Published online Apr 28, 2021. doi: 10.13105/wjma.v9.i2.101
Peer-review started: January 27, 2021
First decision: February 25, 2021
Revised: March 10, 2021
Accepted: April 23, 2021
Article in press: April 23, 2021
Published online: April 28, 2021
Processing time: 88 Days and 5.4 Hours
Insulin resistance (IR) is the common pathophysiological basis of many metabolic diseases. IR is characterized by decreased glucose uptake in skeletal muscle and adipose tissue, especially in skeletal muscle. Skeletal muscle is the main target tissue of glucose uptake under insulin stimulation. Glucose uptake by skeletal muscle is complex, and it is controlled by many pathways. The PI3K/AKt/GSK-1 signaling pathway is not only the main pathway for insulin signal transduction but also an important mechanism for regulating blood glucose. From the binding of insulin to its receptors on the surface of target cells to the transportation of glucose from extracellular fluid to skeletal muscle, a series of signal transduction processes is completed, any of which potentially affects the physiological effects of insulin and leads to IR. Resistance exercise (RT) can reduce skeletal muscle IR and effectively improve blood glucose control and glycosylated hemoglobin level in patients with type 2 diabetes mellitus (T2DM). However, the exact mechanism by which RT improves skeletal muscle IR remains unclear. Therefore, this paper discusses the above problems by tracking the progress of the literature to deepen the correlation between RT and skeletal muscle insulin sensitivity and provide further evidence for the application of exercise therapy in IR. In conclusion, RT mainly improves insulin sensitivity of skeletal muscle by increasing muscle mass, microvascular blood flow, and glucose transporter-4 expression in skeletal muscle, as well as by reducing lipid accumulation and inflammation in skeletal muscle. Thus, it is potentially useful in the prevention and treatment of T2DM.
Core Tip: Both muscle contraction and hypoxia can mobilize the transfer of glucose transporter-4. Resistance exercises (RTs) can significantly increase limb muscle content and body fat content and significantly reduce blood glucose and glycosylated hemoglobin levels in patients with type 2 diabetes mellitus (T2DM). Lipid accumulation in skeletal muscle cells potentially leads to insulin resistance (IR). RT or acute RT can lower intracellular lipid accumulation in muscle, which helps reduce peripheral IR. It is suggested that the RT-induced microvascular blood flow increase helps improve glucose metabolism in patients with skeletal muscle IR and T2DM.
