Published online Mar 28, 2015. doi: 10.3748/wjg.v21.i12.3579
Peer-review started: August 28, 2014
First decision: September 27, 2014
Revised: December 23, 2014
Accepted: January 16, 2015
Article in press: January 16, 2015
Published online: March 28, 2015
Processing time: 215 Days and 7.7 Hours
AIM: To evaluate the techniques, results, and complications related to computed tomography (CT)-guided percutaneous core needle biopsies of solid pancreatic lesions.
METHODS: CT-guided percutaneous biopsies of solid pancreatic lesions performed at a cancer reference center between January 2012 and September 2013 were retrospectively analyzed. Biopsy material was collected with a 16-20 G Tru-Core needle (10-15 cm; Angiotech, Vancouver, CA) using a coaxial system and automatic biopsy gun. When direct access to the lesion was not possible, indirect (transgastric or transhepatic) access or hydrodissection and/or pneumodissection maneuvers were used. Characteristics of the patients, lesions, procedures, and histologic results were recorded using a standardized form.
RESULTS: A total of 103 procedures included in the study were performed on patients with a mean age of 64.8 year (range: 39-94 year). The mean size of the pancreatic lesions was 45.5 mm (range: 15-195 mm). Most (75/103, 72.8%) procedures were performed via direct access, though hydrodissection and/or pneumodissection were used in 22.2% (23/103) of cases and indirect transhepatic or transgastric access was used in 4.8% (5/103) of cases. Histologic analysis was performed on all biopsies, and diagnoses were conclusive in 98.1% (101/103) of cases, confirming 3.9% (4/103) of tumors were benign and 94.2% (97/103) were malignant; results were atypical in 1.9% (2/103) of cases, requiring a repeat biopsy to diagnose a neuroendocrine tumor, and surgical resection to confirm a primary adenocarcinoma. Only mild/moderate complications were observed in 9/103 patients (8.7%), and they were more commonly associated with biopsies of lesions located in the head/uncinate process (n = 8), than of those located in the body/tail (n = 1) of the pancreas, but this difference was not significant.
CONCLUSION: CT-guided biopsy of a pancreatic lesion is a safe procedure with a high success rate, and is an excellent option for minimally invasive diagnosis.
Core tip: Histopathologic analysis is often necessary to confirm the diagnosis of pancreatic tumors and aid in treatment planning. Various techniques, such as imaging-guided percutaneous, endoscopic, and surgical biopsies, can be used to obtain material for the cytologic or histologic analysis. In the present study, computed tomography-guided percutaneous core needle biopsies of pancreatic lesions were associated with few complications and 98.1% diagnostic accuracy. The safety and high diagnostic success rate renders this method an excellent minimally invasive option for diagnostic confirmation of solid pancreatic lesions.
- Citation: Tyng CJ, Almeida MFA, Barbosa PN, Bitencourt AG, Berg JAA, Maciel MS, Coimbra FJ, Schiavon LHO, Begnami MD, Guimarães MD, Zurstrassen CE, Chojniak R. Computed tomography-guided percutaneous core needle biopsy in pancreatic tumor diagnosis. World J Gastroenterol 2015; 21(12): 3579-3586
- URL: https://www.wjgnet.com/1007-9327/full/v21/i12/3579.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i12.3579
Core tip: Histopathologic analysis is often necessary to confirm the diagnosis of pancreatic tumors and aid in treatment planning. Various techniques, such as imaging-guided percutaneous, endoscopic, and surgical biopsies, can be used to obtain material for the cytologic or histologic analysis. In the present study, computed tomography-guided percutaneous core needle biopsies of pancreatic lesions were associated with few complications and 98.1% diagnostic accuracy. The safety and high diagnostic success rate renders this method an excellent minimally invasive option for diagnostic confirmation of solid pancreatic lesions.
