Published online Jun 14, 2014. doi: 10.3748/wjg.v20.i22.6725
Revised: February 8, 2014
Accepted: March 19, 2014
Published online: June 14, 2014
Processing time: 229 Days and 11.4 Hours
According to epidemiological studies, twice as many women as men are affected by irritable bowel syndrome (IBS) in western countries, suggesting a role for sex hormones in IBS pathophysiology. Despite growing evidence about the implications of sex hormones in IBS symptom modulation, data on mechanisms by which they influence disease development are sparse. This review aims to determine the state of knowledge about the role of sex hormones in sensorimotor dysfunctions and to address the possible interplay of sex hormones with common risk factors associated with IBS. The scientific bibliography was searched using the following keywords: irritable bowel syndrome, sex, gender, ovarian hormone, estradiol, progesterone, testosterone, symptoms, pain, sensitivity, motility, permeability, stress, immune system, brain activity, spinal, supraspinal, imaging. Ovarian hormones variations along the menstrual cycle affect sensorimotor gastrointestinal function in both healthy and IBS populations. They can modulate pain processing by interacting with neuromodulator systems and the emotional system responsible for visceral pain perception. These hormones can also modulate the susceptibility to stress, which is a pivotal factor in IBS occurrence and symptom severity. For instance, estrogen-dependent hyper-responsiveness to stress can promote immune activation or impairments of gut barrier function. In conclusion, whereas it is important to keep in mind that ovarian hormones cannot be considered as a causal factor of IBS, they arguably modulate IBS onset and symptomatology. However, our understanding of the underlying mechanisms remains limited and studies assessing the link between IBS symptoms and ovarian hormone levels are needed to improve our knowledge of the disease evolution with regard to gender. Further studies assessing the role of male hormones are also needed to understand fully the role of sex hormones in IBS. Finally, investigation of brain-gut interactions is critical to decipher how stress, ovarian hormones, and female brain processing of pain can translate into gut dysfunctions.
Core tip: This review summarizes the current knowledge on the role of ovarian hormones in the pathophysiology of irritable bowel syndrome (IBS). A better understanding of gender differences in IBS may help unveil some key mechanisms contributing to IBS development. We present data on: (1) the modulatory role of ovarian hormones on IBS symptoms; (2) influence of ovarian hormones on risk factors associated with IBS; and (3) potential mechanisms of action, by which ovarian hormones can modulate and/or induce IBS symptoms.
- Citation: Meleine M, Matricon J. Gender-related differences in irritable bowel syndrome: Potential mechanisms of sex hormones. World J Gastroenterol 2014; 20(22): 6725-6743
- URL: https://www.wjgnet.com/1007-9327/full/v20/i22/6725.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i22.6725
Core tip: This review summarizes the current knowledge on the role of ovarian hormones in the pathophysiology of irritable bowel syndrome (IBS). A better understanding of gender differences in IBS may help unveil some key mechanisms contributing to IBS development. We present data on: (1) the modulatory role of ovarian hormones on IBS symptoms; (2) influence of ovarian hormones on risk factors associated with IBS; and (3) potential mechanisms of action, by which ovarian hormones can modulate and/or induce IBS symptoms.
Irritable bowel syndrome (IBS) is the most common functional gastrointestinal (GI) disorder. IBS is typically characterized by chronic abdominal pain and bowel habit disturbance[1]. The chronicity and the nature of IBS symptoms lead to major impairment in patients’ quality of life and to a significant financial burden for the public healthcare system[2]. However, IBS management remains difficult because of the current lack of appropriate treatment[3]. Consequently, understanding the pathophysiological mechanisms underlying IBS is a key issue for drug development[4,5].
Epidemiological data on sex ratio in IBS provides an intriguing and often overlooked potential avenue for deciphering the pathophysiological mechanisms of IBS: the role of sex hormones in IBS etiology. Indeed, IBS is predominantly diagnosed in women, with a female to male sex ratio ranging from 2:1 (questionnaire-based diagnostics) to 4:1 (practice-based diagnostics)[6]. It is also noteworthy that many if not all the comorbid diseases associated with IBS also share this female predominance. To name the most common, fibromyalgia[7], migraine[8], other functional GI disorders such as functional dyspepsia[9], chronic pelvic pain[10], chronic fatigue syndrome[11], and depression[12] all have sex ratio skewed towards female gender[13].
Ovarian hormones can modulate intestinal function and transit speed[14-18]. These properties make ovarian hormones an interesting suspect for explaining gender differences in IBS. There is a strong correlation between IBS and dysmenorrhea[19] and variations in ovarian hormone levels during the menstrual cycle have been shown to modulate IBS symptomatology in women[17,20]. In particular, exacerbation of abdominal pain during perimenses (i.e., the menstruation phase period characterized by low ovarian hormone levels) has been consistently observed when compared to other phases of the menstrual cycle[6,16,21-23]. Interestingly, the same findings have also been observed in inflammatory bowel disease, another chronic intestinal disease[24]. Lower ovarian hormone levels characterize the perimenses period, therefore, these observations suggest that female sex hormones have a protective role against IBS-associated pain[25]. However, studies in this field of research are scarce and the mechanisms of action of ovarian hormones are unclear. For instance, no correlation has been found between plasma ovarian hormone levels and premenstrual symptoms[16,20,26], suggesting more complex interactions between hormones and the gut sensorimotor system.
The goal of this review is to summarize the current knowledge on the influence of ovarian hormones on the pathophysiology of IBS. A better understanding of gender differences in IBS may be a useful approach to unveil some key mechanisms contributing to its development, and eventually, may provide new therapeutic strategies. In this review, we present data on: (1) the modulatory role of ovarian hormones on IBS symptoms; (2) influence of ovarian hormones on risk factors (e.g., stress, permeability and immune system dysfunction) associated with IBS; and (3) potential mechanisms of action, by which ovarian hormones can modulate and/or induce IBS symptoms.
A multidisciplinary search of the clinical and preclinical bibliography was conducted using Medline/PubMed to identify studies assessing involvement of sex hormones in the pathophysiology of IBS. The search was restricted to studies published in English up to October 2013. The following key words were used to identify original articles with potential relevance to gender differences in IBS, effect of menstrual cycle, menopausal status or hormonal treatments on IBS symptomatology: irritable bowel syndrome, sex, gender, ovarian hormone, estradiol, progesterone, testosterone, symptoms, pain, sensitivity, motility, permeability, stress, immune system, brain activity, spinal, supraspinal, imaging.
The bibliography search yielded a total of 473 articles. The articles retrieved by the Medline/PubMed search were evaluated based on their title and abstract. Studies including patients diagnosed using Rome I, II or III criteria were included. Individual case reports, abstracts and studies published in languages other than English were excluded. Additionally, relevant publications cited in reviews, but which were not captured by the search strategy, were also considered. After selection, 175 papers were analyzed and used to write the review. The outcome measure was the description of ovarian-dependent changes in gut immunity, gut sensori-motricity, gut permeability, or pain processing, which were compared between IBS patients and healthy controls.
The first part of this review describes associations between gender and IBS intestinal symptoms, and aims at deciphering the influence of ovarian hormones on sensorimotor dysfunction. Sensorimotor dysfunction is the key feature of IBS pathophysiology and results in altered bowel habits and visceral hypersensitivity, the two major symptoms leading patients to seek medical attention. In the second part, the review details the impact of gender and ovarian hormones on central and peripheral alterations frequently associated with IBS, namely stress, gut barrier permeability and immune system activation.
Female gender and influence of ovarian hormones on GI motility: Generally, women display slow GI transit compared to men, with delayed gastric emptying[27] and reduced colonic transit time[28]. This gender difference in GI motility could be, at least partially, due to ovarian hormone variations during the menstrual cycle. Indeed, it has been shown in healthy women that GI transit duration tends to be prolonged during the luteal phase and at onset of menses compared to the follicular phase[29], although this finding was not consistently replicated[30,31]. Further support is given to the idea of a modulatory role of ovarian hormones on GI motility by the frequent association between hormonal changes during pregnancy and the co-occurrence of GI motility disorders[32,33]. GI transit time is significantly prolonged in the third trimester of pregnancy when ovarian hormone levels are increased, compared to the postpartum period[32]. Although mechanical causes inherent to morphological changes during pregnancy can account for bowel habit disturbances[34], it seems that the endocrinological changes are more likely to be the accountable causes of GI motor impairments. Preclinical studies showed an increase in the release of NO in the vascular compartment and from the nonadrenergic, noncholinergic nerves, innervating the proximal colon, during late pregnancy compared with mid-pregnancy[35]. This NO increase is responsible for motility decrease in the GI tract and is thought to be mediated by estradiol[36]. Other animal studies confirmed the correlation between hormonal fluctuations and GI motility. In naive female rats, a biphasic transit pattern was observed during estrus cycling with a slow transit during proestrus and estrus (i.e., the period preceding ovulation and during which estrogen levels rise up) compared to metestrus-diestrus phases (i.e., the period following ovulation, characterized by decreased estrogen levels and increased progesterone levels)[37]. Moreover, pregnant animals have a transit time comparable to those of rats in proestrus-estrus phases, when estrogen levels are increased[37], thus supporting clinical observations.
GI complaints during the menstrual cycle can be related to motility changes and/or altered perception of intestinal motor events. These abnormalities can be, at least partially, mediated by changes in ovarian hormone levels as demonstrated by clinical and animal studies highlighting a differential role of these hormones on GI motility. Indeed, it has been shown that estrogens inhibit colonic smooth muscle contractility via a nongenomic mechanism as suggested by the rapid onset and reversible action of the ovarian/sex steroids[38]. Animal studies further showed that estrogens have a peripheral action on smooth muscle contractility. For instance, in ileal tissue isolated from ovariectomized rats, contractions induced by the cholinergic agonist carbachol were impaired in rats treated with estradiol for 3 d. This alteration is due to the inhibition by estradiol of RhoA signaling, a small GTPase protein known to regulate smooth muscle contractility[39]. Inhibitory effects of estrogen on colonic contractile activity have been confirmed in isolated rabbit distal colon in which estradiol potentiates the inhibitory effects of oxytocin on distal colonic contraction[40]. Interestingly, estradiol had no effects on the oxytocin-induced decrease in motility in the proximal colon[41], suggesting a fine regulation of intestinal motility by ovarian hormones depending on GI segments. Oxytocin and estradiol display diverse and sometimes opposite effects depending on species, methods and gut segments considered, adding complexity to our limited understanding of these mechanisms[42,43]. Thus, another report demonstrated that in rats, systemic administration of oxytocin excited colonic motility during proestrus and estrus when estradiol concentration is elevated but not during diestrus when estradiol concentration is low[44].
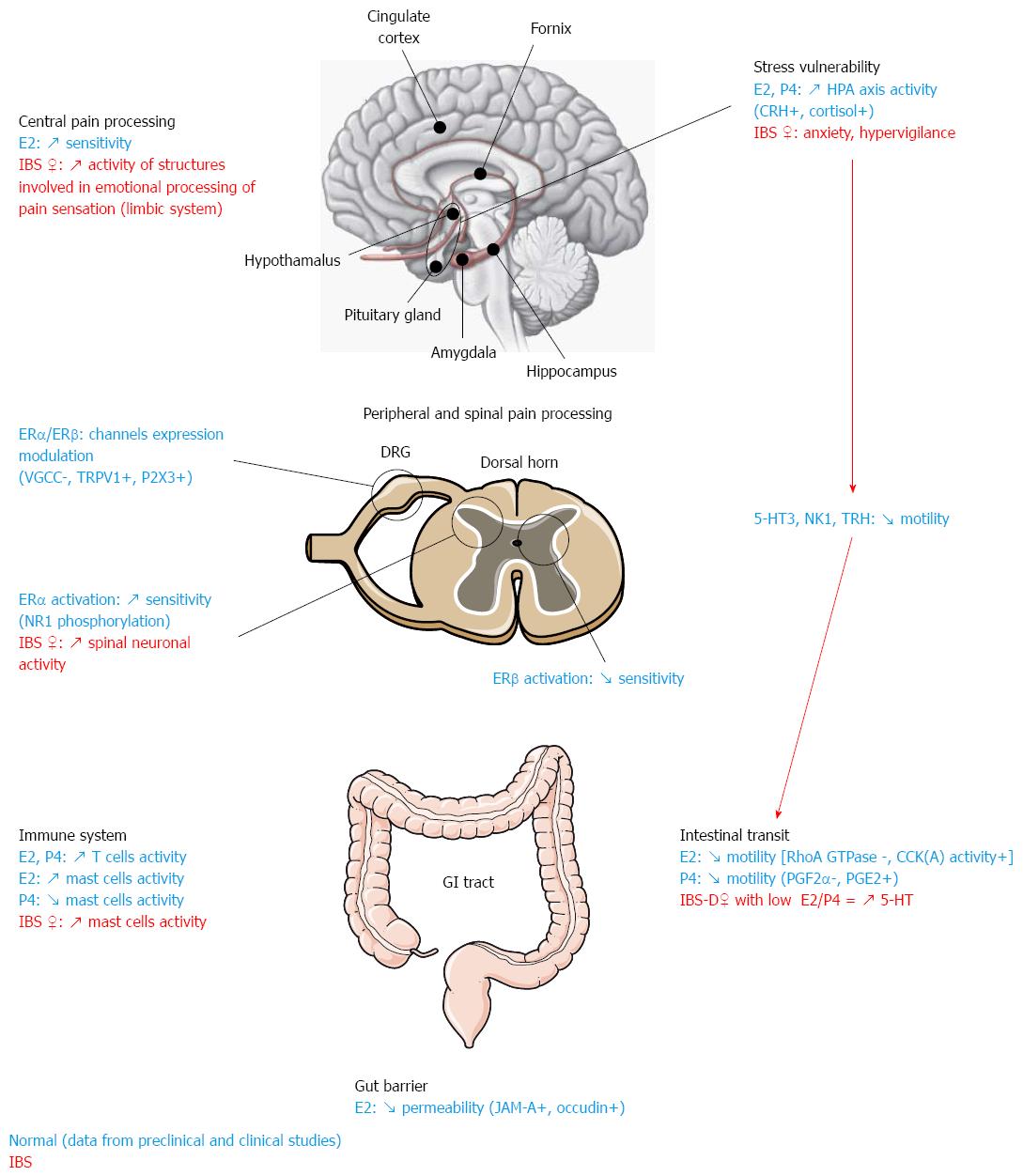
Estrogen can also affect intestinal motility via the involvement of peptidergic hormones. For instance, in ovacter-iomized rats, estradiol benzoate (EB), an estrogen-like hormone, inhibited GI transit and this inhibition was correlated with cholecystokinin (CCK) plasma concentration. CCK(A) receptor antagonists attenuated EB-induced inhibition of GI transit while CCK(B) receptor antagonists had no effects. These results suggest that GI transit estradiol-induced inhibition involves CCK and CCK(A) receptor activation[42].