- Citation: Wang B, Luo X, Li RR, Li YN, Zhao YC. Effect of resistance exercise on insulin sensitivity of skeletal muscle. World J Meta-Anal 2021; 9(2): 101-107
- URL: https://www.wjgnet.com/2308-3840/full/v9/i2/101.htm
- DOI: https://dx.doi.org/10.13105/wjma.v9.i2.101
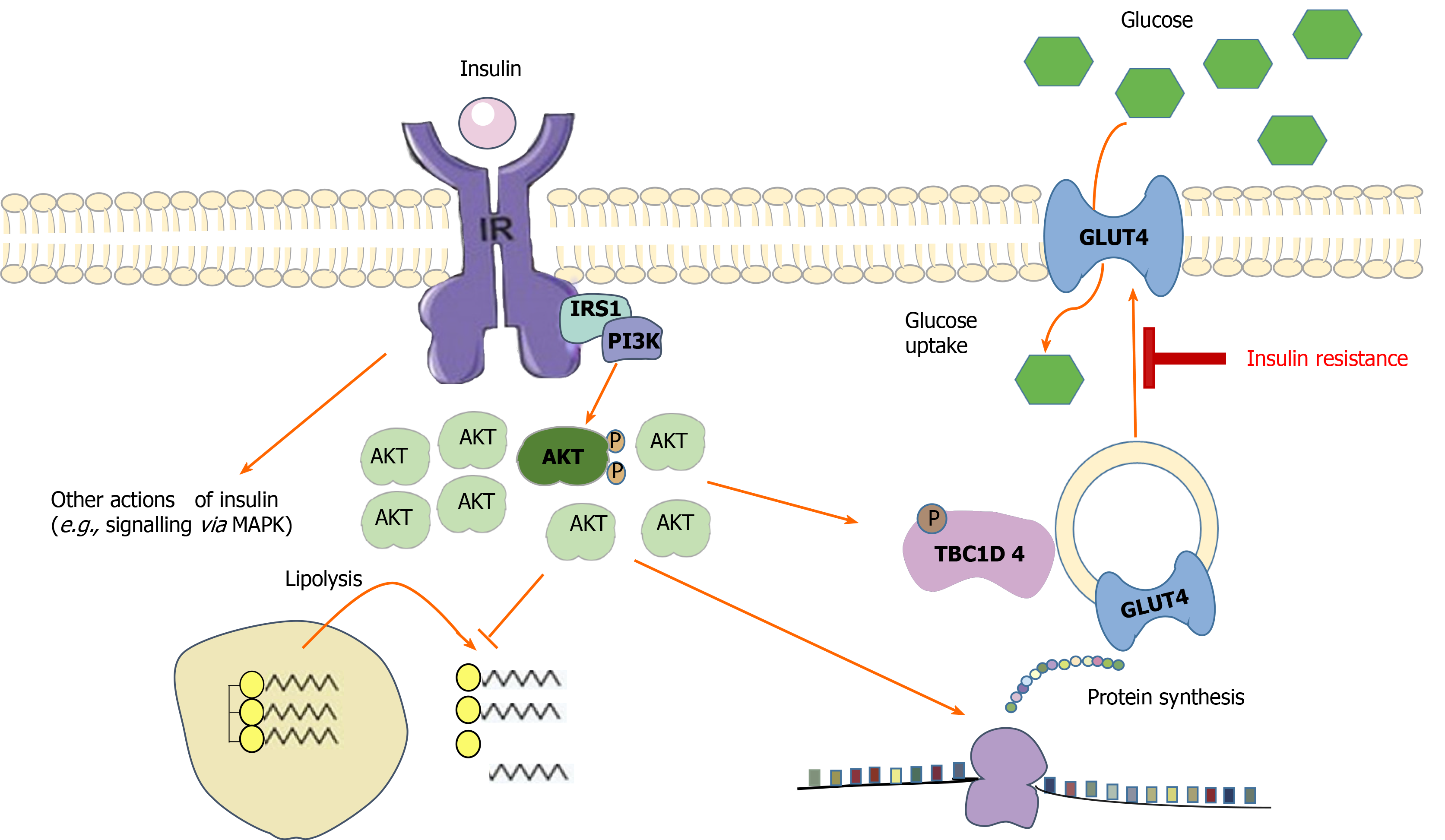
Insulin resistance (IR) is the common pathophysiological basis of many metabolic diseases, and it is a consequence of decreased sensitivity of peripheral tissue to insulin, resulting in a higher demand for insulin to maintain normal metabolic homeostasis. IR is characterized by decreased glucose uptake in skeletal muscle and adipose tissue[1], especially in skeletal muscle[2]. Skeletal muscle is the main target tissue of glucose uptake under insulin stimulation. Eighty to ninety percent of insulin-stimulated glucose uptake occurs in skeletal muscle[3]. Glucose uptake by skeletal muscle is complex, and it is controlled by many pathways. The PI3K/AKt/GSK-1 signaling pathway is not only the main pathway for insulin signal transduction[4], but also an important mechanism for regulating blood glucose. From the binding of insulin to its receptors on the surface of target cells to the transportation of glucose from extracellular fluid to skeletal muscle, a series of signal transduction processes is completed, any of which potentially affects the biological effects of insulin and leads to IR (Figure 1). Genetic susceptibility, lack of physical activity, and obesity all lead to IR[5].
Resistance exercise (RT) is movement in which muscles overcome external resistance to actively contract and relax, which deviates from traditional aerobic exercise in that it exerts external resistance and emphasizes the exercise of muscle strength and endurance. RT can reduce skeletal muscle IR[6] and effectively improve blood glucose control and glycosylated hemoglobin (HbA1c) levels in patients with type 2 diabetes mellitus (T2DM)[7]. However, the exact mechanism by which RT improves skeletal muscle IR remains unclear. Therefore, this paper discusses the above problems by tracking the progress of the literature, locally and abroad, in order to deepen the correlation between RT and skeletal muscle insulin sensitivity and provide further evidence for the application of exercise therapy in IR.
