Published online May 21, 2016. doi: 10.3748/wjg.v22.i19.4638
Peer-review started: January 11, 2016
First decision: March 21, 2016
Revised: April 1, 2016
Accepted: April 20, 2016
Article in press: April 20, 2016
Published online: May 21, 2016
Processing time: 111 Days and 2.8 Hours
Signaling pathways of gastric carcinogenesis and gastric cancer progression are being avidly studied to seek optimal treatment of gastric cancer. Among them, hepatocyte growth factor (HGF)/c-MET, phosphoinositide 3-kinase (PI3K)/Akt/mammalian target of rapamycin (mTOR) and janus kinase 2/signal transducer and activator of transcription 3 (JAK2/STAT3) pathways have been widely investigated. Their aberrant expression or mutation has been significantly associated with advanced stage or poor prognosis of gastric cancer. Recently, aberrations of immune checkpoints including programmed cell death-1/programmed cell death ligand-1 (PD-1/PD-L1) have been suggested as an important step in the formation of a microenvironment favorable for gastric cancer. Accomplishments in basic research have led to the development of novel agents targeting these signaling pathways. However, phase III studies of selective anti-HGF/c-MET antibodies and mTOR inhibitor failed to show significant benefits in terms of overall survival and progression-free survival. Few agents directly targeting STAT3 have been developed. However, this target is still critical issue in terms of chemoresistance, and SH2-containing protein tyrosine phosphatase 1 might be a significant link to effectively inhibit STAT3 activity. Inhibition of PD-1/PD-L1 showed durable efficacy in phase I studies, and phase III evaluation is warranted. Therapeutic strategy to concurrently inhibit multiple tyrosine kinases is a reasonable option, however, lapatinib needs to be further evaluated to identify good responders. Regorafenib has shown promising effectiveness in prolonging progression-free survival in a phase II study. In this topic highlight, we review the biologic roles and outcomes of clinical studies targeting these signaling pathways.
Core tip: Among various cellular signaling pathways, hepatocyte growth factor/c-MET, phosphoinositide 3-kinase/Akt/mammalian target of rapamycin and janus kinase 2/signal transducer and activator of transcription 3 pathways are reportedly important in gastric carcinogenesis and metastasis. Aberrations of immune checkpoints have been vigorously investigated. However, clinical results of their target agents have not always matched the theoretical expectations of efficacy. In this review, we summarize the biologic impacts of the aforementioned signaling pathways, and their recent clinical outcomes including those of multiple kinase inhibitors in gastric cancer.
- Citation: Joo MK, Park JJ, Chun HJ. Recent updates of precision therapy for gastric cancer: Towards optimal tailored management. World J Gastroenterol 2016; 22(19): 4638-4650
- URL: https://www.wjgnet.com/1007-9327/full/v22/i19/4638.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i19.4638
Core tip: Among various cellular signaling pathways, hepatocyte growth factor/c-MET, phosphoinositide 3-kinase/Akt/mammalian target of rapamycin and janus kinase 2/signal transducer and activator of transcription 3 pathways are reportedly important in gastric carcinogenesis and metastasis. Aberrations of immune checkpoints have been vigorously investigated. However, clinical results of their target agents have not always matched the theoretical expectations of efficacy. In this review, we summarize the biologic impacts of the aforementioned signaling pathways, and their recent clinical outcomes including those of multiple kinase inhibitors in gastric cancer.
Gastric cancer is the fourth common malignant tumor worldwide, and the second most common cause of cancer-related mortality[1]. The progress in therapeutic approaches has allowed complete remission of early gastric cancer by surgical or even endoscopic resection of tumors. However, if gastric cancer is advanced when diagnosed, the prognosis is generally poor and survival time is short even after surgical complete resection. Therefore, highly selective and effective chemotherapy remains an important issue for appropriate management of advanced gastric cancer.
A recent notable study provided a comprehensive molecular evaluation of primary gastric adenocarcinoma tissues as part of The Cancer Genome Atlas project[2]. The authors proposed four subtypes of gastric cancer according to the molecular characteristics: Epstein-Barr virus positive tumors, microsatellite instability tumors, genomically stable tumors and chromosomal unstable tumors. This study is a prime example of efforts to develop optimal classification of gastric cancer by analyzing common dysregulated pathways and to provide distinct tailored therapy for individual patients. Since the report of significant clinical benefits of trastuzumab in human epidermal growth factor receptor 2 (HER2)-positive gastric and esophagogastric junction (EGJ) adenocarcinoma[3], various targets have been investigated in the treatment of advanced gastric cancer. These targets include epidermal growth factor receptor (EGFR), vascular endothelial growth factor (VEGF), hepatocyte growth factor(HGF)/c-MET and mammalian target of rapamycin (mTOR)[4]. However, we still have a long way to go before complete conquest of gastric cancer.
In this topic highlights, we aimed to review the biologic roles of several molecular signaling pathways on the basis of recent trials of targeted therapies in advanced gastric cancer. These include the HGF/c-MET, phosphoinositide 3-kinase (PI3K)/Akt/mTOR, janus kinase 2/signal transducer and activator of transcription 3 (JAK2/STAT3) and programmed cell death-1/programmed cell death ligand-1 (PD-1/PD-L1) pathways. In the latter part, we focus on clinical outcomes of newly developed agents targeting the aforementioned pathways and summarize the findings of some clinical studies of multi-kinase inhibitors (MKIs), which can simultaneously multiple receptor tyrosine kinases (RTKs) in advanced gastric cancer.
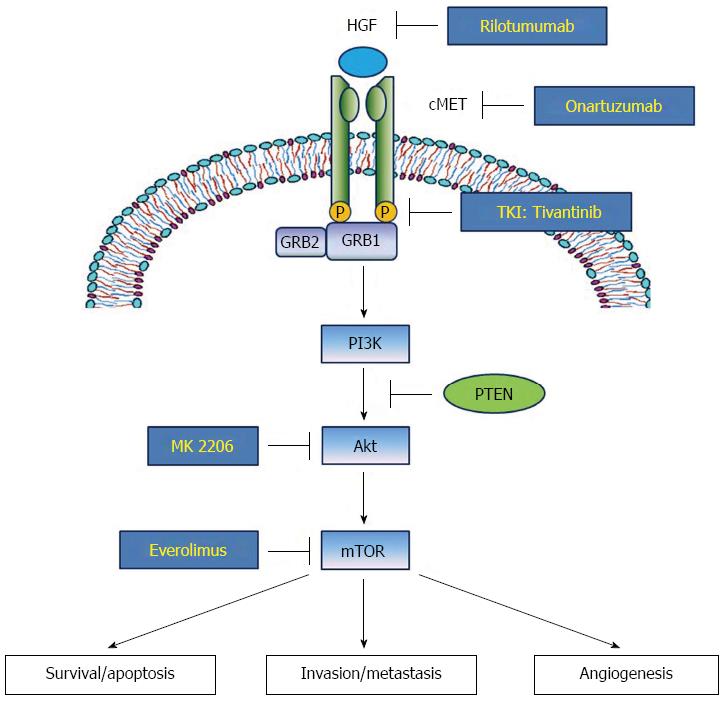
c-MET is a heterodimeric subfamily of RTK. c-MET is composed of an α-chain, which possesses only an extracellular domain, and a β-chain composed of extracellular, transmembrane and intracellular domains[5]. The ligand of c-MET, HGF, is converted into an active form that causes dimerization and activation of the c-MET receptor. The activated HGF/c-MET signal leads to autophosphorylation of multiple tyrosine residues of the intracellular region of c-MET, such as Y1230, Y1234, Y1235, Y1349 and Y1356, which form multi-functional docking sites to recruit several intracellular adaptor proteins[6]. Among them, Grb2-associated binder 1 (GAB1) can directly bind to c-MET or forms a complex with growth factor-bound protein 2 (GRB2) to indirectly interact with c-MET. The c-MET association recruits several main adaptor proteins including STAT3 and PI3K, which in turn activate downstream biologic effects including cellular proliferation, migration/invasion and induction of epithelial-mesenchymal transition (EMT) (Figure 1)[7].
Clinical impact of the HGF/c-MET pathway in gastric cancer has been well documented. High protein expression rate of c-MET in gastric carcinoma tissue has been demonstrated by immunohistochemistry (IHC; 43%-82%)[8,9] and by gene amplification rate (2%-10%)[9-12]. In both approaches, the elevated expression of c-MET has been positively associated with advanced tumor stage and poor survival rate. Consequently, multiple agents targeting the HGF/c-MET signaling pathway are being evaluated. Tivantinib, an anti-c-MET tyrosine kinase inhibitor (TKI), used in combination with the EGFR TKI, erlotinib, has extended progression-free survival (PFS) in patients with locally advanced or metastatic non-squamous non-small-cell lung cancer in a phase III trial[13]. In gastric cancer, phase III studies for rilotumumab, an anti-HGF monoclonal antibody, and onartuzumab, an anti-c-MET monoclonal antibody, have been completed, and clinical outcomes of tivantinib were recently reported[14].
The PI3K-Akt-mTOR pathway plays a pivotal role in oncogenesis and progression including cell growth, survival, invasion/metastasis and angiogenesis, and gastric cancer is no exception. PI3K is usually activated through binding and stimulation of various RTKs by growth factors including HGF and c-MET. Activated PI3K subsequently phosophorylates and activates phosphatidylinositol 3,4-biphosphate (PIP2), phosphatidylinositol 3,4,5-triphosphate (PIP3), phosphoinositide-dependent protein kinase 1 and Akt[15]. Akt, which is also termed protein kinase B, is a major effector protein of the PI3K pathway. Phosphorylated Akt (p-Akt) modulates various biologic functions like cell survival, migration/invasion and angiogenesis through downstream adaptor molecules including mTOR (Figure 1)[16].
