©The Author(s) 2023.
World J Gastroenterol. Oct 28, 2023; 29(40): 5593-5617
Published online Oct 28, 2023. doi: 10.3748/wjg.v29.i40.5593
Published online Oct 28, 2023. doi: 10.3748/wjg.v29.i40.5593
Figure 1 Flow chart of literature search and screening.
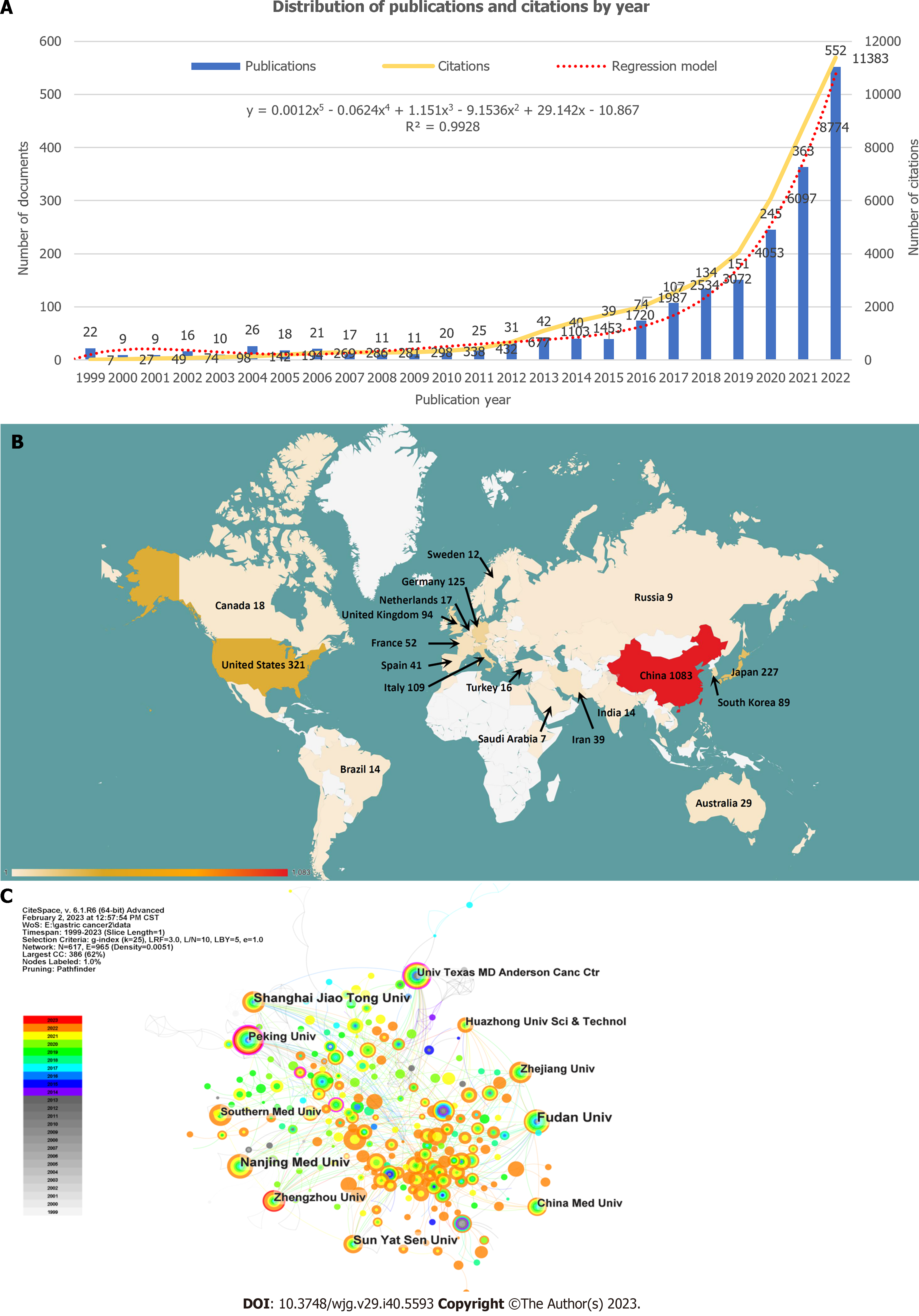
Figure 2 Annual distribution of publications and citations, geographical visualization, and institutions analysis.
