©2012 Baishideng Publishing Group Co.
World J Gastroenterol. Jul 28, 2012; 18(28): 3752-3760
Published online Jul 28, 2012. doi: 10.3748/wjg.v18.i28.3752
Published online Jul 28, 2012. doi: 10.3748/wjg.v18.i28.3752
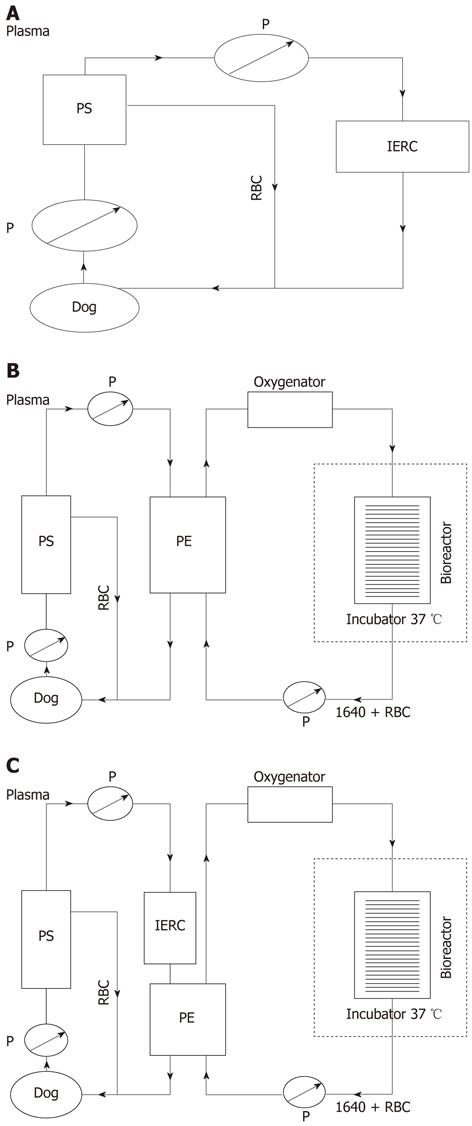
Figure 1 Schematic assembly of three artificial liver systems.
A: Schematic assembly of non-bioartificial liver; B: Schematic assembly of bioartificial liver; C: Schematic assembly of hybrid bioartificial liver. P: Pump; RBC: Red blood cells; PS: Plasma separator; PE: Plasma component exchange column; IERC: Anionic resin adsorption column.
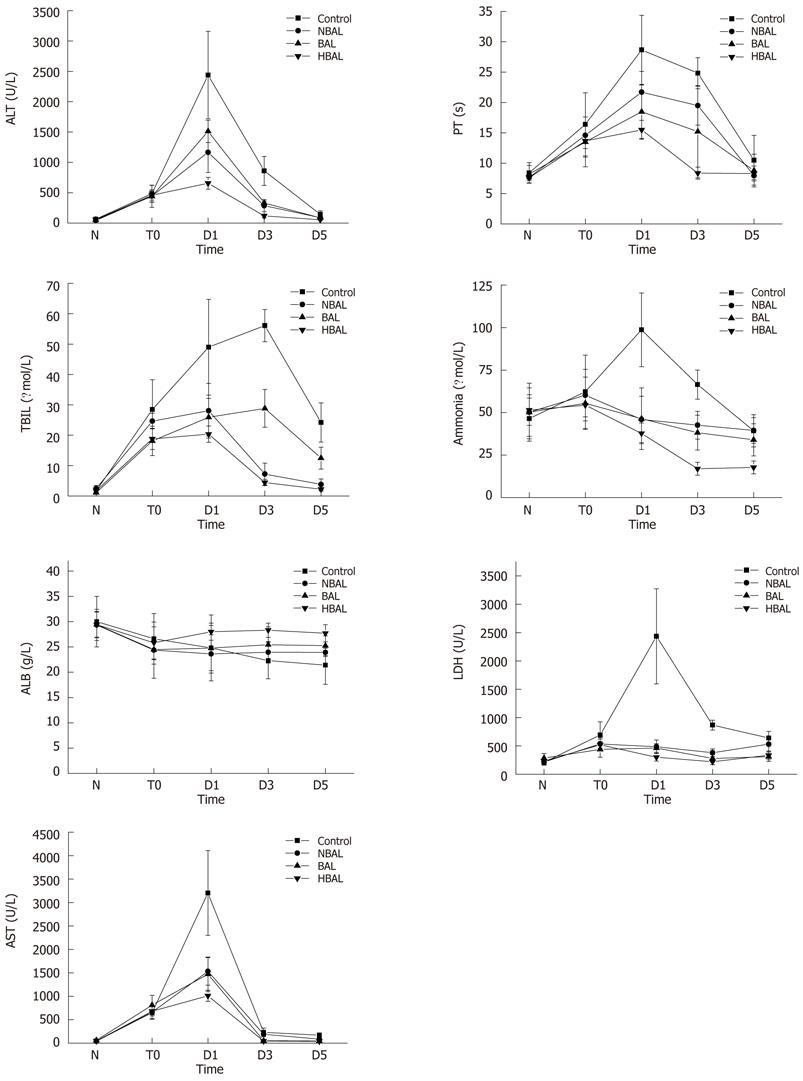
Figure 2 Comparisons of biochemical index between the treatment groups and the control group.