Genetic alteration of biological signals involving the PI3K/Akt/mTOR pathway has been frequently detected in gastric carcinoma. For example, a point mutation of PIK3CA encoding p110 (a class IA subunit of PI3K) is often observed in gastric carcinoma tissues, ranging from 4.3%-25%[17-21], with the point mutation mostly seen in exon 9 and exon 20[17]. Their mutation or gene amplification is positively associated with the T stage of gastric cancer[20,22]. In contrast, PTEN, which encodes phosphatase and tensin homolog and inactivates Akt by converting PIP3 to PIP2, is deleted in 4%-23% of gastric cancers[21,23,24] and loss of heterozygosity (LOH) is observed in 17%-47% cases of gastric cancer. LOH of PTEN is significantly associated with p-Akt level in gastric carcinoma tissues, TNM stage and poor prognosis of survival[25-29]. Activated Akt signaling promotes mTOR protein complexes 1 and 2 (mTORC1 and mTORC2), which can play pivotal roles in cancer cell migration and metastasis. Prevalence of mTOR expression is reported as approximately 50% in gastric cancer tissues, and is negatively associated with PTEN expression[30,31].
Clinical and laboratory evidence indicates the promising potential of targeting the PI3K-Akt-mTOR signaling pathway for efficacious treatment of gastric cancer, and various kinds of inhibitors or antibodies acting on this pathway have been developed and tried. These inhibitors are classified into several categories that include PI3K inhibitors, dual mTOR1/mTOR2 inhibitors, Akt inhibitors, mTOR1 inhibitors and dual PI3K/mTOR inhibitors[15]. Among them, a phase I study of isoform specific PI3K inhibitor (p110α) BYL719 is ongoing (NCT01613950)[32], and clinical outcomes of Akt inhibitor MK 2206 and mTOR1 inhibitor everolimus and rapamycin were previously reported, and are dealt with more fully in the latter part of this review.
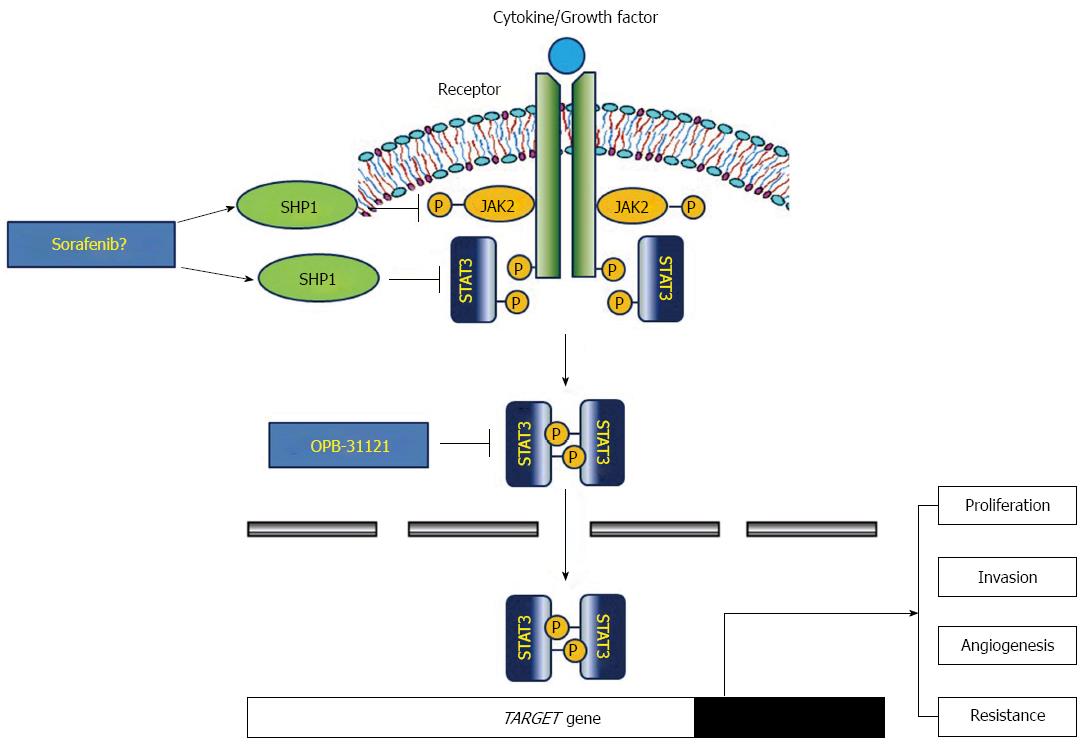
The most established stimulator of STAT3 signaling pathway is the interleukin (IL)-6 family that includes IL-6, IL-11 and leukemia inhibitory factor, which bind to their receptors, and then phosphorylate and activate JAK2. Activated JAK2 recruits and activates STAT3 by phosphorylation, which can dimerize and translocate into the nucleus to act as a transcription factor and up-regulate various target genes involving cellular proliferation, migration/invasion and angiogenesis[33]. Indeed, persistent constitutive activation of JAK2/STAT3 in cancer cells is closely associated with gastric carcinogenesis and poor prognosis[34]. Besides this classic effect of JAK2/STAT3 pathway in cancer development, another pivotal role of STAT3 protein is the tumor microenvironment, where immune cells can be recruited and STAT3 can mediate various interactions with cancer cells to generate tumor progression. In gastric cancer, Helicobacter pylori (H. pylori)-induced cytotoxin-associated antigen (CagA) is closely associated with STAT3 activity in both gastric epithelial cells and mucosal immune cells. For example, H. pylori infection and CagA secretion can lead to IL-23 release from dendritic cells, which binds to their receptor and activates JAK2/STAT3 transmembrane signaling of naïve CD4+ T-cells, and causes differentiation of T-helper (Th)-17 specific lineages to release associated cytokines including IL-17[35]. Up-regulated IL-17 can promote pro-inflammatory and oncogenic environment. Expression level of IL-17 is positively correlated with depth of tumor, lymphovascular invasion and lymph node involvement in gastric cancer tissues[36,37], and IL-17 mediates angiogenesis via up-regulation of VEGF in vivo and in vitro[38]. In gastric epithelial cells, CagA is translocated via the type-IV secretion system and releases IL-11. The released IL-11 bind to their receptor and activate the JAK2/STAT3 cascade[39]. Activated STAT3 functions as a transcription factor to induce many target genes involved in proliferation, invasion/metastasis and angiogenesis including cyclin D1, surviving, matrix metalloproteinase-9, CD44v6 and VEGF[34,40].
Thus, a therapeutic strategy to target the STAT3 signaling pathway appears to be reasonable. Routes of inhibition include blockade of JAK activation by de-phosphorylation, inhibition of STAT3 phosphorylation, dimerization or gene transcription[35]. In terms of de-phosphorylation, several phosphatases have been reported to be associated with STAT3 activity. Among them, SH2-containing protein tyrosine phosphatase 1 (SHP1) may be crucial in the down-regulation of the JAK2/STAT3 pathway by dephosphorylation[41-43]. Several candidate agents including natural compounds were reported to induce SHP1 and inhibit STAT3 activity. Sorafenib and its synthetic analogues also can act as a SHP1 agonist to inhibit phosphor-STAT3 activity and show various anti-cancer effects, such as promotion of apoptosis, overcoming of radio- or chemo-resistance and inhibition of EMT or fibrosis on hepatocellular carcinoma cell lines[44-51]. However, the exact inhibitory role of SHP1 in gastric cancer development and progress is unknown. We recently showed that expression of SHP1 is reduced or ameliorated in various gastric cancer cell lines due to epigenetic silencing, and that reinforced SHP1 expression significantly inhibits cellular proliferation, migration/invasion and induce apoptosis[52]. SHP1 might be a promising target to effectively inhibit JAK2/STAT3 activity in gastric cancer cells (Figure 2).
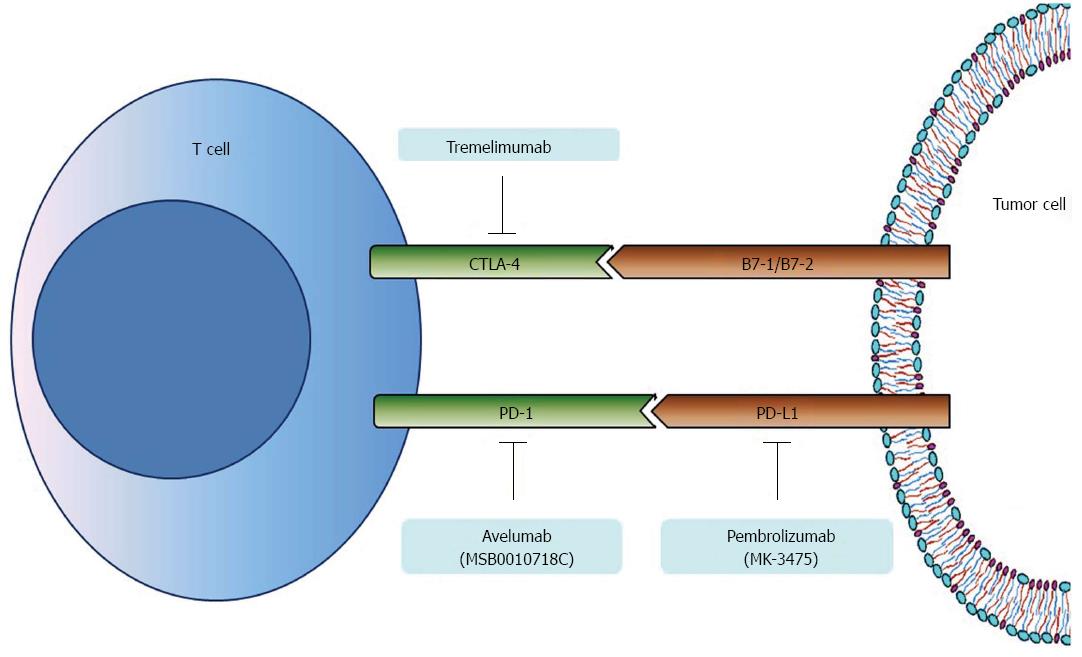
Immune checkpoints regarding tumor infiltrating lymphocytes and immune evasion mechanism associated with carcinogenesis have been studied in the search for alternative therapeutic targets. Among them, cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and PD-1, which are minimally expressed on the surface of resting T-lymphocytes but are widely expressed on activated T-lymphocytes, have been intensively studied for gastric carcinogenesis, and anti-PD-1 antibodies are already in clinical trials of gastric cancer chemotherapy[53]. Ligands for PD-1 (PD-L1) and CTLA-4 (B7-1/B7-2), which are expressed on the surface of tumor cells, bind to PD-1 and CTLA-4 respectively, inhibit pivotal function of effector T-cells for immune surveillance and consequently promote the growth of gastric cancer cells (Figure 3)[54].
