Published online Feb 7, 2017. doi: 10.3748/wjg.v23.i5.763
Peer-review started: July 9, 2016
First decision: September 12, 2016
Revised: November 16, 2016
Accepted: January 11, 2017
Article in press: January 11, 2017
Published online: February 7, 2017
Processing time: 199 Days and 16.2 Hours
Familial intrahepatic cholestasis (FIC) comprises a group of rare cholestatic liver diseases associated with canalicular transport defects resulting predominantly from mutations in ATP8B1, ABCB11 and ABCB4. Phenotypes range from benign recurrent intrahepatic cholestasis (BRIC), associated with recurrent cholestatic attacks, to progressive FIC (PFIC). Patients often suffer from severe pruritus and eventually progressive cholestasis results in liver failure. Currently, first-line treatment includes ursodeoxycholic acid in patients with ABCB4 deficiency (PFIC3) and partial biliary diversion in patients with ATP8B1 or ABCB11 deficiency (PFIC1 and PFIC2). When treatment fails, liver transplantation is needed which is associated with complications like rejection, post-transplant hepatic steatosis and recurrence of disease. Therefore, the need for more and better therapies for this group of chronic diseases remains. Here, we discuss new symptomatic treatment options like total biliary diversion, pharmacological diversion of bile acids and hepatocyte transplantation. Furthermore, we focus on emerging mutation-targeted therapeutic strategies, providing an outlook for future personalized treatment for inherited cholestatic liver diseases.
Core tip: Familial intrahepatic cholestasis (FIC) is a group of autosomal recessive liver diseases characterized by intrahepatic cholestasis. Phenotypes vary from only episodic disease to progressive FIC. Current therapeutic options are often insufficient to prevent progression of the disease. This review will discuss the current therapeutic regimen as well as the development of novel therapeutic strategies, focusing on surgical and pharmacological biliary diversion, hepatocyte transplantation and mutation-specific therapy.
- Citation: van der Woerd WL, Houwen RH, van de Graaf SF. Current and future therapies for inherited cholestatic liver diseases. World J Gastroenterol 2017; 23(5): 763-775
- URL: https://www.wjgnet.com/1007-9327/full/v23/i5/763.htm
- DOI: https://dx.doi.org/10.3748/wjg.v23.i5.763
Core tip: Familial intrahepatic cholestasis (FIC) is a group of autosomal recessive liver diseases characterized by intrahepatic cholestasis. Phenotypes vary from only episodic disease to progressive FIC. Current therapeutic options are often insufficient to prevent progression of the disease. This review will discuss the current therapeutic regimen as well as the development of novel therapeutic strategies, focusing on surgical and pharmacological biliary diversion, hepatocyte transplantation and mutation-specific therapy.
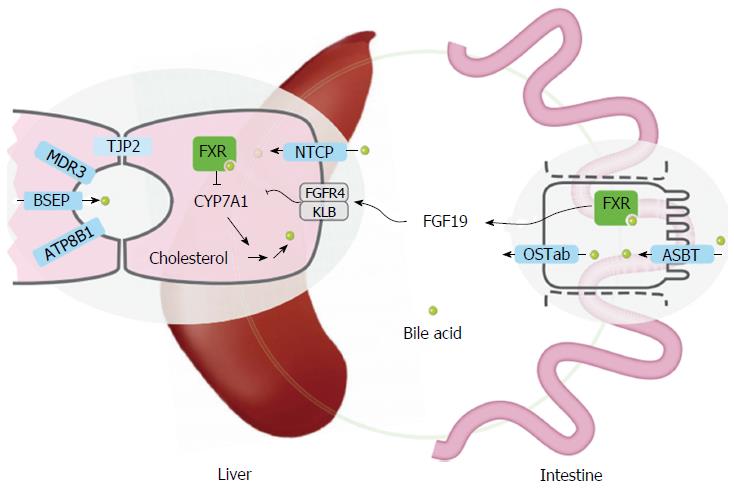
Familial intrahepatic cholestasis (FIC) comprises a group of rare cholestatic diseases with autosomal recessive inheritance, which are classified based on their genetic defect[1]. Three of the responsible mutated genes, namely ATP8B1, ABCB11 and ABCB4, were identified in 1998 and the corresponding diseases are now generally referred to as ATP8B1, ABCB11 and ABCB4 deficiency[2-4]. The gene products are specific canalicular transport proteins (Figure 1). ATP8B1 is an aminophospholipid flippase that is probably important for maintaining lipid asymmetry of the plasma membrane. Disturbances in the membrane composition might interfere with the function of other transmembrane proteins including the bile salt export pump (BSEP, ABCB11) that is encoded by ABCB11. BSEP accounts for the active transport of bile salts into the canalicular lumen thereby generating bile flow. Dysfunction will result in accumulation of bile salts in hepatocytes with subsequent hepatocellular damage. Finally, ABCB4 encodes the multidrug resistance protein 3 (MDR3, ABCB4), a phosphatidylcholine (PC) floppase, responsible for the biliary secretion of phospholipids. Impaired PC secretion prevents proper micelle formation resulting in the presence of potentially harmful free bile salts[5]. ABCB11 and ABCB4 are localized solely at the canalicular membrane of hepatocytes, while ATP8B1 is abundantly expressed in a wide variety of other tissues such as the small intestine, bladder, ear and pancreas.
Patients with FIC usually present in infancy with cholestasis and associated problems like pruritus and malabsorption. Progressive cholestasis eventually results in liver failure. Patients with ATP8B1 deficiency can have multiple phenotypes ranging from benign recurrent intrahepatic cholestasis (BRIC1), associated with recurrent cholestatic attacks, to the more severe form called progressive familial intrahepatic cholestasis (PFIC1)[6]. In some patients the disease may present as a clinical continuum starting with BRIC and evolving into PFIC[6]. Despite cholestasis, patients with ATP8B1 deficiency have normal serum gamma-glutamyl transferase (GGT) activity. In line with its broad expression, multiple organ systems besides the liver can be affected by the loss of function of the ATP8B1 protein. The high incidence of extrahepatic features such as diarrhea, pancreatitis and hearing loss, distinguishes ATP8B1 deficiency from the other FIC subtypes[7-9]. Like in ATP8B1, mutations in ABCB11 can also result in mild (BRIC2) as well as severe cholestasis (PFIC2) and GGT activity is normal. Therefore, these two subtypes are referred to as low-GGT FIC. ABCB11 deficiency specifically is associated with the development of cholelithiasis and a considerable risk for hepatobiliary malignancies[10,11]. In contrast, mutations in ABCB4 predispose to several hepatobiliary disorders, such as transient neonatal cholestasis, low phospholipid-associated cholelithiasis and cholangiocarcinoma, but most frequently PFIC type 3[5,12]. Serum GGT is always elevated and no extrahepatic features or association with malignancies is described.
Recently, the tight junction protein 2 gene (TJP2) was found to be associated with low-GGT PFIC too[13]. This disease is now referred to as PFIC type 4. The TJP2 gene encodes tight junction protein-2, which is not a transporter but involved in the organization of epithelial and endothelial intercellular junctions that, in the liver, separate bile from plasma (Figure 1). Still, some of the patients with low-GGT FIC do not have mutations in either of these genes, indicating that other genes might be involved.
This review will discuss the current therapeutic regimen as well as the development of novel therapeutic strategies for FIC, focusing on surgical and pharmacological biliary diversion, hepatocyte transplantation and mutation-specific therapy.
Medical therapy: Alongside the standard supplementation of fat-soluble vitamins to prevent deficiencies, two drugs can be used: ursodeoxycholic acid (UDCA) and rifampicin. UDCA stimulates hepatobiliary secretion of bile salts. UDCA might enhance bile flow by stimulating the impaired targeting of transport proteins such as BSEP or the conjugate export pump MRP2 (multidrug resistance-associated protein 2, encoded by ABCC2) to the canalicular membrane via activation of a complex signaling network[14,15]. Antiapoptotic effects of UDCA conjugates may also contribute to protection of hepatocytes. Treatment with UDCA is first-line therapy and effective in more than half of the patients with ABCB4 deficiency[16]. In patients with low-GGT PFIC, the response to UDCA therapy was less promising, but still resulted in improvement of serum transaminase levels and pruritus in some patients[16,17]. Since it is safe and without major side effects, treatment can be attempted in these patients, but in case of progressive liver disease, prompt surgical intervention is needed. Rifampicin might work, at least in part, by upregulating detoxification enzymes and export pumps by mechanisms dependent of pregnane X receptor[18]. Increased excretion of bile salts in the urine through stimulation of 6-α hydroxylation could also contribute to the beneficial effect of rifampicin. In low-GGT PFIC patients, rifampicin treatment does not result in improvement of serum transaminases and bilirubin and reduces pruritus in only a few patients[16]. Recently, treatment with serotonin reuptake inhibitors was suggested for the management of refractory cholestatic pruritus in patients with PFIC[19]. Although the mechanism of action is not clear, pruritus improved upon treatment in 14 out of 20 patients with PFIC or Alagille syndrome. Since no severe adverse events were reported in this group, serotonin reuptake inhibitors like sertraline could be considered for uncontrolled pruritus.