Similarly to estrogen, data from studies performed in different species suggest an inhibitory effect of progesterone on GI tract motility[45-49]. However, it is noteworthy that depending on the dose, progesterone displays opposite effects. Indeed, while a decrease in GI motility is generally described with high doses, low dose of progesterone seems to induce a motility increase[47]. Several mechanisms have been proposed to explain the differential effects of progesterone. In the guinea pig distal colon, progesterone inhibits transit by altering the normal levels of prostaglandins that induce contraction (PGF2α) and relaxation (PGE2) of intestinal muscles. This is the result of changes in the pattern of G proteins which regulate prostaglandins expression[48]. Similar mechanisms have been observed in intestinal muscle cells from women with chronic constipation, in which G proteins promoting contractions were downregulated and inhibitory G proteins were, by contrast, upregulated. In this study, these abnormalities were reproduced in vitro by pretreatment of normal colonic muscle cells with progesterone, suggesting a direct involvement of progesterone and its receptor activation on colonic muscle contractility[48]. Interplay between progesterone and the serotoninergic system could also underlie altered bowel habits in women. Serotonin (5-hydroxytryptamine or 5-HT) is known to play a key role in the motor function of the GI tract by regulating smooth muscle contractility[50]. It has been shown that progesterone administration globally increased 5-HT levels by decreasing the level of serotonin transporter (SERT) which participates in 5-HT reuptake[51], monoamine oxidase mRNA[52,53] expression, and increasing the availability of 5-HT precursor, tryptophan[53]. This effect of progesterone on the serotoninergic system is surprising because 5-HT, in many cases, promotes peristalsis. However, recent studies demonstrated that in women with slow transit constipation (STC), progesterone receptors were overexpressed in colonic muscle[54] and epithelial cells[55]. SERT levels were lower and 5-HT concentration was higher than in healthy controls. These results are difficult to explain but the authors hypothesized that increased levels of 5-HT are mostly ineffective in female patients with STC because of the overexpression of progesterone receptors in muscle cells, which impairs the contraction of the circular muscle layer[55]. Nevertheless, the interaction between progesterone, 5-HT and muscle contractions remains to be clarified.
Influence of ovarian hormones on motility in IBS pathophysiology: The aforementioned studies were conducted in healthy volunteers, naive animals or isolated organs. Only few studies assessing the effects of ovarian hormones on intestinal motility are available in IBS patients or in animal models of impaired motility. Female IBS patients are more likely to report constipation-related symptoms including abdominal distension, bloating, infrequent stools and hard stools than men with IBS[56]. In contrast, men report more frequently diarrhea-related symptoms of loose stools and increased stool frequency[57]. Interestingly, at the time of menses, when ovarian hormones are low, diarrhea symptoms become more prevalent in women than constipation does[56]. It is important to notice that menstrual cycle effects on motility symptoms were similar in healthy women and IBS patients but symptom severity was greater in women with IBS[56]. The 5-HT system plays a pivotal role in motility impairment associated with IBS, because it has been shown that postprandial platelet-depleted plasma 5-HT concentration was abnormally elevated in IBS patients with diarrhea (IBS-D)[58-60] but reduced in IBS patients with constipation (IBS-C)[60,61] compared with healthy volunteers. These observations are consistent with animal studies reporting accelerated GI transit induced by increased 5-HT levels[62]. Houghton et al[63] highlighted the influence of the menstrual status on plasma 5-HT concentration by showing that IBS-D women who exhibit lower estrogen and progesterone levels at time of menses have an increased 5-HT plasma concentration compared to IBS-D women with high ovarian hormone levels. Although no correlation with motility symptoms were made in this study, it pointed out differential 5-HT metabolism depending on the cycle phases considered in IBS-D patients. Given the involvement of 5-HT system in GI motor function, these data suggest a dynamic role for ovarian hormones in GI transit impairments observed in IBS-D patients.
Key points: Women display slow GI transit compared to men; transit time is prolonged when ovarian hormone levels are high; estrogen and progesterone inhibit smooth muscle contraction; progesterone modulates the colonic 5HT system, which is known to regulate peristalsis; female IBS patients report constipation-related symptoms more often than men, except at the time of menses, when ovarian hormone levels are low; ovarian hormones may contribute to altered motility in IBS-D patients by interacting with the 5HT system.
Influence of gender and sex hormones on visceral pain: Over the past 20 years, clinical studies regarding the influence of gender on pain responses have built up a body of evidence demonstrating that women are at greater risk for many pain conditions than men. It is now well established that prevalence of most common forms of pain is higher in women than men, and women display increased sensitivity to several forms of experimentally induced pain including visceral pain[13,64,65]. Accordingly, women are more likely to report abdominal pain, increased pain perception and discomfort to colorectal distension (CRD), and pain-related IBS symptoms than men do[56,66,67]. Similar observations have been made in rodents, in which females have greater visceromotor response to CRD than males[68-72]. The influence of gonadal hormones on nociception could explain these gender-related differences. Accordingly, animal studies showed that hormonal status is able to modulate visceral and somatic sensitivity to different nociceptive stimuli[73-77].
Modulation of peripheral pain processing by ovarian hormones: Altered visceromotor response to CRD in rodents after ovariectomization indicate that ovarian hormones modulate gut sensitivity[78-80] and this is corroborated by changes in visceral sensitivity during the estrus cycle. Indeed, in female rats, visceral sensitivity is increased during proestrus compared to met/diestrus[81,82]. In ovariectomized rats, estradiol administration increases visceromotor response to a visceral painful stimulation consisting of colon or bladder distension[75,76,78,79,83-87], whereas progesterone is thought to counteract the effects of estradiol by an antinociceptive action[83]. However, depending on the dose, ovarian hormones can have opposite effects, and if estrogens are thought to be pronociceptive at physiological concentrations, one study showed that estradiol can be antinociceptive at supraphysiological levels[88]. Furthermore, conflicting results come from studies in different species. For instance, unlike rats, ovariectomized mice displayed long lasting visceral hyperalgesia, which was reversed by estradiol administration[26].
Estrogen receptor (ER)α and ERβ are both expressed at a peripheral level in small-diameter dorsal root ganglion (DRG) neurons conducting nociceptive information[89]. ER activation can modulate nociception by regulating the expression of ion channels and receptors in sensory neurons. Estradiol can inhibit high-voltage activated calcium channel of L and N type expressed on primary afferents[90]. Moreover, the ATP purinoceptor P2X3 and the capsaicin-sensitive transient receptor potential vanilloid type 1 (TRPV1) receptor, known to participate in visceral nociceptive integration, are both downregulated in ERα and ERβ knock-out (KO) mice[91]. Further studies demonstrated that ATP-induced [Ca2+] in DRG neurons is attenuated following estradiol administration in wild type and ERβ KO but not in ERα KO mice[92,93], suggesting that this attenuation depends on ERα specifically. Thus, an interaction between P2X3 and ERα in primary sensory neurons could underlie gender differences in visceral nociception. Finally, recent research highlighted the potential role of a new estrogen receptor called GPR30 in estrogen-dependent visceral hypersensitivity induced by 5-HT[94].
Modulation of central pain processing by ovarian hormones: Modulation of visceral nociceptive signal by sex hormones at the spinal cord level is also complex. ERα is expressed predominantly in the superficial dorsal horn, which receives information from nociceptive sensory neurons, while ERβ is expressed in deeper layers suggesting a differential function for these receptors in nociceptive processing. Spinal ERs are thought to play an important role in the modulation of visceral sensitivity as suggested by the finding that administration of the ERα agonist 4,4′,4′′-[4-propyl-(1H)-pyrazole-1,3,5-triyl]tris-phenol via intrathecal route increases visceromotor response to CRD in ovariectomized rats[87]. In contrast, administration of the ERβ agonist diaryl proprionitrile attenuates the response of visceroceptive dorsal horn neurons and the visceromotor response during CRD, suggesting antinociceptive action of ER[95]. The proposed mechanism of action of ERs on visceral sensitivity at the spinal level is gating of synaptic transmission by modulation of ion channel activity[71,84,96]. Some studies have explored the possible interaction between ERα and the NR1 subunit of the glutamate N-methyl-D-aspartate receptor (NMDA). Both receptors are coexpressed in dorsal horn neurons, and it was shown that estradiol increases spinal processing of visceral nociception by upregulating NR1 expression and activity following PKA-mediated NR1 phosphorylation. In contrast, ovariectomy increases the potency of the NMDA receptor antagonist (2R)-amino-5-phosphonovaleric acid in modulating the visceromotor response to CRD[71,84]. Taken together, these data confirm the pronociceptive effect induced by spinal ERα activation in the rat.
In the brain, a recent study showed in ovariectomized rats receiving implants of estradiol that elevated levels of estradiol in the amygdala correlated with increased visceromotor response to CRD. Estradiol administration to adjacent brain areas had no such effect and did not affect somatic sensitivity[80]. The underlying mechanisms of the central action of estrogens in the amygdala have not been investigated yet but could involve opioid receptors, which are highly expressed in this brain area[97-99] and have been implicated in the estrogen-dependent differential effects of morphine[70,100].
Gender differences in peripheral visceral pain processing and IBS pathophysiology: Gender-related differences in somatic and visceral sensitivity in the general population have been widely described, however, the link between gender and pain-related symptoms in IBS is still discussed. The most recent meta-analysis[56] did not reveal any difference in visceral-pain-related symptoms between men and women in the IBS population. This finding contrasts with several studies reporting greater abdominal pain and a reduction in discomfort thresholds during phasic CRD in women compared with men[66,67,101]. Discrepancies between these studies may be attributed to the fact that menstrual cycle phase, menopausal status and hormonal therapy were not taken into account.
Only a limited number of studies have investigated correlations between pain perception and hormonal status. Yet, sex hormones are likely to play an important role in the pathogenesis of visceral hypersensitivity in IBS. This is supported by observations that premenopausal patients present exacerbation of their abdominal pain symptoms at the time of menses[102,103]. Moreover, when considering the different phases of the cycle, worsening abdominal pain and increased rectal perception in response to CRD specifically are observed at the time of menses compared with other phases in IBS patients[25]. Interestingly, it was shown in healthy volunteers that rectal sensitivity was not influenced by the menstrual cycle, suggesting that ovarian hormone fluctuations only affect lower GI sensitivity in pathological conditions[22]. Globally, a negative correlation between female gonadal hormone levels and pain severity is pointed out in several studies supporting a protective role of ovarian hormones. For example, a recent analysis using a population-based questionnaire reported that abdominal pain/discomfort increases after menopause, when ovarian hormones levels fall down, compared to premenopausal women[104]. Nevertheless, some data are conflicting because another study reported that abdominal pain is reduced in women over 50 years old, suggesting a benefit of menstrual cycle cessation[105]. Discrepancies may result from different clinical designs, because the studies used different diagnostic criteria for IBS and different recruitment strategies (i.e., 10-year longitudinal follow-up vs single questionnaire). In a recent meta-analysis comparing relative risk of individual IBS symptoms between men and women at different times of the cycle, Adeyemo et al[56] concluded that sensitivity was globally increased during menses compared to other phases of the cycle but further analysis is needed to conclude definitively on the involvement of hormonal status in visceral pain. It is noteworthy that these clinical observations are not consistent with preclinical results because increased sensitivity in women is observed when estrogen levels are low, whereas estrogen has globally a pronociceptive effect in the rat. These data suggest that some of the effects of estradiol on visceral nociception are species dependent. Moreover, estrogens may have differential effects depending on the organ considered. Indeed, results from experiments investigating the role of ovarian hormones on vaginal sensitivity in a rat model of ureteral stone suggested an anti-nociceptive role of estradiol. The study showed lower vaginal sensitivity during proestrus/estrus (high estradiol levels), than during met/diestrus[73]. These dual effects could result from different functions of ERα and ERβ.
