Published online May 15, 2004. doi: 10.3748/wjg.v10.i10.1476
Revised: October 12, 2003
Accepted: November 19, 2003
Published online: May 15, 2004
AIM: To investigate the effects induced by emodin on single smooth muscle cells from rat colon in vitro, and to determine the signal pathways involved.
METHODS: Cells were isolated from the muscle layers of Wistar rat colon by enzymatic digestion. Cell length was measured by computerized image micrometry. Intracellular Ca2+ ([Ca2+]i) signals were studied using the fluorescent Ca2+ indicator fluo-3 and confocal microscopy. PKCα distribution at rest state or after stimulation was measured with immunofluorescence confocal microscopy.
RESULTS: (1) Emodin dose-dependently caused colonic smooth muscle cells contraction; (2) emodin induced an increase in intracellular Ca2+ concentration; (3) the contractile responses induced by emodin were respectively inhibited by preincubation of the cells with ML-7 (an inhibitor of MLCK) and calphostin C (an inhibitor of PKC); and (4) Incubation of cells with emodin caused translocation of PKCα from cytosolic area to the membrane.
CONCLUSION: Emodin has a direct contractile effect on colonic smooth muscle cell. This signal cascade induced by emodin is initiated by increased [Ca2+]i and PKCα translocation, which in turn lead to the activation of MLCK and the suppression of MLCP. Both of them contribute to the emodin-induced contraction.
- Citation: Ma T, Qi QH, Xu J, Dong ZL, Yang WX. Signal pathways involved in emodin-induced contraction of smooth muscle cells from rat colon. World J Gastroenterol 2004; 10(10): 1476-1479
- URL: https://www.wjgnet.com/1007-9327/full/v10/i10/1476.htm
- DOI: https://dx.doi.org/10.3748/wjg.v10.i10.1476
Gastrointestinal dysmotility underlies frequent clinical entities such as diabetes mellitus, chronic constipation, irritable bowel syndrome, postoperative ileus etc. Currently few drugs have been proven to be efficient at improving motility. Emodin is a naturally occurring anthraquinone present in the roots and bark of numerous plants of the genus Rhamnus[1]. Extracts from the roots, bark, and/or dried leaves of buckthorn, senna, cascara, aloe, frangula, and rhubarb have been used as laxatives since ancient times and currently are widely used in the preparation of herbal laxative preparations in China[2-5]. Anthraquinone glycosides are poorly absorbed from the gastrointestinal tract but are cleaved by gut bacteria to produce aglycones, including emodin which are more readily absorbed and thought to be responsible for the purgative properties of these preparations[1]. The reported biological effects of emodin include antitumor, antibacterial and anti-inflammatory actions[6-9]. Emodin also possesses prokinetic effect on gastrointestinal tract. Stimulatory actions of emodin on gastrointestinal smooth muscle have been described in several studies[10-12]. However, its mechanism in accelerating gastrointestinal motion is not yet clarified.
It is now understood that the contraction of smooth muscle cells involves two processes[13-16]: (a) The concentration of intracellular Ca2+ increases; this Ca2+ increase results in phosphorylation of myosin and consequently an increased contractility. (b) The sensitivity of the myofilaments to Ca2+ increases. Pharmacological agents can stimulate smooth muscle cell contractions by mobilizing intracellular Ca2+ and/or enhancing Ca2+ sensitivity.
Because of the potential therapeutic implications of emodin in gastrointestinal hypomotility disorders, this study was then designed to further characterize the effects of emodin on smooth muscle cells from rat colon in vitro and to investigate the signal transduction cascade leading to cell contraction induced by emodin.
Fluo-3 AM and Pluronic F-127 were from Molecular Probes (USA). Rabbit anti-PKCα IgG and goat anti-rabbit IgG FITC were from Santa Cruz Biotechnology (USA). Collagenase type II, emodin, trypsin inhibitor and HEPES were all purchased from Sigma Co (USA). DMEM was purchased from GIBCO Co., USA. FBS was from Hyclone (USA). All other reagents were from LianXing Co.,Ltd (China). Emodin was dissolved in dimethyl sulfoxide and ethanol mixture (2:8) to make stock solution and the final concentration of the vehicle in the solution did not exceed 0.1%.