PD-1 expression differs between gastric cancer tissues and non-cancerous tissues, with the significantly up-regulated PD-1 level in gastric cancer tissues being significantly correlated with poor clinical parameters including increased tumor size, advanced stage, metastasis and patient survival[55-58]. Furthermore, PD-1 expression on CD4+ and CD8+ T cells from gastric cancer tissues is higher than non-cancer tissues or peripheral blood mononuclear cells from normal subjects, and is significantly associated with disease progression[59]. However, a recent Korean study demonstrated that expression rate of PD-L1 on gastric cancer tissues was 43.6%, and was related to less advanced stage, intestinal type, well/moderately differentiated adenocarcinoma rather than poor differentiation and better overall survival (OS) and disease-free survival[60]. A recent Chinese study investigated PD-L1 expression level using large number of gastric cancer tissues (almost 400 specimens); PD-L1 expression was significantly associated with TIL density, and moderate to high TIL density was closely correlated with better prognosis[61]. Thus, the exact relationship between PD-1/PD-L1 expression and clinical parameters needs to be further evaluated.
The pivotal ToGA study of targeted therapy for the treatment of unresectable gastric/EGJ cancer investigated the synergistic effects of trastuzumab, a monoclonal anti-HER2 antibody, with capecitabine/cisplatin or fluorouracil/cisplatin regimen. OS and PFS were significantly prolonged[3]. Since then, various target agents have been tried for the optimal treatment of advanced gastric cancer. Among them, newly developed drugs targeting the HGF/c-MET, PI3K/Akt/mTOR, JAK2/STAT3 and PD-1/PD-L1 pathways are dealt with here, and MKIs that simultaneously target multiple tyrosine kinases is also introduced in this section. A portion of these studies were presented at the 2015 annual meeting of the American Society of Clinical Oncology (ASCO); their outcomes are summarized in Table 1.
| Author and trial | Line of treatment | Phase of study | n | Treatment arms | Outcomes |
| Anti-HGF/cMET antibodies | |||||
| Cunningham et al[63], RILOMET-1 (2015) | First | III | 609 | ECX + rilotumumab vs ECX + placebo | OS: 9.6 mo vs 11.5 mo (HR = 1.37, P = 0.016) |
| PFS: 5.7 mo vs 5.7 mo (HR = 1.30, P = 0.016) | |||||
| ORR: 30% vs 39.2% (OR 0.67, P = 0.027) | |||||
| Shah et al[65], METGastric (2015) | First | III | 562 | mFOLFOX + ornatuzumab vs mFOLFOX + placebo | OS: 11.0 mo vs 11.3 mo (HR = 0.82, P = 0.244) |
| PFS: 6.7 mo vs 6.8 mo (HR = 0.90, P = 0.429) | |||||
| ORR: 46% vs 41% (P = 0.253) | |||||
| Malka et al[66], PRODIGE 17 ACCORD 20 MEGA (2015) | First | II | 162 | mFOLFOX alone vs mFOLFOX + panitumumab vs mFOLFOX + rilobumumab | 4-mo PFS rate: 71 vs 63 vs 63% |
| PFS: 5.8 mo vs 5.2 mo vs 7.6 mo | |||||
| ORR: 54% vs 44% vs 50% | |||||
| Akt/mTOR inhibitors | |||||
| Hudis et al[68] (2013) | Second/third | I | 34 | Trastuzumab + Akt inhibitor (MK-2206) | RR (including stable disease): 24% |
| Time to progression: 72 d | |||||
| Ohtsu et al[70] GRANITE (2013) | Second/third | III | 646 | Everolimus vs BSC | OS: 5.4 mo vs 4.3 mo (HR = 0.90, P = 0.124) |
| PFS: 1.7 mo vs 1.4 mo (HR = 0.66, P < 0.001) | |||||
| ORR: 4.5% vs 2.1%; DCR: 43.3% vs 22.0% | |||||
| Shen et al[71] (2014) | First | II | 40 | Everolimus + cisplatin + HDFL | OS: 10.5 mo (95%CI: 8.6-12.3) |
| PFS: 6.9 mo (95%CI: 4.9-8.4) | |||||
| STAT3 inhibitor | |||||
| Oh et al[74] (2015) | Second/third | I | 25 | STAT3 inhibitor (OPB-31121) | RR (including stable disease): 44.4% |
| Immune checkpoints inhibitors | |||||
| Ralph et al[89] (2010) | Second | II | 18 | Tremelimumab | OS: 4.8 mo (95%CI: 4.06-5.59) |
| 12 mo OS rate: 33% (95%CI: 14-54) | |||||
| RR (including stable disease): 27.8% | |||||
| Bang et al[90], KEYNOTE-012 (2015) | Second/third | I | 39 | Pembrolizumab (MK-3475) | OS: 11.4 mo; PFS 1.9 mo |
| ORR: 22% (95%CI: 10-39) | |||||
| Yamada et al[93] (2015) | Second/third | I | 20 | Avelumab (MS0010718C) | PFS: 11.9 wk (95%CI: 6.0-12.3) |
| ORR: 15.0% (95%CI: 3.2-37.9) | |||||
| Multikinase inhibitors | |||||
| Sun et al[95] (2010) | First | II | 44 | Sorafenib + docetaxel + cisplatin | OS: 13.6 mo (90%CI: 8.6-16.1) |
| PFS: 5.8 mo (90%CI: 5.4-7.4) | |||||
| PR: 41% (90%CI: 28-54) | |||||
| Martin-Richard et al[96], GERCAD (2013) | First | II | 40 | Sorafenib + oxaliplatin | OS: 6.5 mo (95%CI: 5.2-9.6) |
| PFS: 3 mo (95%CI: 2.3-4.1) | |||||
| RR (including stable disease): 50.0% | |||||
| Hecht et al[100], LOGiC (2015) | First | III | 487 | CapeOx + lapatinib vs CapeOx + placebo | OS: 12.2 mo vs 10.5 mo (HR = 0.91, P = 0.349) |
| PFS: 6.0 mo vs 5.4 mo (HR = 0.82, P = 0.0381) | |||||
| ORR: 53% vs 39% (P = 0.0031) | |||||
| Satoh et al[101], TyTAN (2014) | Second | III | 261 | Lapatinib + paclitaxel vs Paclitaxel alone | OS: 11.0 mo vs 8.9 mo (HR = 0.84, P = 0.1044) |
| PFS: 5.4 mo vs 4.4 mo (HR = 0.85, P = 0.2441) | |||||
| ORR: 27% vs 9% (P < 0.001) | |||||
| Pavlakis et al[103], INTEGRATE (2015) | Second/third | II | 147 | Regorafenib vs placebo | OS: 5.8 mo vs 4.5 mo (HR = 0.74, P = 0.11) |
| PFS: 2.6 mo vs 0.9 mo (HR = 0.40, P < 0.0001) | |||||
| RR (including stable disease): 44% vs 16% | |||||
| Lee et al[106] (2015) | First | II | 66 | CapeOx + pazopanib | PFS: 6.5 mo; OS: 10.5 mo; ORR: 57.6% |
Phase III studies of anti-HGF/c-MET antibodies
Rilotumumab, an anti-HGF monoclonal antibody, and onartuzumab, an anti-c-MET monoclonal antibody, have been tried as first-line treatments for gastric or EGJ adenocarcinoma in phase III studies. Their clinical outcomes were presented at the ASCO 2015 meeting. Rilotumumab significantly increased PFS when combined with ECX (epirubicin/cisplatin/capecitabine) regimen in a phase Ib/II study[62]. From this background, the phase III RILOMET-1 study of first-line therapy of MET-positive, HER2-negative gastric/EGJ cancer compared rilotumumab (15 mg/kg) plus ECX with placebo plus ECX was performed. OS (9.6 mo vs 11.5 mo, HR = 1.37, P = 0.021) and objective response rate (ORR: 30.0% vs 39.2%, OR = 0.67, P = 0.027) were significantly inferior in the rilotumumab group, and subgroup analysis also showed that no subgroups appeared to be benefit with rilotumumab arm regardless of the degree of MET positivity[63]. This result is contrary to that of a phase I/IIa study, which contained a larger number of Asian patients (18%) than the RILOMET-1 population (1%). The different racial distribution may have contributed to the opposite outcomes between the two studies. Thus, the phase III RILOMET-2 study has been performed to investigate the efficacy of rilotumumab in combination with cisplatin/capecitabine regimen as the first line chemotherapy among Asian patients with unresectable gastric/EGJ cancer[64].
