQR) was 1.65 (1.13-2.2), compared to healthy controls with the same age, sex, and ethnicity (Table 1).
| Characteristics | Total | Low risk | High risk | P value |
| median (IQR); n (%) | n = 1122 | n = 800 | n = 322 | N/A |
| Age in yr | 53 (44-60) | 49 (41-55) | 63 (59-67) | < 0.001 |
| Female | 532 (47.4%) | 391 (48.9%) | 141 (43.8%) | 0.12 |
| Weight (kg) | 76 (66-87) | 77 (67-88) | 74 (65-84) | 0.002 |
| Height (m) | 1.63 (1.56-1.7) | 1.64 (1.57-1.71) | 1.61 (1.55-1.67) | < 0.001 |
| BMI (kg/m2) | 28.36 (25.8-31.47) | 28.38 (25.84-31.45) | 28.31 (25.68-31.65) | 0.572 |
| Waist circumference (cm) | 96.5 (89-104) | 96 (88.3-103.5) | 98 (90-106) | 0.017 |
| Hip circumference (cm) | 103 (97-109) | 103 (97-109) | 102.5 (96-109) | 0.502 |
| Ethnic origin: Chinese | 492 (43.9%) | 405 (50.6%) | 87 (27%) | < 0.001 |
| Diabetes | 642 (57.2%) | 365 (45.6%) | 277 (86%) | < 0.001 |
| Hypertension | 639 (57%) | 400 (50.1%) | 239 (74.5%) | < 0.001 |
| Dyslipidemia | 635 (56.6%) | 419 (66.6%) | 216 (75.5%) | 0.007 |
| Albumin (g/L) | 44 (41-46) | 44 (42-46) | 42.5 (39-45) | < 0.001 |
| ALT (U/L) | 63 (41-100) | 69 (43-106) | 49 (30-82) | < 0.001 |
| AST (U/L) | 40 (41-61) | 41 (27-61) | 39 (28-62) | 0.98 |
| ALP (U/L) | 75 (41-96) | 76 (63-95.5) | 74 (58-96.5) | 0.63 |
| GGT (U/L) | 64 (41-105) | 66 (38-103) | 60 (37-113) | 0.563 |
| HbA1c (%) | 6.6 (41-7.5) | 6.2 (5.6-7.2) | 7.2 (6-7.9) | < 0.001 |
| Total cholesterol (mmol/L) | 4.8 (41-5.5) | 4.9 (4.3-5.6) | 4.4 (3-5.113) | < 0.001 |
| LDL cholesterol (mmol/L) | 2.8 (41-3.4) | 2.95 (2.4-3.56) | 2.465 (1-3.1) | < 0.001 |
| HDL cholesterol (mmol/L) | 1.2 (41-1.4) | 1.2 (1-1.4) | 1.14 (0-1.37) | 0.002 |
| Triglycerides (mmol/L) | 1.6 (41-2.1) | 1.6 (1.151-2.1) | 1.6 (1-2.13) | 0.411 |
| Hemoglobin (g/dL) | 14.0 (41-15.2) | 14.2 (13.3-15.3) | 13.6 (12-14.6) | < 0.001 |
| Platelet (109/L) | 243 (41-289) | 249 (205-294.25) | 229 (189-280) | < 0.001 |
| WBC (109/L) | 7.1 (41-8.4) | 7.03 (5.8-8.39) | 7.25 (5-8.4) | 0.28 |
| NAFLD Activity Score1 | 4 (3-5) | 4 (3-5) | 4 (3-5) | 0.684 |
| Significant fibrosis (≥ F2): histopathology | 464 (41.4%) | 286 (35.9%) | 178 (55.3%) | < 0.001 |
| Advanced fibrosis (≥ F3): histopathology | 314 (28%) | 173 (21.7%) | 141 (43.8%) | < 0.001 |
| Cirrhosis | 109 (9.7%) | 50 (6.3%) | 59 (18.3%) | < 0.001 |
| CAP | 325 (294-349) | 323 (297-349) | 326 (286-349) | 0.979 |
| Liver stiffness measurement: TE (kPa) | 9.3 (6.7-13.9) | 8.6 (6.3-12) | 11 (7.6-15.8) | < 0.001 |
| 10-year CVD risk by QRISK2 (%) | 5.9 (2.6-10.9) | 3.8 (1.9-6.4) | 14.7 (12-19.3) | < 0.001 |
| Relative risk of 10-year CVD risk | 1.65 (1.13-2.2) | 1.51 (1.06-2.26) | 1.76 (1-2.08) | < 0.001 |
The median (IQR) 10-year CVD risk in the high-CVD-risk group was 14.7% (12-19.3), compared to 3.8% (1.9-6.4) in the low-risk group, with statistical difference (P < 0.001). In the high-CVD-risk group, the median age was significantly older than that in the low-risk group (63 [59-67] vs 49 [41-55] years; P < 0.001). The high-risk group had a significantly higher prevalence of diabetes (86% vs 45.6%), hypertension (74.5% vs 50.1%), and dyslipidemia (75.5% vs 66.6%), with all P values < 0.01. GGT and aspartate transaminase (AST) were not different between the two groups (GGT [U/L]: median [IQR], 60 [37-113] vs 66 [38-103], P = 0.563; AST [U/L], 39 [28-62] vs 41 [27-61], P = 0.98). However, the alanine transaminase (ALT) level (U/L) was significantly lower in the high-CVD-risk group (49 [30-82] vs 69 [43-106], P < 0.001) (Table 1).
