Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 28, 2026; 32(16): 116781
Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.116781
Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.116781
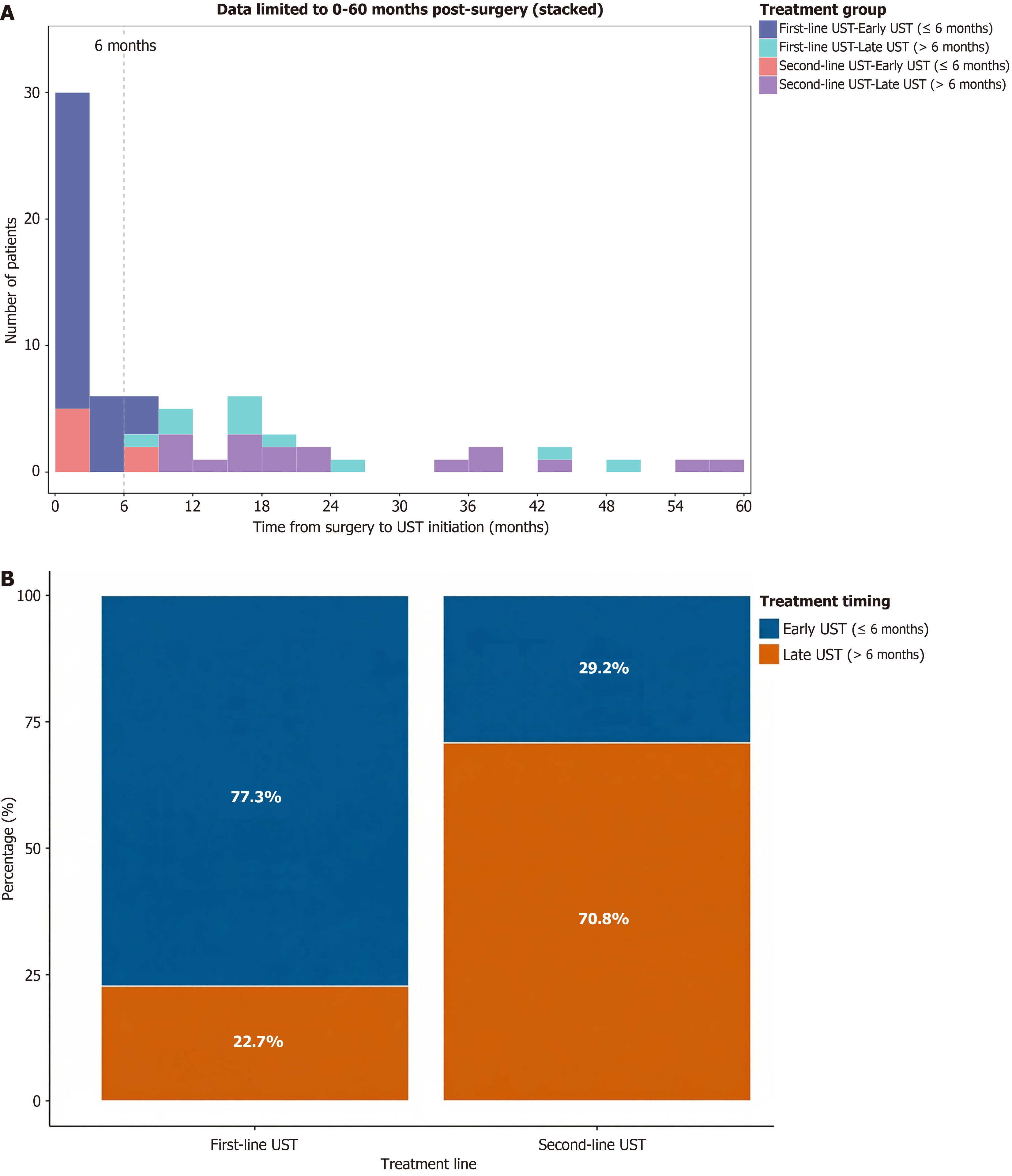
Figure 1 Timing of ustekinumab initiation after surgery by treatment line.
A: The distribution highlights the significantly shorter median time from surgery to ustekinumab (UST) initiation in the first-line treatment group compared to that of the second-line group; B: Bar chart showing the proportion of patients receiving early (≤ 6 months) vs late (> 6 months) UST initiation after intestinal resection, stratified by the treatment line (first-line vs second-line UST). UST: Ustekinumab.