In BRIC patients, cholestatic attacks eventually resolve spontaneously without permanent liver damage. Yet, these episodes can be long-lasting and the pruritus impairs quality of life significantly[7]. In contrast to its marginal effect in PFIC patients, rifampicin is able to reduce pruritus and even completely abort a cholestatic episode in BRIC patients. UDCA, on the other hand, is rarely effective[16].
Nasobiliary drainage: Another non-surgical intervention is nasobiliary drainage, a temporary diversion of bile established by endoscopically introducing a nasobiliary drain[20]. Due to the temporary character, this therapy is especially useful to abort long-term cholestatic episodes in BRIC patients. The risk of procedure-related pancreatitis needs to be considered.
Partial biliary diversion: The purpose of partial biliary diversion (PBD) is to reduce the enterohepatic circulation of bile salts, thereby diminishing the accumulation of these compounds and preventing hepatic injury. Currently, PBD is therapy of choice in all non-cirrhotic children with low-GGT PFIC when permanent cholestasis and/or intractable pruritus is present. This intervention was introduced in 1988, as partial external biliary diversion (PEBD) by cholecystojejunocutaneostomy[21]. In up to 85% of the non-cirrhotic patients, PEBD is successful in improving pruritus as well as biochemical parameters of cholestasis such as serum bile acids, liver enzymes and bilirubin. Also long-term results indicate that PEBD might delay or even reverse hepatic injury[22,23]. However, PEBD is associated with the disadvantage of a permanent stoma[24,25]. Therefore, alternative methods have been explored preventing the presence of a stoma, including ileal exclusion and partial internal biliary diversion (PIBD). The latter procedure is performed by cholecystocolostomy or using an isolated jejunal loop as a conduit from gall bladder to mid ascending colon[26-28]. At present, only a few reports are available that describe the long-term results of these procedures. One study reported inferior long-term outcome of ileal bypass compared to PEBD with recurrence of symptoms in 50% of the patients. The recurrence in this type of procedure is probably the result of increasing re-absorption of bile acids over time[29]. On the contrary, the first long-term results of PIBD were very promising with outcomes comparable to those of PEBD[30]. A potential complication of PIBD however is choleretic diarrhea resulting from large amounts of bile salts entering the colon[30]. This problem occurs regularly but can be managed with the use of oral bile salt binders (cholestyramine). Furthermore, the possible side-effects of the direct bile flow on the colonic mucosa, such as the development of colitis or colon carcinoma, need to be investigated. Although long-term results are still limited, PIBD already is frequently used as a stoma-free alternative for PEBD[30-32].
Liver transplantation: Biliary diversion will not be successful if significant fibrosis or cirrhosis is already present. Therefore, liver transplantation is indicated for patients with advanced liver disease or persistent uncontrolled pruritus despite PBD. Living-donor liver transplantation is also a safe and effective option[33,34]. However, since ATP8B1 deficiency is not confined to the liver, extrahepatic manifestations can cause significant morbidity even after transplantation. Features such as pancreatitis, short stature and hearing loss will persist and with liver transplantation diarrhea can be induced or exacerbate[8,35]. ATP8B1 is highly expressed in the small intestine where it might have a role in the regulation of reabsorption of bile salts[36]. After transplantation, normal amounts of bile salts are excreted by the hepatic graft while intestinal ATP8B1 is still impaired. High concentrations of bile salts in the colon may result in diarrhea which however can be diminished by treatment with bile adsorptive resin. Furthermore, significant hepatic steatosis often occurs after transplantation which can progress to cirrhosis and may require re-transplantation[37,38]. The pathogenesis of this steatosis is not totally clear, but it might also be secondary to the continued intestinal malfunction of ATP8B1 since it coincides with diarrhea in practically all patients[37]. Liver transplantation usually gives complete correction of phenotype in patients with ABCB4 and ABCB11 deficiency. However, in some patients with severe ABCB11 deficiency, recurrence has been observed post liver transplantation as a result of the formation of autoantibodies against BSEP[39]. Up to 8% of transplanted PFIC2 patients develop these antibodies. Most of these patients had more severe mutations, resulting in a total absence of BSEP[40]. Treatment options include changes of immunosuppressive therapy, depletion of anti-BSEP antibodies by plasmapheresis or immunoadsorption or treatment with rituximab[41]. A second liver transplantation may be necessary, although almost all re-transplanted patients again developed recurrence of cholestasis.
Total biliary diversion: In some patients, PBD is not effective in reducing clinical symptoms and improving biochemical parameters. For these patients, total biliary diversion (TBD) might be useful[42]. In four patients with ATP8B1 deficiency, of which three did not benefit from PEBD, total external biliary diversion resulted in a significant reduction or complete disappearance of pruritus and cholestasis. No clinical signs of fat malabsorption were encountered, although fat-soluble vitamin levels turned out to be more difficult to manage than in PBD. If additional long-term results confirm the safety and effectivity of PIBD, this PBD variant can also be converted to a TBD by choledochal duct ligation in patients that do not respond to this procedure. It is likely that at least some residual bile salt export capacity is necessary to respond to TBD and therefore mutation type may be of influence on the effectivity. The four PFIC1 patients that were described all beared the same mutation (c.2932-3C>A), that was shown to predominantly result in aberrant splicing but also in a small percentage of normally spliced product[43]. The exact role of genotype on the outcome of TBD is still unknown. However, in accordance with PBD, TBD might also be beneficial to patients with other mutations in ATP8B1 or JAG1, as well as to patients with mutations in ABCB11. Currently, TBD is not recommended as primary therapy since PBD is already effective in the vast majority of the patients and the experience with TBD is limited to only a few patients. Moreover, the long-term effects of total absence of intestinal bile salts are unknown. This is especially important as during the last 15 years it became clear that bile salts are not only necessary for solubilizing lipophilic nutrients in the small intestine but can also act as regulatory molecules that are important in glucose and lipid metabolism. Dysregulation of these pathways might therefore interfere with various physiologic processes, with unknown consequences in the long run[44]. However, given the good results in the described patients and the alternative being a liver transplantation, TBD can be added to the treatment options for patients with severe low-GGT PFIC and persisting symptoms after PBD.
Liver transplantation combined with biliary diversion in PFIC1 patients: Liver transplantation alone is often not a permanent solution in patients with severe ATP8B1 deficiency since the graft is frequently affected by steatohepatitis requiring re-transplantation, and diarrhea is persistent or even worse after transplantation. Diarrhea and malnutrition seem to play an important role in the development of post-transplant steatosis[37]. Recently, biliary diversion has also been used in combination with liver transplantation to improve diarrhea and nutritional status in two patients, which proved to be effective in terms of clinical, histological and biochemical outcomes[45-47]. Again, the long-term safety of total external drainage needs to be established. Yet, liver transplantation combined with variants of biliary diversion seems a promising solution to reduce the frequent complications post transplantation.
