Published online Mar 14, 2017. doi: 10.3748/wjg.v23.i10.1796
Peer-review started: December 2, 2016
First decision: December 19, 2016
Revised: January 6, 2017
Accepted: January 18, 2017
Article in press: January 18, 2017
Published online: March 14, 2017
Processing time: 102 Days and 20.8 Hours
To explore the anti-tumor effects of esophageal cancer-related gene 2 (ECRG2) in combination with cisplatin (DDP) in DDP-resistant esophageal cancer cells (EC9706/DDP).
A drug-resistant cell model was established, with EC9706/DDP cells being treated with ECRG2 and/or DDP. Cell viability was examined by MTT assay. The rate of cell apoptosis was determined by flow cytometry. The mRNA expression levels of proliferating cell nuclear antigen (PCNA), metallothionein (MT), and p53 were determined by RT-PCR and PCNA, while MT and p53 protein expression levels were determined by western blotting.
The anti-proliferative effect of ECRG2 in combination with DDP was superior when compared to ECRG2 or DDP alone. The inhibition rate for the combination reached its peak (51.33%) at 96 h. The early apoptotic rates of the control, ECRG2 alone, DDP alone, and ECRG2 plus DDP groups were 5.71% ± 0.27%, 12.68% ± 0.61%, 14.15% ± 0.87%, and 27.96% ± 0.36%, respectively. Although all treatment groups were significantly different from the control group (P < 0.05), the combination treatment of ECRG2 plus DDP performed significantly better when compared to either ECRG2 or DDP alone (P < 0.05). The combination of ECRG2 and DDP significantly upregulated p53 mRNA and protein levels and downregulated PCNA mRNA and protein levels compared to ECRG2 or DDP alone (P < 0.05). However, no changes were seen in the expression of MT mRNA or protein.
ECRG2 in combination with DDP can inhibit viability and induce apoptosis in esophageal cancer DDP-resistant cells, possibly via upregulation of p53 expression and downregulation of PCNA expression. These findings suggest that the combination of ECRG2 and DDP may be a promising strategy for the clinical treatment of esophageal cancers that are resistant to DDP.
Core tip: Cisplatin (DDP) is one of the most effective agents in treating esophageal cancer, but drug resistance poses a major impediment. Combination therapy has become an important method for overcoming drug resistance. This study showed that esophageal cancer-related gene 2 (ECRG2) in combination with DDP can inhibit viability and induce apoptosis in DDP-resistant esophageal cancer cells, possibly via upregulation of p53 expression and downregulation of proliferating cell nuclear antigen expression. These findings suggest that the combination of ECRG2 and DDP may be a promising strategy for the clinical treatment of esophageal cancers that are resistant to DDP.
- Citation: Hou XF, Xu LP, Song HY, Li S, Wu C, Wang JF. ECRG2 enhances the anti-cancer effects of cisplatin in cisplatin-resistant esophageal cancer cells via upregulation of p53 and downregulation of PCNA. World J Gastroenterol 2017; 23(10): 1796-1803
- URL: https://www.wjgnet.com/1007-9327/full/v23/i10/1796.htm
- DOI: https://dx.doi.org/10.3748/wjg.v23.i10.1796
Core tip: Cisplatin (DDP) is one of the most effective agents in treating esophageal cancer, but drug resistance poses a major impediment. Combination therapy has become an important method for overcoming drug resistance. This study showed that esophageal cancer-related gene 2 (ECRG2) in combination with DDP can inhibit viability and induce apoptosis in DDP-resistant esophageal cancer cells, possibly via upregulation of p53 expression and downregulation of proliferating cell nuclear antigen expression. These findings suggest that the combination of ECRG2 and DDP may be a promising strategy for the clinical treatment of esophageal cancers that are resistant to DDP.
Esophageal cancer is a severe global health problem with an incidence that ranks eighth among all of the malignant cancers[1]. In spite of improvements in early-stage detection, most tumors are already at an advanced stage upon initial diagnosis[2]. Cisplatin (DDP)-based doublets have been the most commonly used treatment regimen for most patients with locally advanced or metastatic esophageal cancer[3-6]. Unfortunately, DDP resistance and dose-limiting side effects have been a challenge to any further improvement of this treatment’s efficacy. These obstacles call for a novel combination of therapeutic approaches in order to achieve high therapeutic efficacy at a lower drug dose[7].
