Copyright: ©Author(s) 2026.
World J Gastroenterol. Mar 28, 2026; 32(12): 116287
Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.116287
Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.116287
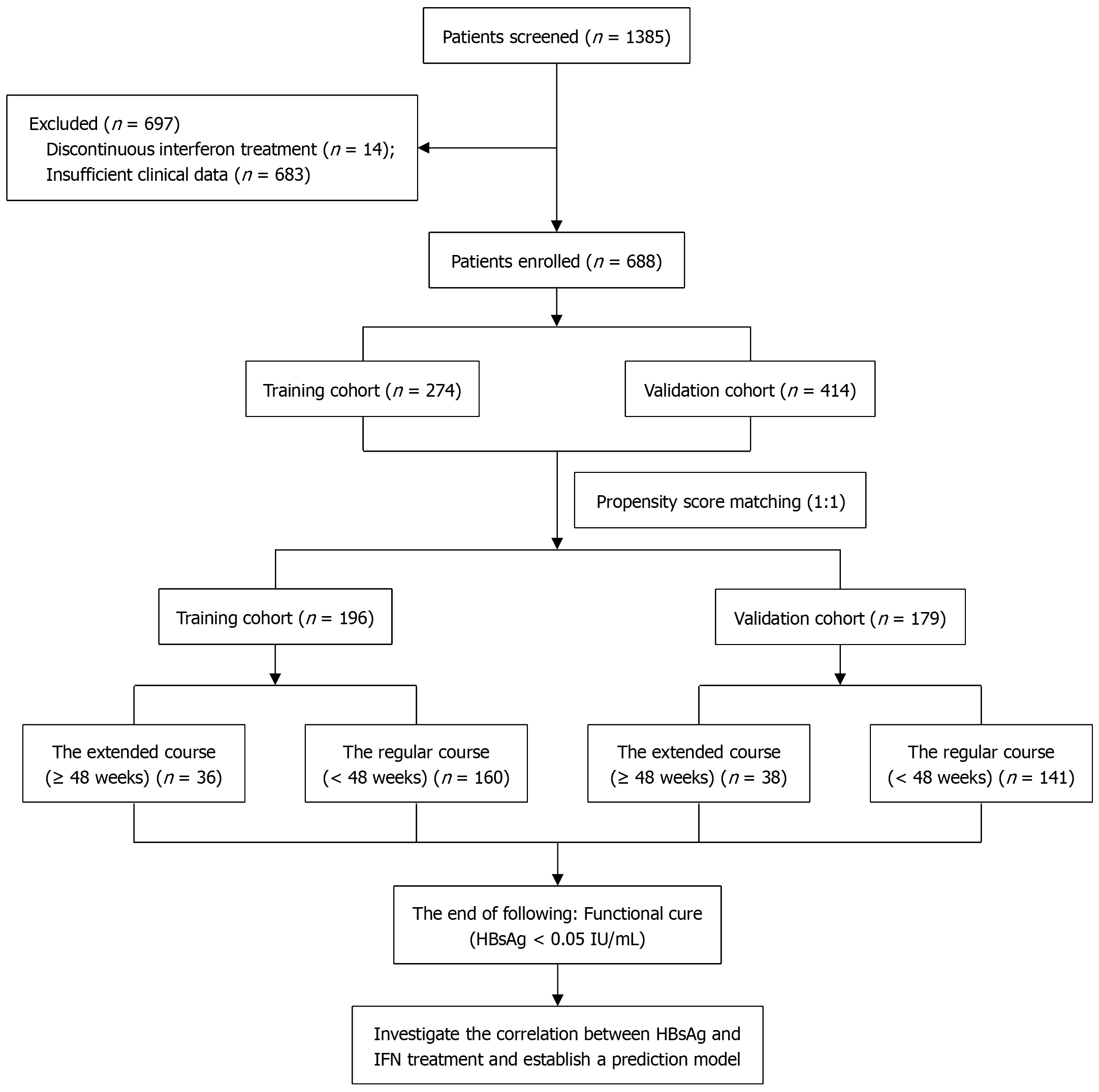
Figure 1 Flow diagram of patients enrolled in this study.