On the fringe of research on the link between IBS and sex hormones, a few authors have raised the question of the possible protective effect of male sex hormones. In a study including 50 male IBS patients compared to 25 controls, serum and free testosterone were measured and correlated to sensitivity to CRD. Sensory thresholds for urgency and discomfort in patients were negatively correlated with testosterone levels, indicating a potential protecting effect of male hormones[106]. In animals, one study evaluated the effects of testosterone administration in a visceral pain model induced by artificial calculus. They concluded that testosterone had no effect on visceral pain because operated animals presented an increase in frequency and duration of ureteral crises independently from testosterone treatment[88].
Peripheral mechanisms underlying visceral hypersensitivity in IBS patients are widely unknown. Clinical trials using pharmacological drugs targeting the serotoninergic system in patients with visceral hypersensitivity have led to the proposal that gender differences may rely upon 5-HT-dependent mechanisms. In particular, the 5-HT3 antagonist alosetron provides abdominal pain relief with a greater effect in women than men[107]. This difference of efficacy could be explained by ovarian-hormone-driven 5-HT3 upregulation, making women more sensitive to this therapeutic agent. Nevertheless, studies are needed to understand the link between 5-HT receptors, estrogens and visceral hypersensitivity in IBS pathology.
Gender differences in central visceral pain processing and IBS pathophysiology: In IBS, the diminished thresholds of perception to visceral experimental stimulations in women compared to men can result not only from sex-related differences in peripheral encoding of the visceral pain message, but also from differences in spinal and supraspinal processing of this message, and/or from a greater reactivity of arousal (attention) and emotional (stress) circuits.
Gender differences in visceral pain perception in IBS have originally been attributed to hypervigilance, and it was suggested that this pronounced hypervigilance was primarily driven by greater anxiety in women[108]. Kilpatrick and colleagues confirmed the involvement of hypervigilance but only in naturally cycling female IBS patients[109], thus stressing the complex involvement of ovarian hormones in the process.
Gender differences in pain perception can also reflect structural and functional differences in central visceral nociceptive pathways. Indeed, women display differences in the processing of pain messages, such as enhanced “wind-up” in the spinal cord for instance[110,111] which corresponds to the progressive increase of the electrical response to repeated nociceptive stimulation in spinal cord neurons, possibly via the involvement of sex hormones and their spinal receptors[112].
Functionally, functional magnetic resonance imaging (fMRI) and positron emission tomography studies showed that colorectal stimulation and expectation of abdominal pain activate preferentially the arousal/emotional circuits (anterior cingulate cortex and amygdala) in female patients, while male patients present greater activation of the cortical regions (insula and dorsal prefrontal cortex)[113-118]. These patterns of activation were replicated with remarkable similarity in rodents using cerebral-blood-flow-related tissue autoradiography after aversive CRD[119,120]. The results indicate that sex-related differences in brain response to visceral stimulation are mainly due to alterations in the emotional arousal circuitry rather than visceral afferent processing circuits. However, in a task-free resting-state fMRI study, although disease-related differences (IBS vs healthy controls) were observed, no differences in emotional arousal circuit activation were evidenced between male and female IBS patients[121]. Moreover, the idea of a greater subjectivity and emotional response in women and of greater cortical control of limbic structures in men was recently challenged by an imaging study monitoring brain activity following the presentation of more male-oriented emotions to female and male patients. In this study, and contrary to previous reports, presentation of faces expressing fear and anger elicited greater activation of the emotional arousal circuits in men and greater activation of the cortical modulatory circuits in women[122]. Thus, the nature and the emotional valence of the stressor are critical to explain gender differences in brain activation patterns.
Structurally, there are widespread gray matter changes in IBS patients although the data is equivocal. Reductions have been reported in the insular and anterior cingulate cortices[123], while another study found increased gray matter thickness in viscera and somatosensory regions (insula and somatosensory cortex S2) with no change in the cingulate[124]. A recent study found no disease-related difference at all between patients and controls[125]. However, the same study reported a sex-related difference in patients, with IBS female subjects showing decreased cortical thickness in the anterior cingulate and insula, and increased cortical thickness in somatosensory and primary motor cortex when compared to female healthy controls. These alterations correlated with IBS symptom severity[125]. White matter and connectivity alterations have also been observed in IBS patients and are more marked in female than male subjects[126,127]. The data indicate that IBS patients have greater connectivity, with sex-specific patterns. Males have stronger connectivity between anterior cingulate subregions, amygdala, and insula, while females have stronger connectivity to and from the prefrontal modulatory regions[127].
If progress has been made in identifying circuits involved in gender-differences in IBS, little is known about their molecular determinants. One study evaluated the impact of serotonin transporter gene polymorphism on brain activation upon CRD and found that subjects with a weak function of the serotonin transporter (s/s genotype) respond with more activation in emotion-regulating brain regions such as the amygdala[128]. Similar alterations of amygdala reactivity were observed during emotional face processing[129]. Further support for the idea of sexual dimorphism of the serotoninergic system came from a study in rodents, which showed that in response to CRD, female but not male rats showed significant changes in cerebral blood flow in the raphe nucleus and in many target regions of its serotoninergic efferent projections[120]. Estrogens can act centrally as steroid neuromodulators to alter the function of glutamatergic or GABAergic systems and modulate synaptic plasticity[72,130]. Moreover, estrogens are known for their ability to shape dendritic spines and modify synaptic plasticity[131-133]. It is therefore likely that gender-related differences in these systems underlie gender-related differences in central processing of pain in IBS, especially during periods when estrogen levels are high. Further studies addressing these questions are warranted.
Key points: In IBS patients, heightened abdominal pain sensation is observed at the time of menses (low ovarian hormone concentrations), which suggests a protective role of estrogens; estrogens modulate peripheral and central nociceptive pathways and have a pro- or antinociceptive action on visceral sensation, depending on the model studied and the neurotransmitter system considered (MOR, NMDA, GABA, NK1); the 5-HT system may contribute to gender differences in pain-related IBS symptoms via modulation of gut sensitivity in the periphery and modulation of pain-associated emotional circuits in the brain; female IBS patients present higher activity of brain structures involved in emotional processing of pain sensation (i.e., insula, cingulate cortex and amygdala) and greater connectivity between brain structures involved in cortical control (i.e., prefrontal cortex).
Stress and female gender in IBS: Women are more vulnerable to life stress[134,135], are more prone to anxiety and depression[136], are exposed to life traumas more often than men[137], and present exacerbated IBS symptoms under stress[16], therefore, it was proposed that the interaction between ovarian and stress hormones may explain female predominance in IBS[138-142].
However, studies assessing gender difference in response to different types of stress failed to show a correlation between women’s vulnerability to stressful adverse life events and neuroendocrine responses. In contrast, it was observed that male IBS patients have greater autonomic responses to a visceral stressor (rectosigmoid balloon distension)[143] and have greater autonomic responses and adrenocorticotropic hormone and cortisol release in response to stress[144,145] than female patients. These results suggest that gender differences in the effects of sex hormones on the autonomic systems and hypothalamo-pituitary axis (HPA) cannot account for differential responses to stress in IBS. However, the parameters of autonomic response assessed in these studies were indirect (e.g., skin conductance and heart rate) and stress hormones levels were determined only in plasma and saliva but not in tissue. Also, methodological confounders such as menstrual phase of the cycle in female patients are not always factored in the data analyses or the clinical designs.
Hence, one cannot rule out the possibility of subtle hormone level changes in specific windows of time, or that molecular and cellular changes occur at the tissue level. Indeed, in rodent models, where the experimental designs can be controlled in a tighter manner and where invasive methodologies (e.g., cerebral infusions of hormones) can be used, it was shown that there is a positive correlation between the levels of estradiol and progesterone and the levels of cortisol, corticotropin releasing hormone (CRH) and its receptors[146-149]. This is particularly true during the proestrus phase, where the levels of estradiol are at their nadir[150-152]. Consequently, these data indicate that ovarian hormones can increase HPA axis activation and that further studies in humans are required. Accordingly, the most recent research has been focusing on the link between ovarian hormones and gut response to stress (see below “Stress, ovarian hormones and IBS pathophysiology”).
Finally, discrepant findings in humans can also be explained by the fact that ovarian hormones may influence stress responses through other ways than a direct action on the HPA axis, primarily by inducing changes in emotion regulation strategies through tuning up of limbic system reactivity[114,122,141]. In IBS patients, it was observed that women are psychologically more reactive to stress as showed by suggestive stress ratings[153]. A study showed that female IBS patients also had increased negative affect both at baseline and in response to a psychological stressor, even though this was not associated with any change in autonomic responses[154]. This altered emotional balance may explain IBS patients’ somatic and psychiatric comorbidity, as well as heightened awareness of bodily sensations[155,156]. In line with the higher prevalence of stress and abuse history in women with IBS, it is thus possible to hypothesize that early traumatic experience can later influence vulnerability to stress mediated by altered limbic system activation[157].
Stress, ovarian hormones and IBS pathophysiology: Preclinical studies performed in rodents have shown that stress can have deleterious effects on gut physiology via mechanisms involving ovarian hormones. For instance, restraint stress can decrease colon motility and increase colon contractility in ovariectomized rats when compared to the sham group and these alterations are reversed by administration of estradiol and progesterone[158]. The effects of ovarian hormones in this paradigm were mediated by the serotonin receptor 5-HT3R[158], the neurokinin receptor NK1[159] and the stress-related hormone thyrotropin-releasing hormone[160]. Most importantly, extensive work by Yvette Taché’s group showed that the interplay between CRH and estrogen signaling pathways, both peripherally and centrally, modulates visceral hypersensitivity induced by colorectal distension in rodents[161-163]. These data suggest that overactivity of CRH signaling in the brain and the gut could explain the comorbidity of stress, depression and IBS in women[162].
In humans, it was shown by comparing healthy women with a life stress history and low-stressed women, equilibrated by menstrual phase, that life stress can confer a gut vulnerability[164]. In this study, women experiencing significant life events presented increased jejunal permeability and diminished secretory ability after being subjected to a mild cold pain stress. Interestingly, the abnormal jejunal epithelial response occurred independently of any autonomic activation or stress hormones release, and was not associated with changes in the levels of ovarian hormones. These findings suggest that a history of stress can induce maladaptive intestinal epithelial response to new stressors, even mild stressors, which do not induce autonomic or HPA activation[164].
A follow-up study by the same group addressed the question whether gender differences underlie maladaptive intestinal response to stress and showed that the mild cold pain stress specifically increased jejunal intestinal macromolecular permeability in women. No differences were detected in the autonomic, hormonal and psychological responses to acute stress parameters between men and women, hence suggesting that the maladaptive gut response is due to gender differences in the intestinal barrier[165]. The molecular mechanisms underlying such a vulnerability are still unclear but animal models suggest that CRH can enhance gut permeability by activating epithelial mast cells[166-170] and subsequent mast cell-dependent decrease of tight junction proteins expression[171,172].
Key points: IBS onset and symptoms severity are associated with acute or chronic stress; IBS female patients present emotional hyper reactivity to stress (limbic system hyper reactivity to stress); levels of estradiol, progesterone and stress hormones are positively correlated in rodent models of IBS; female intestinal barrier vulnerability in response to stress may underlie gender differences in IBS.