Esophageal cancer related gene 2 (ECRG2) was first separated and identified, using the mRNA technique of differential display, in normal esophageal tissues and esophageal cancer tissues in 1998[8]. It has been reported that ECRG2 inhibits tumor cell growth and promotes cell apoptosis in vivo and in vitro[9,10]. In our previous study, we found that ECRG2 combined with DDP had an enhanced inhibitory effect on EC9706 cell proliferation and an increased inductive effect on EC9706 cell apoptosis when compared to ECRG2 alone[11]. This suggests that ECRG2 may be involved in the drug resistance of esophageal cancer. There are currently no known studies in the literature examining the effects of ECRG2 on the drug resistance of esophageal cancer. In the present study, we established a drug-resistant cell model to explore the effects of ECRG2 on drug resistant cells.
PCNA, p53, and MT primers and ECRG2 protein were manufactured by Sangon Biological Engineering Co., Ltd. (Shanghai, China). DDP was bought from Qilu Pharmaceutical Co., Ltd. (Jinan, China). RPMI 1640 medium and fetal bovine serum (FBS) were purchased from HyClone (Logan, UT, United States). 3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT), TRIzol, and annexin V-FITC apoptosis detection kits were purchased from Sigma Co. (St. Louis, MO, United States). The reverse transcription and amplification kits were purchased from Promega Co. (Madison, WI, United States). The rabbit anti-human PCNA and p53 antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, United States). The rabbit anti-human MT and goat anti-rabbit IgG antibodies were purchased from Abcam (Cambridge, United Kingdom). The human esophageal cancer cell line EC9706 was provided by the tumor cell library of the Academy of Medical Sciences in China.
We shocked EC9706 cells in the logarithmic phase with a final concentration of 2 mg/L of DDP. We discarded the culture solution after 48 h, washing 3 times with PBS. The cells were cultured in a fresh nutrient solution and washed every 2 d to remove dead cells. When the cells reached stable growth, we increased the DDP concentration, and repeated until the cells maintained growth in a medium with a concentration of 0.5 mg/L of DDP. The cells were cultured in a RPMI-1640 medium containing 10% fetal bovine serum, 100 U/m penicillin, 100 U/mL streptomycin, and 0.5 mL/L DDP in a humidified incubator at 37 °C with 5% CO2.
Drug sensitivity was analyzed by MTT assay. The cells were plated at 1 × 103 cells/mL in 96-well plates in a volume of 100 μL and incubated for 24 h. The media were discarded and cells were treated with various concentrations of DDP or ECRG2 for 72 h. A total of 20 μL of 5 g/L MTT solution was added to each well, with cells then being incubated for 4 h at 37 °C. The medium was discarded and 150 μL of dimethyl sulfoxide was added to each well, which was then shaken to completely dissolve the blue-purple precipitate from the MTT. A microplate reader was used to test the absorbance (OD) of each well at 490 nm and average values were obtained. Cell viability was calculated using the formula: (OD treated well-OD blank)/(OD control well-OD blank) × 100.
The cells were seeded in a 6-well plate at a density of 4 × 105 cells per well. After overnight attachment, cells were treated with ECRG2 and/or DDP for 24 h. The cells were then harvested by trypsinization, washed with cold PBS, centrifuged at 1 × 103 rpm for 5 min, and resuspended in 1 × binding buffer. 500 μL of each sample solution was then added to 5 μL of FITC-conjugated annexin-V and 10 μL of PI, followed by incubation for 15 min in the dark at room temperature. The samples were then measured by flow cytometry.
