Published online Jul 14, 2014. doi: 10.3748/wjg.v20.i26.8535
Revised: January 28, 2014
Accepted: March 6, 2014
Published online: July 14, 2014
Processing time: 202 Days and 4.8 Hours
AIM: To identify sensitive predictors of clinically relevant postoperative pancreatic fistula (POPF) at the acute phase after pancreatectomy.
METHODS: This study included 153 patients diagnosed as having POPFs at postoperative day (POD) 3 after either open pancreatoduodenectomy or distal pancreatectomy between January 2008 and March 2013. The POPFs were categorized into three grades based on the International Study Group on Pancreatic Fistula Definition, and POPFs of grades B or C were considered to be clinically relevant. The predictive performance for the clinically relevant POPF formation of values at PODs 1, 3 and 5 as well as time-dependent changes in levels of inflammatory markers, including white blood cell count, neutrophil count, total lymphocyte count, C-reactive protein (CRP), procalcitonin level, platelet-to-lymphocyte ratio and neutrophil-to-lymphocyte ratio, and amylase content in the drain fluid were compared using the receiver operating characteristic (ROC) curve and multivariable analyses. A scoring system for the prediction of clinically relevant POPFs was created using five risk factors identified in this study, and its diagnostic performance was also evaluated.
RESULTS: Over time, 77 (50%) of 153 enrolled patients followed a protracted course and were categorized as having clinically relevant POPFs. ROC curve analyses revealed that changes in CRP levels from POD 1 to POD 3 had the greatest area under the curve value (0.767) and that an elevated CRP level of 28.4 mg/L yielded the most optimal predictive value for clinically relevant POPFs. Multivariable analyses for the risk factors of clinically relevant POPFs identified invasive carcinomas of the pancreas and elevation of the CRP level (≥ 28.4 mg/L, from POD 1 to POD 3) as independent diagnostic factors for clinically relevant POPFs (OR 2.94, 95%CI: 1.08-8.55, P = 0.035 and OR 4.82, 95%CI: 1.25-20.2, P = 0.022, respectively). A gradual increase in the prevalence of clinically relevant POPFs in proportion to the risk classification score was confirmed. A highly elevated CRP level and a risk score ≥ 8 were significantly associated with a prolonged duration of drain placement and postoperative hospitalization.
CONCLUSION: A steep rise in the serum CRP level from POD 1 to POD 3 was a highly predictive factor for subsequent clinically relevant POPFs.
Core tip: The diagnostic performance of inflammatory markers and amylase content in drain fluid were compared to identify sensitive predictors of clinically relevant postoperative pancreatic fistulas (POPFs) at the acute phase after pancreatectomy. Receiver operating characteristic curve analyses revealed that the changes in C-reactive protein (CRP) levels from postoperative day (POD) 1 to POD 3 had the greatest area under the curve value for clinically relevant POPFs. Multivariable analyses identified increased elevation of the CRP level as an independent diagnostic factor for clinically relevant POPFs. A steep rise in the CRP level was a highly diagnostic factor for clinically relevant POPFs and may be helpful in selecting the appropriate management for POPFs.
- Citation: Kanda M, Fujii T, Takami H, Suenaga M, Inokawa Y, Yamada S, Kobayashi D, Tanaka C, Sugimoto H, Koike M, Nomoto S, Fujiwara M, Kodera Y. Novel diagnostics for aggravating pancreatic fistulas at the acute phase after pancreatectomy. World J Gastroenterol 2014; 20(26): 8535-8544
- URL: https://www.wjgnet.com/1007-9327/full/v20/i26/8535.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i26.8535
Core tip: The diagnostic performance of inflammatory markers and amylase content in drain fluid were compared to identify sensitive predictors of clinically relevant postoperative pancreatic fistulas (POPFs) at the acute phase after pancreatectomy. Receiver operating characteristic curve analyses revealed that the changes in C-reactive protein (CRP) levels from postoperative day (POD) 1 to POD 3 had the greatest area under the curve value for clinically relevant POPFs. Multivariable analyses identified increased elevation of the CRP level as an independent diagnostic factor for clinically relevant POPFs. A steep rise in the CRP level was a highly diagnostic factor for clinically relevant POPFs and may be helpful in selecting the appropriate management for POPFs.
In general, pancreatectomy is recognized to be a highly invasive surgery. Despite recent advances in surgical methodologies, devices and perioperative management, pancreatectomy is associated with a mortality of approximately 5% and a morbidity of 35%-60%[1,2]. The incidence of postoperative pancreatic fistulas (POPFs), a surgical complication of particular concern, has been reported to be 20%-40% after pancreatoduodenectomy and 30%-55% after distal pancreatectomy, thereby having a decisive influence on the clinical course after pancreatectomy[3-6]. Diverse anastomotic procedures, treatment of the pancreatic stump, and the administration of somatostatin analogs have been evaluated by randomized clinical trials with the aim of reducing the prevalence of POPFs[7-9], but they have not been associated with appreciable improvement.
The aggravation of POPFs can lead to prolonged hospitalization, burgeoning costs of medical treatment, and development into fatal conditions, such as sepsis through abdominal abscesses and intra-abdominal hemorrhages from ruptured aneurysms[4,5]. In addition, clinically relevant POPFs have been reported to be risk factors for peritoneal recurrences[10]. Thus, management of POPFs is a high-priority issue in pancreatic surgery. Sensitive diagnostic factors of clinically relevant POPF are needed to identify high-risk patients and strengthen measures aimed at preventing the aggravation of POPFs. The clinical stratification of POPFs was established by the International Study Group on Pancreatic Fistula Definition (ISGPF) in 2005[11]. The presence or absence of POPFs can be determined at postoperative day (POD) 3 by the amylase content in the drained fluid. The most important issue is whether the POPF subsides promptly or deteriorates into severe leakage, thereby influencing the clinical course.
