Published online May 7, 2013. doi: 10.3748/wjg.v19.i17.2629
Revised: December 3, 2012
Accepted: January 11, 2013
Published online: May 7, 2013
Processing time: 174 Days and 16.9 Hours
AIM: To establish novel intestinal myofibroblast (IMF) cell lines from mouse colonic mucosa and investigate their biological characters.
METHODS: Primary IMFs were isolated from mucosal tissues of mouse colon that was denuded of epithelial cells and smooth muscle layer. For immortalization, primary IMFs were transfected with simian virus 40 large T antigen (designated as LmcMF). We also isolated some primary IMFs that spontaneously became immortalized without transfection (designated as SmcMF). To check immortality and normality of these cells, we examined their proliferative ability and contact inhibition. Moreover, the expression levels of proteins characterizing IMFs [including α-smooth muscle actin (α-SMA), vimentin, desmin, and type I collagen] and proteins associated with the immune response [such as toll-like receptor 4 (TLR-4), CD14, MD2, IκBα, and p-p38] were determined by Western blotting. The localization of several myofibroblast protein markers was also detected by immunofluorescence staining.
RESULTS: The cell growth assay results show that both LmcMF and SmcMF cells proliferated logarithmically at least up to passage 20. In addition, the contact inhibition assays show that LmcMF and SmcMF stopped growing after the cells reached confluence. These data suggest that these 2 types of cells were immortalized without losing contact inhibition of growth. Moreover, both LmcMF and SmcMF, like primary IMFs, showed spindle-shaped appearance. The expression levels of key myofibroblast protein markers, including α-SMA, vimentin, and desmin, were also examined by the Western blotting and immunofluorescence analyses. Our results show that these cells were positive for α-SMA and vimentin, but not desmin, as well as that both LmcMF and SmcMF expressed type I collagen at a lower level than primary IMFs. Finally, we investigated the expression level of lipopolysaccharide (LPS) receptor-related proteins, as well as the response of the cells to LPS treatment. We found that the TLR4, CD14, and MD-2 proteins were present in LmcMF and SmcMF, as well as in primary IMFs, and that all these cells responded to LPS.
CONCLUSION: We established 2 novel IMF cell lines from mouse colonic mucosa, namely, LmcMF and SmcMF, both of which were able to respond to LPS.
- Citation: Kawasaki H, Ohama T, Hori M, Sato K. Establishment of mouse intestinal myofibroblast cell lines. World J Gastroenterol 2013; 19(17): 2629-2637
- URL: https://www.wjgnet.com/1007-9327/full/v19/i17/2629.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i17.2629
The gastrointestinal mucosa is in contact with the extracorporeal environment and is exposed to various antigens and molecules that are mainly derived from ingested food and commensal bacteria[1]. Such antigens do not induce inflammation in healthy individuals, because intestinal epithelial cells form a physical barrier protecting against luminal bacteria and toxic substances. To achieve this protective function, differentiation and proliferation of epithelial cells need to be properly regulated. Accumulating evidence has recently revealed that subepithelial intestinal myofibroblasts (IMFs), which are located subjacent to the epithelium, play crucial roles in regulating epithelial cells[2-4].
IMFs belong to the myofibroblast family, which includes several functionally related cells, such as lung contractile interstitial cells, pancreatic stellate cells, and orbital and synovial fibroblasts[5]. IMFs are spindle-shaped or stellate cells that exhibit phenotypic characteristics of both fibroblasts and smooth muscle cells[6,7]. In addition, it has been shown that IMFs, like other mesenchymal cells, synthesize collagen[6,8,9]. Notably, IMFs orchestrate diverse events in gastrointestinal health and diseases, including epithelial differentiation and development, mucosal repair, carcinogenesis, and inflammatory responses[3,4,10]. IMFs express toll-like receptor 4 (TLR-4), which is activated by lipopolysaccharide (LPS), a component of gram-negative bacteria, subsequently leads to secretion of proinflammatory mediators[10-13]. Moreover, IMFs, as nonprofessional antigen-presenting cells in the intestinal mucosa, induce proliferation of both resting CD4+ T cells and regulatory T cells in a major histocompatibility complex (MHC) class II-dependent manner to maintain intestinal mucosal tolerance[14,15]. Furthermore, enterotoxins are known to engage MHC class II molecules on IMFs, which in turn leads to secretion of inflammatory mediators[16].
Despite the crucial role of IMFs in gastrointestinal health and diseases, the understanding of their underlying regulatory mechanism is limited. One of the major issues that hinder the advance of IMF research is the lack of IMF cell lines that show myofibroblastic phenotypes without stimulation. Typically, primary IMFs isolated from intestine are used within passages 2-6, because their phenotypes change and the cells stop growing after repeated passages. In addition, CCD-18Co cells, which have been used as a human colon myofibroblast cell line, still need to be treated with transforming growth factor-β (TGF-β) for the expression of α-smooth muscle actin (α-SMA)[17]. In this present study, to address these issues, we separated mouse intestinal myofibroblast cell lines (namely, LmcMF and SmcMF) and examined their properties.