Each group of cells was collected after 24 h of drug treatment. The total RNA was extracted from each group of cells using TRIzol reagent according to the manufacturer’s protocol. The cDNA was made via the protocol for the Reverse Transcription System for RT-PCR. The primers for PCNA were 5’-ACCGCTGCGACCGCAATTTG-3’(forward) and 5’-ACGTGCAAATTCACCAGAAGGCATC-3’(reverse). The primers for MT were 5’-CGGATCCAGACTCAAACAGGCTTTTAT-3’ (forward) and 5’-CGGATCCCGGAATGGACCCCAACTGCT-3’ (reverse). The primers for p53 were 5’-ATTTGCGTGTGGAGTATTTGG-3’ (forward) and 5’-GCTGTTCCGTCCCAGTAGATTA-3’ (reverse). The primers for GAPDH were 5’-TCATGGGTGTGAACCATGAGAA-3’ (forward) and 5’-GGCATGGACTGTGGTCATGAG-3’ (reverse). PCR amplification was performed using a kit under the following conditions: 95 °C for 5 min, followed by 30 cycles at 95 °C for 40 s, 55 °C for 40 s, and 72 °C for 1 min. The expression level of mRNA encoding the p53, PCNA, and MT genes were evaluated and normalized relative to that of GAPDH.
The cells were collected from each group and lysed in 200 μL of 1% Tween 20 in PBS by five cycles of freezing and thawing. The insoluble proteins were removed by centrifugation. After quantification of the total protein, the protein was separated on 8% polyacrylamide gel (PAGE), and transferred to a PVDF membrane (Millipore). The membranes were then blocked with 5% non-fat dry milk in PBS-Tween 20 for 1 h at room temperature. After incubation with polyclonal rabbit anti-human MT, PCNA, or p53 antibody overnight at 4 °C at a 1:500 dilution, the membranes were incubated with a secondary HRP-conjugated anti-rabbit antibody at a 1:1000 dilution and then washed again three times with PBST buffer. The transferred proteins were visualized using an ECL detection kit according to the manufacturer’s protocol.
The SPSS 13.0 software system (SPSS, Inc., Chicago, IL, United States) was used for statistical evaluation. The data are expressed as the mean ± SD for at least 3 independent experiments. Statistical significance was evaluated by analysis of variance (ANOVA) with Tukey’s post hoc test. A P value of < 0.05 was considered to indicate a statistically significant result.
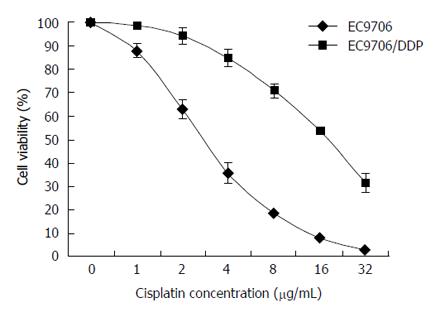
The EC9706/DDP cell line was established after several months of intermittent exposure by the parental cells to a gradually increasing concentration of drugs. After the EC9706 and EC9706/DDP cells were treated with DDP at various concentrations (1, 2, 4, 8, 16, 32 μg/mL) for 48 h, the cell viability rate was examined by MTT assay. As shown in Figure 1, the IC50 for 48 h of DDP in the EC9706/DDP cells was 16.83 ± 1.46 μg/mL and the IC50 for 48 h of DDP in the parental EC9706 cells was 3.34 ± 0.27 μg/mL. Therefore, the EC9706/DDP cells were five times more resistant to DDP compared to the parental cells.
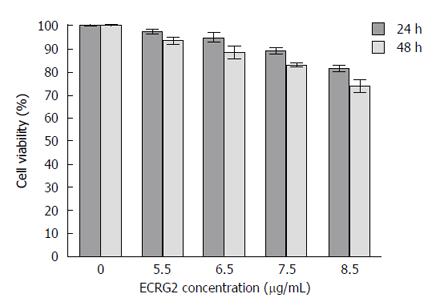
To evaluate the effects of ECRG2 on the growth and viability of the cells, the EC9706/DDP cells were treated with various concentrations of ECRG2 (5.5, 6.5, 7.5, 8.5 μg/mL). The growth rate of the cells was found to be inhibited in a dose- and time- dependent manner by the ECRG2 protein (Figure 2).
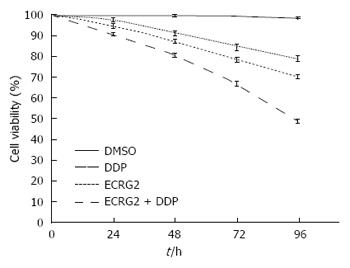
Based on the results above, we used 3 μg/mL of DDP and 6.5 μg/mL of ECRG2 for 48 h in EC9706/DDP cells for further experiments. As shown in Figure 3, the anti-proliferative effect of ECRG2 in combination with DDP was better compared to ECRG2 or DDP alone. These results suggest that a combination treatment of ECRG2 and DDP resulted in a synergistic decrease in cell viability compared to each treatment alone. The inhibition rate for the combination reached its peak (51.33%) at 96 h (Figure 3).