A: Distribution of publications and citations by year. The regression formula is y = 0.0012x5 - 0.0624x4 + 1.151x3 - 9.1536x2 + 29.142x - 10.867, R² = 0.9928; B: Geographical visualization of publications for immunotherapy in gastric cancer; C: Co-occurrence map of the research institutions.
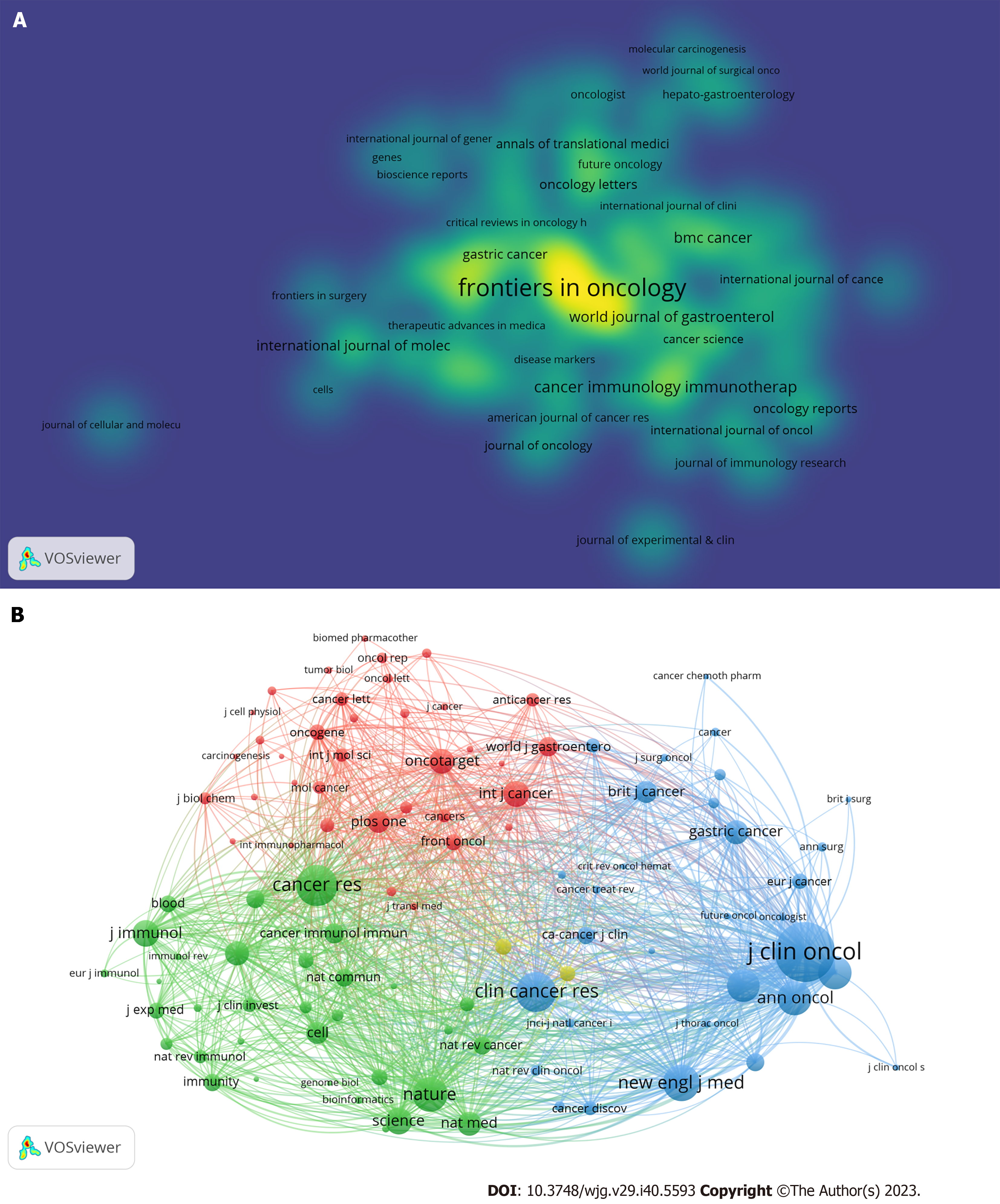
Figure 3 The visualization of journals.
A: Journals with at least five publications in this field; B: Highly cited journals in this field.