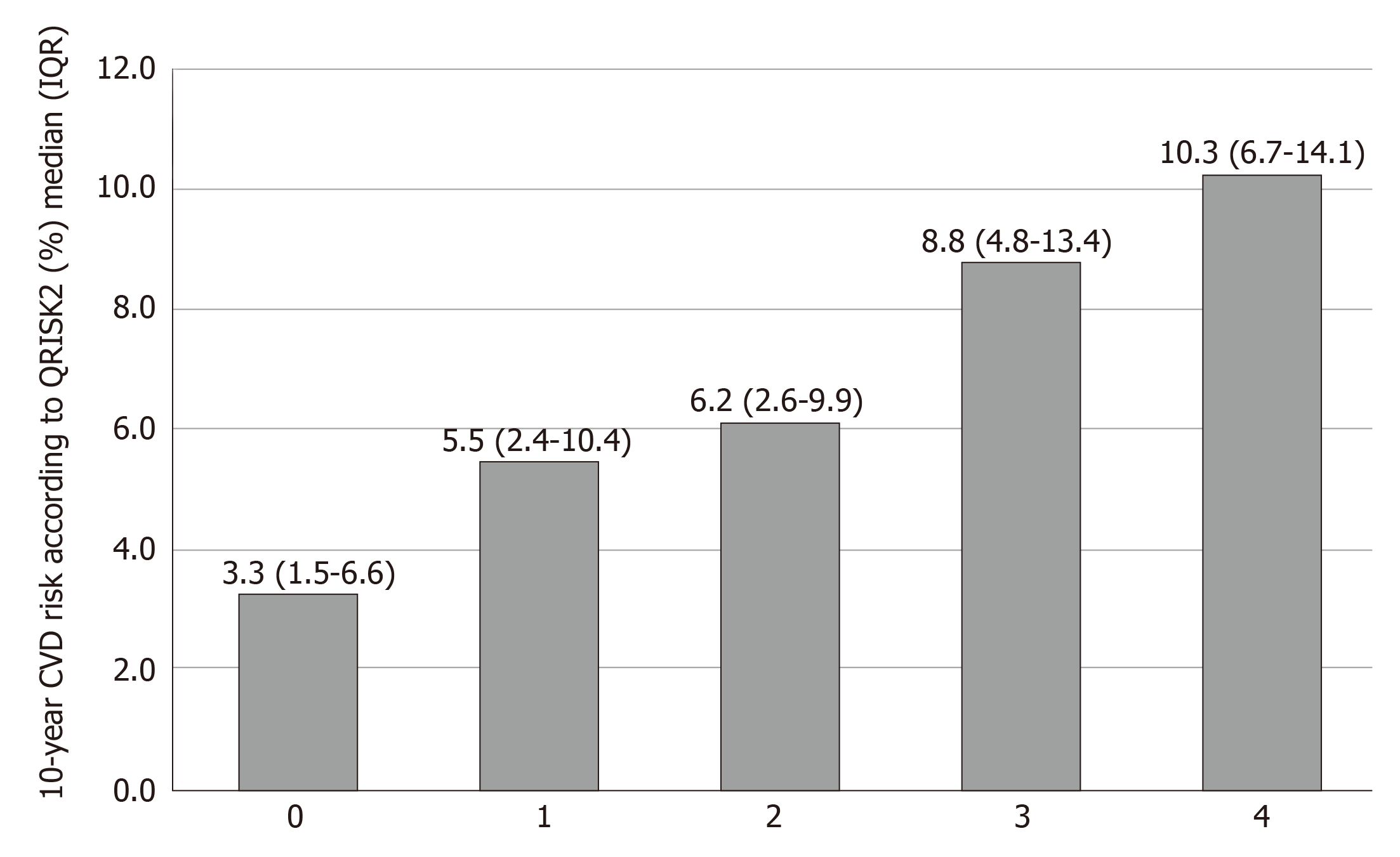
According to the liver biopsy results, NASs were similar between the groups (median [IQR]: high-risk group 4 [3-5] vs low-risk group 4 [3-5], P = 0.684). The high-CVD-risk group was associated with a higher prevalence of advanced fibrosis by liver biopsy (141 [43.8%] vs 173 [21.7%], P < 0.001). Higher fibrosis stage according to liver biopsy significantly was associated with a higher CVD risk (P < 0.001) (Figure 1). LS measurement in the high-CVD-risk group showed higher median TE (IQR) than that in the low-risk group (11 kPa [7.6-15.8] vs 8.6 kPa [6.3-12], P < 0.001). However, the median controlled attenuation parameter was similar between the two groups (P = 0.98) (Table 1).
In the univariate analysis, clinical parameters that predicted higher CVD risk were older age, larger waist circumference, diabetes, hypertension, dyslipidemia, and non-Chinese ethnicity (P < 0.01). Higher HbA1c, lower albumin, ALT, TC, LDL, and HDL were predictors of high CVD risk (P < 0.01). Baseline GGT level did not predict high CVD risk (P = 0.27). Higher fibrosis stage according to liver biopsy and TE were predictors of high CVD risk (P < 0.01). In the multivariate analysis, only older age, non-Chinese ethnicity, diabetes, and lower HDL were predictors of high CVD risk (P < 0.01) (Table 2). All of the variables had no multicollinearity.
| Parameters | Univariate analysis | Multivariate analysis | ||||||
| Exp (B) | 95%CI | P value | Exp (B) | 95%CI | P value | |||
| Lower | Upper | Lower | Upper | |||||
| Age (yr) | 1.39 | 1.33 | 1.45 | < 0.01 | 1.80 | 1.57 | 2.05 | 0.00 |
| BMI (kg/m2) | 1.00 | 0.98 | 1.02 | 0.97 | 0.97 | 0.84 | 1.11 | 0.63 |
| Waist circumference (cm) | 1.01 | 1.00 | 1.03 | 0.02 | 1.03 | 0.97 | 1.10 | 0.37 |
| Ethnic origin: Chinese | 0.36 | 0.27 | 0.48 | < 0.01 | 0.05 | 0.01 | 0.15 | 0.00 |
| Diabetes | 7.34 | 5.20 | 10.35 | < 0.01 | 319.11 | 60.63 | 1679.65 | 0.00 |
| Hypertension | 2.91 | 2.18 | 3.87 | < 0.01 | 1.43 | 0.56 | 3.66 | 0.45 |
| Dyslipidemia | 1.55 | 1.13 | 2.12 | 0.01 | 1.18 | 0.43 | 3.25 | 0.75 |
| Albumin (g/L) | 0.87 | 0.84 | 0.90 | < 0.01 | 0.96 | 0.85 | 1.09 | 0.56 |