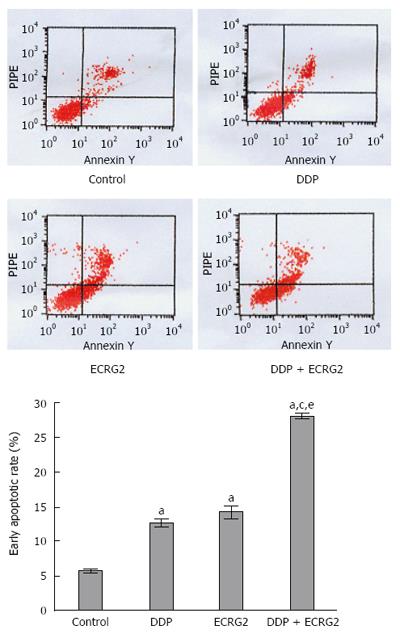
We next examined the effect of ECRG2 and DDP on cell apoptosis by performing annexin V/PI staining. The early apoptotic rates of the control, ECRG2 alone, DDP alone, and ECRG2 plus DDP groups were 5.71% ± 0.27%, 12.68% ± 0.61%, 14.15% ± 0.87%, and 27.96% ± 0.36%, respectively. Although all treatment groups were significantly different from the control group (P < 0.05), the combination treatment of ECRG2 plus DDP performed significantly better when compared to either ECRG2 or DDP alone (P < 0.05) (Figure 4).
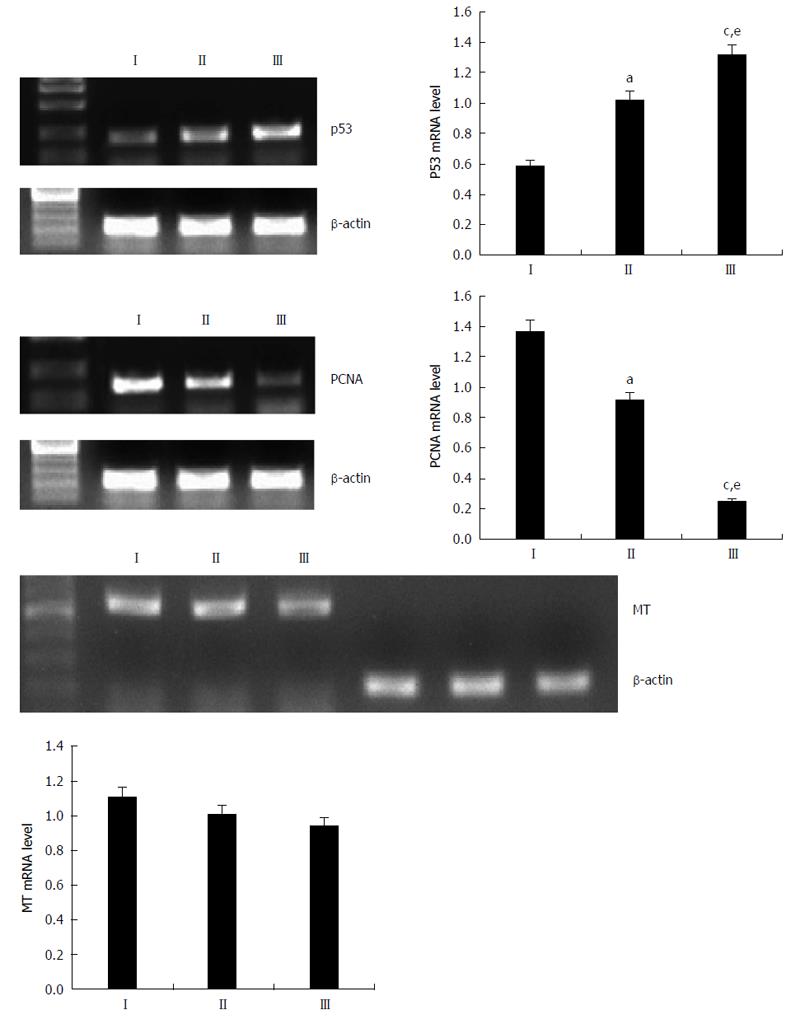
To further elucidate the mechanisms involved in ECRG2 in combination with DDP on the anti-proliferative effects in drug-resistant cells, the expression of p53, PCNA, and MT mRNA were evaluated in cells using RT-PCR analysis. As shown in Figure 5, 3 μg/mL of DDP upregulated the expression of p53 and downregulated the expression of PCNA (P < 0.05). When ECRG2 protein was combined with DDP, p53 mRNA expression significantly increased and PCNA mRNA expression decreased compared to DDP alone (P < 0.05). However, changes in the expression of MT mRNA were not observed (Figure 5).
