Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 7, 2026; 32(13): 117115
Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.117115
Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.117115
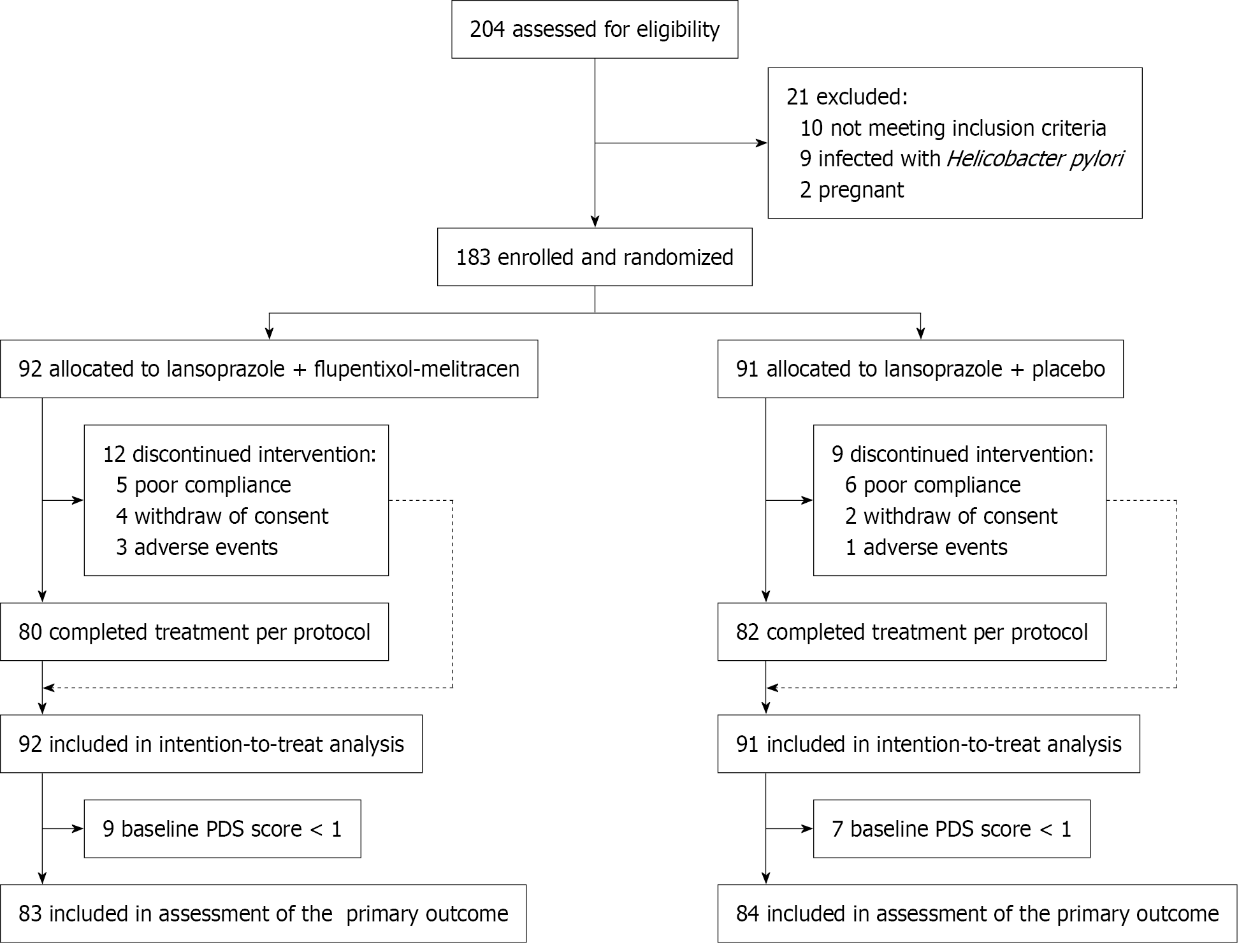
Figure 1 Trial profile.
After excluding ineligible participants, 183 subjects were randomly assigned to the two treatment arms, and 162 completed the treatment course per protocol. 183 were included in the intention-to-treat analysis; 162 were included in the per-protocol analysis. PDS: Postprandial distress syndrome.