C57BL/6J mice purchased from Charles River Japan (Yokohama, Japan) were maintained in compliance with the guidelines of the Animal Care and Use Committee of Yamaguchi University. Mouse IMFs were isolated as previously described, with slight modifications[18]. Briefly, a segment of the proximal colon was detached from the mesenterium, and mucosal layers were completely denuded of epithelial cells by repeated (× 3) 30-min incubation in 1 mmol/L EDTA-Hanks’ balanced salt solution at 37 °C. Subsequently, smooth muscle layers were detached from mucosal layers with tweezers. De-epithelialized mucosal samples were cultured in Dulbecco's Modified Eagle Medium (DMEM; Invitrogen, Tokyo, Japan) containing 100 mL/L fetal bovine serum (FBS) at 37 °C in a 50 mL/L CO2 atmosphere. Consequently, myofibroblast cells that migrated from mucosal tissues formed colonies. In this study, primary IMFs at passages 2-6 were used. Mouse embryonic fibroblasts (MEFs) isolated from embryos of C57BL/6J mice at embryonic day 11.5 were also grown under the same condition described above.
The following antibodies were used in this study: anti-α-SMA and anti-vimentin antibodies from Sigma (Tokyo, Japan); anti-desmin and anti-tubulin antibodies from Thermo Scientific (Yokohama, Japan); anti-TLR4, anti-CD14, and anti-actin antibodies from Santa Cruz (CA, United States); anti-MD2 antibody from AbD Serotec (Kidlington, United Kingdom); anti-IκBα and anti-p-p38 antibodies from Cell Signaling (MA, United States); anti-Collagen type I antibody from Merck (Tokyo, Japan); anti-valosin-containing protein (VCP) antibody from Gene Tex (CA, United States); horseradish peroxidase (HRP)-conjugated mouse anti-rabbit Ig Light Chain from ECM biosciences (KY, United States); and HRP-conjugated donkey anti-mouse IgG from R and D Systems (MN, United States). A mouse mammary gland epithelial cell line, Eph4, and a mouse macrophage-like cell line, RAW264.7, were grown in DMEM supplemented with 100 mL/L FBS at 37 °C in a 50 mL/L CO2 atmosphere.
EGIP-EF1a-Large T-IRES-Puro, a lentiviral plasmid expressing simian virus 40 (SV40) large T antigen (LT) but not small T antigen (ST), was obtained from Addgene (ID No. 18922; MA, United States)[19]. Using Lipofectamine LTX (Invitrogen), lentivirus was produced by transfecting Lenti-X 293T cells in 60-mm dishes with 3 μg of EGIP-EF1a-Large T-IRES-Puro, 2.3 μg of a packaging plasmid (psPAX2), and 1.3 μg of a coat protein plasmid expressing vesicular stomatitis virus G protein (pDM2.G). Viral supernatants were collected after 48 h, and filtered (0.22 μm). Primary IMFs were infected with virus for 8 h. With puromycin treatment for 6 d, immortalized LT-positive cells were selected and designated as LmcMF.
To obtain spontaneously immortalized IMFs, primary IMFs were repeatedly passaged until cells that kept proliferating emerged. These spontaneously immortalized cells were designated as SmcMF.
To determine the proliferative ability of IMF cells, 5 × 104 cells were plated in 35-mm dishes and passaged every 3 d, and the number of cells was subsequently counted using a hemocytometer. For the contact inhibition assay, 5 × 104 cells were plated in 35-mm dishes, and the number of cells was counted on the next day and then every other day.
Primary IMFs, as well as LmcMF and SmcMF, were grown on glass coverslips and subsequently fixed with 4% formaldehyde for 20 min at room temperature. Cells were permeabilized with 0.2% Triton X-100 in PBS-T (PBS containing 0.05% Tween 20) for 60 s and then blocked with 3% skim milk in PBS-T. After incubation with the first antibodies overnight at 4 °C, Alexa594-conjugated secondary antibodies (Alexa Fluor® 594 goat anti-mouse IgG or Alexa Fluor® 594 donkey anti-goat IgG; Invitrogen) were added and incubated for 1 h at room temperature. Nuclei were counterstained with SYTOX Green (Life Technologies, CA, United States). Finally, fluorescence images were captured by a confocal laser-scanning microscope (LSM510; Zeiss, Tokyo, Japan).
