Published online Jan 14, 2018. doi: 10.3748/wjg.v24.i2.216
Peer-review started: September 12, 2017
First decision: October 18, 2017
Revised: November 3, 2017
Accepted: November 22, 2017
Article in press: November 22, 2017
Published online: January 14, 2018
Processing time: 125 Days and 1.2 Hours
To study the role and the possible mechanism of β-arrestin 2 in lipopolysaccharide (LPS)-induced liver injury in vivo and in vitro.
Male β-arrestin 2+/+ and β-arrestin 2-/- C57BL/6J mice were used for in vivo experiments, and the mouse macrophage cell line RAW264.7 was used for in vitro experiments. The animal model was established via intraperitoneal injection of LPS or physiological sodium chloride solution. Blood samples and liver tissues were collected to analyze liver injury and levels of pro-inflammatory cytokines. Cultured cell extracts were collected to analyze the production of pro-inflammatory cytokines and expression of key molecules involved in the TLR4/NF-κB signaling pathway.
Compared with wild-type mice, the β-arrestin 2 knockout mice displayed more severe LPS-induced liver injury and significantly higher levels of pro-inflammatory cytokines, including interleukin (IL)-1β, IL-6, tumor necrosis factor (TNF)-α, and IL-10. Compared with the control group, pro-inflammatory cytokines (including IL-1β, IL-6, TNF-α, and IL-10) produced by RAW264.7 cells in the β-arrestin 2 siRNA group were significantly increased at 6 h after treatment with LPS. Further, key molecules involved in the TLR4/NF-κB signaling pathway, including phospho-IκBα and phosho-p65, were upregulated.
β-arrestin 2 can protect liver tissue from LPS-induced injury via inhibition of TLR4/NF-κB signaling pathway-mediated inflammation.
Core tip: The role and mechanism of β-arrestin 2 in lipopolysaccharide (LPS)-induced liver injury remain unclear. In this study, β-arrestin 2 knockout mice displayed more severe LPS-induced liver injury and significantly higher levels of pro-inflammatory cytokines than wild-type mice. Further, RAW264.7 cells treated with β-arrestin 2 siRNA expressed significantly higher pro-inflammatory cytokines and molecules involved in the TLR4/NF-κB signaling pathway (including phospho-IκBα and phosho-p65) than the control group at 6 h after treatment with LPS. Therefore, β-arrestin 2 could protect liver tissue from LPS-induced injury via inhibition of TLR4/NF-κB-mediated inflammation and may serve as a therapeutic target.
- Citation: Jiang MP, Xu C, Guo YW, Luo QJ, Li L, Liu HL, Jiang J, Chen HX, Wei XQ. β-arrestin 2 attenuates lipopolysaccharide-induced liver injury via inhibition of TLR4/NF-κB signaling pathway-mediated inflammation in mice. World J Gastroenterol 2018; 24(2): 216-225
- URL: https://www.wjgnet.com/1007-9327/full/v24/i2/216.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i2.216
Core tip: The role and mechanism of β-arrestin 2 in lipopolysaccharide (LPS)-induced liver injury remain unclear. In this study, β-arrestin 2 knockout mice displayed more severe LPS-induced liver injury and significantly higher levels of pro-inflammatory cytokines than wild-type mice. Further, RAW264.7 cells treated with β-arrestin 2 siRNA expressed significantly higher pro-inflammatory cytokines and molecules involved in the TLR4/NF-κB signaling pathway (including phospho-IκBα and phosho-p65) than the control group at 6 h after treatment with LPS. Therefore, β-arrestin 2 could protect liver tissue from LPS-induced injury via inhibition of TLR4/NF-κB-mediated inflammation and may serve as a therapeutic target.
Lipopolysaccharide (LPS, also called endotoxin)-induced hepatic injury is the pathological basis of varied hepatic diseases, and Kupffer cells are the key components in LPS-induced injury[1]. Researchers found that endogenous LPS derived from the intestine could promote the production of pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-1β by activating Kupffer cells and accelerate the damage to the liver[2]. Meanwhile, LPS could also aggravate liver damage in nonalcoholic steatohepatitis by increasing the production of TNF-α by Kupffer cells[3]. Additionally, studies of a genetic mouse model of obesity suggested that the mice were more prone to steatohepatitis if they were constantly exposed to LPS[4,5]. When Kupffer cells were eliminated from this model, the mice showed decreased hepatic injury and lower mortality following LPS treatment[6]. LPS and Kupffer cells are the two essential key points in the development of varied hepatic diseases.
β-arrestin 2 is an important protein that plays a well-established role in regulating signaling downstream of the G-protein-coupled receptor (GPCR) pathway. Its recruitment and binding to the ligand-stimulated receptor are essential for signal transduction, sequestration, desensitization, and cell proliferation and differentiation. Recently, accumulating evidence has shown that β-arrestin 2 is a key regulator of not only GPCR-related signaling pathways but also of pathways downstream of major cell surface receptors and receptor tyrosine kinases, including insulin receptor, insulin-like growth factor type 1 receptor, epidermal growth factor receptor, and Toll-like receptor 4 (TLR4). Of these, the TLR4-related signaling pathway has gained much attention for its role in LPS-induced inflammation and host defense. Current views postulate that stimulation with LPS enhances production of pro-inflammatory cytokines from macrophages via the TLR4/NF-κB signaling pathway, while silencing of TLR4 or β-arrestin 2 can both inhibit this increase in pro-inflammatory cytokines and negatively regulate TLR4-mediated inflammatory reactions[7]. Based on these findings, we hypothesized that β-arrestin 2 should have great effects on LPS-induced inflammation and hepatic injury via a TLR4-related signaling pathway. To explore this hypothesis, we investigated the role and the possible mechanisms of β-arrestin 2 in LPS-induced hepatic injury. We found that deletion of β-arrestin 2 in mice aggravated LPS-induced liver injury by increasing macrophage production of pro-inflammatory cytokines including IL-1β, IL-6, TNF-α, and IL-10. Further, this mechanism might be involved in TLR4/NF-κB-mediated inflammation.