Predicting clinically relevant POPFs is often difficult because the amylase content in the drained fluid does not always represent the actual conditions of leakage[12,13]. Numerous pre- or intraoperative risk factors for postoperative POPFs have been reported: a soft pancreas, a small main pancreatic duct, a fatty pancreas, malnutrition, and obesity[14-18]. Nevertheless, physicians frequently vacillate on whether or not a POPF becomes prolonged after pancreatectomy. The prediction of the course of POPFs during the early postoperative phase (i.e., POD 3) would enable the appropriate selection from two contrary treatment strategies: the early removal of drainage tubes or retention of the tubes accompanied by scrutiny of the abdomen by imaging studies or the administration of broad-spectrum antibiotics and the use of somatostatin analogs. The early removal of drainage tubes in patients with clinically irrelevant POPFs and the early recovery of patients from clinically relevant POPFs through optimal management would both lead to shortened hospitalizations.
In terms of monitoring inflammation, measurement of the white blood cell (WBC) count, neutrophil count, total lymphocyte count (TLC), C-reactive protein (CRP) levels, and procalcitonin levels, as well as being alert to the presence of the systemic inflammatory response syndrome (SIRS), have been in widespread use[19-21]. Once a POPF is aggravated, pancreatic fluid leaks into the abdominal cavity and causes local inflammation, secondary infection and, eventually, expansion to a systemic inflammatory response. Thus, the hypothesis that inflammatory markers represent the “momentum” of subsequent POPFs was raised because POPFs induce local and systemic inflammatory responses. No reports have evaluated the associations between the time-dependent changes of postoperative inflammatory markers and the subsequent course of POPF focusing on patients diagnosed as having POPFs at POD 3. To test the hypothesis and to identify sensitive diagnostic factors of clinically relevant POPFs, the inflammatory markers in patients who underwent pancreatectomy were investigated at the acute phase of the postoperative course over time, and their diagnostic values for POPF aggravation were evaluated.
This study conforms to the ethical guidelines of the World Medical Association Declaration of Helsinki-Ethical Principles for Medical Research Involving Human Subjects. Written informed consent for the surgery and the usage of clinical data, as required by the Review Board of Nagoya University (Nagoya, Japan), was obtained from all patients.
Between January 2008 and March 2013, 337 major pancreatectomies (i.e., pancreatoduodenectomy, distal pancreatectomy and total pancreatectomy) were performed at the Department of Gastroenterological Surgery of Nagoya University. Of these, 23 patients who underwent total pancreatectomy were excluded. Patients without POPFs were excluded because the principal purpose of the current study was to identify the diagnostic factors of aggravation among patients with POPFs. Additionally, patients with postoperative infectious complications, such as wound infection and pneumonia, which might influence the inflammatory status, were excluded. Ultimately, 153 patients who were diagnosed as having POPFs at 3 d after pancreatoduodenectomy or distal pancreatectomy were enrolled. The preoperative backgrounds of the patients, intraoperative conditions, and postoperative course (POPF severity, duration of placement of drainage tube, and postoperative inflammatory markers) were investigated to identify sensitive diagnostic factors of clinically relevant POPFs.
For pancreatoduodenectomies, pancreatic reconstruction was performed by a direct anastomosis between the pancreatic duct and mucosal layer of the jejunal loop. Except for patients with a remarkably dilated main pancreatic duct, a stent tube was inserted into the pancreatic duct. Adhesion of the jejunal wall and the pancreatic stump by one-layer sutures was added[22,23]. Silastic flexible drains (Blake® drain, Ethicon, Somerville, NJ, United States) were placed at the anterior and posterior surfaces of the pancreatojejunostomy and choledochojejunostomy. For distal pancreatectomies, closure of the main pancreatic duct was performed using a continuous suture. Silastic flexible drains were placed at the anterior and posterior surface of the pancreatic stump.
Patients were categorized as having developed a grade A, B or C fistula based on the definitions of the ISGPF[11]. Briefly, POPFs were categorized into three grades: biochemical leak without peripancreatic fluid collection, defined as output containing pancreatic amylase on or after the third postoperative day from an operatively positioned drain with a pancreatic amylase level more than three times the upper serum reference value (grade A); leaks with changes in management and a delay of discharge from the hospital (grade B); and leaks that required percutaneous drainage of a fluid collection or re-exploration for critically ill patients and were associated with an extended stay in the hospital (grade C). POPFs of grades B or C were considered to be clinically relevant POPFs in this study.
Cephalosporin was administered immediately before surgery and every 3 h during surgery. In all patients, the administration of antibiotics and H2 blockers was continued routinely thorough POD 3. Oral intake was started routinely 5 d after surgery unless postoperative complications, such as delayed gastric emptying, occurred. Blood-sugar level was routinely monitored, and appropriate levels were maintained (70 mg/dL ≤ blood-sugar level < 200 mg/dL). The amylase content in the discharge from the closed-suction drains was examined at PODs 1, 3 and 5. Continuous low pressure suction was applied to the abdominal drains to minimalize the fluid cavity through POD 4. Abdominal drains were removed at POD 4 in patients without POPFs or replaced as needed until a cure of the POPFs was confirmed in patients with POPFs. Abdominal ultrasonography was performed as needed postoperatively. Percutaneous drainage or the replacement of tubes was employed when signs of inadequate drainage were found.
Blood examinations were conducted routinely before surgery and at PODs 1, 3 and 5. The inflammatory markers included the WBC, neutrophil count, TLC, platelet count, and CRP, procalcitonin and serum amylase levels. The procalcitonin level was tested routinely only at POD 3. In addition to the markers mentioned above, the platelet-to-lymphocyte ratio and neutrophil-to-lymphocyte ratio were calculated. Time-dependent changes of these factors were also investigated. As indications of systemic inflammatory status, the duration of a high fever (≥ 38 °C), SIRS, and the use of catecholamines were evaluated. The amylase content and bacterial culture in the drain fluids were also examined at PODs 1, 3 and 5 or until drain removal.
To identify the appropriate cutoff value of each marker and time-dependent changes, analyses of receiver operating characteristic (ROC) curves were conducted. The diagnostic performance of inflammatory markers for clinically relevant POPFs was compared using optimal cutoff values determined by analyses of the ROC curves. Selected markers with known risk factors for POPFs were included in the multivariable analyses. Moreover, the influence of the selected diagnostic factors on the duration of drain placement and hospitalization was evaluated.