HBsAg: Hepatitis B surface antigen; IFN: Interferon.
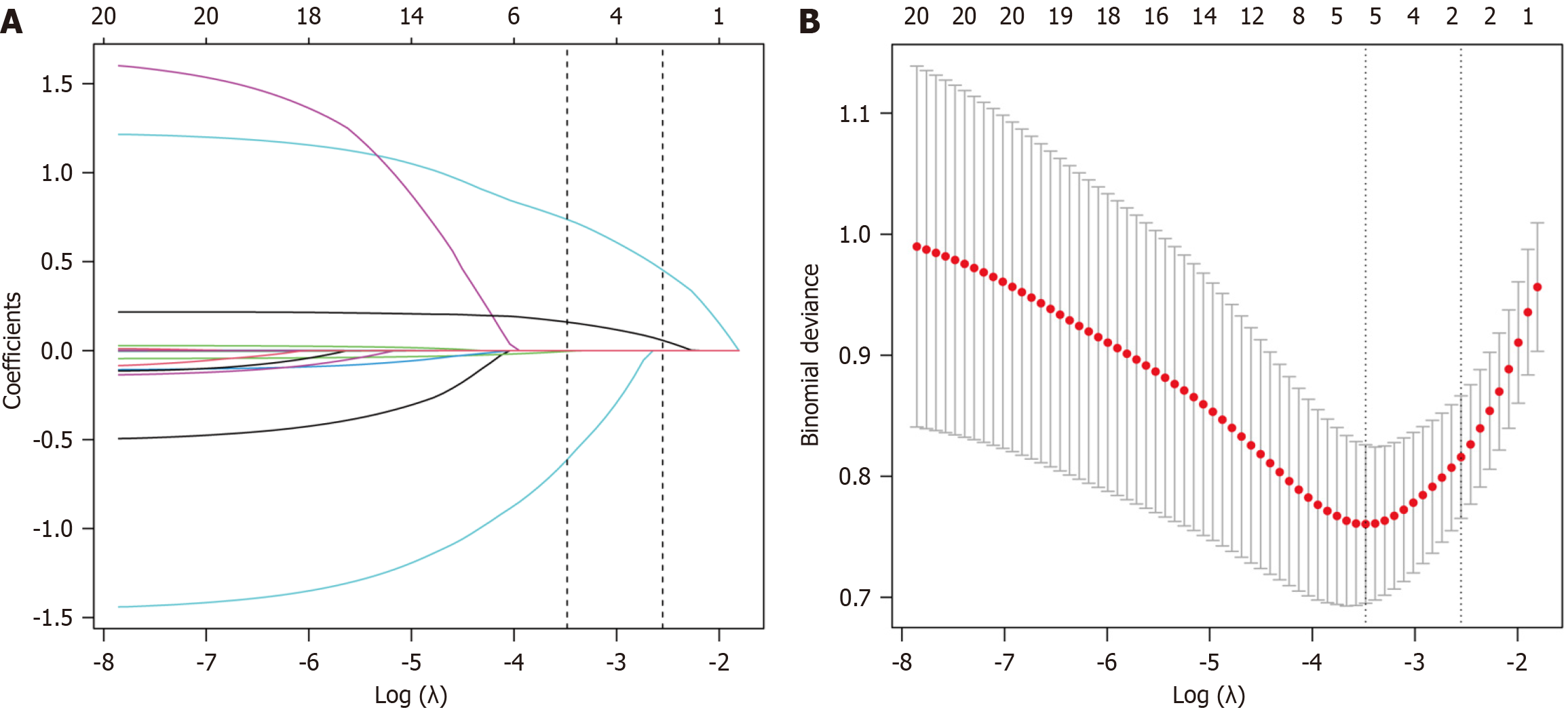
Figure 2 Least absolute shrinkage and selection operator regression-based variable selection.
A: Characteristics of variable coefficient changes; B: Selection process for the optimal value of parameter λ in the least absolute shrinkage and selection operator (LASSO) regression model via cross-validation. At λ = 0.0308 (Log λ = -3.4816), LASSO regression identified four significant variables: Age, baseline hepatitis B surface antigen (HBsAg), 12-week HBsAg, and cirrhosis.