Pancreatic cancer is the fourth leading cause of cancer deaths worldwide. The most common histologic type is adenocarcinoma, which is extremely aggressive and typically presents as unresectable disease at the time of diagnosis, with an average survival time of six months[1,2]. For this reason, solid pancreatic masses should be actively investigated to exclude or confirm a diagnosis of pancreatic cancer. The increasing availability of advanced imaging examinations facilitates the diagnosis and staging of pancreatic masses[2]. However, such methods do not always allow for a precise diagnosis, and noncharacteristic findings can make it difficult to differentiate adenocarcinoma from other causes of focal pancreatic lesions or benign inflammatory conditions. In such cases, histopathologic analysis can confirm the diagnosis and aid treatment planning. Moreover, diagnosis of adenocarcinoma based on imaging results often still requires preoperative histologic confirmation, as a definitive diagnosis for local unresectable or metastatic tumors is essential for the planning of palliative therapy. Other indications for biopsy include locally advanced pancreatic neoplasia (borderline tumor) requiring neoadjuvant therapy, areas suspected of containing lesions for which treatment is primarily nonsurgical (e.g., focal pancreatitis, tuberculosis, lymphoma, metastases), and patients with high surgical risk[3].
Various techniques, such as imaging-guided percutaneous, endoscopic, and surgical biopsies, can be used to obtain material for the cytologic or histologic analysis of these lesions[4]. Surgical biopsies conducted under direct laparotomic or laparoscopic visualization are associated with greater morbidity, mortality, cost, and inpatient time, than are minimally invasive methods. Fine-needle aspiration (FNA) and percutaneous core-needle biopsy guided by endoscopy, ultrasound, or computed tomography (CT), are useful methods for obtaining tissue samples[4,5]. Although core needle biopsies of pancreatic lesions have been performed since the 1980s, very few studies have evaluated their performance[6,7]. The objective of this study was to evaluate the techniques, results, and complications related to CT-guided percutaneous core-needle biopsies of solid pancreatic lesions.
This was a retrospective study analyzing CT-guided percutaneous biopsies of solid pancreatic lesions performed at a cancer reference center between January 2012 and September 2013. The study was approved by the Institutional Ethics Review Board. All study participants or their legal guardians provided informed written consent prior to the procedure.
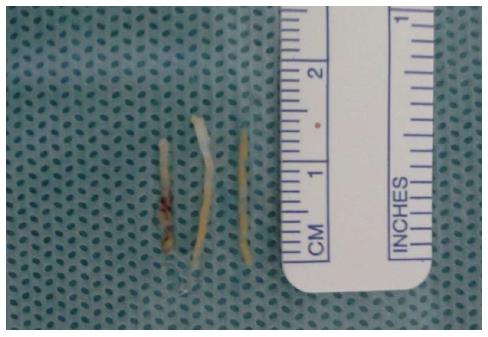
An interventional radiologist or a resident doctor under supervision performed the procedures. Routine coagulation tests were collected and evaluated before the procedures. Each case was individually assessed to determine the best access route, with direct access preferred whenever possible. All procedures were performed under local anesthesia with 2% lidocaine; conscious sedation was used in some cases to increase the patients’ comfort. Depending on the distance between the lesion and the skin, a 16-20 G Tru-Core needle (10-15 cm; Angiotech, Vancouver, CA) was used with a coaxial system and automatic biopsy gun to obtain biopsy specimens. When necessary, iodinated contrast was administered intravenously to better characterize the target lesion and adjacent vascular structures.
When abdominal structures prevented direct access to the lesion, indirect (transgastric or transhepatic) access or hydrodissection and/or pneumodissection maneuvers were used. For these procedures, a coaxial needle was used to inject 0.9% saline (hydrodissection) or 50-100 mL ambient air (pneumodissection) to displace adjacent structures from the needle’s trajectory. The choice between these techniques depended on the position in which the procedure was performed and the structures involved, as injected air tends to concentrate in the uppermost areas of the abdomen.
CT examinations were performed immediately after all procedures to identify any bleeding and/or other complications. Patients remained under observation for ≥ 1 h after the procedure; asymptomatic patients with stable vital signs were released upon receiving guidance concerning later complications. An additional CT was performed for patients exhibiting symptoms or changes in vital signs to rule out further complications.
A standardized form was used to collect patient (age, sex, presence of primary neoplasia), lesion (location in the pancreas, lesion dimensions, and resectability based on imaging), and procedure (indication, needle caliber, access, immediate and subsequent complications) characteristics, as well as histologic results from the biopsy (sufficiency of biopsy material, benign or malignant status, tumor type, and associated surgical findings).