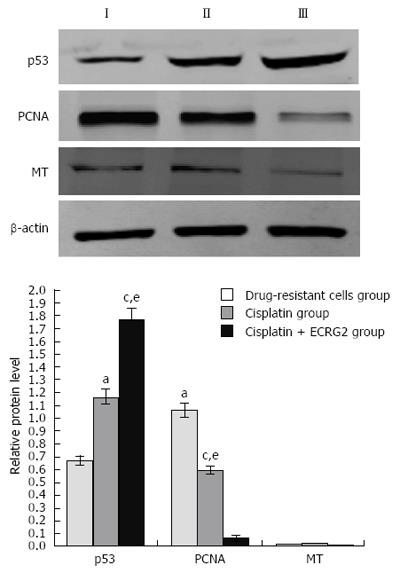
As shown in Figure 6, the results of the western blot were consistent with RT-PCR; 3 μg/mL of DDP upregulated the expression of p53 protein and downregulated the expression of PCNA protein. When ECRG2 protein was combined with DDP, expression of p53 protein was significantly increased and expression of PCNA protein was decreased compared to DDP alone (P < 0.05). However, no changes were seen in the expression of MT protein (Figure 6).
DDP is one of the most effective anti-cancer agents widely used in the treatment of solid tumors, including esophageal cancer. Despite a consistent rate of initial response, DDP treatment often results in the development of chemoresistance, leading to therapeutic failure. Combination therapy has become an important method of overcoming drug resistance. There have been many attempts to overcome DDP resistance in esophageal cancer cells. Lee et al[12] reported that the novel oxygen carrier YQ23 enhanced the DDP response of chemoresistant esophageal tumor xenografts, as well as reducing tumor size and the number of animals with invasive tumors. Lai et al[13] provided evidence that 14-3-3σ, or its related DNA repair molecule, is a promising therapeutic target for counteracting DDP resistance in esophageal squamous cell carcinoma. Furthermore, Yu et al[14] suggested autophagy is a novel target to improve the efficiency of DDP in human esophageal cancers with acquired resistance.
In this study, we first established an EC9706/DDP cell line derived from in vitro treatment of sensitive cells as a model to examine the effect of ECRG2 on DDP resistance. The results demonstrated that a combination of ECRG2 and DDP had synergistic inhibitory effects on the proliferation of EC9706/DDP cells. The inhibition rate was significantly higher in EC9706/DDP cells treated with ECRG2 and DDP compared to cells treated with ECRG2 or DDP alone. This suggests ECRG2 may reduce or inhibit DDP resistance.
The mechanism through which cancer cells develop resistance to chemotherapy is associated with increased resistance to apoptosis. Agents that induce apoptosis have been reported to sensitize tumor cells to DDP[15]. Cui et al[9,10] reported that ECRG2 promoted cell apoptosis in vivo and in vitro. Here, we provided experimental evidence showing the combination of ECRG2 and DDP promoted the apoptosis of esophageal DDP-resistant cancer cells. A study by Li et al[16] showed that ECRG2-overexpressing cells had a higher expression of p53. We therefore compared the p53 expression levels of the control, DDP alone, and ECRG2 plus DDP groups. Our results proved that p53 was involved in this process. The pivotal role of p53 pro-apoptotic function in sensitivity to DDP has also recently been reported[17,18]. The tumor suppressor protein p53 has been shown to mediate the cellular stress response, as well as initiate DNA repair, cell-cycle arrest, senescence, and, most notably, apoptosis. However, p53 can also induce DNA-damage, thereby further promoting cell death. Therefore, we believe the combination of ECRG2 and DDP induced overexpression of p53 and then increased the response to DDP by the p53 pro-apoptotic function in esophageal DDP-resistant cancer cells.
