Published online Apr 28, 2014. doi: 10.3748/wjg.v20.i16.4712
Revised: January 20, 2014
Accepted: March 4, 2014
Published online: April 28, 2014
Processing time: 173 Days and 20.1 Hours
AIM: To analyze the activation of sonic hedgehog (SHh) signaling pathways in a rat model of chronic pancreatitis.
METHODS: Forty Wistar rats were randomly divided into 2 groups: experimental group and control group (20 rats in each group). Dibutyltin dichloride was infused into the tail vein of the rats to induce chronic pancreatitis in the experimental group. The same volume of ethanol and glycerol mixture was infused in the control group. The expression of Ptch, Smo and Gli were analyzed using immunohistochemistry, and real-time reverse transcription polymerase chain reaction (RT-PCR).
RESULTS: Compared with the control group, significant histological changes in terms of the areas of abnormal architecture, glandular atrophy, fibrosis, pseudo tubular complexes, and edema were observed at week 4 in the experimental group. The expression of Ptch1, Smo and Gli1 in the pancreatic tissue increased significantly in the experimental group. Using RT-PCR, mRNA levels of Ptch, Smo and Gli in the experimental group increased significantly compared with the control group.
CONCLUSION: The SHh signaling pathway is aberrantly activated in rats with chronic pancreatitis. The SHh signaling pathway plays an important role in the development of chronic pancreatitis. These results may be helpful in studies focusing on the relationship between chronic pancreatitis and pancreatic cancer.
Core tip: Chronic pancreatitis (CP) is a progressive inflammation of the pancreas in which pancreatic secretory parenchyma is destroyed and replaced by fibrous tissue, eventually leading to impairment of both exocrine and endocrine functions. Hedgehog (Hh) signaling is a developmental signaling pathway that is highly activated in the embryo and in the early postnatal phase. Studies on the Hh signaling pathway in human CP are restricted by limited availability of tissues. Therefore, the present study was carried out to analyze the activation of SHh signaling pathways in a rat model of CP.
- Citation: Wang LW, Lin H, Lu Y, Xia W, Gao J, Li ZS. Sonic hedgehog expression in a rat model of chronic pancreatitis. World J Gastroenterol 2014; 20(16): 4712-4717
- URL: https://www.wjgnet.com/1007-9327/full/v20/i16/4712.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i16.4712
Core tip: Chronic pancreatitis (CP) is a progressive inflammation of the pancreas in which pancreatic secretory parenchyma is destroyed and replaced by fibrous tissue, eventually leading to impairment of both exocrine and endocrine functions. Hedgehog (Hh) signaling is a developmental signaling pathway that is highly activated in the embryo and in the early postnatal phase. Studies on the Hh signaling pathway in human CP are restricted by limited availability of tissues. Therefore, the present study was carried out to analyze the activation of SHh signaling pathways in a rat model of CP.
Chronic pancreatitis (CP) is a progressive inflammation of the pancreas in which pancreatic secretory parenchyma is destroyed and replaced by fibrous tissue, eventually leading to impairment of both exocrine and endocrine functions[1]. On histology, the main findings in this disease are acinar loss, mononuclear cell infiltration, and fibrosis. It is also reported that each inflammatory attack in the pancreas can cause fat necrosis that seems to lead to both pseudocysts and fibrosis[2]. Although CP has received increased attention over the past few years, the pathogenesis of CP has not yet been fully elucidated. Furthermore, patients with CP have a high risk of developing pancreatic cancer, and the molecular mechanism has also not been established[1].
Hedgehog (Hh) signaling is a developmental signaling pathway that is highly activated in the embryo and in the early postnatal phase. Three vertebrate Hh homologs have been identified, including Sonic hedgehog (SHh), Indian hedgehog (IHh), and Desert hedgehog. Of these Hh homologs, SHh has been the most studied in terms of the Hh signaling pathway in vertebrates[3]. Recently, aberrant activation of Hh signaling pathways has been described in various human neoplastic diseases[4-6]. Overexpression of the Hh ligands has also been reported in pancreatic adenocarcinoma, and inhibition of the Hh pathway at the level of Smo can result in blockage of cell proliferation and induction of apoptosis in many pancreatic cell lines in vitro. Kayed et al[7,8] analyzed the expression, distribution, and function of human hedgehog-interacting protein in normal pancreas, CP and pancreatic adenocarcinoma, and found that there was enhanced activation of hedgehog signaling in CP and pancreatic adenocarcinoma. However, studies on the Hh signaling pathway in human CP are restricted by limited availability of tissues. Therefore, the present study was carried out to analyze the activation of SHh signaling pathways in a rat model of CP.