LPS (Cat. L2630), Trizol reagent (Cat. T9424), and rat tail collagen (Cat. L2630) were purchased from Sigma (St Louis, MO, United States). Dulbecco’s modified Eagle’s medium (DMEM, Cat. C11995500B), Roswell Park Memorial Institute 1640 Medium (RPMI-1640, Cat. C11875500BT), fetal bovine serum (Cat. 10270-106), penicillin and streptomycin (Cat. 15140122), and Trypsin (Cat. 25200-056) were obtained from Gibco (Rockville, MD, United States). Real-time PCR Master Mix kit-SYBR Green (Cat. AQ141-04) was from Transgen (Beijing, China). siRNA-β-arrestin 2 (Cat. sc-29208), anti-glyceraldehyde-3-phosphate dehydrogenase antibody (GAPDH, Cat. sc-25778), horseradish peroxidase (HRP) conjugated goat anti-rabbit IgG secondary antibody (Cat. sc-2004), anti-p65 (Cat. sc-372), anti-phospho-IkBα (Cat. sc-8404), anti-IkBα (Cat. sc-371), and anti-TRAF6 (Cat. sc-7221) antibodies were all purchased from Santa Cruz (Santa Cruz, CA, United States). Anti-myeloperoxidase (MPO) primary antibody (Cat. ab9535), anti-β-arrestin 2 (Cat. ab54790), anti-phospho-p65 (Cat. ab86299), and anti-phospho-Akt (Cat. ab38449) antibodies were from Abcam (Abcam, Cambridge, MA, United States). Lipofectamine 3000 (Cat. L3000-015) was from Invitrogen (Invitrogen, Carlsbad, CA,United States). Heparin was from Wanbang (Xuzhou, Jiangsu, China). First Strand cDNA Synthesis Kit ReverTra Ace-α-TM (Cat. FSK-100) was from Toyobo (New York, NY, United States). Alanine transaminase (ALT, Cat. CSB-E16539m), aspartate transaminase (AST, Cat. CSB-E12649m), and TNF-α (Cat. CSB-E04741m) enzyme-linked immunosorbent assay (ELISA) kits were all from Cusabio (Wuhan, Hubei, China).
All animal experiments were approved by the Institutional Animal Care and Use Committee of The Third Affiliated Hospital of Sun Yat-sen University (certification no.: IACUC-F3-17-0801). The original β-arrestin 2+/- heterozygous C57BL/6J mice were a gift from Dr. Robert J Lefkowitz (Duke University Medical Center, Durham, NC). Male β-arrestin 2-/- and β-arrestin 2+/+ mice aged 6 to 8 wk and weighing 20-25 g were randomly divided into four groups with six mice in each group. To establish an LPS-induced liver injury model, the mice were intraperitoneally injected with LPS (5 mg/kg) or physiological solution of sodium chloride. Four hours later, the mice were killed by intraperitoneal injection of 10% chloral hydrate (350 mg/kg). A 0.5 mL blood sample was collected from the inferior vena cava. Serum was stored at -20 °C before testing. The liver was carefully isolated from each mouse. Part of the liver tissue was immediately fixed in 10% neutral buffered formalin before embedding to prepare paraffin sections and the other part was stored at -80 °C for further analysis.
The mouse macrophage cell line RAW264.7 was obtained from American Type Culture Collection. Cells were cultured in DMEM supplemented with 10% fetal bovine serum and 1% antibiotics (penicillin and streptomycin) at 37 °C in a 5% CO2-humidified incubator. Cells at 70% confluence were collected and seeded at 2 × 105 cells per well in a six-well plate for further experimentation. Transfections of cells with β-arrestin 2 siRNA RNAoligo and the control RNAoligo were performed with Lipofectamine 3000 according to the manufacturer’s instructions. At 24 h after transfection, the medium was replaced with regular culture medium and cells were then treated with LPS (1000 ng/mL). At 6 h after administration of LPS, the cells were collected for further experiments.