A scoring scale for prediction of clinically relevant POPFs was introduced using the risk factors identified in the present study, and the diagnostic performance was evaluated. Parameters that demonstrated significant associations with clinically relevant POPFs in univariate analyses were included in this scoring scale. Then, individual scores were allocated to each parameter in accordance with their odds ratios.
The sensitivity, specificity, positive predictive value, negative predictive value, likelihood ratio of a positive result and likelihood ratio of a negative result for clinically relevant POPFs were further evaluated among the potent factors identified in this study to compare diagnostic performance. The CRP level on POD 3, increased elevation of the CRP level from PODs 1 to 3, drain amylase content on POD 3 and the risk score were included in this analysis.
The qualitative χ2 test and quantitative Mann-Whitney’s U test were used to compare the two groups. ROC curve analyses were conducted to evaluate the discrimination of clinically relevant POPFs by the markers of interest. The goodness of fit for clinically relevant POPFs was assessed by comparing the area under the curve (AUC) value, and optimal cutoff values were also determined using the Youden Index. The risk factors for clinically relevant POPFs were evaluated using binomial logistic analyses, and the variables with P < 0.1 were entered into the final model. The statistical analyses were performed using JMP® ver10 (SAS Institute, NC, United States). A P < 0.05 was considered to be significant.
The patients’ characteristics are summarized in Table 1. The ages of the 153 subjects ranged from 28 years to 89 years (65.2 ± 10.3 years; mean ± SD), and the male-to-female ratio was 101:52. Surgery consisted of pancreatoduodenectomy in 103 patients and distal pancreatectomy in 50 patients. In terms of the pathological diagnoses, 68 (44%) subjects had invasive carcinoma of the pancreas. Thirty-nine (25%) patients had diabetes mellitus preoperatively, and 20 (13%) patients had a history of pancreatitis. None of the enrolled participants had active inflammatory disease (e.g., cholangitis or pancreatitis) before surgery, suggesting that the postoperative inflammatory status was assessed accurately. All patients were afebrile, and the CRP levels were < 20 mg/L preoperatively. The postoperative mortality rate was 0%. Eventually, 77 (50%) of the 153 enrolled patients followed a protracted course and were categorized as having clinically relevant POPFs. There was no significant difference in the incidence rate of clinically relevant POPFs between the patients who underwent pancreatoduodenectomy and the patients who underwent distal pancreatectomy (54% vs 42%, respectively, P = 0.151).
| Characteristic | Value |
| Age (yr), mean (range) | 65.2 (28-89) |
| Male/female | 101/52 |
| Preoperative symptom(s) | 37 (24) |
| Diabetes mellitus (preoperative ) | 39 (25) |
| History of pancreatitis | 20 (13) |
| Body mass index, mean ± SD | 22.5 ± 3.5 |
| Procedure | |
| Pancreatoduodenectomy | 103 |
| Distal pancreatectomy | 50 |
| Resection of the portal vein | 21 (14) |
| Pathological diagnosis | |
| Invasive carcinoma of the pancreas | 68 |
| Intraductal papillary mucinous neoplasm | 35 |
| Carcinoma of the ampulla | 13 |
| Cholangiocarcinoma | 10 |
| Pancreatic neuroendocrine tumor | 8 |
| Other | 19 |
| Grade of postoperative pancreatic fistula1 | |
| A | 76 |
| B | 70 |
| C | 7 |
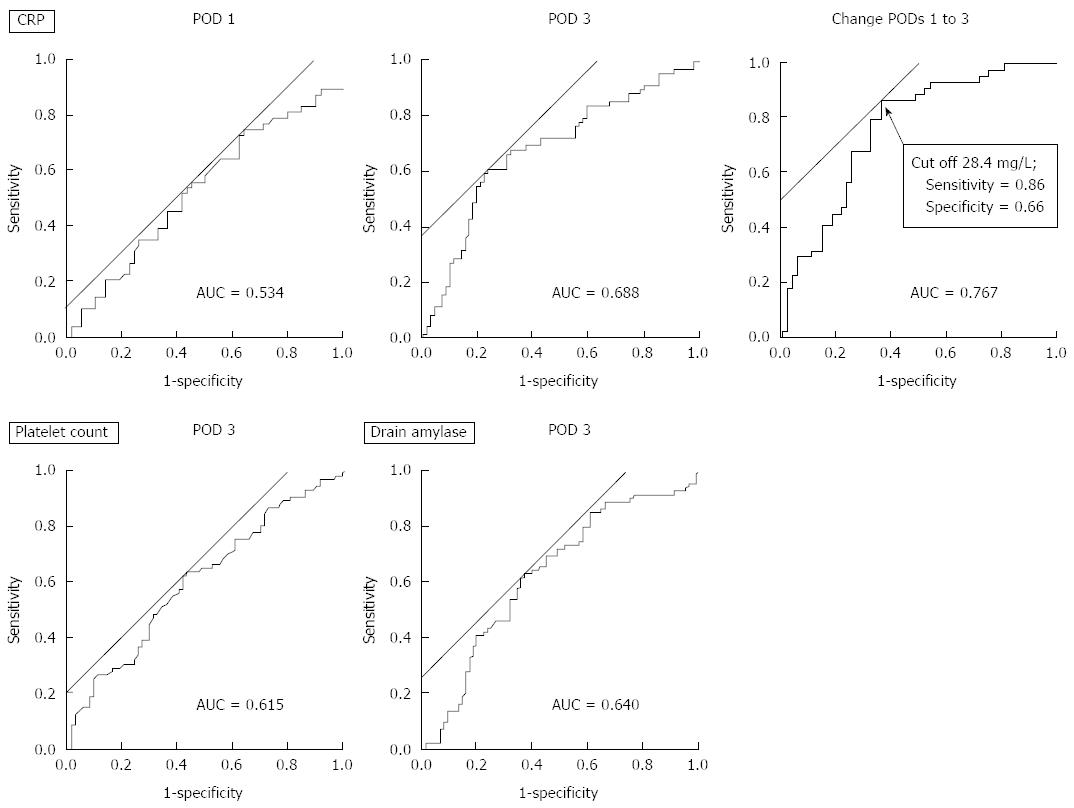
The predictive values of postoperative inflammatory markers at each time point and time-dependent changes were evaluated by ROC curve analyses. The AUC values, P values for differences between grade-A and grade-B/C POPFs, and the most optimal cutoff point to predict clinically relevant POPFs are listed in Table 2. The change in the CRP levels from POD 1 to POD 3 had the highest AUC value (0.767), and a further elevation of the CRP level of 28.4 mg/L yielded the most optimal diagnostic value for clinically relevant POPFs. In general, changes from POD 3 to POD 5 were less diagnostic than those from POD 1 to POD 3. Among the markers determined at POD 3, the platelet count and CRP level showed relatively high AUC values (> 0.6). The amylase content in the drain fluid at POD 3 was significantly associated with clinically relevant POPFs (P = 0.003; AUC = 0.640; optimal cutoff value 1918 IU/L). Representative ROC curves are shown in Figure 1.