All data were analyzed using SPSS software (version 20.0; IBM Corp., Armonk, NY, United States). Descriptive analysis was performed to calculate simple and relative frequencies of the study variables. Dichotomous scalar variables were compared using Student’s t or non-parametric Mann-Whitney U tests. Analysis of variance or the nonparametric Kruskal-Wallis test was used for comparison of variables with three or more possible values. Categorical variables were examined using 2 × 2 and 2 × 3 tables and Pearson’s χ2 test with Yates correction or Fisher’s exact test, as needed, to evaluate statistical significance. P≤ 0.05 was considered as statistically significant. The statistical methods used in this study were reviewed by a biomedical statistician from the AC Camargo Cancer Center.
A total of 103 procedures in patients with a mean age of 64.8 y (range: 39-94 year) were included. Of these, 50.5% (52/103) were women, 20.4% (21/103) had histories of previous primary tumors, most commonly lung cancer (n = 4).
The majority (65/103, 63.1%) of lesions was located in the head/uncinate process, 27.2% (28/103) were in the body, and 9.7% (10/103) were in the tail of the pancreas. The mean size of the pancreatic lesions was 45.5 mm (range: 15-195 mm). Based on imaging criteria, 77.7% (80/103) of lesions were unresectable; 22 (21.4%) were resectable and one (1.0%) was a borderline case. Highly suspected pancreatic masses were found in 6.8% (7/103) of patients (n = 6 located in the head and 1 in the body/tail), for which echoendoscopy-guided FNAs were performed before the biopsies, with negative results.
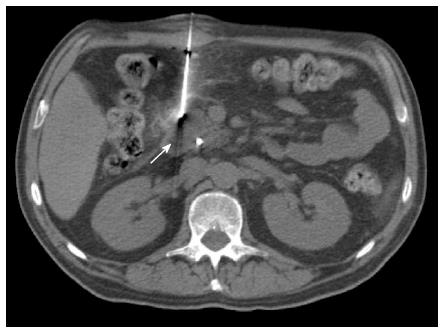
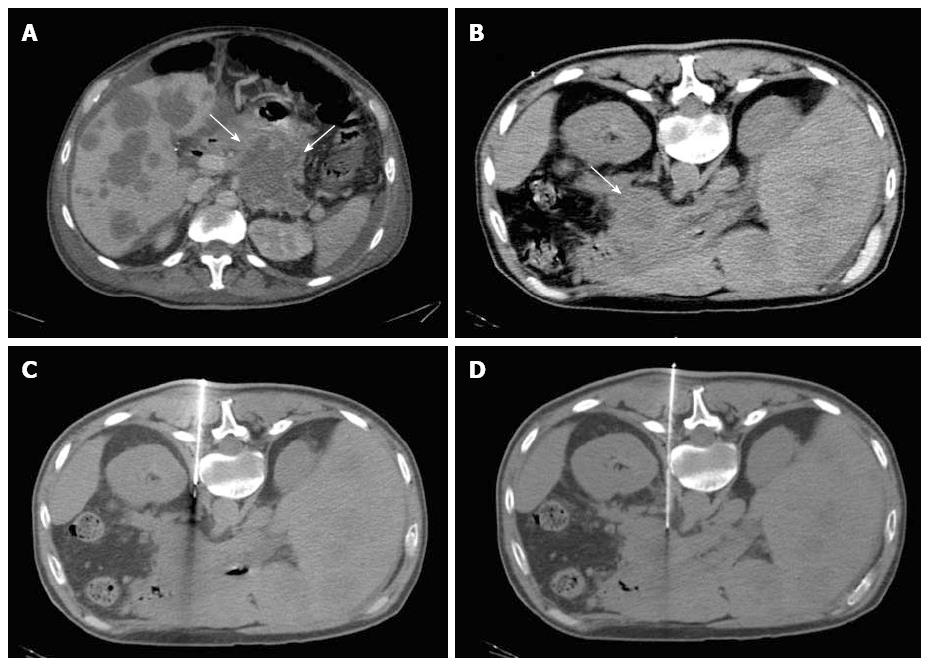
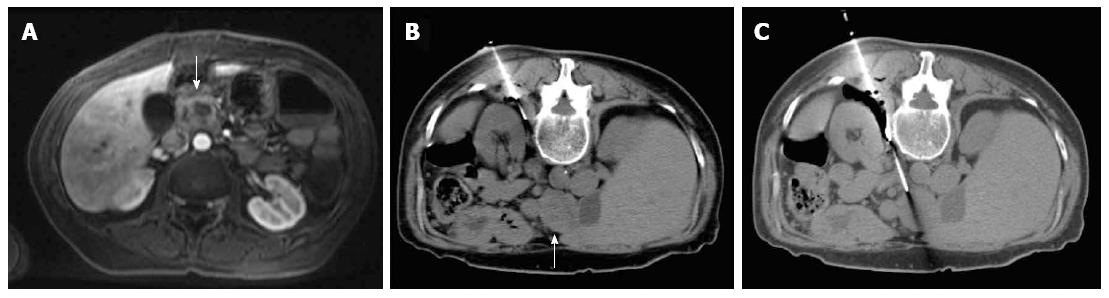
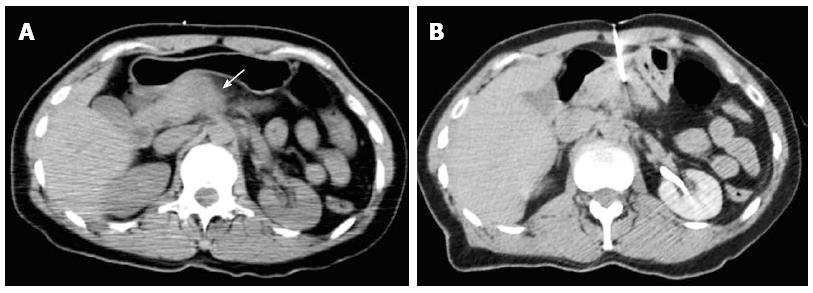
Most (75/103; 72.8%) procedures were performed via direct access (Figure 1). The remaining cases were performed with hydrodissection (n = 16), pneumodissection (n = 5), or hydro/pneumodissection (n = 2) maneuvers, or with indirect transhepatic (n = 3) or transgastric (n = 2) access (Figures 2, 3, 4 and 5). Access was anterior in 77.7% (80/103) of