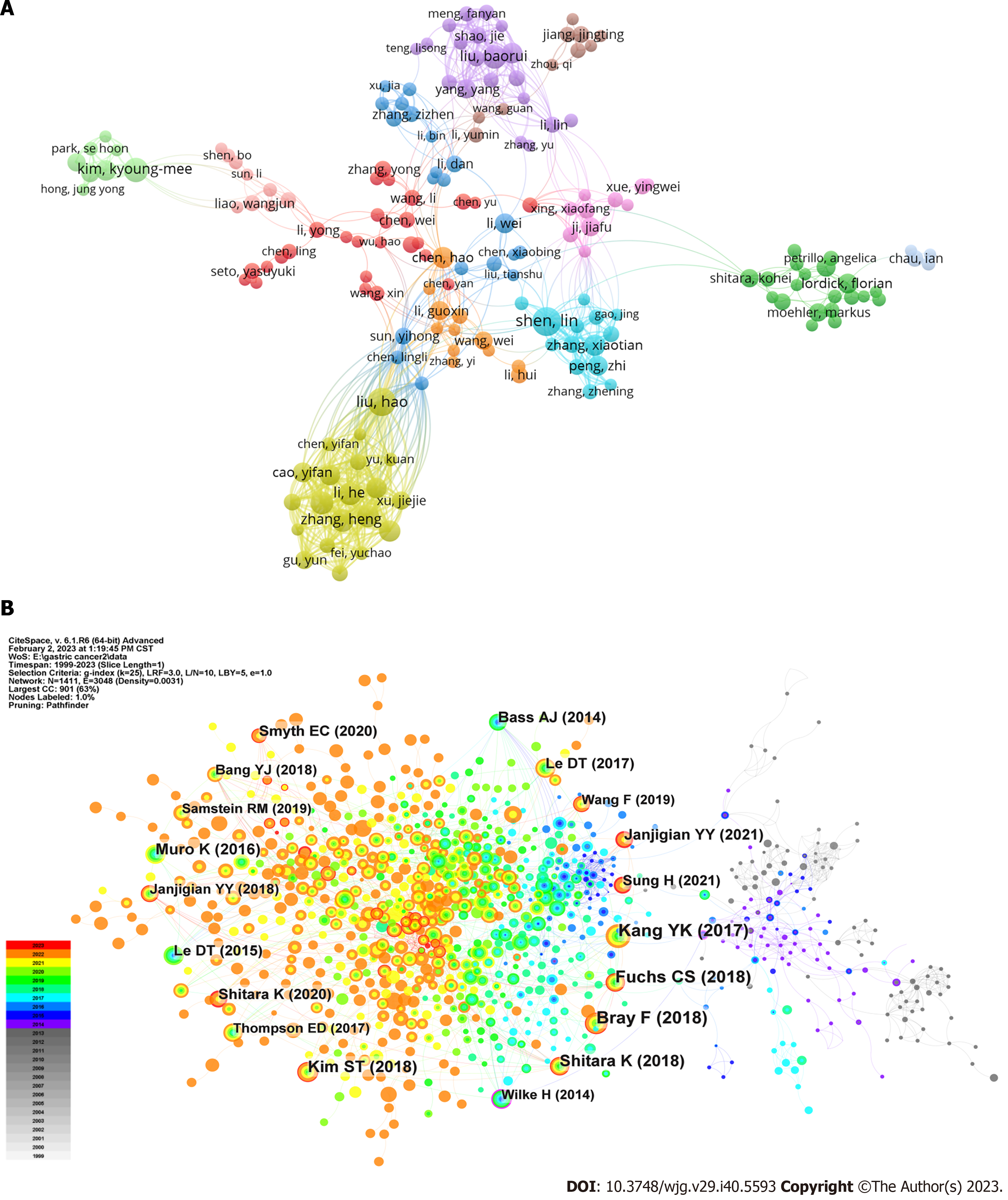
Figure 4 The visualization of authors.
A: Network map of researching authors who had at least five publications; B: Citation analysis of references. Highly cited references are marked by author and publication year.