| Marker | POD 1 | POD 3 | POD 5 | Change from POD 1 to 3 | Change from POD 3 to 5 | |||||||
| AUC | P value | AUC | P value | Cutoff | AUC | P value | AUC | P value | Cutoff | AUC | P value | |
| White blood cell count (/mm3) | 0.545 | 0.343 | 0.524 | 0.615 | 11400 | 0.588 | 0.067 | 0.504 | 0.939 | 600 | 0.557 | 0.242 |
| Neutrophil count (/mm3) | 0.560 | 0.198 | 0.532 | 0.495 | 8200 | 0.591 | 0.060 | 0.506 | 0.898 | -3300 | 0.560 | 0.215 |
| Total lymphocyte count (/mm3) | 0.524 | 0.604 | 0.577 | 0.097 | 600 | 0.541 | 0.397 | 0.550 | 0.285 | 0 | 0.518 | 0.708 |
| Platelet count (/mm3) | 0.540 | 0.399 | 0.615 | 0.014 | 164000 | 0.580 | 0.098 | 0.603 | 0.028 | -24000 | 0.549 | 0.310 |
| Serum amylase (IU/mL) | 0.504 | 0.935 | 0.580 | 0.087 | 136 | 0.600 | 0.039 | 0.502 | 0.966 | -69 | 0.522 | 0.643 |
| C-reactive protein (mg/L) | 0.534 | 0.532 | 0.688 | < 0.001 | 158 | 0.684 | < 0.001 | 0.767 | < 0.001 | 28.4 | 0.565 | 0.200 |
| Procalcitonin (ng/mL) | - | - | 0.584 | 0.451 | 0.3 | - | - | - | - | - | - | - |
| Neutrophil-to-lymphocyte ratio | 0.532 | 0.497 | 0.579 | 0.092 | 9.6 | 0.590 | 0.061 | 0.537 | 0.433 | -0.3 | 0.558 | 0.420 |
| Platelet to lymphocyte ratio | 0.517 | 0.718 | 0.529 | 0.557 | 231 | 0.557 | 0.243 | 0.508 | 0.858 | 67 | 0.539 | 0.231 |
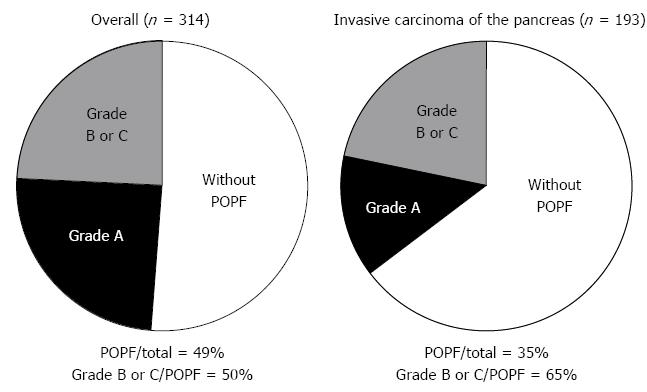
Next, the diagnostic values for clinically relevant POPFs were compared among the well-known risk factors (e.g., the size and texture of the pancreatic duct), postoperative high fever, SIRS, the use of catecholamines, and the inflammatory markers selected by ROC curve analyses and amylase content in the drain fluid at POD 3. Univariate analyses showed that invasive carcinoma of the pancreas, platelet count (< 164000/mm3, POD 3), CRP level (≥ 158 mg/L, POD 3), CRP elevation (≥ 28.4 mg/L, from POD 1 to POD 3) and amylase content in the drain fluid (≥ 1918 IU/L, POD 3) were significant adverse factors for POPFs (Table 3). Multivariable analyses identified invasive carcinoma of the pancreas and elevation of the CRP level (≥ 28.4 mg/L, from POD 1 to POD 3) as independent diagnostic factors for clinically relevant POPFs (OR 2.94, 95%CI: 1.08-8.55, P = 0.035 and OR 4.82, 95% CI: 1.25-20.2, P = 0.022, respectively; Table 3). In patients with invasive carcinoma of the pancreas, the ratio of clinically relevant POPF to mild POPF was higher but the total prevalence of POPFs was lower compared with the whole population (Figure 2).