This study was approved by the Institutional Animal Use and Care Committee of the Second Military Medical University, Shanghai, China.
Forty 6-wk-old male Wistar rats (180-200 g) were obtained from the Animal Center of the Second Military Medical University. Rats were provided with food and water ad libitum and housed in a temperature- and humidity-controlled room with a 12-h dark-light cycle (lights on at 7 AM) for 1 wk before the experiment.
CP was induced as previously described[9]. Briefly, dibutyltin dichloride (DBTC; Sigma-Aldrich, Chemie GmbH, Steinheim, Germany) was dissolved in 100% ethanol (Changshu Yangyuan Chemical Co China, Changshu, China) and mixed with glycerol (Amresco, OH, United States), with a volume ratio of 2:3 (ethanol:glycerol). The final DBTC concentration was 8 mg/mL. DBTC was then infused slowly into the tail vein of rats at a dose of 8 mg/kg body weight. Another 20 rats which acted as controls were infused with the same volume of ethanol and glycerol mixture. One day after the infusion, all rats received 0.6 mL soybean oil (Jinlongyu, China) by orogastric gavage once daily for 4 wk, and body weight was measured before and weekly after the gavage. The rats were killed by exsanguination under pentobarbital anesthesia (50 mg/kg, intraperitoneal) and the pancreas was removed for further analysis 4 wk after the gavage.
The head of the pancreas taken from each rat was fixed in 10% formalin and embedded in paraffin, and then 4-μm-thick sections were cut for histological examination, Sirius red staining, and immunohistochemistry. The caudal part of the pancreas was used for isolation of mRNA for real-time reverse transcription polymerase chain reaction (RT-PCR).
The pancreas sections were stained with hematoxylin and eosin for histological examination, and the histological changes were evaluated by an independent experienced pathologist, as previously described[9]. Briefly, the areas of abnormal architecture were classified as 0, absent; 1, rare; 2, minimal (< 10%); 3, moderate (10%-50%); and 4, severe (> 50%). Glandular atrophy, fibrosis, and pseudo tubular complexes within these areas were each scored as 0, absent; 1, minimal (< 10%); 2, moderate (10%-50%); and 3, severe (> 50%). In addition, the content of inflammatory cells (mainly neutrophils) and edema were also scored on a scale of 0-4. The sections were stained using a Sirius red stain kit (Genmed Inc, Shanghai, China) according to the manufacturer’s protocol for collagen deposition.
Immunostaining was performed to determine the expression of SHh proteins and to identify the activated SHh signaling pathway. The paraffin sections of pancreas were rehydrated and washed 3 times in phosphate buffered saline for 5 min. The sections were incubated with 0.3% H2O2 for 30 min to eliminate intrinsic peroxidase and then washed. The sections were probed with primary antibodies overnight at 4°C. The sections were incubated with goat polyclonal anti-Ptch1 (Patched1) antibody (Santa Cruz Biotechnology Inc, Santa Cruz, CA, United States) diluted to 1:400, goat polyclonal anti-Gli1 antibody (Santa Cruz Biotechnology Inc, Santa Cruz, CA, United States) diluted to 1:500, and rabbit polyclonal anti-Smo (Smoothened) antibody (Abcam, Cambridge, United Kingdom) diluted to 1:400, at room temperature for 3 h.
Total RNA was isolated from pancreatic tissues using TRIzol reagent (TaKaRa, Dalian, China), according to the manufacturer’s instructions. cDNA synthesis was formed using the real-time RT-PCR kit (TaKaRa), according to the manufacturer’s protocol. The specific primers for Ptch, Smo, Gli1 are listed in Table 1. The quantitation of target mRNA expression was performed on the ABI Model 7500 RT-PCR Sequence Detector (Applied Biosystems, Foster City, CA, United States) using a TaKaRa real-time PCR kit. The amplified PCR products were quantified by measuring the target and β-actin mRNA calculated cycle thresholds. The quality of specific mRNA in each sample was calculated from the standard curve and normalized with β-actin mRNA. Results were expressed as normalized values relative to the indicated cell line using the 2−ΔΔCt method.