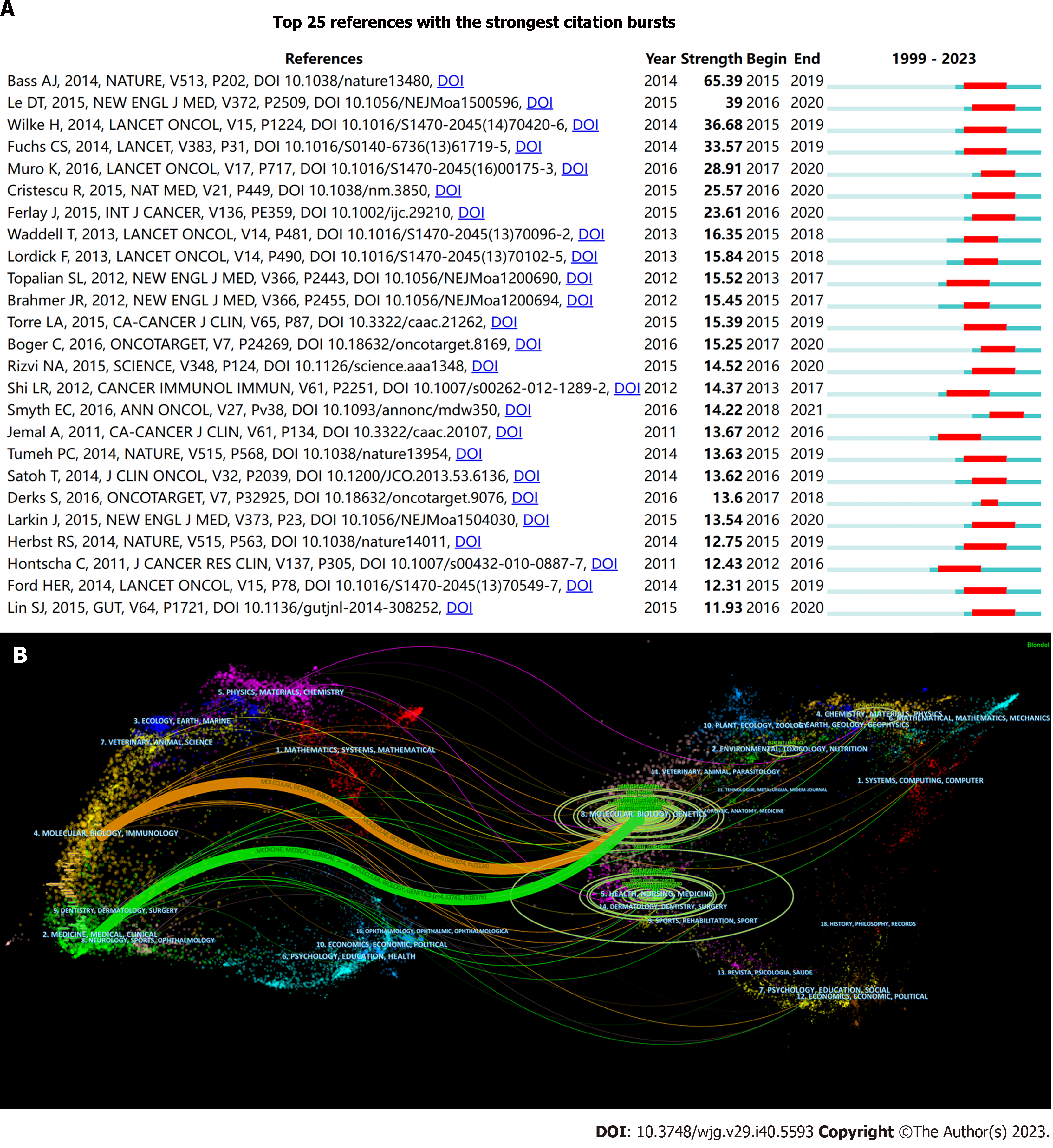
Figure 5 References burst analysis and dual-map overlay of journals.
A: Top 25 references with the strongest citation bursts are sorted by strength; B: Dual-map overlay of journals. The colored curve represents the reference path; the nature of each region is defined by the journal of the corresponding region.