Isolation of smooth muscle cells Smooth muscle cells from rat distal colon were isolated as described previously[13]. Briefly, a segment of 5-cm long distal colon was dissected and digested to yield isolated smooth muscle cells. The tissue was incubated for 2 successive 30-min periods at 31 °C in 10 mL of HEPES (pH7.4) containing 1 g/L collagenase type II and 0.1 g/L soybean trypsin inhibitor. After the second enzymatic incubation, the medium was filtered through 500 μm Nitex mesh. The partially digested tissue left on the filter was washed with collagenase-free buffer solution, and muscle cells were allowed to disperse spontaneously for 30 min. The cells were harvested by centrifugation at 350 r/min for 10 min. After a hemocytometric cell count, the harvested cells were resuspended in collagenase-free buffer solution and diluted as needed.
Measurement of smooth muscle cell contraction Individual cell length was measured by scanning micrometry as described previously[13]. Aliquots consisting of 1 × 104 cells in 0.25 mL of medium were added to 50 μL of a solution containing emodin with or without prior incubation with ML-7(10-5 mol/L) or Calphostin C (10-6 mol/L). The reaction was allowed to proceed for 1 min and terminated by the addition of acrolein at a final concentration of 10 mL/L. The lengths of 30 consecutive intact healthy cells were measured through a phase-contrast microscope fitted with a video camera and connected to a Legend computer. The CIMAS program was used to measure the length. The contractile response was defined as the decrease in the average length of the 30 cells and was expressed as the percent change relative to control length.
Single cell intracellular Ca2+ measurements Changes in [Ca2+]i were estimated as described elsewhere by using laser scanning confocal microscopy (Radiance 2000; Bio-rad, Hertfortdshire, UK)[17] linked to an inverted epifluorescence microscope (Olympus, Japan). The scanning box was set at a resolution of 256 × 256. Only one fixed combination of laser intensity (20% maximum) and photomultiplier gain (1800 of a maximum of 4096) was used during scanning.
To load the cells with fluorescent Ca2+ indicator fluo-3, smooth muscle cells on the coverslips were incubated with 5 μmol/L of the membrane permeant acetoxymethyl ester of the dye (fluo-3 AM; Molecular Probes, USA) and Pluronic F-127 (0.4 g/L; Molecular Probes, USA) dissolved in HEPES-ringer buffer [containing (mmol/L): NaCl 135, KCl 5.9, CaCl2 1.5, MgCl2 1.2, HEPES 11.6 and glucose 11.5, at pH7.3] for 30 min at 37 °C. Cells were subsequently washed twice with HEPES-ringer buffer and further incubated for 20 min to allow de-esterification of the dye.
Coverglass chambers were then mounted on the microscope stage and continuously superfused with HEPES-ringer buffer. The cells loaded with fluo-3 were illuminated at 488 nm, and fluorescent emissions of 525/30 nm were recorded at an intensity of fluo-3. Digital Ca2+ images were collected at 10 s intervals. Because fluo-3 is a single wavelength indicator, it is not possible to apply the ratiometric method to quantitative determination of [Ca2+]i. Therefore, the intensity of fluo-3 fluorescence was normalized in the temporal analysis. The relative values of fluorescence intensity of the dye (Ft) were used to represent the [Ca2+]i.
Cell culture and confocal imaging of PKCα cell culture After enzymatic incubation, the dispersed cells were collected in DMEM with 100 mL/L FBS-medium, then transferred to coverslips coated with poly-L-lysin, and allowed to settle overnight in a humidified 50 mL/L CO2 environment.
Cell fixation and permeabilization Cells were either untreated or treated with emodin (50 μmol/L), and the reaction was stopped by removing the medium and adding 40 g/L formaldehyde to PBS. Cells were fixed in PBS for 10 min. Then the fixative was removed, and the cells were washed with PBS. Thereafter the cells were permeabilized by adding the permeabilization solution [(0.1 mL TritonX-100, 90 mL distilled water, and 10 mL PBS (10 ×)] for 10 min. After permeabilization, the cells were rinsed 3 times with PBS.