| Genes | Forward (5'-3'), reverse (5'-3') |
| Ptch1 | TGGTCACACGAACAATGG |
| TGAACTGGGCAGCTATGAAGTC | |
| Smo | AGTTACATCGCAGCCTTC |
| CACACTACTCCAGCCATC | |
| Gli1 | TGCTGACACTCTGGGATA |
| CAGGGCCATAGTTGGTT |
Statistics was performed using SPSS 16.0 (SPSS Inc, Chicago, IL, United states). All data are expressed as mean ± SD. The Student’s t test was used for statistical analysis. The relationships between the variables were assessed using Spearman rank correlation coefficient. A value of P < 0.05 was considered statistically significant.
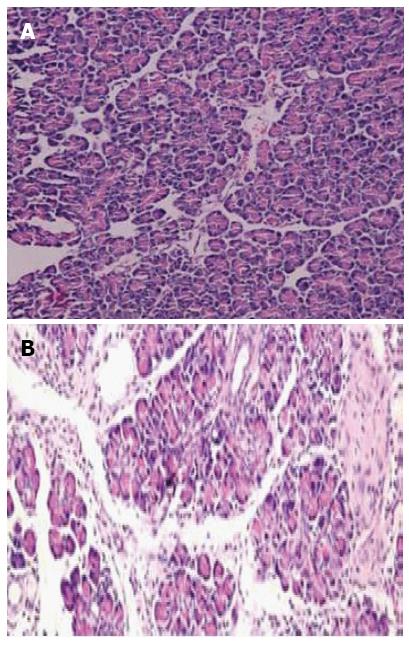
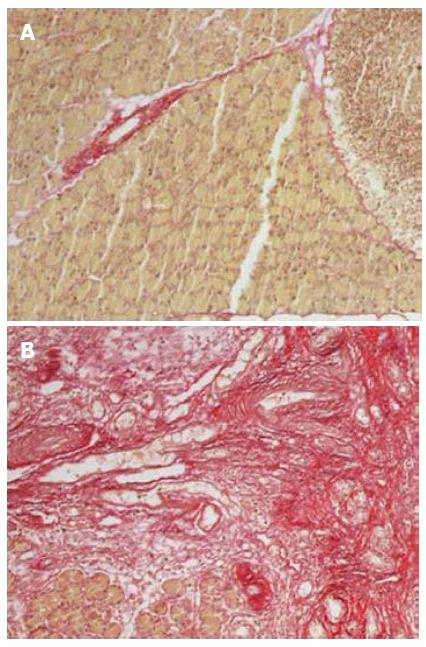
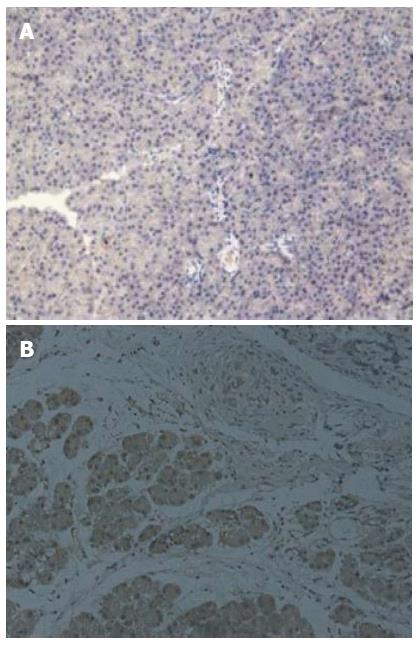
The survival rates of rats were 65% (13/20) and 95% (19/20) for the experimental group (DBTC treated group) and control group (ethanol and glycerol mixture treated group), respectively. There was no difference in body weight at baseline between the two groups, however, a significant reduction in body weight was observed in the experimental group at all time points (week 1, week 2, week 3 and week 4) compared with the controls (all P < 0.05). Histologic examination showed almost normal pancreas in the control rats (Figure 1A), however, dense inflammatory infiltrates, large regions of glandular atrophy, pseudo tubular complexes, edema, and fibrosis replaced the normal pancreas in DBTC treated rats (Figure 1B). Sirius red staining showed a significantly higher expression of lobular and sublobular collagen deposition in DBTC treated rats (P < 0.05) (Figure 2).