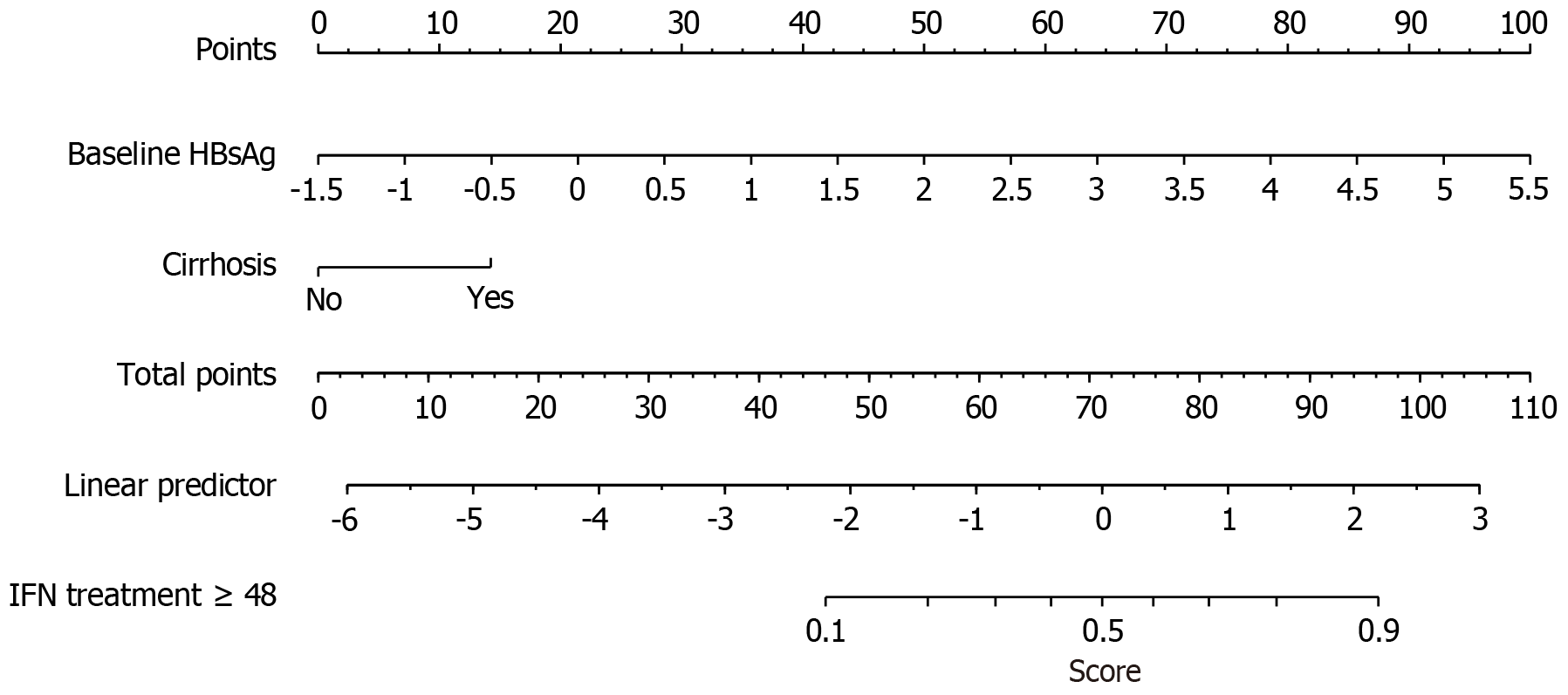
Figure 3 A nomogram for the risk of interferon therapy beyond 48 weeks in patients with chronic hepatitis B.
HBsAg: Hepatitis B surface antigen; IFN: Interferon.