| ALT (U/L) | 0.99 | 0.99 | 0.99 | < 0.01 | 1.00 | 0.99 | 1.01 | 0.80 |
| GGT (U/L) | 1.00 | 1.00 | 1.00 | 0.27 | ||||
| HbA1c (%) | 1.36 | 1.23 | 1.51 | < 0.01 | 0.97 | 0.75 | 1.26 | 0.80 |
| Total cholesterol (mmol/L) | 0.68 | 0.59 | 0.78 | < 0.01 | 1.82 | 0.69 | 4.80 | 0.23 |
| LDL cholesterol (mmol/L) | 0.58 | 0.49 | 0.68 | < 0.01 | 1.05 | 0.35 | 3.13 | 0.93 |
| HDL cholesterol (mmol/L) | 0.52 | 0.35 | 0.76 | < 0.01 | 0.05 | 0.01 | 0.21 | 0.00 |
| Platelet (109/L) | 1.00 | 0.99 | 1.00 | < 0.01 | 1.00 | 0.99 | 1.01 | 0.59 |
| Fibrosis stage by liver biopsy | 1.53 | 1.38 | 1.69 | < 0.01 | 1.12 | 0.74 | 1.68 | 0.60 |
| TE (kPa) | 1.06 | 1.04 | 1.08 | < 0.01 | 1.00 | 0.93 | 1.06 | 0.91 |
There was no correlation between baseline GGT level and 10-year CVD risk by QRISK2 (r = 0.022, P = 0.463) (Figure 2). When baseline GGT (U/L) levels were divided into four quartiles (Q), the median (IQR) 10-year CVD risks according to QRISK2 were as follows: Q1 = 6.1 (2.7-11.2), Q2 = 6.6 (2.9-11.9), Q3 = 4.7 (2.4-9.1) and Q4 = 6.4 (2.6-12.9), with a statistically significant difference (P < 0.001) (Table 3). Post hoc analysis revealed that the differences were between Q3 and Q1 (P = 0.02), Q3 and Q2 (P = 0.009), and Q3 and Q4 (P = 0.003).
| Quartiles | Q1 | Q2 | Q3 | Q4 | P value |
| Patients, n = 1122 | 279 | 273 | 282 | 288 | |
| Median GGT (U/L) | 29 (23-33) | 48 (43-56) | 81 (71-89) | 151 (122-220) | |
| High CVD risk (%) | 84 (30.1) | 86 (31.5) | 61 (21.6) | 91 (31.6) | 0.025 |
| Median 10-year, CVD risk (%) | 6.1 (2.7-11.2) | 6.6 (2.9-11.9) | 4.7 (2.4-9.1) | 6.4 (2.6-12.9) | < 0.001 |
Previous data have shown that NAFLD is associated with an increased risk of CVD[4]. Our study demonstrated a higher median 10-year CVD risk, according to QRISK2, in liver biopsy-confirmed NAFLD patients than in healthy controls with the same age, sex, and ethnicity. In addition, our study found that an increase in fibrosis stage significantly increased CVD risk, and advanced fibrosis was a predictor of a high risk of CVD in univariate analysis. Previous meta-analysis revealed that a higher fibrosis stage was associated