One of the most mature mechanisms of IR is the decreased expression of the glucose transporter-4 (GLUT4) protein in muscle, which may be related to mitochondrial dysfunction and decreased biosynthesis[8]. Sharma et al[9] discovered that the normal phosphorylation of AS160 serine-588 and threonine-642 in the PI3K pathway was significantly decreased in elderly patients with T2DM, and these two sites were considered to have an important effect on the translocation of GLUT4. The decrease or even interruption of the transfer of GLUT4 self-storage vesicles to the surface of muscle cells may be another important reason for the damage caused to the insulin signal pathway. As a single stimulating factor, exercise can promote the translocation of GLUT-4 to the cell membrane and enhance glucose uptake[10]. Both muscle contraction and hypoxia can mobilize the transfer of GLUT4. The energy supply characteristic of RT is that muscle contraction can also cause a hypoxic environment inside the muscle cells, whereas aerobic exercise cannot. According to the degree of IR in T2DM patients, adding the corresponding RT content in the exercise prescription can achieve a better intervention outcome.
The content of GLUT4 in the skeletal muscle of pre-T2DM patients with obesity increased after 16 wk of RT[11]; however, no change in GLUT4 level in the gastrocnemius muscle of T2DM rats was observed in an experiment involving the simulation of RT using an advanced vertical ladder (80° ladder, 50 times/wk) over 7 wk[12]. McMillin et al[13] also demonstrated that rats with skeletal muscle-specific GLUT4 gene knockout did not experience glucose transport impairment in the plantar muscle after 5 d of muscle load (RT model). The above results suggest that RT seems to increase the content of GLUT4 in the skeletal muscle of patients with obesity and/or T2DM and contributes to the improvement of IR; nevertheless, GLUT4 is not the only factor in the RT-induced increase of glucose transport in skeletal muscle. According to existing research results, other types of GLUT, such as GLUT1 and GLUT10, may be involved, which needs to be confirmed through further studies[14].
Skeletal muscle is an important tissue for glucose storage and metabolism. With a decrease in skeletal muscle volume and muscle strength, the number of insulin receptors, insulin sensitivity, and muscle utilization of blood-derived glucose decrease, and this readily induces IR and impaired glucose tolerance[15]. Delaying skeletal muscle loss increases insulin sensitivity[16]. According to the 2011 National Health and Nutrition Survey report, muscle mass reduction was positively correlated with IR and HbA1c levels and negatively correlated with glucose metabolism. There was a significantly negative correlation between muscle strength and IR index in patients with T2DM[17]. RT can significantly increase skeletal muscle strength and increase the cross-sectional area of the quadriceps femoris[18], thus increasing the number of insulin receptors and improving insulin sensitivity. After 8 wk of RT, the upper arm muscle mass and relative muscle strength of patients with T2DM were increased, and the levels of fasting blood glucose and fructosamine were significantly decreased[19]. We observed that resistance and aerobic exercises can significantly increase limb muscle content and body fat content and significantly reduce blood glucose and HbA1c in patients with T2DM[20]. RT can increase muscle mass more effectively than aerobic training[21]. The increase in muscle mass induced by RT is related to the increase in protein synthesis in skeletal muscle cells. The main pathway of muscle protein synthesis is the insulin-like growth factor-1 (IGF-1)/PI3K/Akt/mammalian rapamycin target (mTOR) pathway. RT can upregulate the expression of IGF-1 and eventually activate mTOR[22] to promote mRNA translation and protein synthesis. West et al[23] discovered that the activities of IGF-1 and mTOR in lower limb muscles of female rats were significantly increased after intervention with RT.