Estrogens and the gut barrier: ERα and ERβ have been identified in the GI tract, where ERβ is preferentially expressed on colon epithelial cells[173-177]. Estrogen functions in the gut encompass development and regulation of gut barrier, cell differentiation and proliferation, and architectural maintenance of the intestinal epithelium[177-179]. Studies on the role of estrogens on paracellular permeability and gut barrier integrity are scarce but there is now growing evidence that ERs play a key role in their regulation. Indeed, ERs have been implicated in chloride ion secretion[180] and nutrient transluminal transport[181,182]. Also, indirect evidence from contraceptive use, pregnancy, and hormone replacement therapy in patients with barrier-dysfunction-related pathologies such as inflammatory bowel diseases show that fluctuations in ovarian hormone levels influence the course of and the risk for these diseases[183-185]. Altogether, these observations suggest that ovarian hormones modulate gut permeability. Hence, they have prompted interest in studying the link between estrogens and gut barrier integrity, and especially paracellular permeability. Recent studies in female rodents have provided insights into how estrogens and ERβ signaling can modulate paracellular permeability. These pharmacological studies showed that distal colon epithelial paracellular permeability varies during the sexual cyclic fluctuations of estrogen levels due to variations in tight junction protein expression. Specifically, estrogen level peak during the follicular phase correlated with improved epithelial barrier as shown in the ileum of proestrus rats[186]. Decreased intestinal permeability in male rats was also found following estradiol supplementation[186]. Subsequent investigation of the link between estrogens and the regulation of paracellular spaces found that estradiol influence during the follicular phase resulted in decreased colonic permeability and ERβ-mediated upregulation of the tight junction proteins occludin and junctional adhesion molecule (JAM)-A in epithelial cells[187]. Similarly, the xenoestrogen bisphenol A dose-dependently decreased basal paracellular permeability through upregulation of JAM-A and occludin[188]. Interestingly, progesterone had no effect in these experimental designs. The effects of estrogens on tight junction protein upregulation were also observed in human colon cell line Caco-2[187,188]. In another study, ER expression and colon permeability were negatively correlated in mice and in human HT-29 and T84 colonic epithelial monolayers, thereby confirming a role for estrogens in the maintenance of epithelial permeability in the colon[189]. Finally, chronic oral administration of estrogen compounds (17β-estradiol benzoate and the phytoestrogen-rich soy germ fermented ingredient SG) was associated with less colon permeability during the follicular phase in female rats although the authors did not report any associated effect on occludin expression[190]. Overall, these studies support the idea that estrogens reinforce the gut epithelial barrier through ERβ-mediated upregulation of the tight junction proteins occludin and JAM-A. As such, it is reasonable to propose that estrogens can have a protective role on colon physiology and it has been shown that low ER levels are a vulnerability factor to colon inflammation[186,188,189].
Ovarian hormones, gut barrier and IBS: In the last couple of years, several studies have reported increased intestinal epithelial permeability in IBS patients (for review, see Matricon et al[5]). Intestinal permeability is correlated with IBS symptom severity[191,192]. However, no study has evaluated the link between permeability, symptoms and ovarian hormones so far. Preclinical studies indicate that estrogens are protective but on the contrary, the clear IBS female predominance points to more complex mechanisms. Indeed, the scarce data on this topic suggests the existence of estrogen-dependent maladaptive intestinal epithelial responses to environmental factors such as stress. For instance, Alonso and coworkers specifically showed in healthy women (but not in men) that life or even acute stress could result in decreased jejunal secretory response and increased jejunal permeability[164,165]. Strikingly, these changes were independent of estrogen blood levels. These findings raise the interesting possibility that female over-susceptibility to IBS might be due to a dynamic estrogen-driven maladaptive response of the intestinal barrier to certain risk factors (e.g., stress) rather than a phase-dependent intestinal hyper permeability.
Key points: A subset of IBS patients has increased intestinal permeability; estrogens reduce epithelial barrier permeability by upregulating tight junction proteins; in IBS patients, no correlation between ovarian hormones levels and intestinal permeability has been found.
Effects of ovarian hormones on immune activation: Although immune response is a predominant function of the gut, little is known about the contribution of sex hormones to gut-associated lymphoid tissue activation and most of our knowledge comes from experiments performed in blood samples. A recent immunophenotypic study analyzed lymphocytes population and lymphocyte gene expression in both peripheral blood samples and small intestine biopsies of healthy men and women. This study demonstrated that immune activation and inflammation-associated gene expression were increased in women compared to men[193]. Gender-related differences in immune activation have been pinpointed by the observation of higher incidence of autoimmune diseases in women than in men and are mediated by both humoral and adaptative responses[194]. Gene expression analysis of isolated peripheral blood mononuclear cells recently showed a marked increase in the expression of inflammatory/cytotoxic effector genes, such as interferon, lymphotoxin and interleukin 12 receptor β2, in women compared to men[195]. The differential immune cells gene expression profile detected in women was indicative of enhanced cytotoxic T cell responses compared to men[195]. Sex hormones are thought to play a crucial role in the modulation of immune responses[196-198], possibly by direct action on immune cells. Indeed, a large number of these cells, including T cells and macrophages, expresses ovarian hormone receptors and in particular ERs[199-202]. Thus, estrogen signaling can modulate directly immune responses by regulating immune cell activation and proliferation and cytokine production[203-206]. T cell function is particularly affected by hormonal regulation since both estrogen and progesterone enhance their cytotoxic activity by stimulating reactive oxygen species production[207-209]. In mast cells, progesterone inhibits histamine secretion, which may explain the partial remission in certain inflammatory conditions during pregnancy when progesterone levels are high[210]. In contrast, mast cells express high affinity estrogen receptors and their activation by estradiol has been shown to potentiate histamine and serotonin secretions in mast cells pretreated with the mast cell secretagogue 48/80[211]. Only one study has considered the role of male hormones in immunity. Castration experiments in rats induced a significant decrease in CD4+ and CD25+ T cells and an alteration of CD8+ T cell activation, which could be prevented by testosterone administration[212]. These results show that testosterone can modulate T-cell-mediated immunity and support the notion of gender-related differences in cell-mediated immune response depending on sex hormones.
Ovarian hormones, immune activation and IBS: To the best of our knowledge, only one study established a potential link between gender differences in IBS pathophysiology and immune activation. In this work, Cremon et al[213] performed quantitative immunohistochemistry of colonic immunocytes on endoscopic biopsies of IBS-D and IBS-C patients, and compared the results to patients suffering from microscopic colitis or ulcerative colitis and to healthy controls. IBS patients showed a significant increase in mucosal immune cell counts compared to healthy controls but the magnitude of the immune infiltration was significantly lower than in microscopic and ulcerative colitis. Further analyses concluded that immune activation in IBS patients was characterized by increased CD3+, CD4+, CD8+ T cells and mast cells numbers. Mast cells were significantly increased in female IBS patients, whereas CD3+ and CD8+ T cells were decreased in male patients[213]. It is noteworthy that mast cells are involved in several diseases, among which, some are frequently associated with IBS and affect women particularly (painful bladder syndrome, chronic fatigue syndrome, and fibromyalgia)[214]. As described previously, mast cell degranulation is inhibited by progesterone and stimulated by estradiol[210,211]. An imbalance in ovarian hormone levels in IBS patients could, consequently, explain the mast-cell-mediated immune activation while increased T-cell-mediated activation in men could be due to testosterone modulation[212]. However, further studies are needed to elucidate the mechanisms of interaction between sex hormones and immune cells and to distinguish the differential effects of sex hormones. In this regard, studies assessing changes in GI immune activation along the menstrual cycle in correlation with ovarian hormones levels are warranted.
Key points: Recent findings suggest associations between immune activation and IBS, and especially, GI mucosal mast cell infiltration is consistently reported in IBS patients; estrogens promote mast cell activation; the only study assessing the effect of gender on mast cell infiltration in IBS found a specific increase of mucosal mast cell counts in the colon of female IBS patients.
Sex ratio in IBS is highly skewed towards female gender. This suggests that sex hormones play a key role in IBS physiopathology as suggested by some findings reporting fluctuations in IBS symptoms during the menstrual cycle, especially exacerbation of abdominal pain at menses (i.e., when ovarian hormones levels are low). However, the mechanisms underlying these gender-related differences are unclear. Clinical studies in the field remain scarce and the data obtained in humans are still weak and sometimes conflicting with findings from preclinical models. Moreover, our understanding is hampered by the fact that differences seen between male and female IBS patients are multifactorial and result from intrinsic differences in male and female hormonal systems, stress reactivity and emotionality, nociceptive system and inflammatory response, as well as sociocultural differences.
Nonetheless, the currently available data provide some insights into the potential mechanisms at play (Figure 1). In female IBS patients, estrogens seem to have a modulatory effect on GI pain by slowing down intestinal motor activity via mechanisms that likely involve the 5HT system. Interactions between the 5HT system and ovarian hormones have also been involved in differential peripheral pain processing. Moreover, in line with the frequent history of stress and adverse life events in female IBS patients, altered responsiveness of the HPA axis to stress and abnormal CRH signaling have been pinpointed as major factors responsible for IBS prevalence in women. It is not completely clear whether alterations of the emotional motor system[215] are a cause or consequence of the altered patterns of activity of the limbic system seen in female IBS patients, who display hyperactivity of emotional and attentional circuits (especially the amygdala, a key structure in the processing of aversive experiences). Reciprocal positive feedback in these systems could explain ongoing hyper-reactivity to stress in patients. Indeed, it has been shown in animal models of IBS that stress and hormones can have a synergistic action and result in changes in the periphery leading to IBS-like pathological features such as increased intestinal barrier vulnerability in response to stress or increased intestinal mast cell activation and infiltration.
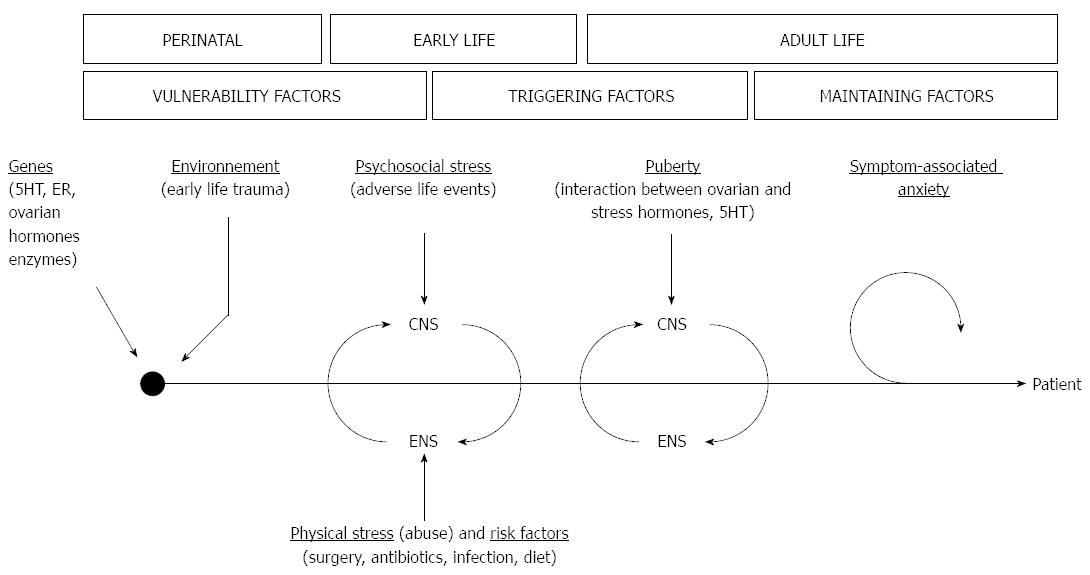
Given the diversity of pathophysiological mechanisms known at currently, it is essential to develop a disease model integrating the multifactorial aspects of gender-related differences in IBS. Such a model could be helpful to identify research avenues leading to a better understanding of IBS etiology. An integrative multiple hits model can thus be proposed to account for the numerous IBS risk factors, their possible interplay, and their time scale in the course of IBS development (Figure 2).
In this model, gender-based genetic differences (e.g., polymorphisms in 5HT genes) confer a vulnerability to women to risk factors such as stress. Subsequent early life adverse events during childhood contribute to develop a hyper-reactive stress system, with hyper-responsiveness of HPA and emotional brain circuits, leading to heightened pain perception and cognition. In these predisposed patients, after puberty, ovarian hormones could act as triggering factors to sensitize further this priming, and ultimately contribute to the adult onset of IBS. Several mechanisms can be put forward to explain the transition to disease. Ovarian hormones may have synergistic actions with stress mediators and receptors to deleteriously impact nociceptive processes (i.e., interaction with key neuromodulator systems such as 5HT and CRH, and changes in neuroplasticity) and gut function (i.e., GI immune system, gut permeability and sensori-motricity). Interestingly, one can also integrate other IBS risk factors in this scenario, and diet, GI infection or dysbiosis can all be considered as possible additional “hits” leading to IBS development. Finally, symptom-related anxiety and lifestyle can explain the lasting and intermittent course of IBS in the long run.