Labeling with PKCα antibody and confocal microscopy Cells were incubated for 1 h with normal goat serum, followed by 3 washes in PBS with gentle agitation. Subsequently, the cells were incubated for 1 h with the primary antibody, a rabbit anti-PKCα IgG followed by 3 washes with PBS with gentle agitation. This step was followed by incubation of the cells for 1 h with secondary antibody (goat anti-rabbit IgG FITC). Then the cells were washed 3 times with PBS with gentle agitation. Finally, the cells on the coverslip were mounted onto a slide. Control slides were made by incubating cells with the secondary antibody only. The excitation parameter for fluorescent probes was as follows: FITC excitation at 488 nm and emission at 520 nm. Immunostained cells were visualized with an Olympus × 40 objective by confocal laser scanning microscopy (Bio-Rad radiance 2000). Image restoration and analysis were performed in Lasersharp2000 and Laserpix.
The data are presented as mean ± SD, and n represents the number of experiments. Statistical analysis was made by Student’s unpaired t test when applicable. P values less than 0.05 were considered to be significant.
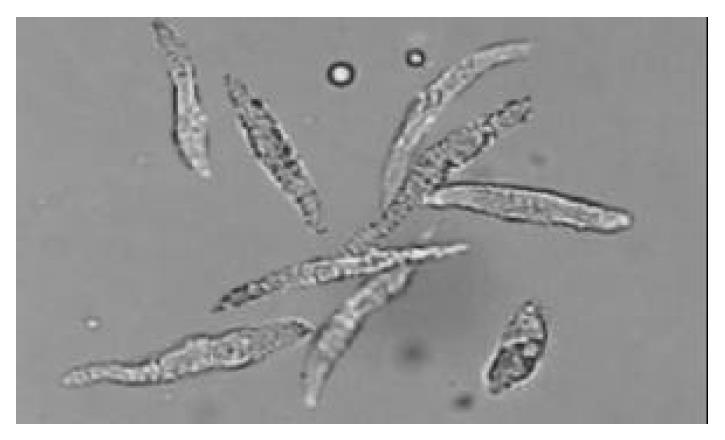
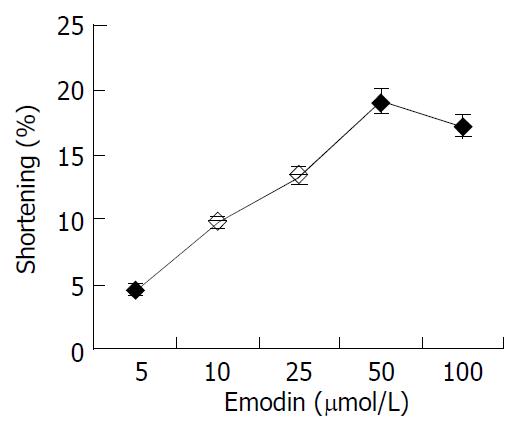
Freshly isolated colonic smooth muscle cells appeared in spindle shape with diverse length (range from 44-121 μm). Some of them were relaxed while others were at different phases of contraction (Figure 1). In resting state, the average cell length was 84.26 μm. The application of emodin to freshly isolated smooth muscle cells induced a reduction in cell length. This reduction in cell length reflected contraction of the smooth muscle cells. Emodin at concentrations of 5 to 100 μmol/L induced a concentration -dependent contraction (Figure 2). Maximal contraction of 19.17% ± 2.59% was observed with 50 μmol/L of emodin.