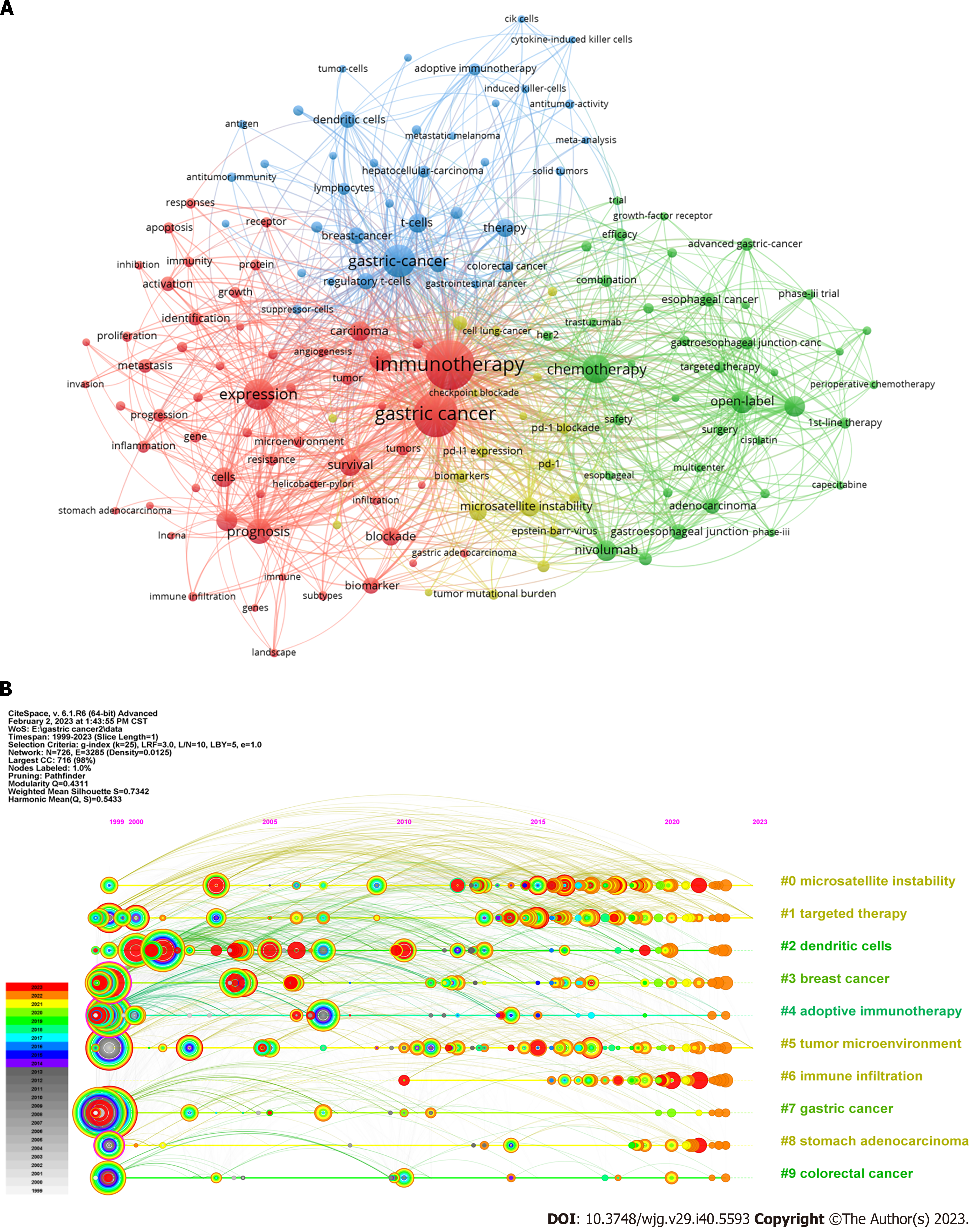
Figure 6 Keywords occurrences and timeline view analysis.
A: Co-occurrence map of the keywords with at least 25 occurrences; B: Timeline view of keywords with the ten leading clusters.
Figure 7 Analysis of keywords burst.
A: Top 25 keywords with the strongest citation bursts are sorted by strength; B: Top 25 keywords with the strongest citation bursts sorted by starting year.