In addition to p53, there are other molecules involved in DDP resistance. Dillehay et al[19] reported a novel small molecule targeting PCNA-induced DNA damage and apoptosis in prostate cancer cells, as well as enhancing DNA damage and apoptosis triggered by DDP in a p53-independent manner. Expression of PCNA genes in all organisms is traditionally associated with cell proliferation and thus with DNA synthesis during genome replication in the S phase of the cell cycle[20]. However, recent developments in PCNA protein interactions, trimer formation, and signaling regulation allow for the potential of possible therapeutic targeting of PCNA[21]. Both peptides and small molecules targeting PCNA signaling have been reported to sensitize cancer cells to chemotherapeutic agents[22,23]. A PCNA inhibitor can increase DNA double-strand breaks and cause the accumulation of DNA damage, which will eventually lead to cell death in cells that lack the ability to repair this damage[19]. In the present study, we demonstrated that the small molecule ECRG2 combined with DDP reversed DDP resistance by downregulating PCNA expression in esophageal DDP-resistant cancer cells. However, the underlying molecular mechanisms of this phenomenon require further future exploration.
MT is a low molecular weight protein that is rich in cysteine[24]. In recent years, the relationship between MT and tumors has received increasing attention, including the role of MT in tumor formation and the differentiation, proliferation, and apoptosis of tumor cells and tumor tissue in ammonia chloride DDP chemotherapy drug resistance[25,26]. In vitro and in vivo studies show that overexpression of MT involved in DDP resistance, MT expression level, and DDP resistance level are related, with MT increasing DDP resistance[27,28]. However, another study demonstrated that the expression of MT was not related to resistance in DDP treatment[29-31]. In this present study, we found no significant change in the expression of MT after increasing DDP and ECRG2. ECRG2 in combination with DDP promotes cell apoptosis and inhibits cell proliferation, but not by reducing the expression of MT.
In conclusion, we showed in the present study that ECRG2 in combination with DDP reduces or inhibits DDP resistance and induces cell apoptosis by upregulating p53 expression and downregulating PCNA expression, but not MT expression. Our results suggest that the combination of ECRG2 and DDP may be a promising strategy for the clinical treatment of esophageal cancers that are resistant to DDP. The effects of combined ECRG2 and DDP treatment on DDP-resistant cancer cells should be further investigated and confirmed in other human cancer cell lines, as well as animal models of cancers.
Cisplatin (DDP) is one of the most effective agents in esophageal cancer, but drug resistance poses a major impediment. Combination therapy has become an important method of overcoming drug resistance. It has been reported that esophageal cancer-related gene 2 (ECRG2) inhibits tumor cell growth and promotes cell apoptosis. In our previous study, we found ECRG2, in combination with DDP, had an enhanced inhibitory effect on EC9706 cell proliferation and an increased inductive effect on EC9706 cell apoptosis. This suggests that ECRG2 may be involved in the drug resistance of esophageal cancer. Currently, there are no known studies in the literature examining the effects of ECRG2 on the drug resistance of esophageal cancer.
Drug resistance and dose-limiting side effects have been a challenge to the further improvement of chemotherapy efficacy. These obstacles call for a novel combination of therapeutic approaches. Anticancer drugs may be combined with a broad spectrum of molecules such as small-molecule inhibitors, interfering RNA molecules, microRNA, oncolytic viruses, and naturally occurring substances. The ECRG2/DDP combination presents a very promising treatment approach.
This is the first known study to investigate the synergistic effect of ECRG2 in combination with DDP. In addition, the current study also explores the synergy mechanisms of ECRG2 and DDP in DDP-resistant esophageal cancer cells.
This study shows that ECRG2 in combination with DDP can inhibit viability and induce apoptosis in esophageal cancer DDP-resistant cells, possibly through upregulation of p53 expression and downregulation of PCNA expression. These findings suggest that the combination of ECRG2 and DDP may be a promising strategy for the clinical treatment of esophageal cancers that are resistant to DDP.
