Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.114269
Revised: October 13, 2025
Accepted: January 7, 2026
Published online: June 20, 2026
Processing time: 220 Days and 17.5 Hours
Neutralizing autoantibodies against interleukin-1 receptor antagonist (IL-1RA) have been identified in patients with Still’s disease, often in association with hy
Core Tip: Still’s disease is a rare, severe systemic inflammatory disorder characterized by elevated levels of neutralizing interleukin-1 receptor antagonist autoantibodies. Their induction is thought to result from a combination of host-related factors, such as human leukocyte antigen class II alleles, and interleukin-1 receptor antagonist intrinsic factors, including hyperphosphorylation. Despite frequent detection of these antibodies using immunoassays and functional cell-based assays, the lack of assay standardization limits their clinical utility. At present, their presence is best regarded as a marker of underlying immune dysregulation rather than a direct pathogenic driver.
- Citation: Bouayad A. Interleukin-1 receptor antagonist autoantibodies in Still’s disease: Mechanistic insights and laboratory testing. World J Methodol 2026; 16(2): 114269
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/114269.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.114269
Still’s disease is a rare, severe systemic inflammatory syndrome that includes systemic juvenile idiopathic arthritis (sJIA) in children and adult-onset Still’s disease (AoSD) in adults. Both forms share characteristic features, including quotidian spiking fever, evanescent rash, and inflammatory arthritis[1,2]. Phenotypic evidence supporting the autoinflammatory nature of Still’s disease includes elevated levels of several inflammatory biomarkers, including neutrophilic leukocytosis, increased ferritin, C-reactive protein, erythrocyte sedimentation rate, interleukin (IL)-18, and S100 proteins[3,4]. Among these, hyperferritinemia represents a key biological hallmark and is thought to reflect increased levels of IL-18 and soluble IL-2 receptor[5]. Recent findings by Jia et al[6] suggest that ferritin may actively contribute to disease pathogenesis by triggering neutrophil extracellular trap-mediated cytokine storms in AoSD.
Although the recurrent fevers and systemic inflammation observed in Still’s disease closely resemble classic autoinflammatory syndromes driven by innate immune dysregulation, accumulating evidence points to an additional role for adaptive immunity. This is supported by the detection of autoantibodies (AAbs)[7-9] and genetic association with human leukocyte antigen class II (HLA-II) alleles[10,11], implicating contributions from B cells, T cells, and AAb-mediated mechanisms. Recently, Hoffmann et al[7] reported the presence of neutralizing AAbs (Nabs) targeting IL-1 receptor antagonist (IL-1RA) in about 18.9% of individuals with Still’s disease. These AAbs have also been implicated in other conditions, such as vaccine-induced myocarditis[12], multisystem inflammatory syndrome in children (MIS-C)[13], and immunoglobulin G4-related disease (IgG4-RD)[14], suggesting a broader role in inflammatory diseases.
Why only a subset of individuals develops these AAbs remains unclear and appears to be only partly explained by factors such as antibody titer, IgG isotype, and depletion of circulating IL-1RA[7]. The reported prevalence, titers, and biological significance of these AAbs are further confounded by methodological heterogeneity in detection assays. This minireview discusses genetic and environmental factors that may predispose individuals to the development of IL-1RA Nabs and their potential biological and clinical significance in Still’s disease. It also summarizes current laboratory methods for their detection, highlighting key challenges and limitations.
The IL-1 cytokine family, which broadly influences Still’s disease, comprises both pro- and anti-inflammatory cytokines that mediate and regulate inflammation. Key proinflammatory members include IL-1α, IL-1β, and IL-18, which promote inflammatory responses[15-17]. In contrast, anti-inflammatory members such as IL-1RA, IL-36RA, and IL-18 binding protein (IL-18BP) act to limit and resolve inflammation[16,17].
IL1-RA is an anti-inflammatory cytokine protein of about 17 kDa composed of two very short 3-10 helices and 12 β-strands[18,19]. Plasma or serum levels of IL-1RA are significantly decreased in anti-IL-1RA seropositive patients with Still’s disease[7]. Structural analyses have identified specific regions of IL-1RA that are critical for antibody recognition. In patients with Still’s disease and MIS-C, residues glycine98-asparagine116 and phenylalanine125-glycine143 located in IL-1RA have been implicated as major epitopes for IL-1RA Nabs[7,13] (Supplementary Table 1). These epitopes partially overlap with residues Met136–Glu152 and Phe100-Gly119 within the IL-1RA/IL-1 type-I receptor (IL-1RI) binding interface, which have been previously associated with Nabs against IL-1RA in the context of IgG4-RD[14] (Supple
IL-36RA is a receptor antagonist with a molecular mass of about 17 kDa that binds to IL-1R6, thereby inhibiting IL-36-mediated signaling[20]. Anti-IL-36RA AAbs have been reported in approximately 7% of patients with AOSD[7]. These AAbs have also been detected in subgroups of patients with psoriasis without arthritic manifestations and in those with psoriatic arthritis, where they may contribute to disease pathogenesis[21].
Another important IL-1 family cytokine is IL-1β, which is moderately elevated in active Still’s disease[17]. The activity of IL-1β and IL-1α is effectively regulated by decoy receptors and soluble antagonists such as IL-1RA. However, when IL-1RA undergoes hyperphosphorylation, IgG-IL-1RA immune complexes may form, preventing IL-1RA from binding IL-1RI and thereby amplifying IL-1 signaling[22,23]. Similar to IL-1β, IL-18 in Still’s disease serves as a marker of heightened inflammation, with elevated circulating levels observed in patients with active disease[17]. This cytokine is considered pathogenic in macrophage activation syndrome (MAS), a complication that commonly occurs in sJIA and possibly also in AOSD[24,25]. Its biological activity is neutralized by IL-18BP, and to date, no anti-IL-18BP AAbs have been reported in patients with AOSD[7].