Pharmacological diversion of bile acids: Hepatic accumulation and toxicity of bile acids likely plays an essential role in the etiology of liver damage in PFIC. Therefore, pharmacological inhibition of bile acid uptake transporters could be beneficial clinically as well. The majority of bile acids are absorbed by active transport in the terminal ileum, mediated by the apical sodium-dependent bile acid transporter (ASBT) (Figure 1). Using a mouse model for genetic cholestasis, two studies recently described significantly reduced liver fibrosis and inflammation upon treatment with small molecules that inhibit ASBT[48,49]. Fecal bile acid excretion largely increased, providing a likely explanation for the effect. Although both studies used Abcb4 knockout mice, which is a model for PFIC3 or inflammation of bile ducts and not for low-GGT cholestasis, it is well possible that patients with ATP8B1 or ABCB11 deficiency could be successfuly treated with such ASBT inhibitors. One possible disadvantage of this strategy is the increased hepatic synthesis of bile acids. In this respect, targeting the ileal basolateral bile acid export, mediated by the organic solute transporter (OST) OSTα-OSTβ (OSTab), could be an interesting alternative approach to reduce hepatic bile acid accumulation (Figure 1). OSTab is a heteromeric protein, consisting of 2 subunits, both of which are essential for bile acid transport. OSTa knockout mice show a less cholestatic phenotype upon ligation of the common bile duct, related to increased loss of bile acids in urine[50]. Notably, OSTa depletion leads to enhanced intestinal activation of the farnesoid X receptor (FXR), the main nuclear receptor for bile acids. FXR activation results in increased secretion of FGF19, which reduces bile acid synthesis in the liver[50,51]. Unfortunately, no OSTab inhibitors have been developed to date. Also pharmacological FXR activation leads to increased production of the FGF19 hormone, which reduces hepatic bile acid synthesis (Figure 1). Recently, a nontumorigenic FGF19-like peptide was designed that does not affect proliferation but reduces bile acid production in humans[52,53]. This peptide effectively reversed cholestatic liver injury in Abcb4-deficient mice[54] and could potentially also be beneficial in ATP8B1 and ABCB11 deficiency. Finally, inhibition of hepatic bile acid uptake could be effective to reduce accumulation of toxic bile acids in the liver. The first results of clinical trials with Myrcludex B, a peptidic inhibitor for one of the major uptake proteins for conjugated bile acids, sodium taurocholate cotransporting polypeptide (NTCP), have recently been published[55,56]. Myrcludex B was designed to treat hepatitis B virus (HBV) infections, as NTCP is the docking platform for HBV uptake[57,58]. NTCP inhibition seems well tolerated, and mice and men lacking NTCP are viable[59,60]. However, effects of Myrcludex B on cholestasis are not yet reported.
Hepatocyte transplantation: Given the shortage of donor organs, hepatocyte transplantation might become an alternative to liver transplantation[61,62]. Additional advantages of hepatocyte transplantation are that the procedure is less invasive, can be repeated several times and leaves the native liver in situ. The procedure may delay or even eliminate the need for liver transplantation. However, scarcity of donor hepatocytes again is the limiting factor. In addition, immunosuppression with its associated morbidity is still necessary. Therefore, alternative cell sources are being investigated such as induced pluripotent stem cells (iPSCs) derived hepatocyte-like cells or hepatocytes generated by transdifferentiation[63]. Still, other important issues remain: poor engraftment, survival and function of the transplanted cells. Since especially cirrhosis impairs engraftment and survival, extrahepatic transplantation sites need to be investigated. Furthermore, progress has been achieved in the organogenesis from iPSCs. Takebe et al[64] succeeded in generating vascularized and functional human liver from iPSCs by transplantation of three-dimensional liver buds created in vitro. These liver buds could also be delivered to extrahepatic sites. However, the hepatocyte-like cells were not fully differentiated, as evidenced by lower albumin secretion and lower expression of hepatocyte specific CYP450 enzymes compared to primary human hepatocytes. Furthermore, the absence of cholangiocytes in the organoid might be a problem, since the bile produced by the organoid will be released into the circulation. A promising alternative approach is the clonal expansion of single Lgr 5+ bipotent liver progenitor cells into transplantable liver organoids[65]. Upon transplantation in mice, these ductal organoids differentiate into apparently functional hepatocytes. In contrast to iPSC derived hepatocytes or liver buds, the organoid cells are obtained directly from the liver without the need for genetic modification or introduction of reprogramming factors, diminishing the risk of malignancies[66,67]. Although several issues need to be addressed, these techniques might enable personalized autologous hepatocyte transplantation in the future. However, for patients with ABCB11 deficiency, hepatocyte transplantation is probably less suitable, since premalignant cells are left in place.
Gene therapy: Despite treatment, the quality of life and life expectancy of many patients with PFIC are still limited. Furthermore, in patients with ATP8B1 deficiency, extra-hepatic features persist, even after liver transplantation. Gene therapy corrects the defective gene responsible for disease development and can be applied to different tissues. However, multiple barriers are still present, including the vector genome and transcriptional persistence as well as the immune response that can limit the viability of transduced cells[68]. In addition, genes like ATP8B1 and ABCB11 are large and therefore difficult to insert into conventional viral vectors. Alternatively, autologous transplant with ex vivo genetically corrected stem cells or hepatocyte-like cells generated from patient derived iPSCs, may hold promise of success. Because liver organoids can be based on the in vitro expansion of a single progenitor cell, these might also be particular suitable for genetic modifications. Also in vivo gene editing using the bacterial CRISPR/Cas system might become an option in the future[69].
Mutation-specific therapy: An alternative approach is molecular rescue of mutation-specific defects. Since these strategies target common disease-causing mechanisms, rare diseases like inherited cholestatic liver diseases can benefit from the experience in other diseases sharing the same underlying mechanisms, such as cystic fibrosis (CF). CF is a multi-organ disease that predominantly affects the lungs, caused by a variety of mutations in the CF transmembrane conductance regulator (CFTR)[70]. CF is particularly suitable to use as a model disease, since it is one of the most common life-threatening monogenic diseases, clinically approved drugs are available and new drugs are being developed.
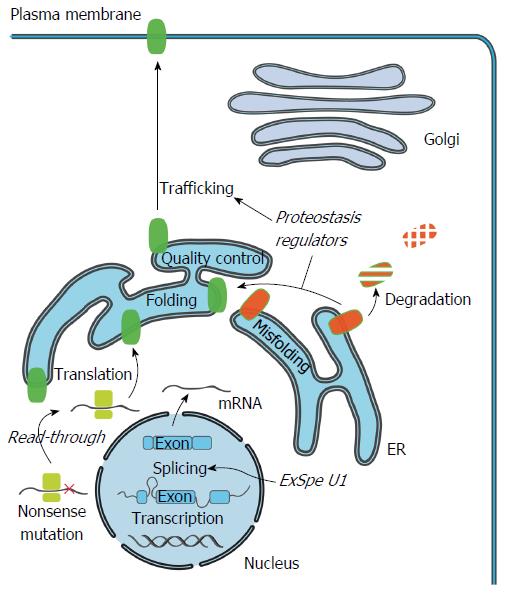
A considerable number of mutations associated with PFIC have been reported so far[2-4,71-73]. Most of them are rare or limited to a specific population. In line with the classification of CFTR mutations, we can subdivide these mutations in four classes[74,75]. The first class consists of mutations that cause defects in full-length protein synthesis resulting in a non-functional protein. Mutations in this group include nonsense mutations, splice site abnormalities causing a premature termination codon (PTC) or frame shift, and frame shifts due to insertions or deletions. The second class includes mutations resulting in a reduced number of normal transcripts such as some splicing mutants, which allow the synthesis of some residual normal mRNA. This class can also include promotor mutations that reduce transcription. The third class comprises mutations associated with impaired trafficking to the plasma membrane or impaired stability. This can be due to misfolding and subsequent degradation but for ATP8B1 might also be the result of disturbed interaction with CDC50A. The last class comprises mutants that exhibit no or reduced functionality.
To develop targeted therapies, elucidation of the molecular effects, caused by the wide-spectrum of mutations that result in PFIC, is very relevant. Recently, the effect of an increasing number of mutations, resulting in one of the FIC subtypes, has been characterized[43,73,76-82]. Knowing the disease mechanism will allow the classification of mutations according to their functional defect and will help to choose the most adequate therapeutic strategy aimed at correcting this defect (Figure 2).
Restoration of full-length protein synthesis: PTCs cause inappropriate termination of translation leading to a truncated protein, unable to fulfill its functions. If a PTC is caused by a nonsense mutation, a potential pharmacological approach is to selectively promote the translational read-through of the PTC by small molecules, aiming to restore the expression of a full-length protein. Compounds that are known to achieve this goal are the aminoglycoside antibiotics. However, high intravenous or intramuscular concentrations are necessary, leading to serious toxicity. Another compound, PTC124 (Ataluren) was reported to selectively induce ribosomal read-through of premature but not normal termination codons. Unfortunately, clinical trials in patients with CF or Duchenne muscular dystrophy have shown limited efficacy[83-85]. However, in CF patients that did not take chronic inhaled tobramycin, small but significant improvements in pulmonary function were reported[84]. Nevertheless, further optimization or discovery of new compounds is required to achieve clinical efficacy in the future.