| Variable | Univariate | Multivariable | ||||
| Odds ratio | 95%CI | P value | Odds ratio | 95%CI | P value | |
| Age (≥ 65 yr) | 1.16 | 0.61-2.23 | 0.654 | |||
| Gender (male) | 1.29 | 0.66-2.53 | 0.459 | |||
| Diabetes mellitus | 1.39 | 0.67-2.92 | 0.378 | |||
| History of pancreatitis | 0.62 | 0.23-1.59 | 0.321 | |||
| Body mass index (≥ 22) | 1.48 | 0.79-2.82 | 0.226 | |||
| Operative procedure (DP) | 1.65 | 0.83-3.28 | 0.151 | |||
| Invasive carcinoma of the pancreas | 2.31 | 1.21-4.47 | 0.011 | 2.94 | 1.08-8.55 | 0.035 |
| Operative time (≥ 360 min) | 1.33 | 0.71-2.53 | 0.376 | |||
| Blood loss (≥ 600 mL) | 1.83 | 0.97-3.51 | 0.063 | 1.73 | 0.62-4.86 | 0.297 |
| Intraoperative transfusion | 1.45 | 0.48-4.59 | 0.511 | |||
| Pancreas texture (soft) | 1.46 | 0.69-3.10 | 0.322 | |||
| Main pancreatic duct (< 4 mm) | 1.36 | 0.70-2.65 | 0.366 | |||
| Persistence of high fever (≥ 38 °C, ≥ 3 d) | 1.10 | 0.57-2.14 | 0.781 | |||
| Persistence of SIRS (≥ 3 d) | 1.68 | 0.88-3.23 | 0.116 | |||
| Usage of catecholamines | 1.73 | 0.68-4.62 | 0.251 | |||
| Total lymphocyte count (< 600/mm3, POD 3) | 1.73 | 0.74-4.23 | 0.201 | |||
| Platelet count (< 164000/mm3, POD 3) | 2.28 | 1.20-4.39 | 0.012 | 1.28 | 0.44-3.64 | 0.646 |
| CRP (≥ 158 mg/L, POD 3) | 4.82 | 2.37-10.2 | < 0.001 | 2.39 | 0.73-8.03 | 0.150 |
| Neutrophil-to-lymphocyte ratio (≥ 9.6, POD 3) | 1.84 | 0.97-3.52 | 0.061 | 1.56 | 0.55-4.40 | 0.401 |
| CRP elevation (≥ 28.4 mg/L, POD 1 to 3) | 11.4 | 4.35-34.3 | < 0.001 | 4.82 | 1.25-20.2 | 0.022 |
| Drain amylase (≥ 1918 IU/mL, POD 3) | 3.00 | 1.57-5.86 | 0.001 | 1.50 | 0.55-4.08 | 0.423 |
| Drain fluid bacterial culture (positive, POD 3) | 1.62 | 0.68-3.87 | 0.278 | |||
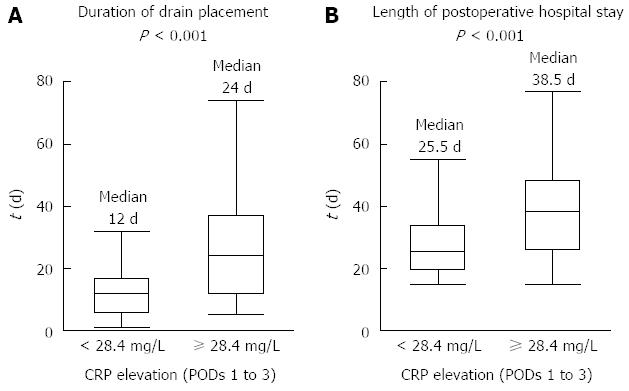
Patients with a further elevation in the CRP levels ≥ 28.4 mg/L from POD 1 to POD 3 (high elevation of the CRP level) had a significantly longer duration of drain placement than patients without elevation in the CRP levels (24 and 12 d, respectively; median, P < 0.001; Figure 3A). Consequently, the duration of postoperative hospitalization was significantly greater in patients with high CRP elevations than in patients without high CRP elevations (38.5 and 25.5 d, respectively; median, P < 0.001; Figure 3B). High CRP elevation was not significantly associated with the WBC count, neutrophil count, procalcitonin level or bacterial culture in the drain fluid at POD 3.
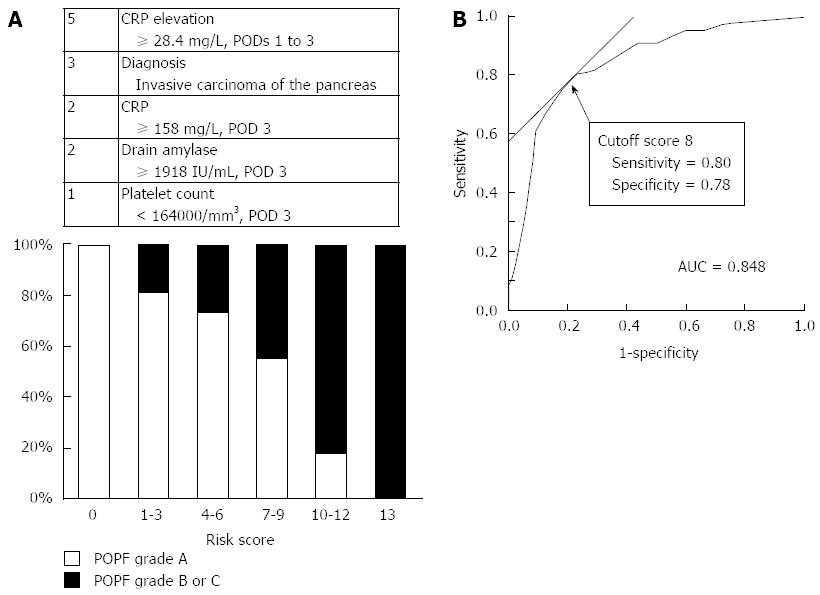
A scoring scale for the prediction of clinically relevant POPFs was developed to enhance the clinical utility of the diagnostic factors detected in this study. Five diagnostic factors (invasive carcinoma of the pancreas, platelet count < 164000/mm3 at POD 3, CRP ≥ 158 mg/L at POD 3, CRP elevation ≥ 28.4 mg/L from POD 1 to POD 3, and amylase content in the drain fluid ≥ 1918 IU/L at POD 3) were included in the scoring scale because these factors demonstrated a significant association with clinically relevant POPFs. The prevalence of clinically relevant POPFs increased gradually in proportion with the score, from 0% to 100%. The score had a remarkably high goodness of fit for clinically relevant POPFs (AUC = 0.848). A score of 8 yielded the most optimal diagnostic value for clinically relevant POPFs, thereby supporting the clinical utility of the scoring scale (Figure 4). In addition, patients with a risk score ≥ 8 had a significantly longer duration of drain placement and postoperative hospitalization than patients with a score < 8 (25.5 ± 18.2 and 12 ± 10.6, 39.5 ± 29.0 and 26.0 ± 10.1 d, respectively; mean ± SD, P < 0.001 for both).