The METGastric study of onartuzumab (10 mg/kg) for the treatment of HER2-negative, MET-positive metastatic gastric or EGJ adenocarcinoma without prior treatment, used onartuzumab in combination with the 5-fluorouracil (5-FU)/leukovorin/oxaliplatin (FOLFOX) regimen and compared outcomes with FOLFOX alone[65]. The addition of onartuzumab to FOLFOX was ineffective in the intention-to-treat analysis and OS, PFS and ORR were not significantly different between the two groups. However, addition of onartuzumab showed a marginal effect in OS for the moderate-to-strong MET positive subgroup (9.7 mo vs 11.0 mo, HR = 0.64, P = 0.062). Grade 3 or 4 adverse events were more common in the onartuzumab arm. Furthermore, a French phase II study that compared FOLFOX plus rilotumumab or panitumumab, an anti-EGFR antibody, with FOLFOX alone for first-line treatment of metastatic, HER2-negative gastric or EGJ adenocarcinoma showed that adding panitumumab or rilotumumab seemed more toxic and was not more effective than mFOLFOX6 alone[66]. Considering recent outcomes of phase II/III studies of rilotumumab and ornatuzumab, targeting HGF/c-MET in gastric cancer has little rationale for further evaluation. However, cMET still has potential for promising biomarkers considering that c-MET-positive gastric/EGJ cancers have strong association with shorter OS and poor prognosis[67], and future research needs to search for other significant predictive factors for response to anti-HGF/c-MET therapy.
A phase I study evaluated the combinatory effect of MK-2206, a potent pan-Akt inhibitor, with trastuzumab for treatment of HER2-positive, refractory gastric carcinoma. The rational was that the PI3K/Akt pathway is a main downstream signaling pathway of HER2 and is closely related with trastuzumab resistance. Oral MKN-2206 was given either 135 mg every week or 60 mg every other day with trastuzumab 8 mg/kg intravenously on day 1 every 3 wk. Clinical benefit response rate including stable disease more than 4 mo was 24%, and median time to progression was 72 d[68].
The PI3K/Akt pathway might be successfully inhibited by targeting mTORC1 kinase, and the development of rapamycin analogs (e.g., everolimus, temsirolimus) have been promoted[15]. A multicenter phase II study of everolimus, an oral inhibitor of mTOR, in patients with refractory metastatic gastric cancer showed a disease control rate of 56.0%, PFS of 2.7 mo (95%CI: 1.6-3.0 mo) and OS of 10.1 mo (95%CI: 6.5-12.1 mo), which warrant further phase III evaluation[69]. However, results of the phase III GRANITE-1 study comparing everolimus with best supportive care for previously treated advanced gastric cancer were disappointing, and researchers failed to demonstrated significant benefit in OS (5.4 mo vs 4.3 mo, HR = 0.90, P = 0.124); indeed, PFS was significantly increased in the everolimus arm (1.7 mo vs 1.4 mo, HR = 0.66, P < 0.001)[70]. A phase II multicenter study of low dose everolimus (10 mg on days 1, 8 and 15) plus cisplatin and a weekly 24-h infusion of high-dose 5-FU and leucovorin (cisplatin 35 mg/m2 intravenous infusion for 24 h on days 1 and 8, 5-FU 2000 mg/m2 and leucovorin 300 mg/m2 intravenous infusion for 24 h on days 1, 8 and 15) for treatment-naïve gastric cancer was conducted but failed to increase ORR as in a preplanned statistical assumption (52.5%)[71]. However, in one case everolimus was tried after failure of 1st and 2nd line chemotherapy for a young male metastatic gastric cancer patient with multiple liver metastases. A subsequent mutational analysis revealed a PIK3CA hotspot mutation and pS6 overexpression in the primary tumor. The patient achieved stable disease for 1 year and pS6 expression was nearly abolished after two cycles of everolimus treatment[72]. Furthermore, a phase II study of everolimus for refractory metastatic gastric and EGJ adenocarcinoma showed that a subgroup with strong pS6 expression (≥ 2 + IHC staining) was significantly correlated with better PFS and disease control rate[73]. Therefore, subgroup analysis for finding of positive predictive biomarkers in patients treated with everolimus needs to be performed.
Few agents capable of directly targeting STAT3 have been developed, and clinical trials of STAT3 inhibitors in the treatment of gastric cancer are lacking. A recent phase I study reported that OPB-31121, an oral STAT3 inhibitor, showed an overall response rate of 44.4% assessed as stable disease in advanced solid tumors including gastric cancer[74]. However, STAT3 not only up-regulates various target oncogenes associated with gastric carcinogenesis and metastasis, but is closely related with drug resistance of standard chemotherapeutic agents including 5-FU, cisplatin and adriamycin in gastric cancer[75-77]. In this regard, targeting of STAT3 remains a critical issue in the treatment of gastric cancer, and development of specific and effective inhibitors of STAT3 should be further investigated. Several natural compounds[76,78-80] and pharmacologic medicines, such as proton pump inhibitors[75,81], inhibit STAT3 activity in in vitro and in vivo studies of gastric cancer. These agents are expected to show a synergetic effect or enhance chemosensitivity when combined with standard chemotherapy agents.
Several natural compounds inhibit the STAT3 activation pathway through induction of SHP1 in hematopoietic cancer cell lines[42,43,82-85] and hepatoceullar carcinoma (HCC) cell lines[86,87]. We recently showed that plumbagin (5-hydroxy-2-methyl-1,4-naphthoquinone), a quinonoid constituent extracted from the roots of the medical plant Plumbago zeylanica L, suppresses STAT3 activity and consequently targets gene expression via induction of SHP1 in gastric cancer cells[88]. Because most gastric cancer cells showed reduced or lack of expression of SHP1, a therapeutic strategy to indirectly inhibit STAT3 pathway might be an alternative option in pharmacologic treatment of gastric cancer and SHP1 may play pivotal roles in this signaling pathway. As mentioned above, several MKIs, such as sorafenib and sunitinib, show a significant link between SHP1 and suppression of STAT3 activity in HCC cells.
The anti-CTLA-4 monoclonal antibody tremelimumab was developed and a phase II study was performed to evaluate its use in second-line chemotherapy in advanced gastric and esophageal adenocarcinoma. However, the results were disappointing and only one patient achieved partial response among 18 enrolled patients, and stable disease was observed only in four patients[89]. Concerning the PD-1/PD-L1 pathway, pembrolizumab (MK-3475), an anti-PD-1 monoclonal antibody, and avelumab (MS0010718C), an anti-PD-L1 monoclonal antibody, have been developed. Pembrolizumab was tried for rescue therapy of recurrent or metastatic gastric or EGJ adenocarcinoma, which were positive for PD-L1, in the KEYNOTE-012 study[90]. ORR by central review was 22.2%, PFS 1.9 mo and OS 11.4 mo, and pembrolizumab showed durable efficacy and manageable safety profile for the heavily pre-treated, PD-L1 positive population. Further studies to support the efficacy of pembrolizumab in advanced gastric cancer are now in progress. For example, KEYNOTE-059 (NCT02335411) is a phase II study of pembrolizumab monotherapy or in combination with standard chemotherapy[91] and KEYNOTE-061 (NCT02370498) is a phase III study to compare pembrolizumab monotherapy with paclitaxel as the second-line therapy[92] (Table 2).
| Trial identifier | Line of treatment | Phase of study | Treatment arms | Primary endpoint |
| Akt/mTOR inhibitors | ||||
| NCT01613950[32] | Second/third | Ib | AUY922/BYL719 | MTD |
| Immune checkpoints inhibitors | ||||
| NCT02335411 (KEYNOTE-059)[91] | Third | II | Cohort 1: pembrolizumab monotherapy | ORR |
| Cohort 2: pembrolizumab + 5-FU/cisplatin or capecitabine/cisplatin | ||||
| NCT02370498 (KEYNOTE-061)[92] | Second | III | Pembrolizumab vs paclitaxel | PFS, OS |
| Multikinase inhibitors | ||||
| NCT02015169[107] | Neoadjuvant | II | XELOX + lapatinib | R0 resection rate |
| NCT01913639[108] | First | II | FOLFOX + regorafenib | PFS |
In Japan, avelumab was tried for refractory stage IV gastric and EGJ adenocarcinoma. A dose of 10 mg/kg was administered intravenously every 2 wk until progression. Most of adverse events were grade 1 or 2, ORR was 15.0% and PFS was 11.9 wk. Additional studies to evaluate the efficacy of avelumab and biomarkers from tumor tissue and blood samples including PD-L1 expression need to be evaluated[93].
RTKs play crucial roles in the development of proliferation, differentiation, migration/invasion and apoptosis in gastric cancer. Currently, various inhibitors targeting the tyrosine kinase motif have been developed, and some display concurrent inhibitory effects of multiple tyrosine kinases. One of the first generation MKIs was sorafenib, which can inhibit BRAF, VEGF receptor (VEGFR) and platelet-derived growth factor receptor (PDGFR)[94]. A phase II study investigated the efficacy of sorafenib in combination with docetaxel and cisplatin as the first-line chemotherapy in metastatic gastric or EGJ adenocarcinoma. Partial response was achieved in 41% (90%CI: 28%-54%), and the median PFS was 5.8 mo (90%CI: 5.4-7.4 mo) and median OS was 13.6 mo (90%CI: 8.6-16.1 mo). No additional toxicities were observed by adding sorafenib to docetaxel/cisplatin regimen. The results of this study warranted further evaluation of sorafenib in chemotherapy of gastric cancer[95]. However, another multicenter phase II study of oxaliplatin and sorafenib as the second-line chemotherapy after failure of cisplatin/fluoropyrimidine regimen in advanced gastric adenocarcinoma revealed a median PFS of 3.0 mo (95%CI: 2.3-4.1 mo) and median OS of 6.5 mo (95%CI: 5.2-9.7 mo), which failed to support the implementation of a phase III study[96]. Sorafenib was also evaluated for combination therapy with oral fluoropyrimidine and cisplatin, such as S-1/cisplatin[97] and capecitabine/cisplatin[98], in phase I studies. Both studies showed tolerable safety profile and acceptable efficacy.