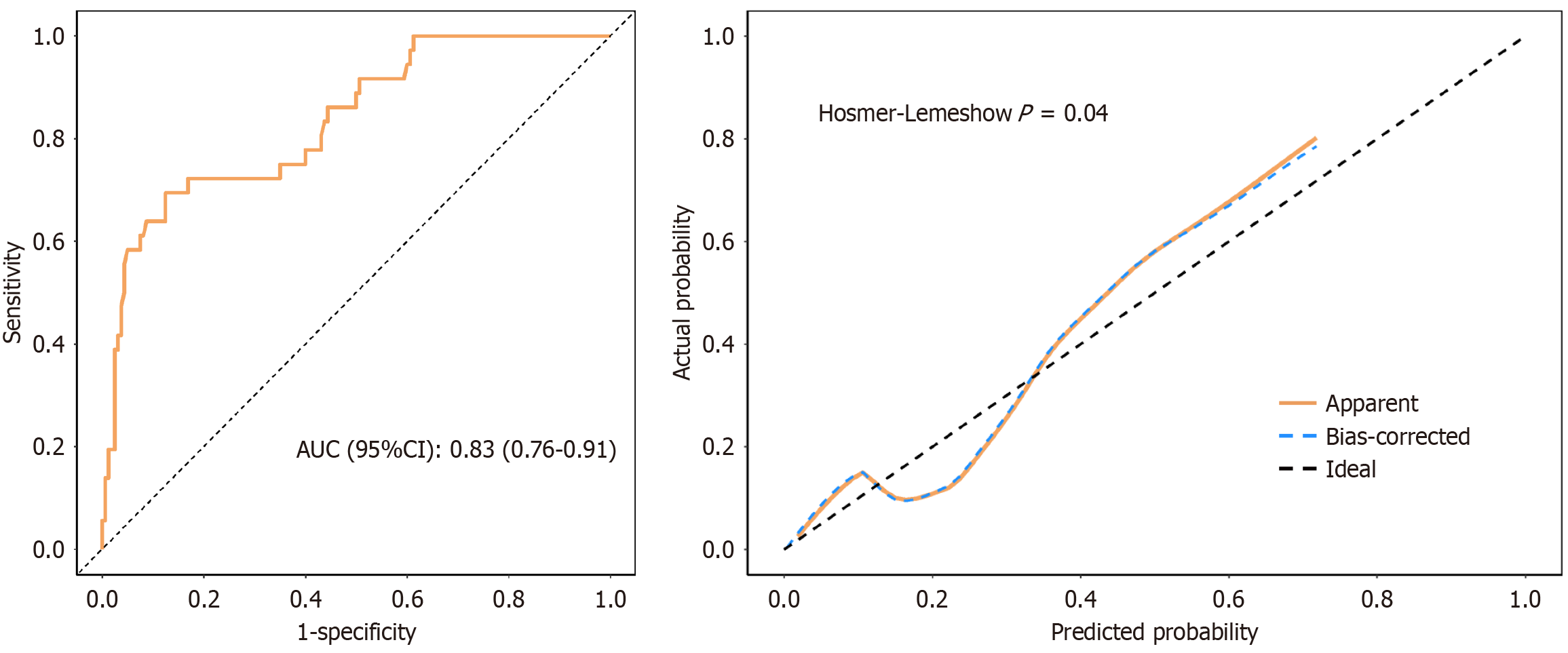
Figure 4 Receiver operating characteristic curve and calibration curve of the training cohort.
The area under the curve was 0.83 (95% confidence interval: 0.76-0.91), with a sensitivity of 0.88 and specificity of 0.69. The result of Hosmer-Lemeshow goodness-of-fit test for the training cohort was χ2 = 16.30, P = 0.04. AUC: Area under the curve; CI: Confidence interval.
