Published online Apr 28, 2008. doi: 10.3748/wjg.14.2596
Revised: February 29, 2008
Published online: April 28, 2008
Chronic pancreatitis characterized by an early onset should be extensively investigated including the search for a mutation of the PRSS1, SPINK-1 or CFTR genes and potential features of autoimmune pancreatitis. We here describe a case of chronic pancreatitis with an onset at a very young age in which a mutation of the PRSS1 and several features of autoimmune pancreatitis were identified.
- Citation: Frossard JL, Dumonceau JM, Pastor C, Spahr L, Hadengue A. Concomitant autoimmune and genetic pancreatitis leads to severe inflammatory conditions. World J Gastroenterol 2008; 14(16): 2596-2598
- URL: https://www.wjgnet.com/1007-9327/full/v14/i16/2596.htm
- DOI: https://dx.doi.org/10.3748/wjg.14.2596
Chronic pancreatitis is a progressive inflammatory disease of the pancreatic gland that will finally result in both exocrine and endocrine insufficiency[1]. The insult to the gland is usually caused by recurrent episodes of pancreatic necro-inflammation, leading to pancreatic atrophy and dysfunction. It manifests clinically as maldigestion, weight loss, constant or intermittent abdominal pain and eventually diabetes. Over the past decade, tremendous advance has been made regarding the etiology of chronic pancreatitis[2]. Alcohol abuse remains the most frequent cause of chronic pancreatitis, while other causes have been delineated such as tobacco abuse, genetically determined pancreatitis and autoimmune pancreatitis[3]. Additional causes such as pancreas divisum are controversial. Cases of recurrent acute pancreatitis and some forms of chronic pancreatitis have been definitively associated with genetic mutations[4]. Indeed, genetic analyses have identified a specific gene for hereditary pancreatitis on the long arm of the chromosome 7 (cationic trypsinogen gene or PRSS1). Some PRSS1 mutations enhance trypsinogen autoactivation, explaining the onset at young age, recurrent acute episodes of pancreatitis and progress into chronic pancreatitis. Other mutations may render some patients more susceptible to pancreatitis in the presence of other insults to the pancreas as shown in AIDS patients receiving potentially harmful drugs for the pancreas[5]. Thus, serine protease inhibitor Kazal type 1 (SPINK1) and cystic fibrosis transmembrane conductance regulator (CFTR) genes have been involved in idiopathic recurrent acute pancreatitis and chronic pancreatitis. Since 1965, several authors have paid attention to particular cases of pancreatitis of an autoimmune pattern and some of the cases were associated with Sjögren’s syndrome or primary biliary cirrhosis[6]. This entity was recognized as autoimmune pancreatitis, a condition responsible for both acute and chronic pancreatitis.
We herein describe a case of chronic pancreatitis with an onset at a very young age, in which a mutation of the PRSS1 and several features of autoimmune pancreatitis were identified.
A 33-year-old man approached to our pancreatic outpatient unit for information because he read on internet about recent advance made in the understanding of the causes of pancreatitis. His disease history started 23 years ago at the age of 10 years with the first episode of acute pancreatitis that required a 21-d hospital stay. The first episode was characterized by an acute onset of abdominal pain localized in the epigastric area accompanied by intractable vomiting associated with increased serum amylase concentrations twice the upper normal values. White blood cell counts were 10 350/mm3 with 19% of band form granulocytes. Calcium, triglyceride levels and liver enzymes were within normal range. Mumps serology confirmed a previous disease. Percutaneous abdominal sonography identified an enlarged gland while the gallbladder was normal without stone. There was no familial history of pancreatic disorders and the patient had no other relevant past medical history. A conservative approach was applied while the extensive search for the etiology of acute pancreatitis remained unsuccessful after exclusion of biiary disease, alcohol abuse, drug-induced acute pancreatitis. Between 1986 and 1990, the patient subsequently developed four recurrent episodes of acute pancreatitis, the cause remains inconclusive. A sweat test was normal excluding cystic fibrosis, while there was a suspicion of pancreas divisum during endoscopic retrograde cholangiopancreatography. In 1990, the patient underwent cholecystectomy and surgical papillotomy. Transcystic cholangiopancreatography showed a normal common bile duct and a normal pancreatic ductular tract, ruling out the previous suspicion of pancreas divisum. The progress between 1990 and 1996 was marked by 5 additional episodes of pancreatitis with calcifications localized in the head and corpus of the pancreas. In 1996, a magnetic resonance imaging (MRI) performed during the 10th episode of pancreatitis identified a ductular stenosis of the caudal part of the main duct associated with distal dilatation of side brench ducts. A caudal pancreatectomy was performed without complications. Histological analysis showed pancreatic atrophy associated with large fibrous strains. Since 1996, the patient has suffered from 9 additional uncomplicated episodes of acute pancreatitis. He never drank alcohol neither was addicted to tobacco use. He was referred to us after the last severe acute pancreatitis in May 2007.