Lapatinib is a MKI that competitively inhibits ATP binding of tyrosine kinase in both HER2 and EGFR, and which is approved for the treatment of HER2-positive breast cancer[99]. Two large-scale, randomized, phase III trials were recently reported. The researchers evaluated the efficacy and safety of lapatinib in HER2-positive, advanced or metastatic gastric and EGJ adenocarcinoma. The LOGiC study addressed lapatinib as the first-line chemotherapy in combination with capecitabine/oxaliplatin, and lapatinib arm was compared with capecitabine/oxaliplatin alone[100]. Median OS was not significant between both arms (12.2 mo vs 10.5 mo, P = 0.349), while PFS was significantly longer (6.0 mo vs 5.3 mo, P = 0.0381) and ORR was higher (53% vs 39%, P = 0.0031) in the lapatinib arm. Subgroup analysis for OS revealed that Asians and younger patients (< 60 years) showed significant benefit. The TyTAN study compared lapatinib plus paclitaxel with paclitaxel alone in the second-line treatment of gastric cancer in an Asian population[101]. This study showed no significant difference of median OS and PFS between both arms (11.0 mo vs 8.9 mo, P = 0.1044; 5.4 mo vs 4.4 mo, P = 0.2441; respectively). However, better efficacy was observed in the lapatinib arm in HER2-3+ subgroup. Further studies are warranted to examine the factors predicting good responders to lapatinib therapy.
Several novel MKIs have been investigated for the treatment of refractory gastric cancer. Findings were presented at the ASCO 2015 meeting. Among them, regorafenib, which inhibits multiple tyrosine kinases related to angiogenesis (VEGFR1-3), tumor microenvironment [PDGFR-β, fibroblast growth factor receptor (FGFR)] and oncogenesis (KIT), was previously developed and reported as effective in colon cancer and gastrointestinal stromal tumors (GISTs)[102]. The phase II INTEGRATE study was designed and performed to investigate the efficacy of regorafenib in refractory, metastatic gastric and EGJ adenocarcinoma by comparing regorafenib 160 mg/d with placebo[103]. PFS was significantly increased in regrafenib group (2.6 mo vs 0.9 mo, HR = 0.40, P < 0.0001), however, OS was not significantly different between two groups (5.8 mo vs 4.5 mo, HR = 0.74, P = 0.11). An interesting thing is that HR = for PFS was significantly lower in Korean patients than in Western patients from Canada and Australia, which indicates that regorafenib might be more effective in Asian patients. Pazopanib is another potent MKI of VEGFR1-3, PDGFRα/β and FGFR1/3, and was previously approved by the United States Food and Drug Administration for the treatment of patients with advanced renal cell carcinoma[104]. A phase II study was performed and reported the combinatory effect of pazopanib with capecitabine/oxaliplatin regimen as the first-line chemotherapy in metastatic gastric and EGJ cancer. ORR was 57.6% and adverse events of grade 3-4 were neutropenia (15.1%), anemia and thrombocytopenia (both 10.6%)[105].
Many studies have focused on revealing biologic relevant mechanism of development and progression of gastric cancer, and many medical agents targeting these pathways have been validated in clinical trials. However, most of them failed to reach significant benefits in phase III trials, and novel therapeutic strategies are necessary in the future. To achieve this goal, individualized and precise target therapy should be planned on the basis of exploration of biologic characteristics of individual gastric cancer patients. In addition, targeting multiple RTKs rather than focusing on single pathway and attempts to overcome chemoresistance and enhance synergism with standard chemotherapeutic agents are expected to be prevalent. These approaches will hopefully lead to a more effective treatment, perhaps even conquest, of gastric cancer.
| 1. | Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137-2150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2679] [Cited by in RCA: 2561] [Article Influence: 128.1] [Reference Citation Analysis (3)] |
| 2. | Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202-209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5511] [Cited by in RCA: 5152] [Article Influence: 429.3] [Reference Citation Analysis (6)] |
| 3. | Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5933] [Cited by in RCA: 5471] [Article Influence: 341.9] [Reference Citation Analysis (5)] |
| 4. | Wadhwa R, Song S, Lee JS, Yao Y, Wei Q, Ajani JA. Gastric cancer-molecular and clinical dimensions. Nat Rev Clin Oncol. 2013;10:643-655. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 335] [Cited by in RCA: 341] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 5. | Blumenschein GR, Mills GB, Gonzalez-Angulo AM. Targeting the hepatocyte growth factor-cMET axis in cancer therapy. J Clin Oncol. 2012;30:3287-3296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 224] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 6. | Samamé Pérez-Vargas JC, Biondani P, Maggi C, Gariboldi M, Gloghini A, Inno A, Volpi CC, Gualeni AV, di Bartolomeo M, de Braud F. Role of cMET in the development and progression of colorectal cancer. Int J Mol Sci. 2013;14:18056-18077. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 7. | Gu H, Neel BG. The “Gab” in signal transduction. Trends Cell Biol. 2003;13:122-130. [PubMed] |
| 8. | Yu S, Yu Y, Zhao N, Cui J, Li W, Liu T. C-Met as a prognostic marker in gastric cancer: a systematic review and meta-analysis. PLoS One. 2013;8:e79137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Peng Z, Li Z, Gao J, Lu M, Gong J, Tang ET, Oliner KS, Hei YJ, Zhou H, Shen L. Tumor MET Expression and Gene Amplification in Chinese Patients with Locally Advanced or Metastatic Gastric or Gastroesophageal Junction Cancer. Mol Cancer Ther. 2015;14:2634-2641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 10. | Nakajima M, Sawada H, Yamada Y, Watanabe A, Tatsumi M, Yamashita J, Matsuda M, Sakaguchi T, Hirao T, Nakano H. The prognostic significance of amplification and overexpression of c-met and c-erb B-2 in human gastric carcinomas. Cancer. 1999;85:1894-1902. [PubMed] |
| 11. | Janjigian YY, Tang LH, Coit DG, Kelsen DP, Francone TD, Weiser MR, Jhanwar SC, Shah MA. MET expression and amplification in patients with localized gastric cancer. Cancer Epidemiol Biomarkers Prev. 2011;20:1021-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 136] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 12. | Lennerz JK, Kwak EL, Ackerman A, Michael M, Fox SB, Bergethon K, Lauwers GY, Christensen JG, Wilner KD, Haber DA. MET amplification identifies a small and aggressive subgroup of esophagogastric adenocarcinoma with evidence of responsiveness to crizotinib. J Clin Oncol. 2011;29:4803-4810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 369] [Cited by in RCA: 369] [Article Influence: 24.6] [Reference Citation Analysis (3)] |
| 13. | Scagliotti G, von Pawel J, Novello S, Ramlau R, Favaretto A, Barlesi F, Akerley W, Orlov S, Santoro A, Spigel D. Phase III Multinational, Randomized, Double-Blind, Placebo-Controlled Study of Tivantinib (ARQ 197) Plus Erlotinib Versus Erlotinib Alone in Previously Treated Patients With Locally Advanced or Metastatic Nonsquamous Non-Small-Cell Lung Cancer. J Clin Oncol. 2015;33:2667-2674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 230] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 14. | Kang YK, Muro K, Ryu MH, Yasui H, Nishina T, Ryoo BY, Kamiya Y, Akinaga S, Boku N. A phase II trial of a selective c-Met inhibitor tivantinib (ARQ 197) monotherapy as a second- or third-line therapy in the patients with metastatic gastric cancer. Invest New Drugs. 2014;32:355-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 98] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 15. | Matsuoka T, Yashiro M. The Role of PI3K/Akt/mTOR Signaling in Gastric Carcinoma. Cancers (Basel). 2014;6:1441-1463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 186] [Cited by in RCA: 175] [Article Influence: 14.6] [Reference Citation Analysis (4)] |
| 16. | Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell. 2007;129:1261-1274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5039] [Cited by in RCA: 4861] [Article Influence: 255.8] [Reference Citation Analysis (4)] |
| 17. | Samuels Y, Wang Z, Bardelli A, Silliman N, Ptak J, Szabo S, Yan H, Gazdar A, Powell SM, Riggins GJ. High frequency of mutations of the PIK3CA gene in human cancers. Science. 2004;304:554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2973] [Cited by in RCA: 2747] [Article Influence: 124.9] [Reference Citation Analysis (3)] |