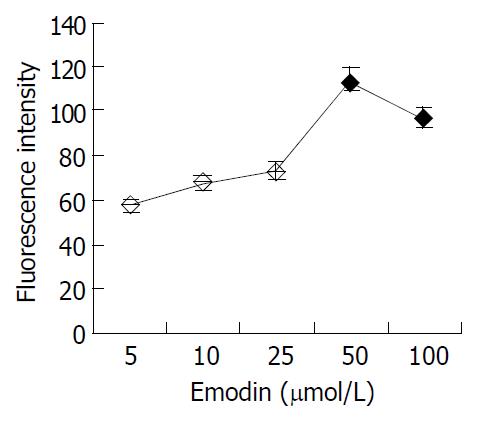
Basal [Ca2+]i levels were not significantly different between cells and the relative fluorescence intensity ranged from 38.46 to 52.59 (46.56 ± 3.79). At each concentration of emodin, the [Ca2+]i response of 20 cells was determined. The application of emodin at concentrations from 5 to 100 μmol/L could trigger an increase in [Ca2+]i. The [Ca2+]i responses to varying emodin concentrations are shown in Figure 3.
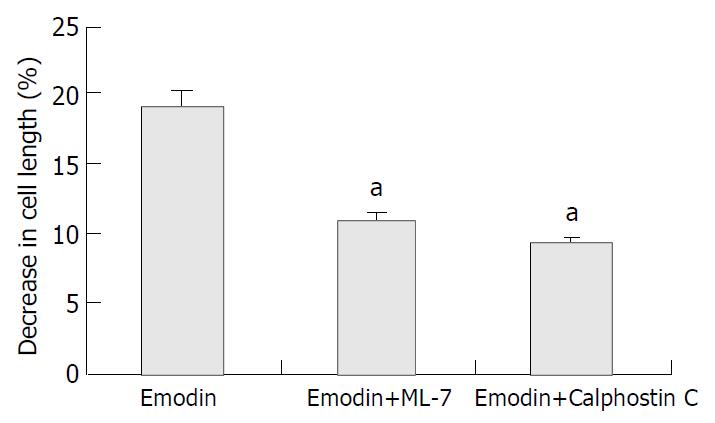
Freshly isolated smooth muscle cells were treated with emodin (50 μmol/L) for 1 min with or without prior incubation with ML-7 (an inhibitor of MLCK, 10-5mol/L) or Calphostin C (an inhibitor of PKC,10-6 mol/L). The application of 50 μmol/L emodin to freshly isolated colonic smooth muscle cells induced a reduction in cell length (19.17% ± 2.59%). And 42.93% ± 6.16% of the contractile response induced by emodin was inhibited by preincubation of the cells with ML-7 (P < 0.05). Similarly, 51.12% ± 3.87% of the contractile response induced by emodin was inhibited by Calphostin C (P < 0.05). (Figure 4).
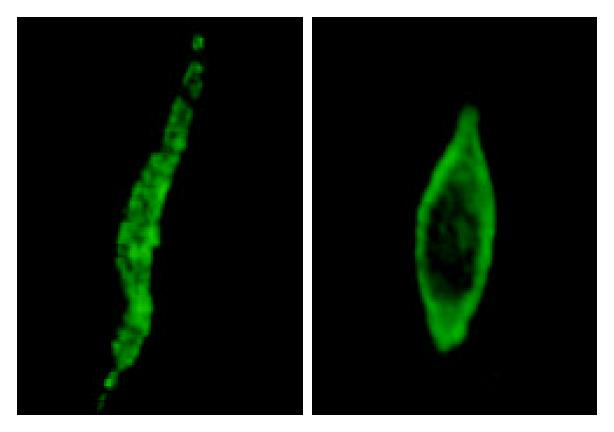
PKCα is thought to be inactive in the cytoplasm and therefore must translocate to the plasma membrane to be activated. Recent studies have demonstrated that PKCα can translocate from the cytoplasm to membrane on stimulation by contractile agonists during smooth muscle cell contraction[18-21]. To determine whether activation of PKCα is involved in contraction of smooth muscle cells induced by emodin, we performed immunofluorescent labeling of PKCα in isolated smooth muscle cells, followed by confocal microscopy. Our data indicate that, at resting state, PKCα is mainly distributed throughout the cytoplasm. This is in contrast to the distribution of PKCα following emodin stimulation, as shown in Figure 5, PKCα translocated from the cytosolic part and aligned itself along the membranes of contracted cells.