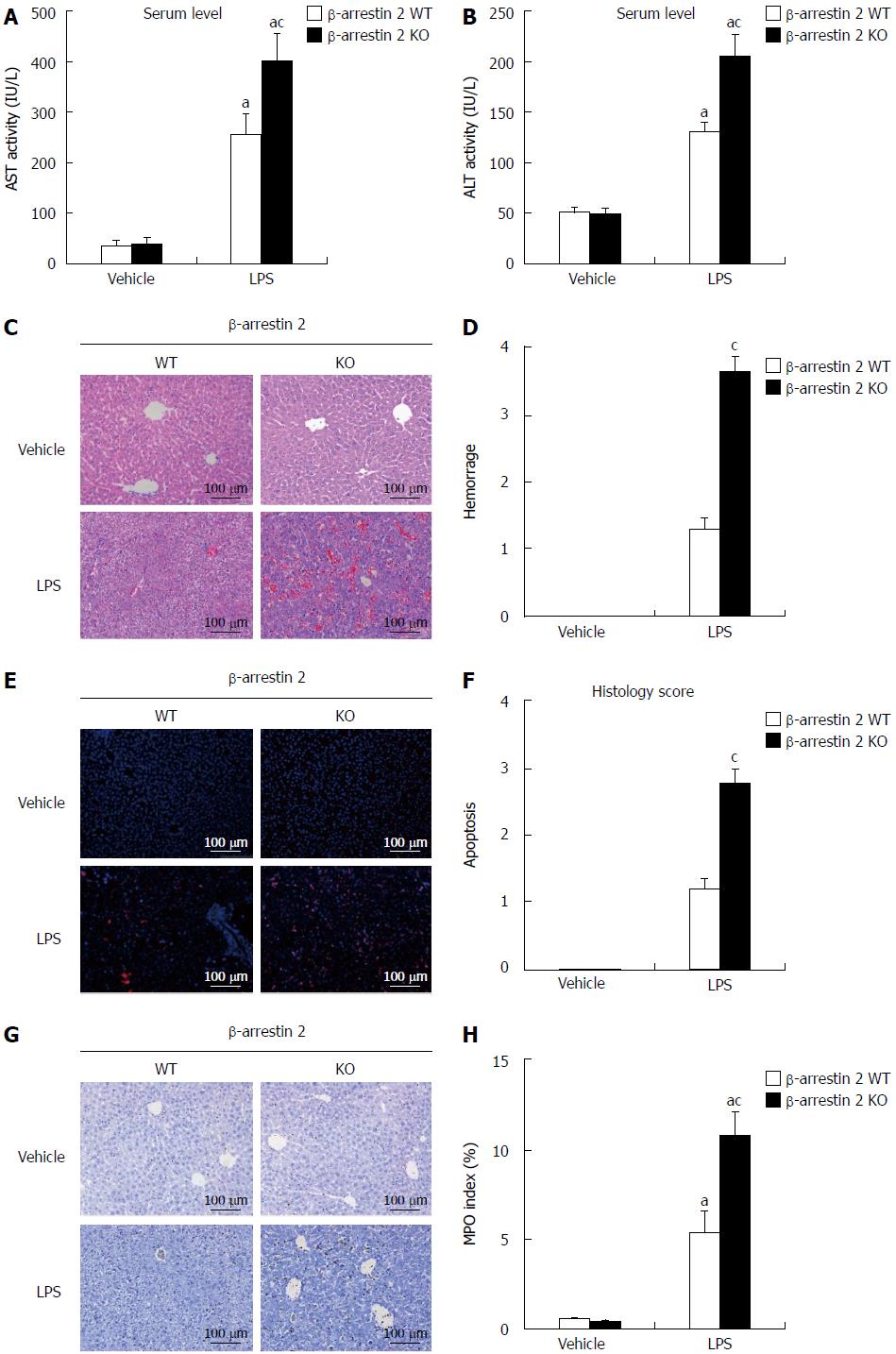
Hematoxylin and eosin (HE) staining and TUNEL staining were performed to generate a histopathology score of liver injury. H&E staining was performed as described in our previous study[8] and TUNEL staining was performed using an in-situ cell death detection kit (Roche, Basel, Switzerland) according to the manufacturer’s instructions. Briefly, sections were scored in a blinded manner for apoptosis and hemorrhage in five 200 × magnified fields according to Hoque’s report[9]. Apoptosis was scored from 0-4 according to the rate of hepatocyte apoptosis (0: ≤1%; 1: 1%-5%; 2: 5%-10%; 3: 10%-20%; and 4: ≥20%) per 200 × field. Hemorrhage was also scored as 0-4 based on the hemorrhage rate (0: 0%; 1: 1%-5%; 2: 5%-20%; 3: 20%-50%; and 4: ≥50%) per 200 × field.
To evaluate LPS-induced hepatic injury, immunohistochemistry was used to detect the expression of MPO in neutrophils, and the number of MPO positive cells was counted in 20 randomly selected 200 × magnified fields of each section. Immunohistochemistry was performed as previously described[10], and MPO was detected using the anti-MPO primary antibody and horseradish peroxidase (HRP) conjugated goat anti-rabbit IgG secondary antibody. The liver tissues of six mice from each group were studied.
Total RNA was collected using Trizol reagent according to the manufacturer’s instructions. Then, 2 μg of RNA was used for reverse transcription to produce first-strand cDNA with the First Strand cDNA Synthesis Kit ReverTra Ace-α-TM according to the manufacturer’s instructions. Real-time PCR was performed for genes of interest on an ABI7700 System (Applied Biosystems, Foster City, CA) using DyNAmo SYBR Green Master Mix. β-actin was used as an internal reference to normalize the genes of interest. The melting curve for each gene was analyzed to ensure the specificity of amplification. The genes of interest and the primers are as follows: TNF-α: forward, 5′-TTCTGTCTACTGAACTTCGGGGTGATCGGTCC-3′ and reverse, 5′- GTATGAGATAGCAAATCGGCTGACGGTGTGGG-3′; IL-1β: forward, 5′-ATGGCAACTGTTCCTGAACTCAACT-3′ and reverse, 5′-CAGGACAGGTATAGATTCTTTCCTTT-3′; IL-6: forward, 5′-AGGATACCACTCCCAACAGACCT-3′ and reverse, 5′-CAAGTGCATCATCGTTGTTCATAC-3′; IL-10: forward, 5′-GCTCTTACTGACTGGCATGAG-3′ and reverse, 5′-CGCAGCTCTAGGAGCATGTG-3′; β-actin: forward, 5′-GGCTGTATTCCCCTCCATCG-3′ and reverse, 5′-CCAGTTGGTAACAATGCCATGT-3′.
Western blot analysis was used to test for the expression of proteins of interest in cultured cells or liver tissues and was performed as previously described[11]. Briefly, equal amounts of protein were separated by electrophoresis and then transferred to polyvinylidene difluoride membranes. After blocking for 1 h at room temperature, the membrane was incubated with primary antibodies against β-arrestin 2, TRAF6, IKKβ, IκBα, phospho-IκBα, p65, phosho-p65, or GAPDH at 4 °C overnight. After washing, the membrane was then incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies for 1 h at room temperature. The bands were visualized using an enhanced chemiluminescence system. Image-Pro Plus 6.0 software (Media Cybernetics, 8484 Georgia Avenue Silver Spring, MD, United States) was used for densitometry analyses.