| 18. | Li VS, Wong CW, Chan TL, Chan AS, Zhao W, Chu KM, So S, Chen X, Yuen ST, Leung SY. Mutations of PIK3CA in gastric adenocarcinoma. BMC Cancer. 2005;5:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 140] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 19. | Velho S, Oliveira C, Ferreira A, Ferreira AC, Suriano G, Schwartz S, Duval A, Carneiro F, Machado JC, Hamelin R. The prevalence of PIK3CA mutations in gastric and colon cancer. Eur J Cancer. 2005;41:1649-1654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 268] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 20. | Shi J, Yao D, Liu W, Wang N, Lv H, Zhang G, Ji M, Xu L, He N, Shi B. Highly frequent PIK3CA amplification is associated with poor prognosis in gastric cancer. BMC Cancer. 2012;12:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 128] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 21. | Tran TN, Brettingham-Moore K, Duong CP, Mitchell C, Clemons NJ, Phillips WA. Molecular changes in the phosphatidylinositide 3-kinase (PI3K) pathway are common in gastric cancer. J Surg Oncol. 2013;108:113-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 22. | Sukawa Y, Yamamoto H, Nosho K, Ito M, Igarashi H, Naito T, Mitsuhashi K, Matsunaga Y, Takahashi T, Mikami M. HER2 expression and PI3K-Akt pathway alterations in gastric cancer. Digestion. 2014;89:12-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 23. | Chong ML, Loh M, Thakkar B, Pang B, Iacopetta B, Soong R. Phosphatidylinositol-3-kinase pathway aberrations in gastric and colorectal cancer: meta-analysis, co-occurrence and ethnic variation. Int J Cancer. 2014;134:1232-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 24. | Mina S, Bohn BA, Simon R, Krohn A, Reeh M, Arnold D, Bokemeyer C, Sauter G, Izbicki JR, Marx A. PTEN deletion is rare but often homogeneous in gastric cancer. J Clin Pathol. 2012;65:693-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 25. | Oki E, Tokunaga E, Nakamura T, Ueda N, Futatsugi M, Mashino K, Yamamoto M, Watanabe M, Ikebe M, Kakeji Y. Genetic mutual relationship between PTEN and p53 in gastric cancer. Cancer Lett. 2005;227:33-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 26. | Byun DS, Cho K, Ryu BK, Lee MG, Park JI, Chae KS, Kim HJ, Chi SG. Frequent monoallelic deletion of PTEN and its reciprocal associatioin with PIK3CA amplification in gastric carcinoma. Int J Cancer. 2003;104:318-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 144] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 27. | Li YL, Tian Z, Wu DY, Fu BY, Xin Y. Loss of heterozygosity on 10q23.3 and mutation of tumor suppressor gene PTEN in gastric cancer and precancerous lesions. World J Gastroenterol. 2005;11:285-288. [PubMed] [DOI] [Full Text] |
| 28. | Oki E, Baba H, Tokunaga E, Nakamura T, Ueda N, Futatsugi M, Mashino K, Yamamoto M, Ikebe M, Kakeji Y. Akt phosphorylation associates with LOH of PTEN and leads to chemoresistance for gastric cancer. Int J Cancer. 2005;117:376-380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 144] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 29. | Oki E, Kakeji Y, Baba H, Tokunaga E, Nakamura T, Ueda N, Futatsugi M, Yamamoto M, Ikebe M, Maehara Y. Impact of loss of heterozygosity of encoding phosphate and tensin homolog on the prognosis of gastric cancer. J Gastroenterol Hepatol. 2006;21:814-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Yu G, Wang J, Chen Y, Wang X, Pan J, Li G, Jia Z, Li Q, Yao JC, Xie K. Overexpression of phosphorylated mammalian target of rapamycin predicts lymph node metastasis and prognosis of chinese patients with gastric cancer. Clin Cancer Res. 2009;15:1821-1829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 124] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 31. | Li M, Sun H, Song L, Gao X, Chang W, Qin X. Immunohistochemical expression of mTOR negatively correlates with PTEN expression in gastric carcinoma. Oncol Lett. 2012;4:1213-1218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 32. | Li M; ClinicalTrials. gov. PI3K Inhibitor BYL719 in Combination With the HSP90 Inhibitor AUY922 in Patients With Advanced or Metastatic Gastric Cancer. (accessed January 7, 2016). Available from: http://www.clinicaltrials.gov/ct/show/NCT01613950. |
| 33. | Yu H, Lee H, Herrmann A, Buettner R, Jove R. Revisiting STAT3 signalling in cancer: new and unexpected biological functions. Nat Rev Cancer. 2014;14:736-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1798] [Cited by in RCA: 1726] [Article Influence: 143.8] [Reference Citation Analysis (3)] |
| 34. | Jackson CB, Giraud AS. STAT3 as a prognostic marker in human gastric cancer. J Gastroenterol Hepatol. 2009;24:505-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 35. | Giraud AS, Menheniott TR, Judd LM. Targeting STAT3 in gastric cancer. Expert Opin Ther Targets. 2012;16:889-901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 36. | Iida T, Iwahashi M, Katsuda M, Ishida K, Nakamori M, Nakamura M, Naka T, Ojima T, Ueda K, Hayata K. Tumor-infiltrating CD4+ Th17 cells produce IL-17 in tumor microenvironment and promote tumor progression in human gastric cancer. Oncol Rep. 2011;25:1271-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 65] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 37. | Liu X, Jin H, Zhang G, Lin X, Chen C, Sun J, Zhang Y, Zhang Q, Yu J. Intratumor IL-17-positive mast cells are the major source of the IL-17 that is predictive of survival in gastric cancer patients. PLoS One. 2014;9:e106834. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (4)] |
| 38. | Wu X, Yang T, Liu X, Guo JN, Xie T, Ding Y, Lin M, Yang H. IL-17 promotes tumor angiogenesis through Stat3 pathway mediated upregulation of VEGF in gastric cancer. Tumour Biol. 2016;37:5493-5501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 39. | Howlett M, Giraud AS, Lescesen H, Jackson CB, Kalantzis A, Van Driel IR, Robb L, Van der Hoek M, Ernst M, Minamoto T. The interleukin-6 family cytokine interleukin-11 regulates homeostatic epithelial cell turnover and promotes gastric tumor development. Gastroenterology. 2009;136:967-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 74] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 40. | Han JC, Zhang KL, Chen XY, Jiang HF, Kong QY, Sun Y, Wu ML, Huang L, Li H, Liu J. Expression of seven gastric cancer-associated genes and its relevance for Wnt, NF-kappaB and Stat3 signaling. APMIS. 2007;115:1331-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 41. | Rhee YH, Jeong SJ, Lee HJ, Lee HJ, Koh W, Jung JH, Kim SH, Sung-Hoon K. Inhibition of STAT3 signaling and induction of SHP1 mediate antiangiogenic and antitumor activities of ergosterol peroxide in U266 multiple myeloma cells. BMC Cancer. 2012;12:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 42. | Sandur SK, Pandey MK, Sung B, Aggarwal BB. 5-hydroxy-2-methyl-1,4-naphthoquinone, a vitamin K3 analogue, suppresses STAT3 activation pathway through induction of protein tyrosine phosphatase, SHP-1: potential role in chemosensitization. Mol Cancer Res. 2010;8:107-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 67] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 43. | Lee JH, Chiang SY, Nam D, Chung WS, Lee J, Na YS, Sethi G, Ahn KS. Capillarisin inhibits constitutive and inducible STAT3 activation through induction of SHP-1 and SHP-2 tyrosine phosphatases. Cancer Lett. 2014;345:140-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 131] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 44. | Chen KF, Tai WT, Liu TH, Huang HP, Lin YC, Shiau CW, Li PK, Chen PJ, Cheng AL. Sorafenib overcomes TRAIL resistance of hepatocellular carcinoma cells through the inhibition of STAT3. Clin Cancer Res. 2010;16:5189-5199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 145] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 45. | Tai WT, Cheng AL, Shiau CW, Huang HP, Huang JW, Chen PJ, Chen KF. Signal transducer and activator of transcription 3 is a major kinase-independent target of sorafenib in hepatocellular carcinoma. J Hepatol. 2011;55:1041-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 134] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 46. | Chen KF, Tai WT, Hsu CY, Huang JW, Liu CY, Chen PJ, Kim I, Shiau CW. Blockade of STAT3 activation by sorafenib derivatives through enhancing SHP-1 phosphatase activity. Eur J Med Chem. 2012;55:220-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 47. | Tai WT, Shiau CW, Chen PJ, Chu PY, Huang HP, Liu CY, Huang JW, Chen KF. Discovery of novel Src homology region 2 domain-containing phosphatase 1 agonists from sorafenib for the treatment of hepatocellular carcinoma. Hepatology. 2014;59:190-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 48. | Huang CY, Tai WT, Hsieh CY, Hsu WM, Lai YJ, Chen LJ, Shiau CW, Chen KF. A sorafenib derivative and novel SHP-1 agonist, SC-59, acts synergistically with radiotherapy in hepatocellular carcinoma cells through inhibition of STAT3. Cancer Lett. 2014;349:136-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 49. | Fan LC, Shiau CW, Tai WT, Hung MH, Chu PY, Hsieh FS, Lin H, Yu HC, Chen KF. SHP-1 is a negative regulator of epithelial-mesenchymal transition in hepatocellular carcinoma. Oncogene. 2015;34:5252-5263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 50. | Su TH, Shiau CW, Jao P, Liu CH, Liu CJ, Tai WT, Jeng YM, Yang HC, Tseng TC, Huang HP. Sorafenib and its derivative SC-1 exhibit antifibrotic effects through signal transducer and activator of transcription 3 inhibition. Proc Natl Acad Sci USA. 2015;112:7243-7248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (3)] |