This is an interesting study about ECRG2 enhancing the anticancer effects of DDP in DDP-resistant esophageal cancer cells.
| 1. | Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18589] [Cited by in RCA: 21017] [Article Influence: 1910.6] [Reference Citation Analysis (2)] |
| 2. | Rustgi AK, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2014;371:2499-2509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1096] [Cited by in RCA: 1038] [Article Influence: 86.5] [Reference Citation Analysis (5)] |
| 3. | Kim M, Keam B, Kim TM, Kim HG, Kim JS, Lee SS, Shin SH, Kim MK, Park KU, Kim DW. Phase II Study of Irinotecan and Cisplatin Combination Chemotherapy in Metastatic, Unresectable Esophageal Cancer. Cancer Res Treat. 2016; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 4. | Millar J, Scullin P, Morrison A, McClory B, Wall L, Cameron D, Philips H, Price A, Dunlop D, Eatock M. Phase II study of gemcitabine and cisplatin in locally advanced/metastatic oesophageal cancer. Br J Cancer. 2005;93:1112-1116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Almhanna K, Hoffe S, Strosberg J, Dinwoodie W, Meredith K, Shridhar R. Concurrent chemoradiotherapy with protracted infusion of 5-fluorouracil (5-FU) and cisplatin for locally advanced resectable esophageal cancer. J Gastrointest Oncol. 2015;6:39-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 6. | Chang H, Shin SK, Cho BC, Lee CG, Kim CB, Kim DJ, Lee JG, Hur J, Lee CY, Bae MK. A prospective phase II trial of S-1 and cisplatin-based chemoradiotherapy for locoregionally advanced esophageal cancer. Cancer Chemother Pharmacol. 2014;73:665-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Pavelic J. Editorial: combined cancer therapy. Curr Pharm Des. 2014;20:6511-6512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 8. | Su T, Liu H, Lu S. [Cloning and identification of cDNA fragments related to human esophageal cancer]. Zhonghua Zhong Liu Za Zhi. 1998;20:254-257. [PubMed] |
| 9. | Cui YP, Wang JB, Zhang XY, Bi MX, Guo LP, Lu SH. Using yeast two-hybrid system to identify ECRG2 associated proteins and their possible interactions with ECRG2 gene. World J Gastroenterol. 2003;9:1892-1896. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Cui Y, Wang J, Zhang X, Lang R, Bi M, Guo L, Lu SH. ECRG2, a novel candidate of tumor suppressor gene in the esophageal carcinoma, interacts directly with metallothionein 2A and links to apoptosis. Biochem Biophys Res Commun. 2003;302:904-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 11. | Song HY, Deng XH, Yuan GY, Hou XF, Zhu ZD, Zhou L, Ren MX. Expression of bcl-2 and p53 in induction of esophageal cancer cell apoptosis by ECRG2 in combination with cisplatin. Asian Pac J Cancer Prev. 2014;15:1397-1401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 12. | Lee NP, Chan KT, Choi MY, Lam HY, Tung LN, Tzang FC, Han H, Lam IP, Kwok SY, Lau SH. Oxygen carrier YQ23 can enhance the chemotherapeutic drug responses of chemoresistant esophageal tumor xenografts. Cancer Chemother Pharmacol. 2015;76:1199-1207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 13. | Lai KK, Chan KT, Choi MY, Wang HK, Fung EY, Lam HY, Tan W, Tung LN, Tong DK, Sun RW. 14-3-3σ confers cisplatin resistance in esophageal squamous cell carcinoma cells via regulating DNA repair molecules. Tumour Biol. 2016;37:2127-2136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Yu L, Gu C, Zhong D, Shi L, Kong Y, Zhou Z, Liu S. Induction of autophagy counteracts the anticancer effect of cisplatin in human esophageal cancer cells with acquired drug resistance. Cancer Lett. 2014;355:34-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 96] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 15. | Zhang R, Li Y, Dong X, Peng L, Nie X. MiR-363 sensitizes cisplatin-induced apoptosis targeting in Mcl-1 in breast cancer. Med Oncol. 2014;31:347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 16. | Li MN, Huang G, Guo LP, Lu SH. [Inhibitory effects of esophageal cancer related gene 2 on proliferation of human esophageal cancer cell EC9706]. Zhonghua Yi Xue Za Zhi. 2005;85:2785-2788. [PubMed] |
