Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.117173
Revised: January 2, 2026
Accepted: January 26, 2026
Published online: March 25, 2026
Processing time: 104 Days and 0.8 Hours
Diabetic kidney disease (DKD) remains a leading cause of end-stage renal disease (ESRD), with persistent proteinuria contributing to renal and cardiovascular complications. Despite dual therapy with renin-angiotensin-aldosterone system (RAAS) blockers and sodium-glucose co-transporter-2 inhibitors (SGLT2is), re
To evaluate the efficacy and safety of finerenone in reducing proteinuria in patients with DKD who were receiving maximally tolerated doses of dapagliflozin and telmisartan.
A retrospective, single-center observational study evaluated 33 patients with DKD and residual proteinuria. All patients received finerenone 10 mg daily for 24 weeks. Key parameters assessed at baseline and follow-up included urinary albumin-to-creatinine ratio (UACR), estimated glomerular filtration rate, and serum potassium. Respon
The mean reduction in UACR was 9.5% (P = 0.256). Twenty-two patients (66.7%) responded to treatment with a reduction in UACR, while 11 patients (33.3%) showed no reduction. When stratified by baseline albuminuria, responders with initial UACR < 1000 mg/g, 1000-3000 mg/g, and > 3000 mg/g showed average reductions of -47.9%, -39.0%, and -32.8%, respectively, suggesting a gradient treatment effect. Finerenone was well tolerated with mild hyperkalemia (serum potassium > 5.0 mmol/L) noted in 15.2% of patients with mean increase in serum po
Finerenone was associated with an overall reduction in albuminuria, with the magnitude of response varying according to baseline UACR. Greater proportional reductions were observed in patients with lower baseline al
Core Tip: Diabetic nephropathy is the leading cause of end-stage renal disease worldwide and proteinuria reduction is the cornerstone in the management. In some patients even with dual therapy with renin-angiotensin-aldosterone system blockers and sodium-glucose co-transporter-2 inhibitors, residual proteinuria persists. Finerenone, a novel nonsteroidal mineralocorticoid receptor antagonist has shown results reducing albuminuria and slowing chronic kidney disease progression. This study evaluated the additive effect of finerenone exclusively in patients receiving maximum tolerable doses of both dapagliflozin and telmisartan unlike previously available studies evaluating its effect on patients with maximum tolerated dose of angiotensin receptor blocker alone.
- Citation: Pasari AS, Gupta S, Gurjar P, Ramteke V, Malde S, Pawar T, Jeyachandran V, Kashiv P, Dubey S, Kurundwadkar M, Sejpal K, Bawankule C, Deshpande N, Balwani MR. Efficacy of finerenone in reducing proteinuria in diabetic kidney disease with maximum tolerable doses of dapagliflozin and telmisartan. World J Nephrol 2026; 15(1): 117173
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/117173.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.117173
Diabetic kidney disease (DKD) remains one of the leading causes of end-stage renal disease (ESRD) worldwide, signi
Telmisartan, an angiotensin receptor blocker, has demonstrated efficacy in reducing proteinuria and slowing DKD progression through its inhibition of RAAS pathway critical in DKD pathogenesis[3]. Dapagliflozin, a potent SGLT2 in
Finerenone, a novel nonsteroidal mineralocorticoid receptor antagonist (MRA) has been effective in reducing pro
The aim of the study was to evaluate the efficacy of finerenone in reducing proteinuria and asses its safety in terms of changes in serum potassium and estimated glomerular filtration rate (eGFR) in patients with DKD who are on maximum tolerable doses of telmisartan and dapagliflozin. This was a retrospective, single-center observational study conducted to evaluate the efficacy of finerenone in reducing proteinuria in patients with diabetic nephropathy. The study included pa
(1) Patients with type 2 diabetes mellitus and DKD diagnosed by having urinary albumin-to-creatinine ratio (UACR) > 30 mg/g creatinine; (2) eGFR > 25 mL/minute/1.73 m2; (3) Serum potassium level < 5.0 mmol/L; and (4) Stable treatment with maximum tolerable doses of telmisartan and dapagliflozin for at least 4 weeks prior to study initiation.
(1) eGFR < 25 mL/minute/1.73 m2; (2) Serum potassium level > 5.0 mmol/L; (3) Pregnancy or lactation; and (4) Patients with type 1 diabetes mellitus.
A total of 35 patients were screened for eligibility. Of these, 2 patients were excluded due to hyperkalaemia resulting in 33 patients meeting the inclusion criteria and participating in the study. Baseline characteristics, including demographic data, eGFR, serum potassium, and proteinuria levels, were recorded for all participants.
All enrolled patients received finerenone at a dose of 10 mg daily. The treatment period lasted for 24 weeks, during which patients continued their existing regimen of telmisartan and dapagliflozin.