The western blot analysis was performed as previously described[18]. Briefly, cells were lysed in a buffer consisting of 50 mmol/L Tris-HCl (pH 8.0), 5 mmol/L EDTA (pH 8.0), 5 mmol/L EGTA (pH 8.0), 1% Triton X-100, 1 mmol/L Na3VO4, 20 mmol/L sodium pyrophosphate, and Roche Complete Protease Inhibitor Cocktail (Roche, Tokyo, Japan). Amersham Hybond ECL Nitrocellulose Membranes (GE Healthcare, Buckinghamshire, United Kingdom) were blocked with 0.5% skim milk and treated with specific antibodies. Protein bands were detected using the ECL Western blotting Detection Reagents (GE Healthcare) or the Western Lightning ECL Pro (PerkinElmer, MA, United States) and visualized using a LAS-3000 mini luminescence imager (Fujifilm, Tokyo, Japan).
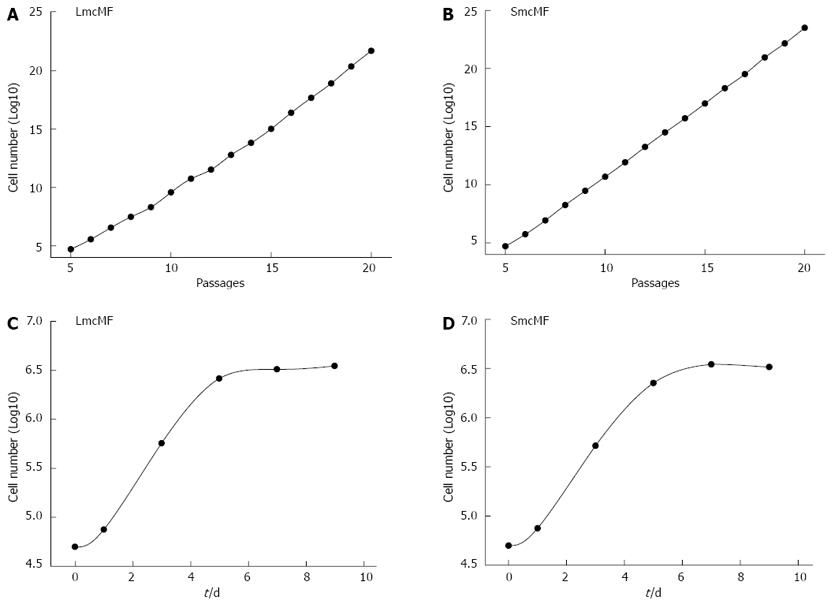
First, we investigated the immortalization of 2 different IMF cell lines, LmcMF and SmcMF. The cells (5 × 104 cells) were seeded in 35-mm dishes and subcultured every 3 d during the examination of the cell growth rate. As shown in Figure 1A and B, both LmcMF and SmcMF cells proliferated logarithmically at least up to passage 20, and their doubling times were 19.1 and 17.2 h, respectively. Primary IMFs, on the other hand, immediately stopped growing under this experimental condition, since they require a high cell density for proliferation.
For the contact inhibition assay, the cells (5 × 104 cells) were seeded in 35-mm dishes and cultured up to 9 d (Figure 1C and D). The cells became confluent around day 5 and reached the plateau phase by day 6 or 7. These data indicate that LmcMF and SmcMF were immortalized without losing contact inhibition of growth.
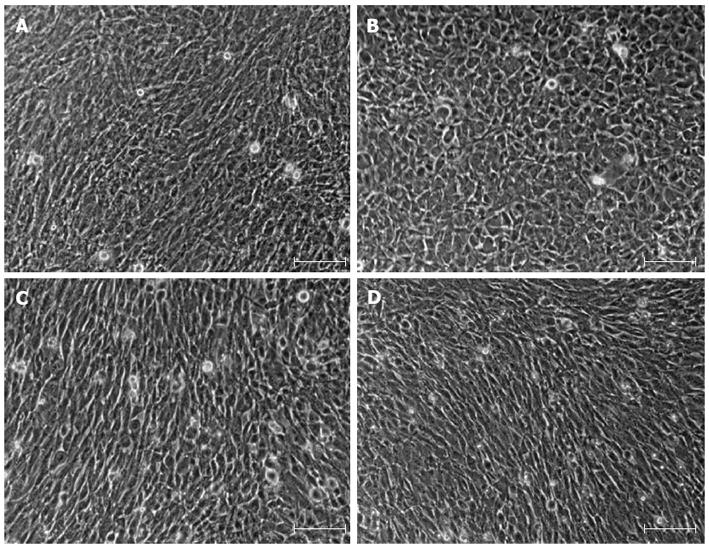
IMFs have been defined as spindle-shaped or stellate cells that are positive for α-SMA and vimentin, and very weakly positive or negative for desmin[7]. As shown in Figure 2A and B, primary IMFs exhibited spindle-shaped appearance, whereas MEFs were round-shaped. Both LmcMF and SmcMF, like primary IMFs, showed spindle-shaped appearance (Figure 2C and D).