with an increase in all-cause mortality and liver-related mortality in NAFLD patients; however, this study did not specifically highlight CVD risk and fibrosis stage[23]. Another study on NAFLD related to CVD confirmed that NAFLD with advanced fibrosis, based on the NAFLD fibrosis score, was associated with subclinical atherosclerosis parameters: Carotid intima-media thickness, and the presence of carotid plaque and arterial stiffness in NAFLD patients, independent of conventional cardiometabolic risk factors[24].
A recent study using ultrasound diagnosis of NAFLD showed a higher risk of CVD in the NAFLD group than in the non-NAFLD group, and a high fibrosis 4 score was independently associated with the risk of CVD[25]. Our study emphasized on the value of fibrosis stage in the prediction of CVD risk, even using noninvasive assessment such as TE. However, in multivariate analysis, only traditional risk factors still played a key role in CVD risk assessment. To date, there is no specific recommendation for CVD risk assessment in NAFLD patients. However, using risk assessment from the general population is still essential for preventive measures for NAFLD patients. In clinical practice, identifying the fibrosis stage in NAFLD patients not only predicts the risk of liver-related complications but also CVD risk. The inclusion of liver assessment parameters in CVD risk assessment may predict CVD in the NAFLD group more precisely. Prospective studies are needed.
According to our findings, lower TC and LDL were associated with higher CVD risk in univariate analysis but not in multivariate analysis. However, this could be explained by the fact that the patients included in the study may have been on medication for dyslipidemia that masked the levels of TC and LDL. In patients with dyslipidemia showed no significant difference in TC and LDL levels from non-dyslipidemia in our study. When we focused on the presence of dyslipidemia, which was defined as an abnormal lipid profile or the use of any drugs as evidence of the corresponding metabolic disorders, we found that dyslipidemia was a risk factor for high-risk CVD.