Lipid accumulation in skeletal muscle cells can lead to IR. The specific mechanism includes the following two aspects. First, intracellular free fatty acids and their potential metabolites can be used as signal molecules to activate protein kinases, such as protein kinase C, JUN kinase, and nuclear factor inhibitor kinase B. These kinases can phosphorylate serine on the insulin receptor substrate (IRS), thus inhibiting the IRS-mediated insulin signal transduction pathway. Finally, it causes IR in skeletal muscle cells and other peripheral target cells. With the continuous development of mass spectrometry, bioactive lipids, such as diacylglycerol, sphingomyelin, and sphingosine glycerol, are more likely to be related to the mechanism of IR[24]. New evidence suggests that the localization of triglycerides, diglycerides, and sphingomyelin seems to play an important role in promoting the reduction of insulin sensitivity[25]. Transmission electron microscopy revealed that submuscular intramyocellular triacylglycerol (IMTG) was negatively correlated with insulin sensitivity of type II fibers, whereas IMTG between muscle fibers was either positively or not correlated with insulin sensitivity. Early work involving diacylglycerol (DAG) in the Bergman Laboratory demonstrated that DAG located on the skeletal muscle membrane was negatively correlated with insulin sensitivity, whereas cytoplasmic DAG was not related to insulin sensitivity[24]. RT or acute RT can affect the accumulation of lipids in muscle[26,27], and this reduction in intracellular lipid accumulation is beneficial in mitigating peripheral IR. It was found that insulin sensitivity increased even after RT[28]. Insulin sensitivity recovered 2 h after RT, and intramyocellular lipid content either did not change[29] or returned to the basic level[30] The utilization rate of intramyocellular triacylglycerol (IMTG) in resistance movement seems to depend partly on the initial concentration; therefore, the higher the content of IMTG, the higher its degradation rate. However, the mechanism still requires further investigation. The effects of acute RT on bioactive lipids (such as DAG and sphingolipids) and other polar lipids (such as acylcarnitine) are unclear. Some scholars believe that exercise can improve the coordination of the transfer, intake, and oxidation of free fatty acids, thus reducing the accumulation of lipids in skeletal muscle cells[31].
Second, free fatty acids can directly activate Toll-like receptor 4 to trigger the natural immune response, thus inducing IR through the inflammatory response pathway. Toll-like receptors are a family of highly conserved proteins that play an important role in the recognition of microbial pathogens. The activation of this receptor signaling pathway can increase the expression of proinflammatory factors and play an important role in the mediation of systemic inflammation. In recent years, studies have shown that high inflammation of skeletal muscle can damage the transduction of the insulin receptor signal, which may play an important role in the occurrence and development of IR[32]. In patients with T2DM, 16 wk of aerobic exercise combined with RT significantly decreased the levels of C-reactive protein, interleukin (IL)-1 β, IL-8, and tumor necrosis factor-α and increased the levels of anti-inflammatory factors, such as IL-6, IL-10, and IL-15[33], thus promoting the conversion of white fat to brown fat[34] and playing a positive regulatory role in insulin secretion and islet β-cell proliferation. However, it is not clear whether this effect has long-term stability. The effects of 12 wk of aerobic exercise combined with RT on cardiovascular risk factors in male adolescents with obesity were observed. It was revealed that aerobic exercise combined with RT could significantly reduce the levels of serum IL-1Ra, interferon-γ-inducible protein 10a, insulin, and the homeostasis model assessment of IR index in men with obesity[35]. Recent studies have reported[36] that RT potentially increases muscle mass and systemic insulin sensitivity, reduces inflammatory response, increases adiponectin, and improves related metabolism in patients with T2DM.
The main function of microvascular units is to provide a site for the exchange of various reaction substrates, oxygen, and hormones between plasma and extracellular fluid in the tissue. At rest, approximately 30% of the skeletal muscle capillaries are perfused. Studies have revealed that changing the endothelial exchange area of skeletal muscle can affect the transport of insulin to and its effect on skeletal muscle[37]. The increase in skeletal muscle microcirculation perfusion caused by many factors, including insulin, mixed meals[38], angiotensin II receptor inhibitor, muscle contraction, and others, is related to the increase in skeletal muscle insulin uptake. This demonstrates that microcirculation plays a very important role in insulin uptake by skeletal muscle. According to Russell et al[39], after systemic RT for 6 wk, IR and fasting blood glucose in patients with T2DM were significantly lower than those before exercise. Further, they were significantly correlated with the increase in microvascular blood flow (MBF) stimulated by RT and independent of the change in muscle capillary density. It is suggested that the increase in MBF induced by RT is beneficial in improving the level of glucose metabolism in patients with skeletal muscle IR and T2DM. Zhao et al[40] discovered that the blood-flow velocity of the low-load blood-flow restriction regimen was significantly higher than that of traditional RT after exercise, which may be related to the stimulation of shear force and metabolic stress during exercise. This enhanced vascular compliance and accelerated tissue perfusion as well as metabolic product transport and clearance after exercise.