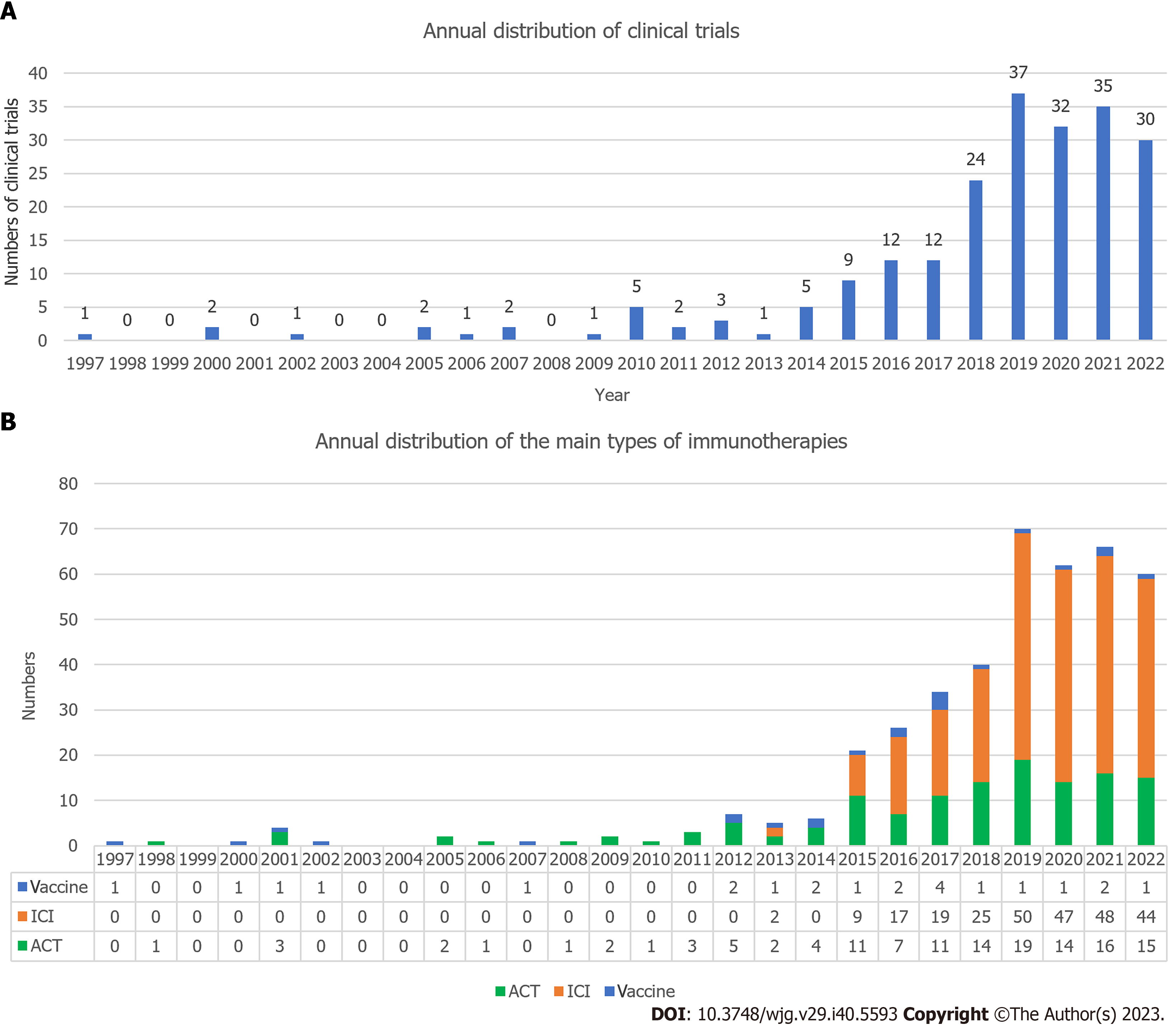
Figure 8 Distribution of clinical trials by year.
A: Annual distribution of clinical trials in this field; B: Annual distribution of the main types of immunotherapies (vaccine, immune checkpoint inhibitor, and adoptive cell therapy). ICI: Immune checkpoint inhibitor; ACT: Adoptive cell therapy.