In previous studies, baseline GGT levels were associated with CVD risk in the general population. A large observational study classified serum GGT into four quartiles, and the highest serum GGT quartile had a significant association with CVD compared to the lowest quartile[17]. A meta-analysis of GGT level and CVD mortality showed that the moderate, high and highest levels of GGT had a higher pooled hazard ratio of CVD mortality than the lowest levels of GGT[26]. A meta-analysis in 2013 reported that elevated plasma GGT significantly increased the risk of cardiovascular and all-cause mortality. However, a subgroup analysis reported no association in an Asian population[27,28].
Our study in patients with biopsy-proven NAFLD reported no relationship between baseline GGT level and CVD risk, which was assessed by QRISK2. This result could be explained by the fact that the patients included in this study were only Asian, and the previously mentioned meta-analysis showed conflicting results in this subgroup. Second, NAFLD might confound GGT levels: Serum GGT levels in patients with NAFLD could be elevated in relation to high visceral fat mass. A study in an obesity clinic demonstrated that GGT level was significantly higher in patients with increased visceral adipose tissue after adjusting for body mass index and C-reactive protein[29]. Another study demonstrated a correlation between GGT levels and liver fat estimated by proton magnetic resonance spectroscopy and changes in GGT levels associated with hepatic steatosis after weight loss[30]. Conversely, a study in the general population showed that baseline and visit to visit variability in GGT levels had a linear relationship with all-cause mortality and cardiovascular events, independent of fatty liver. However, fatty liver in this study was calculated based on the fatty liver index, which incorporated the GGT level in the score and might confound the result[19]. We performed binary logistic regression analysis and found that individuals with higher GGT levels had a higher prevalence of grade 3 steatosis (odds ratio, 1.001; 95% confidence interval, 1.000-1.003, P = 0.028) and individuals in higher GGT quartiles also had a higher proportion of grade 3 steatosis (Supplemental Figure 2). As a result, GGT levels in NAFLD patients may reflect hepatic steatosis rather than long-term CVD risk.
The association between GGT and CVD is still debated in regard to whether GGT plays a direct role in the pathogenesis of atherosclerotic processes or coexists. One of the mechanisms of GGT and the atherosclerotic process is the role of GGT in the breakdown of glutathione which has a function of against oxidative stress, leading to the production of cysteinyl-glycine dipeptide: a strong reducing agent that participates in stepwise peroxidation reactions (including LDLs), and exerts a local pro-oxidant and proinflammatory action[31,32]. A plague analysis study revealed the presence of the enzyme product cysteinyl-glycine in the atherosclerotic plague and correlation between mean plaque GGT concentrations and serum GGT levels[33].
We found that the ALT level was significantly lower in the high-CVD-risk group, but this effect was attenuated in the multivariate analysis. A previous meta-analysis in the general population showed no association between ALT and overall CVD risks[34]. However, in NAFLD patients, lower ALT level is associated with advanced fibrosis stage[35] and ALT level is included in fibrosis prediction scores such as NAFLD Fibrosis Score, and fibrosis 4 index[36]. This may confound the univariate analysis of ALT in our study.
The strengths of our study were patients with NAFLD diagnosed by liver biopsy which is the gold standard for NAFLD diagnosis and a large number of patients were included. However, the limitations of our study were the measurement of CVD risk based on QRISK2 and the lack of long-term follow-up CVD outcome even if this tool was well validated in the general population and included calculations for the Asian ethnicity, it could underestimate actual CVD events compared to Framingham score[22].
In summary, NAFLD patients have higher 10-year-cardiovascular risk than healthy controls with the same age, sex, and ethnicity. Apart from traditional risk factors, advanced fibrosis is a predictor for high risk CVD. However, the baseline GGT level cannot be used to predict cardiovascular risk in patients with established NAFLD.
In general population, gamma-glutamyl transferase (GGT) levels have shown the promising results in cardiovascular disease (CVD) risk prediction. The proposed mechanism of the association between GGT level and CVD risk is GGT function on the metabolism of glutathione, which is an important cellular antioxidant in humans.