cases, posterior or paravertebral in 14.6% (15/103), and left lateral in 7.8% (8/103) of cases. An 18 G needle was used in most (100/103; 97.1%) cases; 20 G (n = 2) and 16 G (n = 1) needles were used in the remaining cases. The mean length of the coaxial needle’s trajectory from the skin to the lesion was 84.3 mm (range: 9-158 mm).
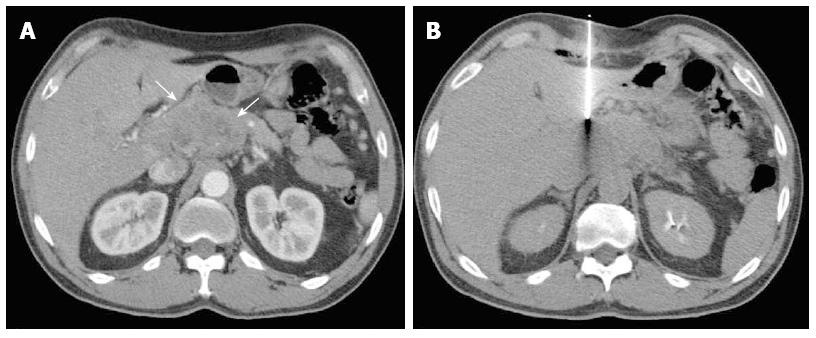
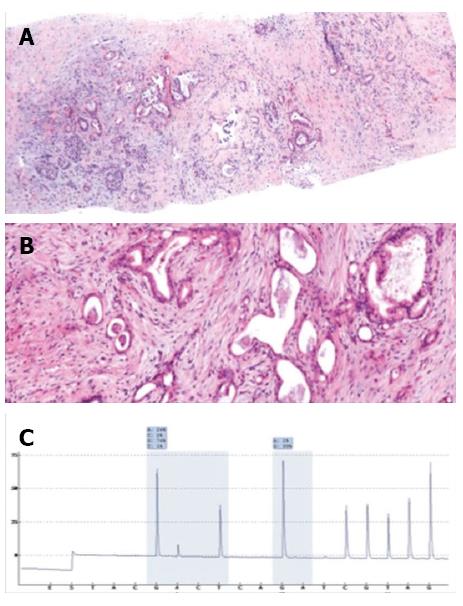
Histologic analyses were performed on all biopsies (Figures 6 and 7). Diagnoses were conclusive in 98.1% (101/103) of cases, confirming 3.9% (4/103) of tumors as benign and 94.2% (97/103) as malignant: 84.5% (87/103) were adenocarcinomas, 2.9% (3/103) were neuroendocrine tumors, 5.8% (6/103) were metastases, and 1.0% (1/103) was a primary leiomyosarcoma. Of the patients with tumors metastasizing to the pancreas, two had pulmonary adenocarcinoma and one each had esophageal cancer, choroidal melanoma, a neuroendocrine tumor, and a solitary fibrous tumor. Only two cases had inconclusive biopsy results, requiring a second biopsy to diagnose a neuroendocrine tumor, and surgical resection to confirm a primary adenocarcinoma. CT-guided biopsy confirmed adenocarcinoma in the seven patients who had negative echoendoscopy-guided FNA results.
No major procedure-related complications were observed. Mild to moderate complications occurred in 8.7% (9/103) of patients, which resolved spontaneously without treatment; retroperitoneal and subcapsular hepatic hematomas (n = 6) occurred immediately after biopsy, and bleeding, symptomatic, and asymptomatic pancreatitis (n = 3) developed later. Patients with complications did not differ from those without complications with respect to age, lesion size, or lesion-skin distance (Table 1). Complications were more commonly associated with biopsies of lesions located in the head/uncinate process, and did not differ according to biopsy technique, access type, or needle caliber (Table 2). There were no suspicious cases of tumor seeding in this case series.
| Variable | Without complications(n = 94) | With complications(n = 9) | P value |