In conclusion, RT predominantly improves insulin sensitivity in skeletal muscle through the following mechanisms: Increasing muscle mass, increasing the expression of GLUT4 in skeletal muscle, reducing the accumulation of lipids and inflammation in skeletal muscle, and increasing MBF. Therefore, it can be used in the prevention and treatment of T2DM. In order to obtain the expected clinical effect, the exercise type, duration, and intensity of RT require further study.
| 1. | Alatrach M, Agyin C, Mehta R, Adams J, DeFronzo RA, Abdul-Ghani M. Glucose-Mediated Glucose Disposal at Baseline Insulin Is Impaired in IFG. J Clin Endocrinol Metab. 2019;104:163-171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 2. | Vichaiwong K, Purohit S, An D, Toyoda T, Jessen N, Hirshman MF, Goodyear LJ. Contraction regulates site-specific phosphorylation of TBC1D1 in skeletal muscle. Biochem J. 2010;431:311-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 83] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 3. | Henriksen EJ. Invited review: Effects of acute exercise and exercise training on insulin resistance. J Appl Physiol (1985). 2002;93:788-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 370] [Cited by in RCA: 308] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 4. | Bird SR, Hawley JA. Update on the effects of physical activity on insulin sensitivity in humans. BMJ Open Sport Exerc Med. 2016;2:e000143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 487] [Cited by in RCA: 418] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 5. | Röhling M, Herder C, Stemper T, Müssig K. Influence of Acute and Chronic Exercise on Glucose Uptake. J Diabetes Res. 2016;2016:2868652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 6. | Holten MK, Zacho M, Gaster M, Juel C, Wojtaszewski JF, Dela F. Strength training increases insulin-mediated glucose uptake, GLUT4 content, and insulin signaling in skeletal muscle in patients with type 2 diabetes. Diabetes. 2004;53:294-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 475] [Cited by in RCA: 399] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 7. | Hangping Z, Xiaona Q, Qi Z, Qingchun L, Na Y, Lijin J, Siying L, Shuo Z, Xiaoming Z, Xiaoxia L, Qian X, Jaimovich D, Yiming L, Bin L. The impact on glycemic control through progressive resistance training with bioDensityTM in Chinese elderly patients with type 2 diabetes: The PReTTy2 (Progressive Resistance Training in Type 2 Diabetes) Trial. Diabetes Res Clin Pract. 2019;150:64-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 8. | Dos Santos JM, Moreli ML, Tewari S, Benite-Ribeiro SA. The effect of exercise on skeletal muscle glucose uptake in type 2 diabetes: An epigenetic perspective. Metabolism. 2015;64:1619-1628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 9. | Sharma P, Arias EB, Cartee GD. Protein Phosphatase 1-α Regulates AS160 Ser588 and Thr642 Dephosphorylation in Skeletal Muscle. Diabetes. 2016;65:2606-2617. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 10. | Fujii N, Jessen N, Goodyear LJ. AMP-activated protein kinase and the regulation of glucose transport. Am J Physiol Endocrinol Metab. 2006;291:E867-E877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 169] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 11. | Stuart CA, Lee ML, South MA, Howell MEA, Stone MH. Muscle hypertrophy in prediabetic men after 16 wk of resistance training. J Appl Physiol (1985). 2017;123:894-901. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | Quines CB, Jardim NS, Araujo PCO, Cechella JL, Prado VC, Nogueira CW. Resistance training restores metabolic alterations induced by monosodium glutamate in a sex-dependent manner in male and female rats. J Cell Biochem. 2019;120:13426-13440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | McMillin SL, Schmidt DL, Kahn BB, Witczak CA. GLUT4 Is Not Necessary for Overload-Induced Glucose Uptake or Hypertrophic Growth in Mouse Skeletal Muscle. Diabetes. 2017;66:1491-1500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 14. | Syu YW, Lai HW, Jiang CL, Tsai HY, Lin CC, Lee YC. GLUT10 maintains the integrity of major arteries through regulation of redox homeostasis and mitochondrial function. Hum Mol Genet. 2018;27:307-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | Nomura T, Kawae T, Kataoka H, Ikeda Y. Aging, physical activity, and diabetic complications related to loss of muscle strength in patients with type 2 diabetes. Phys Ther Res. 2018;21:33-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 16. | Cartee GD, Hepple RT, Bamman MM, Zierath JR. Exercise Promotes Healthy Aging of Skeletal Muscle. Cell Metab. 2016;23:1034-1047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 427] [Cited by in RCA: 367] [Article Influence: 36.7] [Reference Citation Analysis (0)] |