Previous studies have indicated that emodin possesses prokinetic effect on gastrointestinal tract[10-12]. Using confocal microscopy and cell isolation technique, the present study focused on emodin’s cellular effect on smooth muscle cells from rat colon in vitro and investigated the signal mechanisms underlying its effect.
Our study showed that some of the action of emodin on gastrointestinal tract is exerted on the smooth muscle cells, as application of emodin to colonic smooth muscle cells resulted in a decrease in cell length. To our knowledge, this is the first study to show a direct contractile effect of emodin on gastrointestinal smooth muscle cells.
It is well established that Ca2+/calmodulin-mediated phosphorylation of the light chain of smooth muscle myosin is a major regulatory mechanism for smooth muscle contraction[22-26]. Stimulation of smooth muscle cells by specific agonists induces Ca2+ mobilization and activation of myosin light chain (MLC) kinase, which subsequently phosphorylates MLC and activates the myosin ATPase. The cascade of events described above leads to contraction of smooth muscle. Yang et al[12] have demonstrated that emodin could evoke increases in [Ca2+]i in guinea-pigs taenia coli cells. In the present study, we observed the changes in [Ca2+]i induced by emodin in single smooth muscle cell by using confocal microscopy and fluo-3 loading technique. Our data showed that, when applied to isolated smooth muscle cells, emodin induced a significant increase in [Ca2+]i. The effect of emodin on [Ca2+]i indicated that emodin-mediated contractions were Ca2+-dependent and MLCK pathways might be involved in emodin-induced contractions. We then carried out contraction studies in which we tested the effect of ML-7, a MLCK inhibitor, on emodin-induced contraction. As shown in Figure 4, the emodin -mediated contraction was significantly attenuated by ML-7. Taken together, these observations suggested that incubation of smooth muscle cells with emodin seemed to trigger a cascade of events including increase in [Ca2+]i, activation of MLCK and then cell contraction.
However, only 42.93% ± 6.16% of the contractile effect elicited by emodin was inhibited by ML-7. Therefore, it is possible that the rest of the emodin-induced contraction is mediated through an alternative pathway different from myosin light chain phosphorylation. Indeed, the rising of [Ca2+]i has been found to be insufficient to explain excitation-contraction of smooth muscle cell, because the cytosolic concentration of Ca2+ is not always proportional to the extent of MLC phosphorylation and the force of contraction in smooth muscle cells. Other regulatory mechanisms have been proposed[22,2-30]. Evidence accumulated that PKCα activation may contribute to the contraction of smooth muscle cells through a kinase cascade involving suppressing the activity of myosin light chain phosphatase(MLCP) and increasing the sensitivity of contractile apparatus to Ca2+. Therefore, we attempted to study the role of PKCα in emodin-induced contraction. The results of our contractile experiments showed that calphostin C, a PKC inhibitor, significantly suppressed the contraction induced by emodin, which suggested that PKCα might play a role in emodin-induced cell contraction. Lee has demonstrated that PKCα plays a critical role in emodin-induced apoptosis in CH27 and H460 cells[7]. In order to verify the role of PKCα in emodin-induced contraction, we next examined the effect of emodin on the activity of PKCα. Previous studies have demonstrated that PKCα is activated by agonist and translocated from the cytoplasm to the plasma membrane through a complex mechanism during smooth muscle cell contraction. Although the exact nature of the translocation is still unclear, it has been suggested that the recruitment of cytosolic PKCα to the membrane is a pivotal component of signal mechanism which mediates the contraction of smooth muscle cell[31-33]. In our study, it was shown that PKCα distribution at rest appeared homogeneous throughout the cell, which is in agreement with previous studies. After stimulation of colonic smooth muscle cells with emodin, PKCα underwent a distinct translocation to the membrane. These data above strongly suggest that emodin may exert its contractile effects on smooth muscle cells via PKCα activation.