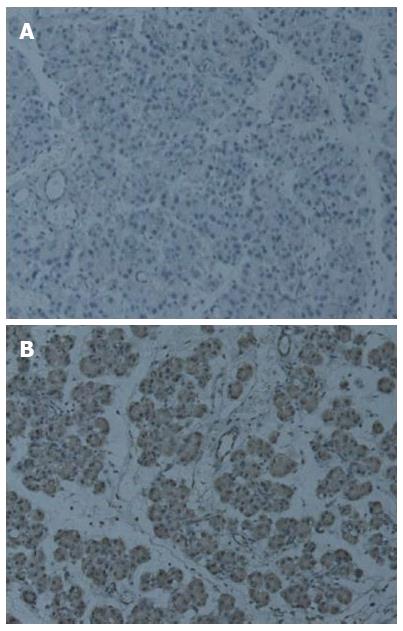
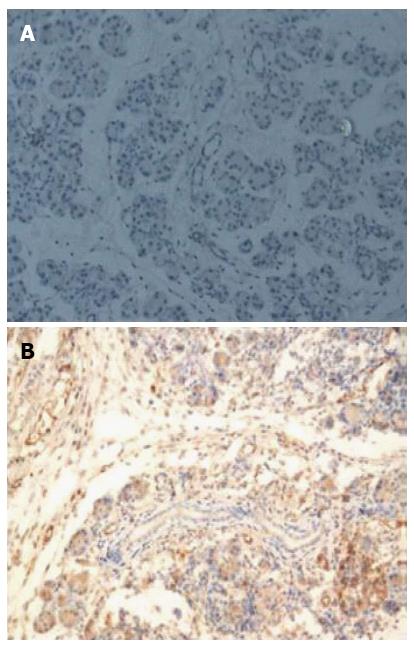
Immunohistochemical staining revealed the expression of Ptch1 in 10 of 13 (76.9%) CP rats (Table 2), while the expression of Ptch1 was negative in control rats. Ptch1 expression was mainly restricted to ductal, acinar, or islet compartments of the pancreas (Figure 3). Smo immunoreactivity was present in 9 (69.2%) rats in the experimental group, and no Smo positive cells were present in the control group. The location of Smo expression was mainly restricted to ductal, acinar, or islet compartments of the pancreas (Figure 4). Eight (61.5%) CP rats displayed strong Gli1 immunoreactivity, and no Gli1 immunoreactivity was observed in the control group. Gli1 expression was mainly restricted to ductal, acinar, or islet compartments of the pancreas (Figure 5).
| Rats | Ptch | Smo | Gli |
| Ethanol and glycerol mixture treated | 0.23 ± 0.16 | 0.14 ± 0.05 | 0.57 ± 0.12 |
| DBTC treated | 2.38 ± 0.42 | 3.85 ± 1.03 | 4.63 ± 1.49 |
These results were confirmed at the mRNA level using quantitative real-time RT-PCR. Compared with the controls, the mRNA expression of Ptch1, Smo, and Gli1 mRNA was significantly increased in rats treated with DBTC (Table 2).
Using immunohistochemistry, we found that SHh proteins are overexpressed in rats with DBTC-induced CP. This overexpression was confirmed by real-time RT-PCR. The SHh signaling pathway may play an important role in the development of CP.
Intercellular communication is essential for morphogenesis and patterning in vertebrates, and the Hh genes encode secreted proteins implicated in cell-cell interaction[10]. Two transmembrane receptors, Ptch1 and Ptch2, have been identified as receptors for processed Hh ligands, and Ptch1 expression is more prominent than Ptch2. In the absence of ligand, Ptch1 represses the activity of Smo. On ligand binding, the repression of Smo is alleviated, and Smo initiates a signaling cascade that results in the translocation of Gli transcription factors into the nucleus[11]. There are three known Gli transcription factors, Gli1, Gli2 and Gli3, in mammals, and Gli1 seems to act as a transcriptional activator and the Hh transcriptional target genes Should there be additional text here?[12].
Although progress has been made toward understanding Hh signaling recently, further research is necessary to clarify the complex nature of this signaling pathway. SHh is the best studied Hh with the broadest expression pattern in organs such as the developing nervous system, limb buds, skin and gut[11].