The primary outcome measure was the change in proteinuria, assessed by the UACR from baseline to 24 weeks. Secondary outcome measures included: Changes in eGFR from baseline to 24 weeks. Changes in serum potassium levels from baseline to 24 weeks. Adverse events, including hyperkalaemia and hypotension, monitored throughout the study.
Urine and blood samples were collected at baseline and after 24 weeks of treatment to assess UACR, eGFR, and serum potassium concentrations. Clinical data were entered into a secure digital database and anonymized prior to analysis.
Statistical analysis was performed using IBM SPSS Statistics (Version 28.0) and R (Version 4.2.2). Visualizations were created using GraphPad Prism (Version 9.5.1) and R. Continuous variables were reported as mean ± SD, and categorical variables as n (%).
Comparisons between groups were conducted using independent t-tests or Mann-Whitney U tests, depending on data distribution. Paired t-tests or Wilcoxon signed-rank tests were used for intra-individual comparisons. Normality was assessed using the Shapiro-Wilk test. Effect sizes were calculated using Cohen’s d, and a two-tailed P value < 0.05 was considered statistically significant. Subgroup analyses and summary visualizations were used to explore differential treatment responses, particularly in relation to baseline UACR categories.
A total of 35 patients were screened for eligibility. Two patients were excluded from the study due to baseline serum potassium levels > 5.0 mmol/L resulting in a final study cohort of 33 patients who met the inclusion criteria. All patients received 10 mg of finerenone daily for a duration of 24 weeks, while continuing background therapy with maximum tolerable doses of dapagliflozin and telmisartan. The mean age of patients was 59.5 ± 10.4 years, with a majority being male patients (90.9%). The average duration of diabetes was 207.7 ± 106.8 months. The mean baseline eGFR was 36.3 ± 10.2 mL/minute/1.73 m2, and the mean serum creatinine level was 2.14 ± 0.52 mg/dL. Median baseline UACR was 1304.0 mg/g, with a wide range (7.53-9699.00 mg/g), indicating considerable variability in baseline proteinuria. The mean baseline serum potassium level was 4.40 ± 0.45 mmol/L (Table 1).
| Characteristic | Value |
| Number of participants | 33 |
| Age (years) | 59.5 ± 10.4 |
| Gender (male) | 30 (90.9) |
| Baseline eGFR (mL/minute/1.73 m2) | 36.3 ± 10.2 |
| Baseline serum creatinine (mg/dL) | 2.11 ± 0.43 |
| Baseline UACR (mg/g) | Median: 1656.0 (range: 5.57-9699.00) |
| Baseline serum potassium (mmol/L) | 4.33 ± 0.53 |
| Duration of diabetes (years) | 17.3 ± 8.9 |
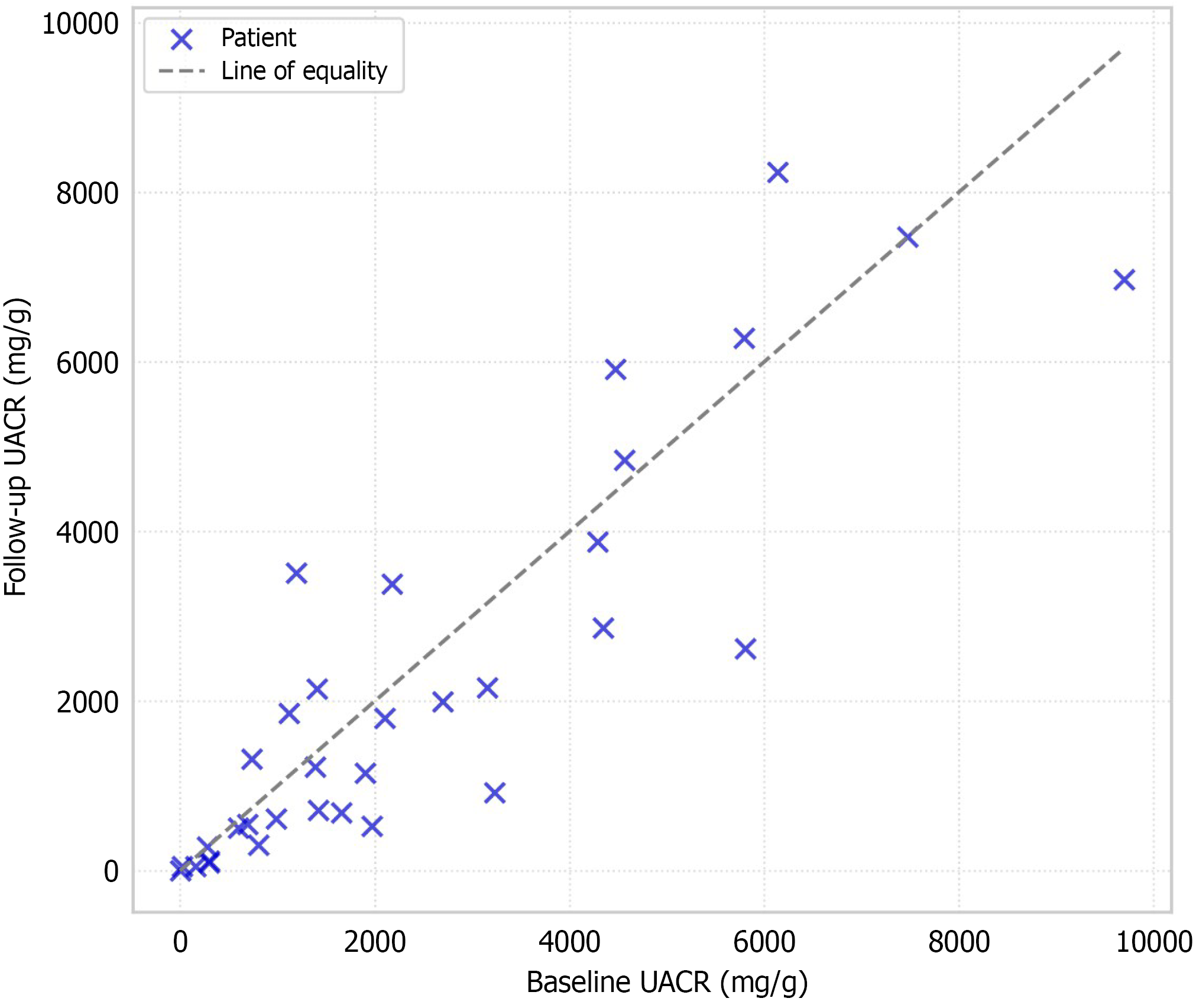
The primary outcome measure i.e. UACR, demonstrated substantial interpatient variability following 24 weeks of treatment with finerenone. The baseline median UACR was 1304.0 mg/g, with a range of 7.53-9699.00 mg/g. The mean UACR at baseline was 2512.7 mg/g (SD ± 2396.2) and on follow up the mean UACR was 2274.6 mg/g (SD ± 2379.8). There was a mean reduction of UACR by 9.5 percent, however, this change was not statistically significant (P = 0.256), and the effect size was small (Cohen’s d = -0.20), with a wide confidence interval (-2820.4 to +2143.9 mg/g) (Table 2). The maximum reduction in UACR observed was -73.3%, while the maximum increase was found to be +193.5% (Figures 1 and 2).
| Parameter | Baseline | Follow-up | Change (%) | P value | Effect size (Cohen’s d) | 95%CI |
| Serum potassium (mmol/L) | 4.33 ± 0.53 | 4.60 ± 0.50 | ↑ 6.4% | 0.016 | 0.45 | -0.75 to 1.55 |
| UACR (mg/g) | 2512.7 ± 2396.2 | 2274.6 ± 2379.8 | -9.5% | 0.256 | -0.20 | -2820.4 to 2143.9 |
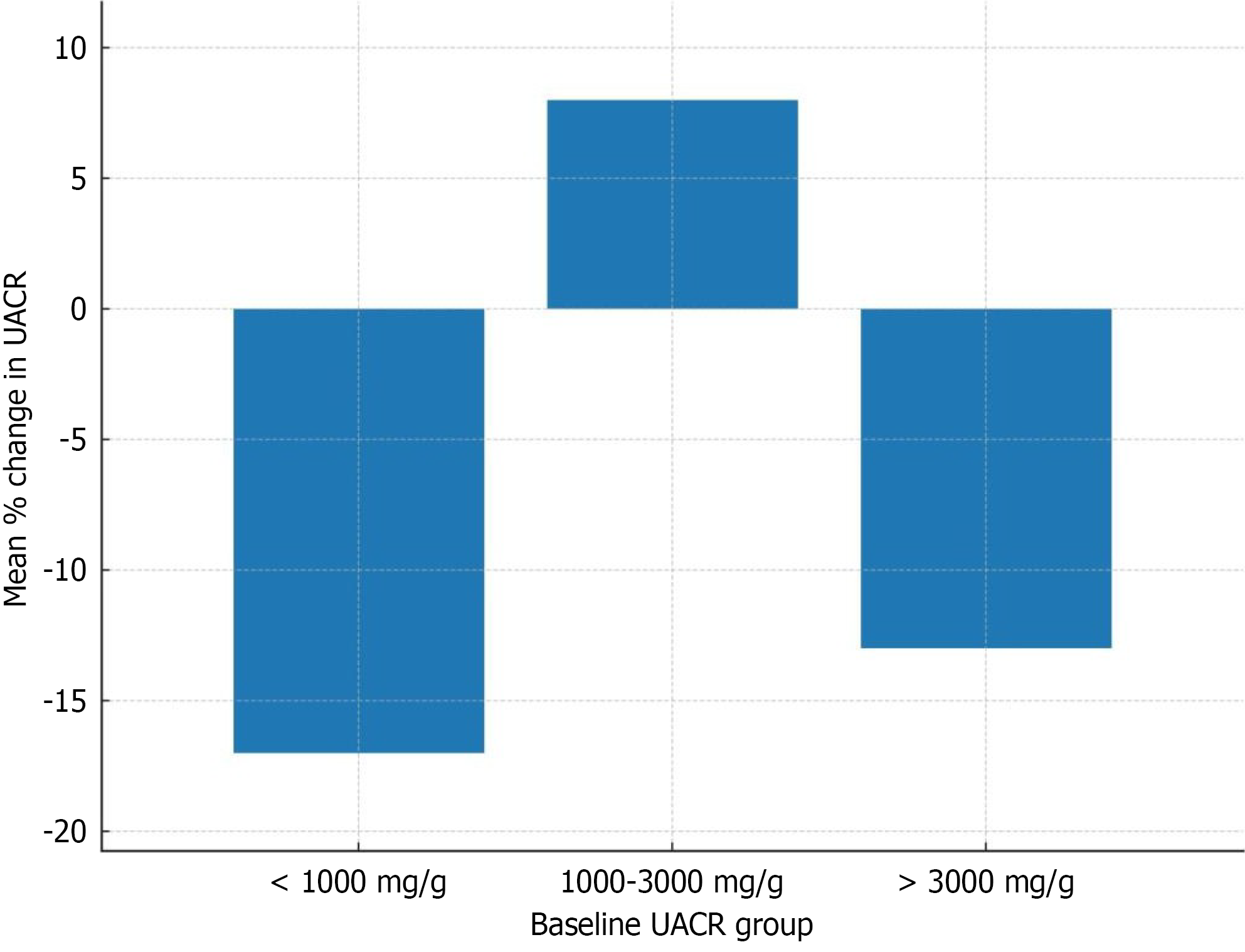
Patients were classified as responders and non-responders if they showed reduction in proteinuria and increase in proteinuria respectively. A total of 22 patients (66.7%) were classified as responders, whereas 11 patients (33.3%) were non-responders. Responders had a significantly higher baseline UACR compared to non-responders (2341.2 ± 1854.3 mg/g vs 1425.5 ± 998.7 mg/g, P = 0.028) (Figure 3). All patients (responders and non-responders) were additionally stratified into three groups according to their baseline UACR: < 1000 mg/g, 1000-3000 mg/g and > 3000 mg/g. Patients with baseline UACR < 1000 mg/g experienced a mean reduction of 17.1%, while those with baseline UACR > 3000 mg/g showed a 13.5% reduction. In contrast, the 1000-3000 mg/g group showed a slight mean increase (+8.6%) in UACR (Figure 4).
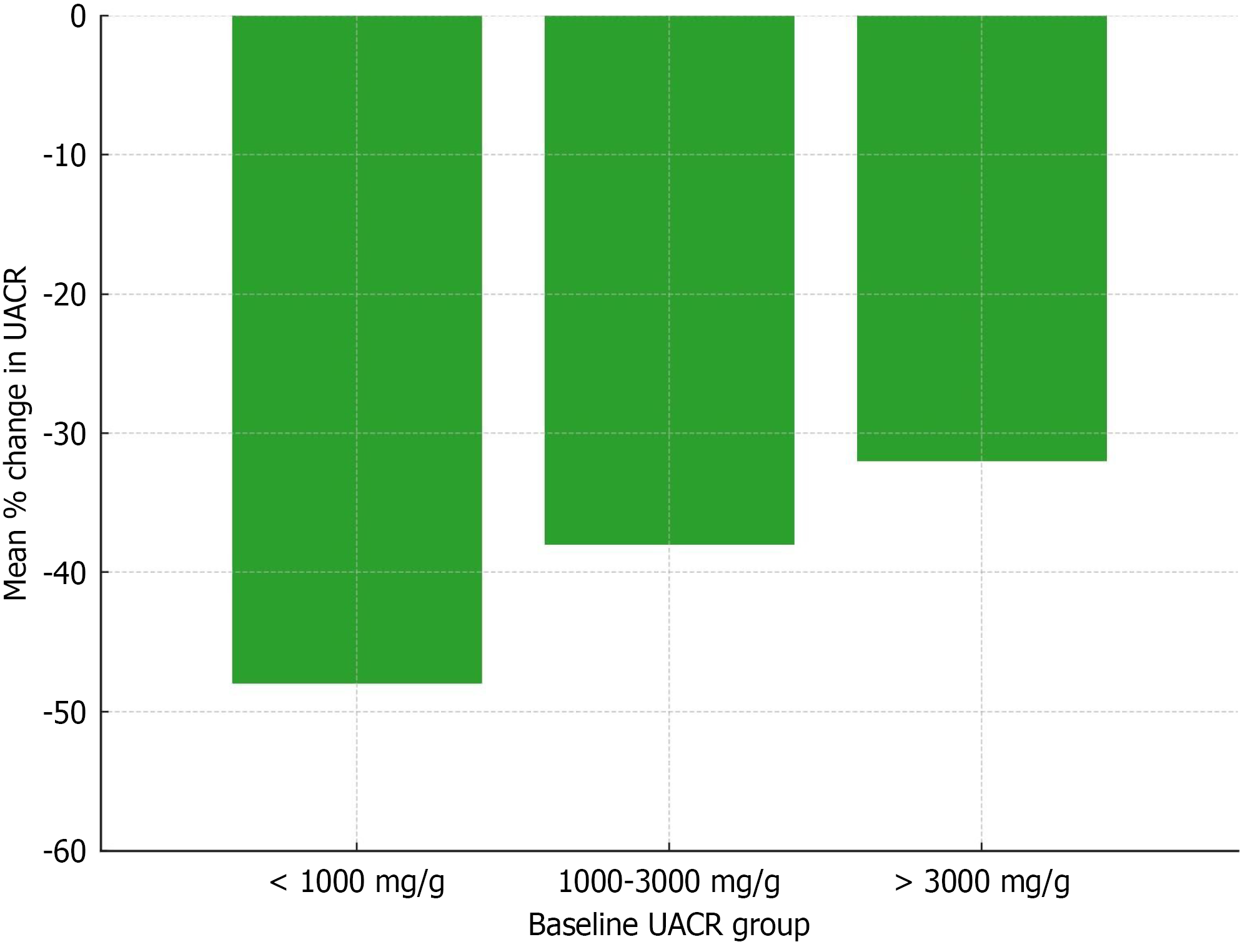