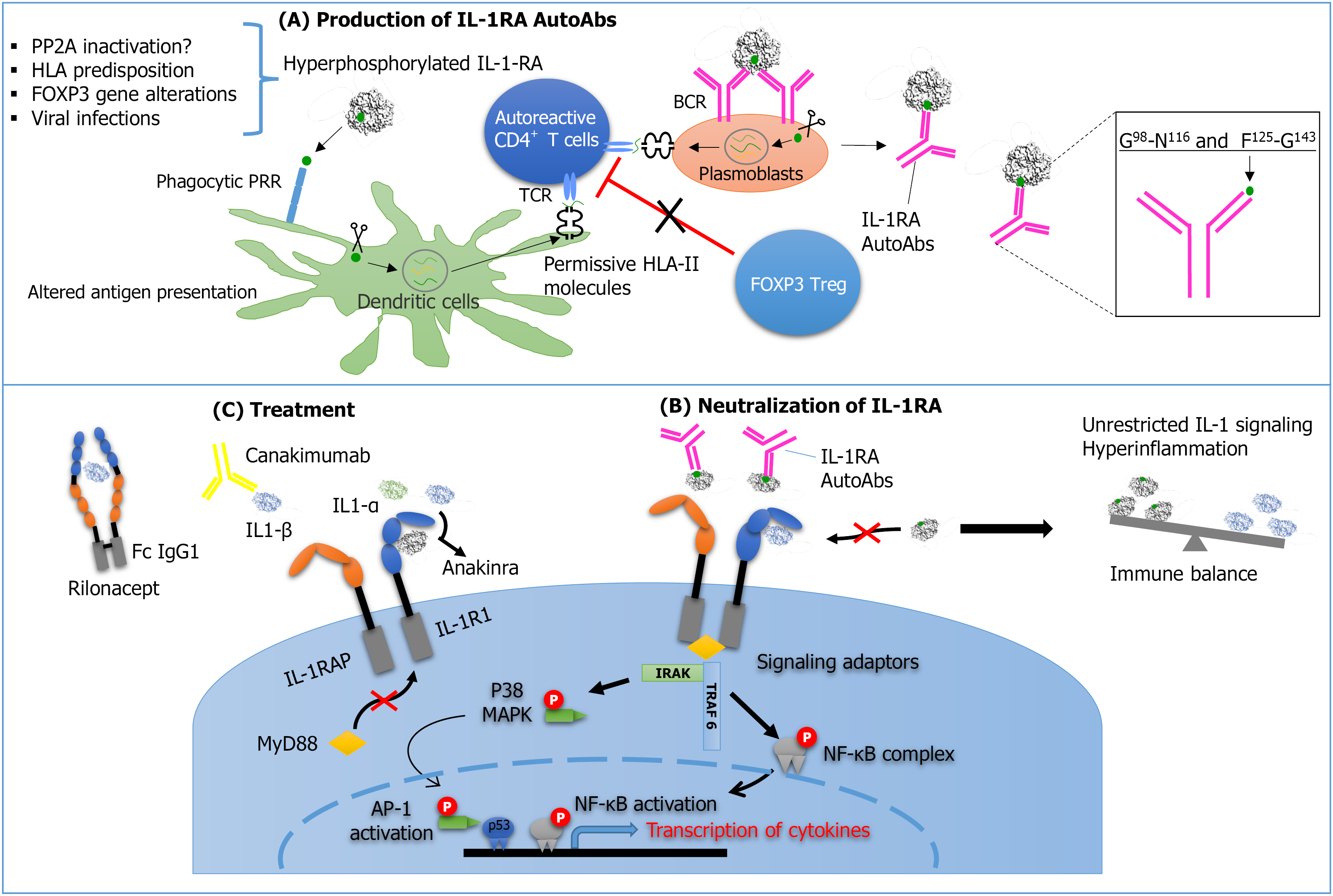
The IL-1α/1β signaling pathway transduction can be divided into two branches, as illustrated in Figure 1. Briefly, upon binding of IL-1α or IL-1β to IL-1RI, a receptor complex forms with the IL-1 receptor accessory protein, recruiting the adaptor myeloid differentiation factor 88[26,27]. This initiates phosphorylation of IL-1R-associated kinases and subsequent activation of tumor necrosis factor receptor-associated factor 6. The nuclear factor kappa B (NF-κB) pathway is then triggered through activation of the IκB kinase complex, which phosphorylates IκB, marking it for ubiquitination and proteasomal degradation. The liberated NF-κB heterodimer translocates to the nucleus to induce transcription of pro-inflammatory cytokine genes. In parallel, the mitogen-activated protein kinases (MAPK) cascade is activated via mammalian MAPK kinase kinase, leading to phosphorylation of p38 MAPK[27]. Activated p38 translocates into the nucleus and promotes cytokine production.
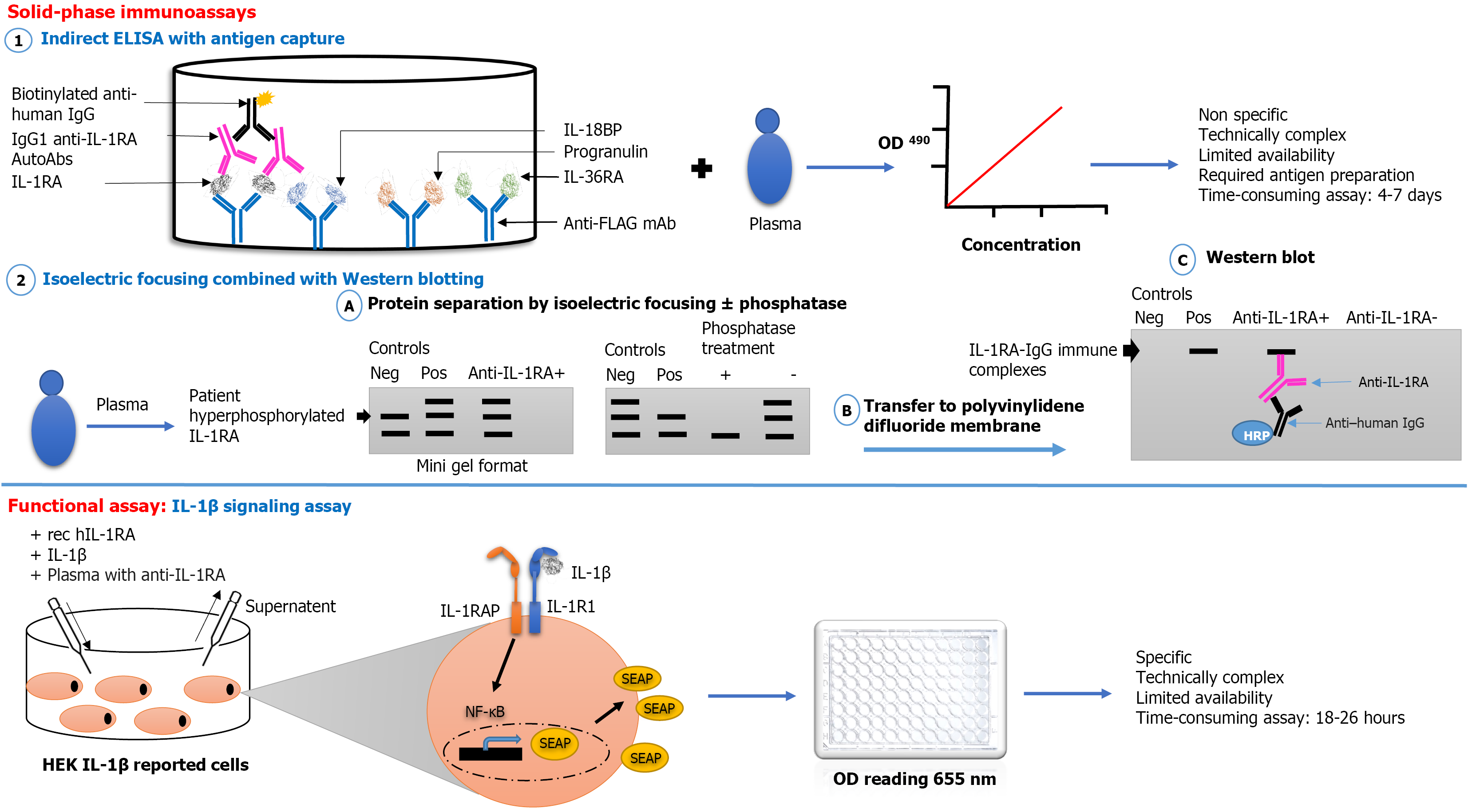
Laboratory detection and characterization of anti-IL-1RA AAbs in Still’s disease patients are performed using solid-phase immunoassays followed by functional IL-1β activation assays[7].