| Patient age, yr | 64.9 ± 12.9 | 64.1 ± 12.3 | 0.86 |
| Lesion size, mm | 46.4 ± 25.9 | 35.5 ± 14.2 | 0.25 |
| Lesion-skin distance, mm | 83.1 ± 31.5 | 97.1 ± 30.2 | 0.20 |
| Variable | Complications | P value |
| Lesion location | ||
| Head/uncinate process | 8 (12.3) | 0.15 |
| Body/tail | 1 (2.6) | |
| Biopsy technique | ||
| Direct | 4 (5.4) | 0.11 |
| Indirect | 5 (17.2) | |
| Access type | ||
| Anterior | 8 (10.0) | 0.60 |
| Posterior/paravertebral | 1 (6.7) | |
| Needle caliber | ||
| 18 G | 9 (9.0) | 0.86 |
CT-guided core needle biopsy is a well-established routine procedure for obtaining samples from various organs[8-10]. Most studies, including this one, demonstrate that this procedure has > 90% diagnostic accuracy for pancreatic lesions (Table 3)[2,4,5,11-13]. In our cohort, histologic diagnosis based on percutaneous biopsy samples was considered indeterminate in only two cases, which were later confirmed to be malignant.
Core needle biopsy is considerably more sensitive than FNA for the diagnosis of pancreatic diseases, particularly when the differential diagnosis includes neuroendocrine tumors and chronic pancreatitis[14,15]. The major advantage is sample quality, as the core needle extracts sufficient material to determine cell type and origin via histologic and immunohistochemical analyses, thus allowing reliable differentiation of pancreatic tumor types[2,11]. In addition, core needle biopsy does not require that a cytopathologist be present during the procedure.
Ultrasound can be used to guide percutaneous core needle and FNA biopsies of pancreatic lesions, with the advantages of lower cost, shorter procedure time, elimination of ionizing radiation, and the ability to accompany the needle’s trajectory in real time[14,16]. However, accessing small lesions and those located in the body/tail of the pancreas is difficult with ultrasound guidance, particularly in the presence of gaseous gastrointestinal interpositioning. The choice of imaging method to guide the procedure should be individualized and depend on the interventional radiologist’s experience and the patient and lesion characteristics.