When the responders were evaluated with respect to baseline UACR < 1000 mg/g, 1000-3000 mg/g and > 3000 mg/g, most pronounced effect was observed in patients with baseline UACR < 1000 mg/g (-47.9%), followed by those in the 1000-3000 mg/g (-39.0%) and > 3000 mg/g (-32.8%) groups (Figure 5).
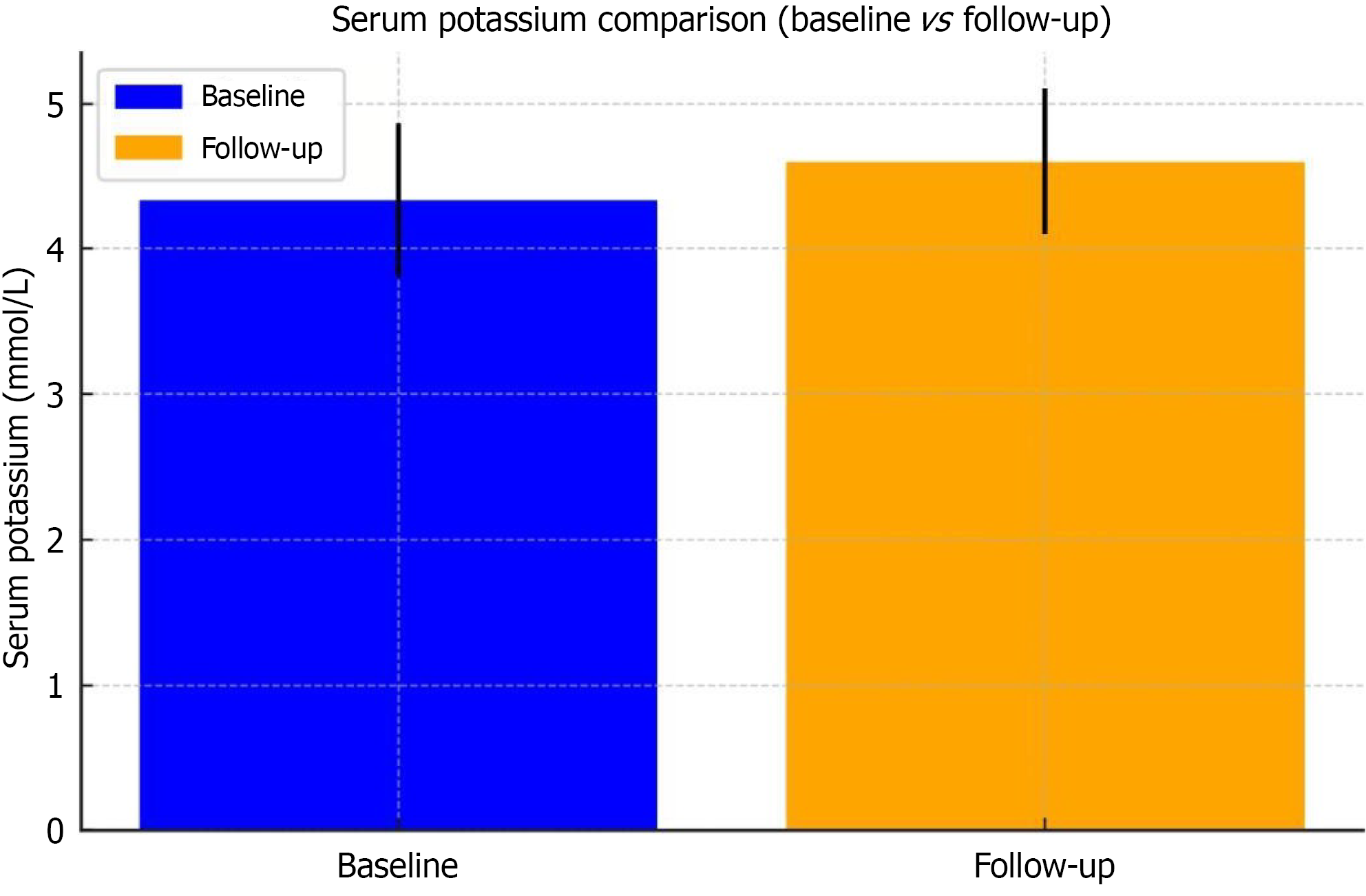
The mean serum potassium level increased from 4.33 mmol/L at baseline to 4.60 mmol/L at 24 weeks, representing a 6.4 percent increase. This change was statistically significant (P = 0.016, paired t-test), with a small-to-moderate effect size (Cohen’s d = 0.45). Despite this increase, serum potassium remained within acceptable safety limits for most patients. Five patients (15.2%) developed mild hyperkalaemia (serum potassium > 5.0 mmol/L), and no cases of severe hyperka
Renal function remained stable throughout the 24-week follow-up period. The mean eGFR at baseline was 36.3 ± 10.2 mL/minute/1.73 m2. No significant deterioration or clinical events necessitating treatment withdrawal were reported during the study period.
Among the 33 patients treated with finerenone, 26 (78.8%) completed the study without any adverse events. Mild hyperkalaemia (serum potassium > 5.0 mmol/L) was observed in 5 patients (15.2%), while hypotension occurred in 2 patients (6.1%) (Figure 6). No cases of severe hyperkalemia or adverse events requiring treatment discontinuation were reported.
The majority of patients did not experience adverse effects. Hyperkalaemia and hypotension were the only docu