Detection and quantification of anti-IL-1RA AAbs are performed using in-house indirect enzyme-linked immunosorbent assays (ELISA) with antigen capture (Figure 2). Patient serum or plasma samples are incubated in microtiter wells coated with recombinant progranulin, IL-1RA, IL-18BP, and IL-36RA antigens[28]. Antibody binding is subsequently quantified by measuring optical density at about 490 nm. Notably, lower anti-IL-1RA titers are generally observed in Still’s disease[7] compared with those reported in vaccine-induced myocarditis[12] and MIS-C[13] (Supplementary Table 1). Despite its widespread use, this method has several limitations, including the need for antigen preparation[29], susceptibility to assay variability, lack of standardized cut-offs, and limited reproducibility. In addition, diagnostic specificity is suboptimal, as IL-1RA AAbs have also been detected in several inflammatory and autoimmune conditions[30] and even in some healthy individuals[12]. Accordingly, interpretation should always be guided by local laboratory protocols and integrated with the broader clinical context.
Isoelectric focusing (IEF) combined with alkaline phosphatase treatment allows identification of free hyperphosphorylated IL-1RA at Glycine98- Asparagine116 and Phenylalanine125- Glycine143[7,31]. Western blot analysis using antibodies directed against IL-1RA further confirmed the presence of IL-1RA-IgG immune complexes in the serum of patients with Still’s disease[7]. However, this approach does not provide information about the amount of complexed IL-1RA/anti-IL-1RA AAbs.
Overall, solid-phase immunoassays provide valuable information on the presence of IL-1RA AAbs but cannot differentiate pathogenic from clinically insignificant antibodies. Therefore, functional assays are essential to confirm their pathogenicity.
Functional IL-1β signaling reporter cell assays confirm that circulating anti-IL-1RA AutoAbs neutralize the inhibitory function of endogenous IL-1RA. This results in dysinhibition of IL-1β activity, which can be experimentally demonstrated by enhanced activation of NF-κB and MAPK signaling in reporter systems (Figure 2). This functional assay measures the secretion of embryonic alkaline phosphatase by human embryonic kidney IL-1β reporter cells following incubation with patient serum containing anti-IL-1RA antibodies, IL-1β, and recombinant human IL-1RA[7,12,13]. In the presence of clinically relevant anti-IL-1RA AutoAbs, binding to recombinant human IL-1RA impedes its antagonistic function, thereby allowing endogenous IL-1β to engage its receptor on human embryonic kidney cells and trigger secretion of embryonic alkaline phosphatase. The quantitative format of the assay enables measurement of optical density at 655 nm[7]. The IL-1β signaling reporter cell assay, however, is a time-consuming assay, expensive to perform, and requires technical expertise. Prospective studies will be required to determine the relationship between antibody levels and observed dysfunction of IL-1RA bioactivity. Given the potential implications of IL-1 targeting therapies, it is crucial to validate clinical measurements of anti-IL1RA antibodies in more than one center, ensuring analytical parameters such as specificity, predictive value, precision, accuracy, and robustness are rigorously assessed. These laboratory performances should adhere to regulatory guidelines such as those outlined by the International Council for Harmonization[32].
Taken together, a combined triple approach using ELISA, IEF/western blot, and functional assays provides the most comprehensive evaluation of anti-IL-1RA AAbs and enhances understanding of their role in Still’s disease.
Still’s disease is characterized by increased levels of several AAbs, including IL-1RA AAbs[7], antinuclear antibodies[7,9,33], DEK proto-oncogene AAbs[34], rheumatoid factors, and anticyclic citrullinated peptide antibodies[35]. However, only a subset of patients develops IgG Nabs against hyperphosphorylated IL-1RA, which can impair IL-1RA levels and bioactivity in vitro[7]. Their generation appears to result from an interplay between intrinsic properties of IL-1RA (e.g., phosphorylation) and host-specific determinants (e.g., genetic susceptibility and immune regulation).
The pivotal importance of class-switched, high-affinity IgG1 anti-IL-1RA AAbs in the pathophysiology of Still’s disease implies cooperation between B and CD4+ T cells (Supplementary Table 1). Activation of IL-1RA-specific CD4+ T lym
Beyond HLA, genetic regulation of immune tolerance also appears relevant. The forkhead box P3 (FOXP3) gene is a key transcription factor required for regulatory T cell (Treg) development and function[39]. Mutations or reduced expression of FOXP3 impair Treg-mediated immune tolerance, leading to the development of AAbs[40,41] and increased susceptibility to several autoimmune diseases[42,43]. In AOSD, FOXP3 expression is reduced in Tregs and inversely correlated with intracellular cytokine levels of interferon gamma, IL-17, and IL-4[44]. Another study found that circulating Treg cells were significantly decreased in AOSD patients[45]. Hence, it is evident that a complete defect in Tregs results in immune system dysfunction in patients with AOSD. However, the role of this defect in the development of IL-1RA AAbs remains unknown.