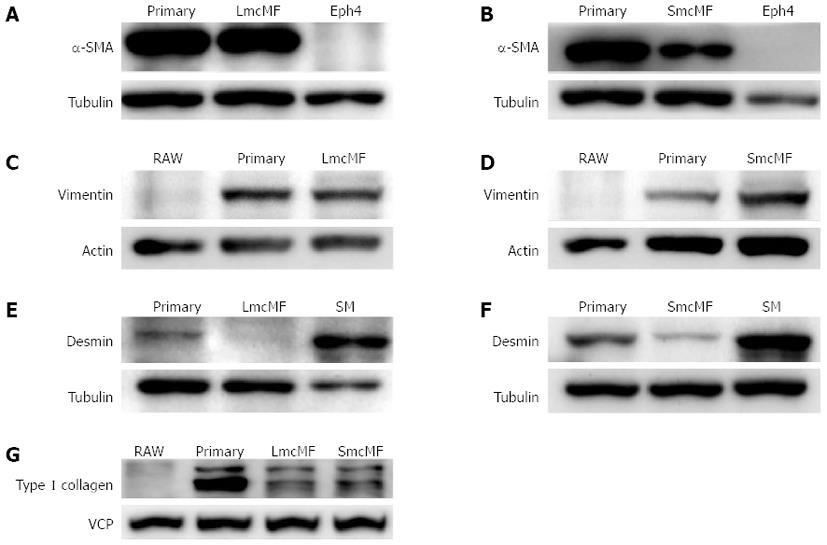
The expression levels of key myofibroblast protein markers, including α-SMA, vimentin, and desmin, were subsequently examined by the Western blotting and immunofluorescence analyses. We observed that primary IMFs and LmcMF expressed an equivalent amount of α-SMA, whereas the α-SMA protein level in SmcMF was relatively low (Figure 3A and B). As expected, α-SMA was not detectable in the mouse mammary gland epithelial cell line, Eph4, which was used as a negative control. We also found that vimentin was present in both LmcMF and SmcMF, almost at the same level as that in primary IMFs, but was not detectable in the negative control RAW 264.7 cells (Figure 3C and D). Moreover, desmin, though not present in LmcMF, was detected at very low levels in SmcMF, compared to its expression level in intestinal smooth muscle tissues (positive control) (Figure 3E and F). Furthermore, both LmcMF and SmcMF expressed type I collagen, however, at a lower level than that in primary IMFs (Figure 3G). As the negative control, RAW 264.7 cells did not express type I collagen. Overall, these results suggest that LmcMF and SmcMF share almost the same characteristics with intestinal myofibroblasts.
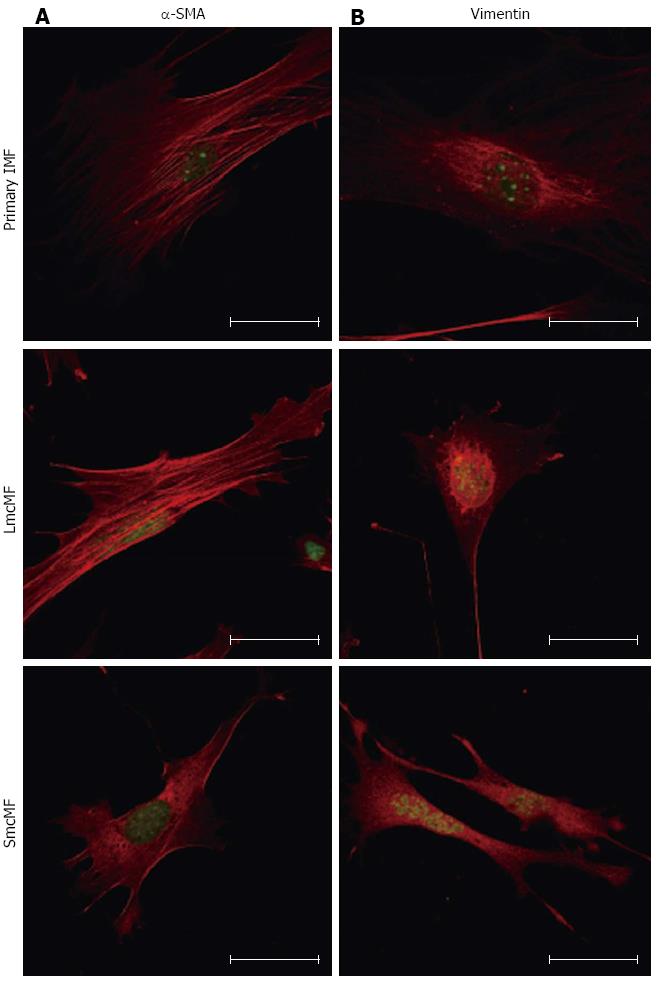
To confirm the expression pattern of α-SMA and vimentin in IMFs, we next performed the immunofluorescence analysis. In a cell, actin transits between its globular (G-) and filamentous (F-actin) states, and G-actin can be visualized by immunofluorescence microscopy in a spreading pattern, because they are monomeric actin[20]. As shown in Figure 4A, we observed strong filamentous α-SMA expression in primary IMFs. While LmcMF also showed filamentous α-SMA fibers, the amount of globular α-SMA was higher than that in primary IMFs. Most of the α-SMA protein in SmcMF existed in a globular α-SMA form. On the other hand, vimentin was predominantly localized around the nucleus of primary IMFs and LmcMF but diffused throughout SmcMF cells (Figure 4B).