This review highlights the complexity of the multiple actions of ovarian hormones in IBS and the key role played by stress. Consequently, it appears crucial to understand better the brain-gut interactions and how ovarian hormones modulate them. Animal models can be highly valuable to this end and could shed light on the role of genes and environment in gender-related differences in IBS. Another element stressed by the data reported herein is the scarcity of information regarding sex hormones involvement in patients. There is a need for powerful longitudinal studies taking into account phases of cycles and correlating symptoms and ovarian hormones levels. These studies should also use pair-matched cohorts and factor IBS subtypes in their analysis to minimize methodological confounders. Finally, assessing the role of male sex hormones has remained an unexplored research avenue even though such studies could prove very insightful.
| 1. | Choung RS, Locke GR. Epidemiology of IBS. Gastroenterol Clin North Am. 2011;40:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 118] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 2. | Sandler RS, Everhart JE, Donowitz M, Adams E, Cronin K, Goodman C, Gemmen E, Shah S, Avdic A, Rubin R. The burden of selected digestive diseases in the United States. Gastroenterology. 2002;122:1500-1511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1186] [Cited by in RCA: 1020] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 3. | Azpiroz F, Bouin M, Camilleri M, Mayer EA, Poitras P, Serra J, Spiller RC. Mechanisms of hypersensitivity in IBS and functional disorders. Neurogastroenterol Motil. 2007;19:62-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 267] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 4. | Camilleri M, Lasch K, Zhou W. Irritable bowel syndrome: methods, mechanisms, and pathophysiology. The confluence of increased permeability, inflammation, and pain in irritable bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2012;303:G775-G785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 282] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 5. | Matricon J, Meleine M, Gelot A, Piche T, Dapoigny M, Muller E, Ardid D. Review article: Associations between immune activation, intestinal permeability and the irritable bowel syndrome. Aliment Pharmacol Ther. 2012;36:1009-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 155] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 6. | Chial HJ, Camilleri M. Gender differences in irritable bowel syndrome. J Gend Specif Med. 2002;5:37-45. [PubMed] |
| 7. | McCarberg BH. Clinical overview of fibromyalgia. Am J Ther. 2012;19:357-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 8. | Lipton RB, Stewart WF, Scher AI. Epidemiology and economic impact of migraine. Curr Med Res Opin. 2001;17 Suppl 1:s4-12. [PubMed] |
| 9. | Flier SN, Rose S. Is functional dyspepsia of particular concern in women? A review of gender differences in epidemiology, pathophysiologic mechanisms, clinical presentation, and management. Am J Gastroenterol. 2006;101:S644-S653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Walker EA, Gelfand AN, Gelfand MD, Green C, Katon WJ. Chronic pelvic pain and gynecological symptoms in women with irritable bowel syndrome. J Psychosom Obstet Gynaecol. 1996;17:39-46. [PubMed] |
| 11. | Dinos S, Khoshaba B, Ashby D, White PD, Nazroo J, Wessely S, Bhui KS. A systematic review of chronic fatigue, its syndromes and ethnicity: prevalence, severity, co-morbidity and coping. Int J Epidemiol. 2009;38:1554-1570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 87] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 12. | Grigoriadis S, Robinson GE. Gender issues in depression. Ann Clin Psychiatry. 2007;19:247-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 175] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 14. | Hinds JP, Stoney B, Wald A. Does gender or the menstrual cycle affect colonic transit? Am J Gastroenterol. 1989;84:123-126. [PubMed] |
| 15. | Laessle RG, Tuschl RJ, Schweiger U, Pirke KM. Mood changes and physical complaints during the normal menstrual cycle in healthy young women. Psychoneuroendocrinology. 1990;15:131-138. [PubMed] |
| 16. | Heitkemper MM, Jarrett M. Pattern of gastrointestinal and somatic symptoms across the menstrual cycle. Gastroenterology. 1992;102:505-513. [PubMed] |
| 17. | Moore J, Barlow D, Jewell D, Kennedy S. Do gastrointestinal symptoms vary with the menstrual cycle? Br J Obstet Gynaecol. 1998;105:1322-1325. [PubMed] |
| 18. | Palomba S, Di Cello A, Riccio E, Manguso F, La Sala GB. Ovarian function and gastrointestinal motor activity. Minerva Endocrinol. 2011;36:295-310. [PubMed] |
| 19. | Altman G, Cain KC, Motzer S, Jarrett M, Burr R, Heitkemper M. Increased symptoms in female IBS patients with dysmenorrhea and PMS. Gastroenterol Nurs. 2006;29:4-11. [PubMed] |
| 20. | Heitkemper MM, Chang L. Do fluctuations in ovarian hormones affect gastrointestinal symptoms in women with irritable bowel syndrome? Gend Med. 2009;6 Suppl 2:152-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 114] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 21. | Heitkemper MM, Jarrett M, Caudell KA, Bond E. Women with gastrointestinal symptoms: implications for nursing research and practice. Gastroenterol Nurs. 1993;15:226-232. [PubMed] |
| 22. | Jackson NA, Houghton LA, Whorwell PJ, Currer B. Does the menstrual cycle affect anorectal physiology? Dig Dis Sci. 1994;39:2607-2611. [PubMed] |
| 23. | Heitkemper MM, Cain KC, Jarrett ME, Burr RL, Crowell MD, Woods NF. Relationship of bloating to other GI and menstrual symptoms in women with irritable bowel syndrome. Dig Dis Sci. 2004;49:88-95. [PubMed] |
| 24. | Okoro NI, Kane SV. Gender-related issues in the female inflammatory bowel disease patient. Expert Rev Gastroenterol Hepatol. 2009;3:145-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 25. | Houghton LA, Lea R, Jackson N, Whorwell PJ. The menstrual cycle affects rectal sensitivity in patients with irritable bowel syndrome but not healthy volunteers. Gut. 2002;50:471-474. [PubMed] |
| 26. | Sanoja R, Cervero F. Estrogen-dependent changes in visceral afferent sensitivity. Auton Neurosci. 2010;153:84-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Teff KL, Alavi A, Chen J, Pourdehnad M, Townsend RR. Muscarinic blockade inhibits gastric emptying of mixed-nutrient meal: effects of weight and gender. Am J Physiol. 1999;276:R707-R714. [PubMed] |
| 28. | Meier R, Beglinger C, Dederding JP, Meyer-Wyss B, Fumagalli M, Rowedder A, Turberg Y, Brignoli R. Influence of age, gender, hormonal status and smoking habits on colonic transit time. Neurogastroenterol Motil. 1995;7:235-238. [PubMed] |
| 29. | Wald A, Van Thiel DH, Hoechstetter L, Gavaler JS, Egler KM, Verm R, Scott L, Lester R. Gastrointestinal transit: the effect of the menstrual cycle. Gastroenterology. 1981;80:1497-1500. [PubMed] |
| 30. | Kamm MA, Farthing MJ, Lennard-Jones JE. Bowel function and transit rate during the menstrual cycle. Gut. 1989;30:605-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 66] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 31. | Bovo P, Paola Brunori M, di Francesco V, Frulloni L, Montesi G, Cavallini G. The menstrual cycle has no effect on gastrointestinal transit time. Evaluation by means of the lactulose H2 breath test. Ital J Gastroenterol. 1992;24:449-451. [PubMed] |
| 32. | Wald A, Van Thiel DH, Hoechstetter L, Gavaler JS, Egler KM, Verm R, Scott L, Lester R. Effect of pregnancy on gastrointestinal transit. Dig Dis Sci. 1982;27:1015-1018. [PubMed] |
| 33. | Baron TH, Ramirez B, Richter JE. Gastrointestinal motility disorders during pregnancy. Ann Intern Med. 1993;118:366-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 110] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 34. | Wald A. Constipation, diarrhea, and symptomatic hemorrhoids during pregnancy. Gastroenterol Clin North Am. 2003;32:309-22, vii. [PubMed] |
| 35. | Shah S, Hobbs A, Singh R, Cuevas J, Ignarro LJ, Chaudhuri G. Gastrointestinal motility during pregnancy: role of nitrergic component of NANC nerves. Am J Physiol Regul Integr Comp Physiol. 2000;279:R1478-R1485. [PubMed] |
| 36. | Shah S, Nathan L, Singh R, Fu YS, Chaudhuri G. E2 and not P4 increases NO release from NANC nerves of the gastrointestinal tract: implications in pregnancy. Am J Physiol Regul Integr Comp Physiol. 2001;280:R1546-R1554. [PubMed] |
| 37. | Ryan JP, Bhojwani A. Colonic transit in rats: effect of ovariectomy, sex steroid hormones, and pregnancy. Am J Physiol. 1986;251:G46-G50. [PubMed] |
| 38. | Hogan AM, Kennelly R, Collins D, Baird AW, Winter DC. Oestrogen inhibits human colonic motility by a non-genomic cell membrane receptor-dependent mechanism. Br J Surg. 2009;96:817-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 39. | Shimomura A, Ohama T, Hori M, Ozaki H. 17Beta-estradiol induces gastrointestinal motility disorder by decreasing CPI-17 phosphorylation via changes in rho-family G-protein Rnd expression in small intestine. J Vet Med Sci. 2009;71:1591-1597. [PubMed] |
| 40. | Xie D, Chen L, Liu C, Liu K. The inhibitory effects of oxytocin on distal colonic contractile activity in rabbits are enhanced by ovarian steroids. Acta Physiol (Oxf). 2006;186:141-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 41. | Xie DP, Chen LB, Liu CY, Liu JZ, Liu KJ. Effect of oxytocin on contraction of rabbit proximal colon in vitro. World J Gastroenterol. 2003;9:165-168. [PubMed] |
| 42. | Wu CL, Hung CR, Chang FY, Pau KY, Wang JL, Wang PS. Involvement of cholecystokinin receptor in the inhibition of gastric emptying by oxytocin in male rats. Pflugers Arch. 2002;445:187-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 43. | Ohlsson B, Truedsson M, Bengtsson M, Torstenson R, Sjölund K, Björnsson ES, Simrèn M. Effects of long-term treatment with oxytocin in chronic constipation; a double blind, placebo-controlled pilot trial. Neurogastroenterol Motil. 2005;17:697-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 44. | Feng M, Qin J, Wang C, Ye Y, Wang S, Xie D, Wang PS, Liu C. Estradiol upregulates the expression of oxytocin receptor in colon in rats. Am J Physiol Endocrinol Metab. 2009;296:E1059-E1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Gill RC, Bowes KL, Kingma YJ. Effect of progesterone on canine colonic smooth muscle. Gastroenterology. 1985;88:1941-1947. [PubMed] |
| 46. | Oh ST, Yedidag E, Bielefeldt K. Differential effects of progesterone and its analogues on the contractility of the murine jejunum in vitro. J Surg Res. 1998;75:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 47. | Liu CY, Chen LB, Liu PY, Xie DP, Wang PS. Effects of progesterone on gastric emptying and intestinal transit in male rats. World J Gastroenterol. 2002;8:338-341. [PubMed] |
| 48. | Xiao ZL, Pricolo V, Biancani P, Behar J. Role of progesterone signaling in the regulation of G-protein levels in female chronic constipation. Gastroenterology. 2005;128:667-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 60] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 49. | Li CP, Ling C, Biancani P, Behar J. Effect of progesterone on colonic motility and fecal output in mice with diarrhea. Neurogastroenterol Motil. 2012;24:392-e174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 50. | Gershon MD, Tack J. The serotonin signaling system: from basic understanding to drug development for functional GI disorders. Gastroenterology. 2007;132:397-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1279] [Cited by in RCA: 1142] [Article Influence: 60.1] [Reference Citation Analysis (0)] |
| 51. | Pecins-Thompson M, Brown NA, Bethea CL. Regulation of serotonin re-uptake transporter mRNA expression by ovarian steroids in rhesus macaques. Brain Res Mol Brain Res. 1998;53:120-129. [PubMed] |
| 52. | Gundlah C, Lu NZ, Bethea CL. Ovarian steroid regulation of monoamine oxidase-A and -B mRNAs in the macaque dorsal raphe and hypothalamic nuclei. Psychopharmacology (Berl). 2002;160:271-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 133] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 53. | Smith LJ, Henderson JA, Abell CW, Bethea CL. Effects of ovarian steroids and raloxifene on proteins that synthesize, transport, and degrade serotonin in the raphe region of macaques. Neuropsychopharmacology. 2004;29:2035-2045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 120] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 54. | Cheng L, Pricolo V, Biancani P, Behar J. Overexpression of progesterone receptor B increases sensitivity of human colon muscle cells to progesterone. Am J Physiol Gastrointest Liver Physiol. 2008;295:G493-G502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 55. | Guarino M, Cheng L, Cicala M, Ripetti V, Biancani P, Behar J. Progesterone receptors and serotonin levels in colon epithelial cells from females with slow transit constipation. Neurogastroenterol Motil. 2011;23:575-e210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 56. | Adeyemo MA, Spiegel BM, Chang L. Meta-analysis: do irritable bowel syndrome symptoms vary between men and women? Aliment Pharmacol Ther. 2010;32:738-755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 143] [Cited by in RCA: 141] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 57. | Herman J, Pokkunuri V, Braham L, Pimentel M. Gender distribution in irritable bowel syndrome is proportional to the severity of constipation relative to diarrhea. Gend Med. 2010;7:240-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 58. | Bearcroft CP, Perrett D, Farthing MJ. Postprandial plasma 5-hydroxytryptamine in diarrhoea predominant irritable bowel syndrome: a pilot study. Gut. 1998;42:42-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 223] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 59. | Houghton LA, Atkinson W, Whitaker RP, Whorwell PJ, Rimmer MJ. Increased platelet depleted plasma 5-hydroxytryptamine concentration following meal ingestion in symptomatic female subjects with diarrhoea predominant irritable bowel syndrome. Gut. 2003;52:663-670. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 143] [Cited by in RCA: 138] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 60. | Atkinson W, Lockhart S, Whorwell PJ, Keevil B, Houghton LA. Altered 5-hydroxytryptamine signaling in patients with constipation- and diarrhea-predominant irritable bowel syndrome. Gastroenterology. 2006;130:34-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 272] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 61. | Dunlop SP, Coleman NS, Blackshaw E, Perkins AC, Singh G, Marsden CA, Spiller RC. Abnormalities of 5-hydroxytryptamine metabolism in irritable bowel syndrome. Clin Gastroenterol Hepatol. 2005;3:349-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 269] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 62. | Spiller R, Aziz Q, Creed F, Emmanuel A, Houghton L, Hungin P, Jones R, Kumar D, Rubin G, Trudgill N. Guidelines on the irritable bowel syndrome: mechanisms and practical management. Gut. 2007;56:1770-1798. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 625] [Cited by in RCA: 533] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 63. | Houghton LA, Brown H, Atkinson W, Morris J, Fell C, Whorwell PJ, Lockhart S, Keevil B. 5-hydroxytryptamine signalling in irritable bowel syndrome with diarrhoea: effects of gender and menstrual status. Aliment Pharmacol Ther. 2009;30:919-929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 64. | Fillingim RB, Ness TJ. Sex-related hormonal influences on pain and analgesic responses. Neurosci Biobehav Rev. 2000;24:485-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 386] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 65. | Fillingim RB, King CD, Ribeiro-Dasilva MC, Rahim-Williams B, Riley JL. Sex, gender, and pain: a review of recent clinical and experimental findings. J Pain. 2009;10:447-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1549] [Reference Citation Analysis (2)] |