As demonstrated in the present study, percutaneous biopsy typically involves anterior access to pancreatic lesions. However, pancreatic masses in the head or uncinate process are frequently obscured by other abdominal structures, including the stomach, duodenum, transverse colon, liver, mesenteric vessels, and/or inferior vena cava. In such cases, indirect (e.g., transhepatic, transgastric, or transcaval) access is considered a safe alternative, though accompanied by risks of bleeding and peritonitis[17,18]. Another alternative includes the hydrodissection and pneumodissection maneuvers[19,20]. The location of the lesion can be confirmed with intravenous contrast administration to improve the diagnostic accuracy of the procedure[21].
Previous reports indicate that serious complications related to this procedure are rare, even when biopsies are conducted via indirect access[2,5,11-15,17,18,21]. Accordingly, no serious complications occurred in the patients in our study, with only a small percentage of patients experiencing mild adverse events that spontaneously resolved. However, Amin et al[2] reported complications in 17/372 (4.6%) percutaneous core needle biopsies, including serious complications such as abscess, duodenal perforation, and large retroperitoneal hematoma. Almost all of the complications in the present study occurred from lesions located in the head/uncinate process of the pancreas, possibly resulting from the large number of vessels and structures involving this portion of the organ. Needle size did not relate to the complication rate, though most procedures were performed with the same size needle (18 G).
Although rare, the peritoneal implantation of tumor cells during biopsy is a complication of concern. Cases of tumor dissemination along the needle trajectory after percutaneous or echoendoscopy-guided FNA have been described[22-24]. This did not occur in any of our cases. Some authors have suggested that the use of a coaxial needle, as used in this study, reduces the risk of implantation along the biopsy trajectory as the adjacent normal tissues are shielded from the needle[25].
Echoendoscopy-guided FNA has received attention because of its accuracy in obtaining cytopathologic material, with a sensitivity of 55%-97%[26-28]. This technique facilitates access to small lesions near the duodenum and stomach. However, despite the increasing availability of this technique, success rates with this method are difficult to reproduce, with high rates of false-negative findings[29]. In comparison, CT-guided biopsy has a greater availability, lower cost, higher success rate, and allows access to lesions in any part of the pancreas. Notably, pancreatic adenocarcinoma was confirmed by CT-guided biopsy in seven patients in our sample who had negative echoendoscopy-guided FNA results. However, those results were obtained from FNA procedures performed at outside centers and were not properly reviewed.
This study has several limitations. Given its retrospective nature, we could not standardize medium- and long-term follow-up evaluations. Moreover, our sample only included patients attending a single cancer reference center that were selected to undergo biopsy based on discussions among the oncologist, surgeon, and interventional radiologist. Furthermore, we were unable to directly compare CT-guided core needle biopsy with echoendoscopy-guided FNA, as this method was not available in our institution during the study period. Future studies are needed to address this issue.
In the present sample, CT-guided percutaneous core needle biopsies of pancreatic lesions were associated with few complications and a 98.1% diagnostic accuracy. The results demonstrate that this procedure is safe when performed by an experienced interventional radiologist, and represents a valuable alternative for preoperative diagnostic confirmation of solid pancreatic lesions.
Pancreatic cancer is the fourth leading cause of cancer deaths worldwide. Histopathologic analysis is often necessary for diagnosis confirmation and treatment planning, for which samples can be obtained using various techniques, such as imaging-guided percutaneous, endoscopic, and surgical biopsies. This study evaluates the techniques, results and complications related to computed tomography (CT)-guided percutaneous core needle biopsies of solid pancreatic lesions.
CT-guided core needle biopsy is a well-established procedure used routinely to obtain samples from various organs. Compared to surgical biopsies, this method is associated with lower morbidity, mortality, cost, and inpatient time.
Although core needle biopsies of pancreatic lesions have been performed since the 1980s, very few studies have evaluated their performance. This work is the first to describe different techniques for CT-guided percutaneous biopsy of pancreatic masses, demonstrating a higher diagnostic accuracy than previously reported.