GGT activities are reportedly abundant in the liver. In nonalcoholic fatty liver disease (NAFLD) patients, whether GGT levels can predict CVD risk is largely debated. Our study focused on the association of GGT level and CVD risk in NAFLD patients.
We aimed to identify the association of baseline GGT level and the QRISK2 score among patients with biopsy-proven NAFLD.
This was a retrospective study involving 1535 biopsy-proven NAFLD patients from 10 Asian centers in 8 countries using data collected by the Gut and Obesity in Asia workgroup. Patients with available baseline GGT level and variables for the QRISK2 calculation were included.
A total of 1122 patients were included in the final analysis. The median 10-year CVD risk (interquartile range [IQR]) was 5.9% (2.6-10.9), and the median relative risk of CVD over 10 years (IQR) was 1.65 (1.13-2.2) compared to healthy individuals with the same age, sex, and ethnicity. Higher fibrosis stages in biopsy-proven NAFLD patients brought a significantly higher CVD risk (P < 0.001). Median GGT level was not different between the two groups (GGT [U/L]: median [IQR], high risk 60 [37-113] vs low risk 66 [38-103], P = 0.56). There was no correlation between baseline GGT level and 10-year CVD risk based on the QRISK2 score (r = 0.02).
NAFLD patients have higher 10-year-CVD risk than healthy controls. Apart from traditional risk factors, advanced fibrosis is a predictor of high-risk CVD. However, the baseline GGT level and GGT quartile cannot be used to predict CVD risk in patients with established NAFLD.
To date, CVD risk assessment in NAFLD patients is based on general population risk assessment tools. Including liver fibrosis parameters may assist in identifying NAFLD patients with high-CVD-risk. Prospective studies are needed.
We would like to thank the supporting team of the Gut and Obesity in Asia workgroup for database. Additionally, we would like to thank research coordinator and statistician, Division of Gastroenterology, King Chulalongkorn Memorial Hospital, Kanokwan Sornsiri and Chonlada Phathong.
| 1. | Younossi Z, Henry L. Contribution of Alcoholic and Nonalcoholic Fatty Liver Disease to the Burden of Liver-Related Morbidity and Mortality. Gastroenterology. 2016;150:1778-1785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 293] [Cited by in RCA: 258] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 2. | Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P, Mills PR, Keach JC, Lafferty HD, Stahler A, Haflidadottir S, Bendtsen F. Liver Fibrosis, but No Other Histologic Features, Is Associated With Long-term Outcomes of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2015;149:389-397.e10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2514] [Cited by in RCA: 2380] [Article Influence: 216.4] [Reference Citation Analysis (5)] |
| 3. | Wong VW, Wong GL, Yip GW, Lo AO, Limquiaco J, Chu WC, Chim AM, Yu CM, Yu J, Chan FK, Sung JJ, Chan HL. Coronary artery disease and cardiovascular outcomes in patients with non-alcoholic fatty liver disease. Gut. 2011;60:1721-1727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 228] [Article Influence: 15.2] [Reference Citation Analysis (3)] |
| 4. | Stepanova M, Younossi ZM. Independent association between nonalcoholic fatty liver disease and cardiovascular disease in the US population. Clin Gastroenterol Hepatol. 2012;10:646-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 261] [Article Influence: 18.6] [Reference Citation Analysis (1)] |
| 5. | Gehrke N, Schattenberg JM. Metabolic Inflammation-A Role for Hepatic Inflammatory Pathways as Drivers of Comorbidities in Nonalcoholic Fatty Liver Disease? Gastroenterology. 2020;Online ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 164] [Article Influence: 27.3] [Reference Citation Analysis (1)] |
| 6. | Sonmez A, Nikolic D, Dogru T, Ercin CN, Genc H, Cesur M, Tapan S, Karslioğlu Y, Montalto G, Banach M, Toth PP, Bagci S, Rizzo M. Low- and high-density lipoprotein subclasses in subjects with nonalcoholic fatty liver disease. J Clin Lipidol. 2015;9:576-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 7. | Chalasani N, Deeg MA, Crabb DW. Systemic levels of lipid peroxidation and its metabolic and dietary correlates in patients with nonalcoholic steatohepatitis. Am J Gastroenterol. 2004;99:1497-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 261] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 8. | Bieghs V, Walenbergh SM, Hendrikx T, van Gorp PJ, Verheyen F, Olde Damink SW, Masclee AA, Koek GH, Hofker MH, Binder CJ, Shiri-Sverdlov R. Trapping of oxidized LDL in lysosomes of Kupffer cells is a trigger for hepatic inflammation. Liver Int. 2013;33:1056-1061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 71] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 9. | Arnett DK, Khera A, Blumenthal RS. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: Part 1, Lifestyle and Behavioral Factors. JAMA Cardiol. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 123] [Article Influence: 20.5] [Reference Citation Analysis (3)] |