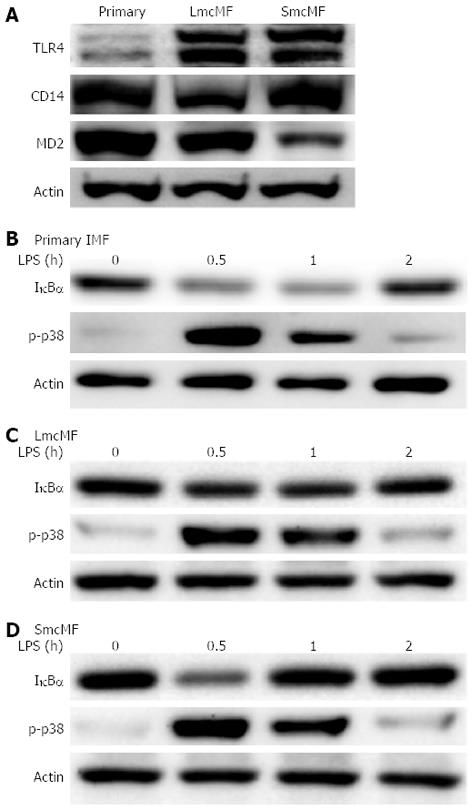
It is well known that the LPS receptor complex is composed of TLR4, CD14, and MD-2 and that both CD14 and MD-2 are required for the activation of TLR4[21]. A study has previously shown that primary IMFs expressed all these 3 components and that the exposure of cells to LPS activated the NF-κB and p38 MAPK pathways[10]; therefore, we assessed whether the same holds true for LmcMF and SmcMF. Our results show that although the expression level of TLR4 was low in primary IMFs, the levels in LmcMF and SmcMF were much higher (Figure 5A). As for CD14, the expression level was lower in LmcMF, compared to primary IMFs and SmcMF, and the expression level of MD-2 was relatively higher in primary IMFs and lower in SmcMF, compared to LmcMF.
Finally, we investigated the reactivity of primary IMFs, LmcMF, and SmcMF to LPS. We found that LPS treatment (20 ng/mL) induced the degradation of IκBα, as well as the phosphorylation of p38 MAPK (Figure 5B-D). While the maximal levels of IκBα degradation and p38 MAPK phosphorylation were detected at 30 min after LPS treatment, their expression was restored to basal levels within 2 h. Consistent with the previous study (performed in primary IMFs)[10], our results demonstrate that both LmcMF and SmcMF preserve myofibroblastic phenotypes.
IMFs play an important role in intestinal injury, inflammation, fibrosis, and tissue repair; however, their exact function and involvement are still not completely understood[6,8,10,22]. One issue that hinders the development of IMF research is the lack of IMF cell lines. In this present study, we established 2 IMF cell lines, namely, LmcMF and SmcMF, both of which were immortalized without losing contact inhibition of growth. LmcMF was established by transfecting primary IMFs with plasmid DNA encoding the LT of replication origin-defective SV40. It is known that 2 oncoproteins, LT and ST, encoded by the SV40 early region play an essential role in inducing transformation of mammalian cells[23]. In addition, previous studies have also shown that LT can immortalize cells but cannot induce neoplastic transformation without ST[23]. Moreover, it has been shown that immortalization of mouse cells can be achieved by transfecting only with LT[24]. Based on these findings, we used a plasmid that expresses LT, but not ST, for our immortalization experiments of IMFs. On the other hand, SmcMF was derived from cells that spontaneously immortalized. It has been reported that murine cells tend to immortalize spontaneously in culture, because mouse somatic cells have the telomerase activity[25].