The expression of Hh signaling pathways is seen in the embryonic mouse pancreas, and is also observed in the adult mouse pancreas. Ptch and Smo are expressed in islet β cells. Activation of the Hh signaling pathway leads to increased insulin production, and this increase in insulin production can be prevented through inhibition of Hh signaling, indicating that Hh signaling in adult pancreatic islets acts mainly to regulate insulin production[13]. In the human pancreas, Ptch and Smo are localized in the islet cells similar to that in mouse pancreas, and weakly expressed in normal ductal cells[14].
Ptch, Smo, and Gli are reported to be overexpressed in human pancreatic cancer tissues and pancreatic cancer cell lines. SHh and its receptors are localized in malignant pancreatic ductal cells as well as in premalignant pancreatic lesions[7,15]. Specific inhibition of Hh activity in Ptch expressing pancreatic cancer cells can reduce pancreatic cancer cell growth in vitro. The inhibition of Hh activity can not only reduce the development of pancreatic tumors, but also the growth of already established tumors. In addition, binding of the Hh ligands can reduce Hh signaling activity in pancreatic cancer cells expressing Ptch[7,15-16].
It is reported that IHh and its receptors, Ptch and Smo, are overexpressed in human CP tissues and are localized in the ductal cells and tubular complexes. Compared to normal islets, the islet cells of CP tissues exhibit an abnormal localization pattern for Ihh[17]. Walter et al[18] established fibroblast cultures from human pancreatic adenocarcinomas and non-neoplastic pancreas tissues to identify differentially expressed genes in cancer-associated stromal fibroblasts. The results showed that Smo was upregulated in cancer-associated stromal fibroblasts, and the expressed Smo could transduce the SHh signal to activate Gli1 expression. Furthermore, small interfering RNA knockdown of Smo blocked the induction of Gli1 in these cells[18].
Due to the high risk of developing pancreatic cancer in CP patients, and the aberrant expression of Hh proteins, we examined the expression of SHh signaling pathways in a rat model of CP. The results showed that the receptors of SHh were overexpressed in CP. Given the role of Hh signaling in pancreatic cancer, these findings may provide a mechanism for the pathogenesis between CP and pancreatic cancer. The role of the microenvironment determining SHh expression and localization, and the role of SHh in CP require further investigation.
Chronic pancreatitis (CP) is a progressive inflammation of the pancreas, and patients with CP have a high risk of developing pancreatic cancer. Recently, aberrant activation of hedgehog (Hh) signaling pathways has been described in pancreatic cancer. Studies on the Hh signaling pathway in CP animal models are lacking.
It has been reported that human hedgehog-interacting protein was activated in CP and pancreatic adenocarcinoma, but little is known about the role of Hh in CP development and its role in the transformation from CP to pancreatic cancer.
Previous studies have reported that SHh and its receptors are localized in malignant pancreatic ductal cells as well as in premalignant pancreatic lesions. Specific inhibition of Hh activity in Ptch-expressing pancreatic cancer cells can reduce pancreatic cancer cell growth in vitro. In this study, they found that SHh proteins are overexpressed in rats with DBTC-induced CP, and this overexpression was confirmed by real-time reverse transcription polymerase chain reaction. The SHh signaling pathway may play an important role in the development of CP.
The study results suggest that the SHh signaling pathway may play an important role in the development of chronic pancreatitis, and these findings may provide a mechanism for the pathogenesis between chronic pancreatitis and pancreatic cancer.
The authors analyzed the expression of SHh and its related protein in a rat CP model, results showed SHh proteins are overexpressed. These findings can provide a new clue in the explanation of CP developing, even the mechanistic pathogenesis from chronic pancreatitis to pancreatic cancer.