This study evaluated the efficacy and safety of finerenone in proteinuric chronic kidney disease (CKD) patients with maximal dose of telmisartan and dapagliflozin in reduction of proteinuria, renal function deterioration, and safety profile over 24 months observation. While there was reduction in proteinuria with addition of finerenone without alarming hyperkalemia or reduction of eGFR, the observation was variable and based on baseline proteinuria.
The mean reduction in UACR across the full cohort was 9.5%. The magnitude of proteinuria reduction though not significant (P = 0.256), was beneficial to the patients who were on already on maximal antiproteinuric medications, with responder analysis demonstrating additional UACR lowering following finerenone initiation. A total of 22 patients (66.7%) were classified as responders, exhibiting reductions in UACR at 24 weeks, while 11 patients (33.3%) were non-responders. This exemplifies the response in majority of patient started on finerenone.
The only statistically significant predictor of response identified was baseline UACR Responders had a significantly higher baseline UACR compared to non-responders (2341.2 ± 1854.3 mg/g vs 1425.5 ± 998.7 mg/g, P = 0.028). This finding reiterates the gradient effect and suggests that patients with greater initial proteinuria are more likely to benefit from finerenone treatment. These results are consistent with subgroup analyses from both the FIDELIO-DKD and FIGARO-DKD trials, where higher baseline albuminuria correlated with greater relative reductions in UACR[9-11]. Thus, a higher baseline UACR is a strong predictor of response, helping guide therapeutic decisions and patient selection.