The levels of ALT, AST, LDH, and TNF-α in the culture supernatants and mouse serum were determined with the ELISA kits according to the manufacturer’s instruction.
SPSS version 13.0 (SPSS Inc., Chicago, IL, United States) was used for statistical analyses. Data are expressed as the mean ± SD and differences between groups were assessed by Student’s t-test or one-way analysis of variance, followed by Bonferroni’s post hoc tests. A two-sided P-value < 0.05 was considered significant.
Deletion of β-arrestin 2 aggravates lipopolysaccharide-induced liver injury in vivo
To investigate the role of β-arrestin 2 in LPS-induced liver injury, we first established an animal model of LPS-induced liver injury by intraperitoneal injection of LPS (5 mg/kg) into β-arrestin 2 wild type (WT) and β-arrestin 2 knockout (KO) mice. Four hours after administration of LPS, the mice were sacrificed and liver tissues and blood were collected for histopathological scoring of liver injury and detection of ALT and AST in serum. As shown in Figure 1, we observed a significant difference in AST and ALT levels (Figure 1A and B) between β-arrestin 2 WT and β-arrestin 2 KO mice treated with LPS, whereas no significant difference was observed in the mice treated with physiological solution of sodium chloride. Similarly, histopathological scores of liver injury, including hemorrhage score (Figure 1D) and apoptosis score (Figure 1F), were significantly higher in β-arrestin 2 KO mice than in β-arrestin 2 WT mice after administration of LPS. Moreover, the MPO index (Figure 1G and H) presented similar results to the AST, ALT, and histopathology scores. These results suggested that decreased β-arrestin 2 aggravated LPS-induced liver injury.
Deletion of β-arrestin 2 facilitates the expression of lipopolysaccharide-induced inflammatory factors
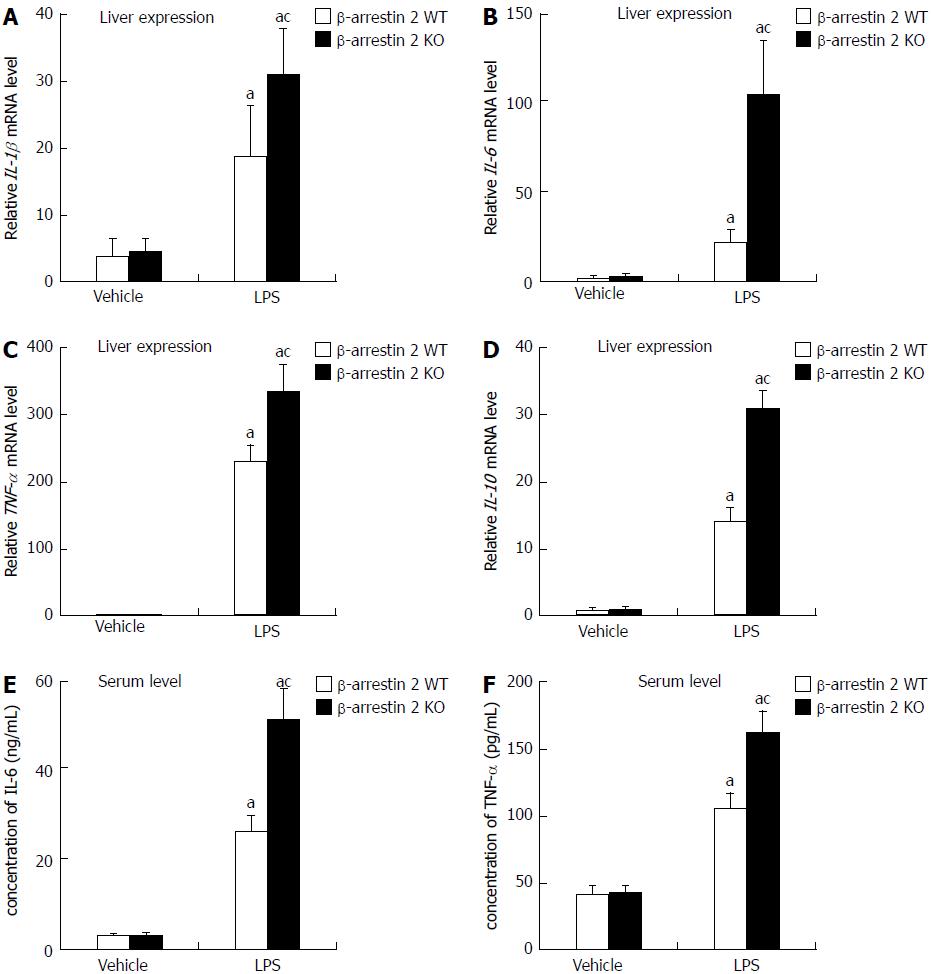
As mentioned in the Introduction section, LPS-induced liver injury involves an increase in pro-inflammatory cytokines via activation of Kupffer cells. We therefore evaluated mRNA levels of IL-1β, IL-6, TNF-α, and IL-10 in liver tissue and serum. We discovered that mRNA levels of IL-1β, IL-6, TNF-α, and IL-10 were noticeably increased after treatment with LPS. The mRNA levels of the four pro-inflammatory cytokines mentioned above were significantly higher in liver tissue from β-arrestin 2 KO mice as compared with liver tissue from the β-arrestin 2 WT mice (Figure 2 A-D). Meanwhile, protein analysis of IL-6 and TNF-α in serum was consistent with the results in liver tissue (Figure 2E and F). These results indicated that increased pro-inflammatory cytokines in both liver tissue and serum might be associated with decreased β-arrestin 2.