Table 4 lists the sensitivity, specificity, positive predictive value, negative predictive value, likelihood ratio of a positive result and likelihood ratio of a negative result of four important factors: the CRP level on POD 3, the increased elevation of the CRP level from POD 1 to 3, the drain amylase content on POD 3 and the risk score. It is worth noting that an increased elevation of the CRP level demonstrated a high negative predictive value (0.857) and a low likelihood ratio of a negative result (0.212), indicating that it may be useful especially for the prediction of the absence of a clinically relevant POPF. The risk score scale was found to have an additional impact on detecting the presence of clinically relevant POPF.
| Odds ratio | 95%CI | P value | Sensitivity | Specificity | Positive predictive value | Negative predictive value | Likelihood ratio of positive result | Likelihood ratio of negative result | |
| CRP ≥ 158 mg/L1 | 4.82 | 2.37-10.2 | < 0.001 | 0.594 | 0.767 | 0.707 | 0.667 | 2.552 | 0.529 |
| CRP elevation ≥ 28.4 mg/L2 | 11.4 | 4.35-34.3 | < 0.001 | 0.864 | 0.643 | 0.655 | 0.857 | 2.418 | 0.212 |
| Drain amylase ≥ 1918 IU/mL1 | 3.00 | 1.57-5.86 | 0.001 | 0.636 | 0.632 | 0.636 | 0.632 | 1.727 | 0.576 |
| Risk score ≥ 8 | 12.9 | 5.13-35.4 | < 0.001 | 0.795 | 0.768 | 0.729 | 0.827 | 3.427 | 0.266 |
Aggravated POPFs can involve critical complications, such as sepsis and intra-abdominal hemorrhage from ruptured aneurysms, resulting in prolonged hospitalization, increased medical expense, and in the most extreme cases, mortality[4,5]. In addition, clinically relevant POPFs can lead to an increased risk of recurrences through peritoneal dissemination[10]. Clinically relevant POPFs are the most serious problem in the field of pancreatic surgery. The realistic goal is to minimize the extent of POPFs and to limit it to grade A, which has less impact on the postoperative clinical course. To achieve this goal, the early identification of high-risk patients with clinically relevant POPFs is necessary for their subsequent management, including the thorough control of infection, effective drainage, and nutritional care with the aim to localize inflammation and achieve early recovery.
When should surgeons manage POPFs, and what are the definitive criteria in clinical practice? An important “crossroads” may occur at POD 3, when POPFs are usually diagnosed. Management (including drainage and antibiotic administration) after the diagnosis of a POPF at POD 3 depends on whether the POPF is likely to be aggravated. Drainage tubes are removed at the surgeon’s request. However, it would be too late to prevent the expansion of a POPF if a course of treatment is determined at POD 5 or later. In the present study, patient inclusion was limited to those diagnosed as having a POPF, and all of the evaluated factors should have been available at POD 3. POPFs are associated with inflammation that is a combination of pancreatic fluid with secondary inflammation by bacterial infection, so inflammatory markers are expected to reflect the momentum of POPFs[6]. Thus, POD 3 was the ideal time point for the early prediction of clinically relevant POPFs, and the inflammatory markers at POD 3 were evaluated by focusing on universally prevalent markers.
Elevation of the CRP level from POD 1 to POD 3 was found to be an independent risk factor for clinically relevant POPFs with the largest odds ratio, suggesting that it was the most sensitive diagnostic factor of a subsequent clinically relevant POPF. Surprisingly, elevation of the CRP level from POD 1 to POD 3 demonstrated a greater diagnostic performance than well-known major risk factors, such as the size and texture of the pancreatic duct and the amylase content of drain fluid. Within 6 h after stimulation, the CRP level exceeded normal values, and it peaked after 48 h[24]. CRP has a nearly constant half-life in serum of 19 h[25]. Therefore, the CRP concentration in serum is determined by its rate of synthesis and reflects the intensity of the stimulus for acute inflammatory responses. The high diagnostic value of elevation of the CRP level from POD 1 to POD 3 could also be explained. First, an inevitable elevation of the CRP level due to surgical stress and local inflammation in resected tissues was confirmed at POD 1 in all patients. The half-life of CRP is 19 h, so the CRP level starts to show a gradual decline at PODs 2-3 in patients without additional inflammatory events, whereas it continues to increase in patients with expanding POPFs. Second, the CRP level can comprehensively reflect the chemical inflammation of the peritoneum due to leakage of the pancreatic fluid as well as coexisting inflammation due to bacterial infection. The well-known inflammatory markers of WBC and neutrophil counts showed less diagnostic value compared with the CRP level, most likely because of their wide normal range, thereby debilitating the decrease by severe infection and influences by factors other than inflammation (e.g., medications)[26-28]. Procalcitonin is specific only to bacterial infection and does not reflect a comprehensive inflammatory status (including chemical inflammation caused by pancreatic fluids)[29]. In addition, elevation in the CRP level from POD 1 to POD 3 is an ideal marker because it is readily measurable in hospitals at low cost and has high diagnostic value for POPFs.
Drain amylase content is superior for determining the presence of POPFs because it directly reflects the leakage of pancreatic fluids. However, the concentration can be strongly influenced by the amount of exudative ascites fluid and the efficacy of drainage, indicating that it does not always increase parallel to the exacerbation of POPFs. Pancreatic texture, which is a well-known risk factor of POPF, is problematic in that it is determined by surgeons subjectively. On the other hand, the CRP level has the advantages that it can be measured objectively and that it enables the evaluation of the degree of comprehensive inflammation easily.
As shown in Figure 2, subjects with invasive carcinoma of the pancreas were less likely to have POPFs, but the POPFs tended to be protracted when they did occur. It has been postulated that the surrounding tissues and the bloodstream around the anastomotic site are diminished by the dissection of lymph nodes, leading to poorer wound healing[30]. Additionally, the size and texture of the pancreatic duct (which have been recognized as major risk factors for POPFs) were not significantly associated with aggravated POPFs in the present study. Patient assessment revealed that the size and texture of the pancreatic duct increased the formation of POPFs but did not always lead directly to persistent POPFs.