| 51. | Chao TI, Tai WT, Hung MH, Tsai MH, Chen MH, Chang MJ, Shiau CW, Chen KF. A combination of sorafenib and SC-43 is a synergistic SHP-1 agonist duo to advance hepatocellular carcinoma therapy. Cancer Lett. 2016;371:205-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 52. | Joo MK, Park JJ, Yoo HS, Lee BJ, Chun HJ, Lee SW, Bak YT. Epigenetic regulation and anti-tumorigenic effects of SH2-containing protein tyrosine phosphatase 1 (SHP1) in human gastric cancer cells. Tumour Biol. 2016;37:4603-4612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 53. | Abdel-Rahman O. Immune checkpoints aberrations and gastric cancer; assessment of prognostic value and evaluation of therapeutic potentials. Crit Rev Oncol Hematol. 2016;97:65-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 54. | Chen L, Han X. Anti-PD-1/PD-L1 therapy of human cancer: past, present, and future. J Clin Invest. 2015;125:3384-3391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1260] [Cited by in RCA: 1190] [Article Influence: 108.2] [Reference Citation Analysis (3)] |
| 55. | Wu C, Zhu Y, Jiang J, Zhao J, Zhang XG, Xu N. Immunohistochemical localization of programmed death-1 ligand-1 (PD-L1) in gastric carcinoma and its clinical significance. Acta Histochem. 2006;108:19-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 394] [Article Influence: 19.7] [Reference Citation Analysis (3)] |
| 56. | Lu B, Chen L, Liu L, Zhu Y, Wu C, Jiang J, Zhang X. T-cell-mediated tumor immune surveillance and expression of B7 co-inhibitory molecules in cancers of the upper gastrointestinal tract. Immunol Res. 2011;50:269-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 57. | Hou J, Yu Z, Xiang R, Li C, Wang L, Chen S, Li Q, Chen M, Wang L. Correlation between infiltration of FOXP3+ regulatory T cells and expression of B7-H1 in the tumor tissues of gastric cancer. Exp Mol Pathol. 2014;96:284-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 118] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 58. | Qing Y, Li Q, Ren T, Xia W, Peng Y, Liu GL, Luo H, Yang YX, Dai XY, Zhou SF. Upregulation of PD-L1 and APE1 is associated with tumorigenesis and poor prognosis of gastric cancer. Drug Des Devel Ther. 2015;9:901-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 155] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 59. | Saito H, Kuroda H, Matsunaga T, Osaki T, Ikeguchi M. Increased PD-1 expression on CD4+ and CD8+ T cells is involved in immune evasion in gastric cancer. J Surg Oncol. 2013;107:517-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 60. | Kim JW, Nam KH, Ahn SH, Park do J, Kim HH, Kim SH, Chang H, Lee JO, Kim YJ, Lee HS. Prognostic implications of immunosuppressive protein expression in tumors as well as immune cell infiltration within the tumor microenvironment in gastric cancer. Gastric Cancer. 2016;19:42-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 206] [Article Influence: 20.6] [Reference Citation Analysis (1)] |
| 61. | Geng R, Dai C, Wong A, Qing M, Hu J, Sun Y, Lo AWI, Li J. Prognostic significance of tumor infiltrating immune cells and PD-L1 expression in gastric carcinoma in Chinese patients. J Clin Oncol. 2015;33 Suppl 15:4042. |
| 62. | Iveson T, Donehower RC, Davidenko I, Tjulandin S, Deptala A, Harrison M, Nirni S, Lakshmaiah K, Thomas A, Jiang Y. Rilotumumab in combination with epirubicin, cisplatin, and capecitabine as first-line treatment for gastric or oesophagogastric junction adenocarcinoma: an open-label, dose de-escalation phase 1b study and a double-blind, randomised phase 2 study. Lancet Oncol. 2014;15:1007-1018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 216] [Article Influence: 18.0] [Reference Citation Analysis (3)] |
| 63. | Cunningham D, Tebbutt NC, Davidenko I, Murad AM, Al-Batran S-E, Ilson DH, Tjulandin S, Gotovkin E, Karaszewska B, Bondarenko I. Phase III, randomized, double-blind, multicenter, placebo (P)-controlled trial of rilotumumab (R) plus epirubicin, cisplatin and capecitabine (ECX) as first-line therapy in patients (pts) with advanced MET-positive (pos) gastric or gastroesophageal junction (G/GEJ) cancer: RILOMET-1 study. J Clin Oncol. 2015;33 Suppl 15:4000. |
| 64. | Doi T, Kang YK, Muro K, Jiang Y, Jain RK, Lizambri R. A phase 3, multicenter, randomized, double-blind, placebo-controlled study of rilotumumab in combination with cisplatin and capecitabine (CX) as first-line therapy for Asian patients (pts) with advanced MET-positive gastric or gastroesophageal junction (G/GEJ) adenocarcinoma: The RILOMET-2 trial. J Clin Oncol. 2015;33 Suppl 15:TPS226. |
| 65. | Shah MA, Bang YJ, Lordick F, Tabernero J, Chen M, Hack SP, Phan SC, Shames DS, Cunningham D. METGastric: A phase III study of onartuzumab plus mFOLFOX6 in patients with metastatic HER2-negative (HER2-) and MET-positive (MET ) adenocarcinoma of the stomach or gastroesophageal junction (GEC). J Clin Oncol. 2015;33 Suppl 15:4012. |
| 66. | Malka D, Castan F, Francois E, Bouche O, Bennouna J, Ghiringhelli F, Fouchardiere CDL, Borg C, Samalin E, Bachet JB, Raoul J-L, Cvitkovic F, Miglianico L, Bengrine-Lefevre L, Dahan L, Lecaille C, Aparicio T, Perrier H, Gourgou S, Taïeb J, Unicancer, (FFCD) FFdCD, (AGEO) AdG-EO. FOLFOX alone or combined to rilotumumab or panitumumab as first-line treatment in patients (pts) with advanced gastroesophageal adenocarcinoma (AGEA): An open-label, randomized phase II trial (PRODIGE 17 ACCORD 20 MEGA). J Clin Oncol. 2015;33 Suppl 15:4013. |
| 67. | Xu RH, Qiu M, Zhou YX, Wang DS, Zhang DS, Wang F, Li YH, Huang J, Zhou H, Tang ET. Evaluation of tumor MET protein expression, MET gene amplification, and HER2 expression in Chinese patients with advanced gastric or gastroesophageal junction (G/GEJ) cancer. J Clin Oncol. 2015;33 Suppl:abstr 4048. |
| 68. | Hudis C, Swanton C, Janjigian YY, Lee R, Sutherland S, Lehman R, Chandarlapaty S, Hamilton N, Gajria D, Knowles J. A phase 1 study evaluating the combination of an allosteric AKT inhibitor (MK-2206) and trastuzumab in patients with HER2-positive solid tumors. Breast Cancer Res. 2013;15:R110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 90] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 69. | Doi T, Muro K, Boku N, Yamada Y, Nishina T, Takiuchi H, Komatsu Y, Hamamoto Y, Ohno N, Fujita Y. Multicenter phase II study of everolimus in patients with previously treated metastatic gastric cancer. J Clin Oncol. 2010;28:1904-1910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 157] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 70. | Ohtsu A, Ajani JA, Bai YX, Bang YJ, Chung HC, Pan HM, Sahmoud T, Shen L, Yeh KH, Chin K. Everolimus for previously treated advanced gastric cancer: results of the randomized, double-blind, phase III GRANITE-1 study. J Clin Oncol. 2013;31:3935-3943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 383] [Cited by in RCA: 380] [Article Influence: 29.2] [Reference Citation Analysis (1)] |
| 71. | Shen YC, Li CP, Yen CJ, Hsu C, Lin YL, Lin ZZ, Chen LT, Su WC, Chao Y, Yeh KH. Phase II multicentered study of low-dose everolimus plus cisplatin and weekly 24-hour infusion of high-dose 5-fluorouracil and leucovorin as first-line treatment for patients with advanced gastric cancer. Oncology. 2014;87:104-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 72. | Park JH, Ryu MH, Park YS, Park SR, Na YS, Rhoo BY, Kang YK. Successful control of heavily pretreated metastatic gastric cancer with the mTOR inhibitor everolimus (RAD001) in a patient with PIK3CA mutation and pS6 overexpression. BMC Cancer. 2015;15:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 73. | Wainberg ZA, Soares HP, Patel R, DiCarlo B, Park DJ, Liem A, Wang HJ, Yonemoto L, Martinez D, Laux I. Phase II trial of everolimus in patients with refractory metastatic adenocarcinoma of the esophagus, gastroesophageal junction and stomach: possible role for predictive biomarkers. Cancer Chemother Pharmacol. 2015;76:61-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 74. | Oh DY, Lee SH, Han SW, Kim MJ, Kim TM, Kim TY, Heo DS, Yuasa M, Yanagihara Y, Bang YJ. Phase I Study of OPB-31121, an Oral STAT3 Inhibitor, in Patients with Advanced Solid Tumors. Cancer Res Treat. 2015;47:607-615. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 108] [Article Influence: 9.8] [Reference Citation Analysis (4)] |
| 75. | Huang S, Chen M, Ding X, Zhang X, Zou X. Proton pump inhibitor selectively suppresses proliferation and restores the chemosensitivity of gastric cancer cells by inhibiting STAT3 signaling pathway. Int Immunopharmacol. 2013;17:585-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 76. | Pandey A, Vishnoi K, Mahata S, Tripathi SC, Misra SP, Misra V, Mehrotra R, Dwivedi M, Bharti AC. Berberine and Curcumin Target Survivin and STAT3 in Gastric Cancer Cells and Synergize Actions of Standard Chemotherapeutic 5-Fluorouracil. Nutr Cancer. 2015;67:1293-1304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 95] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 77. | Zhang B, Yang Y, Shi X, Liao W, Chen M, Cheng AS, Yan H, Fang C, Zhang S, Xu G. Proton pump inhibitor pantoprazole abrogates adriamycin-resistant gastric cancer cell invasiveness via suppression of Akt/GSK-β/β-catenin signaling and epithelial-mesenchymal transition. Cancer Lett. 2015;356:704-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 78. | Chen J, Wang J, Lin L, He L, Wu Y, Zhang L, Yi Z, Chen Y, Pang X, Liu M. Inhibition of STAT3 signaling pathway by nitidine chloride suppressed the angiogenesis and growth of human gastric cancer. Mol Cancer Ther. 2012;11:277-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 131] [Article Influence: 9.4] [Reference Citation Analysis (3)] |
