Copyright: ©Author(s) 2026.
World J Methodol. Jun 20, 2026; 16(2): 114269
Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.114269
Published online Jun 20, 2026. doi: 10.5662/wjm.v16.i2.114269
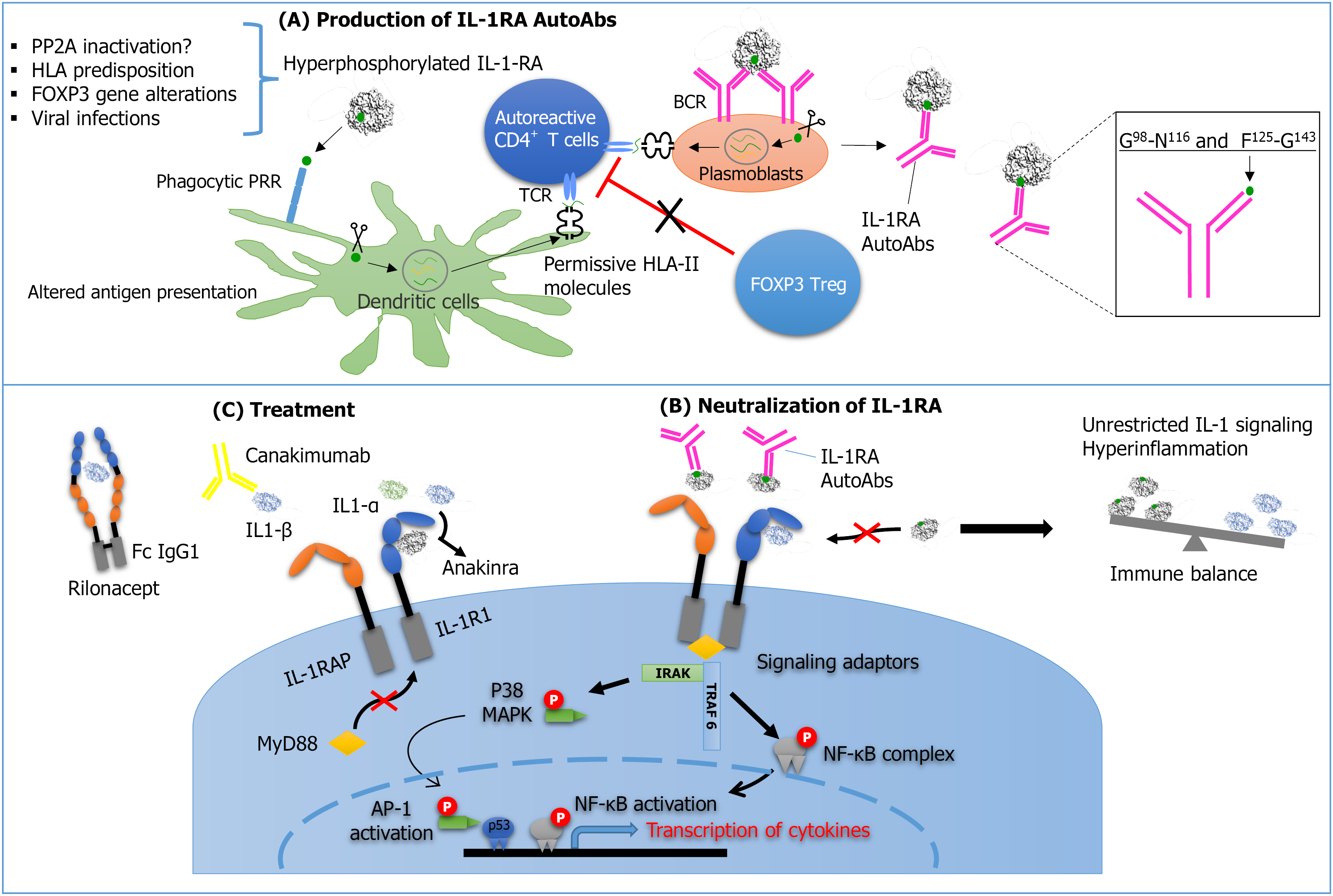
Figure 1 Immune response against hyperphosphorylated interleukin-1 receptor antagonist.
PP2A: Protein phosphatase 2A; HLA: Human leukocyte antigen; FOXP3: Forkhead box P3; IL-1-RA: Interleukin-1-receptor antagonist; BCR: B-cell receptor; PRR: Pattern recognition receptor; TCR: T-cell receptor; HLA-II: Human leukocyte antigen class II; IL-1RA AutoAbs: Autoantibodies against Interleukin-1 receptor antagonist; IL-1RAP: Interleukin-1 receptor accessory protein; p38 MAPK: P38 mitogen-activated protein kinase; NF-κB: Nuclear factor kappa B; IL-1RI: Interleukin-1 type-I receptor; IRAKs: Interleukin-1 receptor-associated kinases; MyD88: Myeloid differentiation factor 88; AP-1: Activator protein 1; TRAF6: Tumor necrosis factor receptor-associated factor 6.