| 17. | Nomura T, Ikeda Y, Nakao S, Ito K, Ishida K, Suehiro T, Hashimoto K. Muscle strength is a marker of insulin resistance in patients with type 2 diabetes: a pilot study. Endocr J. 2007;54:791-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Fonseca RM, Roschel H, Tricoli V, de Souza EO, Wilson JM, Laurentino GC, Aihara AY, de Souza Leão AR, Ugrinowitsch C. Changes in exercises are more effective than in loading schemes to improve muscle strength. J Strength Cond Res. 2014;28:3085-3092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 19. | Egger A, Niederseer D, Diem G, Finkenzeller T, Ledl-Kurkowski E, Forstner R, Pirich C, Patsch W, Weitgasser R, Niebauer J. Different types of resistance training in type 2 diabetes mellitus: effects on glycaemic control, muscle mass and strength. Eur J Prev Cardiol. 2013;20:1051-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 20. | Wang B, Mu XL, Zhao J, Jiang HP, Li SS, Yan G, Hua YY, Ren XY, Xing LX, Liang Y, Zhang SD, Zhao YC. Effects of lifestyle interventions on rural patients with type 2 diabetes mellitus. World J Diabetes. 2020;11:261-268. [PubMed] [DOI] [Full Text] |
| 21. | Yan J, Dai X, Feng J, Yuan X, Li J, Yang L, Zuo P, Fang Z, Liu C, Hsue C, Zhu J, Miller JD, Lou Q. Effect of 12-Month Resistance Training on Changes in Abdominal Adipose Tissue and Metabolic Variables in Patients with Prediabetes: A Randomized Controlled Trial. J Diabetes Res. 2019;2019:8469739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 22. | Vassilakos G, Barton ER. Insulin-Like Growth Factor I Regulation and Its Actions in Skeletal Muscle. Compr Physiol. 2018;9:413-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 23. | West DW, Baehr LM, Marcotte GR, Chason CM, Tolento L, Gomes AV, Bodine SC, Baar K. Acute resistance exercise activates rapamycin-sensitive and -insensitive mechanisms that control translational activity and capacity in skeletal muscle. J Physiol. 2016;594:453-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 124] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 24. | Bergman BC, Hunerdosse DM, Kerege A, Playdon MC, Perreault L. Localisation and composition of skeletal muscle diacylglycerol predicts insulin resistance in humans. Diabetologia. 2012;55:1140-1150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 137] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 25. | Perreault L, Newsom SA, Strauss A, Kerege A, Kahn DE, Harrison KA, Snell-Bergeon JK, Nemkov T, D'Alessandro A, Jackman MR, MacLean PS, Bergman BC. Intracellular localization of diacylglycerols and sphingolipids influences insulin sensitivity and mitochondrial function in human skeletal muscle. JCI Insight. 2018;3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 163] [Cited by in RCA: 139] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 26. | Perseghin G, Price TB, Petersen KF, Roden M, Cline GW, Gerow K, Rothman DL, Shulman GI. Increased glucose transport-phosphorylation and muscle glycogen synthesis after exercise training in insulin-resistant subjects. N Engl J Med. 1996;335:1357-1362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 510] [Cited by in RCA: 436] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 27. | Devries MC, Samjoo IA, Hamadeh MJ, McCready C, Raha S, Watt MJ, Steinberg GR, Tarnopolsky MA. Endurance training modulates intramyocellular lipid compartmentalization and morphology in skeletal muscle of lean and obese women. J Clin Endocrinol Metab. 