Decreasing levels of β-arrestin 2 promote the production of pro-inflammatory factors in RAW264.7 cells in vitro
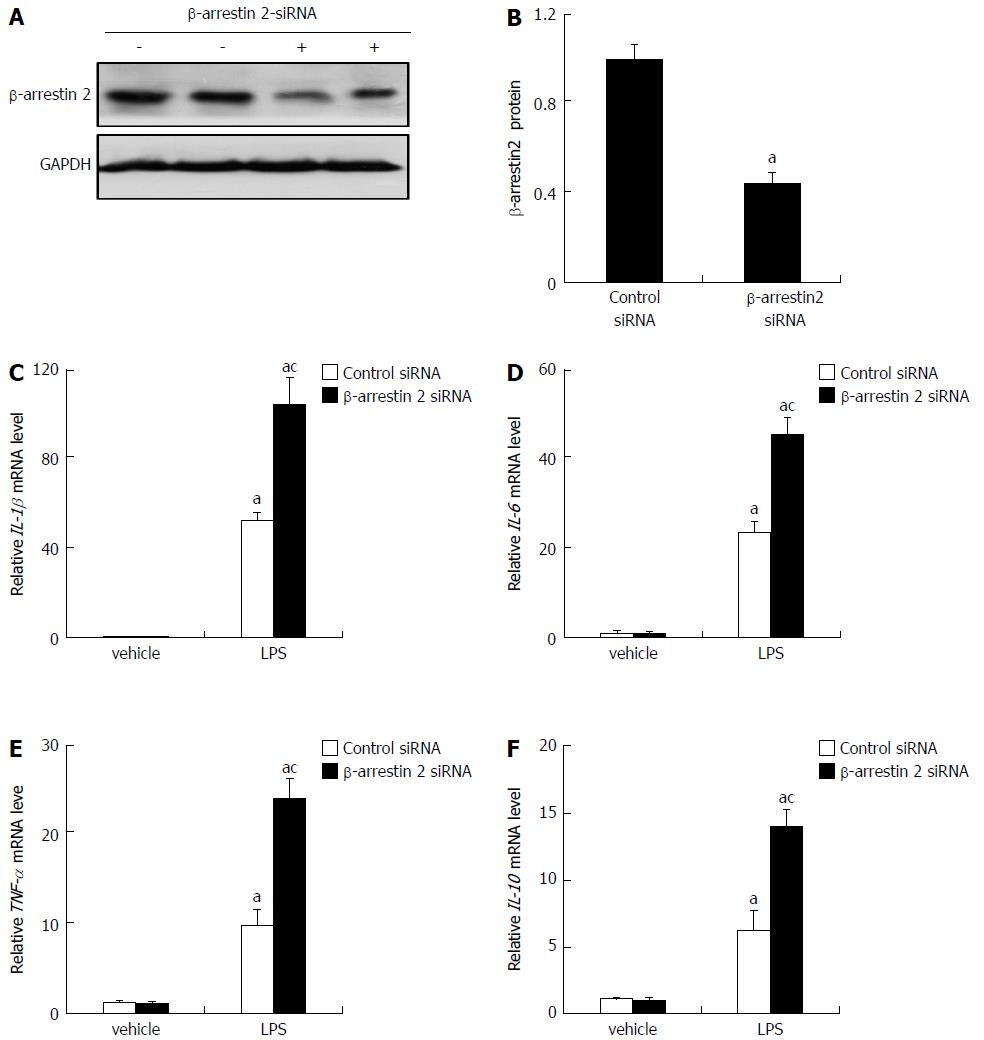
Based on the above results, we supposed that decreasing β-arrestin 2 might promote the production of pro-inflammatory factors via macrophage activation. To confirm this, we investigated whether a genetic reduction of β-arrestin 2 in RAW264.7 cells could increase the production of pro-inflammatory factors. As shown in Figure 3A and B, 6 h after transfection with β-arrestin 2 siRNA, expression of β-arrestin 2 was significantly down-regulated. Meanwhile, at another 6 h after treatment with LPS, RAW264.7 cells treated with β-arrestin 2 siRNA showed significantly increased production of IL-1β, IL-6, TNF-α, and IL-10 (Figure 3C-F). These results revealed that decreasing β-arrestin 2 in RAW264.7 cells promoted the in vitro production of pro-inflammatory factors.
Increased pro-inflammatory factors are involved in activation of the TLR4/NF-κB signaling pathway
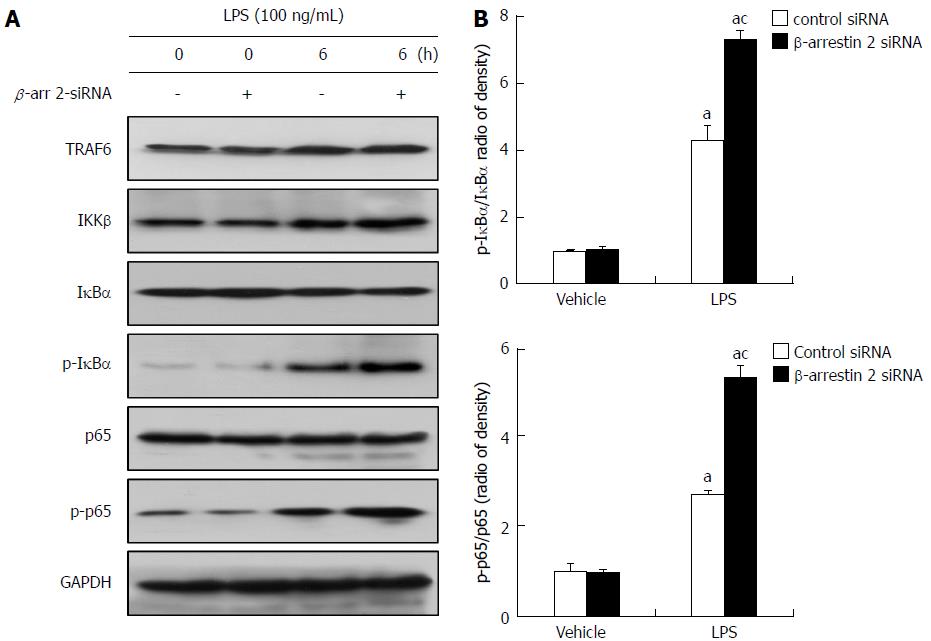
The TLR4/NF-κB signaling pathway is considered to be involved in LPS-induced liver injury, and silencing of β-arrestin 2 can negatively regulate TLR4-mediated inflammatory reactions. To identify the mechanism whereby decreased β-arrestin 2 promotes production of pro-inflammatory factors, we then detected the expression of key molecules in the TLR4/NF-κB signaling pathway. The results showed that key molecules, including TRAF6, IKKβ, phospho-IκBα, and phospho-p65, produced by RAW264.7 cells increased noticeably after treatment with LPS for 6 h (Figure 4A), suggesting that LPS-induced liver injury is related to activation of the TLR4/NF-κB signaling pathway. Moreover, phospho-IκBα and phospho-p65 (but not TRAF6 or IKKβ) were significantly increased in the cells treated with β-arrestin 2 siRNA and LPS (Figure 4B), indicating that decreased β-arrestin 2 might also be involved in activation of the TLR4/NF-κB signaling pathway, therefore promoting the production of pro-inflammatory factors in RAW264.7 cells.