| 66. | Ragnarsson G, Hallböök O, Bodemar G. Abdominal symptoms are not related to anorectal function in the irritable bowel syndrome. Scand J Gastroenterol. 1999;34:250-258. [PubMed] |
| 67. | Chang L, Mayer EA, Labus JS, Schmulson M, Lee OY, Olivas TI, Stains J, Naliboff BD. Effect of sex on perception of rectosigmoid stimuli in irritable bowel syndrome. Am J Physiol Regul Integr Comp Physiol. 2006;291:R277-R284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 82] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 68. | Holdcroft A, Sapsed-Byrne S, Ma D, Hammal D, Forsling ML. Sex and oestrous cycle differences in visceromotor responses and vasopressin release in response to colonic distension in male and female rats anaesthetized with halothane. Br J Anaesth. 2000;85:907-910. [PubMed] |
| 69. | Kamp EH, Jones RC, Tillman SR, Gebhart GF. Quantitative assessment and characterization of visceral nociception and hyperalgesia in mice. Am J Physiol Gastrointest Liver Physiol. 2003;284:G434-G444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 82] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 70. | Ji Y, Murphy AZ, Traub RJ. Sex differences in morphine-induced analgesia of visceral pain are supraspinally and peripherally mediated. Am J Physiol Regul Integr Comp Physiol. 2006;291:R307-R314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 58] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 71. | Ji Y, Tang B, Cao DY, Wang G, Traub RJ. Sex differences in spinal processing of transient and inflammatory colorectal stimuli in the rat. Pain. 2012;153:1965-1973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 72. | Chaloner A, Greenwood-Van Meerveld B. Sexually dimorphic effects of unpredictable early life adversity on visceral pain behavior in a rodent model. J Pain. 2013;14:270-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 73. | Giamberardino MA, Affaitati G, Valente R, Iezzi S, Vecchiet L. Changes in visceral pain reactivity as a function of estrous cycle in female rats with artificial ureteral calculosis. Brain Res. 1997;774:234-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 74. | Bradshaw HB, Temple JL, Wood E, Berkley KJ. Estrous variations in behavioral responses to vaginal and uterine distention in the rat. Pain. 1999;82:187-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 75. | Bradesi S, Eutamene H, Garcia-Villar R, Fioramonti J, Bueno L. Stress-induced visceral hypersensitivity in female rats is estrogen-dependent and involves tachykinin NK1 receptors. Pain. 2003;102:227-234. [PubMed] |
| 76. | Ji Y, Murphy AZ, Traub RJ. Estrogen modulates the visceromotor reflex and responses of spinal dorsal horn neurons to colorectal stimulation in the rat. J Neurosci. 2003;23:3908-3915. [PubMed] |
| 77. | Sanoja R, Cervero F. Estrogen-dependent abdominal hyperalgesia induced by ovariectomy in adult mice: a model of functional abdominal pain. Pain. 2005;118:243-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 78. | Lu CL, Hsieh JC, Tsaur ML, Huang YH, Wang PS, Wu LL, Liu PY, Chang FY, Lee SD. Estrogen rapidly modulates mustard oil-induced visceral hypersensitivity in conscious female rats: A role of CREB phosphorylation in spinal dorsal horn neurons. Am J Physiol Gastrointest Liver Physiol. 2007;292:G438-G446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 79. | Fan J, Yu LH, Zhang Y, Ni X, Ma B, Burnstock G. Estrogen altered visceromotor reflex and P2X(3) mRNA expression in a rat model of colitis. Steroids. 2009;74:956-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 80. | Myers B, Schulkin J, Greenwood-Van Meerveld B. Sex steroids localized to the amygdala increase pain responses to visceral stimulation in rats. J Pain. 2011;12:486-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 81. | Ji Y, Tang B, Traub RJ. The visceromotor response to colorectal distention fluctuates with the estrous cycle in rats. Neuroscience. 2008;154:1562-1567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 86] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 82. | Peng HY, Huang PC, Liao JM, Tung KC, Lee SD, Cheng CL, Shyu JC, Lai CY, Chen GD, Lin TB. Estrous cycle variation of TRPV1-mediated cross-organ sensitization between uterus and NMDA-dependent pelvic-urethra reflex activity. Am J Physiol Endocrinol Metab. 2008;295:E559-E568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (2)] |
| 83. | Ji Y, Tang B, Traub RJ. Modulatory effects of estrogen and progesterone on colorectal hyperalgesia in the rat. Pain. 2005;117:433-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 54] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 84. | Tang B, Ji Y, Traub RJ. Estrogen alters spinal NMDA receptor activity via a PKA signaling pathway in a visceral pain model in the rat. Pain. 2008;137:540-549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 85. | Robbins MT, Mebane H, Ball CL, Shaffer AD, Ness TJ. Effect of estrogen on bladder nociception in rats. J Urol. 2010;183:1201-1205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 86. | Peng HY, Chen GD, Lai CY, Hsieh MC, Hsu HH, Wu HC, Lin TB. PI3K modulates estrogen-dependent facilitation of colon-to-urethra cross-organ reflex sensitization in ovariectomized female rats. J Neurochem. 2010;113:54-66. [RCA] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 87. | Ji Y, Tang B, Traub RJ. Spinal estrogen receptor alpha mediates estradiol-induced pronociception in a visceral pain model in the rat. Pain. 2011;152:1182-1191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 62] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 88. | Aloisi AM, Affaitati G, Ceccarelli I, Fiorenzani P, Lerza R, Rossi C, Pace MC, Chiefari M, Aurilio C, Giamberardino MA. Estradiol and testosterone differently affect visceral pain-related behavioural responses in male and female rats. Eur J Pain. 2010;14:602-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 89. | Papka RE, Storey-Workley M. Estrogen receptor-alpha and -beta coexist in a subpopulation of sensory neurons of female rat dorsal root ganglia. Neurosci Lett. 2002;319:71-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 56] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 90. | Lee DY, Chai YG, Lee EB, Kim KW, Nah SY, Oh TH, Rhim H. 17Beta-estradiol inhibits high-voltage-activated calcium channel currents in rat sensory neurons via a non-genomic mechanism. Life Sci. 2002;70:2047-2059. [PubMed] |
| 91. | Cho T, Chaban VV. Expression of P2X3 and TRPV1 receptors in primary sensory neurons from estrogen receptors-α and estrogen receptor-β knockout mice. Neuroreport. 2012;23:530-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 92. | Chaban VV, Mayer EA, Ennes HS, Micevych PE. Estradiol inhibits atp-induced intracellular calcium concentration increase in dorsal root ganglia neurons. Neuroscience. 2003;118:941-948. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 100] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 93. | Chaban VV, Micevych PE. Estrogen receptor-alpha mediates estradiol attenuation of ATP-induced Ca2+ signaling in mouse dorsal root ganglion neurons. J Neurosci Res. 2005;81:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 62] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 94. | Lu CL, Hsieh JC, Dun NJ, Oprea TI, Wang PS, Luo JC, Lin HC, Chang FY, Lee SD. Estrogen rapidly modulates 5-hydroxytrytophan-induced visceral hypersensitivity via GPR30 in rats. Gastroenterology. 2009;137:1040-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 95. | Cao DY, Ji Y, Tang B, Traub RJ. Estrogen receptor β activation is antinociceptive in a model of visceral pain in the rat. J Pain. 2012;13:685-694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 96. | McRoberts JA, Li J, Ennes HS, Mayer EA. Sex-dependent differences in the activity and modulation of N-methyl-d-aspartic acid receptors in rat dorsal root ganglia neurons. Neuroscience. 2007;148:1015-1020. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 65] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 97. | Chieng BC, Christie MJ, Osborne PB. Characterization of neurons in the rat central nucleus of the amygdala: cellular physiology, morphology, and opioid sensitivity. J Comp Neurol. 2006;497:910-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 103] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 98. | Jaferi A, Pickel VM. Mu-opioid and corticotropin-releasing-factor receptors show largely postsynaptic co-expression, and separate presynaptic distributions, in the mouse central amygdala and bed nucleus of the stria terminalis. Neuroscience. 2009;159:526-539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 99. | Beckerman MA, Glass MJ. The NMDA-NR1 receptor subunit and the mu-opioid receptor are expressed in somatodendritic compartments of central nucleus of the amygdala neurons projecting to the bed nucleus of the stria terminalis. Exp Neurol. 2012;234:112-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 100. | Sandner-Kiesling A, Eisenach JC. Estrogen reduces efficacy of mu- but Not kappa-opioid agonist inhibition in response to uterine cervical distension. Anesthesiology. 2002;96:375-379. [PubMed] |
| 101. | Tang YR, Yang WW, Wang YL, Lin L. Sex differences in the symptoms and psychological factors that influence quality of life in patients with irritable bowel syndrome. Eur J Gastroenterol Hepatol. 2012;24:702-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 102. | Whitehead WE, Cheskin LJ, Heller BR, Robinson JC, Crowell MD, Benjamin C, Schuster MM. Evidence for exacerbation of irritable bowel syndrome during menses. Gastroenterology. 1990;98:1485-1489. [PubMed] |
| 103. | Kane SV, Sable K, Hanauer SB. The menstrual cycle and its effect on inflammatory bowel disease and irritable bowel syndrome: a prevalence study. Am J Gastroenterol. 1998;93:1867-1872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 110] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 104. | Olafsdottir LB, Gudjonsson H, Jonsdottir HH, Björnsson E, Thjodleifsson B. Natural History of Irritable Bowel Syndrome in Women and Dysmenorrhea: A 10-Year Follow-Up Study. Gastroenterol Res Pract. 2012;2012:534204. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 105. | Palsson OS, Whitehead WE, Barghout V, Levy R, Feld A, Von Korff M, Garner M, Drossman DA, Turner MJ. IBS severity and health-related quality of life improve with age in women but not in men. Am J Gastroenterol. 2003;98:S272. [DOI] [Full Text] |
| 106. | Houghton LA, Jackson NA, Whorwell PJ, Morris J. Do male sex hormones protect from irritable bowel syndrome? Am J Gastroenterol. 2000;95:2296-2300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 107. | Camilleri M, Mayer EA, Drossman DA, Heath A, Dukes GE, McSorley D, Kong S, Mangel AW, Northcutt AR. Improvement in pain and bowel function in female irritable bowel patients with alosetron, a 5-HT3 receptor antagonist. Aliment Pharmacol Ther. 1999;13:1149-1159. [PubMed] |
| 108. | Berman SM, Naliboff BD, Chang L, Fitzgerald L, Antolin T, Camplone A, Mayer EA. Enhanced preattentive central nervous system reactivity in irritable bowel syndrome. Am J Gastroenterol. 2002;97:2791-2797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 50] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 109. | Kilpatrick LA, Ornitz E, Ibrahimovic H, Treanor M, Craske M, Nazarian M, Labus JS, Mayer EA, Naliboff BD. Sex-related differences in prepulse inhibition of startle in irritable bowel syndrome (IBS). Biol Psychol. 2010;84:272-278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 110. | Berkley KJ. Sex differences in pain. Behav Brain Sci. 1997;20:371-80; discussion 435-513. [PubMed] |
| 111. | Fillingim RB, Maixner W, Kincaid S, Silva S. Sex differences in temporal summation but not sensory-discriminative processing of thermal pain. Pain. 1998;75:121-127. [PubMed] |
| 112. | Papka RE, Mowa CN. Estrogen receptors in the spinal cord, sensory ganglia, and pelvic autonomic ganglia. Int Rev Cytol. 2003;231:91-127. [PubMed] |