Responders with baseline UACR < 1000 mg/g, 1000-3000 mg/g, and > 3000 mg/g showed a mean reduction in UACR of 47.9%, 39.0% and 32.8% respectively. The heterogeneity in response observed in this study is consistent with findings from Bakris et al[5], who reported considerable variability in albuminuria reduction, even among similarly treated patients. No statistically significant differences were observed between responders and non-responders in terms of age, duration of diabetes, eGFR, or baseline serum potassium levels (P > 0.05 for all comparisons) (Table 3). This finding sug
| Variable | Responders (n = 22) | Non-responders (n = 11) | P value |
| Age (years) | 56.2 ± 8.9 | 59.1 ± 7.6 | 0.257 |
| Duration of diabetes (months) | 128.3 ± 46.7 | 135.9 ± 51.2 | 0.612 |
| eGFR (ml/minute/1.73 m2) | 41.5 (34.2-48.8) | 38.6 (32.1-46.4) | 0.276 |
| Serum potassium (mmol/L) | 4.6 (4.3-4.9) | 4.7 (4.4-5.0) | 0.720 |
The noticed increase in the drug effect with the increase of the degree of proteinuria is likely the underlying mechanism of action of finerenone. Finerenone, a non-steroidal MRA inhibits proinflammatory and profibrotic signalling pathways that are increasingly active in patients with advanced proteinuria. This pathophysiological context may explain why patients with higher baseline UACR experienced greater benefit from treatment. The observed reductions in albuminuria, particularly among responders, are consistent with finerenone’s known ability to reduce glomerular injury and preserve renal structure and function through anti-inflammatory effects.
Our findings are further supported by the work of Mårup et al[12], which demonstrated that finerenone significantly reduced albuminuria both as monotherapy and in combination with SGLT2 inhibitors such as dapagliflozin. Notably, their study also suggested an additive benefit when combining finerenone with dapagliflozin-an approach that may be particularly advantageous in patients with persistent proteinuria despite dual RAAS and SGLT2 inhibition.
Although our study did not include a control arm, the magnitude of albuminuria reduction observed in responders is comparable to that reported in placebo-corrected arms of larger randomized trials[10,12]. It is also noteworthy that the gradient effect did not appear to be strictly linear, with substantial reductions observed even in patients with moderate baseline UACR levels. This finding suggests that finerenone’s benefits may extend across a broader spectrum of albuminuria, though further prospective trials are warranted to confirm efficacy in patients with lower baseline risk.
Finerenone was generally well tolerated in this cohort. Over the 24-week treatment period, 5 out of 33 patients (15.2%) developed mild hyperkalaemia, defined as serum potassium > 5.0 mmol/L. No cases of severe hyperkalaemia or treatment discontinuation due to adverse events were recorded. These findings go with safety outcomes from the FIDELIO-DKD trial, where hyperkalaemia-related discontinuation occurred in 2.3% of participants in spite of being of a higher degree[9]. The higher proportion of hyperkalaemia observed in our study likely reflects the clinician guided frequent monitoring of potassium. The eGFR remained stable throughout the study. The baseline mean eGFR was 36.3 ± 10.2 mL/minute/1.73 m2, and no patient experienced a decline in kidney function that required discontinuation of the
These safety outcomes align with the data from Mårup et al[12], which reported modest declines in measured GFR when finerenone was combined with dapagliflozin. Such declines are thought to reflect transient hemodynamic effects rather than true structural renal injury. Importantly, our findings support the notion that finerenone can be used safely in combination with other renoprotective agents, provided that patients are closely monitored for potassium levels and volume status. Overall, the safety and renal function data from this study strengthen the indication for finerenone as a therapeutic option in DKD, particularly when integrated into a multimodal treatment approach targeting both RAAS and SGLT2 pathways.
This study has several limitations that must be considered when interpreting its findings. First, the observational and non-randomized design precludes any causal inference regarding the effects of finerenone. Without a placebo or com
Finerenone demonstrated a favourable safety profile and variable efficacy in reducing albuminuria among patients with DKD already receiving maximum tolerated doses of dapagliflozin and telmisartan. Patients with higher baseline UACR were more likely to respond than patients with lower baseline UACR in proteinuria. Baseline albuminuria emerged as a potential predictor of treatment response, while other clinical variables did not significantly differentiate responders from non-responders. These findings add to the role of finerenone as an antialbuminuric agent in DKD management and support its integration into multidrug strategies aimed at slowing disease progression. However, the observed heterogeneity in response highlights the need for personalized therapeutic approaches and biomarker-based patient selection.