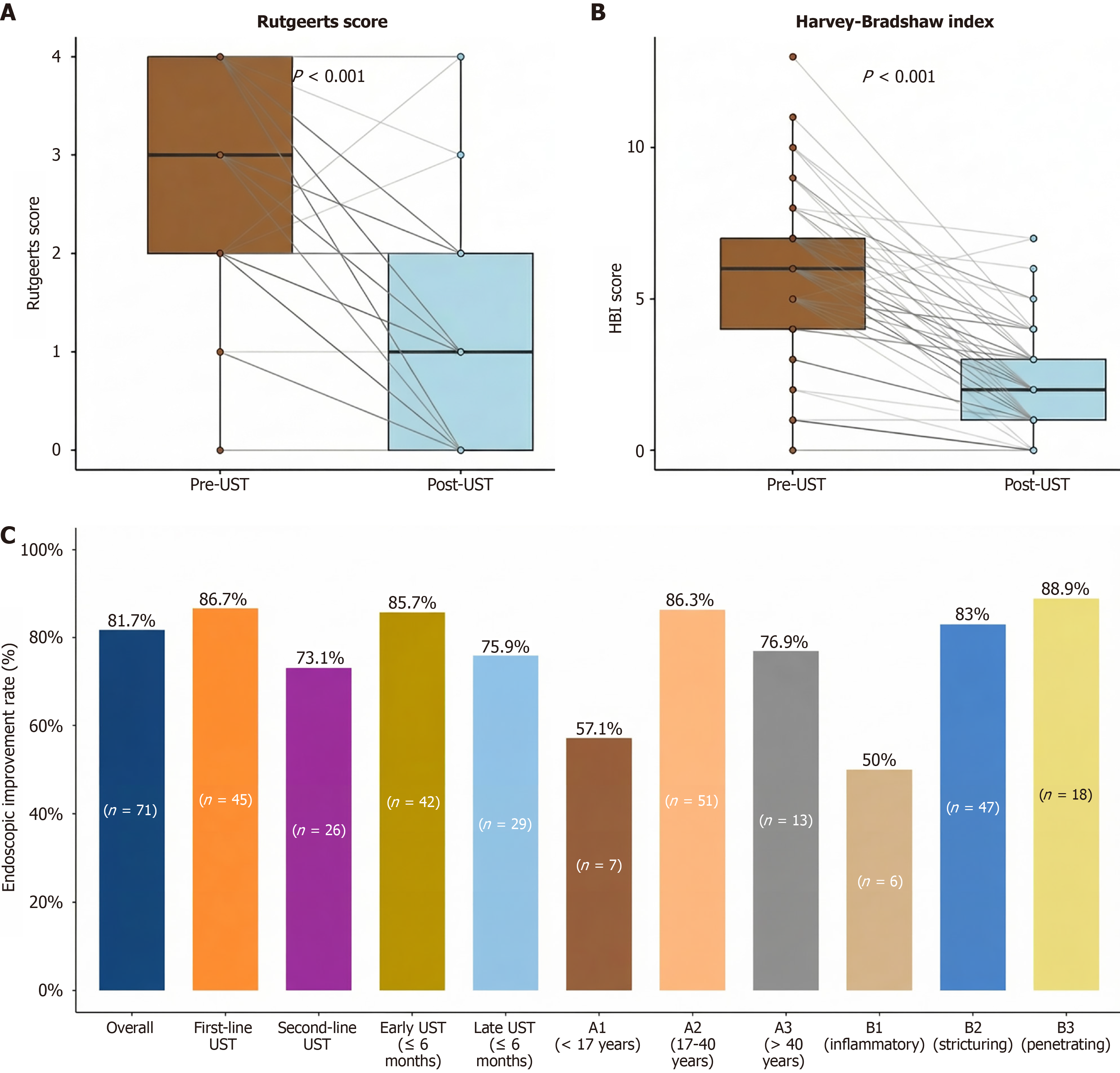
Figure 2 Changes in Rutgeerts score, Harvey-Bradshaw Index, and endoscopic improvement by subgroup following ustekinumab treatment.