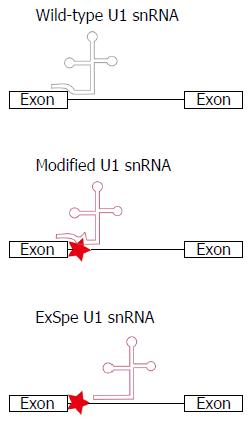
Non-functional truncated proteins can also be caused by mutations interfering with pre-messenger RNA splicing. Most of the identified disease-associated ATP8B1 mutations in exon-intron boundaries were characterized using minigenes, as an alternative to RNA analysis of affected tissue, comparing the splicing patterns of mutant and wild-type exons. All of them resulted to some extent in aberrant splicing resulting in PTCs or large in frame deletions[43]. Also intronic ABCB11 mutations resulting in abnormal splicing, were identified[81]. The splicing process is normally initiated by binding of the U1 small nuclear RNA (snRNA) to the pre-messenger RNA splice donor site (SDS) by specific base-pairing to the last three nucleotides of an exon and the first six nucleotides of an intron (Figure 3). Mutations interfering with the SDS may reduce complementarity with the U1 snRNA and disturb the splicing process. In vitro, modified U1 snRNA constructs, matching the mutated SDS, could rescue splicing very efficiently for a specific group of ATP8B1 splice-site mutations located at splice donor as well as splice acceptor sites[43]. This same strategy can also be used for the splice site mutations in class 2. Two advantages of the U1 technique compared to conventional gene replacement therapy are that i) the endogenous regulation of transcription is preserved and ii) the small size of the U1 snRNA construct is better suited for insertion in viral vectors. Furthermore, it is becoming increasingly evident that also exonic changes are frequently responsible for missplicing. For instance, a considerable number of missense mutations and single nucleotide polymorphisms in ABCB11 resulted in aberrant pre-mRNA splicing[78]. Treatment with modified U1 constructs might therefore be applicable to even more patients. However, the safety and efficacy in vivo of such treatment needs to be investigated. The use of exon-specific U1 constructs, binding at less conserved intronic regions, may reduce the risk of off-target effects (Figure 3)[43,86,87]. The first in vivo data were recently published showing a significant increase of the percentage normal splicing in mice with coagulation factor VII deficiency or spinal muscular atrophy, without obvious harmful side-effects[87-89]. These promising findings indicate that this strategy may have broad clinical potential for the treatment of a large group of genetic disorders associated with abnormal splicing, including inherited cholestatic liver diseases.
Rescue of impaired protein trafficking: The cellular pathways involved in maintaining the integrity of the proteome are collectively referred to as the proteostasis network. This network coordinates protein synthesis, folding, trafficking and degradation[90]. The inability to restore disturbed proteostasis due to misfolded proteins leads to disease. In vitro studies show that some missense mutations, associated with PFIC type 1, 2 and 3, indeed affect protein processing and trafficking, preventing the protein from passing the endoplasmic reticulum (ER) quality control system with subsequent proteasomal breakdown[76-80,82,91,92]. These mutations include the ATP8B1 mutation p.I661T and ABCB11 mutations p.E297G and p.D482G, which are the most frequently found mutations in European patients[76,79]. In addition, nine of the 35 characterized ABCB4 variants also cause retention of ABCB4 in the ER[92]. Cyclosporins were identified as pharmcological chaperones targeting ABCB4 specifically and could improve protein maturation of some of these mutants[80,82]. In contrast to protein-specific compounds like cyclosporins, proteostasis regulators are compounds that can manipulate general proteostasis pathways thereby restoring protein homeostasis and ameliorate disease. These compounds could potentially be beneficial for multiple rare diseases associated with protein misfolding. One of these proteostasis regulator compounds is the histone deacetylase inhibitor 4-phenyl butyric acid (4-PBA). For some of the ATP8B1, ABCB11 and ABCB4 mutants resulting in retention in the ER, 4-PBA has been shown to result in a marked induction of plasma membrane expression in vitro[77,79,92]. Recently, also the first positive in vivo results of treatment with 4-PBA were published[93-97]. Five PFIC2 patients were treated with 4-PBA resulting in partial restoration of protein expression at the plasma membrane as well as clinical improvement with disappearance of pruritus and improved liver tests[94-96]. Furthermore, 4-PBA treatment was successful in aborting a cholestatic attack in a BRIC2 patient[97]. Although no beneficial effect on transaminases, GGT and bilirubin was detected, treatment resulted in significant relief of intractable itch in three patients with the progressive form of ATP8B1 deficiency. However, the effect of 4-PBA on the biochemical markers of cholestasis and liver histology might depend on mutation type and the associated degree of residual ATP8B1 activity.
The most common CFTR mutant F508del also impairs protein folding and various CFTR correctors have been discovered to rescue this misfolding. Some of these CFTR corrector compounds were shown to overcome the trafficking block of ATP8B1 mutant p.I661T at the ER, presumably acting as proteostasis regulators[98]. In addition to 4-PBA, six compounds were able to significantly enhance the plasma membrane expression of p.I661T-ATP8B1 in vitro[98]. Two of these compounds were already approved by the Food and Drug Administration for the management of other diseases. This will facilitate the clinical application of these compounds for other indications. Moreover, these data show that p.I661T-ATP8B1, after leaving the ER, is likely to reach its proper location at the canalicular membrane. Evaluation of the functionality of the corresponding p.L622T mutation in P4-ATPase family member ATP8A2, suggested that p.I661T-ATP8B1 maintains functionality. In line with these findings, the transport function of ABCB11 mutants p.E297G and p.D482G, associated with impaired membrane trafficking, remained largely unchanged[76].
The clinical effect of the most promising CFTR corrector, VX-809, was disappointing in CF patients[99]. Despite very promising pre-clinical results of some proteostasis regulator compounds, it needs to be evaluated whether these will indeed be clinically meaningful in PFIC patients. Combination therapy with multiple proteostasis regulator compounds acting at different steps along the folding pathway may however provide a basis for a more effective intervention. Combination of the two most promising corrector compounds (suberoylanilide hydroxamic acid, SAHA and compound C4) in vitro did result in an additional improvement of ATP8B1 cell surface abundance[98]. Moreover, it is important to consider that modulating protein misfolding by targeting the proteostasis network is one of the few treatments that potentially can also improve the extrahepatic features in patients with ATP8B1 deficiency. Therefore, these proteostasis regulator compounds hold promise for the future treatment of well-defined patients with inherited cholestatic liver disorders.
The new therapeutic options described here hold promise for the future treatment of specific subgroups of patients with FIC, paving the way to the development of personalized treatment based on molecular profiles. Below, we describe our view on the challenging route to deliver the benefits of this in vitro work to the patients.
The majority of the mutations still cannot be assigned to a specific mutational class with certainty, due to the absence of molecular characterization. Furthermore, some mutations may be included in multiple classes, making rescue therapy even more difficult to achieve. Therefore, the first step, after genotyping, is the characterization of the patient-specific underlying disease-mechanism. Molecular analysis can be used to predict disease phenotype and determine the optimal treatment strategy, most likely to benefit the individual patient. Since residual activity might be one of the main predictors of a positive treatment outcome, a complicating factor in this analysis is that representative functional assays for ATP8B1 remain challenging[100,101]. ATP8B1 mutations are often in residues that are conserved across the P4-ATPase subfamily. It is therefore also possible to consider measuring the potential impact of these mutations in another P4-ATPase with well characterized transporter activity and specificity such as family member ATP8A2. This ATP8B1 homologue exhibits ATPase activity that is stimulated by phosphatidylserine and to a lesser degree phosphatidylethanolamine and was previously used to evaluate the possible functional consequences of p.I661T-ATP8B1, showing preserved functionality[98]. In addition, a cellular assay for characterizing P4-ATPase-mediated transport in living yeast cells was developed[102].
The success of personalized medicine depends on having accurate tests that identify patients who can benefit from targeted therapies, avoiding costly treatments with limited benefits and possible side effects. Regular in vitro assays using artificial cell lines can be used as pre-clinical assays. However, it is essential that the results of such an assay have a high predictive value for the actual clinical outcomes. Therefore, it is attractive to evaluate the treatment efficacy in a patient’s native cellular and genetic background. Now, iPSCs from patient specific cell sources, such as skin fibroblast, hair follicle cells, patient blood samples and even urine containing small amount of epithelial cells, can be generated. These iPSCs and iPSC-derived hepatocytes can serve as disease model. iPSC-derived in vitro models can be used to confirm a genetic diagnosis, demonstrate mutation pathogenicity and they can also serve as the basis for drug screening or gene therapy optimization. Patient-specific liver or intestinal organoids could be even better pre-clinical disease models[103]. Although a liver biopsy is necessary, growing an organoid culture from actual patients might show specific defects and will facilitate the determination of specific treatment regimens. Notably, organoids from patients with other liver diseases such as α1-antitrypsin deficiency and Alagille syndrome mirrored the in vivo pathology[67]. It might also be possible to use these organoids for indirect functional assays since they can readily be converted into functional hepatocytes in vitro. Functional organoid cells secrete albumin as well as bile salts into the medium and exhibit adequate detoxifying qualities. By evaluating these parameters, disease severity and therapy response might be predicted. Given the broad tissue expression of ATP8B1, patient-derived rectal organoids might be a suitable alternative modelsystem for evaluation of drug-response in patients with ATP8B1 deficiency, circumventing the risk of a liver biopsy.