We proposed a novel risk scoring scale for clinically relevant POPFs. The score demonstrated good compatibility with the prevalence of clinically relevant POPFs. The score included five parameters that are available at POD 3 (including changes in the CRP level) that achieved a better goodness of fit for the aggravation of POPFs than changes in the CRP level. The scoring scale can be easily calculated at POD 3, indicating that it would be a convenient and useful method for patient selection. After the diagnosis of POPFs at POD 3, changes in the CRP level and the risk score scale were helpful in determining the subsequent management. In patients with a continuous elevation of the CRP level or a high risk score, frequent physical assessment, a computed tomography, and the culture of drainage fluids may be considered to allow early management of aggravated POPFs. Additionally, the strict control of infection, thorough management of drains, and administration of somatostatin analogs might be recommended, depending on a patient’s condition. Conversely, patients without elevations of the CRP level or a high risk score are not at risk of clinically relevant POPFs, so an early removal of the drains and early discharge from the hospital should be considered. The benefits of early drain removal have been reported by Bassi et al[31] and Kawai et al[32], but subsequent abdominal abscesses and in-hospital death have also been observed. Changes in the CRP levels and the risk score scale could help in the selection of patients eligible for the early removal of drains. Welsch et al[33] reported that a high CRP level (140 mg/L) at POD 4 is a diagnostic factor for overall infectious complications after pancreatectomy. Our results would apply better to clinical practice due to their focus on clinically relevant POPFs at an earlier time point.
The present study had limitations, including a low number of enrolled patients and the retrospective determination of cut-off values in an explorative setting. The inflammatory markers and the amylase content in drain fluid were not examined every day. The measurement of the levels of proinflammatory cytokines such as interleukin-6 would have made this study more complete. Because the results of the current study were exploratory, this analysis may lead to prospective studies evaluating the hypothesis that an elevation of the CRP level from PODs 1 to 3 might have a diagnostic value for the identification of patients with clinically relevant POPFs. Well-designed prospective clinical studies based on changes in the CRP level or the use of a high-performance risk scoring scale at the acute phase after pancreatectomy could help to establish the therapeutic strategies for POPFs.
In conclusion, a steep rise in the serum CRP level from 1 POD to 3 PODs was shown to be a highly diagnostic factor for clinically relevant POPFs and could be helpful in selecting the most appropriate management for POPFs. The scoring scale consisting of five diagnostic factors identified in the current study may provide additional assistance for patient classification.
Aggravation of postoperative pancreatic fistulas (POPFs) leads to prolonged hospitalization and burgeoning medical costs and can develop into fatal conditions.
The identification of sensitive diagnostic factors of clinically relevant POPFs are needed to identify high-risk patients and strengthen measures aimed at preventing the aggravation of POPFs.
To date, many pre- and intraoperative risk factors of POPF formation have been reported. However, the selection methods for high-risk patients with aggravated POPFs at the acute phase after pancreatectomy have not been fully discussed. In this study, the elevation of the C-reactive protein (CRP) level was found to be a strong diagnostic factor of a clinically relevant POPF.
The changes in CRP levels and the risk score can provide a good indication for the management of a POPF, including the early removal of the drainage tubes. The inclusion of proinflammatory cytokines (e.g., interleukin-6) may achieve a further refinement of the risk score.
POPF is the most common complication in pancreatic surgeries and can lead to fatal condition. How to control POPFs is one of the most important issues in the field of pancreatic surgery.
This retrospective study identifies an increase in CRP as a good marker for predicting aggravation of a POPF. The conclusion of this study can assist surgeons, especially in the appropriate management of POPFs.
| 1. | Hill JS, Zhou Z, Simons JP, Ng SC, McDade TP, Whalen GF, Tseng JF. A simple risk score to predict in-hospital mortality after pancreatic resection for cancer. Ann Surg Oncol. 2010;17:1802-1807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 82] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 2. | Ouaïssi M, Giger U, Louis G, Sielezneff I, Farges O, Sastre B. Ductal adenocarcinoma of the pancreatic head: a focus on current diagnostic and surgical concepts. World J Gastroenterol. 2012;18:3058-3069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 16] [Cited by in RCA: 15] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 3. | de Castro SM, Busch OR, van Gulik TM, Obertop H, Gouma DJ. Incidence and management of pancreatic leakage after pancreatoduodenectomy. Br J Surg. 2005;92:1117-1123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 154] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 4. | Lee HG, Heo JS, Choi SH, Choi DW. Management of bleeding from pseudoaneurysms following pancreaticoduodenectomy. World J Gastroenterol. 2010;16:1239-1244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 65] [Cited by in RCA: 70] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 5. | Büchler MW, Wagner M, Schmied BM, Uhl W, Friess H, Z’graggen K. Changes in morbidity after pancreatic resection: toward the end of completion pancreatectomy. Arch Surg. 2003;138:1310-134; discussion 1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 496] [Cited by in RCA: 438] [Article Influence: 19.0] [Reference Citation Analysis (3)] |
| 6. | Simons JP, Shah SA, Ng SC, Whalen GF, Tseng JF. National complication rates after pancreatectomy: beyond mere mortality. J Gastrointest Surg. 2009;13:1798-1805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 111] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 7. | McKay A, Mackenzie S, Sutherland FR, Bathe OF, Doig C, Dort J, Vollmer CM, Dixon E. Meta-analysis of pancreaticojejunostomy versus pancreaticogastrostomy reconstruction after pancreaticoduodenectomy. Br J Surg. 2006;93:929-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 141] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 8. | Oláh A, Issekutz A, Belágyi T, Hajdú N, Romics L. Randomized clinical trial of techniques for closure of the pancreatic remnant following distal pancreatectomy. Br J Surg. 2009;96:602-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 79] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 9. | Yeo CJ, Cameron JL, Lillemoe KD, Sauter PK, Coleman J, Sohn TA, Campbell KA, Choti MA. Does prophylactic octreotide decrease the rates of pancreatic fistula and other complications after pancreaticoduodenectomy? Results of a prospective randomized placebo-controlled trial. Ann Surg. 2000;232:419-429. [PubMed] |