On the basis of our findings, we propose the existence of an intracellular signaling cascade leading to cell contraction in colonic smooth muscle induced by emodin. This cascade is initiated by increased [Ca2+]i and PKCα translocation, which in turn lead to the activation of MLCK and the suppression of MLCP, both of which contribute to the emodin-induced contraction. And these results provide evidence for an excitatory role of emodin in colonic smooth muscle cells contraction and indicate that emodin may be a promising prokinetic agent in ameliorating colonic hypomotility.
| 1. | Liang JW, Hsiu SL, Wu PP, Chao PD. Emodin pharmacokinetics in rabbits. Planta Med. 1995;61:406-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 2. | Xie WL, Lin XZ, Ma DL, Li H. Effect of DACHENGQITANG on phosphodiesterase in enteral smooth muscle cell in rats. Chin Traditional Herbal Drug. 2001;4:339-341. |
| 3. | Chen H, Wu X, Guan F. [Protective effects of tongli gongxia herbs on gut barrier in rat with multiple organ dysfunction syndrome]. Zhongguo Zhongxiyi Jiehe Zazhi. 2000;20:120-122. [PubMed] |
| 4. | You SY, Wu XZ, Liu ML. [Effects of dachengqi decoction on gut hormones and intestinal movement after cholecystectomy]. Zhongguo Zhongxiyi Jiehe Zazhi. 1994;14:522-524. [PubMed] |
| 5. | Xia Q, Jiang JM, Gong X, Chen GY, Li L, Huang ZW. Experimental study of Tong Xia purgative method in ameliorating lung injury in acute necrotizing pancreatitis. World J Gastroenterol. 2000;6:115-118. [PubMed] |
| 6. | Lee HZ. Effects and mechanisms of emodin on cell death in human lung squamous cell carcinoma. Br J Pharmacol. 2001;134:11-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 70] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Lee HZ. Protein kinase C involvement in aloe-emodin- and emodin-induced apoptosis in lung carcinoma cell. Br J Pharmacol. 2001;134:1093-1103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 82] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 8. | Srinivas G, Anto RJ, Srinivas P, Vidhyalakshmi S, Senan VP, Karunagaran D. Emodin induces apoptosis of human cervical cancer cells through poly(ADP-ribose) polymerase cleavage and activation of caspase-9. Eur J Pharmacol. 2003;473:117-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 136] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 9. | Kuo YC, Meng HC, Tsai WJ. Regulation of cell proliferation, inflammatory cytokine production and calcium mobilization in primary human T lymphocytes by emodin from Polygonum hypoleucum Ohwi. Inflamm Res. 2001;50:73-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 100] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Jin ZH, Ma DL, Lin XZ. [Study on effect of emodin on the isolated intestinal smooth muscle of guinea-pigs]. Zhongguo Zhongxiyi Jiehe Zazhi. 1994;14:429-431. [PubMed] |
| 11. | Li J, Yang W, Hu W, Wang J, Jin Z, Wang X, Xu W. [Effects of emodin on the activity of K channel in guinea pig taenia coli smooth muscle cells]. Yaoxue Xuebao. 1998;33:321-325. [PubMed] |
| 12. | Yang WX, Wang J, Li JY. Characteristics of emodin evoked [Ca2 ] i and inhibition of GDP in guinea pig taenia coli cells. Acta Biophysica Sinca. 2001;1:165-169. |
| 13. | Wang P, Bitar KN. Rho A regulates sustained smooth muscle contraction through cytoskeletal reorganization of HSP27. Am J Physiol. 1998;275:G1454-G1462. [PubMed] |
| 14. | Fan J, Byron KL. Ca2+ signalling in rat vascular smooth muscle cells: a role for protein kinase C at physiological vasoconstrictor concentrations of vasopressin. J Physiol. 2000;524 Pt 3:821-831. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Taggart MJ, Lee YH, Morgan KG. Cellular redistribution of PKCalpha, rhoA, and ROKalpha following smooth muscle agonist stimulation. Exp Cell Res. 1999;251:92-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 66] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Ibitayo AI, Sladick J, Tuteja S, Louis-Jacques O, Yamada H, Groblewski G, Welsh M, Bitar KN. HSP27 in signal transduction and association with contractile proteins in smooth muscle cells. Am J Physiol. 1999;277:G445-G454. [PubMed] |