On the basis of pre-clinical results, patients can be selected that are suitable for a specific therapy. The ultimate way to verify in vitro drug responses in vivo, is a randomized controlled trial. However, given the low number of patients per mutation, it is practically impossible to conduct such trials. Single patient trials can be considered[104]. Single patient trials, also known as n-of-1 trials, are multi-period crossover experiments comparing two or more treatments within one patient. These trials are only suitable for evaluating long-term treatments for chronic conditions with stable treatment response, quick onset of treatment effect and only modest carryover effects. For most therapies, these data will be unknown and also patients with rapidly progressive disease are not eligible. Furthermore, to evaluate the effect of mutation-specific therapy, we need an objective in vivo read-out of response. For patients with PFIC we can use biochemical parameters such as serum liver enzymes and bile acids. However, this is impossible for patients with episodic disease since biochemical parameters normalize in between the cholestatic attacks. In addition, the duration in between episodes is unpredictable. Currently, treatment effect and drug efficacy might therefore be hard to prove, especially in BRIC patients. Still, a lot can be learned from the experience with these therapies in other, more frequent, diseases with a comparable basic defect, like CF.
Recently, important steps were taken towards a more personalized treatment strategy for FIC patients focusing on mutation-targeted therapeutic strategies. Promising mutation-specific therapies, like the use of modified U1 snRNA and proteostasis regulators, have much broader potential, and can also be applied to other genetic diseases that are associated with defective pre-mRNA splicing or impaired protein folding. This will facilitate the movement from concept to clinical use enormously. Further research should focus on the establishment of a solid functional assay for ATP8B1, representative patient-specific preclinical assays as well as a good clinical read-out, to make personalized therapy a reality.
| 1. | van Mil SW, Houwen RH, Klomp LW. Genetics of familial intrahepatic cholestasis syndromes. J Med Genet. 2005;42:449-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 41] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 2. | Bull LN, van Eijk MJ, Pawlikowska L, DeYoung JA, Juijn JA, Liao M, Klomp LW, Lomri N, Berger R, Scharschmidt BF. A gene encoding a P-type ATPase mutated in two forms of hereditary cholestasis. Nat Genet. 1998;18:219-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 650] [Cited by in RCA: 500] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 3. | Strautnieks SS, Bull LN, Knisely AS, Kocoshis SA, Dahl N, Arnell H, Sokal E, Dahan K, Childs S, Ling V. A gene encoding a liver-specific ABC transporter is mutated in progressive familial intrahepatic cholestasis. Nat Genet. 1998;20:233-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 852] [Cited by in RCA: 670] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 4. | de Vree JM, Jacquemin E, Sturm E, Cresteil D, Bosma PJ, Aten J, Deleuze JF, Desrochers M, Burdelski M, Bernard O. Mutations in the MDR3 gene cause progressive familial intrahepatic cholestasis. Proc Natl Acad Sci USA. 1998;95:282-287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 631] [Cited by in RCA: 455] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 5. | Gordo-Gilart R, Andueza S, Hierro L, Martínez-Fernández P, D’Agostino D, Jara P, Alvarez L. Functional analysis of ABCB4 mutations relates clinical outcomes of progressive familial intrahepatic cholestasis type 3 to the degree of MDR3 floppase activity. Gut. 2015;64:147-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 63] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 6. | van Ooteghem NA, Klomp LW, van Berge-Henegouwen GP, Houwen RH. Benign recurrent intrahepatic cholestasis progressing to progressive familial intrahepatic cholestasis: low GGT cholestasis is a clinical continuum. J Hepatol. 2002;36:439-443. [PubMed] |
| 7. | Tygstrup N, Steig BA, Juijn JA, Bull LN, Houwen RH. Recurrent familial intrahepatic cholestasis in the Faeroe Islands. Phenotypic heterogeneity but genetic homogeneity. Hepatology. 1999;29:506-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 46] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 8. | Lykavieris P, van Mil S, Cresteil D, Fabre M, Hadchouel M, Klomp L, Bernard O, Jacquemin E. Progressive familial intrahepatic cholestasis type 1 and extrahepatic features: no catch-up of stature growth, exacerbation of diarrhea, and appearance of liver steatosis after liver transplantation. J Hepatol. 2003;39:447-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 116] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 9. | Stapelbroek JM, Peters TA, van Beurden DH, Curfs JH, Joosten A, Beynon AJ, van Leeuwen BM, van der Velden LM, Bull L, Oude Elferink RP. ATP8B1 is essential for maintaining normal hearing. Proc Natl Acad Sci USA. 2009;106:9709-9714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 104] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 10. | van Mil SW, van der Woerd WL, van der Brugge G, Sturm E, Jansen PL, Bull LN, van den Berg IE, Berger R, Houwen RH, Klomp LW. Benign recurrent intrahepatic cholestasis type 2 is caused by mutations in ABCB11. Gastroenterology. 2004;127:379-384. [PubMed] |
| 11. | Knisely AS, Strautnieks SS, Meier Y, Stieger B, Byrne JA, Portmann BC, Bull LN, Pawlikowska L, Bilezikçi B, Ozçay F. Hepatocellular carcinoma in ten children under five years of age with bile salt export pump deficiency. Hepatology. 2006;44:478-486. [PubMed] |
| 12. | Jacquemin E, De Vree JM, Cresteil D, Sokal EM, Sturm E, Dumont M, Scheffer GL, Paul M, Burdelski M, Bosma PJ. The wide spectrum of multidrug resistance 3 deficiency: from neonatal cholestasis to cirrhosis of adulthood. Gastroenterology. 2001;120:1448-1458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 335] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 13. | Sambrotta M, Strautnieks S, Papouli E, Rushton P, Clark BE, Parry DA, Logan CV, Newbury LJ, Kamath BM, Ling S. Mutations in TJP2 cause progressive cholestatic liver disease. Nat Genet. 2014;46:326-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 269] [Cited by in RCA: 214] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 14. | Beuers U. Drug insight: Mechanisms and sites of action of ursodeoxycholic acid in cholestasis. Nat Clin Pract Gastroenterol Hepatol. 2006;3:318-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 338] [Cited by in RCA: 282] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 15. | Dombrowski F, Stieger B, Beuers U. Tauroursodeoxycholic acid inserts the bile salt export pump into canalicular membranes of cholestatic rat liver. Lab Invest. 2006;86:166-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 60] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 16. | Stapelbroek JM, van Erpecum KJ, Klomp LW, Houwen RH. Liver disease associated with canalicular transport defects: current and future therapies. J Hepatol. 2010;52:258-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 129] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 17. | Dinler G, Koçak N, Ozen H, Yüce A, Gürakan F. Ursodeoxycholic acid treatment in children with Byler disease. Pediatr Int. 1999;41:662-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 18. | van Dijk R, Kremer AE, Smit W, van den Elzen B, van Gulik T, Gouma D, Lameris JS, Bikker H, Enemuo V, Stokkers PC. Characterization and treatment of persistent hepatocellular secretory failure. Liver Int. 2015;35:1478-1488. [PubMed] |
| 19. | Thébaut A, Habes D, Gottrand F, Rivet C, Cohen J, Debray D, Jacquemin E, Gonzales E. Sertraline as an Additional Treatment for Cholestatic Pruritus in Children. J Pediatr Gastroenterol Nutr. 2016; [Epub ahead of print]. [PubMed] |