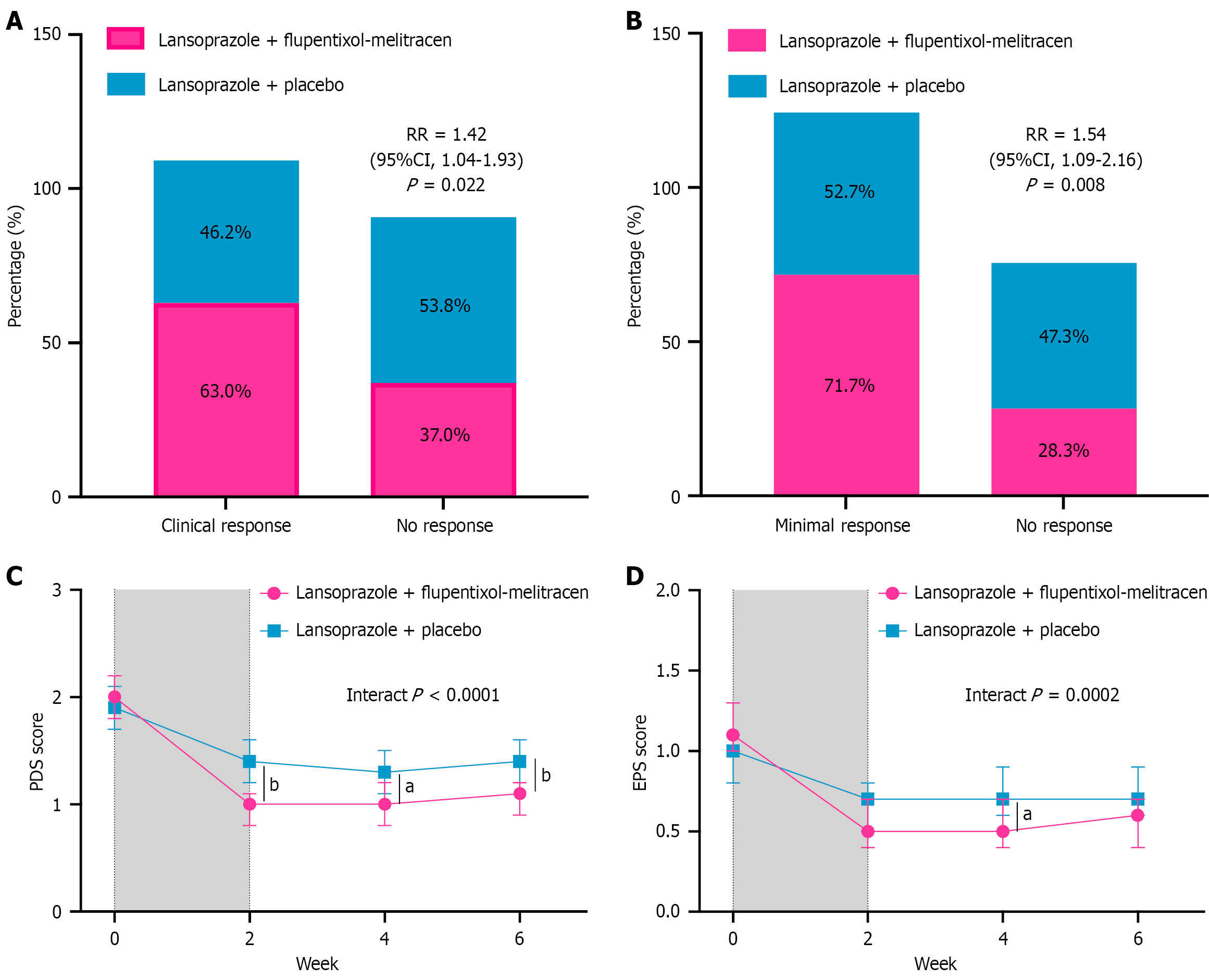
Figure 2 Percentage of treatment response and changes of estimated Leuven Postprandial Distress Scale scores over time between lansoprazole plus flupentixol-melitracen and placebo group.
A and B: Percentage of clinical response and minimal clinical response at the end of treatment. The relative risk, along with its 95% confidence intervals, was calculated to compare the lansoprazole + flupentixol-melitracen group against the lansoprazole + placebo group; C and D: Over-time model-based estimated postprandial distress syndrome score and epigastric pain syndrome score changes. The gray shaded area represents the treatment period. aP < 0.05, and bP < 0.01. RR: Relative risk; CI: Confidence interval; PDS: Postprandial distress syndrome; EPS: Epigastric pain syndrome.