As previously discussed, the balance between IL-1β and its receptor antagonist IL-1RA plays an important role in the development of Still’s disease. IL-1β and IL-1RA gene polymorphisms are not associated with the development or clinical features of AOSD[46]. However, post-translational modifications of IL-1RA, particularly temporary hyperphosphorylation at threonine 111, may act as an additional immunogenic trigger preceding the loss of peripheral tolerance[12]. Dysregulation of protein phosphatase 2A, a key serine/threonine phosphatase involved in pathways such as extra
In addition to genetic factors, various environmental triggers are likely to contribute to the development of IL-1RA AAbs. Recent evidence suggests that severe acute respiratory distress syndrome corona virus-2 infection and vaccination may transiently induce AAb response leading to IL-1RA depletion[12,13,30]. Other infectious agents, particularly viruses, may enhance self-antigen immunogenicity through post-translational modifications such as phosphorylation, promoting loss of immune tolerance and potentially contributing to Still’s disease[51-54].
Anti-IL-1RA AAbs contribute to the depletion of IL-1RA levels and bioactivity in vitro[30]. Nevertheless, their presence has not been associated with disease activity, disability progression, or clinical response to anakinra in Still’s disease[7]. Similarly, no consistent evidence supports an association between these AAbs and clinical presentation or disease severity in patients with MIS-C[13]. By contrast, some studies suggest that these AAbs may predict disease activity in vaccine-associated myocarditis and IgG4-RD[30]. It is therefore likely that the detection of anti-IL-1RA AAbs is best interpreted as a marker of underlying immune dysregulation rather than a direct pathogenic driver. This hypothesis is consistent with the findings of Fijolek et al[55], who reported that antineutrophil cytoplasmic antibody levels were associated with pronounced proinflammatory effects in vitro without a clear in vivo correlate.
The functional consequences of anti-IL-1RA AAbs may also be modulated by additional regulatory factors within the IL-1 pathway, including soluble IL-1RI and soluble or membrane-bound IL-1R2[56]. Further studies are needed to clarify this hypothesis and to define the mechanisms involved.
IL-1-targeted biologics are therapeutics that interfere with IL-1α/β signaling by either blocking the IL-1 receptor or directly neutralizing IL-1 ligands (Figure 1). Anakinra is a recombinant form of the human soluble IL-1RA that competitively blocks IL-1R1 and has been associated with complete remission in patients with refractory AOSD[57]. Because of its short half-life, it requires once-daily subcutaneous administration.
Canakinumab, a fully human IgG1κ monoclonal antibody (about 150 kDa) that binds and neutralizes IL-1β, has also shown efficacy in Still’s disease[58]; its longer half-life allows for less frequent subcutaneous dosing.
Rilonacept, a soluble decoy receptor fusion protein of about 250 kDa that traps both IL-1α and IL-1β, has demonstrated clinical efficacy in Still’s disease[59]. Although antidrug antibodies have been reported during anakinra therapy in patients with rheumatoid arthritis[60], juvenile idiopathic arthritis[61], and cryopyrin-associated periodic syndromes[62], they are typically transient and do not affect safety or efficacy. In Still’s disease, however, anti–IL-1RA AAbs arise independently of prior anakinra exposure, indicating an endogenous origin[7]. Notably, both anakinra and canakinumab remain effective despite autoantibody-mediated reductions in IL-1RA bioactivity, underscoring the preserved efficacy of IL-1 blockade.
The detection of IL-1RA AAbs in a subset of patients with Still’s disease adds valuable insight into the complex immunopathogenesis of this condition. These AAbs are best considered candidate supporting biomarkers that reflect IL-1RA dysregulation in a defined subgroup and therefore require independent validation in larger, prospective cohorts. Their detection may be clinically useful in diagnostically challenging cases, could help biologically stratify patients for IL-1-targeted therapies, and may inform personalized treatment strategies. Multicenter, prospective studies with long-term follow-up are required to establish their true diagnostic and prognostic value and to determine whether AAb status predicts response to IL-1 blockade.
The mechanisms underlying the development of these AAbs remain incompletely understood. Future research should focus on elucidating how HLA polymorphism and the presentation of hyperphosphorylated IL-1RA peptides contribute to AAb generation. These investigations should also explore the interplay between environmental factors, such as viral infections, and HLA class II-associated risk alleles in shaping the IL-1RA autoantibody response.
Currently, laboratory testing for these AAbs is highly dependent on the ELISA protocol employed, underscoring the need for assay standardization to ensure analytical accuracy. Routine detection is not yet implemented in most clinical laboratories, and functional assessments remain confined to specialized, high-complexity research settings. Flow cytometry-based approaches, including phospho-signaling or cytokine readouts, may complement reporter assays. They can demonstrate IL-1β pathway activation by neutralizing antibodies, providing indirect evidence of IL-1RA inhibition in vitro and ex vivo. Although not yet widely available, such approaches have been established for interferon gamma and granulocyte-macrophage colony stimulating factor AAbs by leveraging knowledge of their signaling pathways.
Beyond IL-1-targeted biologics, autoreactive B cells producing pathogenic anti-IL-1RA AAbs may represent a promising therapeutic target in refractory Still’s disease. Although evidence remains limited to case reports and small series, B-cell depletion with anti-CD20 antibodies (e.g., rituximab) has shown potential efficacy in select patients[63,64]. Randomized studies are needed to confirm these observations. Furthermore, tolerance-inducing strategies, such as regulatory T-cell approaches or antigen-specific tolerization, may represent rational next steps and warrant rigorous preclinical and early-phase clinical evaluation to enable personalized therapeutic strategies.