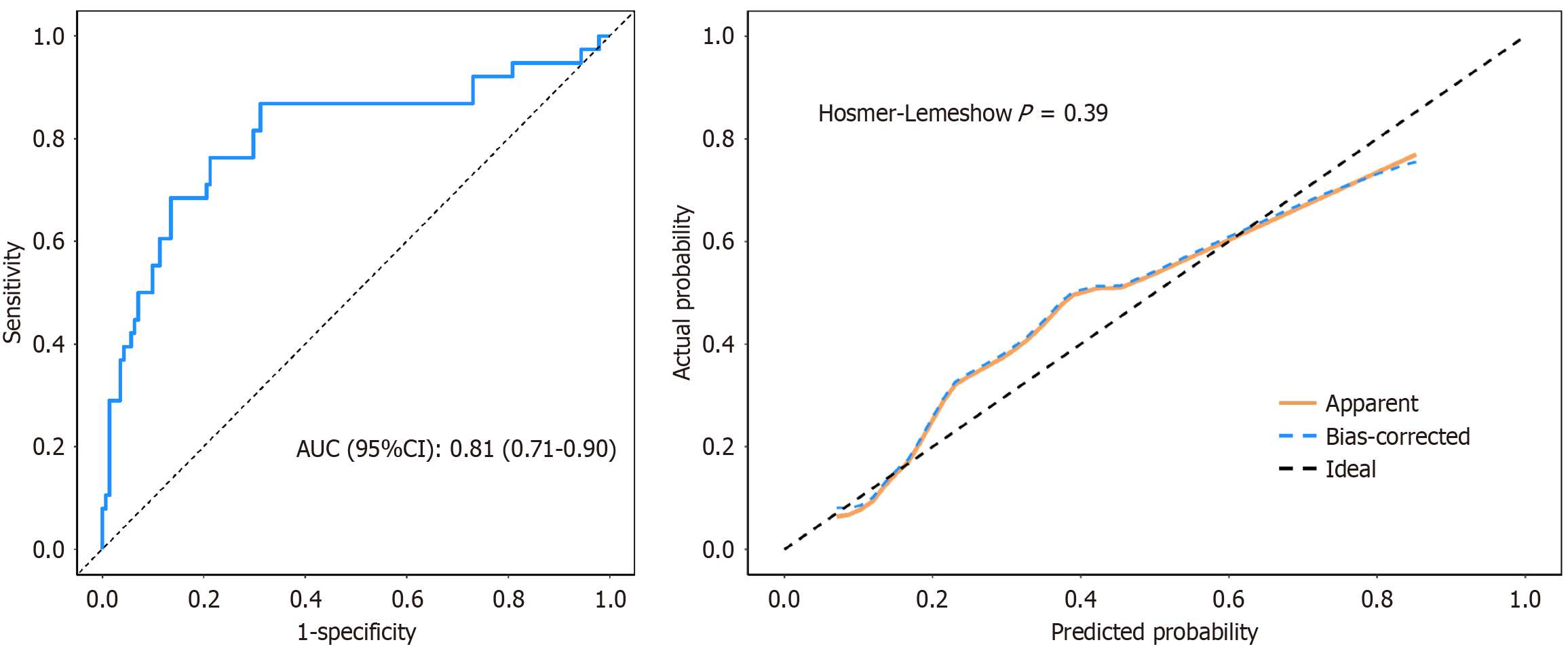
Figure 5 Receiver operating characteristic curve and calibration curve of the validation cohort.
The area under the curve for the external validation cohort was 0.81 (95% confidence interval: 0.71-0.90), and the goodness-of-fit test resulted in χ2 = 8.41 and P = 0.39. AUC: Area under the curve; CI: Confidence interval.