Core needle biopsy is considerably more sensitive than fine-needle aspiration for the diagnosis of pancreatic diseases, particularly when the differential diagnosis includes neuroendocrine tumors and chronic pancreatitis. The major advantage of this technique is sample quality, as the core needle extracts sufficient material to determine cell type and origin via histologic and immunohistochemical analyses, thus allowing reliable differentiation of pancreatic tumor types.
Core needle biopsy involves the use of a long metal needle to collect tissue cores from a lesion. This procedure is safe when guided by imaging methods and performed by an experienced interventional radiologist.
The authors described the usefulness of CT-guided needle biopsy for diagnosing pancreatic tumors. The article is well written with sufficient sample size and acceptable procedure/results. This procedure appears safe and should be widely used for clinical diagnosis of pancreatic cancer. In general, the method is valuable for confirming pancreatic cancer along with pathologic testing.
| 1. | Rocha-Lima CM. New directions in the management of advanced pancreatic cancer: a review. Anticancer Drugs. 2008;19:435-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 2. | Amin Z, Theis B, Russell RC, House C, Novelli M, Lees WR. Diagnosing pancreatic cancer: the role of percutaneous biopsy and CT. Clin Radiol. 2006;61:996-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 33] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 3. | Clarke DL, Clarke BA, Thomson SR, Garden OJ, Lazarus NG. The role of preoperative biopsy in pancreatic cancer. HPB (Oxford). 2004;6:144-153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 4. | Goldin SB, Bradner MW, Zervos EE, Rosemurgy AS. Assessment of pancreatic neoplasms: review of biopsy techniques. J Gastrointest Surg. 2007;11:783-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Paulsen SD, Nghiem HV, Negussie E, Higgins EJ, Caoili EM, Francis IR. Evaluation of imaging-guided core biopsy of pancreatic masses. AJR Am J Roentgenol. 2006;187:769-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 48] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 6. | Dickey JE, Haaga JR, Stellato TA, Schultz CL, Hau T. Evaluation of computed tomography guided percutaneous biopsy of the pancreas. Surg Gynecol Obstet. 1986;163:497-503. [PubMed] |
| 7. | Mitchell CJ, Wai D, Jackson AM, MacFie J. Ultrasound guided percutaneous pancreatic biopsy. Br J Surg. 1989;76:706-707. [PubMed] |
| 8. | Guimaraes MD, de Andrade MQ, da Fonte AC, Chojniak R, Gross JL. CT-guided cutting needle biopsy of lung lesions--an effective procedure for adequate material and specific diagnose. Eur J Radiol. 2011;80:e488-e490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 9. | Chojniak R, Isberner RK, Viana LM, Yu LS, Aita AA, Soares FA. Computed tomography guided needle biopsy: experience from 1,300 procedures. Sao Paulo Med J. 2006;124:10-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 64] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 10. | de Bucourt M, Busse R, Zada O, Kaschke H, Weiss A, Teichgräber U, Rogalla P, Hein PA. CT-guided biopsies: quality, complications and impact on treatment: a retrospective initial quality control. Rofo. 2011;183:842-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 11. | Karlson BM, Forsman CA, Wilander E, Skogseid B, Lindgren PG, Jacobson G, Rastad J. Efficiency of percutaneous core biopsy in pancreatic tumor diagnosis. Surgery. 1996;120:75-79. [PubMed] |
| 12. | Yu YP, Jiang HT, Yao Z, Xia QR, Hong FM, Zeng H, Li S. [Feasibility and safety of CT-guided percutaneous needle biopsy and subsequent iodine-125 seed interstitial implantation for pancreatic cancer]. Zhonghua Zhong Liu Zazhi. 2013;35:608-612. [PubMed] |
| 13. | Yang RY, Ng D, Jaskolka JD, Rogalla P, Sreeharsha B. Evaluation of percutaneous ultrasound-guided biopsies of solid mass lesions of the pancreas: a center’s 10-year experience. Clin Imaging. 2014;39:62-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 14. | Spârchez Z. Ultrasound-guided percutaneous pancreatic biopsy. Indications, performance and complications. Rom J Gastroenterol. 2002;11:335-341. [PubMed] |