| 1. | Braganza JM, Lee SH, McCloy RF, McMahon MJ. Chronic pancreatitis. Lancet. 2011;377:1184-1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 340] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 2. | Klöppel G. Chronic pancreatitis, pseudotumors and other tumor-like lesions. Mod Pathol. 2007;20 Suppl 1:S113-S131. [PubMed] |
| 3. | Martin J, Donnelly JM, Houghton J, Zavros Y. The role of sonic hedgehog reemergence during gastric cancer. Dig Dis Sci. 2010;55:1516-1524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 4. | Lees C, Howie S, Sartor RB, Satsangi J. The hedgehog signalling pathway in the gastrointestinal tract: implications for development, homeostasis, and disease. Gastroenterology. 2005;129:1696-1710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 78] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 5. | Lupi O. Correlations between the Sonic Hedgehog pathway and basal cell carcinoma. Int J Dermatol. 2007;46:1113-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Yang ZJ, Ellis T, Markant SL, Read TA, Kessler JD, Bourboulas M, Schüller U, Machold R, Fishell G, Rowitch DH. Medulloblastoma can be initiated by deletion of Patched in lineage-restricted progenitors or stem cells. Cancer Cell. 2008;14:135-145. [PubMed] |
| 7. | Thayer SP, di Magliano MP, Heiser PW, Nielsen CM, Roberts DJ, Lauwers GY, Qi YP, Gysin S, Fernández-del Castillo C, Yajnik V. Hedgehog is an early and late mediator of pancreatic cancer tumorigenesis. Nature. 2003;425:851-856. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1204] [Cited by in RCA: 1116] [Article Influence: 48.5] [Reference Citation Analysis (4)] |
| 8. | Kayed H, Kleeff J, Esposito I, Giese T, Keleg S, Giese N, Büchler MW, Friess H. Localization of the human hedgehog-interacting protein (Hip) in the normal and diseased pancreas. Mol Carcinog. 2005;42:183-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Jiang F, Liao Z, Hu LH, Du YQ, Man XH, Gu JJ, Gao J, Gong YF, Li ZS. Comparison of antioxidative and antifibrotic effects of α-tocopherol with those of tocotrienol-rich fraction in a rat model of chronic pancreatitis. Pancreas. 2011;40:1091-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 10. | Echelard Y, Epstein DJ, St-Jacques B, Shen L, Mohler J, McMahon JA, McMahon AP. Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell. 1993;75:1417-1430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1778] [Cited by in RCA: 1653] [Article Influence: 50.1] [Reference Citation Analysis (0)] |
| 11. | Ingham PW, McMahon AP. Hedgehog signaling in animal development: paradigms and principles. Genes Dev. 2001;15:3059-3087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2489] [Cited by in RCA: 2307] [Article Influence: 92.3] [Reference Citation Analysis (3)] |
| 12. | Hebrok M, Kim SK, St Jacques B, McMahon AP, Melton DA. Regulation of pancreas development by hedgehog signaling. Development. 2000;127:4905-4913. [PubMed] |
| 13. | Thomas MK, Rastalsky N, Lee JH, Habener JF. Hedgehog signaling regulation of insulin production by pancreatic beta-cells. Diabetes. 2000;49:2039-2047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 109] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 14. | McCarthy RA, Barth JL, Chintalapudi MR, Knaak C, Argraves WS. Megalin functions as an endocytic sonic hedgehog receptor. J Biol Chem. 2002;277:25660-25667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 153] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 15. | Kayed H, Kleeff J, Keleg S, Guo J, Ketterer K, Berberat PO, Giese N, Esposito I, Giese T, Büchler MW. Indian hedgehog signaling pathway: expression and regulation in pancreatic cancer. Int J Cancer. 2004;110:668-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 77] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | Berman DM, Karhadkar SS, Maitra A, Montes De Oca R, Gerstenblith MR, Briggs K, Parker AR, Shimada Y, Eshleman JR, Watkins DN. Widespread requirement for Hedgehog ligand stimulation in growth of digestive tract tumours. Nature. 2003;425:846-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1024] [Cited by in RCA: 947] [Article Influence: 41.2] [Reference Citation Analysis (3)] |
| 17. | Kayed H, Kleeff J, Keleg S, Büchler MW, Friess H. Distribution of Indian hedgehog and its receptors patched and smoothened in human chronic pancreatitis. J Endocrinol. 2003;178:467-478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 18. | Walter K, Omura N, Hong SM, Griffith M, Vincent A, Borges M, Goggins M. Overexpression of smoothened activates the sonic hedgehog signaling pathway in pancreatic cancer-associated fibroblasts. Clin Cancer Res. 2010;16:1781-1789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 145] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
P- Reviewers: Chiang BL, Gregoire M, Shea JA S- Editor: Zhai HH L- Editor: Webster JR E- Editor: Wang CH