| 113. | Berman S, Munakata J, Naliboff BD, Chang L, Mandelkern M, Silverman D, Kovalik E, Mayer EA. Gender differences in regional brain response to visceral pressure in IBS patients. Eur J Pain. 2000;4:157-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 132] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 114. | Naliboff BD, Berman S, Chang L, Derbyshire SW, Suyenobu B, Vogt BA, Mandelkern M, Mayer EA. Sex-related differences in IBS patients: central processing of visceral stimuli. Gastroenterology. 2003;124:1738-1747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 221] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 115. | Wilder-Smith CH, Schindler D, Lovblad K, Redmond SM, Nirkko A. Brain functional magnetic resonance imaging of rectal pain and activation of endogenous inhibitory mechanisms in irritable bowel syndrome patient subgroups and healthy controls. Gut. 2004;53:1595-1601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 308] [Cited by in RCA: 302] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 116. | Berman SM, Naliboff BD, Suyenobu B, Labus JS, Stains J, Bueller JA, Ruby K, Mayer EA. Sex differences in regional brain response to aversive pelvic visceral stimuli. Am J Physiol Regul Integr Comp Physiol. 2006;291:R268-R276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 2.9] [Reference Citation Analysis (2)] |
| 117. | Labus JS, Naliboff BN, Fallon J, Berman SM, Suyenobu B, Bueller JA, Mandelkern M, Mayer EA. Sex differences in brain activity during aversive visceral stimulation and its expectation in patients with chronic abdominal pain: a network analysis. Neuroimage. 2008;41:1032-1043. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 108] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 118. | Tillisch K, Mayer EA, Labus JS. Quantitative meta-analysis identifies brain regions activated during rectal distension in irritable bowel syndrome. Gastroenterology. 2011;140:91-100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 361] [Cited by in RCA: 333] [Article Influence: 22.2] [Reference Citation Analysis (1)] |
| 119. | Wang Z, Bradesi S, Maarek JM, Lee K, Winchester WJ, Mayer EA, Holschneider DP. Regional brain activation in conscious, nonrestrained rats in response to noxious visceral stimulation. Pain. 2008;138:233-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 120. | Wang Z, Guo Y, Bradesi S, Labus JS, Maarek JM, Lee K, Winchester WJ, Mayer EA, Holschneider DP. Sex differences in functional brain activation during noxious visceral stimulation in rats. Pain. 2009;145:120-128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (7)] |
| 121. | Hong JY, Kilpatrick LA, Labus J, Gupta A, Jiang Z, Ashe-McNalley C, Stains J, Heendeniya N, Ebrat B, Smith S. Patients with chronic visceral pain show sex-related alterations in intrinsic oscillations of the resting brain. J Neurosci. 2013;33:11994-12002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 87] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 122. | Labus JS, Gupta A, Coveleskie K, Tillisch K, Kilpatrick L, Jarcho J, Feier N, Bueller J, Stains J, Smith S. Sex differences in emotion-related cognitive processes in irritable bowel syndrome and healthy control subjects. Pain. 2013;154:2088-2099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 123. | Blankstein U, Chen J, Diamant NE, Davis KD. Altered brain structure in irritable bowel syndrome: potential contributions of pre-existing and disease-driven factors. Gastroenterology. 2010;138:1783-1789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 190] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 124. | Seminowicz DA, Labus JS, Bueller JA, Tillisch K, Naliboff BD, Bushnell MC, Mayer EA. Regional gray matter density changes in brains of patients with irritable bowel syndrome. Gastroenterology. 2010;139:48-57.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 248] [Cited by in RCA: 233] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 125. | Jiang Z, Dinov ID, Labus J, Shi Y, Zamanyan A, Gupta A, Ashe-McNalley C, Hong JY, Tillisch K, Toga AW. Sex-related differences of cortical thickness in patients with chronic abdominal pain. PLoS One. 2013;8:e73932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 126. | Ellingson BM, Mayer E, Harris RJ, Ashe-McNally C, Naliboff BD, Labus JS, Tillisch K. Diffusion tensor imaging detects microstructural reorganization in the brain associated with chronic irritable bowel syndrome. Pain. 2013;154:1528-1541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 127. | Labus JS, Dinov ID, Jiang Z, Ashe-McNalley C, Zamanyan A, Shi Y, Hong JY, Gupta A, Tillisch K, Ebrat B. Irritable bowel syndrome in female patients is associated with alterations in structural brain networks. Pain. 2014;155:137-149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 128] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 128. | Fukudo S, Kanazawa M, Mizuno T, Hamaguchi T, Kano M, Watanabe S, Sagami Y, Shoji T, Endo Y, Hongo M. Impact of serotonin transporter gene polymorphism on brain activation by colorectal distention. Neuroimage. 2009;47:946-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 57] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 129. | Kilpatrick LA, Labus JS, Coveleskie K, Hammer C, Rappold G, Tillisch K, Bueller JA, Suyenobu B, Jarcho JM, McRoberts JA. The HTR3A polymorphism c. -42C>T is associated with amygdala responsiveness in patients with irritable bowel syndrome. Gastroenterology. 2011;140:1943-1951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 70] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 130. | Aloisi AM, Bonifazi M. Sex hormones, central nervous system and pain. Horm Behav. 2006;50:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 161] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 131. | Cooke BM, Woolley CS. Gonadal hormone modulation of dendrites in the mammalian CNS. J Neurobiol. 2005;64:34-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 197] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 132. | Mukai H, Kimoto T, Hojo Y, Kawato S, Murakami G, Higo S, Hatanaka Y, Ogiue-Ikeda M. Modulation of synaptic plasticity by brain estrogen in the hippocampus. Biochim Biophys Acta. 2010;1800:1030-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 121] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 133. | Kramár EA, Babayan AH, Gall CM, Lynch G. Estrogen promotes learning-related plasticity by modifying the synaptic cytoskeleton. Neuroscience. 2013;239:3-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 134. | Seeman MV. Psychopathology in women and men: focus on female hormones. Am J Psychiatry. 1997;154:1641-1647. [PubMed] |
| 135. | Young E, Korszun A. Sex, trauma, stress hormones and depression. Mol Psychiatry. 2010;15:23-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 177] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 136. | Altemus M. Sex differences in depression and anxiety disorders: potential biological determinants. Horm Behav. 2006;50:534-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 220] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 137. | Bradford K, Shih W, Videlock EJ, Presson AP, Naliboff BD, Mayer EA, Chang L. Association between early adverse life events and irritable bowel syndrome. Clin Gastroenterol Hepatol. 2012;10:385-90.e1-3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 264] [Cited by in RCA: 232] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 138. | Bennett E, Tennant C, Piesse C, Badcock C, Kellow J. Level of chronic life stress predicts clinical outcome in irritable bowel syndrome. Gut. 1998;43:256. |
| 139. | Mayer EA, Naliboff BD, Chang L, Coutinho SV. V. Stress and irritable bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2001;280:G519-G524. [PubMed] |
| 140. | Spence MJ, Moss-Morris R. The cognitive behavioural model of irritable bowel syndrome: a prospective investigation of patients with gastroenteritis. Gut. 2007;56:1066-1071. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 110] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 141. | Myers B, Greenwood-Van Meerveld B. Role of anxiety in the pathophysiology of irritable bowel syndrome: importance of the amygdala. Front Neurosci. 2009;3:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (2)] |
| 142. | Chaloner A, Greenwood-Van Meerveld B. Early life adversity as a risk factor for visceral pain in later life: importance of sex differences. Front Neurosci. 2013;7:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 143. | Tillisch K, Mayer EA, Labus JS, Stains J, Chang L, Naliboff BD. Sex specific alterations in autonomic function among patients with irritable bowel syndrome. Gut. 2005;54:1396-1401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 114] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 144. | Kudielka BM, Kirschbaum C. Sex differences in HPA axis responses to stress: a review. Biol Psychol. 2005;69:113-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1004] [Cited by in RCA: 1155] [Article Influence: 52.5] [Reference Citation Analysis (0)] |
| 145. | Videlock EJ, Adeyemo M, Licudine A, Hirano M, Ohning G, Mayer M, Mayer EA, Chang L. Childhood trauma is associated with hypothalamic-pituitary-adrenal axis responsiveness in irritable bowel syndrome. Gastroenterology. 2009;137:1954-1962. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 155] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 146. | Young EA. Sex differences in response to exogenous corticosterone: a rat model of hypercortisolemia. Mol Psychiatry. 1996;1:313-319. [PubMed] |
| 147. | Smith RF, Ghuman SP, Evans NP, Karsch FJ, Dobson H. Stress and the control of LH secretion in the ewe. Reprod Suppl. 2003;61:267-282. [PubMed] |
| 148. | Ochedalski T, Subburaju S, Wynn PC, Aguilera G. Interaction between oestrogen and oxytocin on hypothalamic-pituitary-adrenal axis activity. J Neuroendocrinol. 2007;19:189-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 101] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 149. | Logrip ML, Rivier C, Lau C, Im S, Vaughan J, Lee S. Adolescent alcohol exposure alters the rat adult hypothalamic-pituitary-adrenal axis responsiveness in a sex-specific manner. Neuroscience. 2013;235:174-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 150. | Handa RJ, Burgess LH, Kerr JE, O'Keefe JA. Gonadal steroid hormone receptors and sex differences in the hypothalamo-pituitary-adrenal axis. Horm Behav. 1994;28:464-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 592] [Cited by in RCA: 588] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 151. | Nappi RE, Rivest S. Ovulatory cycle influences the stimulatory effect of stress on the expression of corticotropin-releasing factor receptor messenger ribonucleic acid in the paraventricular nucleus of the female rat hypothalamus. Endocrinology. 1995;136:4073-4083. [PubMed] |
| 152. | Nappi RE, Rivest S. Stress-induced genetic expression of a selective corticotropin-releasing factor-receptor subtype within the rat ovaries: an effect dependent on the ovulatory cycle. Biol Reprod. 1995;53:1417-1428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 153. | Suárez-Hitz KA, Otto B, Bidlingmaier M, Schwizer W, Fried M, Ehlert U. Altered psychobiological responsiveness in women with irritable bowel syndrome. Psychosom Med. 2012;74:221-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 154. | Elsenbruch S, Lovallo WR, Orr WC. Psychological and physiological responses to postprandial mental stress in women with the irritable bowel syndrome. Psychosom Med. 2001;63:805-813. [PubMed] |
| 155. | Salmon P, Skaife K, Rhodes J. Abuse, dissociation, and somatization in irritable bowel syndrome: towards an explanatory model. J Behav Med. 2003;26:1-18. [PubMed] |
| 156. | Creed F, Tomenson B, Guthrie E, Ratcliffe J, Fernandes L, Read N, Palmer S, Thompson DG. The relationship between somatisation and outcome in patients with severe irritable bowel syndrome. J Psychosom Res. 2008;64:613-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 157. | Ladd CO, Huot RL, Thrivikraman KV, Nemeroff CB, Meaney MJ, Plotsky PM. Long-term behavioral and neuroendocrine adaptations to adverse early experience. Prog Brain Res. 2000;122:81-103. [PubMed] |
| 158. | Li TJ, Yu BP, Dong WG, Luo HS, Xu L, Li MQ. Ovarian hormone modulates 5-hydroxytryptamine 3 receptors mRNA expression in rat colon with restraint stress-induced bowel dysfunction. World J Gastroenterol. 2004;10:2723-2726. [PubMed] |
| 159. | Bradesi S, Eutamene H, Fioramonti J, Bueno L. Acute restraint stress activates functional NK1 receptor in the colon of female rats: involvement of steroids. Gut. 2002;50:349-354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 160. | Bond EF, Heitkemper MM, Bailey SL. Estrogen suppresses gastric motility response to thyrotropin-releasing hormone and stress in awake rats. Res Nurs Health. 1998;21:221-228. [PubMed] [DOI] [Full Text] |
| 161. | Taché Y, Martinez V, Wang L, Million M. CRF1 receptor signaling pathways are involved in stress-related alterations of colonic function and viscerosensitivity: implications for irritable bowel syndrome. Br J Pharmacol. 2004;141:1321-1330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 198] [Cited by in RCA: 176] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 162. | Taché Y, Million M, Nelson AG, Lamy C, Wang L. Role of corticotropin-releasing factor pathways in stress-related alterations of colonic motor function and viscerosensibility in female rodents. Gend Med. 2005;2:146-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 163. | Larauche M, Mulak A, Kim YS, Labus J, Million M, Taché Y. Visceral analgesia induced by acute and repeated water avoidance stress in rats: sex difference in opioid involvement. Neurogastroenterol Motil. 2012;24:1031-e547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 164. | Alonso C, Guilarte M, Vicario M, Ramos L, Ramadan Z, Antolín M, Martínez C, Rezzi S, Saperas E, Kochhar S. Maladaptive intestinal epithelial responses to life stress may predispose healthy women to gut mucosal inflammation. Gastroenterology. 2008;135:163-172.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 91] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 165. | Alonso C, Guilarte M, Vicario M, Ramos L, Rezzi S, Martínez C, Lobo B, Martin FP, Pigrau M, González-Castro AM. Acute experimental stress evokes a differential gender-determined increase in human intestinal macromolecular permeability. Neurogastroenterol Motil. 2012;24:740-76, e348-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 166. | Meddings JB, Swain MG. Environmental stress-induced gastrointestinal permeability is mediated by endogenous glucocorticoids in the rat. Gastroenterology. 2000;119:1019-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 187] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 167. | Santos J, Perdue MH. Stress and neuroimmune regulation of gut mucosal function. Gut. 2000;47 Suppl 4:iv49-i51; discussion iv52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 168. | Söderholm JD, Perdue MH. Stress and gastrointestinal tract. II. Stress and intestinal barrier function. Am J Physiol Gastrointest Liver Physiol. 2001;280:G7-G13. [PubMed] |