| 17. | Huang H, Chen AY, Ye X, Li B, Rojanasakul Y, Rankin GO, Chen YC. Myricetin inhibits proliferation of cisplatin-resistant cancer cells through a p53-dependent apoptotic pathway. Int J Oncol. 2015;47:1494-1502. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 18. | Li X, Huang JM, Wang JN, Xiong XK, Yang XF, Zou F. Combination of chrysin and cisplatin promotes the apoptosis of Hep G2 cells by up-regulating p53. Chem Biol Interact. 2015;232:12-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 19. | Dillehay KL, Lu S, Dong Z. Antitumor effects of a novel small molecule targeting PCNA chromatin association in prostate cancer. Mol Cancer Ther. 2014;13:2817-2826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Strzalka W, Ziemienowicz A. Proliferating cell nuclear antigen (PCNA): a key factor in DNA replication and cell cycle regulation. Ann Bot. 2011;107:1127-1140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 627] [Cited by in RCA: 557] [Article Influence: 37.1] [Reference Citation Analysis (1)] |
| 21. | Wang SC. PCNA: a silent housekeeper or a potential therapeutic target? Trends Pharmacol Sci. 2014;35:178-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 232] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 22. | Inoue A, Kikuchi S, Hishiki A, Shao Y, Heath R, Evison BJ, Actis M, Canman CE, Hashimoto H, Fujii N. A small molecule inhibitor of monoubiquitinated Proliferating Cell Nuclear Antigen (PCNA) inhibits repair of interstrand DNA cross-link, enhances DNA double strand break, and sensitizes cancer cells to cisplatin. J Biol Chem. 2014;289:7109-7120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 23. | Yu YL, Chou RH, Liang JH, Chang WJ, Su KJ, Tseng YJ, Huang WC, Wang SC, Hung MC. Targeting the EGFR/PCNA signaling suppresses tumor growth of triple-negative breast cancer cells with cell-penetrating PCNA peptides. PLoS One. 2013;8:e61362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 24. | Mehus AA, Muhonen WW, Garrett SH, Somji S, Sens DA, Shabb JB. Quantitation of human metallothionein isoforms: a family of small, highly conserved, cysteine-rich proteins. Mol Cell Proteomics. 2014;13:1020-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 25. | Tian ZQ, Xu YZ, Zhang YF, Ma GF, He M, Wang GY. Effects of metallothionein-3 and metallothionein-1E gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells. Genet Mol Res. 2013;12:4595-4603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 39] [Reference Citation Analysis (0)] |
| 26. | Zamirska A, Matusiak Ł, Dziegiel P, Szybejko-Machaj G, Szepietowski JC. Expression of metallothioneins in cutaneous squamous cell carcinoma and actinic keratosis. Pathol Oncol Res. 2012;18:849-855. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Kelley SL, Basu A, Teicher BA, Hacker MP, Hamer DH, Lazo JS. Overexpression of metallothionein confers resistance to anticancer drugs. Science. 1988;241:1813-1815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 411] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 28. | Kennette W, Collins OM, Zalups RK, Koropatnick J. Basal and zinc-induced metallothionein in resistance to cadmium, cisplatin, zinc, and tertbutyl hydroperoxide: studies using MT knockout and antisense-downregulated MT in mammalian cells. Toxicol Sci. 2005;88:602-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 29. | Surowiak P, Kaplenko I, Spaczyński M, Zabel M. The expression of metallothionein (MT) and proliferation intensity in ovarian cancers treated with cisplatin and paclitaxel. Folia Morphol (Warsz). 2003;62:493-495. [PubMed] |
| 30. | Tuzel E, Yorukoglu K, Ozkara E, Kirkali Z. Association of metallothionein expression and clinical response to cisplatin based chemotherapy in testicular germ cell tumors. Cent European J Urol. 2015;68:45-50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 31. | Gansukh T, Donizy P, Halon A, Lage H, Surowiak P. In vitro analysis of the relationships between metallothionein expression and cisplatin sensitivity of non-small cellular lung cancer cells. Anticancer Res. 2013;33:5255-5260. [PubMed] |
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Kantsevoy S, Ong J, Tokunaga Y S- Editor: Qi Y L- Editor: Rutherford A E- Editor: Wang CH