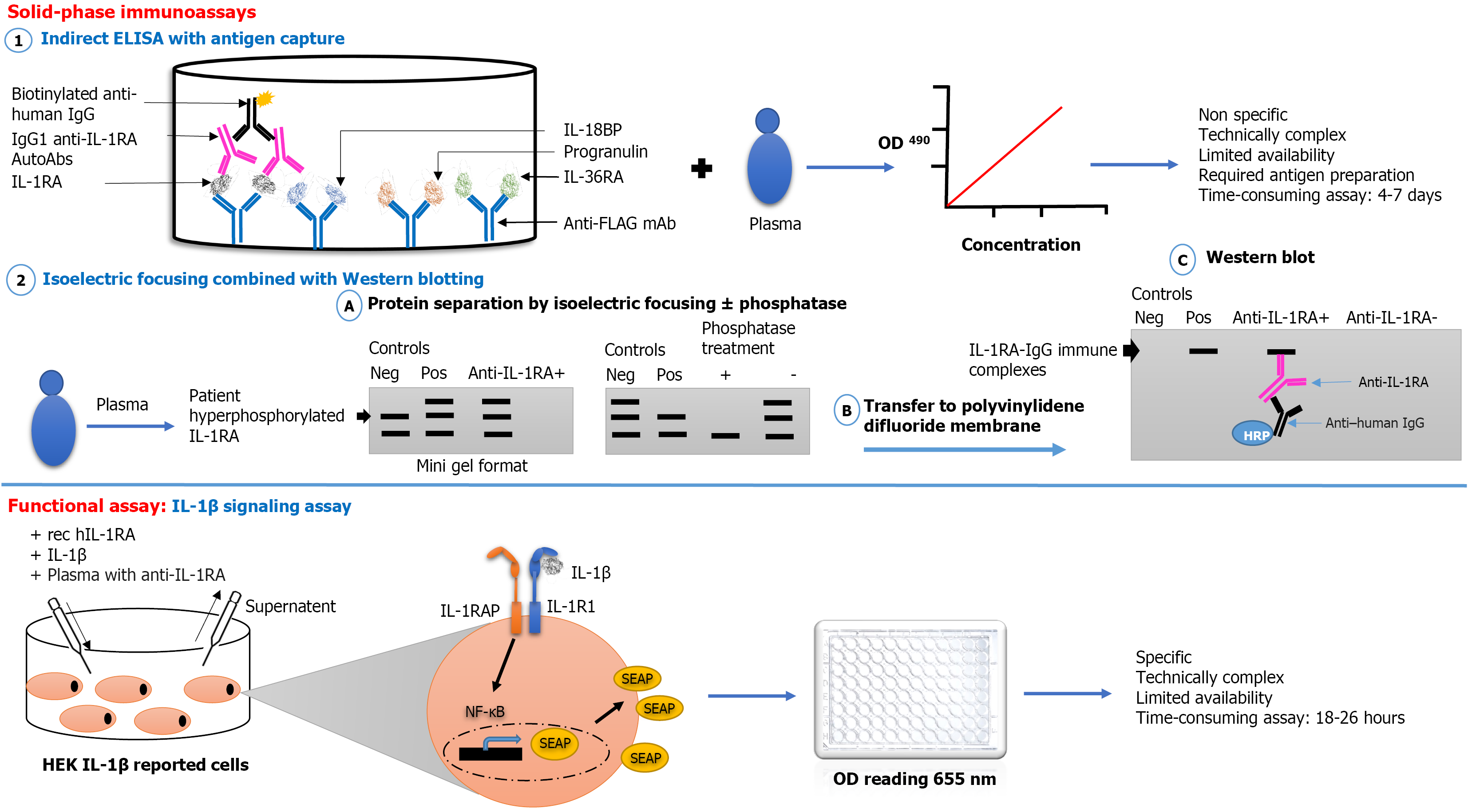
Figure 2 Approaches for the detection and characterization of anti-interleukin-1 receptor accessory autoantibodies.
SEAP: Secretion of embryonic alkaline phosphatase; rec hIL-1RA: Recombinant human interleukin-1 receptor antagonist; IL-1RAP: Interleukin-1 receptor accessory protein; IL-1R1: Interleukin-1 receptor type 1; OD: Optical density; HEK: Human embryonic kidney; Anti-FLAG mAb: Anti-FLAG monoclonal antibody; HRP: Horseradish peroxidase; ELISA: Enzyme-linked immunosorbent.
- Citation: Bouayad A. Interleukin-1 receptor antagonist autoantibodies in Still’s disease: Mechanistic insights and laboratory testing. World J Methodol 2026; 16(2): 114269
- URL: https://www.wjgnet.com/2222-0682/full/v16/i2/114269.htm
- DOI: https://dx.doi.org/10.5662/wjm.v16.i2.114269