| 10. | Hippisley-Cox J, Coupland C, Brindle P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: prospective cohort study. BMJ. 2017;357:j2099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1248] [Cited by in RCA: 1100] [Article Influence: 122.2] [Reference Citation Analysis (4)] |
| 11. | Hippisley-Cox J, Coupland C, Vinogradova Y, Robson J, Minhas R, Sheikh A, Brindle P. Predicting cardiovascular risk in England and Wales: prospective derivation and validation of QRISK2. BMJ. 2008;336:1475-1482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 934] [Cited by in RCA: 1012] [Article Influence: 56.2] [Reference Citation Analysis (0)] |
| 12. | Hippisley-Cox J, Coupland C, Robson J, Brindle P. Derivation, validation, and evaluation of a new QRISK model to estimate lifetime risk of cardiovascular disease: cohort study using QResearch database. BMJ. 2010;341:c6624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 247] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 13. | Lee DH, Blomhoff R, Jacobs DR. Is serum gamma glutamyltransferase a marker of oxidative stress? Free Radic Res. 2004;38:535-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 492] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 14. | Lee DH, Jacobs DR. Association between serum gamma-glutamyltransferase and C-reactive protein. Atherosclerosis. 2005;178:327-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 140] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 15. | Targher G, Byrne CD. Circulating Markers of Liver Function and Cardiovascular Disease Risk. Arterioscler Thromb Vasc Biol. 2015;35:2290-2296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 16. | Koenig G, Seneff S. Gamma-Glutamyltransferase: A Predictive Biomarker of Cellular Antioxidant Inadequacy and Disease Risk. Dis Markers. 2015;2015:818570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 258] [Cited by in RCA: 238] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 17. | Jeon J, Kim DH, Kim W, Choi DW, Jung KJ, Jang SI. Dose-response relationship between gamma-glutamyltransferase and the risk of atherosclerotic cardiovascular diseases in Korean adults. Atherosclerosis. 2020;292:152-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 18. | Yang P, Wu P, Liu X, Feng J, Zheng S, Wang Y, Fan Z. Association Between γ-Glutamyltransferase Level and Cardiovascular or All-Cause Mortality in Patients With Coronary Artery Disease: A Systematic Review and Meta-Analysis. Angiology. 2019;70:844-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Cho EJ, Han K, Lee SP, Shin DW, Yu SJ. Liver enzyme variability and risk of heart disease and mortality: A nationwide population-based study. Liver Int. 2020;Online ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Chan WK, Treeprasertsuk S, Imajo K, Nakajima A, Seki Y, Kasama K, Kakizaki S, Fan JG, Song MJ, Yoon SK, Dan YY, Lesmana L, Ho KY, Goh KL, Wong VW. Clinical features and treatment of nonalcoholic fatty liver disease across the Asia Pacific region-the GO ASIA initiative. Aliment Pharmacol Ther. 2018;47:816-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (3)] |
| 21. | Kleiner DE, Brunt EM, Van Natta M, Behling C, Contos MJ, Cummings OW, Ferrell LD, Liu YC, Torbenson MS, Unalp-Arida A, Yeh M, McCullough AJ, Sanyal AJ; Nonalcoholic Steatohepatitis Clinical Research Network. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9231] [Cited by in RCA: 8612] [Article Influence: 410.1] [Reference Citation Analysis (7)] |