LPS-induced hepatic injury is the pathological basis of varied hepatic diseases. However, the injury is considered indirect and induced by the production of pro-inflammatory cytokines from activated Kupffer cells[12]. In this current study, we found that serum levels of AST and ALT were visibly high in both β-arrestin 2 KO and WT mice treated with LPS for 4 h (Figure 1A and B). Meanwhile, the histopathology scores and count of MPO positive cells were also higher in the LPS group compared with the control group. These results showed obvious liver damage not only in β-arrestin 2 KO mice but also in WT mice, which suggested that our animal model was successful. In addition, mRNA levels of IL-1β, IL-6, TNF-α, and IL-10 in liver tissue and serum levels of IL-6 and TNF-α were significantly higher after treatment with LPS, which also indicated that LPS-induced liver injury was mediated by pro-inflammatory cytokines. All these results were consistent with previous reports by Wang et al[13] and Seregin et al[14].
β-arrestin 2 is an important negative regulator of the TLR4 signaling pathway and could protect mice from TLR4-mediated endotoxic shock and lethality via down-regulation of inflammatory cytokines[13]. However, it is unclear whether β-arrestin 2 could attenuate LPS-induced liver injury via regulation of pro-inflammatory cytokines. Porter et al[7] found that deletion of β-arrestin 2 in mice could decrease serum levels of IL-1β, IL-12p40, interferon-γ, IL-2, IL-3, IL-4, and IL-5. Thus, they hypothesized that β-arrestin 2 could reduce LPS-induced inflammation. Conversely, Li et al[15] found in another study that overexpression (but not deletion) of β-arrestin 2 could reduce the production of pro-inflammatory cytokines such as TNF-α and reduce experimental arthritis severity. Our results showed that ALT and AST levels and histopathology scores were higher in β-arrestin 2 KO mice than in β-arrestin 2 WT mice, which suggested that decreased β-arrestin 2 can aggravate LPS-induced liver damage. Moreover, the pro-inflammatory cytokines in both liver tissues and serum were higher in β-arrestin 2 KO mice, which revealed that decreased β-arrestin 2 might promote production of pro-inflammatory cytokines. We then investigated the correlation between decreased β-arrestin 2 and production of pro-inflammatory cytokines in vitro using siRNA interference technology. As we show in Figure 3, pro-inflammatory cytokines including IL-1β, IL-6, TNF-α, and IL-10 were higher in the cells treated with β-arrestin 2 siRNA and LPS, which revealed that β-arrestin 2 could attenuate LPS-induced liver injury via negative regulation of pro-inflammatory cytokines. Other than negative regulation of pro-inflammatory cytokines, Fong et al[16] found that β-arrestin 2 might modulate the CXCR4-induced chemotactic migration of lymphocytes. Moreover, Basher et al[17] and Fan et al[18] found that β-arrestin 2 inhibited chemotactic migration of neutrophils. In our study, the number of MPO positive cells was larger in liver tissues from β-arrestin 2 KO mice (Figure 1G and H), which indicated that decreased β-arrestin 2 reduce neutrophil infiltration in liver tissue. The results further revealed that inhibition of the chemotactic migration of neutrophils might be another mechanism by which β-arrestin 2 attenuates LPS-induced liver injury.
Kupffer cells are macrophages located in liver tissue, which have been identified as the major cells that produce pro-inflammatory cytokines and play a critical role in LPS-induced inflammation[19-22]. RAW264.7 macrophages are derived from leukemic mice and have similar characteristics to Kupffer cells in liver tissue. Therefore, we used RAW264.7 cells for our in vitro studies. Our results showed that pro-inflammatory cytokines from RAW264.7 cells obviously increased at 6 h after treatment with LPS, which revealed that RAW264.7 could act as Kupffer cells when stimulated by LPS. It is well known that β-arrestin 2 is involved in various cell signaling pathways and plays a key role in the regulation of cell signaling[23]. For example, β-arrestin 2 acts as a scaffold protein and participates in the activation of JNK and ERK in the MAPK pathway[24,25]. Further, β-arrestin 2 can directly bind to TRAF6 and reduce the phosphorylation of NF-κB through inhibition of TRAF6 oligomerization and ubiquitination, further inhibiting activation of the NF-κB pathway[13,26]. A recent study showed that β-arrestin 2 could down-regulate TLR4-mediated production of NLRP3 and IL1-1β, which are involved in inflammation induced by various factors[9]. In our study, we investigated the key molecules involved in the TLR4/NF-κB signaling pathway and found that key molecules, including TRAF6, IKKβ, phospho-IκBα, and phospho-p65, produced by RAW264.7 cells increased noticeably after treatment with LPS (Figure 4). Moreover, siRNA-mediated knockdown of β-arrestin 2 further increased levels of the key molecules mentioned above, including phospho-IκBα and phospho-p65 (Figure 4). This suggested that reductions in β-arrestin 2-induced pro-inflammatory cytokines might be associated with inhibition of the TLR4/NF-κB signaling pathway.