| 15. | Brandt KR, Charboneau JW, Stephens DH, Welch TJ, Goellner JR. CT- and US-guided biopsy of the pancreas. Radiology. 1993;187:99-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 137] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 16. | Lewitowicz P, Matykiewicz J, Heciak J, Koziel D, Gluszek S. Percutaneous fine needle biopsy in pancreatic tumors: a study of 42 cases. Gastroenterol Res Pract. 2012;2012:908963. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (2)] |
| 17. | Tseng HS, Chen CY, Chan WP, Chiang JH. Percutaneous transgastric computed tomography-guided biopsy of the pancreas using large needles. World J Gastroenterol. 2009;15:5972-5975. [PubMed] |
| 18. | Gupta S, Ahrar K, Morello FA, Wallace MJ, Hicks ME. Masses in or around the pancreatic head: CT-guided coaxial fine-needle aspiration biopsy with a posterior transcaval approach. Radiology. 2002;222:63-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 25] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 19. | Tyng CJ, Bitencourt AG, Martins EB, Pinto PN, Chojniak R. Technical note: CT-guided paravertebral adrenal biopsy using hydrodissection--a safe and technically easy approach. Br J Radiol. 2012;85:e339-e342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 20. | Tyng CJ, Bitencourt AGV, Almeida MFA, Barbosa PNV, Martins EBL, Junior JPKM, Chojniak R, Coimbra FJF. Computed tomography-guided percutaneous biopsy of pancreatic masses using pneumodissection. Radiol Bras. 2013;46:139-142. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 21. | Meier-Meitinger M, Anders K, Alibek S, Uder M, Baum U. CT-guided biopsies of pancreatic lesions: impact of contrast application prior to versus following needle placement. Acad Radiol. 2009;16:1386-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 22. | Lundstedt C, Stridbeck H, Andersson R, Tranberg KG, Andrén-Sandberg A. Tumor seeding occurring after fine-needle biopsy of abdominal malignancies. Acta Radiol. 1991;32:518-520. [PubMed] |
| 23. | Caturelli E, Rapaccini GL, Anti M, Fabiano A, Fedeli G. Malignant seeding after fine-needle aspiration biopsy of the pancreas. Diagn Imaging Clin Med. 1985;54:88-91. [PubMed] |
| 24. | Katanuma A, Maguchi H, Hashigo S, Kaneko M, Kin T, Yane K, Kato R, Kato S, Harada R, Osanai M. Tumor seeding after endoscopic ultrasound-guided fine-needle aspiration of cancer in the body of the pancreas. Endoscopy. 2012;44 Suppl 2 UCTN:E160-E161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 25. | Maturen KE, Nghiem HV, Marrero JA, Hussain HK, Higgins EG, Fox GA, Francis IR. Lack of tumor seeding of hepatocellular carcinoma after percutaneous needle biopsy using coaxial cutting needle technique. AJR Am J Roentgenol. 2006;187:1184-1187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 103] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 26. | Qian X, Hecht JL. Pancreatic fine needle aspiration. A comparison of computed tomographic and endoscopic ultrasonographic guidance. Acta Cytol. 2014;47:723-726. [PubMed] |
| 27. | Brugge WR. Fine needle aspiration of pancreatic masses: the clinical impact. Am J Gastroenterol. 2002;97:2701-2702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 28. | Fritscher-Ravens A, Izbicki JR, Sriram PV, Krause C, Knoefel WT, Topalidis T, Jaeckle S, Thonke F, Soehendra N. Endosonography-guided, fine-needle aspiration cytology extending the indication for organ-preserving pancreatic surgery. Am J Gastroenterol. 2000;95:2255-2260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 48] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 29. | Volmar KE, Vollmer RT, Jowell PS, Nelson RC, Xie HB. Pancreatic FNA in 1000 cases: a comparison of imaging modalities. Gastrointest Endosc. 2005;61:854-861. [PubMed] |
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
P- Reviewer: Bujanda L, Ren CL, Yokoyama N, Zhu H S- Editor: Qi Y L- Editor: A E- Editor: Wang CH