A and B: Box plots that demonstrated the significant reduction in both endoscopic Rutgeerts scores and clinical Harvey-Bradshaw index (HBI) scores following ustekinumab (UST) treatment. The statistical significance was determined using paired analyses (P < 0.001 for Rutgeerts score; P < 0.001 for HBI score); C: Histogram illustrating endoscopic improvement rates (defined as ≥ 1-point reduction in the Rutgeerts score) across key patient subgroups, including the treatment line (i.e., first-line vs second-line UST) and disease characteristics based on the Montreal classification. The overall endoscopic improvement rate was 81.7% (58/71 patients). Together, these panels indicate substantial improvement in endoscopic and clinical disease activity following UST treatment, with differential responses observed across the patient subgroups. UST: Ustekinumab.
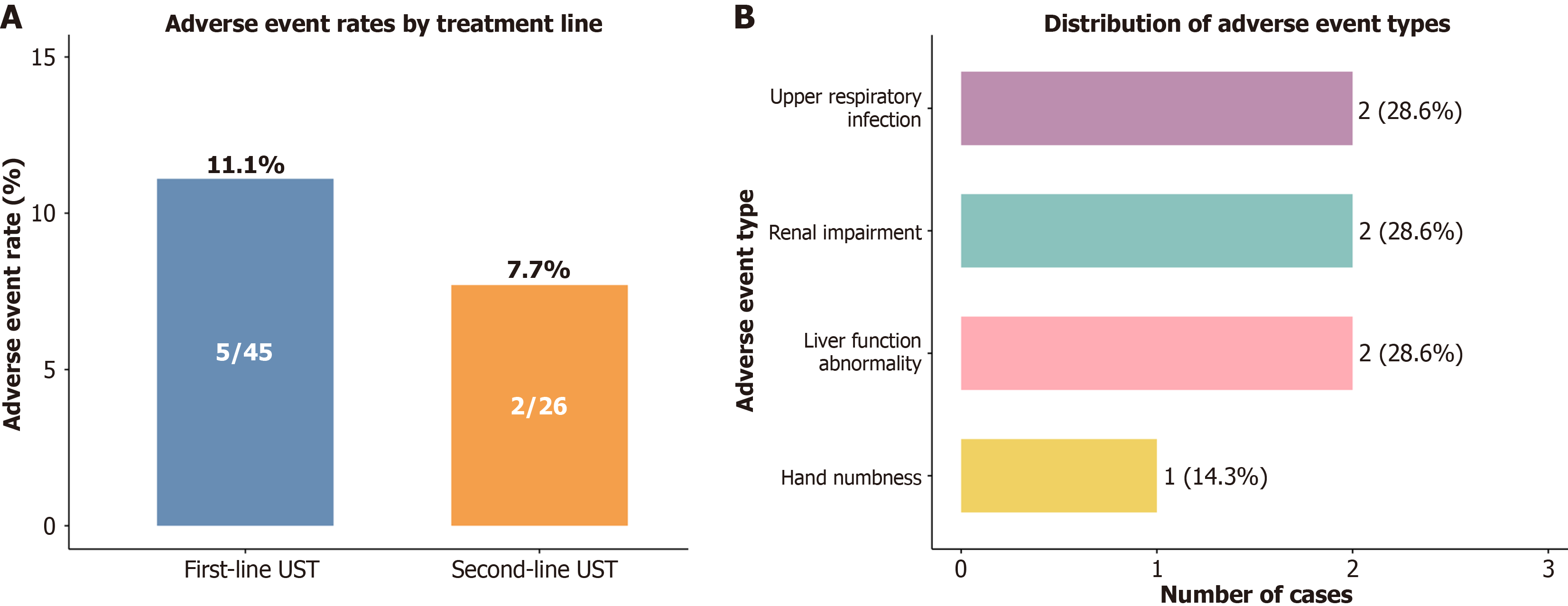
Figure 3 Ustekinumab safety profile in postoperative Crohn's disease.
A: Bar chart comparing the adverse event (AE) rates between the first-line and second-line ustekinumab treatment groups, showing comparable safety profiles; B: Pie chart detailing the distribution of AE types among the seven reported events, including upper respiratory infection, renal impairment, liver function abnormality, and hand numbness. All of the AEs were mild to moderate in severity. UST: Ustekinumab.
- Citation: Ma XZ, Han XZ, Zhang WY, Tian F, Zhou XC, Zhou LY, Teng YS, Lei L, Sheng JQ, Jin P, Zhao XM, Jia Y. Effectiveness of ustekinumab in postoperative Crohn's disease management: Evidence from a Chinese multicenter cohort. World J Gastroenterol 2026; 32(16): 116781
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/116781.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.116781