| 1. | Lee JJY, Schneider R. Systemic Juvenile Idiopathic Arthritis. Pediatr Clin North Am. 2018;65:691-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 104] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 2. | Giacomelli R, Ruscitti P, Shoenfeld Y. A comprehensive review on adult onset Still's disease. J Autoimmun. 2018;93:24-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 303] [Article Influence: 37.9] [Reference Citation Analysis (0)] |
| 3. | Feist E, Mitrovic S, Fautrel B. Mechanisms, biomarkers and targets for adult-onset Still's disease. Nat Rev Rheumatol. 2018;14:603-618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 158] [Cited by in RCA: 283] [Article Influence: 40.4] [Reference Citation Analysis (8)] |
| 4. | Fautrel B, Mitrovic S, De Matteis A, Bindoli S, Antón J, Belot A, Bracaglia C, Constantin T, Dagna L, Di Bartolo A, Feist E, Foell D, Gattorno M, Georgin-Lavialle S, Giacomelli R, Grom AA, Jamilloux Y, Laskari K, Lazar C, Minoia F, Nigrovic PA, Oliveira Ramos F, Ozen S, Quartier P, Ruscitti P, Sag E, Savic S, Truchetet ME, Vastert SJ, Wilhelmer TC, Wouters C, Carmona L, De Benedetti F. EULAR/PReS recommendations for the diagnosis and management of Still's disease, comprising systemic juvenile idiopathic arthritis and adult-onset Still's disease. Ann Rheum Dis. 2024;83:1614-1627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 115] [Article Influence: 57.5] [Reference Citation Analysis (0)] |
| 5. | Choi JH, Suh CH, Lee YM, Suh YJ, Lee SK, Kim SS, Nahm DH, Park HS. Serum cytokine profiles in patients with adult onset Still's disease. J Rheumatol. 2003;30:2422-2427. [PubMed] |
| 6. | Jia J, Wang M, Meng J, Ma Y, Wang Y, Miao N, Teng J, Zhu D, Shi H, Sun Y, Liu H, Cheng X, Su Y, Ye J, Chi H, Liu T, Zhou Z, Wan L, Chen X, Wang F, Zhang H, Ben J, Wang J, Yang C, Hu Q. Ferritin triggers neutrophil extracellular trap-mediated cytokine storm through Msr1 contributing to adult-onset Still's disease pathogenesis. Nat Commun. 2022;13:6804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 63] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 7. | Hoffmann MC, Cavalli G, Fadle N, Cantoni E, Regitz E, Fleser O, Klemm P, Zaks M, Stöger E, Campochiaro C, Tomelleri A, Baldissera E, Bittenbring JT, Zimmer V, Pfeifer J, Fischer Y, Preuss KD, Bewarder M, Thurner B, Fuehner S, Foell D, Dagna L, Kessel C, Thurner L. Autoantibody-Mediated Depletion of IL-1RA in Still's Disease and Potential Impact of IL-1 Targeting Therapies. J Clin Immunol. 2024;44:45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 8. | Nagpure K, Raju P, Dube AH, Verma I, Kumbhalkar S. Chronic Adult-Onset Still's Disease With Positive Antinuclear Antibodies: Navigating Diagnostic Dilemmas and Clinical Implications. Cureus. 2024;16:e56776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 9. | Hirooka Y, Okuda S, Sugiyama M, Shiga T, Nozaki Y, Kinoshita K, Funauchi M, Matsumura I. Case Report: A Rare Case of Elderly-Onset Adult-Onset Still's Disease in a Patient With Systemic Lupus Erythematosus. Front Immunol. 2022;13:822169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 10. | Fujita Y, Furukawa H, Asano T, Sato S, Yashiro Furuya M, Kobayashi H, Watanabe H, Suzuki E, Koga T, Shimizu T, Ueki Y, Eguchi K, Tsuchiya N, Kawakami A, Migita K. HLA-DQB1 DPB1 alleles in Japanese patients with adult-onset Still's disease. Mod Rheumatol. 2019;29:843-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Ombrello MJ, Remmers EF, Tachmazidou I, Grom A, Foell D, Haas JP, Martini A, Gattorno M, Özen S, Prahalad S, Zeft AS, Bohnsack JF, Mellins ED, Ilowite NT, Russo R, Len C, Hilario MO, Oliveira S, Yeung RS, Rosenberg A, Wedderburn LR, Anton J, Schwarz T, Hinks A, Bilginer Y, Park J, Cobb J, Satorius CL, Han B, Baskin E, Signa S, Duerr RH, Achkar JP, Kamboh MI, Kaufman KM, Kottyan LC, Pinto D, Scherer SW, Alarcón-Riquelme ME, Docampo E, Estivill X, Gül A; British Society of Pediatric and Adolescent Rheumatology (BSPAR) Study Group; Childhood Arthritis Prospective Study (CAPS) Group; Randomized Placebo Phase Study of Rilonacept in sJIA (RAPPORT) Investigators; Sparks-Childhood Arthritis Response to Medication Study (CHARMS) Group; Biologically Based Outcome Predictors in JIA (BBOP) Group, de Bakker PI, Raychaudhuri S, Langefeld CD, Thompson S, Zeggini E, Thomson W, Kastner DL, Woo P; International Childhood Arthritis Genetics (INCHARGE) Consortium. HLA-DRB1*11 and variants of the MHC class II locus are strong risk factors for systemic juvenile idiopathic arthritis. Proc Natl Acad Sci U S A. 2015;112:15970-15975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 135] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 12. | Thurner L, Kessel C, Fadle N, Regitz E, Seidel F, Kindermann I, Lohse S, Kos I, Tschöpe C, Kheiroddin P, Kiblboeck D, Hoffmann MC, Bette B, Carbon G, Cetin O, Preuss KD, Christofyllakis K, Bittenbring JT, Pickardt T, Fischer Y, Thiele H, Baldus S, Stangl K, Steiner S, Gietzen F, Kerber S, Deneke T, Jellinghaus S, Linke A, Ibrahim K, Grabmaier U, Massberg S, Thilo C, Greulich S, Gawaz M, Mayatepek E, Meyer-Dobkowitz L, Kindermann M, Birk E, Birk M, Lainscak M, Foell D, Lepper PM, Bals R, Krawczyk M, Mevorach D, Hasin T, Keren A, Kabesch M, Abdul-Khaliq H, Smola S, Bewarder M, Thurner B, Böhm M, Pfeifer J, Klingel K. IL-1RA Antibodies in Myocarditis after SARS-CoV-2 Vaccination. N Engl J Med. 2022;387:1524-1527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 13. | Pfeifer J, Thurner B, Kessel C, Fadle N, Kheiroddin P, Regitz E, Hoffmann MC, Kos IA, Preuss KD, Fischer Y, Roemer K, Lohse S, Heyne K, Detemple MC, Fedlmeier M, Juenger H, Sauer H, Meyer S, Rohrer T, Wittkowski H, Becker SL, Masjosthusmann K, Bals R, Gerling S, Smola S, Bewarder M, Birk E, Keren A, Böhm M, Jakob A, Abdul-Khaliq H, Anton J, Kabesch M, Pino-Ramirez RM, Foell D, Thurner L. Autoantibodies against interleukin-1 receptor antagonist in multisystem inflammatory syndrome in children: a multicentre, retrospective, cohort study. Lancet Rheumatol. 2022;4:e329-e337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (1)] |