| 20. | Stapelbroek JM, van Erpecum KJ, Klomp LW, Venneman NG, Schwartz TP, van Berge Henegouwen GP, Devlin J, van Nieuwkerk CM, Knisely AS, Houwen RH. Nasobiliary drainage induces long-lasting remission in benign recurrent intrahepatic cholestasis. Hepatology. 2006;43:51-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 74] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 21. | Whitington PF, Whitington GL. Partial external diversion of bile for the treatment of intractable pruritus associated with intrahepatic cholestasis. Gastroenterology. 1988;95:130-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 191] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 22. | Davis AR, Rosenthal P, Newman TB. Nontransplant surgical interventions in progressive familial intrahepatic cholestasis. J Pediatr Surg. 2009;44:821-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 23. | Schukfeh N, Metzelder ML, Petersen C, Reismann M, Pfister ED, Ure BM, Kuebler JF. Normalization of serum bile acids after partial external biliary diversion indicates an excellent long-term outcome in children with progressive familial intrahepatic cholestasis. J Pediatr Surg. 2012;47:501-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 49] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 24. | Rebhandl W, Felberbauer FX, Turnbull J, Paya K, Barcik U, Huber WD, Whitington PF, Horcher E. Biliary diversion by use of the appendix (cholecystoappendicostomy) in progressive familial intrahepatic cholestasis. J Pediatr Gastroenterol Nutr. 1999;28:217-219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 25. | Schukfeh N, Gerner P, Paul A, Kathemann S, Metzelder M. Laparoscopic button cholecystostomy for progressive familial intrahepatic cholestasis in two children. Eur J Pediatr Surg. 2014;24:433-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 26. | Bustorff-Silva J, Sbraggia Neto L, Olímpio H, de Alcantara RV, Matsushima E, De Tommaso AM, Brandão MA, Hessel G. Partial internal biliary diversion through a cholecystojejunocolonic anastomosis--a novel surgical approach for patients with progressive familial intrahepatic cholestasis: a preliminary report. J Pediatr Surg. 2007;42:1337-1340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 56] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 27. | Diao M, Li L, Zhang JS, Ye M, Cheng W. Laparoscopic cholecystocolostomy: a novel surgical approach for the treatment of progressive familial intrahepatic cholestasis. Ann Surg. 2013;258:1028-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 28. | Jankowska I, Czubkowski P, Kaliciński P, Ismail H, Kowalski A, Ryżko J, Pawłowska J. Ileal exclusion in children with progressive familial intrahepatic cholestasis. J Pediatr Gastroenterol Nutr. 2014;58:92-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 29. | Kaliciński PJ, Ismail H, Jankowska I, Kamiński A, Pawłowska J, Drewniak T, Markiewicz M, Szymczak M. Surgical treatment of progressive familial intrahepatic cholestasis: comparison of partial external biliary diversion and ileal bypass. Eur J Pediatr Surg. 2003;13:307-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 79] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Ramachandran P, Shanmugam NP, Sinani SA, Shanmugam V, Srinivas S, Sathiyasekaran M, Tamilvanan V, Rela M. Outcome of partial internal biliary diversion for intractable pruritus in children with cholestatic liver disease. Pediatr Surg Int. 2014;30:1045-1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 31. | Gün F, Erginel B, Durmaz O, Sökücü S, Salman T, Celik A. An outstanding non-transplant surgical intervention in progressive familial intrahepatic cholestasis: partial internal biliary diversion. Pediatr Surg Int. 2010;26:831-834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 32. | Mochizuki K, Obatake M, Takatsuki M, Nakatomi A, Hayashi T, Okudaira S, Eguchi S. Partial internal biliary diversion for patients with progressive familial intrahepatic cholestasis type 1. Pediatr Surg Int. 2012;28:51-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 33. | Cutillo L, Najimi M, Smets F, Janssen M, Reding R, de Ville de Goyet J, Sokal EM. Safety of living-related liver transplantation for progressive familial intrahepatic cholestasis. Pediatr Transplant. 2006;10:570-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 34. | Hori T, Egawa H, Takada Y, Ueda M, Oike F, Ogura Y, Sakamoto S, Kasahara M, Ogawa K, Miyagawa-Hayashino A. Progressive familial intrahepatic cholestasis: a single-center experience of living-donor liver transplantation during two decades in Japan. Clin Transplant. 2011;25:776-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 35. | Egawa H, Yorifuji T, Sumazaki R, Kimura A, Hasegawa M, Tanaka K. Intractable diarrhea after liver transplantation for Byler’s disease: successful treatment with bile adsorptive resin. Liver Transpl. 2002;8:714-716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 61] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 36. | van der Mark VA, de Waart DR, Ho-Mok KS, Tabbers MM, Voogt HW, Oude Elferink RP, Knisely AS, Paulusma CC. The lipid flippase heterodimer ATP8B1-CDC50A is essential for surface expression of the apical sodium-dependent bile acid transporter (SLC10A2/ASBT) in intestinal Caco-2 cells. Biochim Biophys Acta. 2014;1842:2378-2386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 2.8] [Reference Citation Analysis (2)] |
| 37. | Miyagawa-Hayashino A, Egawa H, Yorifuji T, Hasegawa M, Haga H, Tsuruyama T, Wen MC, Sumazaki R, Manabe T, Uemoto S. Allograft steatohepatitis in progressive familial intrahepatic cholestasis type 1 after living donor liver transplantation. Liver Transpl. 2009;15:610-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 61] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 38. | Berumen J, Feinberg E, Todo T, Bonham CA, Concepcion W, Esquivel C. Complications following liver transplantation for progressive familial intrahepatic cholestasis. Dig Dis Sci. 2014;59:2649-2652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 39. | Keitel V, Burdelski M, Vojnisek Z, Schmitt L, Häussinger D, Kubitz R. De novo bile salt transporter antibodies as a possible cause of recurrent graft failure after liver transplantation: a novel mechanism of cholestasis. Hepatology. 2009;50:510-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 84] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 40. | Siebold L, Dick AA, Thompson R, Maggiore G, Jacquemin E, Jaffe R, Strautnieks S, Grammatikopoulos T, Horslen S, Whitington PF. Recurrent low gamma-glutamyl transpeptidase cholestasis following liver transplantation for bile salt export pump (BSEP) disease (posttransplant recurrent BSEP disease). Liver Transpl. 2010;16:856-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 41. | Kubitz R, Dröge C, Kluge S, Stross C, Walter N, Keitel V, Häussinger D, Stindt J. Autoimmune BSEP disease: disease recurrence after liver transplantation for progressive familial intrahepatic cholestasis. Clin Rev Allergy Immunol. 2015;48:273-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 42. | van der Woerd WL, Kokke FT, van der Zee DC, Houwen RH. Total biliary diversion as a treatment option for patients with progressive familial intrahepatic cholestasis and Alagille syndrome. J Pediatr Surg. 2015;50:1846-1849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 43. | van der Woerd WL, Mulder J, Pagani F, Beuers U, Houwen RH, van de Graaf SF. Analysis of aberrant pre-messenger RNA splicing resulting from mutations in ATP8B1 and efficient in vitro rescue by adapted U1 small nuclear RNA. Hepatology. 2015;61:1382-1391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 44. | Zhou H, Hylemon PB. Bile acids are nutrient signaling hormones. Steroids. 2014;86:62-68. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 257] [Cited by in RCA: 217] [Article Influence: 18.1] [Reference Citation Analysis (3)] |
| 45. | Usui M, Isaji S, Das BC, Kobayashi M, Osawa I, Iida T, Sakurai H, Tabata M, Yorifuji T, Egawa H. Liver retransplantation with external biliary diversion for progressive familial intrahepatic cholestasis type 1: a case report. Pediatr Transplant. 2009;13:611-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 46. | Nicastro E, Stephenne X, Smets F, Fusaro F, de Magnée C, Reding R, Sokal EM. Recovery of graft steatosis and protein-losing enteropathy after biliary diversion in a PFIC 1 liver transplanted child. Pediatr Transplant. 2012;16:E177-E182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 47. | Mali VP, Fukuda A, Shigeta T, Uchida H, Hirata Y, Rahayatri TH, Kanazawa H, Sasaki K, de Ville de Goyet J, Kasahara M. Total internal biliary diversion during liver transplantation for type 1 progressive familial intrahepatic cholestasis: a novel approach. Pediatr Transplant. 2016;20:981-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 48. | Miethke AG, Zhang W, Simmons J, Taylor AE, Shi T, Shanmukhappa SK, Karns R, White S, Jegga AG, Lages CS. Pharmacological inhibition of apical sodium-dependent bile acid transporter changes bile composition and blocks progression of sclerosing cholangitis in multidrug resistance 2 knockout mice. Hepatology. 2016;63:512-523. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 120] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 49. | Baghdasaryan A, Fuchs CD, Österreicher CH, Lemberger UJ, Halilbasic E, Påhlman I, Graffner H, Krones E, Fickert P, Wahlström A. Inhibition of intestinal bile acid absorption improves cholestatic liver and bile duct injury in a mouse model of sclerosing cholangitis. J Hepatol. 2016;64:674-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 50. | Soroka CJ, Mennone A, Hagey LR, Ballatori N, Boyer JL. Mouse organic solute transporter alpha deficiency enhances renal excretion of bile acids and attenuates cholestasis. Hepatology. 2010;51:181-190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 51. | Rao A, Haywood J, Craddock AL, Belinsky MG, Kruh GD, Dawson PA. The organic solute transporter alpha-beta, Ostalpha-Ostbeta, is essential for intestinal bile acid transport and homeostasis. Proc Natl Acad Sci USA. 2008;105:3891-3896. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 174] [Article Influence: 9.7] [Reference Citation Analysis (3)] |