2013;98:4852-4862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 67] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 28. | Malin SK, Hinnerichs KR, Echtenkamp BG, Evetovich TK, Engebretsen BJ. Effect of adiposity on insulin action after acute and chronic resistance exercise in non-diabetic women. Eur J Appl Physiol. 2013;113:2933-2941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 29. | Harber MP, Crane JD, Douglass MD, Weindel KD, Trappe TA, Trappe SW, Fink WF. Resistance exercise reduces muscular substrates in women. Int J Sports Med. 2008;29:719-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 30. | Koopman R, Manders RJ, Jonkers RA, Hul GB, Kuipers H, van Loon LJ. Intramyocellular lipid and glycogen content are reduced following resistance exercise in untrained healthy males. Eur J Appl Physiol. 2006;96:525-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 91] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 31. | Horowitz JF. Fatty acid mobilization from adipose tissue during exercise. Trends Endocrinol Metab. 2003;14:386-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 130] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 32. | Mesinovic J, Zengin A, De Courten B, Ebeling PR, Scott D. Sarcopenia and type 2 diabetes mellitus: a bidirectional relationship. Diabetes Metab Syndr Obes. 2019;12:1057-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 399] [Article Influence: 57.0] [Reference Citation Analysis (1)] |
| 33. | Tomeleri CM, Souza MF, Burini RC, Cavaglieri CR, Ribeiro AS, Antunes M, Nunes JP, Venturini D, Barbosa DS, Sardinha LB, Cyrino ES. Resistance training reduces metabolic syndrome and inflammatory markers in older women: A randomized controlled trial. J Diabetes. 2018;10:328-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 86] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 34. | Annibalini G, Lucertini F, Agostini D, Vallorani L, Gioacchini A, Barbieri E, Guescini M, Casadei L, Passalia A, Del Sal M, Piccoli G, Andreani M, Federici A, Stocchi V. Concurrent Aerobic and Resistance Training Has Anti-Inflammatory Effects and Increases Both Plasma and Leukocyte Levels of IGF-1 in Late Middle-Aged Type 2 Diabetic Patients. Oxid Med Cell Longev. 2017;2017:3937842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 35. | Zhi JX. Effect of aerobic exercise combined with resistance training on cardiovascular risk factors in obese male adolescents. Zhonghua Wuli Yixue Yu Kangfu Zazhi. 2019;41:447-452. [DOI] [Full Text] |
| 36. | Barrett EJ, Eggleston EM, Inyard AC, Wang H, Li G, Chai W, Liu Z. The vascular actions of insulin control its delivery to muscle and regulate the rate-limiting step in skeletal muscle insulin action. Diabetologia. 2009;52:752-764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 237] [Cited by in RCA: 222] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 37. | Clark MG. Impaired microvascular perfusion: a consequence of vascular dysfunction and a potential cause of insulin resistance in muscle. Am J Physiol Endocrinol Metab. 2008;295:E732-E750. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 144] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 38. | Chai W, Wang W, Liu J, Barrett EJ, Carey RM, Cao W, Liu Z. Angiotensin II type 1 and type 2 receptors regulate basal skeletal muscle microvascular volume and glucose use. Hypertension. 2010;55:523-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 73] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 39. | Russell RD, Hu D, Greenaway T, Blackwood SJ, Dwyer RM, Sharman JE, Jones G, Squibb KA, Brown AA, Otahal P, Boman M, Al-Aubaidy H, Premilovac D, Roberts CK, Hitchins S, Richards SM, Rattigan S, Keske MA. Skeletal Muscle Microvascular-Linked Improvements in Glycemic Control From Resistance Training in Individuals With Type 2 Diabetes. Diabetes Care. 2017;40:1256-1263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 40. | Zhao Y, Li JN, Sun B, Zhou B. Effect of short-term arterial blood flow occlusion training on skeletal muscle performance of healthy college students. Beijing Tiyu Daxue Xuebao. 2013;36:54-58. |
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Manuscript source: Invited manuscript
Specialty type: Endocrinology and metabolism
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Wang CR S-Editor: Wang JL L-Editor: Wang TQ P-Editor: Li JH