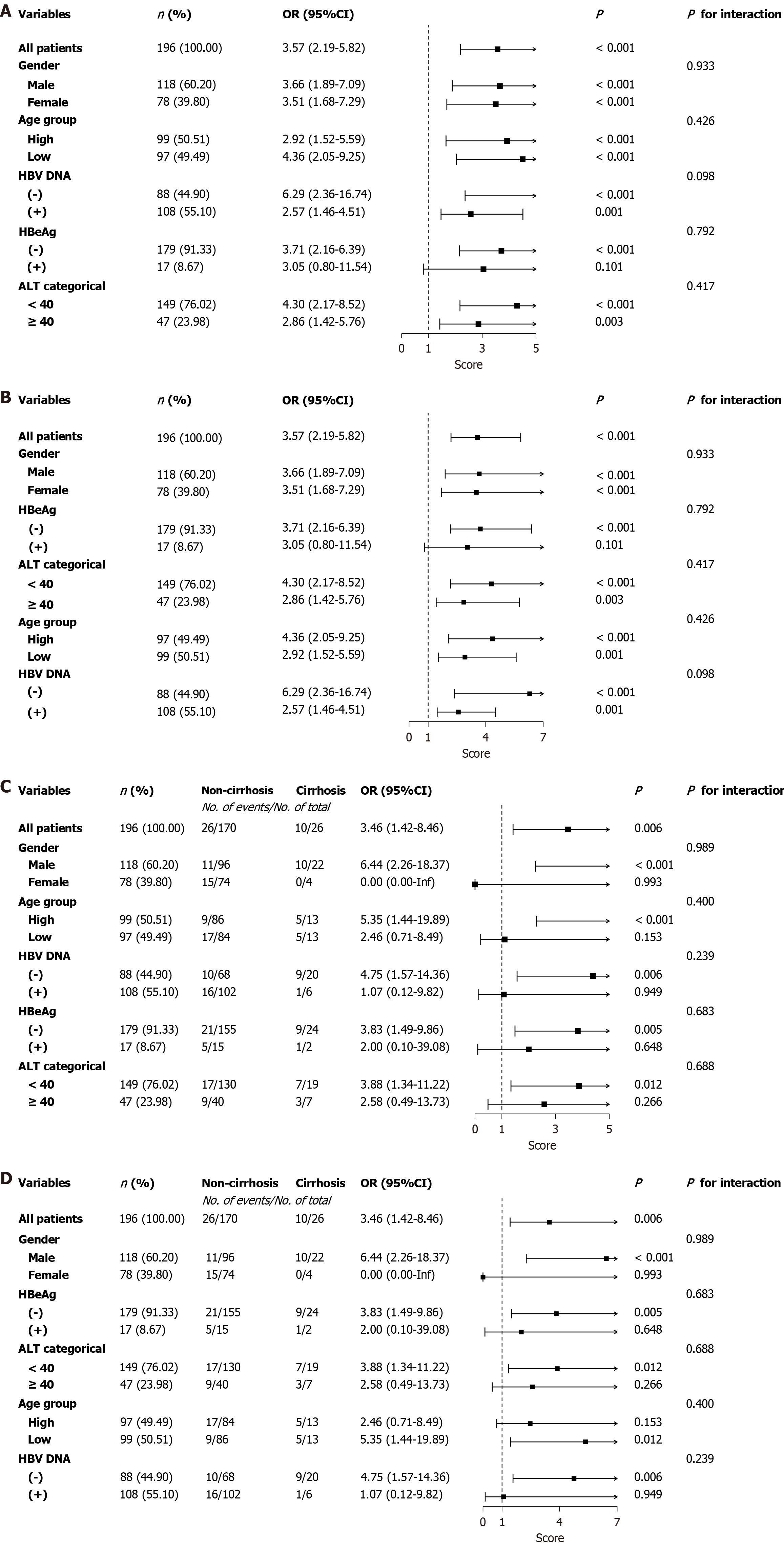
Figure 6 Subgroup analyses stratified by gender, age, baseline hepatitis B envelope antigen, baseline hepatitis B virus DNA, and baseline alanine aminotransferase in the training and validation cohorts.
The relationships of both baseline hepatitis B surface antigen (HBsAg) and liver cirrhosis with extended course interferon (≥ 48 weeks) were consistent with the primary outcome in most subgroups. Furthermore, no significant interactions were observed concerning the association with extended course interferon (≥ 48 weeks). A and B: Baseline HBsAg: Training cohort (A); Validation cohort (B); C and D: Cirrhosis: Training cohort (C); Validation cohort (D). OR: Odds ratio; CI: Confidence interval; HBeAg: Hepatitis B envelope antigen; HBV: Hepatitis B virus; ALT: Alanine aminotransferase.
- Citation: Yan F, Xue XL, Guo Y, Zhang QR, You RR, Shang J, Wu XP, Geng JW, Gao XH, Ye Q, Liang J, Wang XY, Zeng JY, Chen J, Lin YC, Chen XY, Du Q, Yin WL, Liu L, Wang F, Xu BG, Zhang WH, Xiang HL. Baseline hepatitis B surface antigen and cirrhosis predict extended interferon therapy in chronic hepatitis B: A retrospective study. World J Gastroenterol 2026; 32(12): 116287
- URL: https://www.wjgnet.com/1007-9327/full/v32/i12/116287.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i12.116287