Altogether, our results showed that deletion of β-arrestin 2 in mice aggravated LPS-induced liver injury via increasing macrophage production of pro-inflammatory cytokines including IL-1β, IL-6, TNF-α, and IL-10. This mechanism might be involved in TLR4/NF-κB-mediated inflammation. We therefore concluded that β-arrestin 2 could protect liver tissue from LPS-induced injury via inhibition of TLR4/NF-κB-mediated inflammation. However, further study of the exact role and possible mechanism of β-arrestin 2 is needed.
Lipopolysaccharide (LPS)-induced liver injury serves as the pathological basis of varied hepatic diseases. LPS does not directly harm hepatocytes, while Kupffer cells serve as the key components of LPS-induced injury through secretion of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β. β-arrestin 2 is a protein that plays an important role in regulating the TLR4/NF-κB signaling pathway, which plays a critical role in inflammation. However, the role of β-arrestin 2 in LPS-induced liver injury remains unclear.
The inhibition of LPS-induced inflammation via regulation of the TLR4/NF-κB signaling pathway may be a therapeutic method for modulating LPS-induced injury. β-arrestin 2 is a protein that plays an important role in regulating the TLR4/NF-κB signaling pathway. Therefore, we hypothesized that β-arrestin 2 can play a role in the prevention of LPS-induced liver injury.
The objective of this study was to investigate the role and the possible mechanism of β-arrestin 2 in LPS-induced liver injury in vivo and in vitro. This is the first study to show that β-arrestin 2 attenuated LPS-induced liver injury in a mouse model induced by injection of pure LPS. β-arrestin 2 may serve as a therapeutic target for the prevention and treatment of LPS-induced liver injury.
The animal model was established via intraperitoneal injection of LPS or physiological sodium chloride solution in male β-arrestin 2+/+ and β-arrestin 2-/- C57BL/6J mice. Blood samples and liver tissues were collected for analysis of liver injury and levels of pro-inflammatory cytokines. Extracts from the cultured mouse macrophage cell line RAW264.7 treated with various conditions were collected to analyze the production of pro-inflammatory cytokines and expression of key molecules involved in the TLR4/NF-κB signaling pathway.
The β-arrestin 2 knockout mice displayed more severe LPS-induced liver injury and significantly higher levels of pro-inflammatory cytokines, including IL-1β, IL-6, TNF-α, and IL-10, than the wild-type mice. Compared with the control group, pro-inflammatory cytokines, including IL-1β, IL-6, TNF-α, and IL-10, produced by the β-arrestin 2 siRNA-treated RAW264.7 cells were significantly higher at 6 h after treatment with LPS. The key molecules involved in the TLR4/NF-κB signaling pathway were also increased, including phospho-IκBα and phosho-p65.
We hypothesized that β-arrestin 2 could protect liver tissue from LPS-induced injury via inhibition of TLR4/NF-κB-mediated inflammation. This hypothesis was proven using an animal model of LPS-induced liver injury in male β-arrestin 2+/+ and β-arrestin 2-/- C57BL/6J mice and a cell model using the mouse macrophage cell line RAW264.7. These findings may be helpful for the prevention and treatment of LPS-induced liver injury in future clinical practice via strengthening the function of β-arrestin 2. However, further study on the exact role and possible mechanism is still needed.
Studies of the role of β-arrestin 2 agonists and methods of up-regulation of β-arrestin 2 in the prevention and treatment of LPS-induced liver injury should be performed.
| 1. | Nolan JP. The role of intestinal endotoxin in liver injury: a long and evolving history. Hepatology. 2010;52:1829-1835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 196] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 2. | Enomoto N, Ikejima K, Yamashina S, Hirose M, Shimizu H, Kitamura T, Takei Y, Sato And N, Thurman RG. Kupffer cell sensitization by alcohol involves increased permeability to gut-derived endotoxin. Alcohol Clin Exp Res. 2001;25:51S-54S. [PubMed] [DOI] [Full Text] |
| 3. | Kudo H, Takahara T, Yata Y, Kawai K, Zhang W, Sugiyama T. Lipopolysaccharide triggered TNF-alpha-induced hepatocyte apoptosis in a murine non-alcoholic steatohepatitis model. J Hepatol. 2009;51:168-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 4. | Ye D, Li FY, Lam KS, Li H, Jia W, Wang Y, Man K, Lo CM, Li X, Xu A. Toll-like receptor-4 mediates obesity-induced non-alcoholic steatohepatitis through activation of X-box binding protein-1 in mice. Gut. 2012;61:1058-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 162] [Article Influence: 11.6] [Reference Citation Analysis (4)] |
| 5. | Yang SQ, Lin HZ, Lane MD, Clemens M, Diehl AM. Obesity increases sensitivity to endotoxin liver injury: implications for the pathogenesis of steatohepatitis. Proc Natl Acad Sci USA. 1997;94:2557-2562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 594] [Cited by in RCA: 569] [Article Influence: 19.6] [Reference Citation Analysis (1)] |
| 6. | Budagov RS, Ul’ianova LP. [Effects of modulators of cytokine levels on mice and rats survival under combined radiation/thermal injuries]. Radiats Biol Radioecol. 2004;44:392-397. [PubMed] |