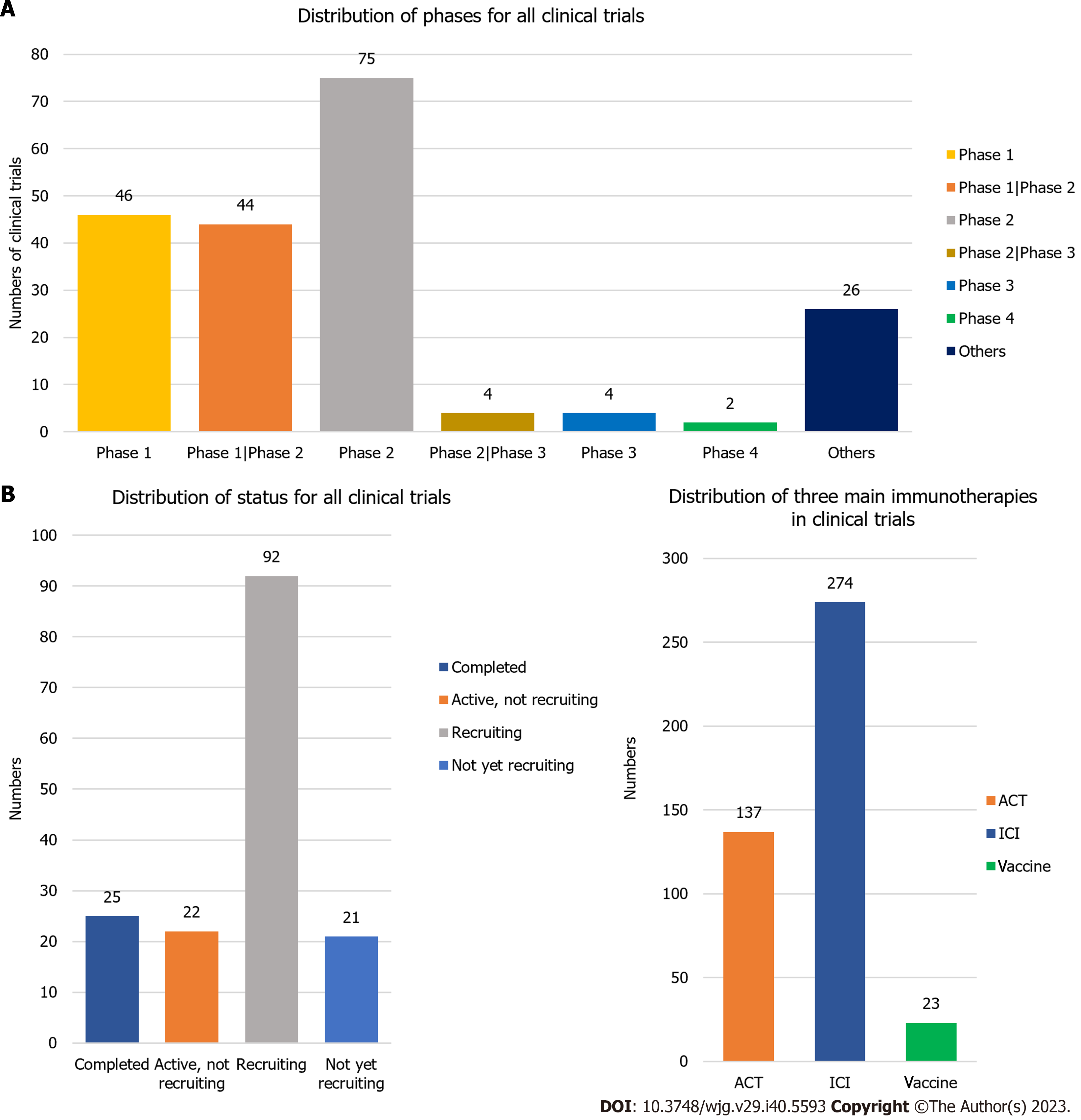
Figure 9 Phases and status distribution across all clinical trials.
A: Distribution of phases for all clinical trials. Phases include: Phase 1, phase 1/phase 2, phase 2, phase 2/phase 3, phase 3, phase 4, and others; B: Distribution of status for all clinical trials. The status includes: Completed, active but not recruiting, recruiting, and not yet recruiting; C: Distribution of adoptive cell therapy, immune checkpoint inhibitor, and vaccine in clinical trials. ICI: Immune checkpoint inhibitor; ACT: Adoptive cell therapy.
- Citation: Li YN, Xie B, Zhang Y, He MH, Xing Y, Mu DM, Wang H, Guo R. Advances and key focus areas in gastric cancer immunotherapy: A comprehensive scientometric and clinical trial review (1999-2023). World J Gastroenterol 2023; 29(40): 5593-5617
- URL: https://www.wjgnet.com/1007-9327/full/v29/i40/5593.htm
- DOI: https://dx.doi.org/10.3748/wjg.v29.i40.5593