| 10. | Nagai S, Fujii T, Kodera Y, Kanda M, Sahin TT, Kanzaki A, Hayashi M, Sugimoto H, Nomoto S, Takeda S. Recurrence pattern and prognosis of pancreatic cancer after pancreatic fistula. Ann Surg Oncol. 2011;18:2329-2337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 54] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 11. | Bassi C, Dervenis C, Butturini G, Fingerhut A, Yeo C, Izbicki J, Neoptolemos J, Sarr M, Traverso W, Buchler M. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138:8-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3690] [Cited by in RCA: 3345] [Article Influence: 159.3] [Reference Citation Analysis (4)] |
| 12. | Bassi C, Butturini G, Molinari E, Mascetta G, Salvia R, Falconi M, Gumbs A, Pederzoli P. Pancreatic fistula rate after pancreatic resection. The importance of definitions. Dig Surg. 2004;21:54-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 223] [Article Influence: 9.7] [Reference Citation Analysis (4)] |
| 13. | Tani M, Kawai M, Hirono S, Hatori T, Imaizumi T, Nakao A, Egawa S, Asano T, Nagakawa T, Yamaue H. Use of omentum or falciform ligament does not decrease complications after pancreaticoduodenectomy: nationwide survey of the Japanese Society of Pancreatic Surgery. Surgery. 2012;151:183-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 62] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 14. | Choe YM, Lee KY, Oh CA, Lee JB, Choi SK, Hur YS, Kim SJ, Cho YU, Ahn SI, Hong KC. Risk factors affecting pancreatic fistulas after pancreaticoduodenectomy. World J Gastroenterol. 2008;14:6970-6974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 35] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 15. | Kanda M, Fujii T, Kodera Y, Nagai S, Takeda S, Nakao A. Nutritional predictors of postoperative outcome in pancreatic cancer. Br J Surg. 2011;98:268-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 475] [Article Influence: 31.7] [Reference Citation Analysis (5)] |
| 16. | Warschkow R, Ukegjini K, Tarantino I, Steffen T, Müller SA, Schmied BM, Marti L. Diagnostic study and meta-analysis of C-reactive protein as a predictor of postoperative inflammatory complications after pancreatic surgery. J Hepatobiliary Pancreat Sci. 2012;19:492-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 17. | Callery MP, Pratt WB, Kent TS, Chaikof EL, Vollmer CM. A prospectively validated clinical risk score accurately predicts pancreatic fistula after pancreatoduodenectomy. J Am Coll Surg. 2013;216:1-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1091] [Cited by in RCA: 985] [Article Influence: 75.8] [Reference Citation Analysis (2)] |
| 18. | Ahmad SA, Edwards MJ, Sutton JM, Grewal SS, Hanseman DJ, Maithel SK, Patel SH, Bentram DJ, Weber SM, Cho CS. Factors influencing readmission after pancreaticoduodenectomy: a multi-institutional study of 1302 patients. Ann Surg. 2012;256:529-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 229] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 19. | Chua TC, Chong CH, Liauw W, Zhao J, Morris DL. Inflammatory markers in blood and serum tumor markers predict survival in patients with epithelial appendiceal neoplasms undergoing surgical cytoreduction and intraperitoneal chemotherapy. Ann Surg. 2012;256:342-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Lippi G, Meschi T, Cervellin G. Inflammatory biomarkers for the diagnosis, monitoring and follow-up of community-acquired pneumonia: clinical evidence and perspectives. Eur J Intern Med. 2011;22:460-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 21. | Herzum I, Renz H. Inflammatory markers in SIRS, sepsis and septic shock. Curr Med Chem. 2008;15:581-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 111] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 22. | Kakita A, Takahashi T, Yoshida M, Furuta K. A simpler and more reliable technique of pancreatojejunal anastomosis. Surg Today. 1996;26:532-535. [PubMed] |
| 23. | Kleespies A, Rentsch M, Seeliger H, Albertsmeier M, Jauch KW, Bruns CJ. Blumgart anastomosis for pancreaticojejunostomy minimizes severe complications after pancreatic head resection. Br J Surg. 2009;96:741-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 133] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 24. | Vigushin DM, Pepys MB, Hawkins PN. Metabolic and scintigraphic studies of radioiodinated human C-reactive protein in health and disease. J Clin Invest. 1993;91:1351-1357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 390] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 25. | Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest. 2003;111:1805-1812. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3343] [Cited by in RCA: 1242] [Article Influence: 54.0] [Reference Citation Analysis (0)] |
| 26. | Warschkow R, Beutner U, Steffen T, Müller SA, Schmied BM, Güller U, Tarantino I. Safe and early discharge after colorectal surgery due to C-reactive protein: a diagnostic meta-analysis of 1832 patients. Ann Surg. 2012;256:245-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 133] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 27. | Kolaczkowska E, Kubes P. Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol. 2013;13:159-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4621] [Cited by in RCA: 4070] [Article Influence: 313.1] [Reference Citation Analysis (3)] |
| 28. | Becker KL, Snider R, Nylen ES. Procalcitonin assay in systemic inflammation, infection, and sepsis: clinical utility and limitations. Crit Care Med. 2008;36:941-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 374] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 29. | Aikawa N, Fujishima S, Endo S, Sekine I, Kogawa K, Yamamoto Y, Kushimoto S, Yukioka H, Kato N, Totsuka K. Multicenter prospective study of procalcitonin as an indicator of sepsis. J Infect Chemother. 2005;11:152-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 86] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 30. | Kanda M, Fujii T, Nagai S, Kodera Y, Kanzaki A, Sahin TT, Hayashi M, Yamada S, Sugimoto H, Nomoto S. Pattern of lymph node metastasis spread in pancreatic cancer. Pancreas. 2011;40:951-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 87] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 31. | Bassi C, Molinari E, Malleo G, Crippa S, Butturini G, Salvia R, Talamini G, Pederzoli P. Early versus late drain removal after standard pancreatic resections: results of a prospective randomized trial. Ann Surg. 2010;252:207-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 384] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 32. | Kawai M, Tani M, Terasawa H, Ina S, Hirono S, Nishioka R, Miyazawa M, Uchiyama K, Yamaue H. Early removal of prophylactic drains reduces the risk of intra-abdominal infections in patients with pancreatic head resection: prospective study for 104 consecutive patients. Ann Surg. 2006;244:1-7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 354] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 33. | Welsch T, Frommhold K, Hinz U, Weigand MA, Kleeff J, Friess H, Büchler MW, Schmidt J. Persisting elevation of C-reactive protein after pancreatic resections can indicate developing inflammatory complications. Surgery. 2008;143:20-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 93] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
P- Reviewers: El-Sayed M, Mathew A S- Editor: Gou SX L- Editor: A E- Editor: Wang CH