IMFs can be distinguished from other types of mesenchymal cells, such as smooth muscle cells and fibroblasts, by the expression of 3 protein markers: α-SMA, vimentin, and desmin. It was reported that IMFs are positive for α-SMA and vimentin, but negative or very weakly positive for desmin[7]. In contrast, smooth muscle cells are α-SMA-positive, vimentin-negative, and strongly positive for desmin, whereas typical fibroblasts, such as dermal fibroblasts, are negative for α-SMA and desmin, but positive for vimentin[7,8,26]. In the present study, we observed that all 3 cell lines (primary IMFs, LmcMF, and SmcMF) expressed both α-SMA and vimentin, suggesting that these cells share the same characteristics and that LmcMF and SmcMF can be used as intestinal myofibroblast cell lines. It is also noted that the doubling time of these cells is within a day, whereas that of human primary IMFs is about 5 d[27]. Therefore, the use of LmcMF and SmcMF will be helpful for improving the efficiency of IMF research.
There are, however, some differences among these cells. For instance, the α-SMA expression level was slightly lower in SmcMF, and α-SMA was organized into stress fibers in primary IMFs and LmcMF, but not in SmcMF. The vimentin expression level was slightly higher in SmcMF, whereas the type I collagen levels in LmcMF and SmcMF were relatively lower than that in primary IMFs. We propose that these differences may depend on the differentiation stage of the cells. In the present study, very low levels of desmin were detected in primary IMFs and SmcMF. It has been reported that primary human colonic myofibroblasts and CCD-18Co cells do not express desmin[28], whereas some lesion myofibroblasts do[7]. These differences in the desmin expression may occur by cell differentiation or by the differences between animal species tested (for example, mice vs humans); however, to clarify this, additional experiments are necessary.
The differentiation of fibroblasts into myofibroblasts is an important step in tissue repair and fibrosis[29]; however, it is a complex process and has not been completely elucidated. It was reported that CCD-18Co cells exhibit fibroblastic and myofibroblastic phenotypes and that TGF-β stimulates their differentiation into myofibroblasts[17]. CCD-18Co cells have also been used to investigate the mechanism of differentiation into myofibroblasts during wound healing processes. However, CCD-18Co cells poorly differentiate into myofibroblasts[30]; therefore, LmcMF and SmcMF may be useful to help reveal the role of differentiated myofibroblasts in tissue repair.
Accumulating evidence has shown that IMFs play a crucial role in local immune regulation in the intestine[10,14,15]; therefore, it is important that the developed cell lines show immune responses to external factors. Consistent with previous report[10], all three types of IMF expressed TLR4, CD14, and MD-2. We also demonstrate that LmcMF and SmcMF, similar to primary IMFs, were able to respond to LPS stimulation.
In the present study, we observed that the TLR4 expression levels in LmcMF and SmcMF were higher than that in primary IMFs. It is possible that TLR4 was downregulated in primary IMFs and then recovered in LmcMF and SmcMF, since during the cell isolation procedure the intestinal tissues and cells were exposed to a large amount of LPS that existed in the intestinal lumen. Interestingly, although the TLR4 expression level in primary IMFs was low, LPS induced almost the same responses, including IκBα degradation and p38 MAPK phosphorylation, in all 3 types of IMFs. Together, our results indicate that the amount of TLR4 protein in primary IMFs was sufficient to respond to LPS.
In conclusion, we have successfully established 2 novel types of myofibroblast cell lines from mouse colon, namely, LmcMF and SmcMF, both of which expressed the key myofibroblast markers. Moreover, these cell lines were immortalized (rather than transformed to neoplastic cells) and were able to respond to LPS, like primary IMFs. Furthermore, these cell lines proliferate much faster than primary IMFs, and therefore they can be used efficiently. Nevertheless, for the development of IMF research using LmcMF and SmcMF, more experiments are necessary to determine their properties.
With respect to the gastrointestinal mucosa, accumulating evidence has shown that subepithelial intestinal myofibroblasts (IMFs), which are located subjacent to the epithelium, play crucial roles in regulating epithelial cells. IMFs orchestrate diverse events in gastrointestinal health and diseases, including epithelial differentiation and development, mucosal repair, carcinogenesis, and inflammatory responses. Therefore, it is important to establish IMF cell lines, which can be used for clarifying the role of IMFs.
Despite the crucial role played by IMFs in gastrointestinal health and diseases, the understanding of their underlying regulatory mechanism is limited. A major issue that hinders the advance of IMF research is the lack of IMF cell lines that exhibit myofibroblastic phenotypes. In this study, authors established 2 IMF cell lines, namely, LmcMF and SmcMF, both of which were immortalized without losing contact inhibition of growth, and were able to respond to lipopolysaccharide, like primary IMFs.