| 14. | Jarrell JA, Baker MC, Perugino CA, Liu H, Bloom MS, Maehara T, Wong HH, Lanz TV, Adamska JZ, Kongpachith S, Sokolove J, Stone JH, Pillai SS, Robinson WH. Neutralizing anti-IL-1 receptor antagonist autoantibodies induce inflammatory and fibrotic mediators in IgG4-related disease. J Allergy Clin Immunol. 2022;149:358-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 15. | Girard-Guyonvarc'h C, Rodriguez E, Mueller YM, Caruso A, Katsikis PD, Gabay C; ImmunAID consortium. Elevated serum levels of interleukin-18 discriminate Still's disease from other autoinflammatory conditions: results from the European ImmunAID cohort. RMD Open. 2025;11:e005388. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 16. | Girard-Guyonvarc'h C, Harel M, Gabay C. The Role of Interleukin 18/Interleukin 18-Binding Protein in Adult-Onset Still's Disease and Systemic Juvenile Idiopathic Arthritis. J Clin Med. 2022;11:430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Kötter I, Wacker A, Koch S, Henes J, Richter C, Engel A, Günaydin I, Kanz L. Anakinra in patients with treatment-resistant adult-onset Still's disease: four case reports with serial cytokine measurements and a review of the literature. Semin Arthritis Rheum. 2007;37:189-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 99] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 18. | Vigers GP, Caffes P, Evans RJ, Thompson RC, Eisenberg SP, Brandhuber BJ. X-ray structure of interleukin-1 receptor antagonist at 2.0-A resolution. J Biol Chem. 1994;269:12874-12879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 46] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 19. | Fields JK, Günther S, Sundberg EJ. Structural Basis of IL-1 Family Cytokine Signaling. Front Immunol. 2019;10:1412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 231] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 20. | Zhou L, Todorovic V, Kakavas S, Sielaff B, Medina L, Wang L, Sadhukhan R, Stockmann H, Richardson PL, DiGiammarino E, Sun C, Scott V. Quantitative ligand and receptor binding studies reveal the mechanism of interleukin-36 (IL-36) pathway activation. J Biol Chem. 2018;293:403-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 21. | Hoffmann MC, Fadle N, Regitz E, Kos IA, Cetin O, Lesan V, Preuss KD, Zaks M, Stöger E, Zimmer V, Klemm P, Assmann G, Pfeifer J, Bittenbring JT, Bewarder M, Vogt T, Pföhler C, Thurner B, Kessel C, Thurner L. Autoantibody mediated deficiency of IL-36-receptor antagonist in a subset of patients with psoriasis and psoriatic arthritis. Immunol Lett. 2024;270:106926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 22. | Hannum CH, Wilcox CJ, Arend WP, Joslin FG, Dripps DJ, Heimdal PL, Armes LG, Sommer A, Eisenberg SP, Thompson RC. Interleukin-1 receptor antagonist activity of a human interleukin-1 inhibitor. Nature. 1990;343:336-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 805] [Cited by in RCA: 782] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 23. | Tahtinen S, Tong AJ, Himmels P, Oh J, Paler-Martinez A, Kim L, Wichner S, Oei Y, McCarron MJ, Freund EC, Amir ZA, de la Cruz CC, Haley B, Blanchette C, Schartner JM, Ye W, Yadav M, Sahin U, Delamarre L, Mellman I. IL-1 and IL-1ra are key regulators of the inflammatory response to RNA vaccines. Nat Immunol. 2022;23:532-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 382] [Article Influence: 95.5] [Reference Citation Analysis (0)] |
| 24. | Maeno N, Takei S, Imanaka H, Yamamoto K, Kuriwaki K, Kawano Y, Oda H. Increased interleukin-18 expression in bone marrow of a patient with systemic juvenile idiopathic arthritis and unrecognized macrophage-activation syndrome. Arthritis Rheum. 2004;50:1935-1938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 58] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 25. | Arlet JB, Le TH, Marinho A, Amoura Z, Wechsler B, Papo T, Piette JC. Reactive haemophagocytic syndrome in adult-onset Still's disease: a report of six patients and a review of the literature. Ann Rheum Dis. 2006;65:1596-1601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 161] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 26. | Auron PE. The interleukin 1 receptor: ligand interactions and signal transduction. Cytokine Growth Factor Rev. 1998;9:221-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 122] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 27. | O'Neill LA. Interleukin-1 signal transduction. Int J Clin Lab Res. 1995;25:169-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Thurner L, Preuss KD, Fadle N, Regitz E, Klemm P, Zaks M, Kemele M, Hasenfus A, Csernok E, Gross WL, Pasquali JL, Martin T, Bohle RM, Pfreundschuh M. Progranulin antibodies in autoimmune diseases. J Autoimmun. 2013;42:29-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Bornkamm GW, Berens C, Kuklik-Roos C, Bechet JM, Laux G, Bachl J, Korndoerfer M, Schlee M, Hölzel M, Malamoussi A, Chapman RD, Nimmerjahn F, Mautner J, Hillen W, Bujard H, Feuillard J. Stringent doxycycline-dependent control of gene activities using an episomal one-vector system. Nucleic Acids Res. 2005;33:e137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 119] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 30. | Bouayad A. IL-1RA autoantibodies: insights into mechanisms and associated diseases. Am J Transl Res. 2024;16:374-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 31. | Anderson JC, Peck SC. A simple and rapid technique for detecting protein phosphorylation using one-dimensional isoelectric focusing gels and immunoblot analysis. Plant J. 2008;55:881-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 32. | Sugiyama A, Goto A, Izumi-Nakaseko H, Takei Y, Takahara A, Kambayashi R. Drug-induced long QT syndrome: Concept and nonclinical models for predicting the onset of drug-induced torsade de pointes in patients in compliance with International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) E14/S7B guidance. J Pharmacol Exp Ther. 2025;392:100023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 33. | Glerup M, Herlin T, Twilt M. Remission rate is not dependent on the presence of antinuclear antibodies in juvenile idiopathic arthritis. Clin Rheumatol. 