| 169. | Ferrier L, Mazelin L, Cenac N, Desreumaux P, Janin A, Emilie D, Colombel JF, Garcia-Villar R, Fioramonti J, Bueno L. Stress-induced disruption of colonic epithelial barrier: role of interferon-gamma and myosin light chain kinase in mice. Gastroenterology. 2003;125:795-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 140] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 170. | Barreau F, Cartier C, Leveque M, Ferrier L, Moriez R, Laroute V, Rosztoczy A, Fioramonti J, Bueno L. Pathways involved in gut mucosal barrier dysfunction induced in adult rats by maternal deprivation: corticotrophin-releasing factor and nerve growth factor interplay. J Physiol. 2007;580:347-56. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 111] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 171. | Santos J, Yang PC, Söderholm JD, Benjamin M, Perdue MH. Role of mast cells in chronic stress induced colonic epithelial barrier dysfunction in the rat. Gut. 2001;48:630-636. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 236] [Cited by in RCA: 225] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 172. | Demaude J, Salvador-Cartier C, Fioramonti J, Ferrier L, Bueno L. Phenotypic changes in colonocytes following acute stress or activation of mast cells in mice: implications for delayed epithelial barrier dysfunction. Gut. 2006;55:655-661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 110] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 173. | Enmark E, Pelto-Huikko M, Grandien K, Lagercrantz S, Lagercrantz J, Fried G, Nordenskjöld M, Gustafsson JA. Human estrogen receptor beta-gene structure, chromosomal localization, and expression pattern. J Clin Endocrinol Metab. 1997;82:4258-4265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 279] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 174. | Campbell-Thompson M, Reyher KK, Wilkinson LB. Immunolocalization of estrogen receptor alpha and beta in gastric epithelium and enteric neurons. J Endocrinol. 2001;171:65-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 175. | Konstantinopoulos P, Kominea A, Vandoros G, Sykiotis G. Andricopoulos P, Varakis I, Sotiropoulou-Bonikou G, Papavassiliou A. Oestrogen receptor beta (ERβ) is abundantly expressed in normal colonic mucosa, but declines in colon adenocarcinoma paralleling the tumour’s dedifferentiation. Eur J Cancer. 2003;39:1251-1258. [RCA] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 247] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 176. | Kawano N, Koji T, Hishikawa Y, Murase K, Murata I, Kohno S. Identification and localization of estrogen receptor alpha- and beta-positive cells in adult male and female mouse intestine at various estrogen levels. Histochem Cell Biol. 2004;121:399-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 177. | Wada-Hiraike O, Warner M, Gustafsson JA. New developments in oestrogen signalling in colonic epithelium. Biochem Soc Trans. 2006;34:1114-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 178. | Enmark E, Gustafsson JA. Oestrogen receptors - an overview. J Intern Med. 1999;246:133-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 224] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 179. | Wada-Hiraike O, Imamov O, Hiraike H, Hultenby K, Schwend T, Omoto Y, Warner M, Gustafsson JA. Role of estrogen receptor beta in colonic epithelium. Proc Natl Acad Sci USA. 2006;103:2959-2964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 201] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 180. | Condliffe SB, Doolan CM, Harvey BJ. 17beta-oestradiol acutely regulates Cl- secretion in rat distal colonic epithelium. J Physiol. 2001;530:47-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 73] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 181. | Cotter AA, Cashman KD. Effect of 17beta-oestradiol on transepithelial calcium transport in human intestinal-like Caco-2 cells and its interactions with 1,25-dihydroxycholecalciferol and 9-cis retinoic acid. Eur J Nutr. 2006;45:234-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 182. | Pirani T, Chen J, Vieira A. Effects of estradiol on the endocytic transport of vitamin D carrier protein in hepatocytes. Biochim Biophys Acta. 2013;1830:3421-3426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 183. | Boyko EJ, Theis MK, Vaughan TL, Nicol-Blades B. Increased risk of inflammatory bowel disease associated with oral contraceptive use. Am J Epidemiol. 1994;140:268-278. [PubMed] |
| 184. | Cornish JA, Tan E, Simillis C, Clark SK, Teare J, Tekkis PP. The risk of oral contraceptives in the etiology of inflammatory bowel disease: a meta-analysis. Am J Gastroenterol. 2008;103:2394-2400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 185] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 185. | Kane SV, Reddy D. Hormonal replacement therapy after menopause is protective of disease activity in women with inflammatory bowel disease. Am J Gastroenterol. 2008;103:1193-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 97] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 186. | Homma H, Hoy E, Xu DZ, Lu Q, Feinman R, Deitch EA. The female intestine is more resistant than the male intestine to gut injury and inflammation when subjected to conditions associated with shock states. Am J Physiol Gastrointest Liver Physiol. 2005;288:G466-G472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 103] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 187. | Braniste V, Leveque M, Buisson-Brenac C, Bueno L, Fioramonti J, Houdeau E. Oestradiol decreases colonic permeability through oestrogen receptor beta-mediated up-regulation of occludin and junctional adhesion molecule-A in epithelial cells. J Physiol. 2009;587:3317-3328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 130] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 188. | Braniste V, Jouault A, Gaultier E, Polizzi A, Buisson-Brenac C, Leveque M, Martin PG, Theodorou V, Fioramonti J, Houdeau E. Impact of oral bisphenol A at reference doses on intestinal barrier function and sex differences after perinatal exposure in rats. Proc Natl Acad Sci USA. 2010;107:448-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 136] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 189. | Looijer-van Langen M, Hotte N, Dieleman LA, Albert E, Mulder C, Madsen KL. Estrogen receptor-β signaling modulates epithelial barrier function. Am J Physiol Gastrointest Liver Physiol. 2011;300:G621-G626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 158] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 190. | Moussa L, Bézirard V, Salvador-Cartier C, Bacquié V, Houdeau E, Théodorou V. A new soy germ fermented ingredient displays estrogenic and protease inhibitor activities able to prevent irritable bowel syndrome-like symptoms in stressed female rats. Clin Nutr. 2013;32:51-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 191. | Zhou Q, Zhang B, Verne GN. Intestinal membrane permeability and hypersensitivity in the irritable bowel syndrome. Pain. 2009;146:41-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 304] [Cited by in RCA: 286] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 192. | Kerckhoffs AP, Akkermans LM, de Smet MB, Besselink MG, Hietbrink F, Bartelink IH, Busschers WB, Samsom M, Renooij W. Intestinal permeability in irritable bowel syndrome patients: effects of NSAIDs. Dig Dis Sci. 2010;55:716-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 52] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 193. | Sankaran-Walters S, Macal M, Grishina I, Nagy L, Goulart L, Coolidge K, Li J, Fenton A, Williams T, Miller MK. Sex differences matter in the gut: effect on mucosal immune activation and inflammation. Biol Sex Differ. 2013;4:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 121] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 194. | Beeson PB. Age and sex associations of 40 autoimmune diseases. Am J Med. 1994;96:457-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 241] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 195. | Hewagama A, Patel D, Yarlagadda S, Strickland FM, Richardson BC. Stronger inflammatory/cytotoxic T-cell response in women identified by microarray analysis. Genes Immun. 2009;10:509-516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 234] [Cited by in RCA: 235] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 196. | Kovats S. Estrogen receptors regulate an inflammatory pathway of dendritic cell differentiation: mechanisms and implications for immunity. Horm Behav. 2012;62:254-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 113] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 197. | Oertelt-Prigione S. The influence of sex and gender on the immune response. Autoimmun Rev. 2012;11:A479-A485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 379] [Cited by in RCA: 338] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 198. | Cutolo M, Sulli A, Straub RH. Estrogen metabolism and autoimmunity. Autoimmun Rev. 2012;11:A460-A464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 95] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 199. | Stygar D, Westlund P, Eriksson H, Sahlin L. Identification of wild type and variants of oestrogen receptors in polymorphonuclear and mononuclear leucocytes. Clin Endocrinol (Oxf). 2006;64:74-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 200. | Rider V, Li X, Peterson G, Dawson J, Kimler BF, Abdou NI. Differential expression of estrogen receptors in women with systemic lupus erythematosus. J Rheumatol. 2006;33:1093-1101. [PubMed] |
| 201. | Inui A, Ogasawara H, Naito T, Sekigawa I, Takasaki Y, Hayashida Y, Takamori K, Ogawa H. Estrogen receptor expression by peripheral blood mononuclear cells of patients with systemic lupus erythematosus. Clin Rheumatol. 2007;26:1675-1678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 202. | Cunningham M, Gilkeson G. Estrogen receptors in immunity and autoimmunity. Clin Rev Allergy Immunol. 2011;40:66-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 208] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 203. | Bonnelye E, Laurin N, Jurdic P, Hart DA, Aubin JE. Estrogen receptor-related receptor-alpha (ERR-alpha) is dysregulated in inflammatory arthritis. Rheumatology (Oxford). 2008;47:1785-1791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 204. | Chen FP, Wang KC, Huang JD. Effect of estrogen on the activity and growth of human osteoclasts in vitro. Taiwan J Obstet Gynecol. 2009;48:350-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 205. | Subramanian M, Shaha C. Oestrogen modulates human macrophage apoptosis via differential signalling through oestrogen receptor-alpha and beta. J Cell Mol Med. 2009;13:2317-2329. [PubMed] [DOI] [Full Text] |
| 206. | Giannoni E, Guignard L, Knaup Reymond M, Perreau M, Roth-Kleiner M, Calandra T, Roger T. Estradiol and progesterone strongly inhibit the innate immune response of mononuclear cells in newborns. Infect Immun. 2011;79:2690-2698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 101] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 207. | Egger B, Bajaj-Elliott M, MacDonald TT, Inglin R, Eysselein VE, Büchler MW. Characterisation of acute murine dextran sodium sulphate colitis: cytokine profile and dose dependency. Digestion. 2000;62:240-248. [PubMed] |
| 208. | Burger D, Dayer JM. Cytokines, acute-phase proteins, and hormones: IL-1 and TNF-alpha production in contact-mediated activation of monocytes by T lymphocytes. Ann N Y Acad Sci. 2002;966:464-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 108] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 209. | Priyanka HP, Krishnan HC, Singh RV, Hima L, Thyagarajan S. Estrogen modulates in vitro T cell responses in a concentration- and receptor-dependent manner: effects on intracellular molecular targets and antioxidant enzymes. Mol Immunol. 2013;56:328-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 210. | Vasiadi M, Kempuraj D, Boucher W, Kalogeromitros D, Theoharides TC. Progesterone inhibits mast cell secretion. Int J Immunopathol Pharmacol. 2006;19:787-794. [PubMed] |
| 211. | Vliagoftis H, Dimitriadou V, Boucher W, Rozniecki JJ, Correia I, Raam S, Theoharides TC. Estradiol augments while tamoxifen inhibits rat mast cell secretion. Int Arch Allergy Immunol. 1992;98:398-409. [PubMed] |
| 212. | Page ST, Plymate SR, Bremner WJ, Matsumoto AM, Hess DL, Lin DW, Amory JK, Nelson PS, Wu JD. Effect of medical castration on CD4+ CD25+ T cells, CD8+ T cell IFN-gamma expression, and NK cells: a physiological role for testosterone and/or its metabolites. Am J Physiol Endocrinol Metab. 2006;290:E856-E863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 130] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 213. | Cremon C, Gargano L, Morselli-Labate AM, Santini D, Cogliandro RF, De Giorgio R, Stanghellini V, Corinaldesi R, Barbara G. Mucosal immune activation in irritable bowel syndrome: gender-dependence and association with digestive symptoms. Am J Gastroenterol. 2009;104:392-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 268] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 214. | Theoharides TC, Cochrane DE. Critical role of mast cells in inflammatory diseases and the effect of acute stress. J Neuroimmunol. 2004;146:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 307] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 215. | Mayer EA, Berman S, Chang L, Naliboff BD. Sex-based differences in gastrointestinal pain. Eur J Pain. 2004;8:451-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 78] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
P- Reviewers: Krogsgaard LR, O’Malley D S- Editor: Qi Y L- Editor: Kerr C E- Editor: Ma S