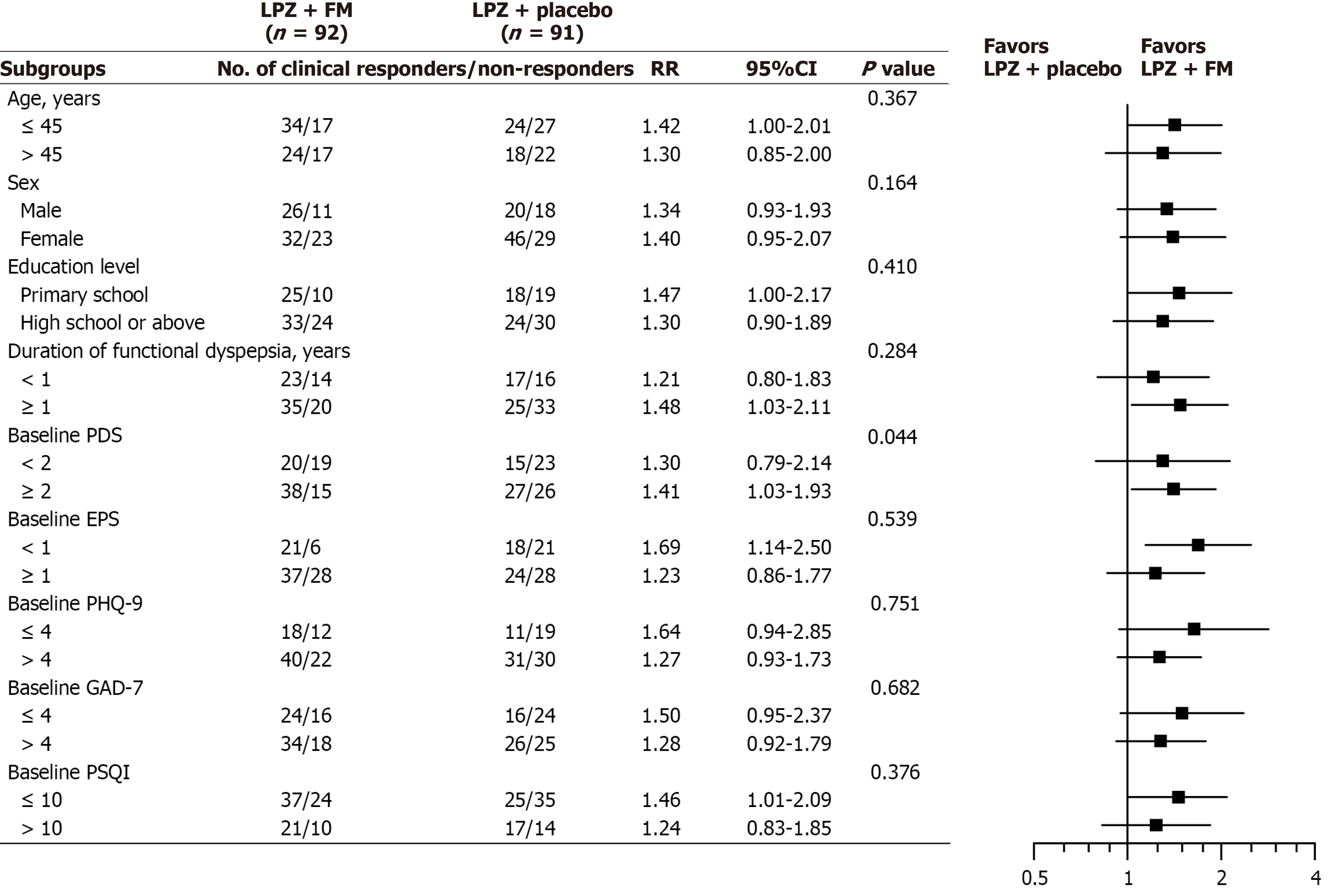
Figure 3 Predefined subgroup analyses for primary endpoint.
Significance is given for interaction effect. LPZ: Lansoprazole; FM: Flupentixol-melitracen; RR: Relative risk; CI: Confidence interval; PDS: Postprandial distress syndrome; EPS: Epigastric pain syndrome; PHQ-9: Patient Health Questionnaire-9; GAD-7: Generalized Anxiety Disorder-7; PSQI: Pittsburgh Sleep Quality Index.
- Citation: Wang XY, Yin KH, Cheng L, Wang XY, Qiao Y, Tang XR, Wang B, Yan XJ, Chen SL. Efficacy and safety of lansoprazole combined with flupentixol-melitracen for functional dyspepsia: A randomized, double-blinded, placebo-controlled clinical trial. World J Gastroenterol 2026; 32(13): 117115
- URL: https://www.wjgnet.com/1007-9327/full/v32/i13/117115.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i13.117115