2017;36:671-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 34. | Mor-Vaknin N, Rivas M, Legendre M, Mohan S, Yuanfan Y, Mau T, Johnson A, Huang B, Zhao L, Kimura Y, Spalding SJ, Morris PW, Gottlieb BS, Onel K, Olson JC, Edelheit BS, Shishov M, Jung LK, Cassidy EA, Prahalad S, Passo MH, Beukelman T, Mehta J, Giannini EH, Adams BS, Lovell DJ, Markovitz DM. High Levels of DEK Autoantibodies in Sera of Patients With Polyarticular Juvenile Idiopathic Arthritis and With Early Disease Flares Following Cessation of Anti-Tumor Necrosis Factor Therapy. Arthritis Rheumatol. 2018;70:594-605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 35. | Syed RH, Gilliam BE, Moore TL. Rheumatoid factors and anticyclic citrullinated peptide antibodies in pediatric rheumatology. Curr Rheumatol Rep. 2008;10:156-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 36. | Teng JL, Chen X, Chen J, Zeng T, He L, Li M, Luo CN, Liu S, Ding TT, Yimaiti K, Li X, Ding Y, Cheng XB, Zhou J, Ye JN, Ji J, Su YT, Shi H, Sun Y, Gao C, Hu QY, Chi HH, Yuan X, Zhou ZC, Wang D, Wang K, Feng D, Li C, Sun Y, Niu Y, Xu X, Chen LJ, Xu J, Wu LJ, Zhou Z, Pan D, Niu H, Yang CD, Yongyong Shi, Li Z, Liu HL. The amino acid variants in HLA II molecules explain the major association with adult-onset Still's disease in the Han Chinese population. J Autoimmun. 2021;116:102562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 37. | Joung CI, Lee HS, Lee SW, Kim CG, Song YH, Jun JB, Chung WT, Choe JY, Kim TG, Yoo DH. Association between HLA-DR B1 and clinical features of adult onset Still's disease in Korea. Clin Exp Rheumatol. 2003;21:489-492. [PubMed] |
| 38. | Asano T, Furukawa H, Sato S, Yashiro M, Kobayashi H, Watanabe H, Suzuki E, Ito T, Ubara Y, Kobayashi D, Iwanaga N, Izumi Y, Fujikawa K, Yamasaki S, Nakamura T, Koga T, Shimizu T, Umeda M, Nonaka F, Yasunami M, Ueki Y, Eguchi K, Tsuchiya N, Tohma S, Yoshiura KI, Ohira H, Kawakami A, Migita K. Effects of HLA-DRB1 alleles on susceptibility and clinical manifestations in Japanese patients with adult onset Still's disease. Arthritis Res Ther. 2017;19:199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 39. | Lan Q, Fan H, Quesniaux V, Ryffel B, Liu Z, Zheng SG. Induced Foxp3(+) regulatory T cells: a potential new weapon to treat autoimmune and inflammatory diseases? J Mol Cell Biol. 2012;4:22-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 129] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 40. | Zhang W, Sharma R, Ju ST, He XS, Tao Y, Tsuneyama K, Tian Z, Lian ZX, Fu SM, Gershwin ME. Deficiency in regulatory T cells results in development of antimitochondrial antibodies and autoimmune cholangitis. Hepatology. 2009;49:545-552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 41. | Stadtlober NP, Flauzino T, da Rosa Franchi Santos LF, Iriyoda TMV, Costa NT, Lozovoy MAB, Dichi I, Reiche EMV, Simão ANC. Haplotypes of FOXP3 genetic variants are associated with susceptibility, autoantibodies, and TGF-β1 in patients with systemic lupus erythematosus. Sci Rep. 2021;11:5406. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 42. | Ehrenstein MR, Evans JG, Singh A, Moore S, Warnes G, Isenberg DA, Mauri C. Compromised function of regulatory T cells in rheumatoid arthritis and reversal by anti-TNFalpha therapy. J Exp Med. 2004;200:277-285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 985] [Cited by in RCA: 948] [Article Influence: 43.1] [Reference Citation Analysis (5)] |
| 43. | Lindley S, Dayan CM, Bishop A, Roep BO, Peakman M, Tree TI. Defective suppressor function in CD4(+)CD25(+) T-cells from patients with type 1 diabetes. Diabetes. 2005;54:92-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 616] [Cited by in RCA: 647] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 44. | Shimojima Y, Ichikawa T, Kishida D, Takamatsu R, Sekijima Y. Circulating regulatory T cells in adult-onset Still's disease: Focusing on their plasticity and stability. Clin Exp Immunol. 2021;206:184-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 45. | Chen DY, Chen YM, Chen HH, Hsieh CW, Lin CC, Lan JL. The associations of circulating CD4+CD25high regulatory T cells and TGF-β with disease activity and clinical course in patients with adult-onset Still's disease. Connect Tissue Res. 2010;51:370-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 46. | Youm JY, Woo JH, Kim TH, Bae SC, Yoo DH. Interleukin-1beta and interleukin-1 receptor antagonist gene polymorphisms in Korean patients with adult-onset Still's disease. Scand J Rheumatol. 2007;36:390-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 47. | Yu LG, Packman LC, Weldon M, Hamlett J, Rhodes JM. Protein phosphatase 2A, a negative regulator of the ERK signaling pathway, is activated by tyrosine phosphorylation of putative HLA class II-associated protein I (PHAPI)/pp32 in response to the antiproliferative lectin, jacalin. J Biol Chem. 2004;279:41377-41383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 48. | Preuss KD, Pfreundschuh M, Fadle N, Regitz E, Raudies S, Murwaski N, Ahlgrimm M, Bittenbring J, Klotz M, Schäfer KH, Held G, Neumann F, Grass S. Hyperphosphorylation of autoantigenic targets of paraproteins is due to inactivation of PP2A. Blood. 2011;118:3340-3346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 49. | Grass S, Preuss KD, Wikowicz A, Terpos E, Ziepert M, Nikolaus D, Yang Y, Fadle N, Regitz E, Dimopoulos MA, Treon SP, Hunter ZR, Pfreundschuh M. Hyperphosphorylated paratarg-7: a new molecularly defined risk factor for monoclonal gammopathy of undetermined significance of the IgM type and Waldenstrom macroglobulinemia. Blood. 