| 17. | Claing A, Shbaklo H, Plante M, Bkaily G, D'Orléans-Juste P. Comparison of the contractile and calcium-increasing properties of platelet-activating factor and endothelin-1 in the rat mesenteric artery and vein. Br J Pharmacol. 2002;135:433-443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 18. | Li L, Eto M, Lee MR, Morita F, Yazawa M, Kitazawa T. Possible involvement of the novel CPI-17 protein in protein kinase C signal transduction of rabbit arterial smooth muscle. J Physiol. 1998;508:871-881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 140] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 19. | Sanders KM. Invited review: mechanisms of calcium handling in smooth muscles. J Appl Physiol (1985). 2001;91:1438-1449. [PubMed] |
| 20. | Cao W, Sohn UD, Bitar KN, Behar J, Biancani P, Harnett KM. MAPK mediates PKC-dependent contraction of cat esophageal and lower esophageal sphincter circular smooth muscle. Am J Physiol Gastrointest Liver Physiol. 2003;285:G86-G95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 47] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Ratz PH, Meehl JT, Eddinger TJ. RhoA kinase and protein kinase C participate in regulation of rabbit stomach fundus smooth muscle contraction. Br J Pharmacol. 2002;137:983-992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Somlyo AP, Somlyo AV. Signal transduction and regulation in smooth muscle. Nature. 1994;372:231-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1550] [Cited by in RCA: 1420] [Article Influence: 44.4] [Reference Citation Analysis (0)] |
| 23. | Vorotnikov AV, Krymsky MA, Shirinsky VP. Signal transduction and protein phosphorylation in smooth muscle contraction. Biochemistry (Mosc). 2002;67:1309-1328. [PubMed] |
| 24. | Makhlouf GM, Murthy KS. Signal transduction in gastrointestinal smooth muscle. Cell Signal. 1997;9:269-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 90] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Bolton TB, Prestwich SA, Zholos AV, Gordienko DV. Excitation-contraction coupling in gastrointestinal and other smooth muscles. Annu Rev Physiol. 1999;61:85-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 178] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 26. | Harnett KM, Biancani P. Calcium-dependent and calcium-independent contractions in smooth muscles. Am J Med. 2003;115 Suppl 3A:24S-30S. [PubMed] |
| 27. | Kitazawa T, Eto M, Woodsome TP, Brautigan DL. Agonists trigger G protein-mediated activation of the CPI-17 inhibitor phosphoprotein of myosin light chain phosphatase to enhance vascular smooth muscle contractility. J Biol Chem. 2000;275:9897-9900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 255] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 28. | Sato A, Hattori Y, Sasaki M, Tomita F, Kohya T, Kitabatake A, Kanno M. Agonist-dependent difference in the mechanisms involved in Ca2+ sensitization of smooth muscle of porcine coronary artery. J Cardiovasc Pharmacol. 2000;35:814-821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 29. | Yamada A, Ohya S, Hirano M, Watanabe M, Walsh MP, Imaizumi Y. Ca2+ sensitization of smooth muscle contractility induced by ruthenium red. Am J Physiol. 1999;276:C566-C575. [PubMed] |
| 30. | Gokina NI, Osol G. Temperature and protein kinase C modulate myofilament Ca2+ sensitivity in pressurized rat cerebral arteries. Am J Physiol. 1998;274:H1920-H1927. [PubMed] |
| 31. | Bitar KN. HSP27 phosphorylation and interaction with actin-myosin in smooth muscle contraction. Am J Physiol Gastrointest Liver Physiol. 2002;282:G894-G903. [PubMed] |
| 32. | Li C, Fultz ME, Wright GL. PKC-alpha shows variable patterns of translocation in response to different stimulatory agents. Acta Physiol Scand. 2002;174:237-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 33. | Taggart MJ, Leavis P, Feron O, Morgan KG. Inhibition of PKCalpha and rhoA translocation in differentiated smooth muscle by a caveolin scaffolding domain peptide. Exp Cell Res. 2000;258:72-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
Edited by Zhu LH and Xu FM