| 52. | Zhou M, Wang X, Phung V, Lindhout DA, Mondal K, Hsu JY, Yang H, Humphrey M, Ding X, Arora T. Separating Tumorigenicity from Bile Acid Regulatory Activity for Endocrine Hormone FGF19. Cancer Res. 2014;74:3306-3316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 162] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 53. | Luo J, Ko B, Elliott M, Zhou M, Lindhout DA, Phung V, To C, Learned RM, Tian H, DePaoli AM. A nontumorigenic variant of FGF19 treats cholestatic liver diseases. Sci Transl Med. 2014;6:247ra100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 135] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 54. | Zhou M, Learned RM, Rossi SJ, DePaoli AM, Tian H, Ling L. Engineered fibroblast growth factor 19 reduces liver injury and resolves sclerosing cholangitis in Mdr2-deficient mice. Hepatology. 2016;63:914-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 112] [Article Influence: 11.2] [Reference Citation Analysis (2)] |
| 55. | Blank A, Markert C, Hohmann N, Carls A, Mikus G, Lehr T, Alexandrov A, Haag M, Schwab M, Urban S. First-in-human application of the novel hepatitis B and hepatitis D virus entry inhibitor myrcludex B. J Hepatol. 2016;65:483-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 182] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 56. | Bogomolov P, Alexandrov A, Voronkova N, Macievich M, Kokina K, Petrachenkova M, Lehr T, Lempp FA, Wedemeyer H, Haag M. Treatment of chronic hepatitis D with the entry inhibitor myrcludex B: First results of a phase Ib/IIa study. J Hepatol. 2016;65:490-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 310] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 57. | Yan H, Zhong G, Xu G, He W, Jing Z, Gao Z, Huang Y, Qi Y, Peng B, Wang H. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. Elife. 2012;1:e00049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1734] [Cited by in RCA: 1660] [Article Influence: 118.6] [Reference Citation Analysis (0)] |
| 58. | Ni Y, Lempp FA, Mehrle S, Nkongolo S, Kaufman C, Fälth M, Stindt J, Königer C, Nassal M, Kubitz R. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology. 2014;146:1070-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 703] [Cited by in RCA: 647] [Article Influence: 53.9] [Reference Citation Analysis (0)] |
| 59. | Vaz FM, Paulusma CC, Huidekoper H, de Ru M, Lim C, Koster J, Ho-Mok K, Bootsma AH, Groen AK, Schaap FG. Sodium taurocholate cotransporting polypeptide (SLC10A1) deficiency: conjugated hypercholanemia without a clear clinical phenotype. Hepatology. 2015;61:260-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 169] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 60. | Slijepcevic D, Kaufman C, Wichers CG, Gilglioni EH, Lempp FA, Duijst S, de Waart DR, Elferink RP, Mier W, Stieger B. Impaired uptake of conjugated bile acids and hepatitis b virus pres1-binding in na(+) -taurocholate cotransporting polypeptide knockout mice. Hepatology. 2015;62:207-219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 115] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 61. | Dhawan A, Mitry RR, Hughes RD. Hepatocyte transplantation for liver-based metabolic disorders. J Inherit Metab Dis. 2006;29:431-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 136] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 62. | Mitry RR, Hughes RD, Dhawan A. Hepatocyte transplantation. J Clin Exp Hepatol. 2011;1:109-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 63. | Sanal MG. Cell therapy from bench to bedside: Hepatocytes from fibroblasts - the truth and myth of transdifferentiation. World J Gastroenterol. 2015;21:6427-6433. [PubMed] [DOI] [Full Text] |
| 64. | Takebe T, Sekine K, Enomura M, Koike H, Kimura M, Ogaeri T, Zhang RR, Ueno Y, Zheng YW, Koike N. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature. 2013;499:481-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1915] [Cited by in RCA: 1574] [Article Influence: 121.1] [Reference Citation Analysis (3)] |
| 65. | Huch M, Dorrell C, Boj SF, van Es JH, Li VS, van de Wetering M, Sato T, Hamer K, Sasaki N, Finegold MJ. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature. 2013;494:247-250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1394] [Cited by in RCA: 1223] [Article Influence: 94.1] [Reference Citation Analysis (4)] |
| 66. | Huch M, Boj SF, Clevers H. Lgr5(+) liver stem cells, hepatic organoids and regenerative medicine. Regen Med. 2013;8:385-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 73] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 67. | Huch M, Gehart H, van Boxtel R, Hamer K, Blokzijl F, Verstegen MM, Ellis E, van Wenum M, Fuchs SA, de Ligt J. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell. 2015;160:299-312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1424] [Cited by in RCA: 1224] [Article Influence: 111.3] [Reference Citation Analysis (5)] |
| 68. | Aravalli RN, Belcher JD, Steer CJ. Liver-targeted gene therapy: Approaches and challenges. Liver Transpl. 2015;21:718-737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 69. | Wang L, Wu J, Fang W, Liu GH, Izpisua Belmonte JC. Regenerative medicine: targeted genome editing in vivo. Cell Res. 2015;25:271-272. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 70. | Riordan JR, Rommens JM, Kerem B, Alon N, Rozmahel R, Grzelczak Z, Zielenski J, Lok S, Plavsic N, Chou JL. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science. 1989;245:1066-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5809] [Cited by in RCA: 5135] [Article Influence: 138.8] [Reference Citation Analysis (2)] |
| 71. | Klomp LW, Vargas JC, van Mil SW, Pawlikowska L, Strautnieks SS, van Eijk MJ, Juijn JA, Pabón-Peña C, Smith LB, DeYoung JA. Characterization of mutations in ATP8B1 associated with hereditary cholestasis. Hepatology. 2004;40:27-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 187] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 72. | Strautnieks SS, Byrne JA, Pawlikowska L, Cebecauerová D, Rayner A, Dutton L, Meier Y, Antoniou A, Stieger B, Arnell H. Severe bile salt export pump deficiency: 82 different ABCB11 mutations in 109 families. Gastroenterology. 2008;134:1203-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 268] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 73. | Degiorgio D, Colombo C, Seia M, Porcaro L, Costantino L, Zazzeron L, Bordo D, Coviello DA. Molecular characterization and structural implications of 25 new ABCB4 mutations in progressive familial intrahepatic cholestasis type 3 (PFIC3). Eur J Hum Genet. 2007;15:1230-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 74] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 74. | Ikpa PT, Bijvelds MJ, de Jonge HR. Cystic fibrosis: toward personalized therapies. Int J Biochem Cell Biol. 2014;52:192-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 57] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 75. | Amaral MD. Novel personalized therapies for cystic fibrosis: treating the basic defect in all patients. J Intern Med. 2015;277:155-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 66] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 76. | Hayashi H, Takada T, Suzuki H, Akita H, Sugiyama Y. Two common PFIC2 mutations are associated with the impaired membrane trafficking of BSEP/ABCB11. Hepatology. 2005;41:916-924. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 98] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 77. | Hayashi H, Sugiyama Y. 4-phenylbutyrate enhances the cell surface expression and the transport capacity of wild-type and mutated bile salt export pumps. Hepatology. 2007;45:1506-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 139] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 78. | Byrne JA, Strautnieks SS, Ihrke G, Pagani F, Knisely AS, Linton KJ, Mieli-Vergani G, Thompson RJ. Missense mutations and single nucleotide polymorphisms in ABCB11 impair bile salt export pump processing and function or disrupt pre-messenger RNA splicing. Hepatology. 