2011;117:2918-2923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 50. | Grass S, Preuss K, Wikowicz A, Terpos E, Ziepert M, Ahlgrimm M, Fadle N, Regitz E, Murawski N, Dimopoulos MA, Treon SP, Hunter Z, Pfreundschuh M. Hyperphosphorylated Paratarg-7 Is a Frequent Antigenic Target of IgM Paraproteins, Is Dominantly Inherited and Represents a Highly Significant Risk Factor for Monoclonal Gammopathy of Undetermined Significance of the IgM Type (IgM-MGUS) and Waldenstrom's Macroglobulinemia (WM), Allowing for the Identification of Family Members at Risk in Cases of Familial IgM-MGUS and WM. Blood. 2009;114:3935-3935. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 51. | Altuzarra-Ranedo Y, Gómez-Ramírez D, Rodríguez-Laguna M, Lois-Bermejo PM, López-Pelaez B, Lorenzo-Villalba N, Méndez-Bailon M. Parvovirus Infection Triggering Still's Disease. Eur J Case Rep Intern Med. 2022;9:003122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 52. | Fet-He S, Ibarra Lecompte G, Quiroz Alfaro AJ. Association between adult-onset still's disease and COVID-19: A report of two cases and brief review. SAGE Open Med Case Rep. 2024;12:2050313X241233197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 53. | Jia J, Shi H, Liu M, Liu T, Gu J, Wan L, Teng J, Liu H, Cheng X, Ye J, Su Y, Sun Y, Gong W, Yang C, Hu Q. Cytomegalovirus Infection May Trigger Adult-Onset Still's Disease Onset or Relapses. Front Immunol. 2019;10:898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 54. | Wang JY, Zhang W, Roehrl MW, Roehrl VB, Roehrl MH. An autoantigen profile of human A549 lung cells reveals viral and host etiologic molecular attributes of autoimmunity in COVID-19. J Autoimmun. 2021;120:102644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 55. | Fijolek J, Wiatr E, Petroniec V, Augustynowicz-Kopec E, Bednarek M, Gawryluk D, Roszkowski-Sliz K. Antineutrophil cytoplasmic antibodies and their relationship with disease activity and presence of staphylococcal superantigens in nasal swabs in patients having granulomatosis with polyangiitis: results of a study involving 115 patients from a single center. Clin Rheumatol. 2019;38:3297-3305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 56. | Gabay C, Lamacchia C, Palmer G. IL-1 pathways in inflammation and human diseases. Nat Rev Rheumatol. 2010;6:232-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 773] [Cited by in RCA: 686] [Article Influence: 42.9] [Reference Citation Analysis (0)] |
| 57. | Giacomelli R, Sota J, Ruscitti P, Campochiaro C, Colafrancesco S, Dagna L, Iacono D, Iannone F, Lopalco G, Sfriso P, Cantarini L. The treatment of adult-onset Still's disease with anakinra, a recombinant human IL-1 receptor antagonist: a systematic review of literature. Clin Exp Rheumatol. 2021;39:187-195. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 58. | Vitale A, Caggiano V, Sfikakis PP, Dagna L, Lopalco G, Ragab G, La Torre F, Almaghlouth IA, Maggio MC, Sota J, Tufan A, Hinojosa-Azaola A, Iannone F, Loconte R, Laskari K, Direskeneli H, Ruscitti P, Morrone M, Mayrink Giardini HA, Panagiotopoulos A, Di Cola I, Martín-Nares E, Monti S, De Stefano L, Kardas RC, Duran R, Campochiaro C, Tomelleri A, Alabdulkareem AM, Gaggiano C, Tarsia M, Bartoloni E, Romeo M, Hussein MA, Laymouna AH, Parente de Brito Antonelli I, Dagostin MA, Fotis L, Bindoli S, Navarini L, Alibaz-Oner F, Sevik G, Frassi M, Ciccia F, Iacono D, Crisafulli F, Portincasa P, Jaber N, Kawakami-Campos PA, Wiesik-Szewczyk E, Iagnocco A, Simonini G, Sfriso P, Balistreri A, Giacomelli R, Conti G, Frediani B, Fabiani C, Cantarini L. Efficacy of canakinumab in patients with Still's disease across different lines of biologic therapy: real-life data from the International AIDA Network Registry for Still's Disease. Front Med (Lausanne). 2023;10:1256243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 59. | Arnold DD, Yalamanoglu A, Boyman O. Systematic Review of Safety and Efficacy of IL-1-Targeted Biologics in Treating Immune-Mediated Disorders. Front Immunol. 2022;13:888392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 100] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 60. | Cohen S, Hurd E, Cush J, Schiff M, Weinblatt ME, Moreland LW, Kremer J, Bear MB, Rich WJ, McCabe D. Treatment of rheumatoid arthritis with anakinra, a recombinant human interleukin-1 receptor antagonist, in combination with methotrexate: results of a twenty-four-week, multicenter, randomized, double-blind, placebo-controlled trial. Arthritis Rheum. 2002;46:614-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 436] [Cited by in RCA: 424] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 61. | Ilowite N, Porras O, Reiff A, Rudge S, Punaro M, Martin A, Allen R, Harville T, Sun YN, Bevirt T, Aras G, Appleton B. Anakinra in the treatment of polyarticular-course juvenile rheumatoid arthritis: safety and preliminary efficacy results of a randomized multicenter study. Clin Rheumatol. 2009;28:129-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 100] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 62. | Wikén M, Hallén B, Kullenberg T, Koskinen LO. Development and effect of antibodies to anakinra during treatment of severe CAPS: sub-analysis of a long-term safety and efficacy study. Clin Rheumatol. 2018;37:3381-3386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 63. | Belfeki N, Smiti Khanfir M, Said F, Hamzaoui A, Ben Salem T, Ben Ghorbel I, Lamloum M, Houman MH. Successful treatment of refractory adult onset Still's disease with rituximab. Reumatismo. 2016;68:159-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 64. | Ahmadi-Simab K, Lamprecht P, Jankowiak C, Gross WL. Successful treatment of refractory adult onset Still's disease with rituximab. Ann Rheum Dis. 2006;65:1117-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 1.8] [Reference Citation Analysis (0)] |