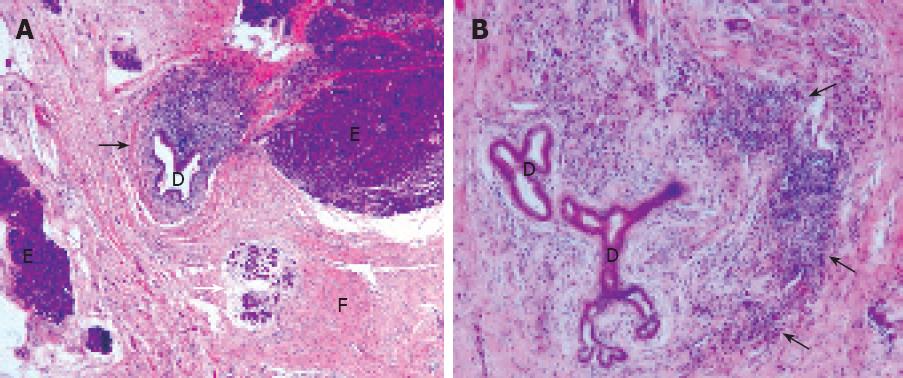
Based on the clinical presentation of the patient, we choose to investigate point mutations in the cationic trypsinogen gene (PRSS1) that underlie hereditary pancreatitis, although we had no data suggestive of familial pancreatitis in this patient. For CFTR mutations and SPINK-1 polymorphisms, we only analysed the most frequent 33 mutations associated with a more severe clinical importance. Whereas genetic testing revealed normal CFTR and SPINK1 genes, PRSS1 analysis revealed the mutation p.A16V which represents the third most common mutation in PRSS1 besides R122H and N29I. Serum auto-antibodies including antilactoferrin antibodies, anticarbonic anhydrase antibodies typeIand II were strongly positive, whereas serum gammaglobulin levels IgG4 reached 12.8% (normal range: 2.5%-5.9%). A second interpretation of the pancreatic specimen collected in 1996 was performed in our University Hospital and clearly showed a periductular lymphoplasmacytic infiltrate and a diffuse pancreatic fibrosis (Figure 1), and all features characterized autoimmune pancreatitis.
Personal and familial histories, clinical symptoms, laboratory tests, and histological studies when available may help identify some of the etiologies of chronic pancreatitis, but 15%-25% of the cases remain of unknown origin. Recently, autoimmune diseases and genetic mutations have been described in association with pancreatic diseases. Particularly, the PRSS1, R122H and N29I mutations have been found to be responsible for hereditary pancreatitis, a rare cause of pancreatitis characterized by an autosomal dominant inheritance pattern and high penetrance reaching 70%-80%[7]. Whereas the R122H and N29I mutations enhance trypsinogen autoactivation, the PRSS1 variant p.A16V displays another harmful mechanism by increasing the activation rate of peptide processing regulated by chymotrypsin, which finally accelerates the trypsinogen activation. Moreover, the p.A16V is found most exclusively in patients without a familial history of pancreatitis, a feature shared by our patient. Molecular test represents a very effective tool to investigate the acute and chronic pancreatitis after having ruled out more common causes of pancreatitis such as biliary stones migration, alcohol-abuse and pancreatitis-induced pancreatitis[8]. Indeed, genetic test can be used to identify unsuspected cases of genetically transmitted disease, particularly in patients without a strong history in pedigrees. Surprisingly, the patient concomitantly presented with several biological and histological features that confirm the diagnosis of autoimmune pancreatitis. To what extent autoimmune pancreatitis has influenced the course of the disease in the patient remains speculative, but two key points should be stressed. The median age of the onset of hereditary pancreatitis is 12 years[9], while the patient became symptomatic at age 10. The pain usually improves or resolves with time in the majority of patients while the patient still had severe attacks that need a long hospital stay. The steroids would be used to treat next attack and the clinical response will be observed. If positive, the attributable role of autoimmune pancreatitis will be definitively delineated.
In summary, we present for the first time a patient with a chronic pancreatic disease that may result from a combination of genetic mutations and autoimmune diseases.
| 1. | Frossard JL, Pastor CM. Experimental acute pancreatitis: new insights into the pathophysiology. Front Biosci. 2002;7:d275-d287. |
| 2. | Steer ML, Waxman I, Freedman S. Chronic pancreatitis. N Engl J Med. 1995;332:1482-1490. |
| 3. | Etemad B, Whitcomb DC. Chronic pancreatitis: diagnosis, classification, and new genetic developments. Gastroenterology. 2001;120:682-707. |
| 4. | Le Bodic L, Bignon JD, Raguenes O, Mercier B, Georgelin T, Schnee M, Soulard F, Gagne K, Bonneville F, Muller JY. The hereditary pancreatitis gene maps to long arm of chromosome 7. Hum Mol Genet. 1996;5:549-554. |
| 5. | Felley C, Morris MA, Wonkam A, Hirschel B, Flepp M, Wolf K, Furrer H, Battegay M, Bernasconi E, Telenti A. The role of CFTR and SPINK-1 mutations in pancreatic disorders in HIV-positive patients: a case-control study. AIDS. 2004;18:1521-1527. |
| 6. | Sarles H, Sarles JC, Camatte R, Muratore R, Gaini M, Guien C, Pastor J, Le Roy F. Observations on 205 confirmed cases of acute pancreatitis, recurring pancreatitis, and chronic pancreatitis. Gut. 1965;6:545-559. |
| 7. | Whitcomb DC, Ulrich CD, Lerch MM, Durie P, Neoptolemos JP, Maisonneuve P, Lowenfels AB. Third International Symposium on Inherited Diseases of the Pancreas. Pancreatology. 2001;1:423-431. |
| 8. | Ellis I, Lerch MM, Whitcomb DC. Genetic testing for hereditary pancreatitis: guidelines for indications, counselling, consent and privacy issues. Pancreatology. 2001;1:405-415. |
| 9. | Howes N, Lerch MM, Greenhalf W, Stocken DD, Ellis I, Simon P, Truninger K, Ammann R, Cavallini G, Charnley RM. Clinical and genetic characteristics of hereditary pancreatitis in Europe. Clin Gastroenterol Hepatol. 2004;2:252-261. |