| 22. | Hippisley-Cox J, Coupland C, Vinogradova Y, Robson J, Brindle P. Performance of the QRISK cardiovascular risk prediction algorithm in an independent UK sample of patients from general practice: a validation study. Heart. 2008;94:34-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 135] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 23. | Dulai PS, Singh S, Patel J, Soni M, Prokop LJ, Younossi Z, Sebastiani G, Ekstedt M, Hagstrom H, Nasr P, Stal P, Wong VW, Kechagias S, Hultcrantz R, Loomba R. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: Systematic review and meta-analysis. Hepatology. 2017;65:1557-1565. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1666] [Cited by in RCA: 1548] [Article Influence: 172.0] [Reference Citation Analysis (5)] |
| 24. | Chen Y, Xu M, Wang T, Sun J, Sun W, Xu B, Huang X, Xu Y, Lu J, Li X, Wang W, Bi Y, Ning G. Advanced fibrosis associates with atherosclerosis in subjects with nonalcoholic fatty liver disease. Atherosclerosis. 2015;241:145-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 25. | Baratta F, Pastori D, Angelico F, Balla A, Paganini AM, Cocomello N, Ferro D, Violi F, Sanyal AJ, Del Ben M. Nonalcoholic Fatty Liver Disease and Fibrosis Associated With Increased Risk of Cardiovascular Events in a Prospective Study. Clin Gastroenterol Hepatol. 2019;Online ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 26. | Wang J, Zhang D, Huang R, Li X, Huang W. Gamma-glutamyltransferase and risk of cardiovascular mortality: A dose-response meta-analysis of prospective cohort studies. PLoS One. 2017;12:e0172631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 27. | Hozawa A, Okamura T, Kadowaki T, Murakami Y, Nakamura K, Hayakawa T, Kita Y, Nakamura Y, Okayama A, Ueshima H; NIPPON DATA90 Research Group. gamma-Glutamyltransferase predicts cardiovascular death among Japanese women. Atherosclerosis. 2007;194:498-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 28. | Du G, Song Z, Zhang Q. Gamma-glutamyltransferase is associated with cardiovascular and all-cause mortality: a meta-analysis of prospective cohort studies. Prev Med. 2013;57:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 29. | Verrijken A, Francque S, Mertens I, Talloen M, Peiffer F, Van Gaal L. Visceral adipose tissue and inflammation correlate with elevated liver tests in a cohort of overweight and obese patients. Int J Obes (Lond). 2010;34:899-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 30. | Phillips ML, Boase S, Wahlroos S, Dugar M, Kow L, Stahl J, Slavotinek JP, Valentine R, Toouli J, Thompson CH. Associates of change in liver fat content in the morbidly obese after laparoscopic gastric banding surgery. Diabetes Obes Metab. 2008;10:661-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 31. | Ndrepepa G, Kastrati A. Gamma-glutamyl transferase and cardiovascular disease. Ann Transl Med. 2016;4:481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 96] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 32. | Mason JE, Starke RD, Van Kirk JE. Gamma-glutamyl transferase: a novel cardiovascular risk biomarker. Prev Cardiol. 2010;13:36-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 153] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 33. | Franzini M, Corti A, Martinelli B, Del Corso A, Emdin M, Parenti GF, Glauber M, Pompella A, Paolicchi A. Gamma-glutamyltransferase activity in human atherosclerotic plaques--biochemical similarities with the circulating enzyme. Atherosclerosis. 2009;202:119-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 98] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 34. | Kunutsor SK, Apekey TA, Khan H. Liver enzymes and risk of cardiovascular disease in the general population: a meta-analysis of prospective cohort studies. Atherosclerosis. 2014;236:7-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 209] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 35. | Treeprasertsuk S, Björnsson E, Enders F, Suwanwalaikorn S, Lindor KD. NAFLD fibrosis score: a prognostic predictor for mortality and liver complications among NAFLD patients. World J Gastroenterol. 2013;19:1219-1229. [PubMed] [DOI] [Full Text] |
| 36. | Angulo P, Hui JM, Marchesini G, Bugianesi E, George J, Farrell GC, Enders F, Saksena S, Burt AD, Bida JP, Lindor K, Sanderson SO, Lenzi M, Adams LA, Kench J, Therneau TM, Day CP. The NAFLD fibrosis score: a noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology. 2007;45:846-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2677] [Cited by in RCA: 2392] [Article Influence: 125.9] [Reference Citation Analysis (5)] |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See:
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Thailand
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Fan JG, Popovic DS S-Editor: Tang JZ L-Editor: Filipodia E-Editor: Ma YJ