| 79. | Liu SH, Wang KB, Lan KH, Lee WJ, Pan HC, Wu SM, Peng YC, Chen YC, Shen CC, Cheng HC. Calpain/SHP-1 interaction by honokiol dampening peritoneal dissemination of gastric cancer in nu/nu mice. PLoS One. 2012;7:e43711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 80. | Zhu BH, Chen HY, Zhan WH, Wang CY, Cai SR, Wang Z, Zhang CH, He YL. (-)-Epigallocatechin-3-gallate inhibits VEGF expression induced by IL-6 via Stat3 in gastric cancer. World J Gastroenterol. 2011;17:2315-2325. [PubMed] [DOI] [Full Text] |
| 81. | Huang S, Chen M, Shen Y, Shen W, Guo H, Gao Q, Zou X. Inhibition of activated Stat3 reverses drug resistance to chemotherapeutic agents in gastric cancer cells. Cancer Lett. 2012;315:198-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 82. | Gupta SC, Phromnoi K, Aggarwal BB. Morin inhibits STAT3 tyrosine 705 phosphorylation in tumor cells through activation of protein tyrosine phosphatase SHP1. Biochem Pharmacol. 2013;85:898-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 83. | Ahn KS, Sethi G, Sung B, Goel A, Ralhan R, Aggarwal BB. Guggulsterone, a farnesoid X receptor antagonist, inhibits constitutive and inducible STAT3 activation through induction of a protein tyrosine phosphatase SHP-1. Cancer Res. 2008;68:4406-4415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 112] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 84. | Prasad S, Pandey MK, Yadav VR, Aggarwal BB. Gambogic acid inhibits STAT3 phosphorylation through activation of protein tyrosine phosphatase SHP-1: potential role in proliferation and apoptosis. Cancer Prev Res (Phila). 2011;4:1084-1094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 85. | Pandey MK, Sung B, Ahn KS, Aggarwal BB. Butein suppresses constitutive and inducible signal transducer and activator of transcription (STAT) 3 activation and STAT3-regulated gene products through the induction of a protein tyrosine phosphatase SHP-1. Mol Pharmacol. 2009;75:525-533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 94] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 86. | Rajendran P, Li F, Manu KA, Shanmugam MK, Loo SY, Kumar AP, Sethi G. γ-Tocotrienol is a novel inhibitor of constitutive and inducible STAT3 signalling pathway in human hepatocellular carcinoma: potential role as an antiproliferative, pro-apoptotic and chemosensitizing agent. Br J Pharmacol. 2011;163:283-298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 113] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 87. | Tan SM, Li F, Rajendran P, Kumar AP, Hui KM, Sethi G. Identification of beta-escin as a novel inhibitor of signal transducer and activator of transcription 3/Janus-activated kinase 2 signaling pathway that suppresses proliferation and induces apoptosis in human hepatocellular carcinoma cells. J Pharmacol Exp Ther. 2010;334:285-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 118] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 88. | Joo MK, Park JJ, Kim SH, Yoo HS, Lee BJ, Chun HJ, Lee SW, Bak YT. Antitumorigenic effect of plumbagin by induction of SH2-containing protein tyrosine phosphatase 1 in human gastric cancer cells. Int J Oncol. 2015;46:2380-2388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 89. | Ralph C, Elkord E, Burt DJ, O’Dwyer JF, Austin EB, Stern PL, Hawkins RE, Thistlethwaite FC. Modulation of lymphocyte regulation for cancer therapy: a phase II trial of tremelimumab in advanced gastric and esophageal adenocarcinoma. Clin Cancer Res. 2010;16:1662-1672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 202] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 90. | Bang YJ, Chung HC, Shankaran V, Geva R, Catenacci DVT, Gupta S, Eder JP, Berger R, Gonzalez EJ, Ray A. Relationship between PD-L1 expression and clinical outcomes in patients with advanced gastric cancer treated with the anti-PD-1 monoclonal antibody pembrolizumab (MK-3475) in KEYNOTE-012. J Clin Oncol. 2015;33 Suppl 15:4001. |
| 91. | Bang YJ; ClinicalTrials. gov. A Study of Pembrolizumab (MK-3475) in Participants With Recurrent or Metastatic Gastric or Gastroesophageal Junction Adenocarcinoma (MK-3475-059/KEYNOTE-059) (accessed January 4, 2016). Available from: http://www.clinicaltrials.gov/ct/show/NCT02335411. |
| 92. | Bang YJ; ClinicalTrials. gov. A Study of Pembrolizumab (MK-3475) Versus Paclitaxel for Participants With Advanced Gastric/Gastroesophageal Junction Adenocarcinoma That Progressed After Therapy With Platinum and Fluoropyrimidine (MK-3475-061/KEYNOTE-061) (accessed January 4, 2016). Available from: http://www.clinicaltrials.gov/ct/show/NCT02370498. |
| 93. | Yamada Y, Nishina T, Iwasa S, Shitara K, Muro K, Esaki T, Hironaka S, Yamaguchi K, Machida N, Satoh T. A phase I dose expansion trial of avelumab (MSB0010718C), an anti-PD-L1 antibody, in Japanese patients with advanced gastric cancer. J Clin Oncol. 2015;33 Suppl 15:4047. |
| 94. | Morishita A, Gong J, Masaki T. Targeting receptor tyrosine kinases in gastric cancer. World J Gastroenterol. 2014;20:4536-4545. [PubMed] [DOI] [Full Text] |
| 95. | Sun W, Powell M, O’Dwyer PJ, Catalano P, Ansari RH, Benson AB. Phase II study of sorafenib in combination with docetaxel and cisplatin in the treatment of metastatic or advanced gastric and gastroesophageal junction adenocarcinoma: ECOG 5203. J Clin Oncol. 2010;28:2947-2951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 172] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 96. | Martin-Richard M, Gallego R, Pericay C, Garcia Foncillas J, Queralt B, Casado E, Barriuso J, Iranzo V, Juez I, Visa L. Multicenter phase II study of oxaliplatin and sorafenib in advanced gastric adenocarcinoma after failure of cisplatin and fluoropyrimidine treatment. A GEMCAD study. Invest New Drugs. 2013;31:1573-1579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 97. | Yamada Y, Kiyota N, Fuse N, Kato K, Minami H, Hashizume K, Kuroki Y, Ito Y, Ohtsu A. A phase I study of sorafenib in combination with S-1 plus cisplatin in patients with advanced gastric cancer. Gastric Cancer. 2014;17:161-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 98. | Kim C, Lee JL, Choi YH, Kang BW, Ryu MH, Chang HM, Kim TW, Kang YK. Phase I dose-finding study of sorafenib in combination with capecitabine and cisplatin as a first-line treatment in patients with advanced gastric cancer. Invest New Drugs. 2012;30:306-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 99. | Zheng J, de Guia T, Wang-Jairaj J, Newlands AH, Wang C, Crim C, Zhong N. Efficacy and safety of fluticasone furoate/vilanterol (50/25 mcg; 100/25 mcg; 200/25 mcg) in Asian patients with chronic obstructive pulmonary disease: a randomized placebo-controlled trial. Curr Med Res Opin. 2015;31:1191-1200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 100. | Hecht JR, Bang YJ, Qin SK, Chung HC, Xu JM, Park JO, Jeziorski K, Shparyk Y, Hoff PM, Sobrero A. Lapatinib in Combination With Capecitabine Plus Oxaliplatin in Human Epidermal Growth Factor Receptor 2-Positive Advanced or Metastatic Gastric, Esophageal, or Gastroesophageal Adenocarcinoma: TRIO-013/LOGiC-A Randomized Phase III Trial. J Clin Oncol. 2016;34:443-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 469] [Article Influence: 46.9] [Reference Citation Analysis (1)] |
| 101. | Satoh T, Xu RH, Chung HC, Sun GP, Doi T, Xu JM, Tsuji A, Omuro Y, Li J, Wang JW. Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN--a randomized, phase III study. J Clin Oncol. 2014;32:2039-2049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 541] [Cited by in RCA: 515] [Article Influence: 42.9] [Reference Citation Analysis (6)] |
| 102. | Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381:303-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2394] [Cited by in RCA: 2212] [Article Influence: 170.2] [Reference Citation Analysis (6)] |
| 103. | Pavlakis N, Sjoquist KM, Tsobanis E, Martin AJ, Kang YK, Bang YJ, O’Callaghan CJ, Tebbutt NC, Rha SY, Lee J. INTEGRATE: A randomized, phase II, double-blind, placebo-controlled study of regorafenib in refractory advanced oesophagogastric cancer (AOGC): A study by the Australasian Gastrointestinal Trials Group (AGITG)-Final overall and subgroup results. J Clin Oncol. 2015;33 Suppl 15:4003. |
| 104. | Bukowski RM, Yasothan U, Kirkpatrick P. Pazopanib. Nat Rev Drug Discov. 2010;9:17-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 93] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 105. | Thuss-Patience PC, Al-Batran SE, Siveke JT, Homann N, Malfertheiner P, Glaeser D, Stein A, Tamm I, Daum S, Potenberg J. Pazopanib and 5-FU/oxaliplatin as first-line treatment in advanced gastric cancer: PaFLO, a randomized phase II study from the AIO (Arbeitsgemeinschaft Internistische Onkologie). J Clin Oncol. 2015;33 Suppl 15:4033. |
| 106. | Lee MY, Kim ST, Lee J, Park SH, Park YS, Lim HY, Kang WK, Park JO. Prospective phase II trial of pazopanib plus CapeOX (capecitabine and oxaliplatin) in previously untreated patients with advanced gastric cancer. J Clin Oncol. 2015;33 Suppl 15:4049. |
| 107. | Lee MY; ClinicalTrials. gov. Phase II Study of Neoadjuvant XELOX Lapatinib in HER2(+) Gastric Cancer Patients With Liver Metastasis (accessed January 14, 2016). Available from: http://www.clinicaltrials.gov/ct/show/NCT02015169. |
| 108. | Lee MY; ClinicalTrials. gov. FOLFOX Plus Regorafenib in Patients With Unresectable or Metastatic Esophagogastric Cancer (accessed January 14, 2016). Available from: http://www.clinicaltrials.gov/ct/show/NCT01913639. |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Balla A, Kondoh C S- Editor: Yu J L- Editor: A E- Editor: Ma S