ALT: Alanine aminotransferase; AST: Aspartate aminotransferase; LDH: Lactate dehydrogenase; TBIL: Total bilirubin; ALB: Albumin; PT: Prothrombin time; HBAL: Hybrid bioartificial liver; BAL: Bioartificial liver; NBAL: Non-bioartificial liver; N: Normal; T0: Before treatment; D1: Day 1 after treatment; D3: Day 3 after treatment; D5: Day 5 after treatment.
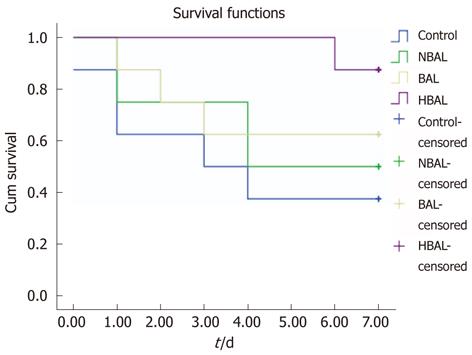
Figure 3 Kaplan-Meier analysis of survival of two groups.
HBAL: Hybrid bioartificial liver; BAL: Bioartificial liver; NBAL: Non-bioartificial liver.
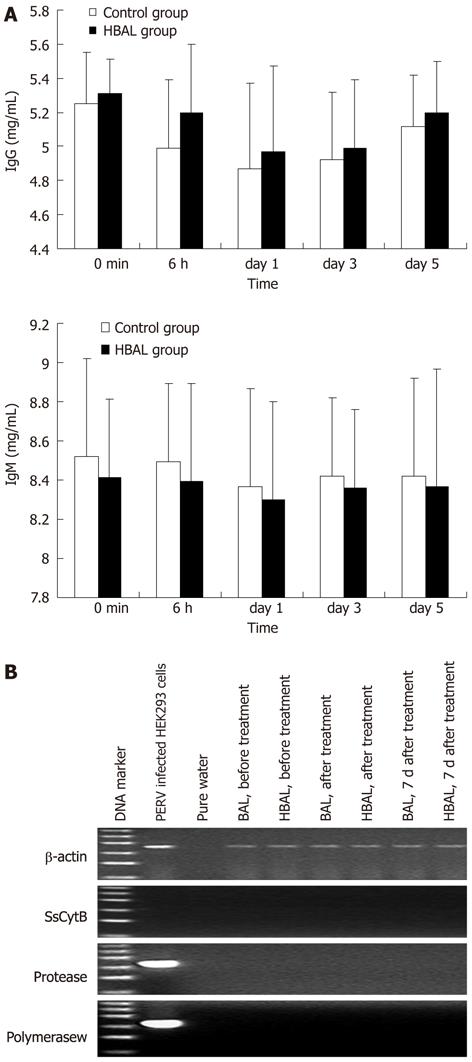
Figure 4 Safety evaluation.
A: Xenoreactive antibodies levels during bioartificial liver treatments; B: Representative results of reverse transcription-polymerase chain reaction electrophoresis with the RNA extracted from the plasma. The ladder ranged from 100 bp to 600 bp. NBAL: Hybrid bioartificial liver; BAL: Bioartificial liver; PERV: Porcine endogenous retrovirus.
- Citation: Shi XL, Zhang Y, Chu XH, Han B, Gu JY, Xiao JQ, Tan JJ, Gu ZZ, Ren HZ, Yuan XW, Ding YT. Evaluation of a novel hybrid bioartificial liver based on a multi-layer flat-plate bioreactor. World J Gastroenterol 2012; 18(28): 3752-3760
- URL: https://www.wjgnet.com/1007-9327/full/v18/i28/3752.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i28.3752