| 7. | Porter KJ, Gonipeta B, Parvataneni S, Appledorn DM, Patial S, Sharma D, Gangur V, Amalfitano A, Parameswaran N. Regulation of lipopolysaccharide-induced inflammatory response and endotoxemia by beta-arrestins. J Cell Physiol. 2010;225:406-416. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 8. | Guo YW, Gu HY, Abassa KK, Lin XY, Wei XQ. Successful treatment of ileal ulcers caused by immunosuppressants in two organ transplant recipients. World J Gastroenterol. 2016;22:5616-5622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 9. | Hoque R, Farooq A, Ghani A, Gorelick F, Mehal WZ. Lactate reduces liver and pancreatic injury in Toll-like receptor- and inflammasome-mediated inflammation via GPR81-mediated suppression of innate immunity. Gastroenterology. 2014;146:1763-1774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 470] [Cited by in RCA: 449] [Article Influence: 37.4] [Reference Citation Analysis (1)] |
| 10. | Wei XQ, Guo YW, Liu JJ, Wen ZF, Yang SJ, Yao JL. The significance of Toll-like receptor 4 (TLR4) expression in patients with chronic hepatitis B. Clin Invest Med. 2008;31:E123-E130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 11. | Wei X, Ke B, Zhao Z, Ye X, Gao Z, Ye J. Regulation of insulin degrading enzyme activity by obesity-associated factors and pioglitazone in liver of diet-induced obese mice. PLoS One. 2014;9:e95399. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 12. | Reiling J, Bridle KR, Schaap FG, Jaskowski L, Santrampurwala N, Britton LJ, Campbell CM, Jansen PLM, Damink SWMO, Crawford DHG. The role of macrophages in the development of biliary injury in a lipopolysaccharide-aggravated hepatic ischaemia-reperfusion model. Biochim Biophys Acta. 2017;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 13. | Wang Y, Tang Y, Teng L, Wu Y, Zhao X, Pei G. Association of beta-arrestin and TRAF6 negatively regulates Toll-like receptor-interleukin 1 receptor signaling. Nat Immunol. 2006;7:139-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 196] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 14. | Seregin SS, Appledorn DM, Patial S, Bujold M, Nance W, Godbehere S, Parameswaran N, Amalfitano A. beta-Arrestins modulate Adenovirus-vector-induced innate immune responses: differential regulation by beta-arrestin-1 and beta-arrestin-2. Virus Res. 2010;147:123-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Li P, Cook JA, Gilkeson GS, Luttrell LM, Wang L, Borg KT, Halushka PV, Fan H. Increased expression of beta-arrestin 1 and 2 in murine models of rheumatoid arthritis: isoform specific regulation of inflammation. Mol Immunol. 2011;49:64-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 16. | Fong AM, Premont RT, Richardson RM, Yu YR, Lefkowitz RJ, Patel DD. Defective lymphocyte chemotaxis in beta-arrestin2- and GRK6-deficient mice. Proc Natl Acad Sci USA. 2002;99:7478-7483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 257] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 17. | Basher F, Fan H, Zingarelli B, Borg KT, Luttrell LM, Tempel GE, Halushka PV, Cook JA. beta-Arrestin 2: a Negative Regulator of Inflammatory Responses in Polymorphonuclear Leukocytes. Int J Clin Exp Med. 2008;1:32-41. [PubMed] |
| 18. | Fan H, Bitto A, Zingarelli B, Luttrell LM, Borg K, Halushka PV, Cook JA. Beta-arrestin 2 negatively regulates sepsis-induced inflammation. Immunology. 2010;130:344-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Van Rooijen N, Sanders A. Kupffer cell depletion by liposome-delivered drugs: comparative activity of intracellular clodronate, propamidine, and ethylenediaminetetraacetic acid. Hepatology. 1996;23:1239-1243. [PubMed] [DOI] [Full Text] |
| 20. | Su GL, Klein RD, Aminlari A, Zhang HY, Steinstraesser L, Alarcon WH, Remick DG, Wang SC. Kupffer cell activation by lipopolysaccharide in rats: role for lipopolysaccharide binding protein and toll-like receptor 4. Hepatology. 2000;31:932-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 209] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 21. | Seki E, Tsutsui H, Nakano H, Tsuji N, Hoshino K, Adachi O, Adachi K, Futatsugi S, Kuida K, Takeuchi O. Lipopolysaccharide-induced IL-18 secretion from murine Kupffer cells independently of myeloid differentiation factor 88 that is critically involved in induction of production of IL-12 and IL-1beta. J Immunol. 2001;166:2651-2657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 186] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 22. | Kopydlowski KM, Salkowski CA, Cody MJ, van Rooijen N, Major J, Hamilton TA, Vogel SN. Regulation of macrophage chemokine expression by lipopolysaccharide in vitro and in vivo. J Immunol. 1999;163:1537-1544. [PubMed] |
| 23. | Porter-Stransky KA, Weinshenker D. Arresting the Development of Addiction: The Role of β-Arrestin 2 in Drug Abuse. J Pharmacol Exp Ther. 2017;361:341-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 24. | McDonald PH, Chow CW, Miller WE, Laporte SA, Field ME, Lin FT, Davis RJ, Lefkowitz RJ. Beta-arrestin 2: a receptor-regulated MAPK scaffold for the activation of JNK3. Science. 2000;290:1574-1577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 660] [Cited by in RCA: 662] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 25. | Cervantes D, Crosby C, Xiang Y. Arrestin orchestrates crosstalk between G protein-coupled receptors to modulate the spatiotemporal activation of ERK MAPK. Circ Res. 2010;106:79-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 26. | Sun W, Yang J. Molecular basis of lysophosphatidic acid-induced NF-κB activation. Cell Signal. 2010;22:1799-1803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Manautou JE, Roychowdhury S S- Editor: Chen K L- Editor: Wang TQ E- Editor: Li RF