2009;49:553-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 130] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 79. | van der Velden LM, Stapelbroek JM, Krieger E, van den Berghe PV, Berger R, Verhulst PM, Holthuis JC, Houwen RH, Klomp LW, van de Graaf SF. Folding defects in P-type ATP 8B1 associated with hereditary cholestasis are ameliorated by 4-phenylbutyrate. Hepatology. 2010;51:286-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 80. | Gautherot J, Durand-Schneider AM, Delautier D, Delaunay JL, Rada A, Gabillet J, Housset C, Maurice M, Aït-Slimane T. Effects of cellular, chemical, and pharmacological chaperones on the rescue of a trafficking-defective mutant of the ATP-binding cassette transporter proteins ABCB1/ABCB4. J Biol Chem. 2012;287:5070-5078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 81. | Dröge C, Schaal H, Engelmann G, Wenning D, Häussinger D, Kubitz R. Exon-skipping and mRNA decay in human liver tissue: molecular consequences of pathogenic bile salt export pump mutations. Sci Rep. 2016;6:24827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 82. | Delaunay JL, Durand-Schneider AM, Dossier C, Falguières T, Gautherot J, Davit-Spraul A, Aït-Slimane T, Housset C, Jacquemin E, Maurice M. A functional classification of ABCB4 variations causing progressive familial intrahepatic cholestasis type 3. Hepatology. 2016;63:1620-1631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 82] [Article Influence: 8.2] [Reference Citation Analysis (2)] |
| 83. | Welch EM, Barton ER, Zhuo J, Tomizawa Y, Friesen WJ, Trifillis P, Paushkin S, Patel M, Trotta CR, Hwang S. PTC124 targets genetic disorders caused by nonsense mutations. Nature. 2007;447:87-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 982] [Cited by in RCA: 869] [Article Influence: 45.7] [Reference Citation Analysis (0)] |
| 84. | Kerem E, Konstan MW, De Boeck K, Accurso FJ, Sermet-Gaudelus I, Wilschanski M, Elborn JS, Melotti P, Bronsveld I, Fajac I. Ataluren for the treatment of nonsense-mutation cystic fibrosis: a randomised, double-blind, placebo-controlled phase 3 trial. Lancet Respir Med. 2014;2:539-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 280] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 85. | Bushby K, Finkel R, Wong B, Barohn R, Campbell C, Comi GP, Connolly AM, Day JW, Flanigan KM, Goemans N. Ataluren treatment of patients with nonsense mutation dystrophinopathy. Muscle Nerve. 2014;50:477-487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 350] [Cited by in RCA: 319] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 86. | Fernandez Alanis E, Pinotti M, Dal Mas A, Balestra D, Cavallari N, Rogalska ME, Bernardi F, Pagani F. An exon-specific U1 small nuclear RNA (snRNA) strategy to correct splicing defects. Hum Mol Genet. 2012;21:2389-2398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 93] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 87. | Rogalska ME, Tajnik M, Licastro D, Bussani E, Camparini L, Mattioli C, Pagani F. Therapeutic activity of modified U1 core spliceosomal particles. Nat Commun. 2016;7:11168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 88. | Balestra D, Faella A, Margaritis P, Cavallari N, Pagani F, Bernardi F, Arruda VR, Pinotti M. An engineered U1 small nuclear RNA rescues splicing-defective coagulation F7 gene expression in mice. J Thromb Haemost. 2014;12:177-185. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 89. | Dal Mas A, Rogalska ME, Bussani E, Pagani F. Improvement of SMN2 pre-mRNA processing mediated by exon-specific U1 small nuclear RNA. Am J Hum Genet. 2015;96:93-103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 90. | Balch WE, Morimoto RI, Dillin A, Kelly JW. Adapting proteostasis for disease intervention. Science. 2008;319:916-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2215] [Cited by in RCA: 1953] [Article Influence: 108.5] [Reference Citation Analysis (0)] |
| 91. | Delaunay JL, Durand-Schneider AM, Delautier D, Rada A, Gautherot J, Jacquemin E, Aït-Slimane T, Maurice M. A missense mutation in ABCB4 gene involved in progressive familial intrahepatic cholestasis type 3 leads to a folding defect that can be rescued by low temperature. Hepatology. 2009;49:1218-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 55] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 92. | Gordo-Gilart R, Andueza S, Hierro L, Jara P, Alvarez L. Functional Rescue of Trafficking-Impaired ABCB4 Mutants by Chemical Chaperones. PLoS One. 2016;11:e0150098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 93. | Hasegawa Y, Hayashi H, Naoi S, Kondou H, Bessho K, Igarashi K, Hanada K, Nakao K, Kimura T, Konishi A. Intractable itch relieved by 4-phenylbutyrate therapy in patients with progressive familial intrahepatic cholestasis type 1. Orphanet J Rare Dis. 2014;9:89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 94. | Gonzales E, Grosse B, Cassio D, Davit-Spraul A, Fabre M, Jacquemin E. Successful mutation-specific chaperone therapy with 4-phenylbutyrate in a child with progressive familial intrahepatic cholestasis type 2. J Hepatol. 2012;57:695-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 95. | Naoi S, Hayashi H, Inoue T, Tanikawa K, Igarashi K, Nagasaka H, Kage M, Takikawa H, Sugiyama Y, Inui A. Improved liver function and relieved pruritus after 4-phenylbutyrate therapy in a patient with progressive familial intrahepatic cholestasis type 2. J Pediatr. 2014;164:1219-1227.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 96. | Gonzales E, Grosse B, Schuller B, Davit-Spraul A, Conti F, Guettier C, Cassio D, Jacquemin E. Targeted pharmacotherapy in progressive familial intrahepatic cholestasis type 2: Evidence for improvement of cholestasis with 4-phenylbutyrate. Hepatology. 2015;62:558-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 84] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 97. | Hayashi H, Naoi S, Hirose Y, Matsuzaka Y, Tanikawa K, Igarashi K, Nagasaka H, Kage M, Inui A, Kusuhara H. Successful treatment with 4-phenylbutyrate in a patient with benign recurrent intrahepatic cholestasis type 2 refractory to biliary drainage and bilirubin absorption. Hepatol Res. 2016;46:192-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 98. | van der Woerd WL, Wichers CG, Vestergaard AL, Andersen JP, Paulusma CC, Houwen RH, van de Graaf SF. Rescue of defective ATP8B1 trafficking by CFTR correctors as a therapeutic strategy for familial intrahepatic cholestasis. J Hepatol. 2016;64:1339-1347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 99. | Clancy JP, Rowe SM, Accurso FJ, Aitken ML, Amin RS, Ashlock MA, Ballmann M, Boyle MP, Bronsveld I, Campbell PW. Results of a phase IIa study of VX-809, an investigational CFTR corrector compound, in subjects with cystic fibrosis homozygous for the F508del-CFTR mutation. Thorax. 2012;67:12-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 431] [Cited by in RCA: 399] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 100. | Paulusma CC, Folmer DE, Ho-Mok KS, de Waart DR, Hilarius PM, Verhoeven AJ, Oude Elferink RP. ATP8B1 requires an accessory protein for endoplasmic reticulum exit and plasma membrane lipid flippase activity. Hepatology. 2008;47:268-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 184] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 101. | Cai SY, Gautam S, Nguyen T, Soroka CJ, Rahner C, Boyer JL. ATP8B1 deficiency disrupts the bile canalicular membrane bilayer structure in hepatocytes, but FXR expression and activity are maintained. Gastroenterology. 2009;136:1060-1069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 78] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 102. | Stone A, Chau C, Eaton C, Foran E, Kapur M, Prevatt E, Belkin N, Kerr D, Kohlin T, Williamson P. Biochemical characterization of P4-ATPase mutations identified in patients with progressive familial intrahepatic cholestasis. J Biol Chem. 2012;287:41139-41151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 103. | Dekkers JF, Berkers G, Kruisselbrink E, Vonk A, de Jonge HR, Janssens HM, Bronsveld I, van de Graaf EA, Nieuwenhuis EE, Houwen RH. Characterizing responses to CFTR-modulating drugs using rectal organoids derived from subjects with cystic fibrosis. Sci Transl Med. 2016;8:344ra84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 433] [Article Influence: 43.3] [Reference Citation Analysis (3)] |
| 104. | Duan N, Kravitz RL, Schmid CH. Single-patient (n-of-1) trials: a pragmatic clinical decision methodology for patient-centered comparative effectiveness research. J Clin Epidemiol. 2013;66:S21-S28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 159] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: The Netherlands
Peer-review report classification
Grade A (Excellent): A, A
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Delaunay JL, Kubitz R, Lee HC, Wang JS S- Editor: Gong ZM L- Editor: A E- Editor: Zhang FF