We acknowledge the Department of Nephrology, Jawaharlal Nehru Medical College-Wardha, Saraswati Kidney Care Centre-Nagpur, Max Superspeciality Hospital-Nagpur and All India Institute of Medical Sciences- Nagpur for supporting the conduct of this study. We are also grateful to the administrative and technical staff for their assistance.
| 1. | Moriya S, Iwata H, Chikata Y, Funamizu T, Doi S, Koike T, Abe K, Matsuo R, Wada H, Naito R, Ogita M, Okai I, Dohi T, Kasai T, Okazaki S, Miyauchi K, Daida H, Minamino T. Prognostic impact of proteinuria in diabetic patients without chronic kidney disease undergoing percutaneous coronary intervention. Cardiovasc Diabetol Endocrinol Rep. 2025;11:34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 2. | Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull S, Cannon CP, Capuano G, Chu PL, de Zeeuw D, Greene T, Levin A, Pollock C, Wheeler DC, Yavin Y, Zhang H, Zinman B, Meininger G, Brenner BM, Mahaffey KW; CREDENCE Trial Investigators. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N Engl J Med. 2019;380:2295-2306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5174] [Cited by in RCA: 4474] [Article Influence: 639.1] [Reference Citation Analysis (5)] |
| 3. | Ferrè S, Li X, Adams-Huet B, Maalouf NM, Sakhaee K, Toto RD, Moe OW, Neyra JA. Association of serum magnesium with all-cause mortality in patients with and without chronic kidney disease in the Dallas Heart Study. Nephrol Dial Transplant. 2018;33:1389-1396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 4. | Heerspink HJL, Stefánsson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, Mann JFE, McMurray JJV, Lindberg M, Rossing P, Sjöström CD, Toto RD, Langkilde AM, Wheeler DC; DAPA-CKD Trial Committees and Investigators. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2020;383:1436-1446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4470] [Cited by in RCA: 3828] [Article Influence: 638.0] [Reference Citation Analysis (7)] |
| 5. | Bakris GL, Agarwal R, Anker SD, Pitt B, Ruilope LM, Rossing P, Kolkhof P, Nowack C, Schloemer P, Joseph A, Filippatos G; FIDELIO-DKD Investigators. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383:2219-2229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2319] [Cited by in RCA: 1935] [Article Influence: 322.5] [Reference Citation Analysis (10)] |
| 6. | Wheeler DC, Toto RD, Stefánsson BV, Jongs N, Chertow GM, Greene T, Hou FF, McMurray JJV, Pecoits-Filho R, Correa-Rotter R, Rossing P, Sjöström CD, Umanath K, Langkilde AM, Heerspink HJL; DAPA-CKD Trial Committees and Investigators. A pre-specified analysis of the DAPA-CKD trial demonstrates the effects of dapagliflozin on major adverse kidney events in patients with IgA nephropathy. Kidney Int. 2021;100:215-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 309] [Article Influence: 61.8] [Reference Citation Analysis (3)] |
| 7. | Tardif JC, Marquis-Gravel G. Low-Dose Colchicine for the Management of Coronary Artery Disease. J Am Coll Cardiol. 2021;78:867-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 8. | Pitt B, Filippatos G, Agarwal R, Anker SD, Bakris GL, Rossing P, Joseph A, Kolkhof P, Nowack C, Schloemer P, Ruilope LM; FIGARO-DKD Investigators. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med. 2021;385:2252-2263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1436] [Cited by in RCA: 1151] [Article Influence: 230.2] [Reference Citation Analysis (8)] |
| 9. | Agarwal R, Green JB, Heerspink HJL, Mann JFE, McGill JB, Mottl AK, Rosenstock J, Rossing P, Vaduganathan M, Brinker M, Edfors R, Li N, Scheerer MF, Scott C, Nangaku M; CONFIDENCE Investigators. Finerenone with Empagliflozin in Chronic Kidney Disease and Type 2 Diabetes. N Engl J Med. 2025;393:533-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 151] [Article Influence: 151.0] [Reference Citation Analysis (0)] |
| 10. | Ruilope LM, Pitt B, Anker SD, Rossing P, Kovesdy CP, Pecoits-Filho R, Pergola P, Joseph A, Lage A, Mentenich N, Scheerer MF, Bakris GL. Kidney outcomes with finerenone: an analysis from the FIGARO-DKD study. Nephrol Dial Transplant. 2023;38:372-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 59] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 11. | Curovic VR, Jongs N, Kroonen MYAM, Zobel EH, Hansen TW, Sen T, Laverman GD, Kooy A, Persson F, Rossing P, Heerspink HJL. Optimization of Albuminuria-Lowering Treatment in Diabetes by Crossover Rotation to Four Different Drug Classes: A Randomized Crossover Trial. Diabetes Care. 2023;46:593-601. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 12. | Mårup FH, Thomsen MB, Birn H. Additive effects of dapagliflozin and finerenone on albuminuria in non-diabetic CKD: an open-label randomized clinical trial. Clin Kidney J. 2024;17:sfad249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (0)] |