For the IMF research, the 2 following cell types are available: freshly isolated IMFs (primary IMFs) and IMF-like cell lines (such as CCD-18Co cells). However, primary IMFs typically change phenotypes within passage 6 and stop growing after repeated passages. In addition, CCD-18Co cells need to be treated with TGF-β for the expression of key myofibroblast protein markers. To the best of our knowledge, this report is the first to establish novel types of myofibroblast cell lines from the mouse colon. Authors developed 2 cell lines, namely, LmcMF and SmcMF, both of which expressed the key myofibroblast markers without stimulation.
Because IMFs play crucial roles in regulating epithelial cells, it is important to develop IMF cell lines. The establishment of 2 cell lines is helpful for improving the efficiency of IMF research.
This study characterizes 2 IMF cell lines isolated from the mouse colon. This is a straightforward descriptive report and is methodological in nature; essentially, the authors have generated a potentially useful cell line resource that may be used to further myofibroblast research into tissue repair and fibrosis, immune regulation within the intestine, and other aspects of myofibroblast function. Because only a few good myofibroblast cell lines are currently available, it is important to create new myofibroblast cell lines for in vitro studies, and the authors have generated 2 IMF cell lines by using 2 different approaches.
| 1. | Sartor RB. Microbial influences in inflammatory bowel diseases. Gastroenterology. 2008;134:577-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1560] [Cited by in RCA: 1382] [Article Influence: 76.8] [Reference Citation Analysis (0)] |
| 2. | Powell DW, Mifflin RC, Valentich JD, Crowe SE, Saada JI, West AB. Myofibroblasts. II. Intestinal subepithelial myofibroblasts. Am J Physiol. 1999;277:C183-C201. [PubMed] |
| 3. | Powell DW, Adegboyega PA, Di Mari JF, Mifflin RC. Epithelial cells and their neighbors I. Role of intestinal myofibroblasts in development, repair, and cancer. Am J Physiol Gastrointest Liver Physiol. 2005;289:G2-G7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 161] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 4. | Andoh A, Bamba S, Fujiyama Y, Brittan M, Wright NA. Colonic subepithelial myofibroblasts in mucosal inflammation and repair: contribution of bone marrow-derived stem cells to the gut regenerative response. J Gastroenterol. 2005;40:1089-1099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 61] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 5. | Powell DW, Mifflin RC, Valentich JD, Crowe SE, Saada JI, West AB. Myofibroblasts. I. Paracrine cells important in health and disease. Am J Physiol. 1999;277:C1-C9. [PubMed] |
| 6. | Mifflin RC, Pinchuk IV, Saada JI, Powell DW. Intestinal myofibroblasts: targets for stem cell therapy. Am J Physiol Gastrointest Liver Physiol. 2011;300:G684-G696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 117] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 7. | Eyden B. The myofibroblast: phenotypic characterization as a prerequisite to understanding its functions in translational medicine. J Cell Mol Med. 2008;12:22-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 189] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 8. | Pucilowska JB, McNaughton KK, Mohapatra NK, Hoyt EC, Zimmermann EM, Sartor RB, Lund PK. IGF-I and procollagen alpha1(I) are coexpressed in a subset of mesenchymal cells in active Crohn’s disease. Am J Physiol Gastrointest Liver Physiol. 2000;279:G1307-G1322. [PubMed] |
| 9. | Theiss AL, Simmons JG, Jobin C, Lund PK. Tumor necrosis factor (TNF) alpha increases collagen accumulation and proliferation in intestinal myofibroblasts via TNF receptor 2. J Biol Chem. 2005;280:36099-36109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 176] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 10. | Walton KL, Holt L, Sartor RB. Lipopolysaccharide activates innate immune responses in murine intestinal myofibroblasts through multiple signaling pathways. Am J Physiol Gastrointest Liver Physiol. 2009;296:G601-G611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 82] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 11. | Zhang Z, Andoh A, Inatomi O, Bamba S, Takayanagi A, Shimizu N, Fujiyama Y. Interleukin-17 and lipopolysaccharides synergistically induce cyclooxygenase-2 expression in human intestinal myofibroblasts. J Gastroenterol Hepatol. 2005;20:619-627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 12. | Rogler G, Gelbmann CM, Vogl D, Brunner M, Schölmerich J, Falk W, Andus T, Brand K. Differential activation of cytokine secretion in primary human colonic fibroblast/myofibroblast cultures. Scand J Gastroenterol. 2001;36:389-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 60] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 13. | Otte JM, Rosenberg IM, Podolsky DK. Intestinal myofibroblasts in innate immune responses of the intestine. Gastroenterology. 2003;124:1866-1878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 190] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 14. | Saada JI, Pinchuk IV, Barrera CA, Adegboyega PA, Suarez G, Mifflin RC, Di Mari JF, Reyes VE, Powell DW. Subepithelial myofibroblasts are novel nonprofessional APCs in the human colonic mucosa. J Immunol. 2006;177:5968-5979. [PubMed] [DOI] [Full Text] |
| 15. | Pinchuk IV, Beswick EJ, Saada JI, Boya G, Schmitt D, Raju GS, Brenmoehl J, Rogler G, Reyes VE, Powell DW. Human colonic myofibroblasts promote expansion of CD4+ CD25high Foxp3+ regulatory T cells. Gastroenterology. 2011;140:2019-2030. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 16. | Pinchuk IV, Beswick EJ, Saada JI, Suarez G, Winston J, Mifflin RC, Di Mari JF, Powell DW, Reyes VE. Monocyte chemoattractant protein-1 production by intestinal myofibroblasts in response to staphylococcal enterotoxin a: relevance to staphylococcal enterotoxigenic disease. J Immunol. 2007;178:8097-8106. [PubMed] |
| 17. | Simmons JG, Pucilowska JB, Keku TO, Lund PK. IGF-I and TGF-beta1 have distinct effects on phenotype and proliferation of intestinal fibroblasts. Am J Physiol Gastrointest Liver Physiol. 2002;283:G809-G818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 96] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Ohama T, Okada M, Murata T, Brautigan DL, Hori M, Ozaki H. Sphingosine-1-phosphate enhances IL-1{beta}-induced COX-2 expression in mouse intestinal subepithelial myofibroblasts. Am J Physiol Gastrointest Liver Physiol. 2008;295:G766-G775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Mali P, Ye Z, Hommond HH, Yu X, Lin J, Chen G, Zou J, Cheng L. Improved efficiency and pace of generating induced pluripotent stem cells from human adult and fetal fibroblasts. Stem Cells. 2008;26:1998-2005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 208] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 20. | Lee SH, Dominguez R. Regulation of actin cytoskeleton dynamics in cells. Mol Cells. 2010;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 306] [Cited by in RCA: 258] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 21. | Komatsu H, Shimose A, Shimizu T, Mukai Y, Kobayashi J, Ohama T, Sato K. Trypsin inhibits lipopolysaccharide signaling in macrophages via toll-like receptor 4 accessory molecules. Life Sci. 2012;91:143-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 22. | van Tol EA, Holt L, Li FL, Kong FM, Rippe R, Yamauchi M, Pucilowska J, Lund PK, Sartor RB. Bacterial cell wall polymers promote intestinal fibrosis by direct stimulation of myofibroblasts. Am J Physiol. 1999;277:G245-G255. [PubMed] |
| 23. | Chen W, Possemato R, Campbell KT, Plattner CA, Pallas DC, Hahn WC. Identification of specific PP2A complexes involved in human cell transformation. Cancer Cell. 2004;5:127-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 264] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 24. | Rangarajan A, Hong SJ, Gifford A, Weinberg RA. Species- and cell type-specific requirements for cellular transformation. Cancer Cell. 2004;6:171-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 407] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 25. | Blasco MA, Funk W, Villeponteau B, Greider CW. Functional characterization and developmental regulation of mouse telomerase RNA. Science. 1995;269:1267-1270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 257] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 26. | Ramadori G, Veit T, Schwögler S, Dienes HP, Knittel T, Rieder H, Meyer zum Büschenfelde KH. Expression of the gene of the alpha-smooth muscle-actin isoform in rat liver and in rat fat-storing (ITO) cells. Virchows Arch B Cell Pathol Incl Mol Pathol. 1990;59:349-357. [PubMed] |
| 27. | Mahida YR, Beltinger J, Makh S, Göke M, Gray T, Podolsky DK, Hawkey CJ. Adult human colonic subepithelial myofibroblasts express extracellular matrix proteins and cyclooxygenase-1 and -2. Am J Physiol. 1997;273:G1341-G1348. [PubMed] |
| 28. | Peiris D, Pacheco I, Spencer C, MacLeod RJ. The extracellular calcium-sensing receptor reciprocally regulates the secretion of BMP-2 and the BMP antagonist Noggin in colonic myofibroblasts. Am J Physiol Gastrointest Liver Physiol. 2007;292:G753-G766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 29. | Brenmoehl J, Miller SN, Hofmann C, Vogl D, Falk W, Schölmerich J, Rogler G. Transforming growth factor-beta 1 induces intestinal myofibroblast differentiation and modulates their migration. World J Gastroenterol. 2009;15:1431-1442. [PubMed] [DOI] [Full Text] |
| 30. | Desmoulière A, Chaponnier C, Gabbiani G. Tissue repair, contraction, and the myofibroblast. Wound Repair Regen. 2005;13:7-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 735] [Cited by in RCA: 649] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
P- Reviewers MacLeod RJ, Spring KJ S- Editor Gou SX L- Editor A E- Editor Zhang DN